Abstract
Graphene, fullerenes, diamond, carbon nanotubes, and carbon dots are just a few of the carbon-based nanomaterials that have gained enormous popularity in a variety of scientific disciplines and industrial uses. As a two-dimensional material in the creation of therapeutic delivery systems for many illnesses, nanosized graphene oxide (NGO) is now garnering a large amount of attention among these materials. In addition to other benefits, NGO functions as a drug nanocarrier with remarkable biocompatibility, high pharmaceutical loading capacity, controlled drug release capability, biological imaging efficiency, multifunctional nanoplatform properties, and the power to increase the therapeutic efficacy of loaded agents. Thus, NGO is a perfect nanoplatform for the development of drug delivery systems (DDSs) to both detect and treat a variety of ailments. This review article’s main focus is on investigating surface functionality, drug-loading methods, and drug release patterns designed particularly for smart delivery systems. The paper also examines the relevance of using NGOs to build DDSs and considers prospective uses in the treatment of diseases including cancer, infection by bacteria, and bone regeneration medicine. These factors cover the use of naturally occurring medicinal substances produced from plant-based sources.
1. Introduction
Applications for carbon-based nanomaterials of various dimensions may be found in nanotechnology. Diamond, amorphous carbon, and graphite are the three main carbon allotropes found in nature. Additionally, carbon has the extraordinary capacity to hybridize its orbitals in sp, sp2, and sp3 configurations, producing a variety of allotropes [1]. The abundance of allotropes is closely related to the fact that carbon is the most adaptable substance in the periodic table of elements [2]. The development of nanotechnology has produced a wide variety of carbon-based tiny materials, each with a unique nanostructure designed for a particular use. These include fullerenes, small diamonds, carbon nanocones, carbon nanohorns, single-walled carbon nanotubes (SWCNTs), multi-walled carbon nanotubes (MWCNTs), graphene oxide (GO), and nanosized graphene oxide (NGO) [3,4,5,6,7]. The utilization of carbon-derived nanotechnologies in biomedical applications for nanomedicine and drug delivery systems (DDSs) has accelerated significantly, as indicated by trends throughout time and extensive reviews in more recent years [8,9,10,11]. These nanostructures have remarkable properties, including flexibility, electrical conductivity, mechanical, electrical, and structural variety. They are thus shown to be incredibly well-suited for diagnosing and treating a wide range of illnesses [12,13,14]. It is important to note that graphene is a two-dimensional (2D) material characterized by a uniform, flat monolayer consisting of carbon atoms organized in a hexagonal honeycomb lattice, which is the focus of our specific attention in this study on NGO [15]. Notably, graphene’s unique properties make it an interesting substance on the frontier of 21st-century innovation, rising quickly to prominence in the field of materials research and practical applications [16,17,18]. The synthesis of GO at the micro- and nanoscales has been accomplished using a variety of techniques. Some of these techniques are known to effectively create graphite oxide, a step to creating GO. These techniques include the Hofmann method, Brodie approach, Staudenmaier method, and Hummers’ method, in addition to several variations that developed from these [18]. Due to its extensive use and efficiency, Hummers’ approach has become the standard method for producing graphene oxide nanoparticles [19,20]. The first separation of single-layer graphene from graphite was accomplished in 2004 by University of Manchester scientists Novoselov and Geim [21] using a simple mechanical peeling method. Following this discovery, graphene saw a rise in attention and a variety of uses across several industries [22]. There are several different ways to create GO, including two-layered GO (2 layers), few-layered GO (3–4 layers), multi-layered GO (5–10 layers), and graphite oxide (>10 layers) [23].
NGO is increasingly being used in a variety of delivery systems, including drugs, proteins, small therapeutic molecules, and genes, along with diverse biomedical applications including bone regeneration, implants, and more. These applications are in addition to its fundamental properties, which it shares with other carbon-based nanomaterials, including electrical, optical, and thermal characteristics. The numerous characteristics of NGO are mostly responsible for this widespread use. In this article, we discuss a number of reasons that highlight the increased interest in using NGO in pharma and biological contexts (Figure 1).
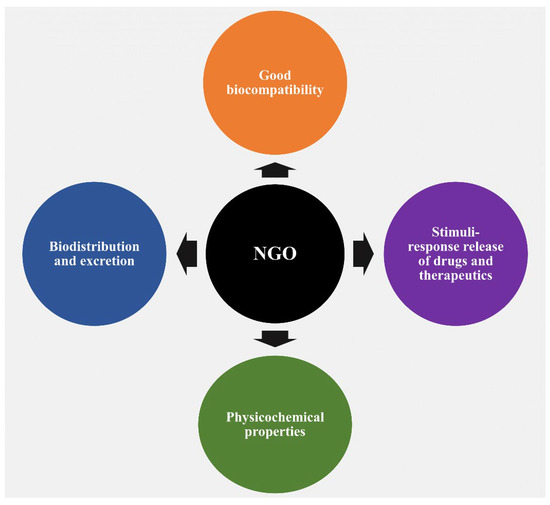
Figure 1.
A schematic depiction illustrating the diverse properties of NGOs and their applicability in drug delivery systems, tissue engineering, and biomedical applications.
One noteworthy reason for this interest is the observed good biocompatibility of NGOs, which is largely related to dose-dependent toxicity [24,25,26]. The biocompatibility of any nanocarrier should be understood as it is a key factor in determining the best way to include it in DDSs. After administration, graphene, a typical carbon-based nanomaterial, has the capacity to pass through physiological barriers or cellular structures, allowing it to enter the body or cells. It is crucial to note that this process can cause toxicity, both in vitro and in vivo [27]. A wide range of factors, such as various administration routes, entry mechanisms, distribution patterns, excretion pathways, cell uptake behaviors, and particular locations within the biological system, have a significant impact on the degree of toxicity, or conversely, biocompatibility and safety [28,29,30]. Regarding this essential characteristic, Wang et al.’s earlier research thoroughly evaluated the biocompatibility of GO, both in vitro employing human fibroblast cells and in vivo using mice [24]. The results showed that GOs at concentrations below 20 μg/mL exhibited no cellular toxic effects, and the intermediate dose of 0.25 mg showed no detectable harm to mice after cells were exposed to various concentrations for a period of up to 5 days, followed by injection of mice with varying concentrations over a period of 30 days. Notably, different hazardous responses were produced in cellular and animal models at greater dosages and in certain disease stages. In another study conducted by Zhang et al., administration of GO to mice at a dosage of 1 mg/kg of body weight for 14 days did not cause any detectable pathological alterations in the organs evaluated [31]. Furthermore, in a different study, Liao et al. found that aggregated graphene sheets had a stronger cytotoxic effect on human skin fibroblast cells than graphene oxide [25]. Similar to this, Zhang et al. showed that ultrasmall GO nanosheets differ from bigger, randomly sized nanosheets in that they display reduced cytotoxicity while permitting improved cellular uptake [32]. The surface functionalization strategy has recently gained acceptance among scientists as a means of improving the biocompatibility of graphene nanomaterials. NGO-NH2 (aminated), NGO-polyacrylamide functionalization (NGO-PAM), NGO-polyacrylic acid functionalization (NGO-PAA), NGO-pegylated functionalization (NGO-PEG), NGO-chitosan, and other modifications are noteworthy among these functionalizations (Figure 2).
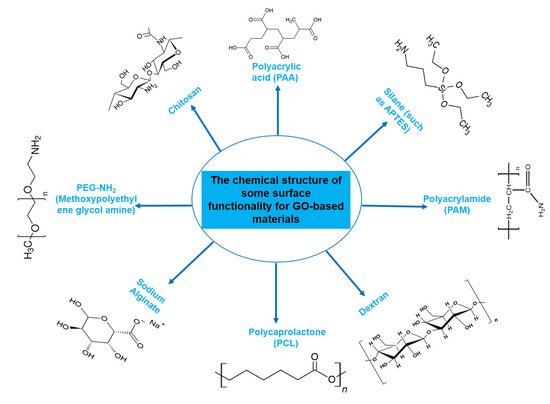
Figure 2.
Chemical structures of molecules frequently used for functionalization of GO-based materials. Chemical structures of chitosan [33], polyacrylamide (PAM) [34], dextran [35], and polycaprolactone (PCL) [36]. Polyacrylic acid (PAA), APTES, dextran, and other chemical structures were obtained from chemspider.com, the website of Royal Society of Chemistry (http://www.chemspider.com/Chemical-Structure, accessed on 21 September 2023). PEG-NH2 (methoxypolyethylene glycol amine) was taken from the Sigma Aldrich website (https://www.sigmaaldrich.com, accessed on 21 September 2023).
In comparison with graphene oxide nanoparticles, these functionalized versions exhibit decreased toxicity [37,38,39,40,41,42,43,44,45]. Kiew et al. briefly summarized the in vitro and in vivo biocompatibility aspects; they noticed that surface functionalization further improves biocompatibility and that NGO displays lower toxicity than micro-sized graphene oxide. This improvement is a result of less material interaction with biological barriers in vitro. Additionally, as compared to their micro-sized counterparts, NGO exhibits higher biocompatibility, and coating the surface helps to prevent accumulation and ameliorate toxic effects in vivo, improving overall biocompatibility [30].
In addition, Bussy et al. have provided a series of recommendations for the use and advancement of graphene and its derivatives, with a view to boosting general safety and reducing the possibility of negative responses in individuals exposed to graphene [46]. These regulations are divided into three main categories: (1) Using small-scale graphene and distinct sheets makes it easier for body macrophages to efficiently internalize and remove graphene from deposition locations. (2) Utilizing hydrophilic, reliable, colloidal dispersions reduces the chance of graphene aggregation within the body and ensures stability for in vivo applications. (3) The use of excretable or chemically modified graphene guarantees that the material is either able to be excreted from the body or is efficiently engineered to decay. Together, these regulations improve safety profiles and reduce the possibility of adverse effects in situations involving human exposure.
According to a review undertaken by Zhang et al. [47], the contact of NGO with plasma membrane and its subsequent translocation across this membrane comprise crucial processes determining the induction of biological effects. The physical and chemical characteristics of NGO, including its size, shape, and surface chemistry, substantially influence the nature of these effects. As a result, it becomes possible to improve transit efficiency while also reducing potential toxicity or negative effects by strategically manipulating these characteristics [47]. A wide range of evaluations on numerous topics, such as biocompatibility, biosafety issues, and contradictions about the impact of graphene nanoparticles, have been offered for a more thorough investigation of this topic [27,30,47,48,49,50,51]. These reviews more deeply examine the complex dynamics that control how graphene nanoparticles interact with biological systems.
The second rationale for utilizing NGOs revolves around their biodistribution and subsequent excretion from the body, which offers insights into potential biomedical applications. Notably, studies on GO sheets that were supplied intravenously and originally had just a few layers (5 nm) showed that these sheets remained in the bodies of living animals for 24 h, mostly localizing in the liver and spleen. Additionally, over 75 percent of the sheets injected into mice were excreted in urine within 24 h of leaving the body, primarily through the kidneys [52]. In a different study, Swiss albino mice were intravenously given (20 mg/kg) non-functionalized, carboxylated, and pegylated nanographene with 2–4 layers (about 100–200 nm) over the course of three months to determine its acute and long-term toxicity [53]. The results showed that no toxicity, organ damage, or substantial modifications were seen despite the continued presence of NGO-PEG inside the liver and spleen tissues throughout the course of the three-month timeframe. These results overwhelmingly point to the material’s potential for biodegradation. However, there are discrepancies in the published data on biodistribution and excretion pathways at the level of animal research. These variances are seen in a variety of organs and are directly related to the specific features of the synthesized NGOs [30,46,51,54,55,56]. This heterogeneity in findings underscores the complex interplay between the properties of the nanomaterials and their interaction with biological systems.
The third rationale for employing NGOs stems from their numerous physicochemical properties, which have been uncovered since their discovery in 2004. These characteristics include a remarkably large surface area, π−π stacking interactions, electrostatic and hydrophobic interactions, streamlined biofunctionalization, efficient water dispersibility, pH-sensitive zeta potential, distinctive intrinsic optical properties, drug-loading capacities, controlled drug release in response to stimuli, applications in photodynamic therapy, and optical imaging functionalities [15,30,51,57]. This multifaceted range of properties positions NGOs as versatile candidates for a wide spectrum of biomedical applications.
The fourth incentive for utilizing NGOs is rooted in their capacity for stimuli-responsive drug and therapeutic release. This material has been extensively used in applications that respond to stimuli. It is noteworthy that several DDSs have been developed with the aid of NGOs to enable the regulated release of medications or therapeutic chemicals in response to internal or external stimuli. These stimuli include variables such as pH, glutathione concentrations, light, magnetic and electric fields, and temperature changes [58,59,60]. The versatility of NGOs in responding to diverse triggers enhances their potential for tailored drug delivery strategies.
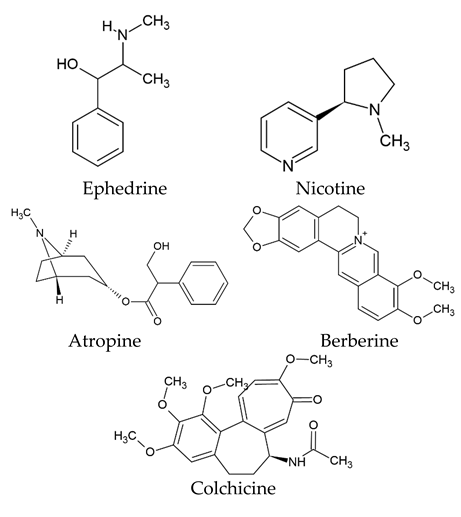
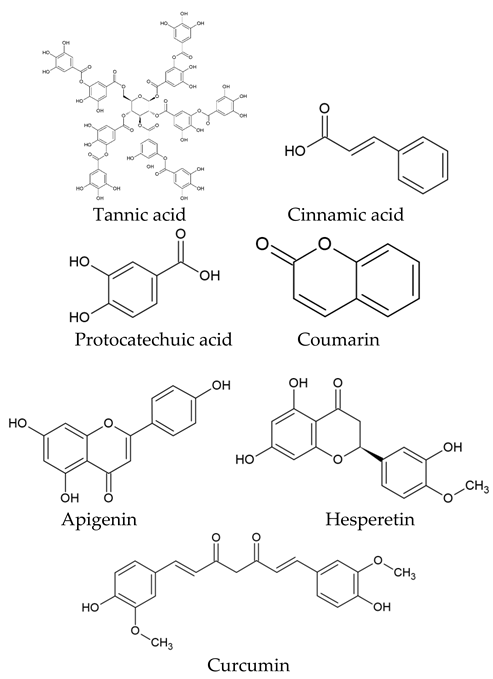
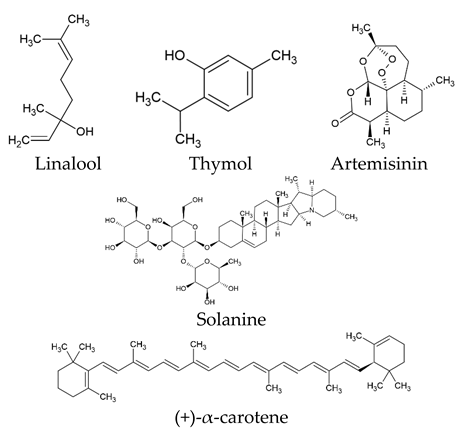
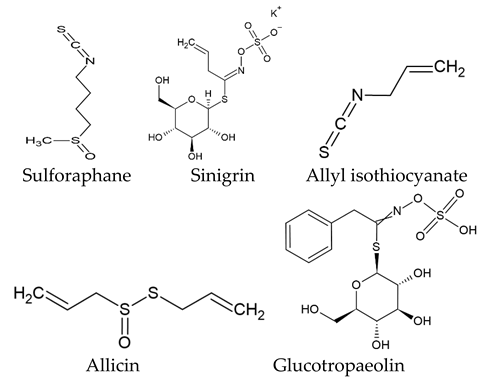
The world of nature provides a variety of helpful chemicals originating from numerous living species, such as plants, fungi, and bacteria, when it comes to medicinal agents. Natural products, also known as natural chemicals or secondary metabolites, take on a crucial role in the context of plants. These chemicals, which are referred to as secondary metabolites, are mostly tiny organic molecules with a variety of chemical configurations. Their categorization is frequently dependent on the precise paths they follow throughout the synthesis [61]. Therefore, these natural products can be classified into four major groups: alkaloids (including pyrrolidines, pyrrolizidines, pyridines, tropanes, isoquinolines, indoles, quinolines, and the terpenoids and steroids), phenolic compounds (including phenolic acids, flavonoids, stilbenes, tannins, and lignans), terpenoids (monoterpenes, sesquiterpenes, diterpenes, sesterpenes, and triterpenes), and glucosinolates (sulfur-containing compounds) [62]. In Table 1, several examples of these compounds with their chemical structures are presented. Another division into three major groups includes terpenoids, which are polymeric isoprene derivatives biosynthesized from acetate via the mevalonic acid pathway; phenolics, which contain one or more hydroxylated aromatic rings; and alkaloids, which are nitrogen-containing compounds that are not proteins and are biosynthesized from amino acids [63]. Natural products display a variety of biological and pharmacological actions, making them excellent candidates for the treatment of an array of disorders. These actions include, but are not limited to, a wide range of purposes such as anticancer and antimicrobial activities, and the encouragement of bone formation. The rich spectrum of biological and pharmacological activities exhibited by natural products underscores their potential in developing therapeutic interventions for a variety of ailments [63,64,65,66,67,68,69,70,71,72,73,74,75,76]. Data from the Food and Drug Administration (FDA) emphasize the importance of these natural compounds and highlight their crucial role in the development of therapeutic medications for human use. Notably, almost 40% of the authorized medicinal compounds used in clinical applications have natural product origins or inspiration. It is interesting to note that this tendency is especially noticeable in the area of anticancer therapy, where over 74% of therapeutic agents have their origins in or are inspired by natural substances [70]. Paclitaxel, a popular treatment for breast cancer, serves as an instructive example of how natural compounds may be used to treat cancer. Initially extracted from the bark of Taxus brevifolia, paclitaxel has been sold under the trade name Taxol® since 1993. To address the need on a worldwide basis, it is currently manufactured synthetically on a larger scale. Because of this substance’s exceptional anticancer effectiveness, chemotherapy has come to rely heavily on it [77,78]. This is an example of how using natural ingredients may lead to important therapeutic improvements. In fact, as shown in Table 2, natural substances are not only used in medical settings, but are also being studied through clinical studies [70,79]. Table 3 also provides a thorough overview of the factors involved in treatment techniques and lists the benefits and drawbacks of using natural vs. synthetic remedies.

Table 1.
Major classes of plant-derived natural products with their chemical structures.

Table 2.
Plant-derived natural products approved for clinical applications and clinical trials in the treatment of various diseases [70,79].

Table 3.
Advantages and disadvantages of natural and synthetic drugs [80,81,82,83,84].
Our main objectives in this review are to investigate surface functioning, drug-loading methods, and approaches to achieving controlled drug release patterns. The focus is on possible pharmaceutical and medical uses, particularly in relation to bacterial infections, cancer, and bone regeneration therapy. Our review’s main focus is the use of DDSs created by NGOs constructed using medicinal compounds derived from plant-derived substances. It is notable that, as far as we are aware, no review paper has yet been published that focuses on the delivery systems designed for natural therapies using NGO materials. Our study seeks to provide useful insight and a thorough grasp of this developing topic by filling this research gap.
2. Surface Functionality of NGO
Due to its oxygen functions, graphene oxide can be functionalized more easily than other carbon nanomaterials [85]. According to the literature, there are several variations of this functionalization process. For instance, Han et al. [86] divided functionalization techniques for graphene oxide-based materials used in gene and drug delivery into organic and inorganic functionalization, each having distinct subcategories (as illustrated in Figure 3a). Additionally, as shown in Figure 3b, Guo et al. [87] classified the surface functionalization techniques into covalent, non-covalent, plasma hydrogenation, and nanoparticle functionalization. As shown in Figure 4, multiple techniques may be used in the context of NGOs to implement various functionalization methodologies for applications in biomedicine, tissue engineering, drug delivery, and functional material development. This large range of functionalization options illustrates the flexibility and versatility of NGOs for a variety of applications.
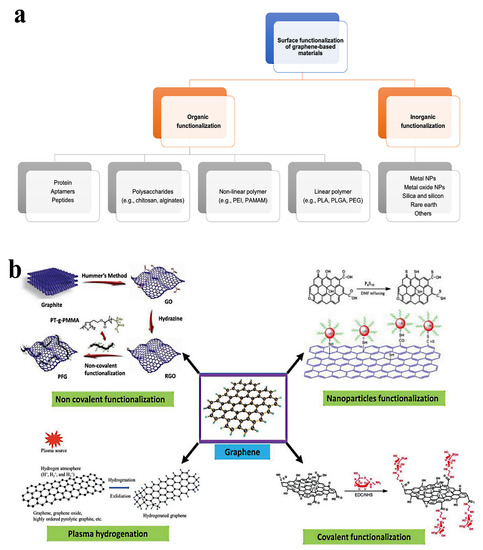
Figure 3.
The surface functionalization strategies for GO-based nanomaterial. (a) Surface functionalization methods for graphene oxide-based nanostructures: organic and inorganic functionalization. Reproduced from Ref. [86], e-Century Publishing Corporation, an open-access source. (b) Surface functionalization approaches for graphene oxide-based nanostructures: non-covalent, covalent, nanoparticles, and plasma hydrogen functionalization. Adapted from Ref. [87]. This is an open-access article distributed under the terms of the Creative Commons CC BY license, published by Advanced Biology, John Wiley and Sons.
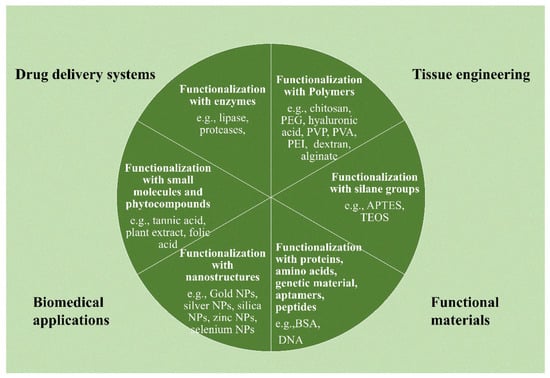
Figure 4.
Surface functionalization strategies can be possible for the functionalization of graphene oxide-based nanostructures: functionalization with polymers; with silane functional groups; with proteins, genetic materials, amino acids, peptides, and aptamers; with nanostructures, small molecules, and phyto-compounds; and with enzymes.
The possibility of functionalizing NGOs to create medicine delivery systems is demonstrated in this section with several examples. For instance, Pei et al. [88] showed a co-delivery strategy for two anticancer medicines using a PEGylated-functionalized NGO. In the first stage, NGO containing carboxylic acid groups underwent functionalization with four-armed PEG-NH2 by creating a covalent amide connection. The PEGylated nanoparticles were then coupled with N-hydroxysuccinimide (NHS) and 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) to deliver cisplatin. Following that, more doxorubicin was loaded, which was made possible by a stacking interaction (Figure 5-1).
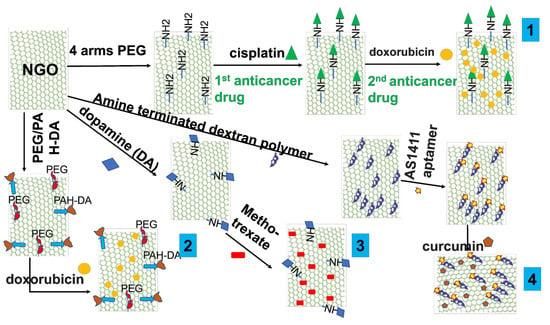
Figure 5.
Schematic illustration of feasible functionalization of NGO for cancer delivery designs. (1) Schematic illustration for co-delivery of cisplatin and doxorubicin for cancer therapy [88]. (2) Schematic illustration for combined chemo and photothermal therapy to overcome drug resistance [89]. (3) Schematic illustration for methotrexate anticancer drug [90]. (4) Schematic illustration for targeted delivery system by aptamer with curcumin natural model anticancer therapy [91].
A smart pH-responsive drug delivery system using NGO was also reported by Feng et al. [89] in order to combat drug resistance using a combination of chemo and photothermal treatment. In this design, PEG and positively charged poly(allylamine hydrochloride) (PAH) were added to NGO to modify it. 2,3-Dimethylmaleic anhydride (DA) was used to further modify PAH (Figure 5-2). The doxorubicin-loaded functionalized NGO then demonstrated a pH-responsive capacity to cause cell death in both drug-sensitive MCF-7/ADR cells and wild-type MCF-7/WT cells. Another noteworthy example is the work of Masoudipour et al. [90], who constructed a dopamine-conjugated NGO by EDC/NHS coupling chemistry. The antitumor medication methotrexate was delivered using this combination as a nanocarrier. The delivery method provided a targeted and tailored approach for drug administration by specifically targeting the DA receptor in human breast cancer cell lines that were positive (Figure 5-3). Furthermore, Alibolandi et al. [91] reported covalently functionalized NGO with an amine-terminated dextran (EDA-DEX) polymer using EDC reagent. Then, the functionalized NGO was coupled to the AS1411 aptamer and curcumin was loaded with the assistance of π−π stacking interactions. When tested against the cancer cells 4T1 and MCF-7, this complete design demonstrated noteworthy results, including effective cellular uptake and strong anticancer effects (Figure 5-4).
In fact, the growing problem of organisms’ resistance to conventional antibiotics has become a major obstacle. To overcome this issue, researchers are now looking into NGOs’ use in medication delivery systems. For instance, Erdal et al. [92] described a process that included grafting porous poly(ε-caprolactone) (PCL) scaffolds with cellulose-derived NGO on their surfaces. The localized delivery of the antibiotic ciprofloxacin was the goal of this concept. Meanwhile, Lu et al. [93] demonstrated the potential of GO nanosheets with amine functionalization for powerful antibacterial applications. Polyethylenimine (PEI)-functionalized NGO was designed to transport the antisense walR plasmid in a work by Wu et al. [94]. The proposed method successfully inhibited Enterococcus faecalis bacteria, demonstrating the promise of functionalized NGOs for focused antimicrobial therapies. Another noteworthy instance involves the creation of a strategy for photodynamic therapy to treat microorganisms. In this instance, curcumin-functionalized graphene quantum dots were used [95]. Pseudomonas aeruginosa, methicillin-resistant Staphylococcus aureus (MRSA), Escherichia coli, and Candida albicans were only some of the pathogens that this system showed improved antimicrobial activity against. It is important to note that materials based on NGO, even those with very small dimensions, such as quantum dots, have displayed astounding efficiency against viruses. This relevance is illustrated by a study that showed functionalized graphene quantum dots and boric acid have the potential to inhibit the highly pathogenic human coronavirus (HCoV-229E) by impairing both viral entrance and reproduction processes [96]. In a previous study, Kim et al. [97] described a delivery method intended particularly to target and control hepatitis C. This strategy made use of NGO conjugated with DNAzyme (deoxyribose) and loaded onto its surface. GO materials provide specific benefits for use in bone regeneration therapy in addition to having superior electrical and mechanical characteristics in comparison to other materials. These benefits are primarily attributable to their innate capacity to support various functionalities. These include giving simple access to facilities for fabrication, the development of several composites, and the construction of scaffolds, all of which contribute to their enormous potential in the field of biomedicine [98,99]. The use of the NGO matrix is central to three widely used approaches in the field of bone regeneration medicine: coatings, controlled release of medicines, and composites [99]. For example, electrospun scaffolds were produced by integrating graphene oxide dots into polymers (such as polylactic acid (PLA) or PCL) during the electrospinning process. This novel strategy is intended to improve mechanical qualities, as well as encourage osteobioactivity [100]. In another instance, researchers added GO nanosheets to methacrylated gelatin (GelMA)-based scaffolds to help human mesenchymal stem cells produce bone [101]. Additionally, it was shown that functionalized scaffolds with NGO on the surface allowed for effective ciprofloxacin loading and release for a longer period of time depending on the predominance of π−π interactions between the antibiotic and NGO; thus, using NGO has led to enhanced compressive strength and bioactivity of the scaffolds [92]. Furthermore, the release of the medication displayed a regulated pattern that was significantly impacted by pH levels when doxorubicin was incorporated into polyethylene oxide/chitosan/GO nanofibrous scaffolds. A share of 40% of the drug was released within 72 h, indicating a substantial pH dependence in the release kinetics [102].
3. Drug Loading and Drug Release Profile
3.1. Drug-Loading Strategies
3.1.1. Drug Solubility
Due to their effective drug-loading capacities, GO and its derivatives exhibit possibilities in the field of medication delivery. However, it is difficult to successfully load medicines with high solubility onto these materials [103]. The two-dimensional aromatic surface of GO and its derivatives makes them ideal for adsorbing pharmaceuticals that have poor solubility using methods such π−π stacking and hydrophobic interactions [104]. An advantageous aspect of utilizing GO-based materials in controlled drug release is their capacity to pass through cell membranes, coupled with their substantial specific surface area [105]. This feature opens up a wide range of possible connections with tailored medication delivery systems. Consequently, a workable method includes releasing medications that have been placed on GO sheets by putting them in biological fluids. The hydroxyl and epoxy groups on GO can form hydrogen bonds with other molecules, while the carboxyl group gives the surface of the compound a negative charge. Because of this quality, GO-based compounds are easily dispersed in polar solvents including water. By adding polymers and other nanoparticles to the surfaces of graphene nanoparticles and their derivatives, one can increase their effectiveness in DDSs [106]. Loading drugs with significant solubility, in particular strong hydrophilic agents, presents challenges due to the lack of robust interactions [103]. This limitation severely limits the possibility of using graphene and its analogs as carriers for highly soluble water-based medications without the use of surface functionalization techniques.
3.1.2. Drug-Loading Approaches
The technique of incorporating pharmaceutical substances or active ingredients (APIs) into a carrier matrix, such as nanoparticles, microparticles, liposomes, or implants, with the goal of delivering them to specific areas inside the body is known as drug loading. For drug loading, a variety of methods are used, including physical encapsulation, co-precipitation, adsorption, coating, solvent evaporation/extraction, and supercritical fluid technology. The above processes make it possible to provide therapeutic ingredients in a focused and regulated manner. The primary objective of incorporating a drug into a carrier material such as carbon, NGO, or mesoporous silica nanoparticles is to maintain the therapeutic agent within the carrier and facilitate its delivery to a particular target site where its therapeutic effects are required. As it affects fundamental characteristics such as loading capacity, the interaction between therapeutic molecules (such as drugs) and NGO is vital in this context. The pharmacological substances must possess these characteristics in order to achieve regulated release patterns and elicit the appropriate therapeutic response. Currently, methods such as molecular dynamics simulation (MDS) are used to investigate how the drug interacts with the carrier substance (such as NGO). Understanding and improving the drug-loading process requires an understanding of binding energies, hydrogen bonds, and dynamic behavior, all of which are rendered possible by MDS. For instance, MDS can shed light on the most efficient ways to load drugs onto NGO when evaluating a medication such as mitoxantrone [107]. It is crucial to remember that GO, which may be used to load drugs, is made up of layers that are formed of six-atom rings organized in a honeycomb structure. This makes GO a genuine planar aromatic macromolecule [17,108]. The basal plane of GO sheets is rich in hydroxyl and epoxy groups, whereas the edges are abundant with carboxyl functional groups [109]. The distinctive functional groups and distinctive hexagonal carbon structural composition contribute to the diverse surface chemistry of GO. Covalent and non-covalent bonds can be created because of this chemistry [110]. The loading technique for pharmaceuticals with NGO may be divided into three main categories: non-covalent bonds, covalent bonds, and mixtures of these two methods (Figure 6). For a wide range of therapeutic substances, including drugs and genetic materials, the non-covalent method of drug loading onto NGO/GO materials has been thoroughly investigated. These studies show that simple physisorption processes, such as π−π stacking (which is thought to exert a considerable force on these materials) or hydrophobic contacts, are the main drug-loading mechanisms that are used to attach drugs to NGO/GO materials. These interactions help to significantly adsorb pharmaceuticals onto GO-based materials, which leads to large drug-loading capabilities [15,111]. The presence of free π electrons on GO’s surface facilitates distinctive interactions with molecules [112]. Drugs and therapeutics can either be water-soluble or hydrophobic (which is a substantial class). Therefore, the free electrons in GO provide a hydrophobic area that is crucial for loading hydrophobic therapeutics through van der Waals forces [113]. Additionally, interactions between medicines and NGO that form hydrogen bonds allow for the process of drug loading [107]. These many interactions, which involve both hydrogen bonds and hydrophobic forces, highlight how adaptable GO and its derivatives are as efficient carriers for a variety of therapeutic treatments. In fact, the loading process is mostly carried out by the adsorption of drugs or other therapeutic molecules onto NGO/GO, which is fueled by a variety of interactions. These interactions include weak and strong van der Waals forces, hydrogen bonds, hydrophobic contacts, and π−π stacking interactions. Together, these pressures make it easier to successfully include pharmaceuticals in NGO/GO carriers. It is also important to note that the covalent bonding strategy is a successful method for medication loading. Between medicines and GO, covalent bonds can develop, offering solid and reliable adhesion [88,114]. As an example, branching polyethyleneimine and NGO-functionalized PEG were used as carriers to covalently load the chemotherapy drug cisplatin [115]. Another work by Wei Zhuang et al. demonstrates a dual strategy using both adsorption and covalent attachment for loading the cancer-targeting medication paclitaxel (PTX) onto functionalized NGO [116]. NGO is an excellent vehicle for combination therapy in the treatment of illness because of its large specific surface area, which allows for a substantial loading capacity. In a work by Pei et al. [88], a novel application is shown where two anticancer medications are coupled on NGO-PEG using a covalent approach. NGO-PEG-Pt is created by conjugating the first medication, cisplatin (Pt), through an amino-bonding process. The second drug, doxorubicin (DOX), was then loaded by a non-covalent method utilizing π−π interactions, creating the dual drug delivery system NGO-PEG-Pt/Dox.
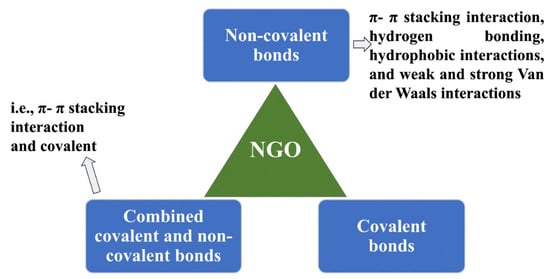
Figure 6.
Possible loading approaches of drugs/therapeutic molecules onto NGO and GO-based materials. There are three approaches: non-covalent bonds (π−π stacking interaction, hydrogen bonding, hydrophobic interactions, and weak and strong van der Waals interactions), covalent bonds (such as amino, amide bonding), and combined non-covalent and covalent bonds (π−π stacking interaction and amino covalent bonds) [15,88,111,115,116].
3.2. Release from Nanographene Oxide
When a drug is contained inside a nanographene oxide, its release is controlled by the rate at which the drug is released from the graphene oxide [117]. Drug solubility, drug dispersion inside the nanocarrier, bonding interactions, release rate, coatings, and other variables are among the many elements that influence the release of pharmaceuticals. The final profile of drug release from the carrier system is greatly influenced by these variables taken together.
Triggering Drug Release from Nanographene Oxide
Materials based on NGO/GO provide a variety of options for producing flexible drug release effects. Both internal and external cues are used to exercise control over and maintain therapeutic advantages from these effects [15,59]. Drug release from NGO-based materials can be governed using a variety of stimulus-responsive release methods. Each of these systems contributes to tailored and controlled drug release patterns, and they may be designated as internal, external, or mixed effects (Table 4).

Table 4.
Summary of possible controlled responsive release of drugs from NGO.
A large number of research studies conducted in the framework of NGOs have focused on cancer therapy, including the use of both natural and approved chemotherapy drugs. A collection of research results from diverse studies is shown in Table 5 and Table 6, emphasizing the possibility of drug loading and controlled release made possible by NGO-based systems.

Table 5.
Summary of studies on the loading and release of NGO-based materials incorporating approved anticancer drugs.

Table 6.
Summary of studies on loading and release of NGO-based materials incorporating natural agents for different diseases.
The drug release profiles that are seen in research involving NGOs show signs of controlled and sustained drug release. This has significant implications since it can improve patient compliance by delivering a consistent therapeutic dose and tailoring the therapy’s effects. These positive research findings have significant potential for the creation of more effective and customized therapies. As a result, the delivery of drugs is becoming safer and more efficient in the field of medicine.
Even though these results are motivating, it is vital to recognize that further research and clinical trials are required. Regarding NGOs, these next initiatives are crucial for properly understanding the long-term safety, general effectiveness, and possible uses of synthetic drugs. These studies will advance our knowledge of the therapeutic applications of these cutting-edge drug delivery technologies for the treatment of different diseases. This extensive research will ultimately open the door for the appropriate and knowledgeable integration of NGO-based remedies into medical practice.
4. Nanographene Oxide for Natural Anticancer Therapy
Recent studies have emphasized a number of beneficial outcomes that result from the use of graphene-based materials in anticancer applications. These processes result in a variety of outcomes, including the induction of apoptosis for anticancer effects, acting as critical nanocarriers that promote cellular uptake and internal release within cells and tissues, and enabling the transport of specific molecules for uses such as imaging, photodynamic/photothermal therapy, and targeted site delivery (Figure 7) [143,144]. Here, we give a summary of the achieved active targeting ligands used in the development of targeted delivery systems using GO nanomaterials. Peptides, small molecules (such as folic acid, a frequently utilized ligand), proteins, and antibodies are the available ligands for active cancer targeting through non-covalent and covalent bonding (Table 7). The delivery of natural medicines through NGO is a growing area of research interest. Although there are currently very few studies that have reported the use of natural medicinal agents via NGO, it is hoped that more thorough investigations and attempts in this regard will be launched in the future.
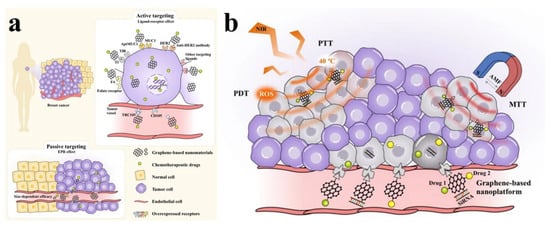
Figure 7.
The schematic presentation shows the active and passive targeting based on graphene-based nanomaterials. (a) The active site targeting ligands displays the direct attachment with many site-specific receptors, overexpressing on the surface of cancer cells (in this case breast cancer) or tumor vessels, and the targeting effects through receptor-mediated endocytosis and the enhanced permeability and retention (EPR) effect. (b) A multifunctional platform established by graphene oxide-based nanomaterials, showing the ability to simultaneously load many therapeutics (drugs and genes), and allow the photothermal therapy (PTT), photodynamic therapy (PDT), magnetothermal therapy (MTT), alternating magnetic field (AMF), and generation of reactive oxygen species (ROS) at the same time. Reproduced from [144], an open-access article, published under a Creative Commons Attribution 4.0 International License, Springer Nature.

Table 7.
Achieved active targeting ligands used in the development of targeted delivery systems using GO nanomaterials.
Curcumin, a natural polyphenol produced from the rhizomes of turmeric (Curcuma longa), has in fact shown extraordinary anticancer properties. An aptamer was used to decorate a dextran-functionalized NGO to actively target cancer cells in research by Alibolandi et al. [91]. Dextran, a biocompatible polymer, was added covalently to the surface of NGO sheets as part of the design. The AS1411 aptamer was also conjugated to this dextran-modified NGO to enable active site targeting. Following that, the system was filled with curcumin thanks to stacking interactions. The high loading capacity of 29 wt% produced by this method highlights how well drugs are loaded onto NGO. The proposed nanoconstruction design effectively internalized into 4T1 and MCF-7 cells, two types of cancer cells that overexpress nucleolin. The increased internalization resulted in noticeably stronger cytotoxic effects. The potential for developing cutting-edge drug delivery systems that take advantage of natural substances and nanomaterials for improved therapeutic outcomes in cancer treatment is demonstrated by the combination of the targeting strategy using aptamer and the loading of curcumin onto the functionalized NGO. A nanoformulation including GO−gold nanoparticles−curcumin was also prepared by Al-Ani et al. [153]. In vitro testing of this nanoformulation was conducted on colon cancer cells. Based on the study’s findings, this formulation promoted the transport of curcumin into cells and caused cytotoxicity in HT-29 and SW-948 colon cancer cell lines via activating apoptosis through redox. This shows the possibility of integrating graphene-based materials such as GO, gold nanoparticles, and curcumin to produce efficient nanoformulations with the capacity to target and elicit devastating effects on certain cancer cell types. A significant advancement was made possible by the establishment of a dual nanoformulation that combines the well-known anticancer drug doxorubicin with the natural anticancer ingredient curcumin. The main objectives of this formulation were to reduce the adverse effects of doxorubicin while concurrently increasing its therapeutic effectiveness [154]. The AGS human gastric cancer cell line, the PC3 prostate cancer cell line, and the A2780 ovarian cancer cell line were among the cancer cell lines used by the researchers to assess the anticancer properties of this dual formulation. The results of the experiment revealed numerous important discoveries. Both doxorubicin and curcumin can be transported by nanoformulation because of their high drug-loading capacities. Additionally, the medicines’ release patterns were pH-dependent, with the highest release taking place at pH 5.5. Contrary to the other cell lines examined, PC3 cells showed higher sensitivity. Due to the fact that only a portion of the loaded drugs were released, the researchers reached the conclusion that using photodynamic therapy may improve the therapeutic effects even more. This approach shows the possibility of integrating many therapeutic compounds into a single nanoformulation in order to maximize efficacy and reduce side effects. The natural flavonoid quercetin, known for its high antioxidant qualities and well-documented anticancer effects, has drawn considerable interest in the field of medication administration. Researchers are exploring its possible use in various drug delivery systems because of its unique features and potential health advantages. In a study on the delivery of quercetin, 0.4 µm carboxylated-GO sheets were utilized as nanocarriers to deliver quercetin to PC3 prostate cancer cells. This approach demonstrated regulated release mechanisms that were affected by both pH sensitivity and temperature. High cellular internalization, controlled release profile, and efficient cytotoxicity against the intended cancer cells were among the significant properties of the nanoformulation [155]. Likewise, Tiwari et al. [156] developed a co-delivery system. In this system, GO functionalized with polyvinylpyrrolidone polymer was used to produce a nanocarrier that contained both the antioxidant quercetin and the cancer fighter gefitinib. The goal of this hybrid nanoformulation was to give ovarian cancer cells a synergistic boost. According to the findings, the co-delivery technology dramatically increased toxicity when compared to administering each drug individually and free natural components. Comparing PA-1 ovarian cancer cells to IOSE-364 normal ovarian epithelial cells made this enhanced cytotoxicity clear. Protocatechuic acid (PCA) and chlorogenic acid (CA) were combined in a delivery nanoformulation by Buskaran et al. [157]. A PEG-functionalized NGO was used in the formulation approach for the delivery system. EDC/NHS coupling chemistry was used during the synthesis to join the carboxylic acid groups of NGO with those of PEG4000 to create PEG-NGO. Then, via adsorption, this PEG-NGO was employed as a carrier to encapsulate both PCA and CA. Folic acid was then added to the resulting functionalized material, creating a folic acid-modified NGO that enhanced the active targeting of cancer cells. To evaluate cellular absorption, the researchers also created a FITC-labeled folic acid-functionalized NGO. In the nanoformulation, PCA and CA made up around 23.82% and 19.55%, respectively. Notably, after approximately 120 and 130 h, respectively, the release of PCA and CA appeared to be completed. Changes in pH (7.4 and 4.8) affected these release patterns. The researchers highlighted the impact of these parameters on release kinetics by attributing the variations in drug release to factors such as drug arrangement on NGO nanocarriers and drug size. Studies on cellular absorption employing FITC-labeled folic acid-functionalized NGO demonstrated clear uptake in HepG2 hepatocellular cancer cells. Additionally, as compared to separate substances and free natural agents, the combined nanoformulation demonstrated notable cytotoxicity. The computed IC50 values for PCA and CA were 40.78 ± 1.92 g/mL and 43.61 ± 1.74 µg/mL, respectively. Further examination of the cell death pathway revealed that HepG2 cells treated with the developed nanosystem induced late apoptosis, G2/M phase cell cycle arrest, depolarized mitochondrial membrane potential, and enhanced generation of reactive oxygen species. Scientists designed an actively targeted nanoformulation for chemo-photothermal synergistic cancer treatment using GO nanosheets and a berberine derivative [131]. The suggested strategy attempted to utilize both the near-infrared (NIR) laser application and the acidic microenvironment of the tumor for dual-responsive medication release. Through an amide reaction, S1411 aptamers were attached to the surface of GO nanosheets to enable active cancer targeting. The endocytosis of the AS1411-GO nanosheets into cancer cells was considerably aided by the conjugation of AS1411 aptamers. The AS1411-conjugated nanosheets were then covered with a berberine 9-O-pyrazole alkyl derivative (as an anticancer drug) via conjugate π−π stacking. The loading capacity of 68.3% was remarkable for this technique. Release experiments were carried out under simulated physiologically normal settings (pH 7.4) and under simulated tumor-acidic conditions (pH 5.3). The findings showed a larger cumulative release at the acidic pH (18%) compared to the physiological pH (12%), indicating a more regulated circulatory release pattern and a faster release in the acidic tumor environment. Notably, the cumulative release effect was much improved when laser radiation was used. An important goal was active targeting, and the AS1411-GO/B3 nanoformulation showed higher accumulation in tumor tissues, especially in A549 cells. Additionally, after being exposed to 128 µg/mL of the nanoformulation, the viability of A549 cells dropped to 81%. The therapeutic response was further enhanced by the addition of laser treatment, leading to higher cytotoxicity and more prominent cell-killing effects. This work serves as an example of the possibility of utilizing sophisticated nanomaterials, such as GO, in conjunction with exact targeting techniques and synergistic therapeutic mechanisms to obtain improved cancer therapy results. GO nanoscrolls were used as carriers for administering gallic acid in a study by Sontakke et al. [158] According to the study’s findings, the pH factor had an impact on how much gallic acid was loaded onto and released from the nanoscrolls. Comparing the A549 lung cancer cell line to the HEK293 healthy cell line, the developed GONS-GA system showed increased toxicity. This approach may be able to efficiently target and destroy cancer cells while preserving healthy cells, according to the differential cytotoxicity. In another study [141], Juniperus squamata root essential oil was loaded onto polyvinylpyrrolidone-functionalized GO (GO-PVP) through π−π and hydrophobic interactions. This nanoformulation improved anticancer efficacy, especially against MDA-MB-231 human breast cancer cells.
The accumulated evidence highlights the possibility of using NGO or GO nanoparticles in combination with naturally occurring substances generated from plants for anticancer applications across diverse cancer types. This strategy provides a promising way to increase the therapeutic benefits of cancer-fighting medicines and specifically target cancer cells. The interaction of natural products with nanomaterials creates new opportunities for innovative cancer therapy. Several technological considerations are advised for the development of these smart delivery systems for natural medicinal compounds, which are presented in Table 8.

Table 8.
Active cancer targeting by combining GO-based materials and natural therapeutics and some remarks.
5. Antibacterial Delivery Systems
Nanomaterials based on graphene, such as GO, reduced graphene oxide (rGO), and their composites with metals, metal oxides, and polymers, show promise as antibacterial agents. Because of their antibacterial action, this has attracted a lot of attention from researchers who are investigating their potential as active therapeutic nanomedicine platforms against pathogenic microorganisms [159]. These materials’ potential to fight infectious disorders, especially those brought on by bacteria resistant to many drugs, is being studied [160]. Several crucial considerations must be made while exploring the use of graphene-based nanomaterials as nano-antimicrobial therapeutics. Enhancing effectiveness and lowering possible toxicity require surface modification and functionalization. It is feasible to maximize these nanoparticles’ antibacterial effectiveness while reducing any negative consequences by adding inorganic nanostructures, proteins, or polymers to their surfaces [161]. In previous research, the antibacterial potential of GO was shown to be effective against a number of bacterial species, including Klebsiella pneumoniae, E. coli, and Pseudomonas aeruginosa [162]. A concentration-dependent pattern of the antibacterial actions was seen. In particular, this study discovered that GO was especially successful in eliminating Klebsiella pneumoniae. A 250.0 µg/mL GO therapy resulted in an in vitro eradication rate of more than 95%. These results are important not just in vitro but potentially have implications in vivo. These results demonstrate that GO itself is an appropriate material with an antimicrobial function. The capacity of GO to physically damage bacterial membranes is what gives it its first antibacterial capabilities [163].
It has been progressively reported that the NGO’s antibacterial activity is highly promising in the biomedical field, and some studies have shown possible antibacterial mechanisms. In a study by Robb et al. [164], GO antibacterial mechanisms were reported to be unclear, but three hypotheses were presented about the aspects that contribute to antibacterial activity: (i) the edges of the 2D sheets dissolve and destroy the bacterial membrane; (ii) the sheets cover the cell, preventing access to essential nutrients; and (iii) GO results in metabolic oxidative damage to the cells. In their experiments, they indicated that NGO significantly inhibits the development of E. coli, but the micrometer-sized GO exhibits no significantly inhibited growth, illustrating the antibacterial efficacy dependent on size features. Additionally, when lipid membrane vesicles are exposed to GO or NGO, a change in lipid composition and tension is made possible. Pulingam et al. revealed the differences in interactions between GO and Gram-positive and Gram-negative bacteria [165]. When GO interacts with bacteria, especially when concentrations rise, bacteria lose membrane integrity by increasing the release of lactose dehydrogenase (LDH) into the reaction medium. The surface morphology of bacterial culture-treated GO demonstrates apparent differences in the GO mechanism of action against Gram-positive and Gram-negative bacteria, where cell entrapment was primarily observed for Gram-positive S. aureus and Enterococcus faecalis, but membrane disruption was found for Gram-negative E. coli and Pseudomonas aeruginosa, which is related to the physical contact. By means of ATR-FTIR analysis, the results reveal that treated bacterial cells display changes in fatty acids, amide I and amide II of proteins, peptides, and amino acid regions compared to untreated bacterial cells. In an earlier study by Tu et al. [166], they demonstrated experimentally and theoretically that GO and NGO sheets induce the degradation of the inner and outer cell membranes of E. coli, as well as reduce their growth viability. They demonstrated that the process occurs in three general steps using TEM observations. By using molecular dynamics simulations, they were able to identify the precise atomic details involved. They demonstrated that NGO can enter cell membranes and remove significant amounts of phospholipids from them due to the powerful dispersion interactions between nanosheets and lipid molecules. Therefore, disruptive extraction enables nanosheets’ potential mechanisms for cytotoxicity and antibacterial actions against bacteria. Similar to this, Mao et al. [167] investigated the processes behind the cytotoxicity and antibacterial activity of GO in stimulating theoretical and experimental methods. Through theoretical research, they showed the cellular interaction of nanosheets with different levels of oxidation. Four typical states of nanosheet−membrane interactions were seen based on the degree of oxidation and size of graphene nanosheets: superstructures made of sandwiched graphene, graphene that adheres to the membrane, graphene that is spread out over the membrane, and hemispheric vesicle structures. Intriguingly, these stages were found in the experimental data. Importantly, their studies revealed that graphene nanosheets with a higher degree of oxidation may cause a greater degree of irregular membrane disruption, damaging the membrane’s integrity. This is due to these nanosheets, especially when they have increased edge length. As a result of cellular internalization and the cytotoxicity and nanosheets with bacteria, these actions aid in realizing the main antibacterial cytotoxicity actions.
GO has a number of benefits over other commonly utilized nanomaterials when it comes to manufacturing antibacterial drug delivery systems and performing as an antibacterial [161]. The following characteristics best describe these benefits: (1) physical and chemical oxidation has an impact on GO’s antibacterial mechanism, which reduces the resistance of many bacteria; (2) low-concentration doses of GO have a mild cytotoxic effect on mammalian cells; and (3) GO is easier to process, can be produced in large quantities, and is less expensive to produce than other carbon nanomaterials [168,169]. Here we discus some examples showing the advantages of GO-based material delivery systems for drug-resistant bacteria. To treat bacteria that are resistant to antibiotics, near-infrared (NIR) photothermal antibacterial materials based on NGOs were developed. These materials have the biocompatibility property of bovine serum albumin (BSA) and photothermal molecules of aggregation-induced emission fluorogen (AIEgen) [170]. In comparison to single-model phototherapy, the results demonstrate a superior antibacterial effect (>99%) against amoxicillin-resistant E. coli and S. aureus, making this delivery system a desirable choice for tracking antibiotic resistance in bacteria. In order to treat Porphyromonas gingivalis using antimicrobial photodynamic treatment, Pourhajibagher et al. [171] developed and assessed a targeted biotheragnostic system based on DNA-aptamer-NGO. As demonstrated by flow cytometry, they reached the conclusion that the proposed system binds specifically to P. gingivalis, and that PDT exposure causes photoactivation and the production of ROS, which greatly reduce P. gingivalis numbers. In addition, the DNA-aptamer-NGO-mediated photodynamics display a significant reduction in the biofilms and metabolic activity of bacteria as compared to the control group. When treated bacteria were exposed to radiation, a slight rise in the number of apoptotic cells and significant changes in the expression of specific genes (related to bacterial biofilm formation and response to oxidative stress) occurred at the molecular level. For vancomycin-resistant Enterococcus (VRE), a hybrid nanocomposite using GO consisting of vancomycin and photosensitizer phthalocyanine was made via π–π stacking interactions to achieve a phototherapy-based combination strategy [172]. Treating bacteria with this delivery system and application of photothermal and photodynamic treatment result in almost 2–3 logs of bacterial reduction when tested in vitro. The treatment also exhibits infection regression on VRE-infected BALB/C mice, along with accelerating wound healing and negligible damage to normal cells and major organs.
Regarding natural delivery systems, a smart medicine delivery system was developed employing NGO-capped PEG loaded with Nigella sativa seed extract [173]. The antibacterial efficacy against E. coli and S. aureus was examined. Investigation into the antibacterial mode of action revealed that the GO-based system efficiently penetrated bacterial cytoplasmic and nucleic acid membranes. As a result, the integrity of the cell wall was damaged, nucleic acids were harmed, and the permeability of the cell wall increased. In another study, the antibacterial efficacy of a composite made of curcumin and GO against methicillin-resistant S. aureus (MRSA) was examined [163]. The results demonstrated this composite’s effective antibacterial ability against MRSA, with activity at doses of less than 2 µg/mL. The fact that this antibacterial activity was attained with no fibroblast damage is significant since it emphasizes the antibacterial action’s selectivity and suggests safe use. Yan et al. [174] focused on creating an efficient antibacterial composite nanomaterial, which resulted in an excellent antimicrobial composite nanomaterial. They accomplished it through π−π stacking interactions to load polydopamine-curcumin onto the surface of GO. This nanocomposite system showed excellent promise for photodynamic treatment in terms of its antibacterial activities, resulting in an incredible decrease in Gram-positive S. aureus by four orders of magnitude and an excellent bactericidal rate of 99.99%. This effectiveness was directly related to the production of ROS brought on by white light irradiation (405–780 nm). Bacterial outer membranes were shattered and bacterial cells were deformed as a result of the ROS. This work emphasizes the significance of integrating polydopamine-curcumin with GO to create a viable photodynamic antibacterial material. In a study by Cacaci et al. [175], curcumin was used to formulate an antibacterial strategy that targeted the biofilm formation of Candida parapsilosis on medical equipment. Healthcare-associated infections are complicated by Candida parapsilosis biofilm production, which adds to worldwide public health problems. The results of the study showed that the combination of curcumin and GO demonstrated a range of antibacterial activities, including antiplanktonic, antiadhesive, and antibiofilm capabilities. Cell viability dropped by 72% as a result of the therapy. After 72 h, the release of extracellular substances significantly decreased by 85%. These findings demonstrate that the combination of curcumin and GO offers a vital defense against microbial colonization of medical equipment. By mitigating the threat of biofilm-related infections, this approach addresses a critical concern in healthcare settings. In order to develop antibacterial coatings suited for diverse applications, Oves et al. [176] tested the effects of a composite system made of copper, curcumin, and GO on the contact killing of microbes and disinfection. Zone inhibition and scanning electron microscopy examinations of the nanocomposite, which was made into paintable coatings, showed antimicrobial characteristics through contact-killing processes. At a concentration of 25 µg/mL, they discovered that the nanocomposite exhibited the strongest antibacterial effects against E. coli with a zone of inhibition of 24 ± 0.50 mm and Pseudomonas aeruginosa with a zone of inhibition of 18 ± 0.25 mm. Additionally, it was found that the bactericidal concentration was 160 µg/mL and the minimal inhibitory concentration (MIC) was 80 µg/mL. This study concluded that when coated on various contact surfaces such as medical devices, the nanocomposite material exhibits outstanding antibacterial activity against E. coli and P. aeruginosa. This suggests that the likelihood of nosocomial infections, which are diseases acquired in healthcare settings, may be significantly reduced. In a study conducted by Shamsi and colleagues [177], the efficacy of gallic acid-loaded GO against several MRSA strains was investigated using various methods. The results showed that this nanocomposite has antibacterial activity equivalent to that of the drug cefoxitin, specifically against MRSA and methicillin-sensitive S. aureus (MSSA) strains at concentrations of less than 150 µg/mL. The nanocomposite also showed a considerably delayed response against both MRSA strains, especially in terms of enhanced inhibition after exposure over a duration of 8 to 24 h. Importantly, the outcomes showed that the nanocomposite was more potent than free natural gallic acid. According to this work, the gallic acid-loaded GO nanocomposite has potential antibacterial properties against MRSA strains. However, further research is necessary to acquire a greater understanding of the potential applications and mechanisms of action of this nanocomposite as a potential antibacterial agent. Another instance is the research by Arfat et al. [178], which generated antibacterial nanopackaging films by combining clove essential oil and GO nanosheets with PLA. S. aureus and E. coli germs were effectively combated by these films. With this novel technique, it may be possible to develop packaging materials that are effectively antibacterial and aid in maintaining the quality and safety of diverse items. In an earlier study, Liu et al. [179] incorporated the garlic-derived chemical allicin into composites of chitosan, polyvinyl alcohol, and GO to create nanofibrous membranes. The controlled release of allicin from the membrane provided this composite with both sustained releasing behavior and significant antibacterial activity. This system’s antibacterial properties were particularly efficient against S. aureus, indicating its potential utility in tissue engineering and antibacterial wound dressings, among other applications. Successful fabrication of a multifunctional covering for orthopedic implants was demonstrated by Xia-Ying et al. [180]. They used GO to apply berberine alkaloid to the surface of a biomedical titanium implant. The outcomes showed that this coating has extraordinary antibacterial properties, which were especially noticeable in its enhanced antibacterial activity against S. aureus. As opposed to the antibacterial activity of berberine alone, this improvement was due to the synergistic impact of GO and berberine. These results, which emphasize the potential of this coated implant to reduce bacterial infections after implantation, were verified by both in vitro and in vivo experiments. Ning et al. [181] also suggested a targeted strategy to prevent MRSA from forming biofilms. They used GO, functionalized it using an aptamer, and loaded it with berberine to create a bifunctional complex. A DNA aptamer known as PBP2 was coupled with berberine to create an aptamer/berberine bifunctional complex. Through π−π interactions, this complex was then adsorbed onto GO. By lowering cell-surface attachment, the approach drastically reduced MRSA biofilm development. Additionally, the simultaneous delivery of berberine and the PBP2a-targeted aptamer via the GO platform revealed efficient antibiofilm action.
Undoubtedly, the existing findings collectively indicate that using natural therapeutic agents in combination with GO-based materials is a viable strategy to treat microbial diseases, including but not limited to bacterial infections. This new sector has significant promise to tackle problems with biofilm development and antibiotic resistance. Several important suggestions may be provided, as shown in Table 9, for further research and use of natural therapies with GO-based materials.

Table 9.
Antimicrobial delivery strategies based on GO-based materials and natural therapeutics and some remarks.
6. Bone Regeneration Systems
Recent developments in bone tissue engineering have been impressive, with an emphasis on creating solid connections between grafts and bone components. Osteoconduction and osseointegration are both aspects of connection that are essential for facilitating the repair and regeneration of damaged bone tissues [182]. Therefore, the improvement in bone tissue regeneration depends on the delivery of signaling molecules and the sustained release of bone growth factors. Here are a few instances that highlight the advantages of NGO composite scaffolds and provide an overview of the function of GO-based materials in this field of research. Shen et al. [183] evaluated the effects of adding NGO sheets to a photopolymerizable poly-D, L-lactic acid (PDLLA)/polyethylene glycol hydrogel in order to promote the long-term release of chondroinductive growth factor (TGF-3). Increased initial mechanical strength, sustained release of TGF-3 (for up to 4 weeks), high cell viability of human bone marrow-derived mesenchymal stem cells (hBMSCs), and higher expression of chondrogenic genes (including aggrecan, collagen type II, SOX9, and cartilage matrix production) as compared to GO-free scaffolds of TGF-3, are all made possible by the incorporation of sheets. In vivo experiments show that hBMSC-seeded TGF-3/PDLLA exhibits a much greater cartilage matrix than their TGF-3/PDLLA counterparts without GO. Bone morphogenetic protein-2 is continuously released through NGO-based administration, which affects the activation of the NF-B signal transduction system, as stated by Zhong et al. [184]. Effectively, bone morphogenetic protein 2 (BMP2) was adsorbed onto GO flakes with diameters ranging from 81.1 nm to 45,749.7 nm. The findings showed that GO continuously prolonged BMP2 release (for at least 40 days). The biocompatibility of chondrocytes or bone marrow stem cells was not significantly altered by BMP2-GO. Evaluated in vivo in a rat model of osteoarthritis also showed a considerable improvement, suggesting that GO may be able to regulate the release of this protein. They concluded that by promoting continuous BMP2 release, the BMP2-GO approach might reduce the activity of the NF-B pathway. The study carried out by La et al. [185] reported the GO delivery of substance P (SP) and BMP2 coated on titanium (Ti) implants. According to their research, the coated GO permits constant release of BMP2. It is anticipated that the combined delivery of BMP2 and SP will improve osteointegration of dental or orthopedic implants since it promotes bone growth on Ti placed in mice calvaria.
Natural substances have significant potential as medicinal products because of their wide range of pharmacological and therapeutic effects. Researchers need to address issues with microbial infections, the possibility of malignant development, and other unfavorable consequences that might obstruct the scaffold’s successful integration inside the human body when it comes to bone production scaffolds and implants. The following paragraphs provide an overview of the literature on this particular study issue, despite the fact that it is currently limited. Faraji et al. [186] constructed an electrospun scaffold by electrospinning a nanofibrous scaffold (PCL/GO/Q) after first loading quercetin onto PCL. The scaffold’s minimal or insignificant toxicity toward cell development is a critical factor in bone repair. According to the results, PCL/GO/Q had a favorable effect on NIH/3T3 fibroblast cells, which demonstrated high cell viability (about 95%), proliferation, and adhesion. This shows that the material is favorable for cell development and wound healing. Additionally, even at low concentrations, the scaffolds showed noticeably improved antibacterial activities, which are crucial for avoiding bacterial infections. The use of artificial bone substitutes as a potential treatment for defects in bones led Guo et al. to develop a novel approach for treating bone defects with artificial bone substitutes [187]. They created a hybrid scaffold known as GO-BAI/DBM using GO nanosheets, flavonoid baicalin (BAI), and demineralized bone matrix (DBM). By steadily releasing the bioactive BAI via GO nanoparticles, this scaffold intends to promote bone repair. According to their findings, baicalin was released over a period of 14 days at a rate of 81.7%. Additionally, the scaffold had two effects: it increased bone marrow stromal cells’ ability to differentiate into osteoblasts and caused macrophages to take on a pro-healing M2 phenotype as a result of the baicalin released. The scaffold increased tissue integration and the inflammatory microenvironment after being implanted in rat subcutaneous tissue. The scaffold has most impressively shown remarkable abilities in fostering considerable bone repair in rats over a 12-week duration. These results show the possibility of scaffolds that combine natural molecules with diverse effects. Through the provision of new replacements with advantageous multifunctional properties, this creative technique shows potential for enhancing bone tissue engineering. An osteoinductive scaffold designed by Kashte et al. [188] used a naturally occurring bioactive substance to stimulate the production of osteoblasts. PCL-GO-CQ is the outcome of the layer-by-layer construction of this scaffold utilizing electrospun PCL, Cissus quadrangularis extract, and GO. According to their research, the synergistic effects of GO and plant extract increased the potential for osteoblastic differentiation, osteoconduction, and osteoinduction. This exciting combination has significant potential for use in tissue engineering and bone regeneration.
Despite the previously conducted research, little is known about how NGO formulations combined with natural substances might improve bone regeneration and their use in daily life. When developing intelligent delivery systems for this particular utilization, researchers need to consider a number of important benefits and considerations (Figure 8 and Table 10).
Figure 8.
The advantages of using delivery systems for natural agents for bone regeneration filed.

Table 10.
Bone regeneration by combining GO-based materials and natural therapeutics and some remarks.
7. Conclusions and Future Prospects
Over the years, materials based on graphene and NGO have demonstrated tremendous promise for use in biomedical applications. Although the initial focus of the research was mostly on cancer therapy, these materials have been shown to have a wide range of applications in tissue engineering, treatment of microbial infections, and a variety of biomedical and pharmaceutical settings. Their attractive physicochemical characteristics, which include a large surface area and a range of structural attributes (stability, mechanics, electrical properties, flexibility, and conductivity), fit well with biomedical demands. These materials have increased therapeutic agent efficacy, surface functionality, efficient drug carrier capability, high drug-loading capacity, biocompatibility, controlled drug release, bioimaging, and multifunctional nanoplatforms. Therefore, they are particularly suitable nanomaterials for the controlled release of drugs in a variety of disease therapies. This review provides an overview of the many surface modification techniques that are necessary to develop multifunctional systems that enhance biocompatibility, loading, release, and targeting efficiency of therapeutic medicines. It also reviews drug-loading and control methods used for NGO and GO-based materials, primarily anticancer medicines, highlighting their outstanding potential as carriers for efficient drug delivery. The final sections of this review provide insight into different approaches to delivery using natural therapeutic substances, including those for bone regeneration, bacterial infections, and cancer. To address the limited published evidence and establish a realistic approach for clinical therapy across a range of diseases, noteworthy ideas are offered to broaden insights into building pharmaceutical formulations integrating natural medicinal agents with NGO. In this context, we highlighted the advantages of employing NGO to deliver herbal remedies in the treatment of bacterial infections, cancer, and bone regeneration, providing exciting new research directions. NGO-based nanocarriers’ remarkable aqueous solubility enables the efficient distribution of naturally insoluble substances while maintaining their therapeutic potential. The problem of the restricted solubility of natural substances can be solved using this method. Additionally, NGO offers a perfect platform for combining several natural compounds to achieve synergistic effects. One major benefit is its ability to encapsulate and load a variety of compounds, each with different pharmacological activities, including alkaloids, flavonoids, phenolics, and essential oils. With the help of non-covalent interactions (such as π−π stacking, hydrogen bonding, and hydrophobic interactions), covalent bonding, and a mixture of both, the NGO’s high surface area facilitates effective medication administration. Additionally, using natural agents into scaffolds or implants has considerable potential in specialized sectors such as bone regeneration therapy. The scaffold’s NGO material allows these integrated natural therapeutic compounds to release gradually over a lengthy period of time (such as weeks). By simultaneously encouraging bone cell development and exerting antimicrobial properties to prevent infections in the early stages of implantation, for example, this dual action strategy can have a variety of advantages. Furthermore, it may have osteoinductive and antifibrosis effects, promoting bone repair following the first implantation. Through innovative techniques for delivery, potent monotherapy can be achieved by utilizing these latent therapeutic properties of natural medical products in combination with the NGO nanocarrier.
Author Contributions
Conceptualization, K.A.; writing original draft, K.A. and F.S.; writing—review and editing, K.A. and B.S.K.; Supervision, B.S.K.; funding acquisition, B.S.K. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Brain Pool (BP) program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science and ICT (Grant No. 2022H1D3A2A02090010).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
No data were created in this review.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Rauti, R.; Musto, M.; Bosi, S.; Prato, M.; Ballerini, L. Properties and behavior of carbon nanomaterials when interfacing neuronal cells: How far have we come? Carbon 2019, 143, 430–446. [Google Scholar] [CrossRef]
- Silva, G.A. Nanotechnology approaches to crossing the blood-brain barrier and drug delivery to the CNS. BMC Neurosci. 2008, 9 (Suppl. S3), S4. [Google Scholar] [CrossRef] [PubMed]
- Mahor, A.; Singh, P.P.; Bharadwaj, P.; Sharma, N.; Yadav, S.; Rosenholm, J.M.; Bansal, K.K. Carbon-Based Nanomaterials for Delivery of Biologicals and Therapeutics: A Cutting-Edge Technology. C 2021, 7, 19. [Google Scholar] [CrossRef]
- Patel, K.D.; Singh, R.K.; Kim, H.-W. Carbon-based nanomaterials as an emerging platform for theranostics. Mater. Horiz. 2019, 6, 434–469. [Google Scholar] [CrossRef]
- Armenta, S.; Esteve-Turrillas, F.A. Carbon-Based Nanomaterials in Analytical Chemistry. In Handbook of Smart Materials in Analytical Chemistry; Wiley: Hoboken, NJ, USA, 2019; pp. 345–374. [Google Scholar]
- Sheoran, K.; Kaur, H.; Siwal, S.S.; Saini, A.K.; Vo, D.-V.N.; Thakur, V.K. Recent advances of carbon-based nanomaterials (CBNMs) for wastewater treatment: Synthesis and application. Chemosphere 2022, 299, 134364. [Google Scholar] [CrossRef]
- Dutta, V.; Verma, R.; Gopalkrishnan, C.; Yuan, M.-H.; Batoo, K.M.; Jayavel, R.; Chauhan, A.; Lin, K.-Y.A.; Balasubramani, R.; Ghotekar, S. Bio-Inspired Synthesis of Carbon-Based Nanomaterials and Their Potential Environmental Applications: A State-of-the-Art Review. Inorganics 2022, 10, 169. [Google Scholar] [CrossRef]
- Maiti, D.; Tong, X.; Mou, X.; Yang, K. Carbon-Based Nanomaterials for Biomedical Applications: A Recent Study. Front. Pharmacol. 2019, 9, 1401. [Google Scholar] [CrossRef]
- Kościk, I.; Jankowski, D.; Jagusiak, A. Carbon Nanomaterials for Theranostic Use. C 2022, 8, 3. [Google Scholar] [CrossRef]
- Kumar, N.; Chamoli, P.; Misra, M.; Manoj, M.K.; Sharma, A. Advanced metal and carbon nanostructures for medical, drug delivery and bio-imaging applications. Nanoscale 2022, 14, 3987–4017. [Google Scholar] [CrossRef] [PubMed]
- Chakroborty, S.; Sahoo, S.K. Carbon-Based Nanomaterials in Drug Delivery Systems. In Environmental Applications of Carbon Nanomaterials-Based Devices; Wiley: Hoboken, NJ, USA, 2021; pp. 375–394. [Google Scholar]
- Wu, H.-C.; Chang, X.; Liu, L.; Zhao, F.; Zhao, Y. Chemistry of carbon nanotubes in biomedical applications. J. Mater. Chem. 2010, 20, 1036–1052. [Google Scholar] [CrossRef]
- Shanbhag, V.K.L.; Prasad, K.S. Graphene based sensors in the detection of glucose in saliva—A promising emerging modality to diagnose diabetes mellitus. Anal. Methods 2016, 8, 6255–6259. [Google Scholar] [CrossRef]
- He, Y.; Hu, C.; Li, Z.; Wu, C.; Zeng, Y.; Peng, C. Multifunctional carbon nanomaterials for diagnostic applications in infectious diseases and tumors. Mater. Today Bio 2022, 14, 100231. [Google Scholar] [CrossRef] [PubMed]
- Hoseini-Ghahfarokhi, M.; Mirkiani, S.; Mozaffari, N.; Abdolahi Sadatlu, M.A.; Ghasemi, A.; Abbaspour, S.; Akbarian, M.; Farjadian, F.; Karimi, M. Applications of Graphene and Graphene Oxide in Smart Drug/Gene Delivery: Is the World Still Flat? Int. J. Nanomed. 2020, 15, 9469–9496. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nat. Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Joung, D.; Zhai, L.; Das, S.; Khondaker, S.I.; Seal, S. Graphene based materials: Past, present and future. Prog. Mater. Sci. 2011, 56, 1178–1271. [Google Scholar] [CrossRef]
- Singh, R.K.; Kumar, R.; Singh, D.P. Graphene oxide: Strategies for synthesis, reduction and frontier applications. RSC Adv. 2016, 6, 64993–65011. [Google Scholar] [CrossRef]
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef] [PubMed]
- Zaaba, N.I.; Foo, K.L.; Hashim, U.; Tan, S.J.; Liu, W.-W.; Voon, C.H. Synthesis of Graphene Oxide using Modified Hummers Method: Solvent Influence. Procedia Eng. 2017, 184, 469–477. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Song, J.; Wang, X.; Chang, C.-T. Preparation and Characterization of Graphene Oxide. J. Nanomater. 2014, 2014, 276143. [Google Scholar] [CrossRef]
- Kumar, V.; Kumar, A.; Lee, D.J.; Park, S.S. Estimation of Number of Graphene Layers Using Different Methods: A Focused Review. Materials 2021, 14, 4590. [Google Scholar] [CrossRef]
- Wang, K.; Ruan, J.; Song, H.; Zhang, J.; Wo, Y.; Guo, S.; Cui, D. Biocompatibility of Graphene Oxide. Nanoscale Res. Lett. 2011, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Liao, K.-H.; Lin, Y.-S.; Macosko, C.W.; Haynes, C.L. Cytotoxicity of Graphene Oxide and Graphene in Human Erythrocytes and Skin Fibroblasts. ACS Appl. Mater. Interfaces 2011, 3, 2607–2615. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Yang, S.-T.; Liu, J.-H.; Dong, E.; Wang, Y.; Cao, A.; Liu, Y.; Wang, H. In vitro toxicity evaluation of graphene oxide on A549 cells. Toxicol. Lett. 2011, 200, 201–210. [Google Scholar] [CrossRef]
- Ou, L.; Song, B.; Liang, H.; Liu, J.; Feng, X.; Deng, B.; Sun, T.; Shao, L. Toxicity of graphene-family nanoparticles: A general review of the origins and mechanisms. Part. Fibre Toxicol. 2016, 13, 57. [Google Scholar] [CrossRef]
- Xu, S.; Zhang, Z.; Chu, M. Long-term toxicity of reduced graphene oxide nanosheets: Effects on female mouse reproductive ability and offspring development. Biomaterials 2015, 54, 188–200. [Google Scholar] [CrossRef]
- Wu, W.; Yan, L.; Wu, Q.; Li, Y.; Li, Q.; Chen, S.; Yang, Y.; Gu, Z.; Xu, H.; Yin, Z.Q. Evaluation of the toxicity of graphene oxide exposure to the eye. Nanotoxicology 2016, 10, 1329–1340. [Google Scholar] [CrossRef] [PubMed]
- Kiew, S.F.; Kiew, L.V.; Lee, H.B.; Imae, T.; Chung, L.Y. Assessing biocompatibility of graphene oxide-based nanocarriers: A review. J. Control. Release 2016, 226, 217–228. [Google Scholar] [CrossRef]
- Zhang, X.; Yin, J.; Peng, C.; Hu, W.; Zhu, Z.; Li, W.; Fan, C.; Huang, Q. Distribution and biocompatibility studies of graphene oxide in mice after intravenous administration. Carbon 2011, 49, 986–995. [Google Scholar] [CrossRef]
- Zhang, H.; Peng, C.; Yang, J.; Lv, M.; Liu, R.; He, D.; Fan, C.; Huang, Q. Uniform Ultrasmall Graphene Oxide Nanosheets with Low Cytotoxicity and High Cellular Uptake. ACS Appl. Mater. Interfaces 2013, 5, 1761–1767. [Google Scholar] [CrossRef]
- Teixeira-Costa, B.E.; Andrade, C.T. Chitosan as a Valuable Biomolecule from Seafood Industry Waste in the Design of Green Food Packaging. Biomolecules 2021, 11, 1599. [Google Scholar] [CrossRef] [PubMed]
- Pinho de Aguiar, K.L.N.; Frias de Oliveira, P.; Elias Mansur, C.R. A comprehensive review of in situ polymer hydrogels for conformance control of oil reservoirs. Oil Gas Sci. Technol. Rev. IFP Energ. Nouv. 2020, 75, 8. [Google Scholar] [CrossRef]
- Bie, B.-J.; Zhao, X.-R.; Yan, J.-R.; Ke, X.-J.; Liu, F.; Yan, G.-P. Dextran Fluorescent Probes Containing Sulfadiazine and Rhodamine B Groups. Molecules 2022, 27, 6747. [Google Scholar] [CrossRef]
- Jirkovec, R.; Erben, J.; Samkova, A.; Chaloupek, J.; Chvojka, J. The effect of the electrospinning setup on the surface energy of polycaprolactone nanofibre layers. J. Ind. Text. 2022, 51, 8517S–8527S. [Google Scholar] [CrossRef]
- Yang, K.; Gong, H.; Shi, X.; Wan, J.; Zhang, Y.; Liu, Z. In vivo biodistribution and toxicology of functionalized nano-graphene oxide in mice after oral and intraperitoneal administration. Biomaterials 2013, 34, 2787–2795. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, K.; Feng, L.; Liu, Z. In vitro and in vivo behaviors of dextran functionalized graphene. Carbon 2011, 49, 4040–4049. [Google Scholar] [CrossRef]
- Xu, M.; Zhu, J.; Wang, F.; Xiong, Y.; Wu, Y.; Wang, Q.; Weng, J.; Zhang, Z.; Chen, W.; Liu, S. Improved In Vitro and In Vivo Biocompatibility of Graphene Oxide through Surface Modification: Poly(Acrylic Acid)-Functionalization is Superior to PEGylation. ACS Nano 2016, 10, 3267–3281. [Google Scholar] [CrossRef] [PubMed]
- Vila, M.; Portolés, M.T.; Marques, P.A.A.P.; Feito, M.J.; Matesanz, M.C.; Ramírez-Santillán, C.; Gonçalves, G.; Cruz, S.M.A.; Nieto, A.; Vallet-Regi, M. Cell uptake survey of pegylated nanographene oxide. Nanotechnology 2012, 23, 465103. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.T.; Tabakman, S.M.; Liang, Y.; Wang, H.; Sanchez Casalongue, H.; Vinh, D.; Dai, H. Ultrasmall Reduced Graphene Oxide with High Near-Infrared Absorbance for Photothermal Therapy. J. Am. Chem. Soc. 2011, 133, 6825–6831. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Chatterjee, K. Poly(Ethylene Glycol) Functionalized Graphene Oxide in Tissue Engineering: A Review on Recent Advances. Int. J. Nanomed. 2020, 15, 5991–6006. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, S.; Li, Y.; Wang, M.; Shi, P.; Huang, X. Covalent Functionalization of Graphene Oxide with Biocompatible Poly(ethylene glycol) for Delivery of Paclitaxel. ACS Appl. Mater. Interfaces 2014, 6, 17268–17276. [Google Scholar] [CrossRef]
- Mao, N.D.; Jeong, H.; Ngan Nguyen, T.K.; Loan Nguyen, T.M.; Vi Do, T.V.; Ha Thuc, C.N.; Perré, P.; Ko, S.C.; Kim, H.G.; Tran, D.T. Polyethylene glycol functionalized graphene oxide and its influences on properties of Poly(lactic acid) biohybrid materials. Compos. Part B Eng. 2019, 161, 651–658. [Google Scholar] [CrossRef]
- Yan, T.; Zhang, H.; Huang, D.; Feng, S.; Fujita, M.; Gao, X.D. Chitosan-Functionalized Graphene Oxide as a Potential Immunoadjuvant. Nanomaterials 2017, 7, 59. [Google Scholar] [CrossRef] [PubMed]
- Bussy, C.; Ali-Boucetta, H.; Kostarelos, K. Safety Considerations for Graphene: Lessons Learnt from Carbon Nanotubes. Acc. Chem. Res. 2013, 46, 692–701. [Google Scholar] [CrossRef]
- Zhang, B.; Wei, P.; Zhou, Z.; Wei, T. Interactions of graphene with mammalian cells: Molecular mechanisms and biomedical insights. Adv. Drug Deliv. Rev. 2016, 105, 145–162. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Kim, J.H. Synthesis, toxicity, biocompatibility, and biomedical applications of graphene and graphene-related materials. Int. J. Nanomed. 2016, 11, 1927–1945. [Google Scholar] [CrossRef] [PubMed]
- Liao, C.; Li, Y.; Tjong, S.C. Graphene Nanomaterials: Synthesis, Biocompatibility, and Cytotoxicity. Int. J. Mol. Sci. 2018, 19, 3564. [Google Scholar] [CrossRef]
- Bullock, C.J.; Bussy, C. Biocompatibility Considerations in the Design of Graphene Biomedical Materials. Adv. Mater. Interfaces 2019, 6, 1900229. [Google Scholar] [CrossRef]
- Rhazouani, A.; Gamrani, H.; El Achaby, M.; Aziz, K.; Gebrati, L.; Uddin, M.S.; Aziz, F. Synthesis and Toxicity of Graphene Oxide Nanoparticles: A Literature Review of In Vitro and In Vivo Studies. Biomed Res. Int. 2021, 2021, 5518999. [Google Scholar] [CrossRef]
- Jasim, D.A.; Boutin, H.; Fairclough, M.; Ménard-Moyon, C.; Prenant, C.; Bianco, A.; Kostarelos, K. Thickness of functionalized graphene oxide sheets plays critical role in tissue accumulation and urinary excretion: A pilot PET/CT study. Appl. Mater. Today 2016, 4, 24–30. [Google Scholar] [CrossRef]
- Sasidharan, A.; Swaroop, S.; Koduri, C.K.; Girish, C.M.; Chandran, P.; Panchakarla, L.S.; Somasundaram, V.H.; Gowd, G.S.; Nair, S.; Koyakutty, M. Comparative in vivo toxicity, organ biodistribution and immune response of pristine, carboxylated and PEGylated few-layer graphene sheets in Swiss albino mice: A three month study. Carbon 2015, 95, 511–524. [Google Scholar] [CrossRef]
- Dasari Shareena, T.P.; McShan, D.; Dasmahapatra, A.K.; Tchounwou, P.B. A Review on Graphene-Based Nanomaterials in Biomedical Applications and Risks in Environment and Health. Nano-Micro Lett. 2018, 10, 53. [Google Scholar] [CrossRef] [PubMed]
- Ema, M.; Gamo, M.; Honda, K. A review of toxicity studies on graphene-based nanomaterials in laboratory animals. Regul. Toxicol. Pharmacol. 2017, 85, 7–24. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, V.; Perini, G.; De Spirito, M.; Papi, M. Graphene oxide touches blood: In vivo interactions of bio-coronated 2D materials. Nanoscale Horiz. 2019, 4, 273–290. [Google Scholar] [CrossRef]
- Bai, R.G.; Husseini, G.A. Chapter 11—Graphene-based drug delivery systems. In Biomimetic Nanoengineered Materials for Advanced Drug Delivery; Unnithan, A.R., Sasikala, A.R.K., Park, C.H., Kim, C.S., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 149–168. [Google Scholar]
- Patil, T.V.; Patel, D.K.; Dutta, S.D.; Ganguly, K.; Lim, K.T. Graphene Oxide-Based Stimuli-Responsive Platforms for Biomedical Applications. Molecules 2021, 26, 2797. [Google Scholar] [CrossRef]
- Yang, K.; Feng, L.; Liu, Z. Stimuli responsive drug delivery systems based on nano-graphene for cancer therapy. Adv. Drug Deliv. Rev. 2016, 105, 228–241. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.K.; Seo, Y.-R.; Lim, K.-T. Stimuli-Responsive Graphene Nanohybrids for Biomedical Applications. Stem Cells Int. 2019, 2019, 9831853. [Google Scholar] [CrossRef]
- Delgoda, R. Pharmacognosy: Fundamentals, Applications and Strategies; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Stella Omokhefe, B. Secondary Metabolites from Natural Products. In Secondary Metabolites; Ramasamy, V., Suresh Selvapuram Sudalaimuthu, R., Eds.; IntechOpen: Rijeka, Croatia, 2022; Chapter 4. [Google Scholar]
- Kabera, J.N.; Semana, E.; Mussa, A.R.; He, X. Plant secondary metabolites: Biosynthesis, classification, function and pharmacological properties. J. Pharm. Pharmacol. 2014, 2, 377–392. [Google Scholar]
- Andrade, M.A.; Braga, M.A.; Cesar, P.H.S.; Trento, M.V.C.; Espósito, M.A.; Silva, L.F.; Marcussi, S. Anticancer Properties of Essential Oils: An Overview. Curr. Cancer Drug Targets 2018, 18, 957–966. [Google Scholar] [CrossRef]
- Herr, I.; Büchler, M.W. Dietary constituents of broccoli and other cruciferous vegetables: Implications for prevention and therapy of cancer. Cancer Treat. Rev. 2010, 36, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Mahavy, C.E.; Duez, P.; ElJaziri, M.; Rasamiravaka, T. African Plant-Based Natural Products with Antivirulence Activities to the Rescue of Antibiotics. Antibiotics 2020, 9, 830. [Google Scholar] [CrossRef] [PubMed]
- Miranda, L.L.; Guimaraes-Lopes, V.P.; Altoe, L.S.; Sarandy, M.M.; Melo, F.; Novaes, R.D.; Goncalves, R.V. Plant Extracts in the Bone Repair Process: A Systematic Review. Mediat. Inflamm. 2019, 2019, 1296153. [Google Scholar] [CrossRef]
- Mondal, A.; Gandhi, A.; Fimognari, C.; Atanasov, A.G.; Bishayee, A. Alkaloids for cancer prevention and therapy: Current progress and future perspectives. Eur. J. Pharmacol. 2019, 858, 172472. [Google Scholar] [CrossRef]
- Saghatelyan, T.; Tananyan, A.; Janoyan, N.; Tadevosyan, A.; Petrosyan, H.; Hovhannisyan, A.; Hayrapetyan, L.; Arustamyan, M.; Arnhold, J.; Rotmann, A.-R.; et al. Efficacy and safety of curcumin in combination with paclitaxel in patients with advanced, metastatic breast cancer: A comparative, randomized, double-blind, placebo-controlled clinical trial. Phytomedicine 2020, 70, 153218. [Google Scholar] [CrossRef]
- Seca, A.M.L.; Pinto, D. Plant Secondary Metabolites as Anticancer Agents: Successes in Clinical Trials and Therapeutic Application. Int. J. Mol. Sci. 2018, 19, 263. [Google Scholar] [CrossRef]
- Springob, K.; Kutchan, T.M. Introduction to the Different Classes of Natural Products. In Plant-Derived Natural Products: Synthesis, Function, and Application; Osbourn, A.E., Lanzotti, V., Eds.; Springer US: New York, NY, USA, 2009; pp. 3–50. [Google Scholar]
- Thoppil, R.J.; Bishayee, A. Terpenoids as potential chemopreventive and therapeutic agents in liver cancer. World J. Hepatol. 2011, 3, 228–249. [Google Scholar] [CrossRef]
- Angioni, A.; Barra, A.; Cereti, E.; Barile, D.; Coïsson, J.D.; Arlorio, M.; Dessi, S.; Coroneo, V.; Cabras, P. Chemical composition, plant genetic differences, antimicrobial and antifungal activity investigation of the essential oil of Rosmarinus officinalis L. J. Agric. Food Chem. 2004, 52, 3530–3535. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Orhan, I.E.; Banach, M.; Rollinger, J.M.; Barreca, D.; Weckwerth, W.; Bauer, R.; Bayer, E.A.; et al. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- De, R.; Kundu, P.; Swarnakar, S.; Ramamurthy, T.; Chowdhury, A.; Nair, G.B.; Mukhopadhyay, A.K. Antimicrobial activity of curcumin against Helicobacter pylori isolates from India and during infections in mice. Antimicrob. Agents Chemother. 2009, 53, 1592–1597. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.S.L.; Tan, L.T.-H.; Chan, K.-G.; Yap, W.H.; Pusparajah, P.; Chuah, L.-H.; Ming, L.C.; Khan, T.M.; Lee, L.-H.; Goh, B.-H. Resveratrol-Potential Antibacterial Agent against Foodborne Pathogens. Front. Pharmacol. 2018, 9, 102. [Google Scholar] [CrossRef] [PubMed]
- Weaver, B.A. How Taxol/paclitaxel kills cancer cells. Mol. Biol. Cell 2014, 25, 2677–2681. [Google Scholar] [CrossRef]
- Bernabeu, E.; Cagel, M.; Lagomarsino, E.; Moretton, M.; Chiappetta, D.A. Paclitaxel: What has been done and the challenges remain ahead. Int. J. Pharm. 2017, 526, 474–495. [Google Scholar] [CrossRef]
- Hashemi, H.; Hand, A.; Florentinus-Mefailoski, A.; Kerrigan, P.; Samuel, P.; Friedberg, J. Plants as medical tools. Cannabis Med. Asp. 2016, 9, 493–504. [Google Scholar]
- Dembitsky, V.M.; Dzhemileva, L.; Gloriozova, T.; D’Yakonov, V. Natural and synthetic drugs used for the treatment of the dementia. Biochem. Biophys. Res. Commun. 2020, 524, 772–783. [Google Scholar] [CrossRef]
- Khamjan, N.A.; Beigh, S.; Algaissi, A.; Megha, K.; Lohani, M.; Darraj, M.; Kamli, N.; Madkhali, F.; Dar, S.A. Natural and synthetic drugs and formulations for intravaginal HPV clearance. J. Infect. Public Health 2023, 16, 1471–1480. [Google Scholar] [CrossRef]
- Huygens, S.; Vellekoop, H.; Versteegh, M.; Santi, I.; Szilberhorn, L.; Zelei, T.; Nagy, B.; Tsiachristas, A.; Koleva-Kolarova, R.; Wordsworth, S.; et al. Cost-Effectiveness Analysis of Treating Patients With NTRK-Positive Cancer With the Histology-Independent Therapy Entrectinib. Value Health 2023, 26, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Atta, H.; Alzahaby, N.; Hamdy, N.M.; Emam, S.H.; Sonousi, A.; Ziko, L. New trends in synthetic drugs and natural products targeting 20S proteasomes in cancers. Bioorganic Chem. 2023, 133, 106427. [Google Scholar] [CrossRef] [PubMed]
- Ulrich-Merzenich, G.S. Combination screening of synthetic drugs and plant derived natural products—Potential and challenges for drug development. Synergy 2014, 1, 59–69. [Google Scholar] [CrossRef]
- Jiříčková, A.; Jankovský, O.; Sofer, Z.; Sedmidubský, D. Synthesis and Applications of Graphene Oxide. Materials 2022, 15, 920. [Google Scholar] [CrossRef]
- Han, X.M.; Zheng, K.W.; Wang, R.L.; Yue, S.F.; Chen, J.; Zhao, Z.W.; Song, F.; Su, Y.; Ma, Q. Functionalization and optimization-strategy of graphene oxide-based nanomaterials for gene and drug delivery. Am. J. Transl. Res. 2020, 12, 1515–1534. [Google Scholar] [PubMed]
- Guo, Z.; Chakraborty, S.; Monikh, F.A.; Varsou, D.-D.; Chetwynd, A.J.; Afantitis, A.; Lynch, I.; Zhang, P. Surface Functionalization of Graphene-Based Materials: Biological Behavior, Toxicology, and Safe-By-Design Aspects. Adv. Biol. 2021, 5, 2100637. [Google Scholar] [CrossRef]
- Pei, X.; Zhu, Z.; Gan, Z.; Chen, J.; Zhang, X.; Cheng, X.; Wan, Q.; Wang, J. PEGylated nano-graphene oxide as a nanocarrier for delivering mixed anticancer drugs to improve anticancer activity. Sci. Rep. 2020, 10, 2717. [Google Scholar] [CrossRef] [PubMed]
- Feng, L.; Li, K.; Shi, X.; Gao, M.; Liu, J.; Liu, Z. Smart pH-Responsive Nanocarriers Based on Nano-Graphene Oxide for Combined Chemo- and Photothermal Therapy Overcoming Drug Resistance. Adv. Healthc. Mater. 2014, 3, 1261–1271. [Google Scholar] [CrossRef] [PubMed]
- Masoudipour, E.; Kashanian, S.; Maleki, N. A targeted drug delivery system based on dopamine functionalized nano graphene oxide. Chem. Phys. Lett. 2017, 668, 56–63. [Google Scholar] [CrossRef]
- Alibolandi, M.; Mohammadi, M.; Taghdisi, S.M.; Ramezani, M.; Abnous, K. Fabrication of aptamer decorated dextran coated nano-graphene oxide for targeted drug delivery. Carbohydr. Polym. 2017, 155, 218–229. [Google Scholar] [CrossRef]
- Erdal, N.B.; Yao, J.G.; Hakkarainen, M. Cellulose-Derived Nanographene Oxide Surface-Functionalized Three-Dimensional Scaffolds with Drug Delivery Capability. Biomacromolecules 2019, 20, 738–749. [Google Scholar] [CrossRef]
- Lu, B.-Y.; Zhu, G.-Y.; Yu, C.-H.; Chen, G.-Y.; Zhang, C.-L.; Zeng, X.; Chen, Q.-M.; Peng, Q. Functionalized graphene oxide nanosheets with unique three-in-one properties for efficient and tunable antibacterial applications. Nano Res. 2021, 14, 185–190. [Google Scholar] [CrossRef]
- Wu, S.; Liu, Y.; Zhang, H.; Lei, L. Nano-graphene oxide with antisense walR RNA inhibits the pathogenicity of Enterococcus faecalis in periapical periodontitis. J. Dent. Sci. 2020, 15, 65–74. [Google Scholar] [CrossRef]
- Mushtaq, S.; Yasin, T.; Saleem, M.; Dai, T.; Yameen, M.A. Potentiation of Antimicrobial Photodynamic Therapy by Curcumin-loaded Graphene Quantum Dots. Photochem. Photobiol. 2022, 98, 202–210. [Google Scholar] [CrossRef]
- Łoczechin, A.; Séron, K.; Barras, A.; Giovanelli, E.; Belouzard, S.; Chen, Y.-T.; Metzler-Nolte, N.; Boukherroub, R.; Dubuisson, J.; Szunerits, S. Functional Carbon Quantum Dots as Medical Countermeasures to Human Coronavirus. ACS Appl. Mater. Interfaces 2019, 11, 42964–42974. [Google Scholar] [CrossRef]
- Kim, S.; Ryoo, S.-R.; Na, H.-K.; Kim, Y.-K.; Choi, B.-S.; Lee, Y.; Kim, D.-E.; Min, D.-H. Deoxyribozyme-loaded nano-graphene oxide for simultaneous sensing and silencing of the hepatitis C virus gene in liver cells. Chem. Commun. 2013, 49, 8241–8243. [Google Scholar] [CrossRef]
- Nanda, S.S.; Papaefthymiou, G.C.; Yi, D.K. Functionalization of Graphene Oxide and its Biomedical Applications. Crit. Rev. Solid State Mater. Sci. 2015, 40, 291–315. [Google Scholar] [CrossRef]
- Holt, B.D.; Wright, Z.M.; Arnold, A.M.; Sydlik, S.A. Graphene oxide as a scaffold for bone regeneration. WIREs Nanomed. Nanobiotechnol. 2017, 9, e1437. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Samanta, A.; Srivastava, R.K.; Hakkarainen, M. Nano-Graphene Oxide Functionalized Bioactive Poly(lactic acid) and Poly(ε-caprolactone) Nanofibrous Scaffolds. Materials 2018, 11, 566. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Xiang, S.; Lin, Z.; Li, E.N.; Yagi, H.; Cao, G.; Yocum, L.; Li, L.; Hao, T.; Bruce, K.K.; et al. Graphene oxide-functionalized nanocomposites promote osteogenesis of human mesenchymal stem cells via enhancement of BMP-SMAD1/5 signaling pathway. Biomaterials 2021, 277, 121082. [Google Scholar] [CrossRef]
- Ardeshirzadeh, B.; Anaraki, N.A.; Irani, M.; Rad, L.R.; Shamshiri, S. Controlled release of doxorubicin from electrospun PEO/chitosan/graphene oxide nanocomposite nanofibrous scaffolds. Mater. Sci. Eng. C 2015, 48, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Nan, X.; Shi, W.; Sun, Y.; Su, H.; He, Y.; Liu, X.; Zhang, Z.; Ge, D. Polydopamine-functionalized nanographene oxide: A versatile nanocarrier for chemotherapy and photothermal therapy. Nanotechnology 2017, 28, 295102. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.S.; Lee, M.Y.; Kong, W.H.; Do, I.H.; Hahn, S.K. Nano graphene oxide-hyaluronic acid conjugate for target specific cancer drug delivery. RSC Adv. 2014, 4, 14197–14200. [Google Scholar] [CrossRef]
- Campbell, E.; Hasan, M.T.; Pho, C.; Callaghan, K.; Akkaraju, G.R.; Naumov, A.V. Graphene Oxide as a Multifunctional Platform for Intracellular Delivery, Imaging, and Cancer Sensing. Sci. Rep. 2019, 9, 416. [Google Scholar] [CrossRef] [PubMed]
- Mahdavian, L. DFT studies of the drug carrier of anti-migraine (sumatriptan) on nano-graphene oxide (NGO) and graphene oxide/polyethylene glycol polymer nano-composite. Diam. Relat. Mater. 2020, 104, 107745. [Google Scholar] [CrossRef]
- Mirhosseini, M.M.; Khordad, R.; Vaseghi, B. Effect of hydrogen bonding on drug loading using a nanographene surface: A molecular dynamics study. Chin. J. Phys. 2019, 62, 99–105. [Google Scholar] [CrossRef]
- Huang, X.; Yin, Z.; Wu, S.; Qi, X.; He, Q.; Zhang, Q.; Yan, Q.; Boey, F.; Zhang, H. Graphene-based materials: Synthesis, characterization, properties, and applications. Small 2011, 7, 1876–1902. [Google Scholar] [CrossRef] [PubMed]
- Chua, C.K.; Pumera, M. Chemical reduction of graphene oxide: A synthetic chemistry viewpoint. Chem. Soc. Rev. 2014, 43, 291–312. [Google Scholar] [CrossRef]
- Khakpour, E.; Salehi, S.; Naghib, S.M.; Ghorbanzadeh, S.; Zhang, W. Graphene-based nanomaterials for stimuli-sensitive controlled delivery of therapeutic molecules. Front. Bioeng. Biotechnol. 2023, 11, 1129768. [Google Scholar] [CrossRef]
- Cho, Y.; Min, S.K.; Yun, J.; Kim, W.Y.; Tkatchenko, A.; Kim, K.S. Noncovalent Interactions of DNA Bases with Naphthalene and Graphene. J. Chem. Theory Comput. 2013, 9, 2090–2096. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Li, L.; Lee, W.B.; Ng, M.C. Structure of graphene and its disorders: A review. Sci. Technol. Adv. Mater. 2018, 19, 613–648. [Google Scholar] [CrossRef]
- Ghawanmeh, A.A.; Ali, G.A.M.; Algarni, H.; Sarkar, S.M.; Chong, K.F. Graphene oxide-based hydrogels as a nanocarrier for anticancer drug delivery. Nano Res. 2019, 12, 973–990. [Google Scholar] [CrossRef]
- Kim, H.; Lee, D.; Kim, J.; Kim, T.-i.; Kim, W.J. Photothermally Triggered Cytosolic Drug Delivery via Endosome Disruption Using a Functionalized Reduced Graphene Oxide. ACS Nano 2013, 7, 6735–6746. [Google Scholar] [CrossRef]
- Zhang, X.; Guo, L.; Sun, Z.; Han, D.; Zhao, L. Research on the Targeted Treatment of Squamous Cell Carcinoma by Nanographene Drug Delivery System. Adv. Mater. Sci. Eng. 2023, 2023, 6336569. [Google Scholar] [CrossRef]
- Zhuang, W.; He, L.; Wang, K.; Ma, B.; Ge, L.; Wang, Z.; Huang, J.; Wu, J.; Zhang, Q.; Ying, H. Combined Adsorption and Covalent Linking of Paclitaxel on Functionalized Nano-Graphene Oxide for Inhibiting Cancer Cells. ACS Omega 2018, 3, 2396–2405. [Google Scholar] [CrossRef]
- Zeng, Y.; Yang, Z.; Li, H.; Hao, Y.; Liu, C.; Zhu, L.; Liu, J.; Lu, B.; Li, R. Multifunctional Nanographene Oxide for Targeted Gene-Mediated Thermochemotherapy of Drug-resistant Tumour. Sci. Rep. 2017, 7, 43506. [Google Scholar] [CrossRef]
- Hu, H.; Yu, J.; Li, Y.; Zhao, J.; Dong, H. Engineering of a novel pluronic F127/graphene nanohybrid for pH responsive drug delivery. J. Biomed. Mater. Res. Part A 2012, 100A, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Xia, J.; Zhao, Q.; Liu, L.; Zhang, Z. Functional Graphene Oxide as a Nanocarrier for Controlled Loading and Targeted Delivery of Mixed Anticancer Drugs. Small 2010, 6, 537–544. [Google Scholar] [CrossRef]
- Alhourani, A.; Førde, J.-L.; Eichacker, L.A.; Herfindal, L.; Hagland, H.R. Improved pH-Responsive Release of Phenformin from Low-Defect Graphene Compared to Graphene Oxide. ACS Omega 2021, 6, 24619–24629. [Google Scholar] [CrossRef]
- Li, R.; Liu, C.; Wan, C.; Liu, T.; Zhang, R.; Du, J.; Wang, X.; Jiao, X.; Gao, R.; Li, B. A Targeted and pH-Responsive Nano-Graphene Oxide Nanoparticle Loaded with Doxorubicin for Synergetic Chemo-Photothermal Therapy of Oral Squamous Cell Carcinoma. Int. J. Nanomed. 2023, 18, 3309–3324. [Google Scholar] [CrossRef]
- Wen, H.; Dong, C.; Dong, H.; Shen, A.; Xia, W.; Cai, X.; Song, Y.; Li, X.; Li, Y.; Shi, D. Engineered Redox-Responsive PEG Detachment Mechanism in PEGylated Nano-Graphene Oxide for Intracellular Drug Delivery. Small 2012, 8, 760–769. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.; Lu, T.; Nong, Z.; Li, G.; Pan, X.; Wei, Y.; Yang, Y.; Wu, N.; Huang, J.; Pan, M.; et al. Reductive response and RGD targeting nano-graphene oxide drug delivery system. J. Drug Deliv. Sci. Technol. 2019, 53, 101202. [Google Scholar] [CrossRef]
- Yin, T.; Liu, J.; Zhao, Z.; Zhao, Y.; Dong, L.; Yang, M.; Zhou, J.; Huo, M. Redox Sensitive Hyaluronic Acid-Decorated Graphene Oxide for Photothermally Controlled Tumor-Cytoplasm-Selective Rapid Drug Delivery. Adv. Funct. Mater. 2017, 27, 1604620. [Google Scholar] [CrossRef]
- Havanur, S.; Batish, I.; Cheruku, S.P.; Gourishetti, K.; JagadeeshBabu, P.E.; Kumar, N. Poly(N,N-diethyl acrylamide)/functionalized graphene quantum dots hydrogels loaded with doxorubicin as a nano-drug carrier for metastatic lung cancer in mice. Mater. Sci. Eng. C 2019, 105, 110094. [Google Scholar] [CrossRef]
- Khoee, S.; Karimi, M.R. Dual-drug loaded Janus graphene oxide-based thermoresponsive nanoparticles for targeted therapy. Polymer 2018, 142, 80–98. [Google Scholar] [CrossRef]
- Xie, M.; Zhang, F.; Peng, H.; Zhang, Y.; Li, Y.; Xu, Y.; Xie, J. Layer-by-layer modification of magnetic graphene oxide by chitosan and sodium alginate with enhanced dispersibility for targeted drug delivery and photothermal therapy. Colloids Surf. B Biointerfaces 2019, 176, 462–470. [Google Scholar] [CrossRef]
- Bardajee, G.R.; Hooshyar, Z. Thermo/pH/magnetic-triple sensitive poly(N-isopropylacrylamide-co-2-dimethylaminoethyl) methacrylate)/sodium alginate modified magnetic graphene oxide nanogel for anticancer drug delivery. Polym. Bull. 2018, 75, 5403–5419. [Google Scholar] [CrossRef]
- Wang, L.; Yu, D.; Dai, R.; Fu, D.; Li, W.; Guo, Z.; Cui, C.; Xu, J.; Shen, S.; Ma, K. PEGylated doxorubicin cloaked nano-graphene oxide for dual-responsive photochemical therapy. Int. J. Pharm. 2019, 557, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Wang, C.; Zhang, S.; Feng, L.; Liu, Z. Photothermally Enhanced Photodynamic Therapy Delivered by Nano-Graphene Oxide. ACS Nano 2011, 5, 7000–7009. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Yan, J.; Long, S.; Xiong, H.; Wen, N.; Cai, S.; Wang, Y.; Peng, D.; Liu, Z.; Liu, Y. Tumor microenvironment and NIR laser dual-responsive release of berberine 9-O-pyrazole alkyl derivative loaded in graphene oxide nanosheets for chemo-photothermal synergetic cancer therapy. J. Mater. Chem. B 2020, 8, 4046–4055. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Huang, C.; Xing, Y.; Fang, W.; Ren, J.; Yu, H.; Wang, G. NIR-Light- and pH-Responsive Graphene Oxide Hybrid Cyclodextrin-Based Supramolecular Hydrogels. Langmuir 2019, 35, 1021–1031. [Google Scholar] [CrossRef]
- Thapa, R.K.; Byeon, J.H.; Choi, H.-G.; Yong, C.S.; Kim, J.O. PEGylated lipid bilayer-wrapped nano-graphene oxides for synergistic co-delivery of doxorubicin and rapamycin to prevent drug resistance in cancers. Nanotechnology 2017, 28, 295101. [Google Scholar] [CrossRef]
- Yang, D.; Feng, L.; Dougherty, C.A.; Luker, K.E.; Chen, D.; Cauble, M.A.; Banaszak Holl, M.M.; Luker, G.D.; Ross, B.D.; Liu, Z.; et al. In vivo targeting of metastatic breast cancer via tumor vasculature-specific nano-graphene oxide. Biomaterials 2016, 104, 361–371. [Google Scholar] [CrossRef]
- Mitra, T.; Manna, P.J.; Raja, S.T.K.; Gnanamani, A.; Kundu, P.P. Curcumin loaded nano graphene oxide reinforced fish scale collagen—A 3D scaffold biomaterial for wound healing applications. RSC Adv. 2015, 5, 98653–98665. [Google Scholar] [CrossRef]
- Charmi, J.; Nosrati, H.; Mostafavi Amjad, J.; Mohammadkhani, R.; Danafar, H. Polyethylene glycol (PEG) decorated graphene oxide nanosheets for controlled release curcumin delivery. Heliyon 2019, 5, e01466. [Google Scholar] [CrossRef]
- Rahman, M.; Singh, J.G.; Afzal, O.; Altamimi, A.S.A.; Alrobaian, M.; Haneef, J.; Barkat, M.A.; Almalki, W.H.; Handa, M.; Shukla, R.; et al. Preparation, Characterization, and Evaluation of Curcumin–Graphene Oxide Complex-Loaded Liposomes against Staphylococcus aureus in Topical Disease. ACS Omega 2022, 7, 43499–43509. [Google Scholar] [CrossRef]
- Hai, L.; He, D.; He, X.; Wang, K.; Yang, X.; Liu, J.; Cheng, H.; Huang, X.; Shangguan, J. Facile fabrication of a resveratrol loaded phospholipid@reduced graphene oxide nanoassembly for targeted and near-infrared laser-triggered chemo/photothermal synergistic therapy of cancer in vivo. J. Mater. Chem. B 2017, 5, 5783–5792. [Google Scholar] [CrossRef]
- Rahmanian, N.; Hamishehkar, H.; Dolatabadi, J.E.N.; Arsalani, N. Nano graphene oxide: A novel carrier for oral delivery of flavonoids. Colloids Surf. B Biointerfaces 2014, 123, 331–338. [Google Scholar] [CrossRef]
- Najafabadi, A.P.; Pourmadadi, M.; Yazdian, F.; Rashedi, H.; Rahdar, A.; Díez-Pascual, A.M. pH-sensitive ameliorated quercetin delivery using graphene oxide nanocarriers coated with potential anticancer gelatin-polyvinylpyrrolidone nanoemulsion with bitter almond oil. J. Drug Deliv. Sci. Technol. 2023, 82, 104339. [Google Scholar] [CrossRef]
- Rana, A.; Matiyani, M.; Tewari, C.; Negi, P.B.; Chandra Arya, M.; Das, V.; Pal, M.; Sahoo, N.G. Functionalized graphene oxide based nanocarrier for enhanced cytotoxicity of Juniperus squamata root essential oil against breast cancer cells. J. Drug Deliv. Sci. Technol. 2022, 72, 103370. [Google Scholar] [CrossRef]
- Figueroa, T.; Aguayo, C.; Fernández, K. Design and Characterization of Chitosan-Graphene Oxide Nanocomposites for the Delivery of Proanthocyanidins. Int. J. Nanomed. 2020, 15, 1229–1238. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Meng, D.; Zheng, X.; Zhang, Y.; Chen, H. Graphene-based materials: A new tool to fight against breast cancer. Int. J. Pharm. 2021, 603, 120644. [Google Scholar] [CrossRef] [PubMed]
- Cui, G.; Wu, J.; Lin, J.; Liu, W.; Chen, P.; Yu, M.; Zhou, D.; Yao, G. Graphene-based nanomaterials for breast cancer treatment: Promising therapeutic strategies. J. Nanobiotechnol. 2021, 19, 211. [Google Scholar] [CrossRef]
- Li, R.; Wang, Y.; Du, J.; Wang, X.; Duan, A.; Gao, R.; Liu, J.; Li, B. Graphene oxide loaded with tumor-targeted peptide and anti-cancer drugs for cancer target therapy. Sci. Rep. 2021, 11, 1725. [Google Scholar] [CrossRef]
- Qin, X.C.; Guo, Z.Y.; Liu, Z.M.; Zhang, W.; Wan, M.M.; Yang, B.W. Folic acid-conjugated graphene oxide for cancer targeted chemo-photothermal therapy. J. Photochem. Photobiol. B Biol. 2013, 120, 156–162. [Google Scholar] [CrossRef]
- Kumari, S.; Nehra, A.; Gupta, K.; Puri, A.; Kumar, V.; Singh, K.P.; Kumar, M.; Sharma, A. Chlorambucil-Loaded Graphene-Oxide-Based Nano-Vesicles for Cancer Therapy. Pharmaceutics 2023, 15, 649. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.-H.; Pi, J.; Jin, H.; Cai, J.-Y. Functional graphene oxide as cancer-targeted drug delivery system to selectively induce oesophageal cancer cell apoptosis. Artif. Cells Nanomed. Biotechnol. 2018, 46, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wei, Y.; Zhai, S.; Chen, Q.; Xing, D. Dihydroartemisinin and transferrin dual-dressed nano-graphene oxide for a pH-triggered chemotherapy. Biomaterials 2015, 62, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Fong, Y.T.; Chen, C.-H.; Chen, J.-P. Intratumoral Delivery of Doxorubicin on Folate-Conjugated Graphene Oxide by In-Situ Forming Thermo-Sensitive Hydrogel for Breast Cancer Therapy. Nanomaterials 2017, 7, 388. [Google Scholar] [CrossRef] [PubMed]
- Hong, H.; Yang, K.; Zhang, Y.; Engle, J.W.; Feng, L.; Yang, Y.; Nayak, T.R.; Goel, S.; Bean, J.; Theuer, C.P.; et al. In Vivo Targeting and Imaging of Tumor Vasculature with Radiolabeled, Antibody-Conjugated Nanographene. ACS Nano 2012, 6, 2361–2370. [Google Scholar] [CrossRef]
- Yan, J.; Zhang, Y.; Zheng, L.; Wu, Y.; Wang, T.; Jiang, T.; Liu, X.; Peng, D.; Liu, Y.; Liu, Z. Let-7i miRNA and platinum loaded nano-graphene oxide platform for detection/reversion of drug resistance and synergetic chemical-photothermal inhibition of cancer cell. Chin. Chem. Lett. 2022, 33, 767–772. [Google Scholar] [CrossRef]
- Al-Ani, L.A.; Kadir, F.A.; Hashim, N.M.; Julkapli, N.M.; Seyfoddin, A.; Lu, J.; AlSaadi, M.A.; Yehye, W.A. The impact of curcumin-graphene based nanoformulation on cellular interaction and redox-activated apoptosis: An in vitro colon cancer study. Heliyon 2020, 6, e05360. [Google Scholar] [CrossRef]
- Yaghoubi, F.; Motlagh, N.S.H.; Naghib, S.M.; Haghiralsadat, F.; Jaliani, H.Z.; Moradi, A. A functionalized graphene oxide with improved cytocompatibility for stimuli-responsive co-delivery of curcumin and doxorubicin in cancer treatment. Sci. Rep. 2022, 12, 1959. [Google Scholar] [CrossRef]
- Yaghoubi, F.; Hosseini Motlagh, N.S.; Moradi, A.; Haghiralsadat, F. Carboxylated Graphene Oxide as a Nanocarrier for Drug Delivery of Quercetin as an Effective Anticancer Agent. Iran. Biomed. J. 2022, 26, 324–329. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, H.; Karki, N.; Pal, M.; Basak, S.; Verma, R.K.; Bal, R.; Kandpal, N.D.; Bisht, G.; Sahoo, N.G. Functionalized graphene oxide as a nanocarrier for dual drug delivery applications: The synergistic effect of quercetin and gefitinib against ovarian cancer cells. Colloids Surf. B Biointerfaces 2019, 178, 452–459. [Google Scholar] [CrossRef] [PubMed]
- Buskaran, K.; Hussein, M.Z.; Moklas, M.A.M.; Masarudin, M.J.; Fakurazi, S. Graphene Oxide Loaded with Protocatechuic Acid and Chlorogenic Acid Dual Drug Nanodelivery System for Human Hepatocellular Carcinoma Therapeutic Application. Int. J. Mol. Sci. 2021, 22, 5786. [Google Scholar] [CrossRef] [PubMed]
- Sontakke, A.D.; Fopase, R.; Pandey, L.M.; Purkait, M.K. Development of graphene oxide nanoscrolls imparted nano-delivery system for the sustained release of gallic acid. Appl. Nanosci. 2022, 12, 2733–2751. [Google Scholar] [CrossRef]
- Bhatt, S.; Punetha, V.D.; Pathak, R.; Punetha, M. Graphene in nanomedicine: A review on nano-bio factors and antibacterial activity. Colloids Surf. B Biointerfaces 2023, 226, 113323. [Google Scholar] [CrossRef] [PubMed]
- Ng, I.M.J.; Shamsi, S. Graphene Oxide (GO): A Promising Nanomaterial against Infectious Diseases Caused by Multidrug-Resistant Bacteria. Int. J. Mol. Sci. 2022, 23, 9096. [Google Scholar] [CrossRef] [PubMed]
- Yousefi, M.; Dadashpour, M.; Hejazi, M.; Hasanzadeh, M.; Behnam, B.; de la Guardia, M.; Shadjou, N.; Mokhtarzadeh, A. Anti-bacterial activity of graphene oxide as a new weapon nanomaterial to combat multidrug-resistance bacteria. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 74, 568–581. [Google Scholar] [CrossRef]
- Wu, X.; Tan, S.; Xing, Y.; Pu, Q.; Wu, M.; Zhao, J.X. Graphene oxide as an efficient antimicrobial nanomaterial for eradicating multi-drug resistant bacteria in vitro and in vivo. Colloids Surf. B Biointerfaces 2017, 157, 1–9. [Google Scholar] [CrossRef]
- Bugli, F.; Cacaci, M.; Palmieri, V.; Di Santo, R.; Torelli, R.; Ciasca, G.; Di Vito, M.; Vitali, A.; Conti, C.; Sanguinetti, M.; et al. Curcumin-loaded graphene oxide flakes as an effective antibacterial system against methicillin-resistant Staphylococcus aureus. Interface Focus 2018, 8, 20170059. [Google Scholar] [CrossRef] [PubMed]
- Robb, S.M.; Li, M.; Dahl, K.N.; Islam, M.F. Antibacterial Mechanisms of Nano-Graphene Oxide By Measuring Membrane Interactions. Electrochem. Soc. Meet. Abstr. 2016, MA2016-01, 616. [Google Scholar] [CrossRef]
- Pulingam, T.; Thong, K.L.; Ali, M.E.; Appaturi, J.N.; Dinshaw, I.J.; Ong, Z.Y.; Leo, B.F. Graphene oxide exhibits differential mechanistic action towards Gram-positive and Gram-negative bacteria. Colloids Surf. B Biointerfaces 2019, 181, 6–15. [Google Scholar] [CrossRef]
- Tu, Y.; Lv, M.; Xiu, P.; Huynh, T.; Zhang, M.; Castelli, M.; Liu, Z.; Huang, Q.; Fan, C.; Fang, H.; et al. Destructive extraction of phospholipids from Escherichia coli membranes by graphene nanosheets. Nat. Nanotechnol. 2013, 8, 594–601. [Google Scholar] [CrossRef]
- Mao, J.; Guo, R.; Yan, L.-T. Simulation and analysis of cellular internalization pathways and membrane perturbation for graphene nanosheets. Biomaterials 2014, 35, 6069–6077. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wang, X.; Han, H. A new function of graphene oxide emerges: Inactivating phytopathogenic bacterium Xanthomonas oryzae pv. Oryzae. J. Nanopart. Res. 2013, 15, 1658. [Google Scholar] [CrossRef]
- Bolotin, K.I.; Sikes, K.J.; Jiang, Z.; Klima, M.; Fudenberg, G.; Hone, J.; Kim, P.; Stormer, H.L. Ultrahigh electron mobility in suspended graphene. Solid State Commun. 2008, 146, 351–355. [Google Scholar] [CrossRef]
- Zhang, Y.; Fu, H.; Liu, D.-E.; An, J.; Gao, H. Construction of biocompatible bovine serum albumin nanoparticles composed of nano graphene oxide and AIEgen for dual-mode phototherapy bacteriostatic and bacterial tracking. J. Nanobiotechnol. 2019, 17, 104. [Google Scholar] [CrossRef]
- Pourhajibagher, M.; Etemad-Moghadam, S.; Alaeddini, M.; Miri Mousavi, R.s.; Bahador, A. DNA-aptamer-nanographene oxide as a targeted bio-theragnostic system in antimicrobial photodynamic therapy against Porphyromonas gingivalis. Sci. Rep. 2022, 12, 12161. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yu, X.; Li, Q.; Zhu, J.; Ding, J.; Jiang, T. Vancomycin modified graphene oxide as carriers to load phthalocyanine for synergistic phototherapy of vancomycin-resistant bacteria. J. Nanopart.Res. 2022, 24, 234. [Google Scholar] [CrossRef]
- Jihad, M.A.; Noori, F.T.M.; Jabir, M.S.; Albukhaty, S.; AlMalki, F.A.; Alyamani, A.A. Polyethylene Glycol Functionalized Graphene Oxide Nanoparticles Loaded with Nigella sativa Extract: A Smart Antibacterial Therapeutic Drug Delivery System. Molecules 2021, 26, 3067. [Google Scholar] [CrossRef]
- Yan, H.; Li, P.; Jiang, X.; Wang, X.; Hu, Y.; Zhang, Y.; Su, R.; Su, W. Preparation of graphene oxide/polydopamine-curcumin composite nanomaterials and its antibacterial effect against Staphylococcus aureus induced by white light. Biomater. Adv. 2022, 139, 213040. [Google Scholar] [CrossRef] [PubMed]
- Cacaci, M.; Squitieri, D.; Palmieri, V.; Torelli, R.; Perini, G.; Campolo, M.; Di Vito, M.; Papi, M.; Posteraro, B.; Sanguinetti, M.; et al. Curcumin-Functionalized Graphene Oxide Strongly Prevents Candida parapsilosis Adhesion and Biofilm Formation. Pharmaceuticals 2023, 16, 275. [Google Scholar] [CrossRef]
- Oves, M.; Ansari, M.O.; Ansari, M.S.; Memić, A. Graphene@Curcumin-Copper Paintable Coatings for the Prevention of Nosocomial Microbial Infection. Molecules 2023, 28, 2814. [Google Scholar] [CrossRef]
- Shamsi, S.; Abdul Ghafor, A.A.H.; Norjoshukrudin, N.H.; Ng, I.M.J.; Abdullah, S.N.S.; Sarchio, S.N.E.; Md Yasin, F.; Abd Gani, S.; Mohd Desa, M.N. Stability, Toxicity, and Antibacterial Potential of Gallic Acid-Loaded Graphene Oxide (GAGO) Against Methicillin-Resistant Staphylococcus aureus (MRSA) Strains. Int. J. Nanomed. 2022, 17, 5781–5807. [Google Scholar] [CrossRef] [PubMed]
- Arfat, Y.A.; Ahmed, J.; Ejaz, M.; Mullah, M. Polylactide/graphene oxide nanosheets/clove essential oil composite films for potential food packaging applications. Int. J. Biol. Macromol. 2018, 107, 194–203. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Song, R.; Zhang, X.; Zhang, D. Enhanced antimicrobial activity and pH-responsive sustained release of chitosan/poly (vinyl alcohol)/graphene oxide nanofibrous membrane loading with allicin. Int. J. Biol. Macromol. 2020, 161, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Han, X.-Y.; Meng, T.; Ye, J.-X.; Yin, H.-B.; Song, D.-W. Enhanced Antibacterial and Osteogenic Properties of Graphene Oxide Loaded with Berberine on Biomedical Titanium. J. Biomed. Nanotechnol. 2022, 18, 849–859. [Google Scholar] [CrossRef] [PubMed]
- Ning, Y.; Wang, X.; Chen, P.; Liu, S.; Hu, J.; Xiao, R.; Li, L.; Lu, F. Targeted inhibition of methicillin-resistant Staphylococcus aureus biofilm formation by a graphene oxide-loaded aptamer/berberine bifunctional complex. Drug Deliv. 2022, 29, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.S.; Park, R.; Jo, H.J.; Shin, Y.C.; Kim, C.S.; Hyon, S.H.; Hong, S.W.; Oh, J.; Han, D.W. Spontaneous Osteogenic Differentiation of Human Mesenchymal Stem Cells by Tuna-Bone-Derived Hydroxyapatite Composites with Green Tea Polyphenol-Reduced Graphene Oxide. Cells 2023, 12, 1448. [Google Scholar] [CrossRef]
- Shen, H.; Lin, H.; Sun, A.X.; Song, S.; Wang, B.; Yang, Y.; Dai, J.; Tuan, R.S. Acceleration of chondrogenic differentiation of human mesenchymal stem cells by sustained growth factor release in 3D graphene oxide incorporated hydrogels. Acta Biomater. 2020, 105, 44–55. [Google Scholar] [CrossRef]
- Zhong, C.; Feng, J.; Lin, X.; Bao, Q. Continuous release of bone morphogenetic protein-2 through nano-graphene oxide-based delivery influences the activation of the NF-κB signal transduction pathway. Int. J. Nanomed. 2017, 12, 1215–1226. [Google Scholar] [CrossRef]
- La, W.-G.; Jin, M.; Park, S.; Yoon, H.-H.; Jeong, G.-J.; Bhang, S.H.; Park, H.; Char, K.; Kim, B.-S. Delivery of bone morphogenetic protein-2 and substance P using graphene oxide for bone regeneration. Int. J. Nanomed. 2014, 9, 107–116. [Google Scholar] [CrossRef]
- Faraji, S.; Nowroozi, N.; Nouralishahi, A.; Shabani Shayeh, J. Electrospun poly-caprolactone/graphene oxide/quercetin nanofibrous scaffold for wound dressing: Evaluation of biological and structural properties. Life Sci. 2020, 257, 118062. [Google Scholar] [CrossRef] [PubMed]
- Guo, B.; Feng, X.; Wang, Y.; Wang, X.; He, Y. Biomimetic and immunomodulatory baicalin-loaded graphene oxide-demineralized bone matrix scaffold for in vivo bone regeneration. J. Mater. Chem. B 2021, 9, 9720–9733. [Google Scholar] [CrossRef] [PubMed]
- Kashte, S.; Sharma, R.K.; Kadam, S. Layer-by-layer decorated herbal cell compatible scaffolds for bone tissue engineering: A synergistic effect of graphene oxide and Cissus quadrangularis. J. Bioact. Compat. Polym. 2020, 35, 57–73. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).