Aramid Fibers Modulated Polyethylene Separator as Efficient Polysulfide Barrier for High-Performance Lithium-Sulfur Batteries
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
2.2. Preparation of AF-PE Separators
2.3. Characterization
2.4. Electrochemical Measurements
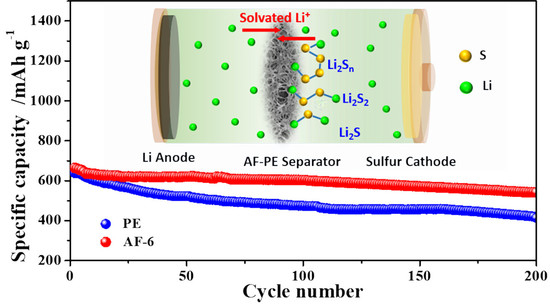
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Balach, J.; Linnemann, J.; Jaumann, T.; Giebeler, L. Metal-based nanostructured materials for advanced lithium-sulfur batteries. J. Mater. Chem. A 2018, 6, 23127–23168. [Google Scholar] [CrossRef] [Green Version]
- Ponraj, R.; Kannan, A.G.; Ahn, J.H.; Lee, J.H.; Kang, J.; Han, B.; Kim, D.W. Effective trapping of lithium polysulfides using a functionalized carbon nanotube-coated separator for lithium-sulfur cells with enhanced cycling stability. ACS Appl. Mater. Inter. 2017, 9, 38445–38454. [Google Scholar] [CrossRef] [PubMed]
- Kong, L.; Li, B.-Q.; Peng, H.-J.; Zhang, R.; Xie, J.; Huang, J.-Q.; Zhang, Q. Porphyrin-derived graphene-based nanosheets enabling strong polysulfide chemisorption and rapid kinetics in lithium-sulfur batteries. Adv. Energy Mater. 2018, 8, 1800849. [Google Scholar] [CrossRef]
- Huang, S.; Lim, Y.V.; Zhang, X.; Wang, Y.; Zheng, Y.; Kong, D.; Ding, M.; Yang, S.A.; Yang, H.Y. Regulating the polysulfide redox conversion by iron phosphide nanocrystals for high-rate and ultrastable lithium-sulfur battery. Nano Energy 2018, 51, 340–348. [Google Scholar] [CrossRef]
- He, Y.; Chang, Z.; Wu, S.; Qiao, Y.; Bai, S.; Jiang, K.; He, P.; Zhou, H. Simultaneously inhibiting lithium dendrites growth and polysulfides shuttle by a flexible MOF-based membrane in Li-S batteries. Adv. Energy Mater. 2018, 8, 1802130. [Google Scholar] [CrossRef]
- Chung, S.-H.; Manthiram, A. Rational design of statically and dynamically stable lithium-sulfur batteries with high sulfur loading and low electrolyte/sulfur ratio. Adv. Mater. 2018, 30, 1705951. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Pan, H.; Zhong, H.; Xiao, Z.; Li, X.; Wang, R. Porous organic polymers for polysulfide trapping in lithium-sulfur batteries. Adv. Funct. Mater. 2018, 28, 1707597. [Google Scholar] [CrossRef]
- Ji, Y.; Liu, X.; Xiu, Y.; Indris, S.; Njel, C.; Maibach, J.; Ehrenherg, H.; Fichtner, M.; Zhao-Karger, Z. Magnesium-sulfur batteries: Polyoxometalate modified separator for performance enhancement of magnesium-sulfur batteries. Adv. Funct. Mater. 2021, 31, 2100868. [Google Scholar] [CrossRef]
- Zhou, W.; Zhao, D.; Wu, Q.; Fan, B.; Dan, J.; Han, A.; Ma, L.; Zhang, X.; Li, L. Amorphous CoP nanoparticle composites with nitrogen-doped hollow carbon nanospheres for synergetic anchoring and catalytic conversion of polysulfides in Li-S batteries. J. Colloid Interf. Sci. 2021, 603, 1–10. [Google Scholar] [CrossRef]
- Yu, H.; Zeng, P.; Liu, H.; Zhou, X.; Guo, C.; Li, Y.; Liu, S.; Chen, M.; Guo, X.; Chang, B.; et al. Li2S in situ grown on three-dimensional porous carbon architecture with electron/ion channels and dual active sites as cathodes of Li-S batteries. ACS Appl. Mater. Inter. 2021, 13, 32968–32977. [Google Scholar] [CrossRef]
- Wu, Z.; Wang, W.; Wang, Y.; Chen, C.; Li, K.; Zhao, G.; Sun, C.; Chen, W.; Ni, L.; Diao, G. Three-dimensional graphene hollow spheres with high sulfur loading for high-performance lithium-sulfur batteries. Electrochim. Acta 2017, 224, 527–533. [Google Scholar] [CrossRef]
- Deng, C.; Wang, Z.; Wang, S.; Yu, J. Inhibition of polysulfide diffusion in lithium–sulfur batteries: Mechanism and improvement strategies. J. Mater. Chem. A 2019, 7, 12381–12413. [Google Scholar] [CrossRef]
- Ogoke, O.; Wu, G.; Wang, X.; Casimir, A.; Ma, L.; Wu, T.; Lu, J. Effective strategies for stabilizing sulfur for advanced lithium–sulfur batteries. J. Mater. Chem. A 2017, 5, 448–469. [Google Scholar] [CrossRef]
- Fu, A.; Wang, C.; Pei, F.; Cui, J.; Fang, X.; Zheng, N. Recent advances in hollow porous carbon materials for lithium-sulfur batteries. Small 2019, 15, 1804786. [Google Scholar] [CrossRef]
- Lee, C.; Kim, I. A hierarchical carbon nanotube-loaded glass-filter composite paper interlayer with outstanding electrolyte uptake properties for high-performance lithium-sulphur batteries. Nanoscale 2015, 7, 10362–10367. [Google Scholar] [CrossRef] [Green Version]
- Eroglu, O.; Kiai, M.S.; Kizil, H. Glass fiber separator coated by boron doped anatase TiO2 for high-rate Li–S battery. Mater. Res. Bull. 2020, 129, 110917. [Google Scholar] [CrossRef]
- Wu, J.; Zeng, H.; Li, X.; Xiang, X.; Liao, Y.; Xue, Z.; Ye, Y.; Xie, X. Ultralight layer-by-layer self-assembled MoS2-polymer modified separator for simultaneously trapping polysulfides and suppressing lithium dendrites. Adv. Energy Mater. 2018, 8, 1802430. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, J.; Lou, X.W. Hollow carbon nanofibers filled with MnO2 nanosheets as efficient sulfur hosts for lithium-sulfur batteries. Angew. Chem. Int. Ed. Engl. 2015, 54, 12886–12890. [Google Scholar] [CrossRef]
- Chen, X.; Huang, Y.; Li, J.; Wang, X.; Zhang, Y.; Guo, Y.; Ding, J.; Wang, L. Bifunctional separator with sandwich structure for high-performance lithium-sulfur batteries. J. Colloid Interf. Sci. 2020, 559, 13–20. [Google Scholar] [CrossRef]
- Ni, L.; Zhao, G.; Wang, Y.; Wu, Z.; Wang, W.; Liao, Y.; Yang, G.; Diao, G.A.-O. Coaxial Carbon/MnO2 Hollow Nanofibers as Sulfur Hosts for High-Performance Lithium-Sulfur Batteries. Chem.-Asian. J. 2017, 12, 3128–3134. [Google Scholar] [CrossRef]
- Thompson, L.T.; Li, J.; Wang, Q.; Wang, Z.; Cao, Y.; Zhu, J.; Lou, Y.; Zhao, Y.; Shi, L.; Yuan, S. Evaporation and in-situ gelation induced porous hybrid film without template enhancing the performance of lithium ion battery separator. Nat. Commun. 2021, 595, 142–150. [Google Scholar]
- Yang, Y.; Huang, C.; Gao, G.; Hu, C.; Luo, L.; Xu, J. Aramid nanofiber/bacterial cellulose composite separators for lithium-ion batteries. Carbohyd. Polym. 2020, 247, 116702. [Google Scholar] [CrossRef]
- Tung, S.O.; Fisher, S.L.; Kotov, N.A. Nanoporous aramid nanofibre separators for nonaqueous redox flow batteries. Nat. Commun. 2018, 9, 4193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoon, E.; Park, J.W.; Kang, J.K.; Kim, S.; Jung, Y. High-energy-density Li-S batteries with additional elemental sulfur coated on a thin-film separator. J. Nanosci. Nanotechnol. 2019, 19, 4715–4718. [Google Scholar] [CrossRef] [PubMed]
- Babu, D.B.; Giribabu, K. Permselective SPEEK/Nafion Composite-Coated Separator as a Potential Polysulfide Crossover Barrier Layer for Li-S Batteries. ACS Appl. Mater. Inter. 2018, 10, 19721–19729. [Google Scholar] [CrossRef]
- Zhang, Z.; Lai, Y.; Zhang, Z.; Zhang, K.; Li, J. Al2O3-coated porous separator for enhanced electrochemical performance of lithium sulfur batteries. Electrochim. Acta 2014, 129, 55–61. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, J.; Zhang, J.; Su, Y.; Yu, X. Aramid Fibers Modulated Polyethylene Separator as Efficient Polysulfide Barrier for High-Performance Lithium-Sulfur Batteries. Nanomaterials 2022, 12, 2513. https://doi.org/10.3390/nano12152513
Gu J, Zhang J, Su Y, Yu X. Aramid Fibers Modulated Polyethylene Separator as Efficient Polysulfide Barrier for High-Performance Lithium-Sulfur Batteries. Nanomaterials. 2022; 12(15):2513. https://doi.org/10.3390/nano12152513
Chicago/Turabian StyleGu, Jifeng, Jiaping Zhang, Yun Su, and Xu Yu. 2022. "Aramid Fibers Modulated Polyethylene Separator as Efficient Polysulfide Barrier for High-Performance Lithium-Sulfur Batteries" Nanomaterials 12, no. 15: 2513. https://doi.org/10.3390/nano12152513
APA StyleGu, J., Zhang, J., Su, Y., & Yu, X. (2022). Aramid Fibers Modulated Polyethylene Separator as Efficient Polysulfide Barrier for High-Performance Lithium-Sulfur Batteries. Nanomaterials, 12(15), 2513. https://doi.org/10.3390/nano12152513