Evaluation of the Corrosion Resistance Properties of Electroplated Chitosan-Zn1−xCuxO Composite Thin Films
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Beecroft, L.L.; Ober, C.K. Nanocomposite materials for optical applications. Chem. Mater. 1997, 9, 1302–1317. [Google Scholar] [CrossRef]
- Sanchez, C.; Soler-Illia, G.J.D.A.A.; Ribot, F.; Lalot, T.; Mayer, C.R.; Cabuil, V. Designed hybrid organic−inorganic nanocomposites from functional nanobuilding blocks. Chem. Mater. 2001, 13, 3061–3083. [Google Scholar] [CrossRef]
- Gangopadhyay, R.; De, A. Conducting polymer nanocomposites: A brief overview. Chem. Mater. 2000, 12, 608–622. [Google Scholar] [CrossRef]
- Mitzi, D.B. Thin-film deposition of organic-inorganic hybrid materials. Chem. Mater. 2001, 13, 3283–3298. [Google Scholar] [CrossRef]
- Merachtsaki, D.; Xidas, P.; Giannakoudakis, P.; Triantafyllidis, K.; Spathis, P. Corrosion protection of steel by epoxy-organoclay nanocomposite coatings. Coatings 2017, 7, 84. [Google Scholar] [CrossRef]
- Zhitomirsky, I. Electrophoretic deposition of organic–inorganic nanocomposites. J. Mater. Sci. 2006, 41, 8186–8195. [Google Scholar] [CrossRef]
- Mirak, M.; Alizadeh, M.; Ghaffari, M.; Ashtiani, M.N. Characterization, mechanical properties and corrosion resistance of biocompatible Zn-HA/TiO2 nanocomposite coatings. J. Mech. Behav. Biomed. Mater. 2016, 62, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Zhitomirsky, I. Electrodeposition of composite hydroxyapatite–chitosan films. Mater. Chem. Phys. 2005, 94, 245–251. [Google Scholar] [CrossRef]
- Dhanasekaran, V.; Mahalingam, T.; Chandramohan, R. Morphology selection for cupric oxide thin films by electrodeposition. Microsc. Res. Tech. 2011, 74, 980–983. [Google Scholar] [CrossRef] [PubMed]
- Boccaccini, A.R.; Zhitomirsky, I. Application of electrophoretic and electrolytic deposition techniques in ceramics processing. Curr. Opin. Solid State Mater. Sci. 2002, 6, 251–260. [Google Scholar] [CrossRef]
- Fernandes, R.; Wu, L.-Q.; Chen, T.; Yi, H.; Rubloff, G.W.; Ghodssi, R.; Bentley, W.E.; Payne, G.F. Electrochemically induced deposition of a polysaccharide hydrogel onto a patterned surface. Langmuir 2003, 19, 4058–4062. [Google Scholar] [CrossRef]
- Yi, H.; Wu, L.-Q.; Bentley, W.E.; Ghodssi, R.; Rubloff, G.W.; Culver, J.N.; Payne, G.F. Biofabrication with chitosan. Biomacromolecules 2005, 6, 2881–2894. [Google Scholar] [CrossRef] [PubMed]
- Cheong, M.; Zhitomirsky, I. Electrodeposition of alginic acid and composite films. Colloids Surf. A 2008, 328, 73–78. [Google Scholar] [CrossRef]
- Gorelikov, I.; Kumacheva, E. Electrodeposition of polymer-semiconductor nanocomposite films. Chem. Mater. 2004, 16, 4122–4127. [Google Scholar] [CrossRef]
- Grandfield, K.; Sun, F.; FitzPatrick, M.; Cheong, M.; Zhitomirsky, I. Electrophoretic deposition of polymer-carbon nanotube–hydroxyapatite composites. Surf. Coat. Technol. 2009, 203, 1481–1487. [Google Scholar] [CrossRef]
- Lee, J.-H.; Leu, C.; Chung, Y.-W.; Hon, M.-H. Fabrication of ordered ZnO hierarchical structures controlled via surface charge in the electrophoretic deposition process. Nanotechnology 2006, 17, 4445. [Google Scholar] [CrossRef]
- Park, J.J.; Luo, X.; Yi, H.; Valentine, T.M.; Payne, G.F.; Bentley, W.E.; Ghodssi, R.; Rubloff, G.W. Chitosan-mediated in situ biomolecule assembly in completely packaged microfluidic devices. Lab Chip 2006, 6, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zhitomirsky, I. Electrodeposition of hyaluronic acid and composite films. Surf. Eng. 2013, 25, 621–627. [Google Scholar] [CrossRef]
- Wu, L.-Q.; Payne, G.F. Biofabrication: Using biological materials and biocatalysts to construct nanostructured assemblies. Trends Biotechnol. 2004, 22, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Anandhavelu, S.; Dhanasekaran, V.; Sethuraman, V.; Park, H.J. Chitin and chitosan based hybrid nanocomposites for super capacitor applications. J. Nanosci. Nanotechnol. 2017, 17, 1321–1328. [Google Scholar] [CrossRef]
- Sanmugam, A.; Vikraman, D.; Venkatesan, S.; Park, H.J. Optical and structural properties of solvent free synthesized starch/chitosan-ZnO nanocomposites. J. Nanomater. 2017, 2017, 7536364. [Google Scholar] [CrossRef]
- Li, Y.; Wu, K.; Zhitomirsky, I. Electrodeposition of composite zinc oxide–chitosan films. Colloids Surf. A 2010, 356, 63–70. [Google Scholar] [CrossRef]
- Gu, B.; Xu, C.; Zhu, G.; Liu, S.; Chen, L.; Wang, M.; Zhu, J. Layer by layer immobilized horseradish peroxidase on zinc oxide nanorods for biosensing. J. Phys. Chem. B 2009, 113, 6553–6557. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.; Kaushik, A.; Solanki, P.R.; Ansari, A.A.; Pandey, M.K.; Malhotra, B. Zinc oxide nanoparticles-chitosan composite film for cholesterol biosensor. Anal. Chim. Acta 2008, 616, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Solanki, P.R.; Kaushik, A.; Ansari, A.A.; Sumana, G.; Malhotra, B. Zinc oxide-chitosan nanobiocomposite for urea sensor. Appl. Phys. Lett. 2008, 93, 163903. [Google Scholar] [CrossRef]
- Wang, L.; Li, D.-B.; Li, K.; Chen, C.; Deng, H.-X.; Gao, L.; Zhao, Y.; Jiang, F.; Li, L.; Huang, F.; et al. Stable 6%-efficient Sb2Se3 solar cells with a ZnO buffer layer. Nat. Energy 2017, 2, 17046. [Google Scholar] [CrossRef]
- Gholizadeh, A.; Reyhani, A.; Parvin, P.; Mortazavi, S.Z. Efficiency enhancement of ZnO nanostructure assisted Si solar cell based on fill factor enlargement and UV-blue spectral down-shifting. J. Phys. D Appl. Phys. 2017, 50, 185501. [Google Scholar] [CrossRef]
- Yu, X.; Ma, J.; Ji, F.; Wang, Y.; Zhang, X.; Ma, H. Influence of annealing on the properties of ZnO:Ga films prepared by radio frequency magnetron sputtering. Thin Solid Films 2005, 483, 296–300. [Google Scholar] [CrossRef]
- Hu, J.; Gordon, R.G. Atmospheric pressure chemical vapor deposition of gallium doped zinc oxide thin films from diethyl zinc, water, and triethyl gallium. J. Appl. Phys. 1992, 72, 5381–5392. [Google Scholar] [CrossRef]
- Kotlyarchuk, B.; Savchuk, V.; Oszwaldowski, M. Preparation of undoped and indium doped ZnO thin films by pulsed laser deposition method. Cryst. Res. Technol. 2005, 40, 1118–1123. [Google Scholar] [CrossRef]
- Omri, K.; Najeh, I.; Dhahri, R.; El Ghoul, J.; El Mir, L. Effects of temperature on the optical and electrical properties of ZnO nanoparticles synthesized by sol–gel method. Microelectron. Eng. 2014, 128, 53–58. [Google Scholar] [CrossRef]
- Chandramohan, R.; Vijayan, T.A.; Arumugam, S.; Ramalingam, H.B.; Dhanasekaran, V.; Sundaram, K.; Mahalingam, T. Effect of heat treatment on microstructural and optical properties of CBD grown Al-doped ZnO thin films. Mater. Sci. Eng. B Solid 2011, 176, 152–156. [Google Scholar] [CrossRef]
- Ben Belgacem, R.; Chaari, M.; Braña, A.F.; Garcia, B.J.; Matoussi, A. Structural, electric modulus and complex impedance analysis of ZnO/TiO2 composite ceramics. J. Am. Chem. Soc. 2017, 100, 2045–2058. [Google Scholar] [CrossRef]
- Dai, L.; Chen, X.L.; Wang, W.J.; Zhou, T.; Hu, B.Q. Growth and luminescence characterization of large-scale zinc oxide nanowires. J. Phys. Condens. Matter. 2003, 15, 2221–2226. [Google Scholar] [CrossRef]
- Tuomisto, F.; Saarinen, K.; Grasza, K.; Mycielski, A. Observation of Zn vacancies in ZnO grown by chemical vapor transport. Phys. Status Solidi B 2006, 243, 794–798. [Google Scholar] [CrossRef]
- Janotti, A.; Walle, C.G.V.D. Oxygen vacancies in ZnO. Appl. Phys. Lett. 2005, 87, 122102. [Google Scholar] [CrossRef]
- Look, D.C.; Hemsky, J.W.; Sizelove, J.R. Residual native shallow donor in ZnO. Phys. Rev. Lett. 1999, 82, 2552–2555. [Google Scholar] [CrossRef]
- Wang, M.; Ren, F.; Zhou, J.; Cai, G.; Cai, L.; Hu, Y.; Wang, D.; Liu, Y.; Guo, L.; Shen, S. N doping to ZnO nanorods for photoelectrochemical water splitting under visible light: Engineered impurity distribution and terraced band structure. Sci. Rep. 2015, 5, 12925. [Google Scholar] [CrossRef] [PubMed]
- Fathollahi, V.; Amini, M.M. Sol–gel preparation of highly oriented gallium-doped zinc oxide thin films. Mater. Lett. 2001, 50, 235–239. [Google Scholar] [CrossRef]
- Kılınç, N.; Arda, L.; Öztürk, S.; Öztürk, Z.Z. Structure and electrical properties of mg-doped ZnO nanoparticles. Cryst. Res. Technol. 2010, 45, 529–538. [Google Scholar] [CrossRef]
- Pawar, S.M.; Kim, J.; Inamdar, A.I.; Woo, H.; Jo, Y.; Pawar, B.S.; Cho, S.; Kim, H.; Im, H. Multi-functional reactively-sputtered copper oxide electrodes for supercapacitor and electro-catalyst in direct methanol fuel cell applications. Sci. Rep. 2016, 6, 21310. [Google Scholar] [CrossRef] [PubMed]
- Vikraman, D.; Park, H.J.; Kim, S.-I.; Thaiyan, M. Magnetic, structural and optical behavior of cupric oxide layers for solar cells. J. Alloys Compd. 2016, 686, 616–627. [Google Scholar] [CrossRef]
- Chandrappa, K.G.; Venkatesha, T.V. Generation of nanostructured CuO by electrochemical method and its Zn–Ni–CuO composite thin films for corrosion protection. Mater. Corros. 2013, 64, 831–839. [Google Scholar] [CrossRef]
- Momeni, M.M.; Nazari, Z.; Kazempour, A.; Hakimiyan, M.; Mirhoseini, S.M. Preparation of CuO nanostructures coating on copper as supercapacitor materials. Surf. Eng. 2014, 30, 775–778. [Google Scholar] [CrossRef]
- Bu, I.Y.Y.; Huang, R. Fabrication of CuO-decorated reduced graphene oxide nanosheets for supercapacitor applications. Ceram. Int. 2017, 43, 45–50. [Google Scholar] [CrossRef]
- Gholivand, M.B.; Heydari, H.; Abdolmaleki, A.; Hosseini, H. Nanostructured CuO/PANI composite as supercapacitor electrode material. Mater. Sci. Semicond. Process. 2015, 30, 157–161. [Google Scholar] [CrossRef]
- Arena, A.; Scandurra, G.; Ciofi, C. Copper oxide chitosan nanocomposite: Characterization and application in non-enzymatic hydrogen peroxide sensing. Sensors 2017, 17, 2198. [Google Scholar] [CrossRef] [PubMed]
- Dai, N.; Zhang, L.-C.; Zhang, J.; Chen, Q.; Wu, M. Corrosion behavior of selective laser melted Ti-6Al-4V alloy in NaCl solution. Corros. Sci. 2016, 102, 484–489. [Google Scholar] [CrossRef]
- Dai, N.; Zhang, L.-C.; Zhang, J.; Zhang, X.; Ni, Q.; Chen, Y.; Wu, M.; Yang, C. Distinction in corrosion resistance of selective laser melted Ti-6Al-4V alloy on different planes. Corros. Sci. 2016, 111, 703–710. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, J.; Dai, N.; Qin, P.; Attar, H.; Zhang, L.-C. Corrosion behaviour of selective laser melted Ti-TiB biocomposite in simulated body fluid. Electrochim. Acta 2017, 232, 89–97. [Google Scholar] [CrossRef]
- Lu, H.-B.; Li, Y.; Wang, F.-H. Dealloying behaviour of Cu–20Zr alloy in hydrochloric acid solution. Corros. Sci. 2006, 48, 2106–2119. [Google Scholar] [CrossRef]
- Lin, S.; Shih, H.; Mansfeld, F. Corrosion protection of aluminum alloys and metal matrix composites by polymer coatings. Corros. Sci. 1992, 33, 1331–1349. [Google Scholar] [CrossRef]
- Singh, B.P.; Jena, B.K.; Bhattacharjee, S.; Besra, L. Development of oxidation and corrosion resistance hydrophobic graphene oxide-polymer composite coating on copper. Surf. Coat. Technol. 2013, 232, 475–481. [Google Scholar] [CrossRef]
- Miao, J.; Zhang, L.-C.; Lin, H. A novel kind of thin film composite nanofiltration membrane with sulfated chitosan as the active layer material. Chem. Eng. Sci. 2013, 87, 152–159. [Google Scholar] [CrossRef]
- Miao, J.; Lin, H.; Wang, W.; Zhang, L.-C. Amphoteric composite membranes for nanofiltration prepared from sulfated chitosan crosslinked with hexamethylene diisocyanate. Chem. Eng. J. 2013, 234, 132–139. [Google Scholar] [CrossRef]
- Mahalingam, T.; Dhanasekaran, V.; Chandramohan, R.; Rhee, J.-K. Microstructural properties of electrochemically synthesized ZnSe thin films. J. Mater. Sci. 2012, 47, 1950–1957. [Google Scholar] [CrossRef]
- Anandhavelu, S.; Thambidurai, S. Effect of zinc chloride and sodium hydroxide concentration on the optical property of chitosan–ZnO nanostructure prepared in chitin deacetylation. Mater. Chem. Phys. 2011, 131, 449–454. [Google Scholar] [CrossRef]
- Kiriakidou, F.; Kondarides, D.I.; Verykios, X.E. The effect of operational parameters and TiO2-doping on the photocatalytic degradation of azo-dyes. Catal. Today 1999, 54, 119–130. [Google Scholar] [CrossRef]
- Poulios, I.; Tsachpinis, I. Photodegradation of the textile dye reactive black 5 in the presence of semiconducting oxides. J. Chem. Technol. Biotechnol. 1999, 74, 349–357. [Google Scholar] [CrossRef]
- Liu, G.; Wu, T.; Zhao, J.; Hidaka, H.; Serpone, N. Photoassisted degradation of dye pollutants. 8. Irreversible degradation of alizarin red under visible light radiation in air-equilibrated aqueous TiO2 dispersions. Environ. Sci. Technol. 1999, 33, 2081–2087. [Google Scholar] [CrossRef]
- Egelhaaf, H.-J.; Oelkrug, D. Luminescence and nonradiative deactivation of excited states involving oxygen defect centers in polycrystalline ZnO. J. Cryst. Growth 1996, 161, 190–194. [Google Scholar] [CrossRef]
- Chaari, M.; Ben Belgacem, R.; Matoussi, A. Impedance analysis, dielectric relaxation and modulus behaviour of ZnO-Sn2O3 ceramics. J. Alloys Compd. 2017, 726, 49–56. [Google Scholar] [CrossRef]
- Lee, L.; Kao, R.-H.; Yang, C.-S.; Ku, C.-S.; Lee, H.-Y. An insulating-conductive transition driven by partial crystallization of amorphous Zn-Sn-O alloy. J. Alloys Compd. 2016, 672, 636–642. [Google Scholar] [CrossRef]
- Wan, Y.; Wang, X.; Sun, H.; Li, Y.; Zhang, K.; Wu, Y. Corrosion behavior of copper at elevated temperature. Int. J. Electrochem. Sci. 2012, 7, 7902–7914. [Google Scholar]
- Hosseini, M.; Bagheri, R.; Najjar, R. Electropolymerization of polypyrrole and polypyrrole-ZnO nanocomposites on mild steel and its corrosion protection performance. J. Appl. Polym. Sci. 2011, 121, 3159–3166. [Google Scholar] [CrossRef]
- Aramaki, K.; Hagiwara, M.; Nishihara, H. The synergistic effect of anions and the ammonium cation on the inhibition of iron corrosion in acid solution. Corros. Sci. 1987, 27, 487–497. [Google Scholar] [CrossRef]
- Hihara, L.; Latanision, R. Suppressing galvanic corrosion in graphite/aluminum metal-matrix composites. Corros. Sci. 1993, 34, 655–665. [Google Scholar] [CrossRef]
- Brichau, F.; Deconinck, J. A numerical model for cathodic protection of buried pipes. Corrosion 1994, 50, 39–49. [Google Scholar] [CrossRef]
- Metwally, I.; Al-Mandhari, H.; Gastli, A.; Nadir, Z. Factors affecting cathodic-protection interference. Eng. Anal. Bound. Elem. 2007, 31, 485–493. [Google Scholar] [CrossRef]




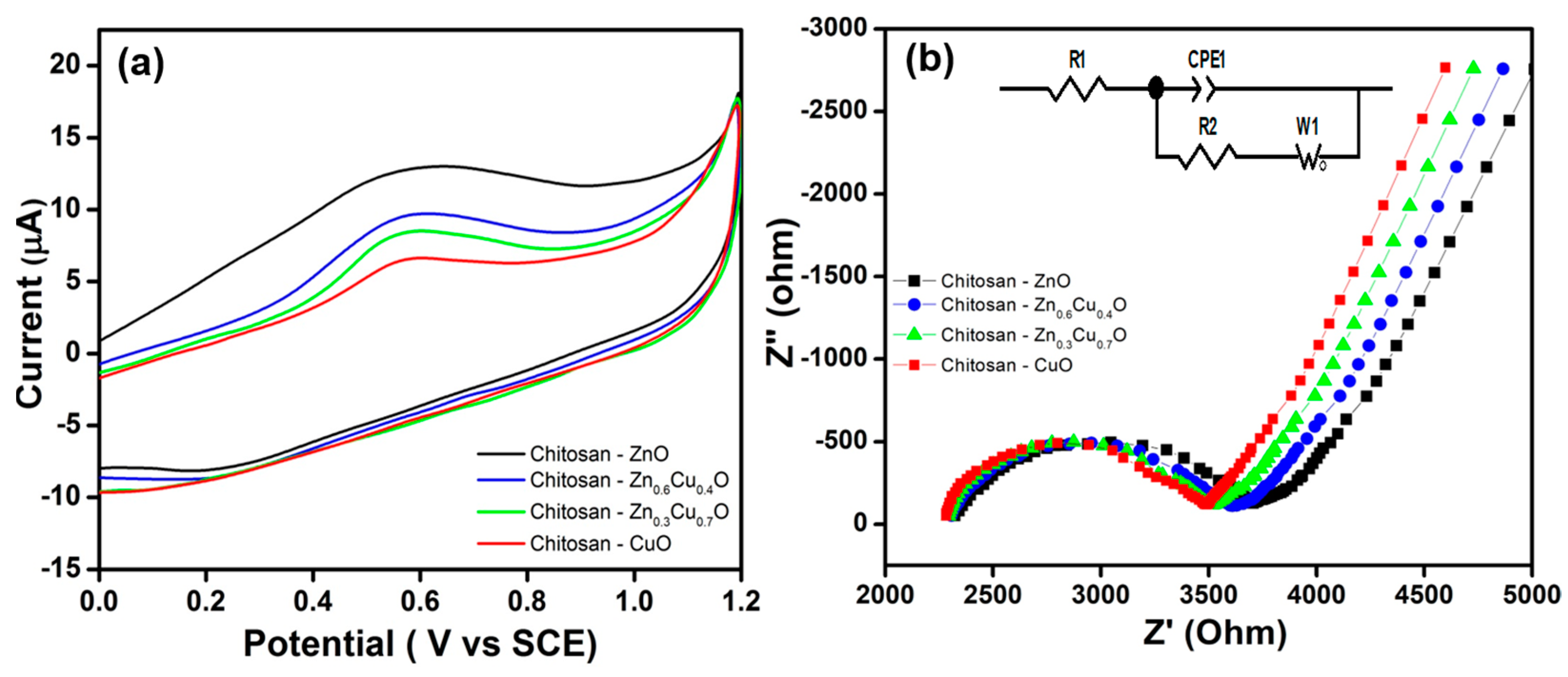
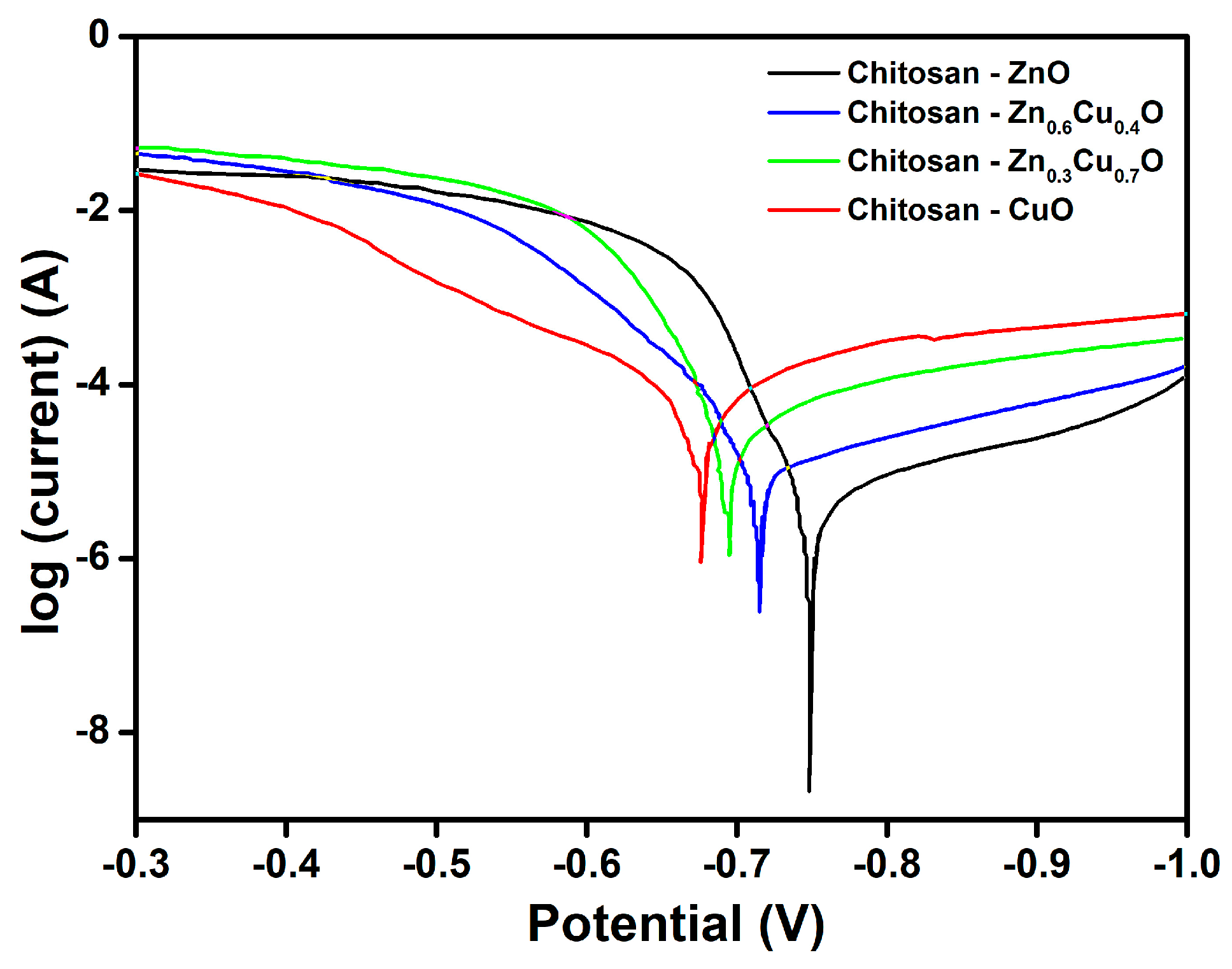
| Composite | EIS Parameters | Corrosion Parameters | |||||
|---|---|---|---|---|---|---|---|
| Electrochemical Method | Weight Loss Method | ||||||
| R1 (Ohm) | R2 (Ohm) | CPE1 μF | Corrosion Current Density (cm−2) | Corrosion Rate (mmpy) 10−3 | Weight Loss (mg) | Corrosion Rate (mmpy) 10−3 | |
| Chitosan-ZnO | 2321 | 3699 | 0.63 | 2.81 × 10−6 | 34 | 160 | 35 |
| Chitosan-Zn0.6Cu0.4O | 2308 | 3610 | 0.58 | 8.12 × 10−6 | 99 | 260 | 58 |
| Chitosan-Zn0.3Cu0.7O | 2290 | 3510 | 0.52 | 2.63 × 10−5 | 320 | 415 | 93 |
| Chitosan-CuO | 2282 | 3488 | 0.31 | 5.12 × 10−5 | 623 | 635 | 141 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sanmugam, A.; Vikraman, D.; Karuppasamy, K.; Lee, J.Y.; Kim, H.-S. Evaluation of the Corrosion Resistance Properties of Electroplated Chitosan-Zn1−xCuxO Composite Thin Films. Nanomaterials 2017, 7, 432. https://doi.org/10.3390/nano7120432
Sanmugam A, Vikraman D, Karuppasamy K, Lee JY, Kim H-S. Evaluation of the Corrosion Resistance Properties of Electroplated Chitosan-Zn1−xCuxO Composite Thin Films. Nanomaterials. 2017; 7(12):432. https://doi.org/10.3390/nano7120432
Chicago/Turabian StyleSanmugam, Anandhavelu, Dhanasekaran Vikraman, K. Karuppasamy, Ji Young Lee, and Hyun-Seok Kim. 2017. "Evaluation of the Corrosion Resistance Properties of Electroplated Chitosan-Zn1−xCuxO Composite Thin Films" Nanomaterials 7, no. 12: 432. https://doi.org/10.3390/nano7120432
APA StyleSanmugam, A., Vikraman, D., Karuppasamy, K., Lee, J. Y., & Kim, H.-S. (2017). Evaluation of the Corrosion Resistance Properties of Electroplated Chitosan-Zn1−xCuxO Composite Thin Films. Nanomaterials, 7(12), 432. https://doi.org/10.3390/nano7120432







