The Influence of Anti-C3aR and Anti-C5aR Antibody Levels on the Course of Specific Glomerulonephritis Types
Abstract
1. Introduction
- -
- Podocyte injury and glomerular membrane leakage in membranous nephropathy [11];
- -
- Decay-accelerating factor expression diminishment in podocytes enhances C3 convertase, which activates C3aR [12]. C3a/C3aR ligation on podocytes [12] activates the interleukin-1beta/interleukin 1 receptor signaling loop, which results in a reduction in nephrin expression in focal and segmental glomerulosclerosis;
- -
- Versican 1 expression activated by C3aR through the AKT/beta cathenin pathway in tubular cells induces fibroblast activation and, as a result, interstitial fibrosis [18] in focal and segmental glomerulosclerosis;
- -
- -
- C3aR caused cell proliferation and inflammation propagation through IL-6 (interleukin-6) and MCP-1 (monocyte chemoattractant protein 1) production enhancement in the course of IgA nephropathy [14];
- -
- C3aR promotes macrophage infiltration in p-ANCA vasculitis but does not promote lymphocyte T activation [15];
- -
- Widespread C3 glomerular deposition activates the complement system, including C3aR in C3 glomerulopathy [16];
- -
- C3 convertase dysregulation leads to inflammation development in membranoproliferative glomerulonephritis [17].
- -
- -
- Increasing Il-1 beta and MIP-2 mRNA (mitochondrial ribonucleic acid) expression encourages the development of inflammation and apoptosis enhancement [25], promoting mitochondrial fission and dysfunction in podocytes, which results in podocytes injury [30] and the modulation of lymphocytes’ Th1 responses in lupus nephritis [39];
- -
- Mesangial cells’ proliferation is promoted and inflammation is activated through Il-6 and MCP-1 [31] in IgA nephropathy;
- -
- C5aR plays a role in amplification loop and neutrophil priming [35] in the development of ANCA vasculitis;
- -
- A deficiency of C5aR ameliorates pANCA vasculitis [34];
- -
- A C5aR deficiency reduces C3 glomerulopathy severity [36];
- -
- C5a convertase regulation disorders enhance inflammation development in membranoproliferative glomerulonephritis [17];
- -
- C5aR is connected with vascular thrombosis in hemolytic uremic syndrome [37];
- -
- C5aR plays a role in lipopolysaccharide-induced kidney injury through N-acetyl-β-D-glucosaminidase activation, ferroptosis, and mitochondrial damage [38].
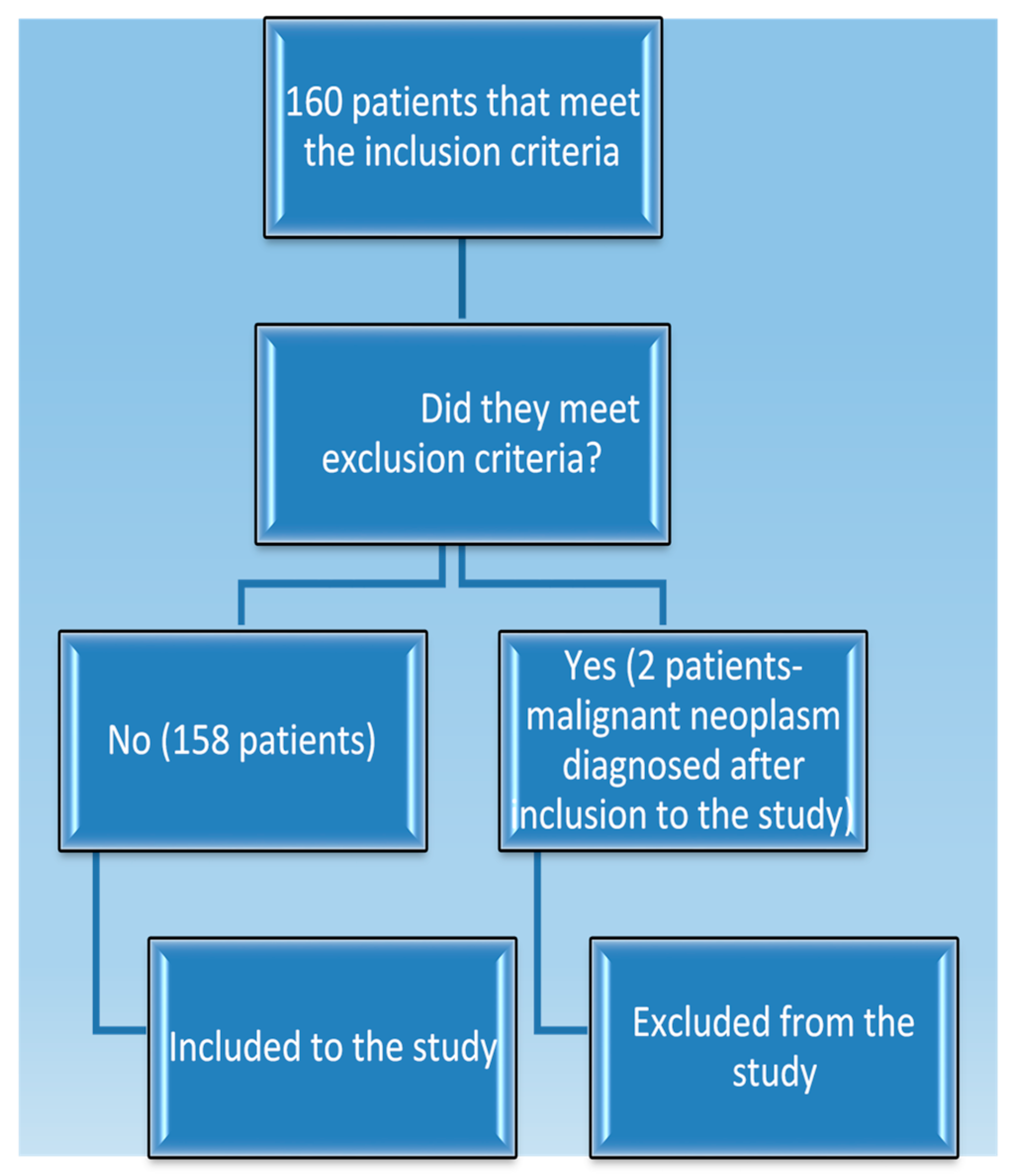
2. Materials and Methods
3. Results
3.1. Patient Clinical Data
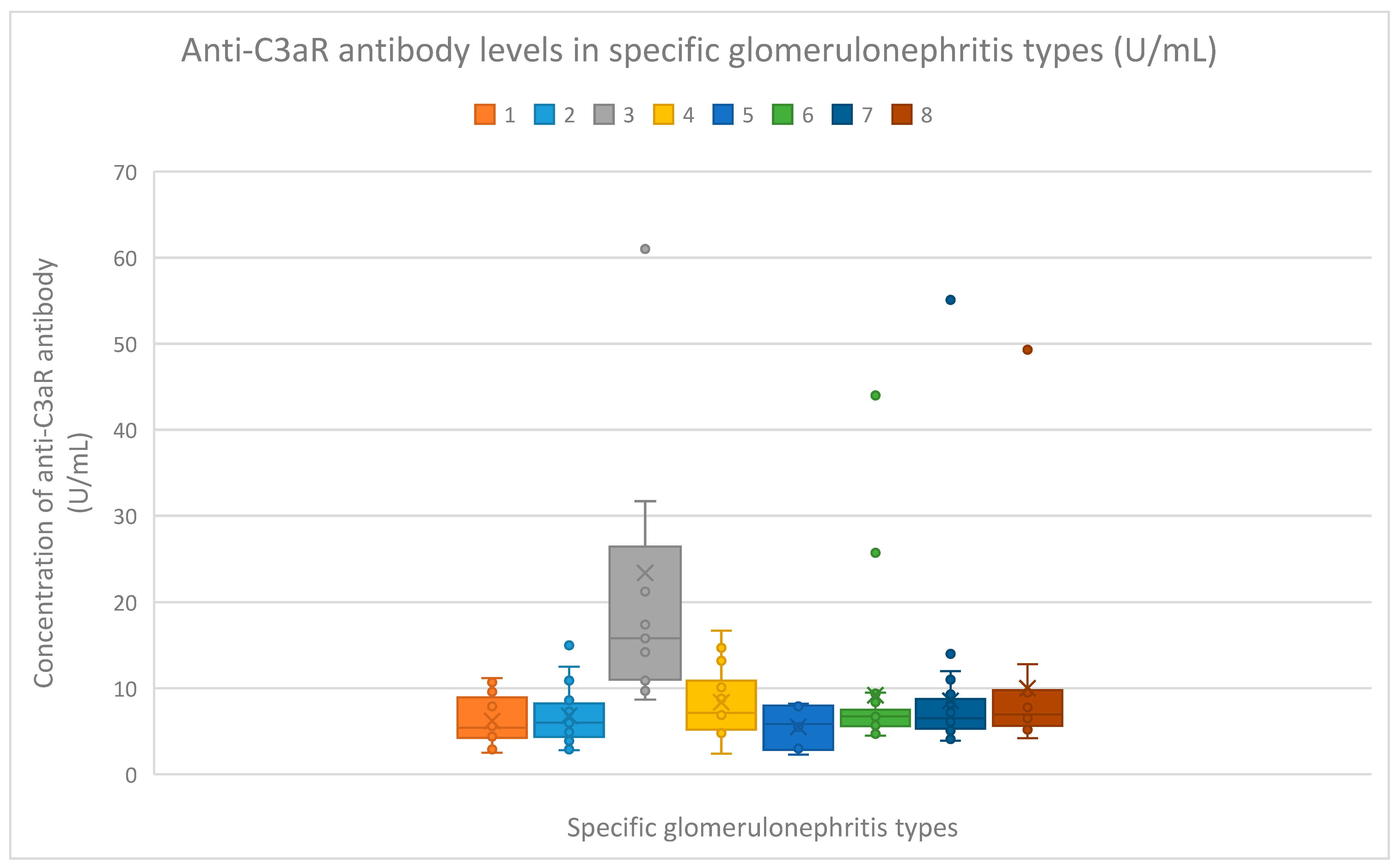
3.2. Anti-C3aR Antibody Evaluation Results
- 5.4 (range: 2.5–11.2) U/mL in the membranous nephropathy group;
- 6 (range: 2.8–15.3) U/mL in the FSGS group;
- 15.8 (range: 8.7–61) U/mL in the lupus nephritis group;
- 7.15 (range: 2.4–16.7) U/mL in the IgA nephropathy group;
- 5.85 (range: 2.3–8.2) U/mL in the mesangial proliferative (non-IgA) glomerulonephritis group;
- 6.75 (range:4.5–44) U/mL in the control group;
- 6.5 (range: 3.9–55.1) U/mL in the c-ANCA vasculitis group;
- 6.95 (range: 4.2–49.3) U/mL in the p-ANCA vasculitis group.
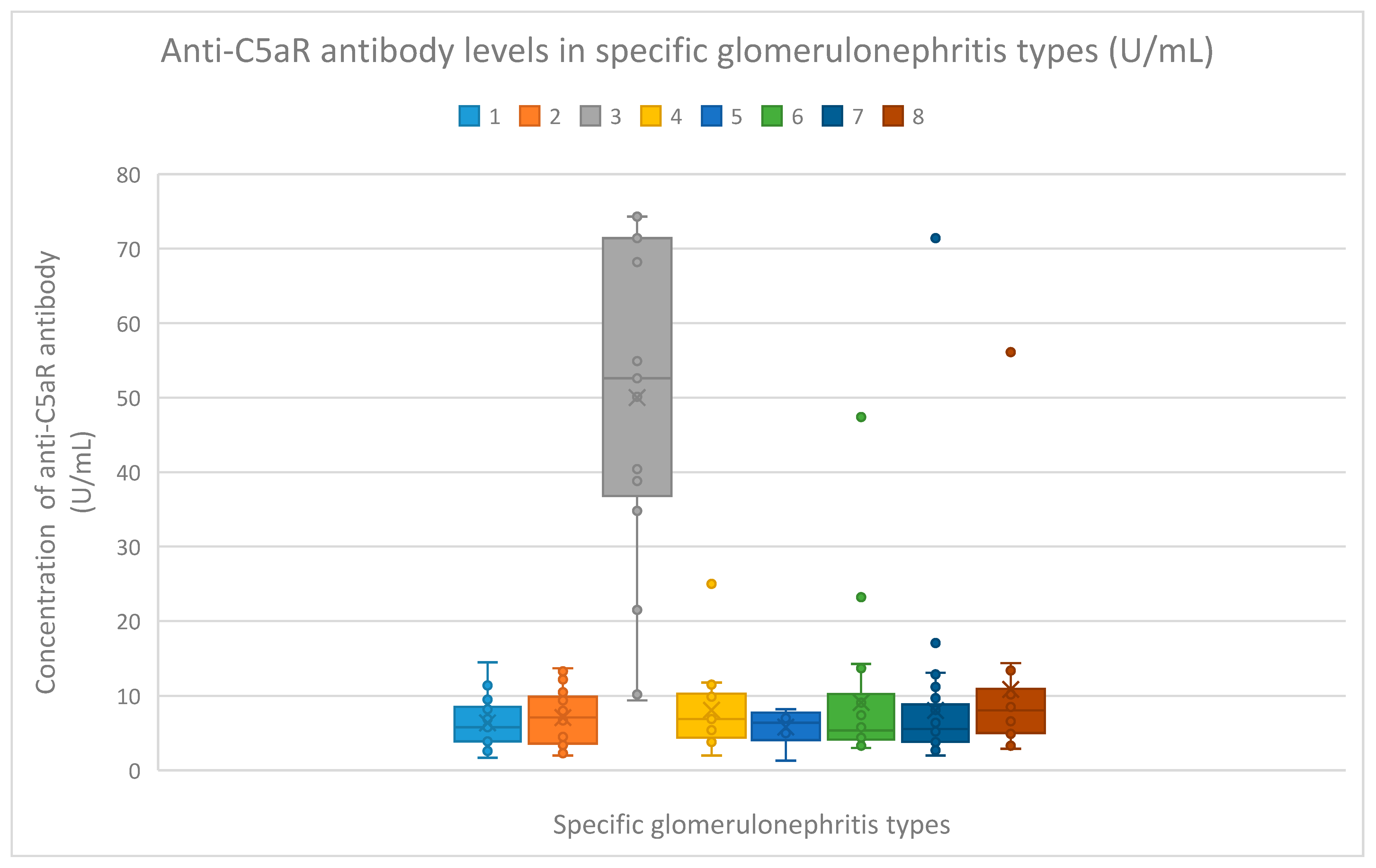
3.3. Anti-C5aR Antibody Evaluation Results
- 5.8 (range: 1.7–14.5) U/mL in the membranous nephropathy group;
- 7.1 (range: 2–13.7) U/mL in the FSGS group;
- 52.6 (range: 9.4–74.3) U/mL in the lupus nephritis group;
- 6.9 (range: 2–25) U/mL in the IgA nephropathy group;
- 6.4 (range: 1.3–8.2) U/mL in the mesangial proliferative (non-IgA) glomerulonephritis group;
- 5.35 (range: 3–47.4) U/mL in the control group;
- 5.55 (range: 2.7–17.1) U/mL in the c-ANCA vasculitis group;
- 8.05 (range: 2.9–56.1) U/mL in the p-ANCA vasculitis group.
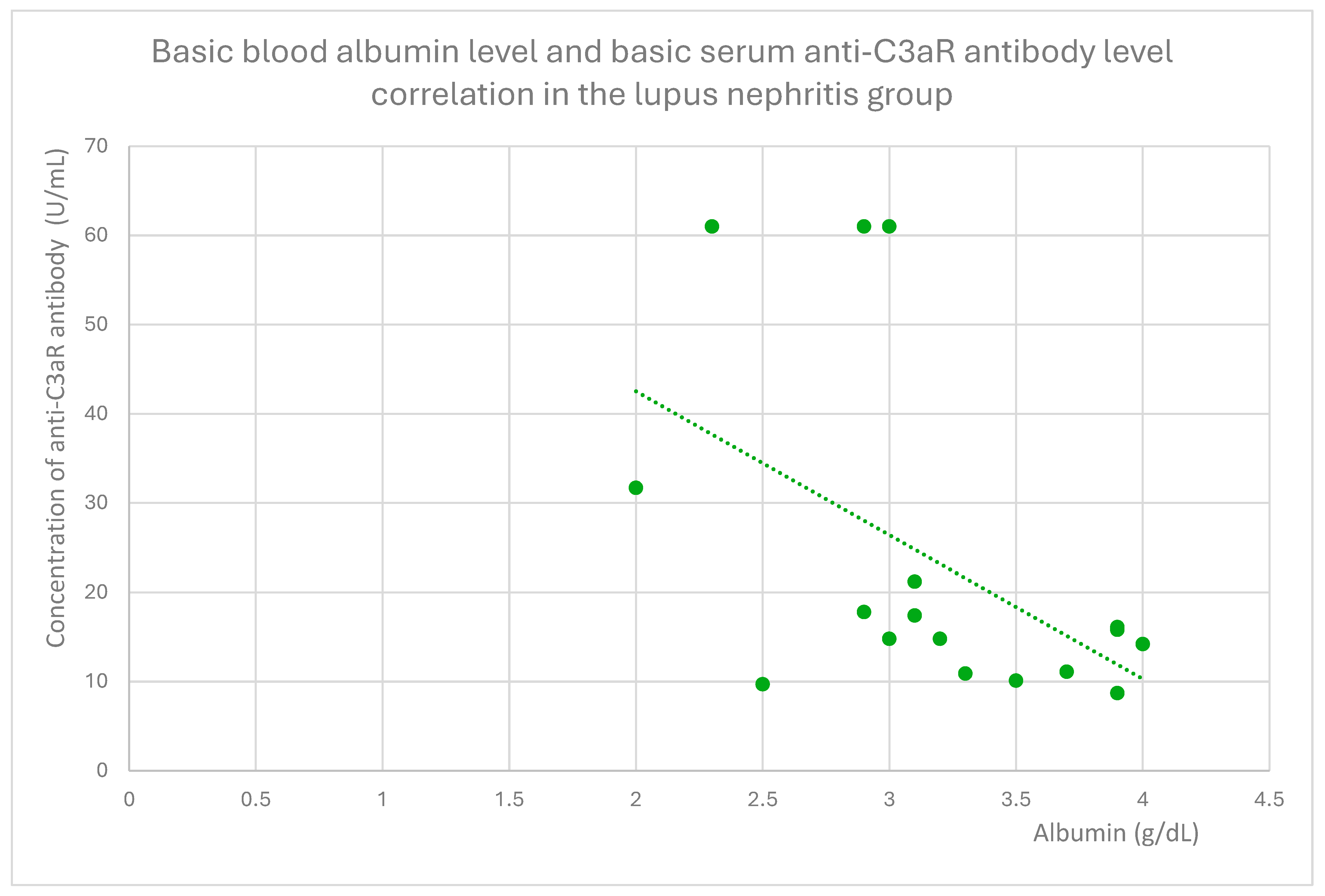
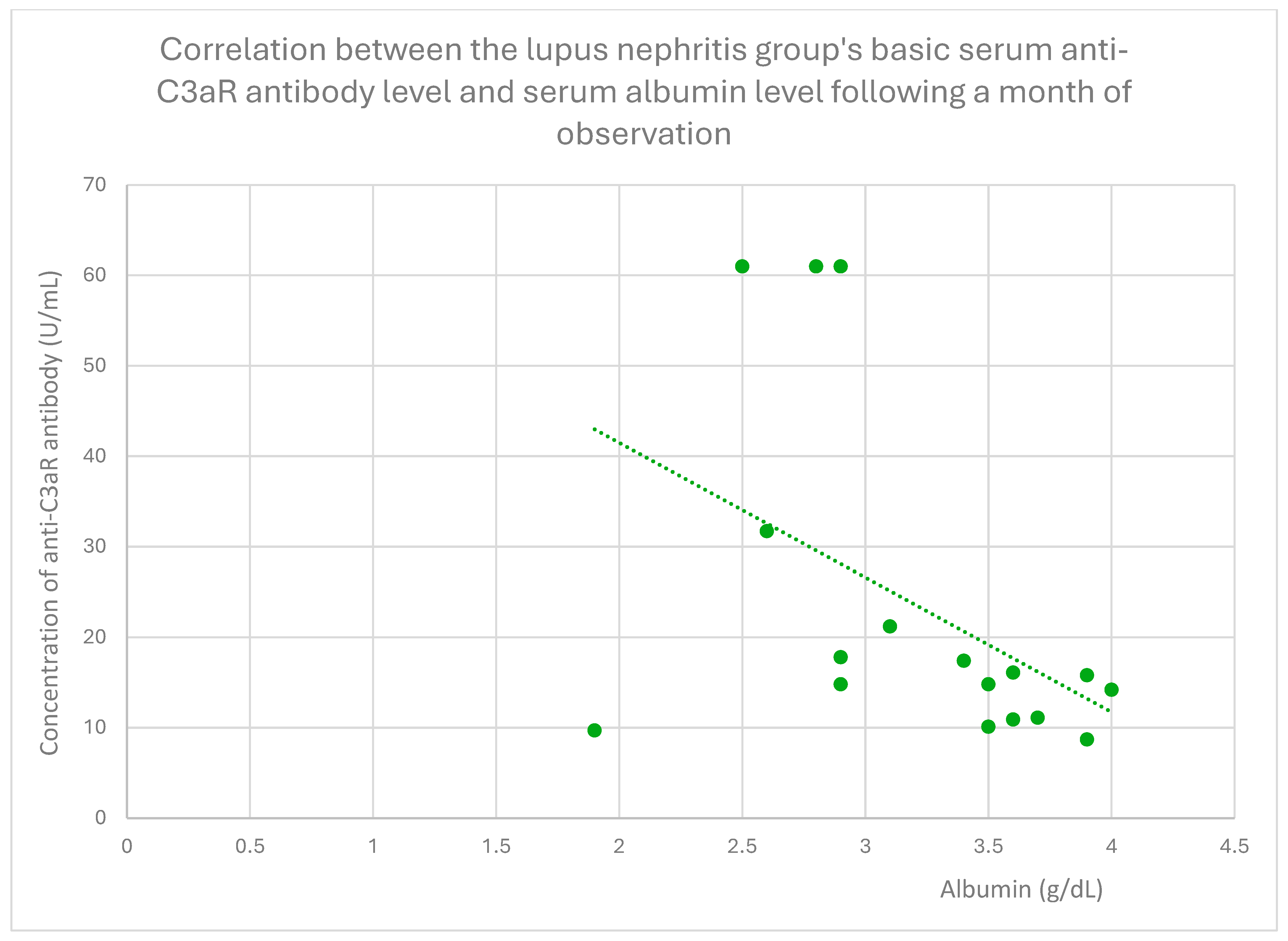
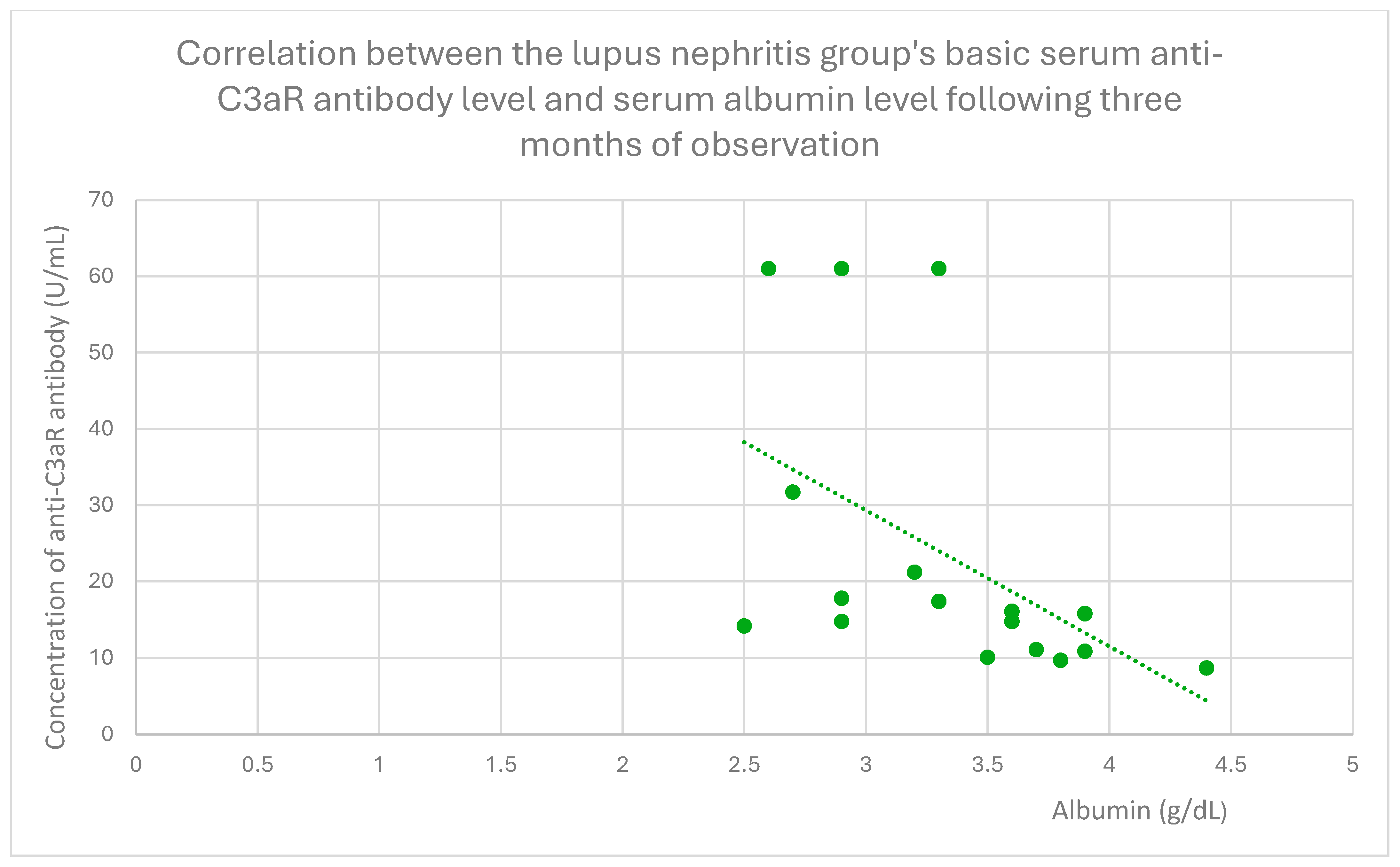
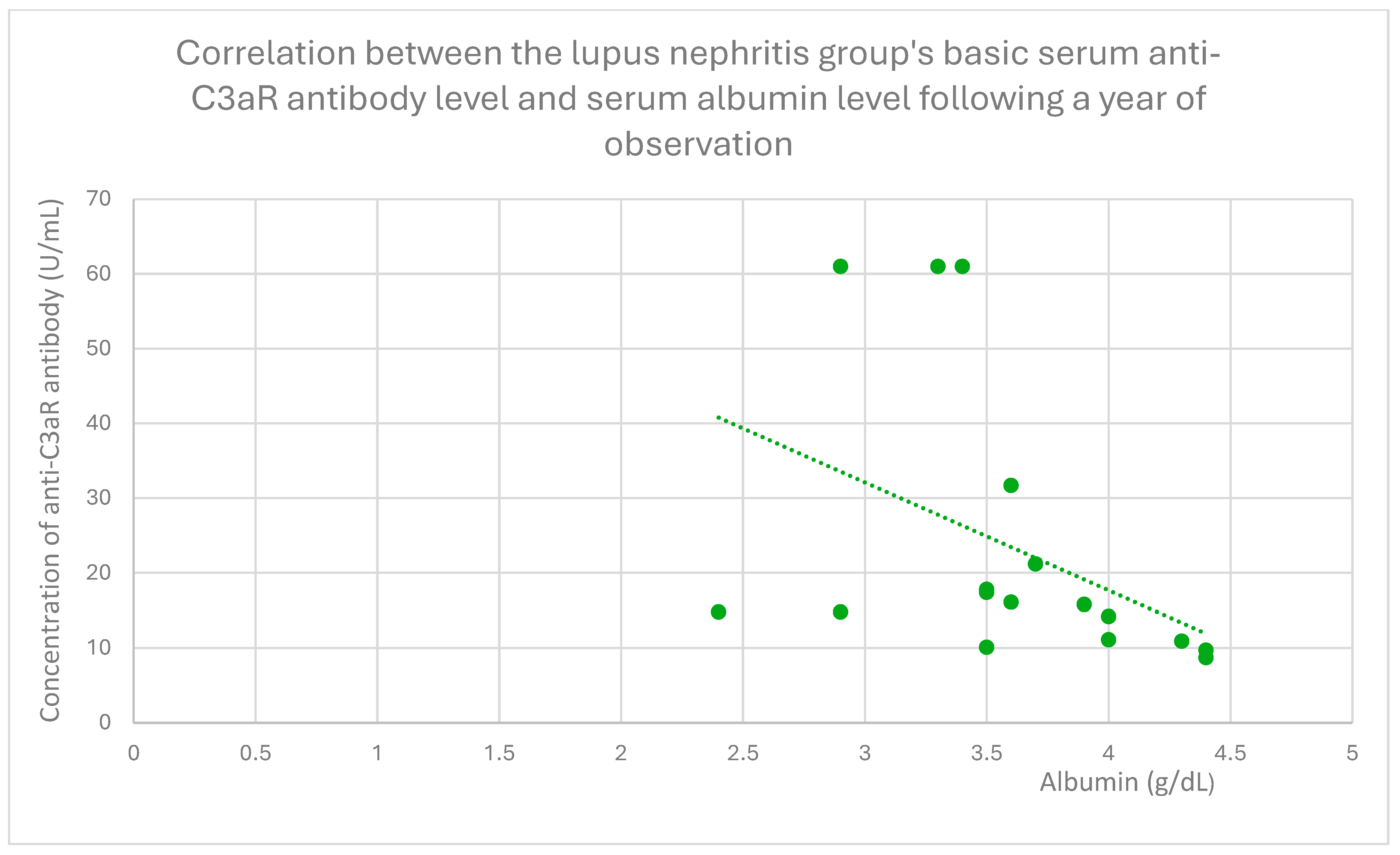
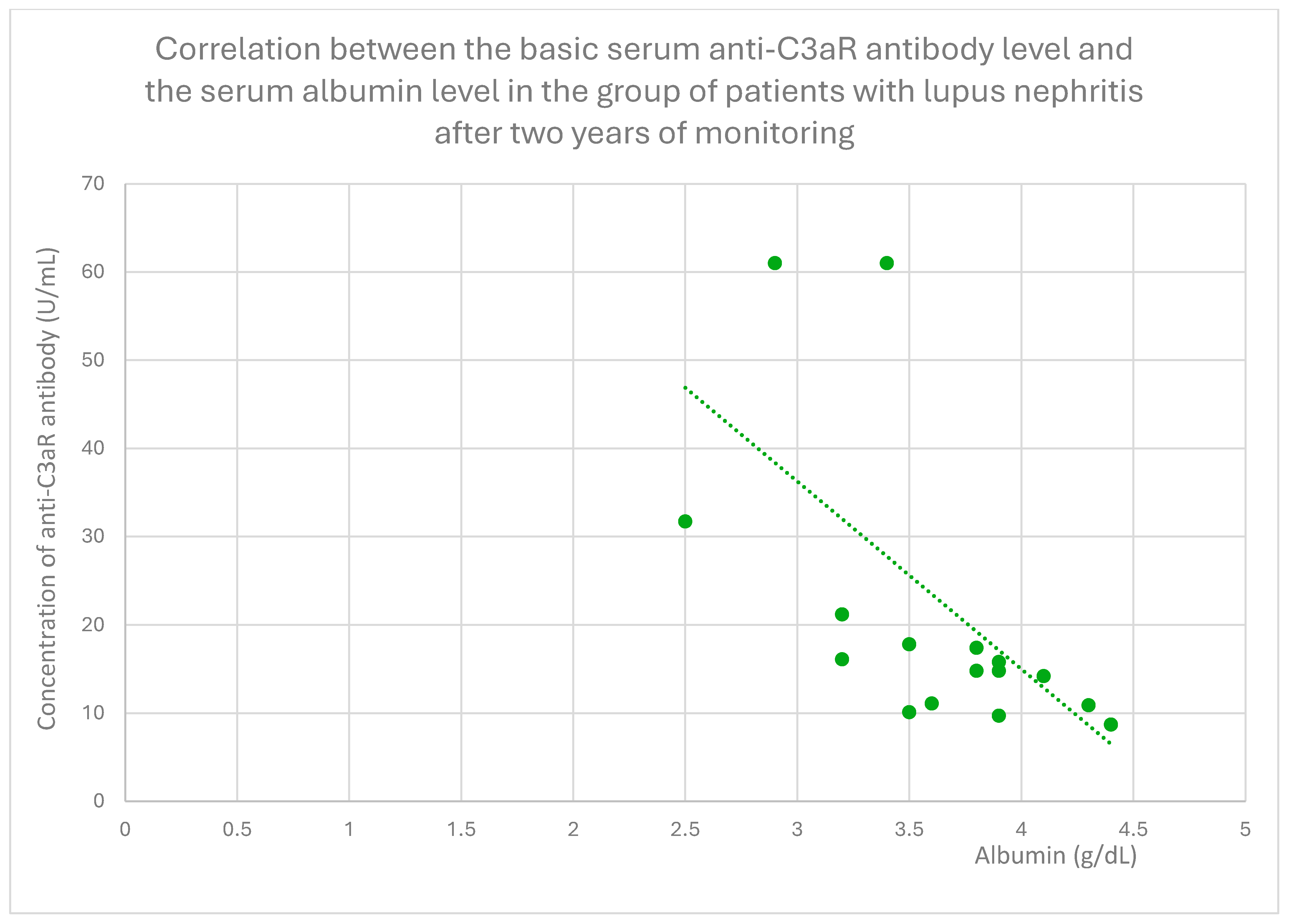
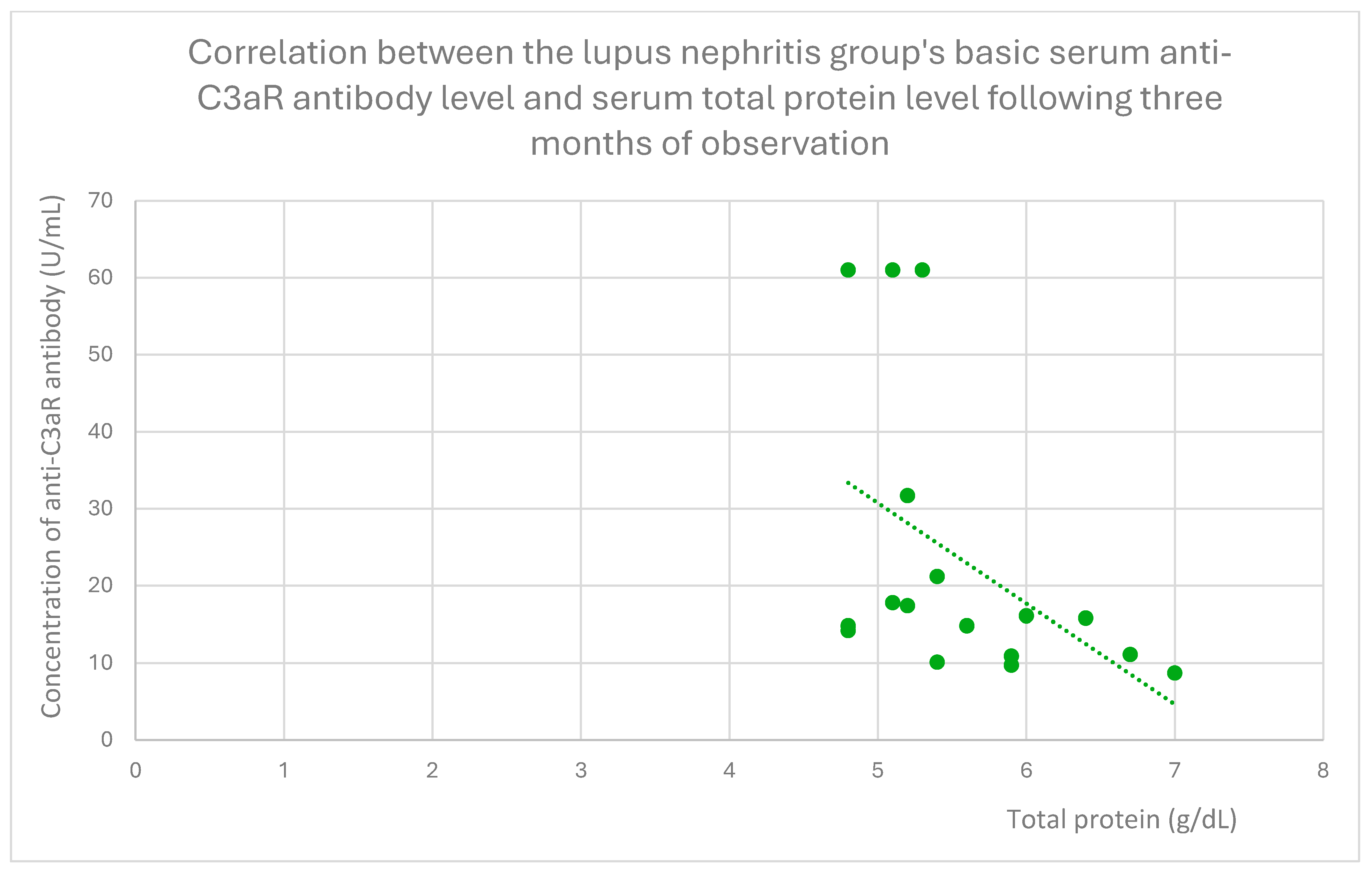
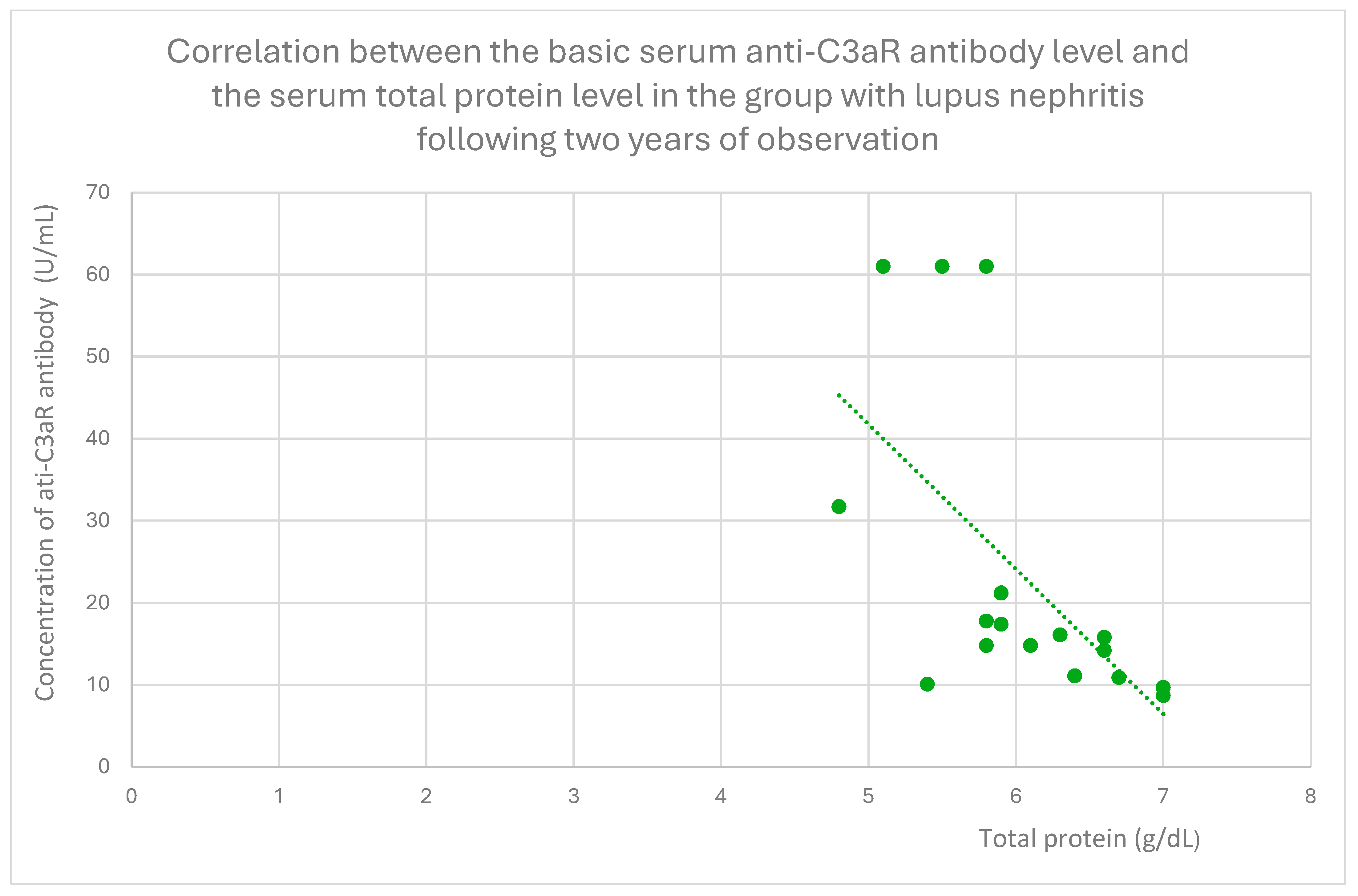
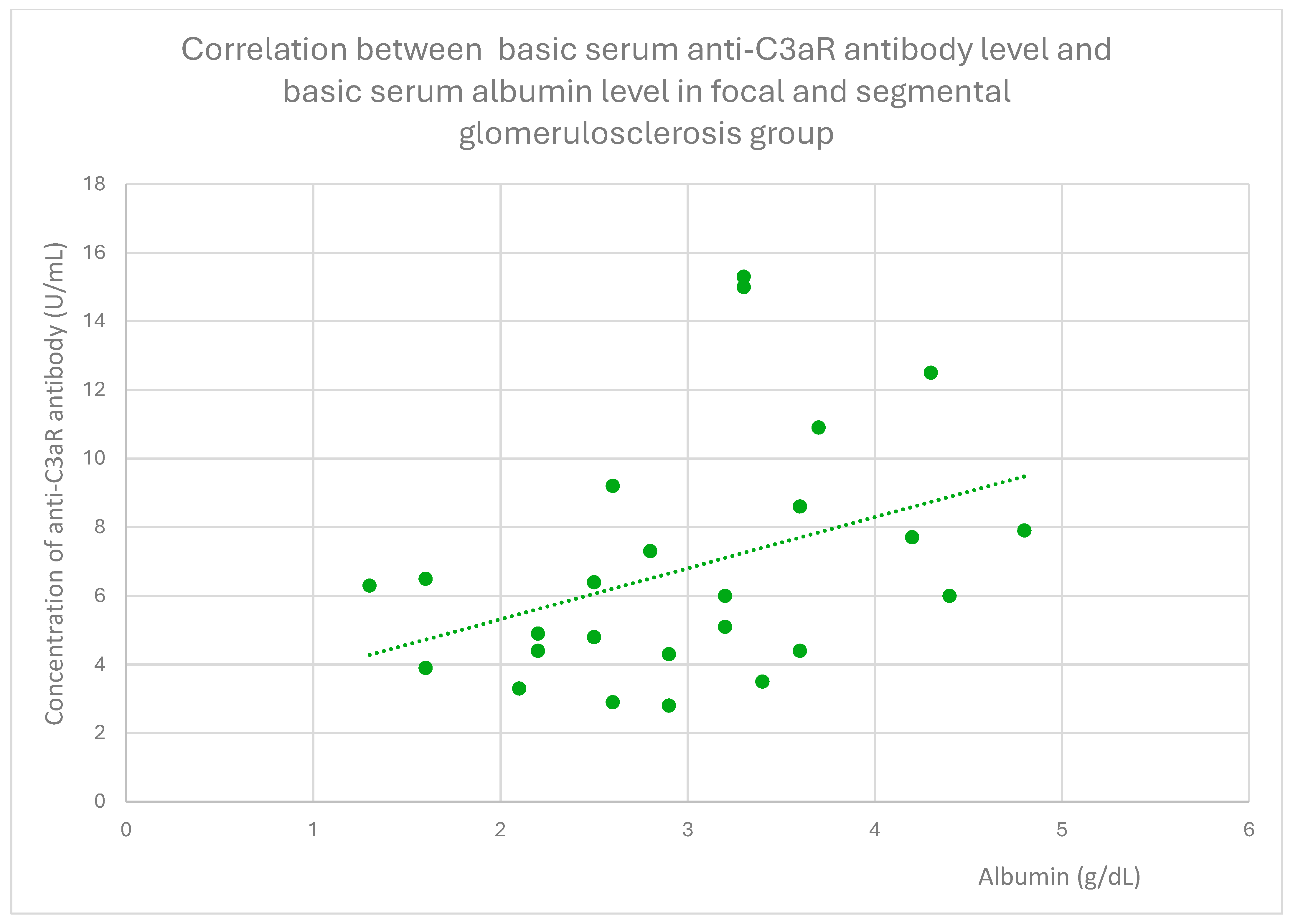
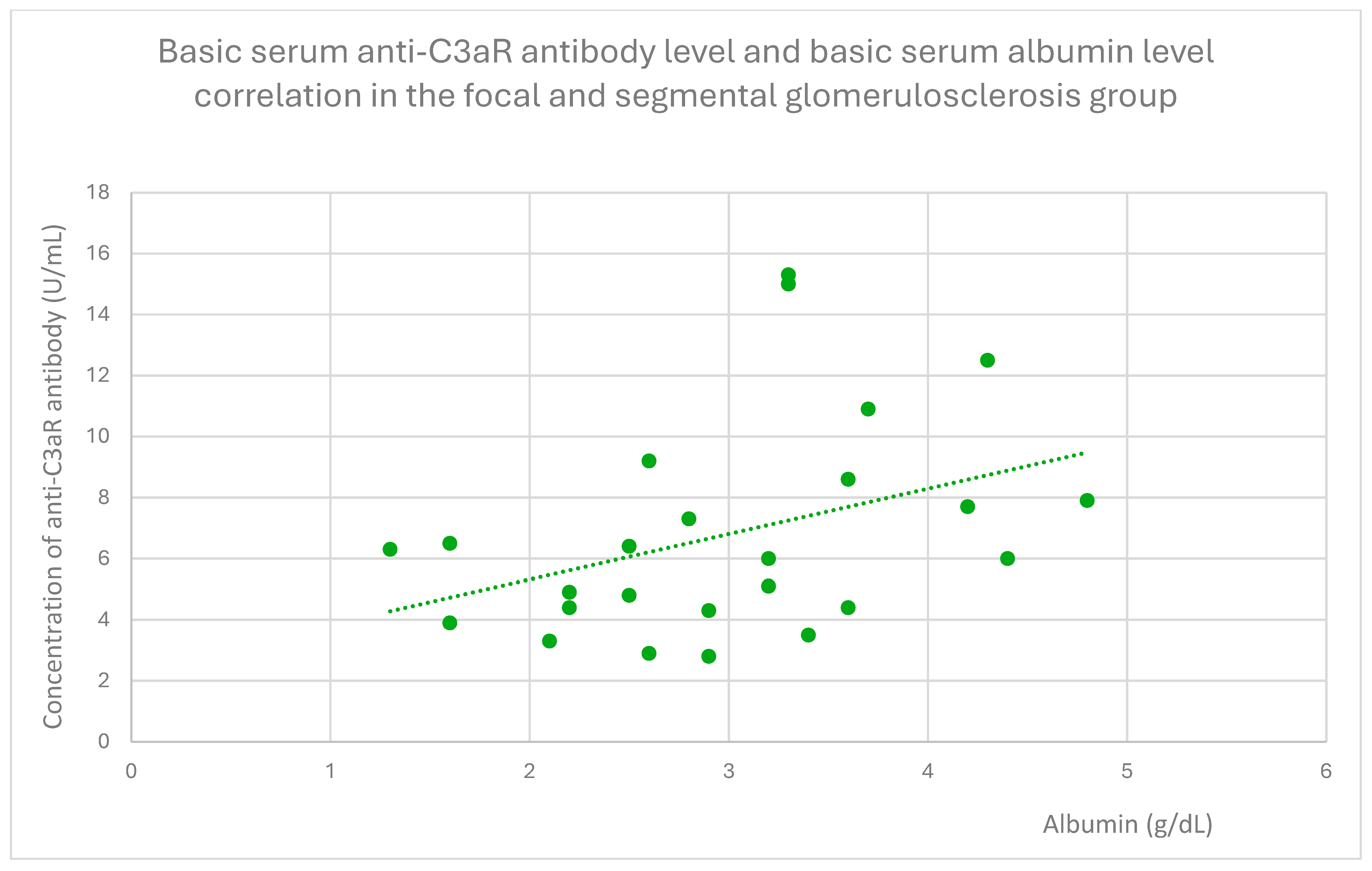
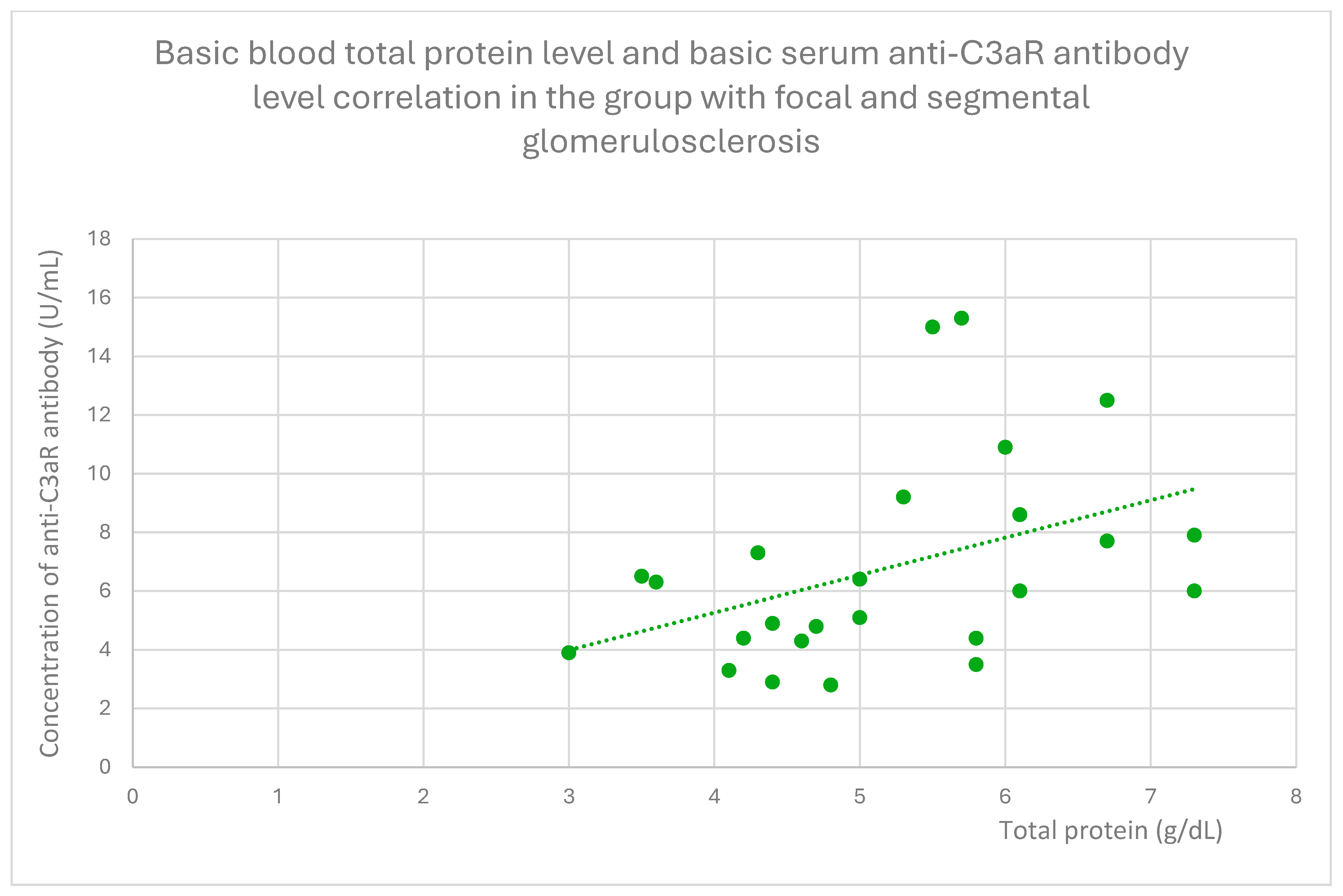
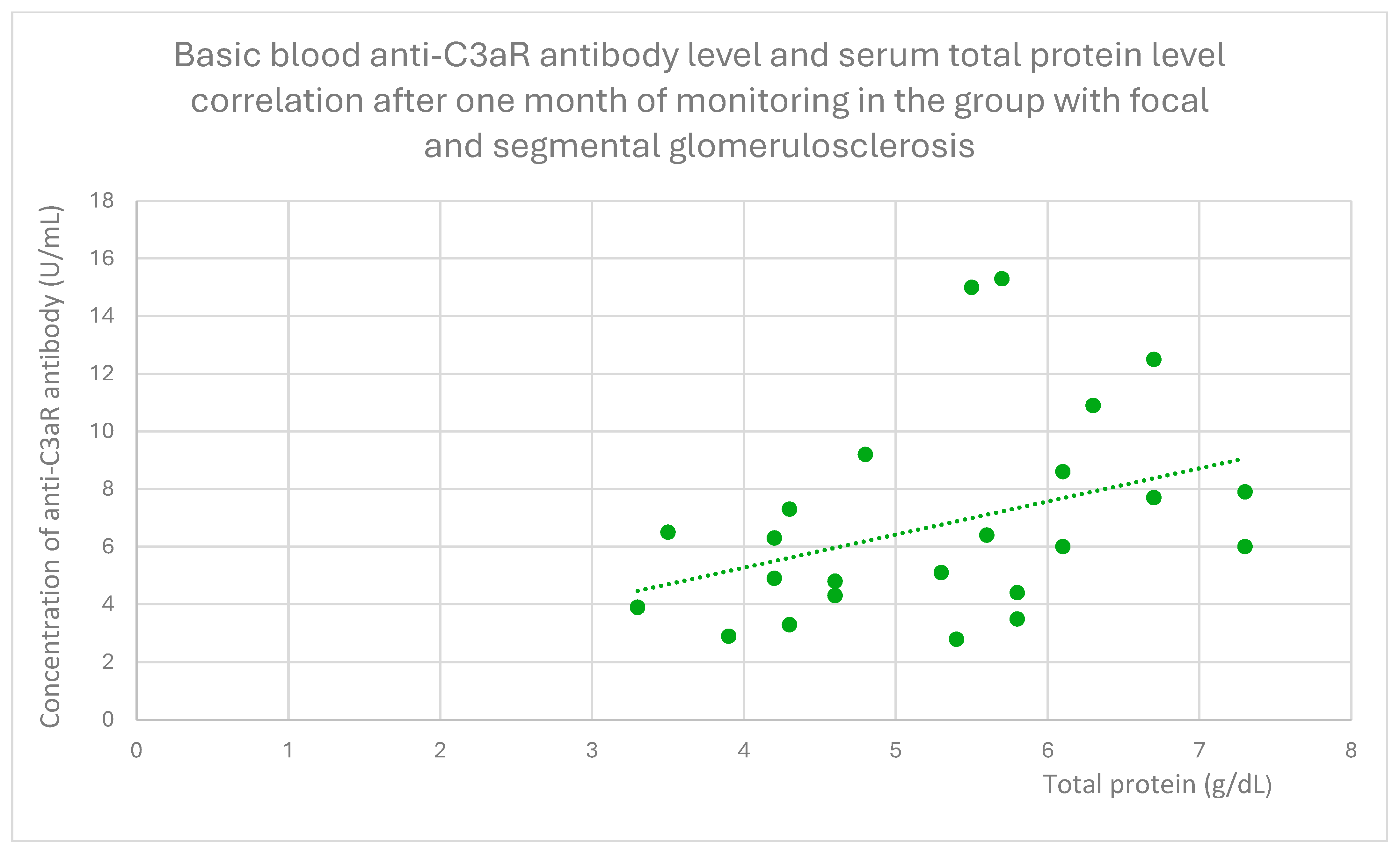
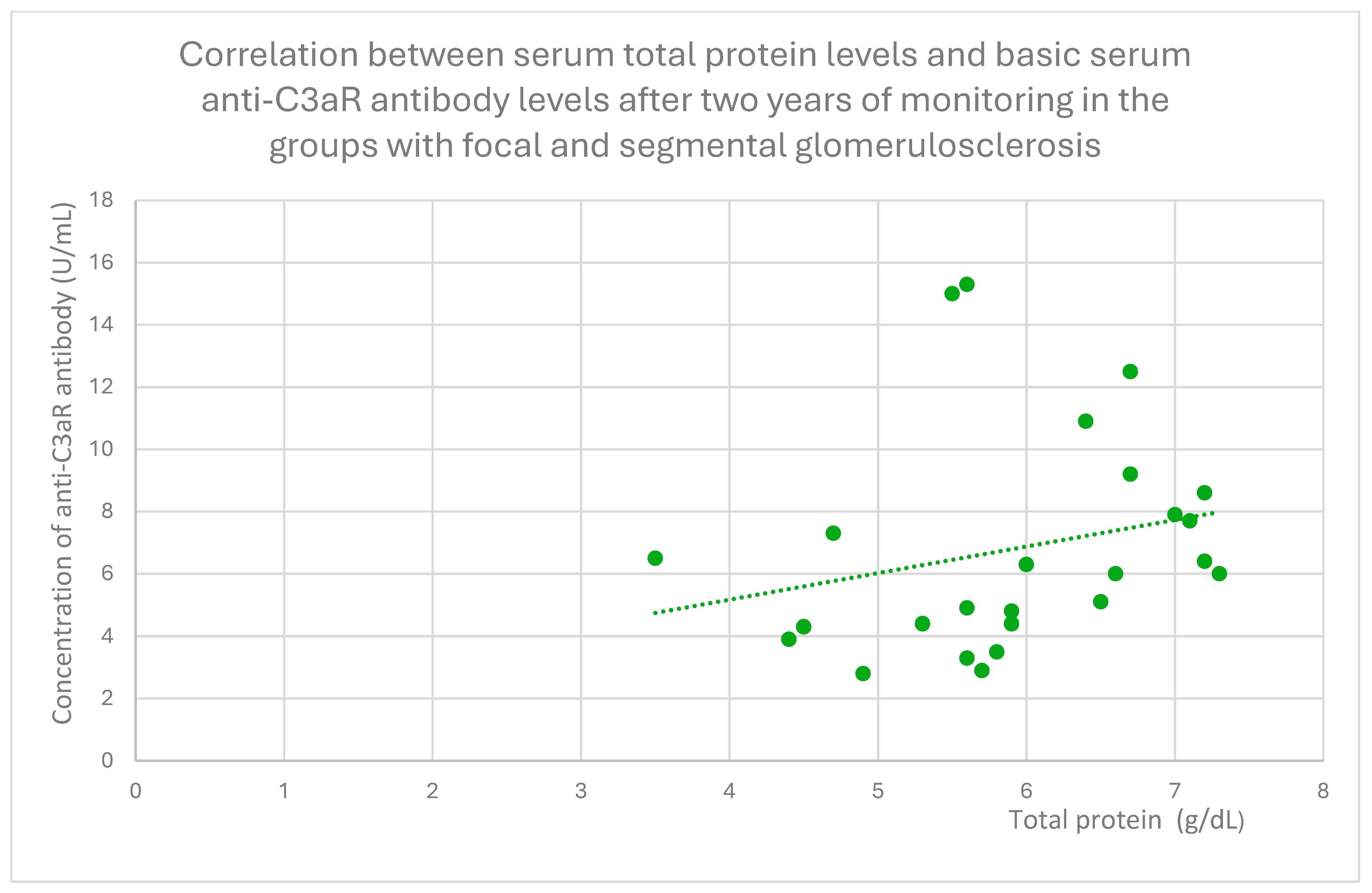
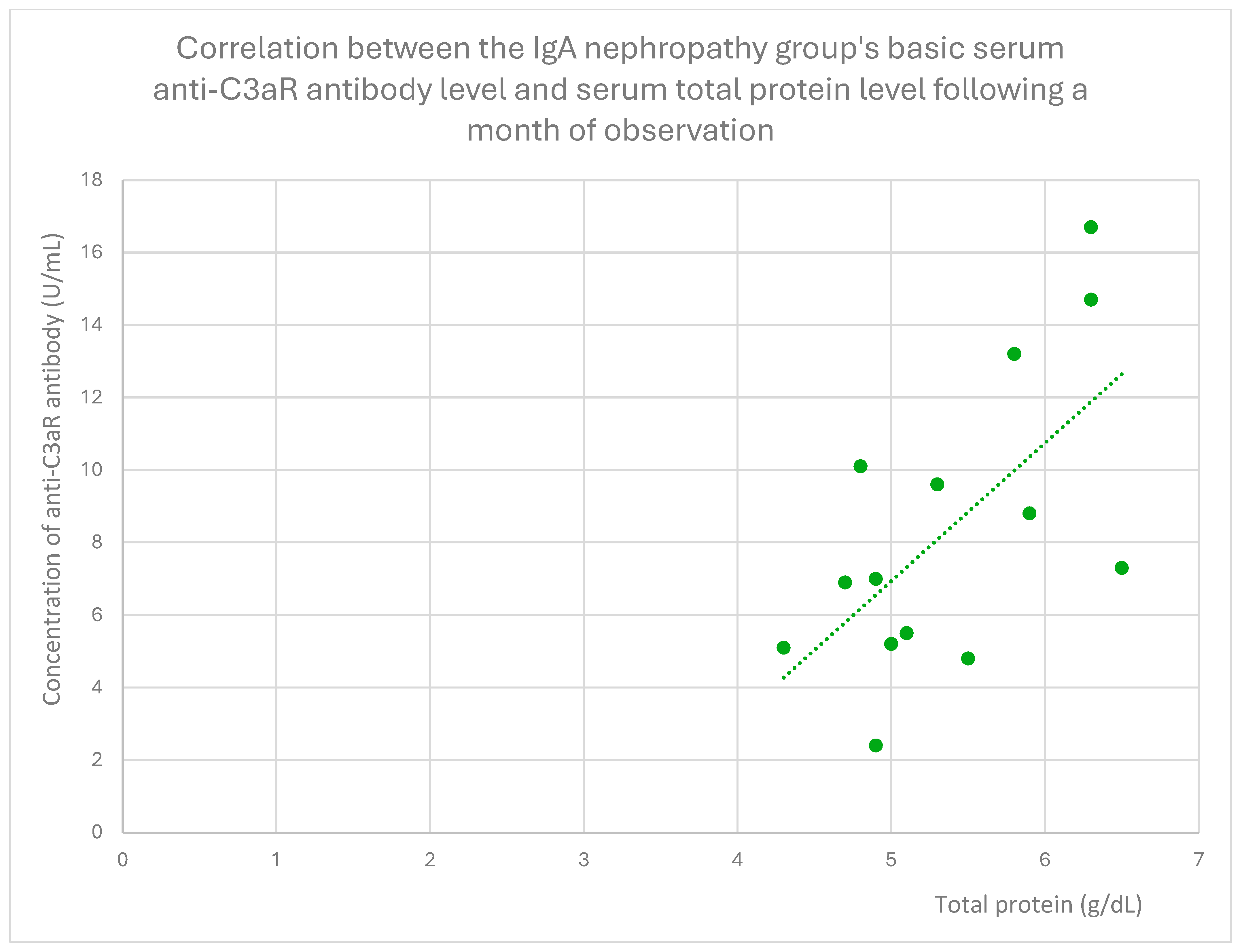
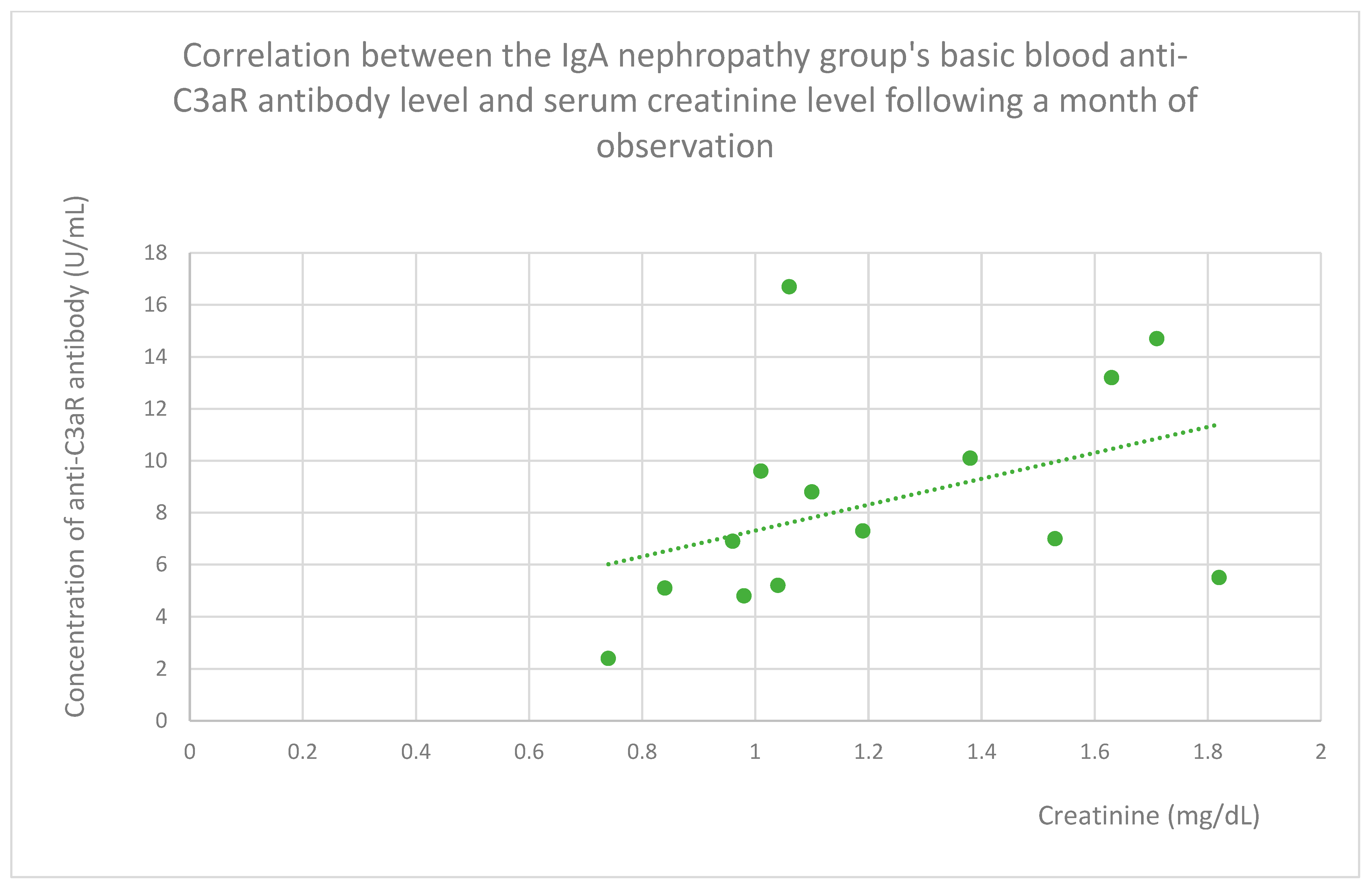
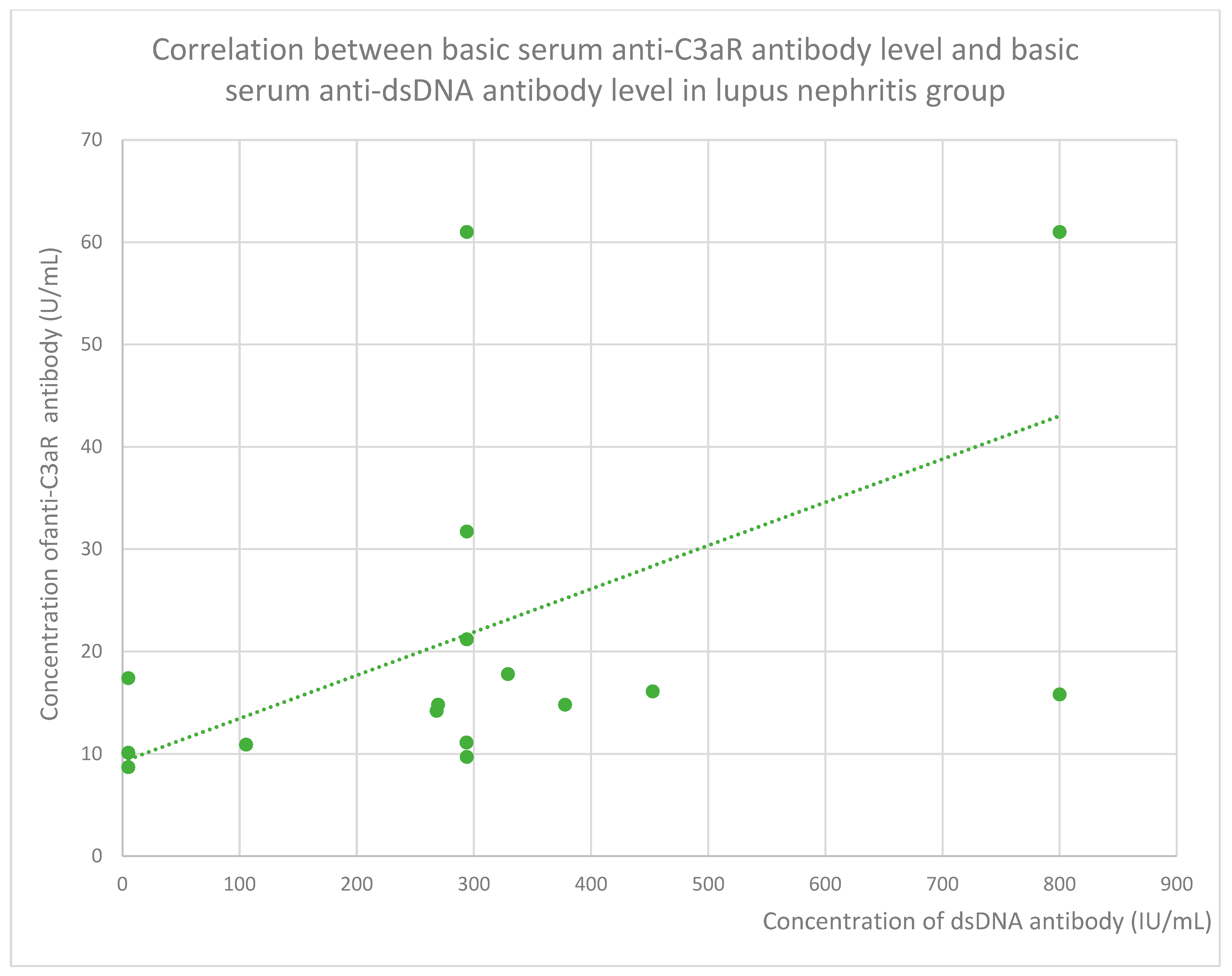
3.4. Relationships Between Patient Clinical Information and Anti-C3aR Antibody Levels
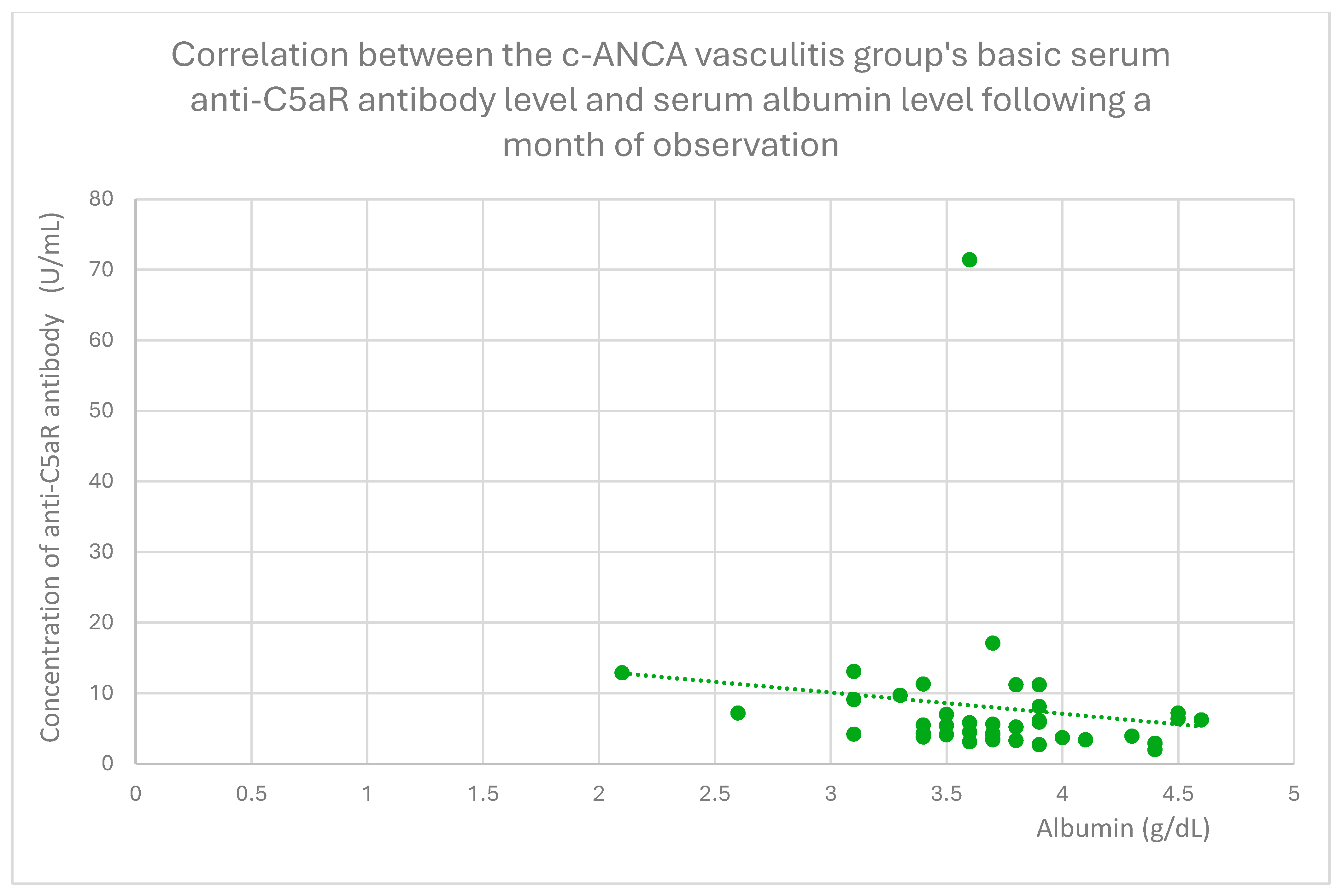
3.5. Relationships Between Patient Clinical Data and Anti-C5aR Antibody Level
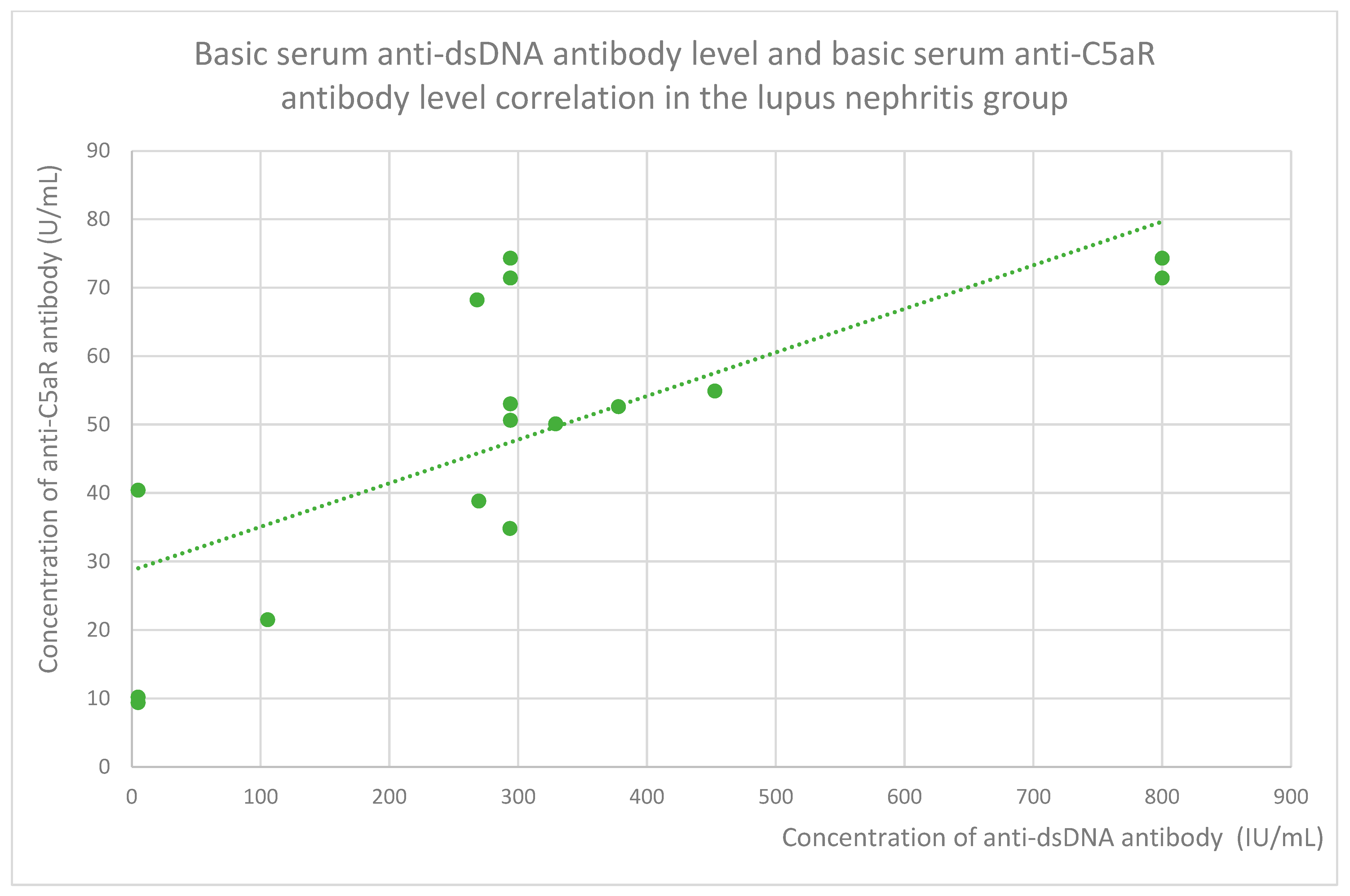
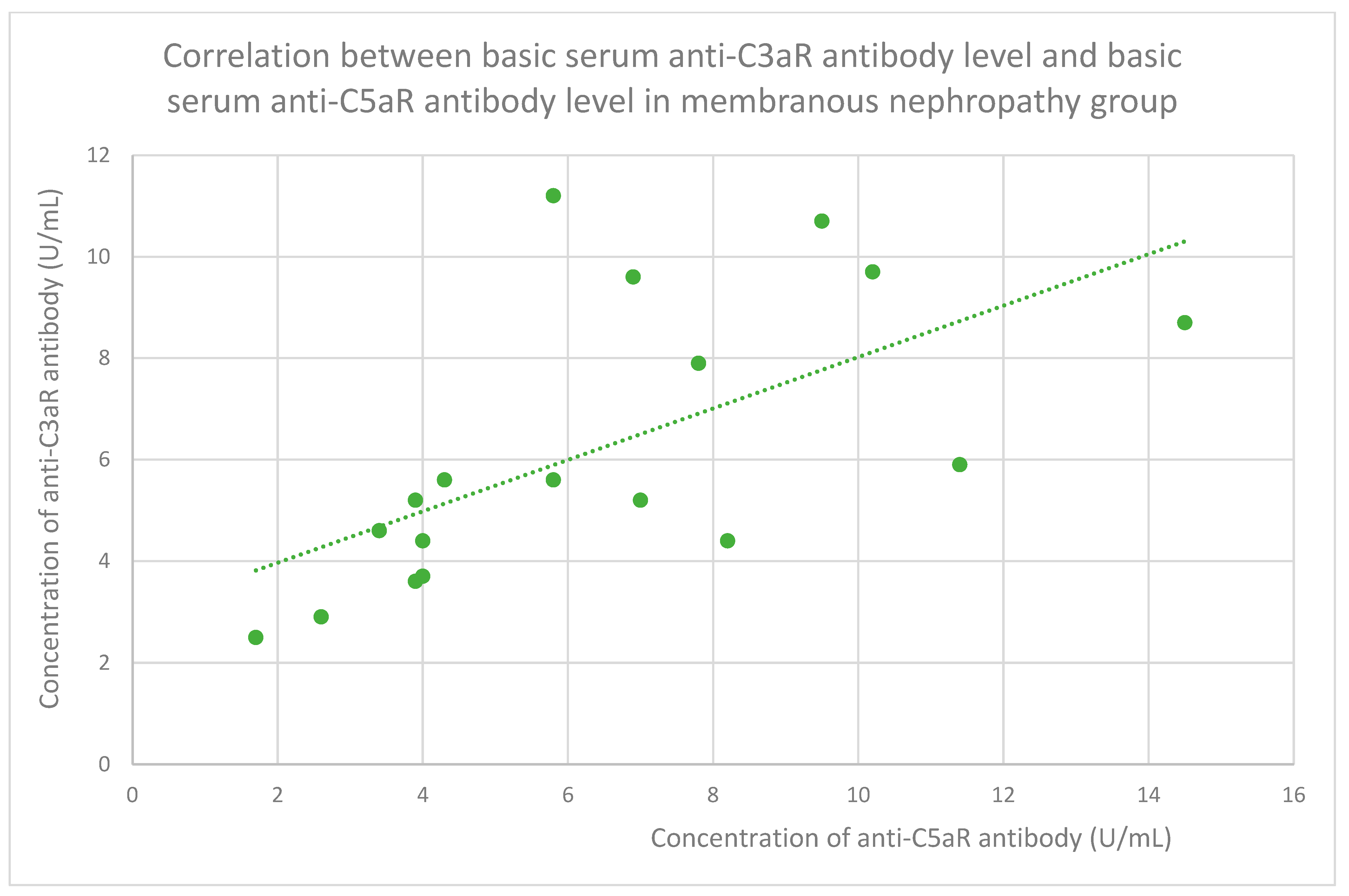
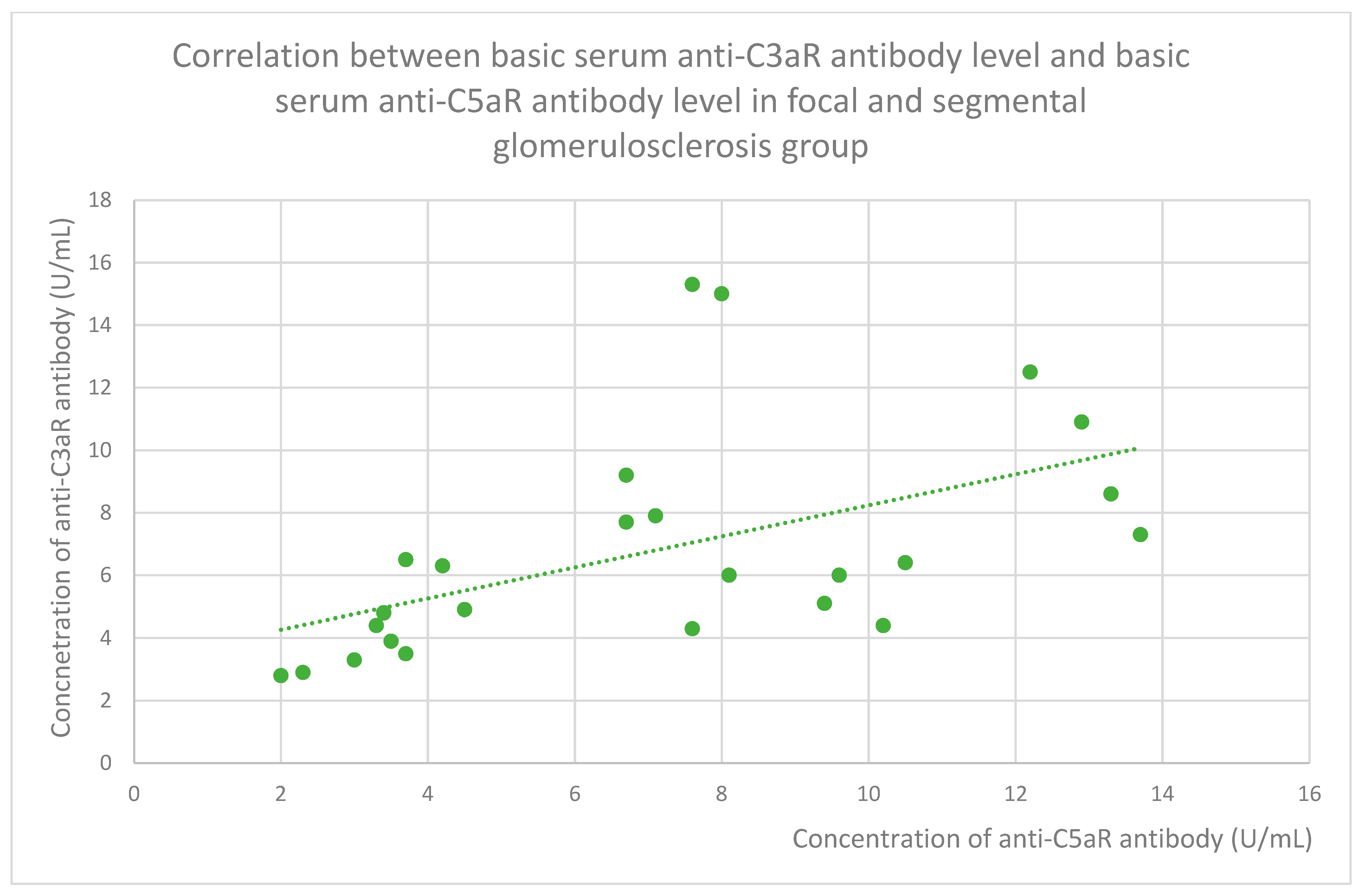
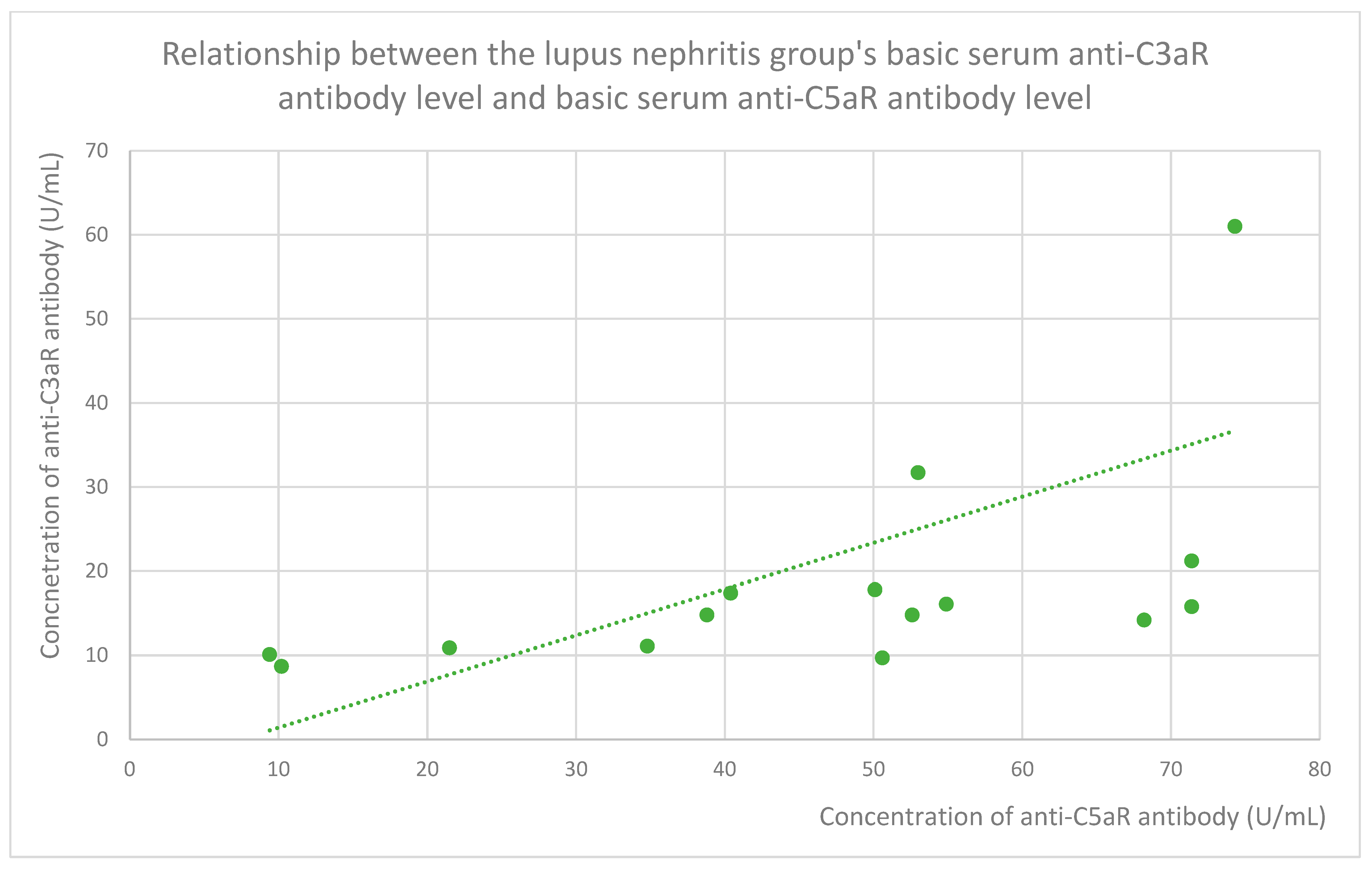
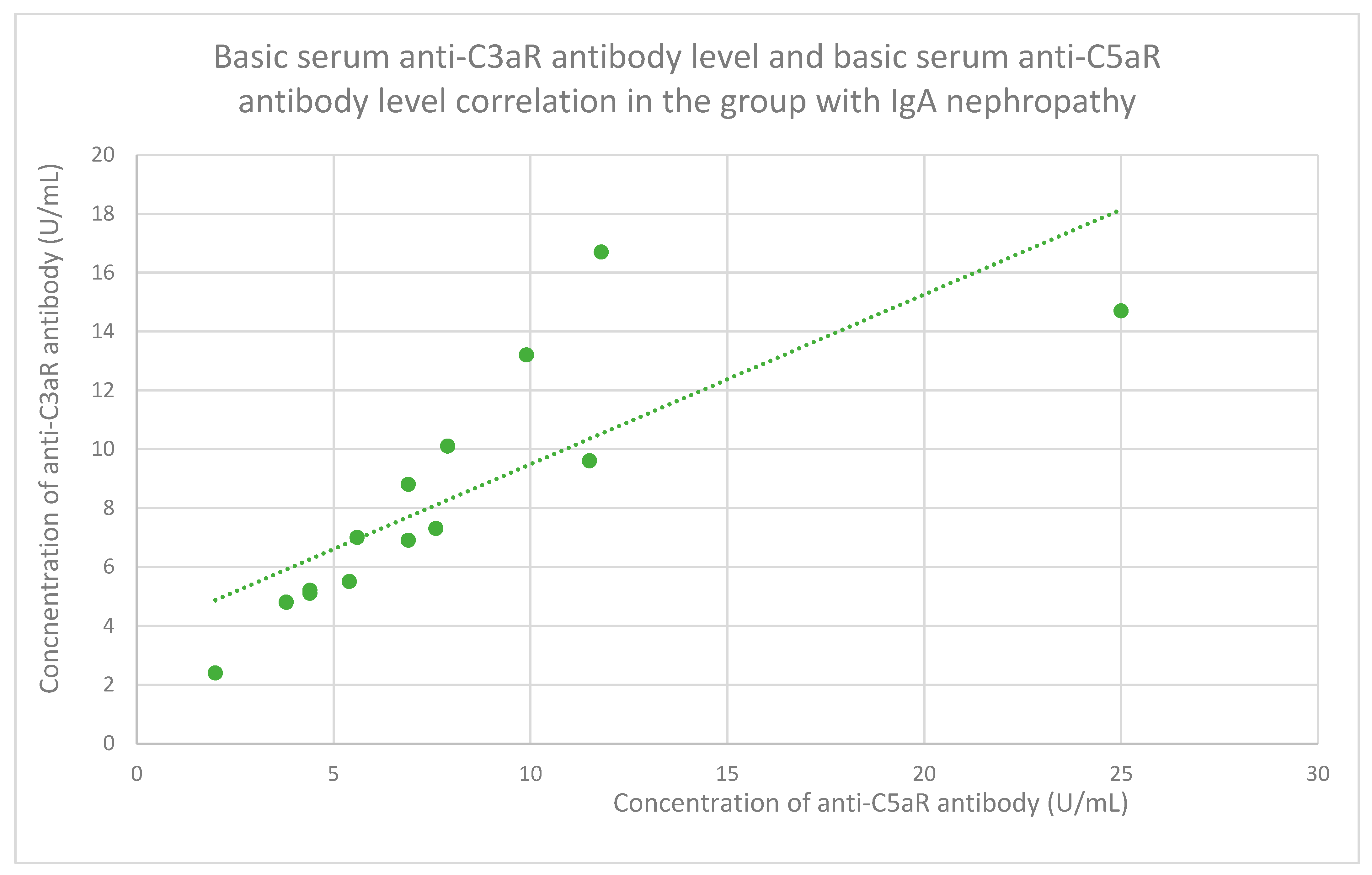
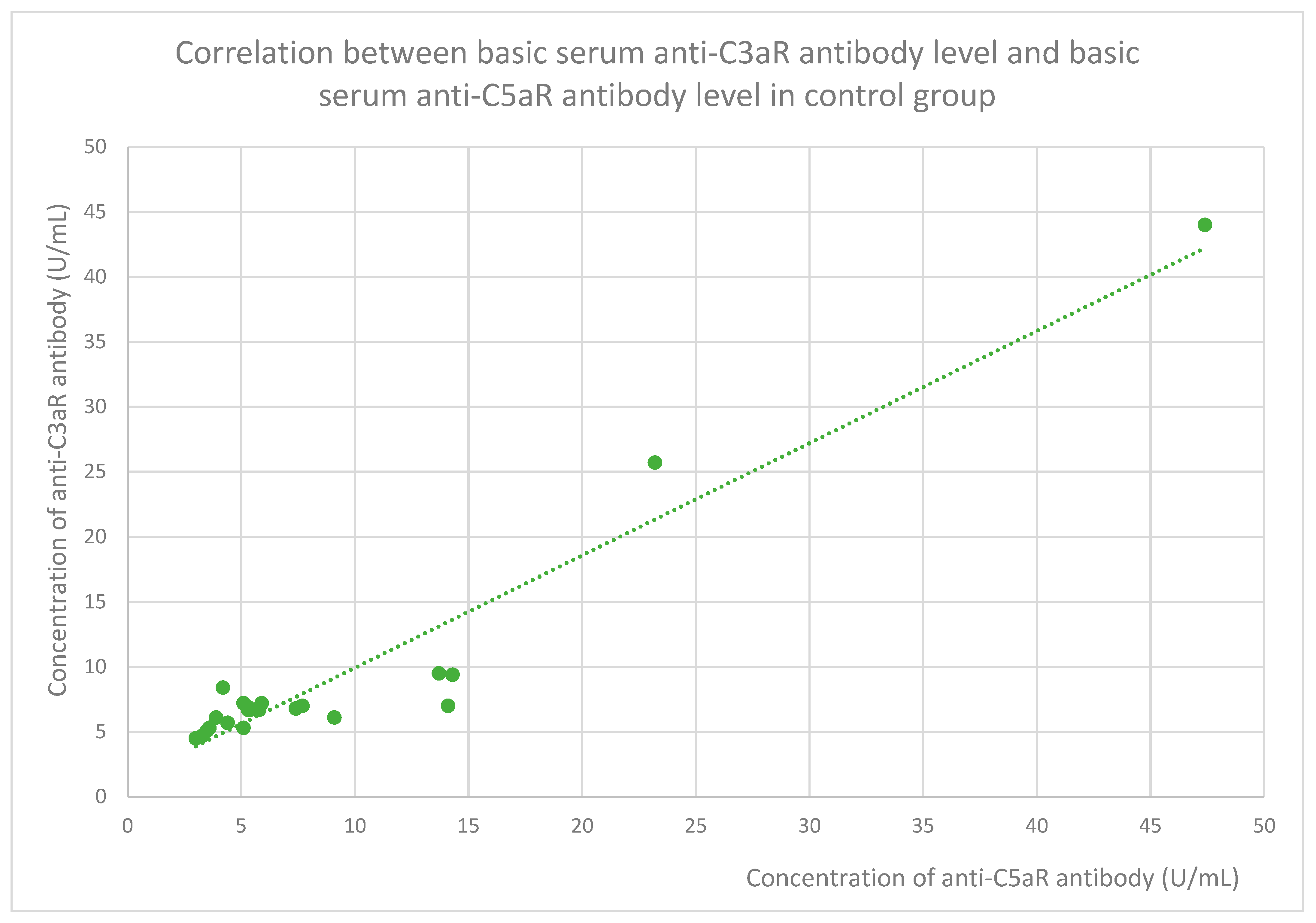
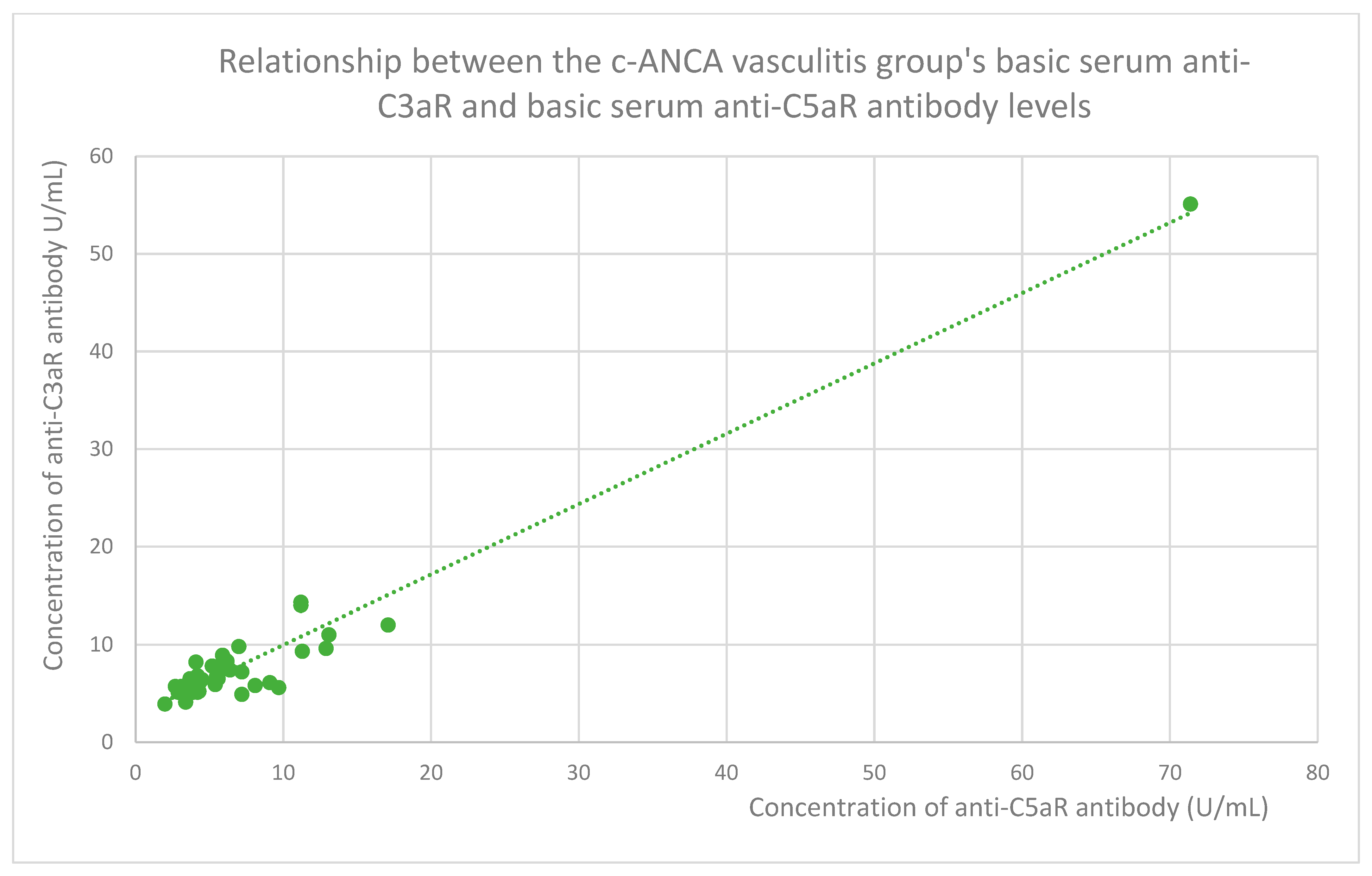
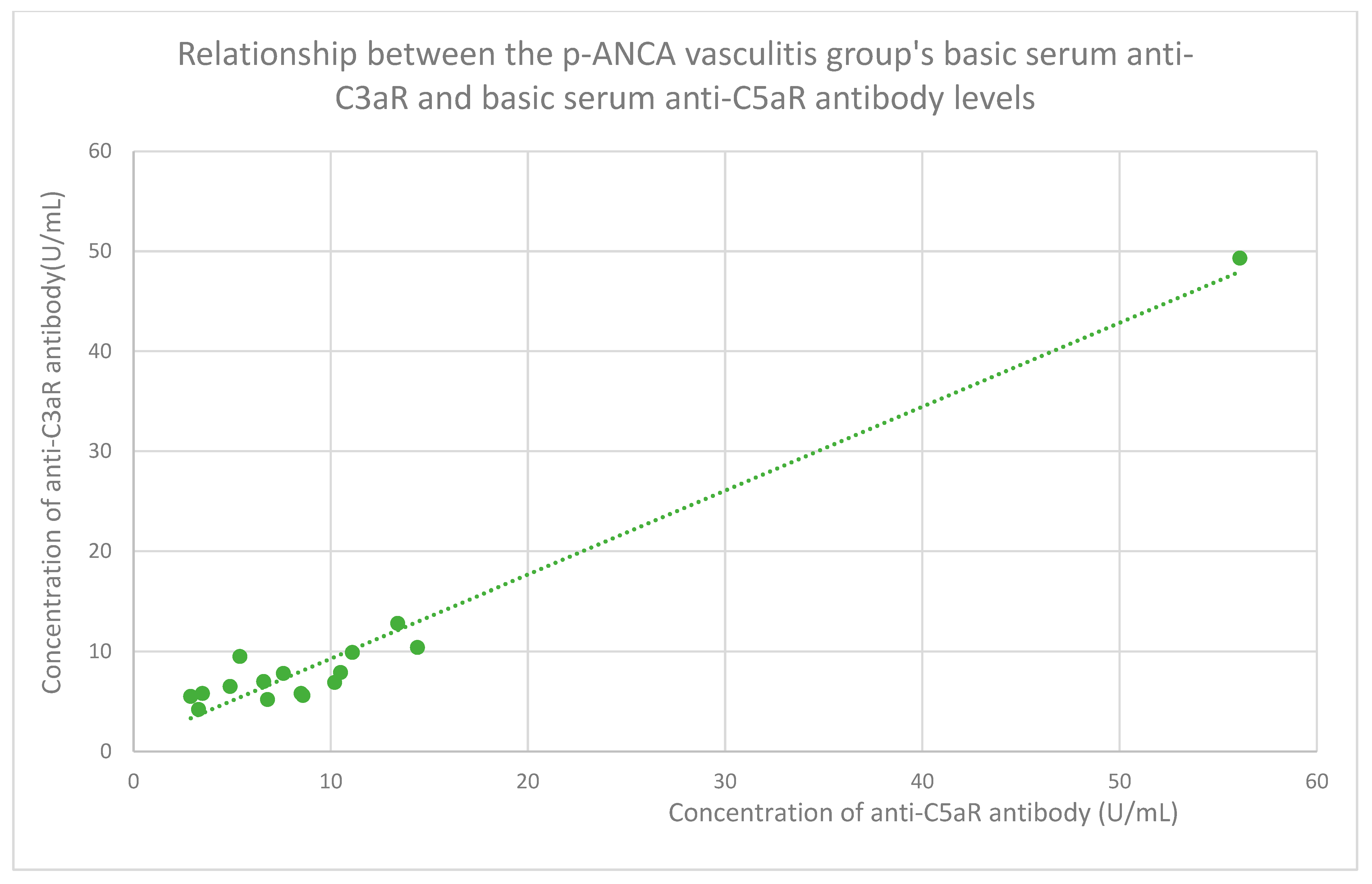
3.6. Anti-C3aR and Anti-C5aR Antibody Level Correlation
4. Discussion
4.1. Study Limitations
4.2. Future Perspectives
5. Conclusions
6. Disclosures
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANAs | antinuclear antibodies |
| ANOVA | analysis of variance |
| Anti-ACE2 | anti-angiotensin 2 converting enzyme antibody |
| Anti-AT1R | anti-angiotensin 2 type 1 receptor antibody |
| Anti-AT2R | anti-angiotensin 2 type 2 receptor antibody |
| Anti-CXCR3 | anti-C-X-C motif chemokine receptor 3 antibody |
| Anti-ETAR | anti endothelin A receptor antibody |
| Anti-PAR1 | anti proteinase-activated receptor type 1 antibody |
| cANCA | cytoplasmic anti-neutrophil cytoplasmic antibody |
| c3aR | complement 3a factor receptor |
| c5aR | complement 5a factor receptor |
| dsDNA | anti-double-stranded deoxyribonucleic acid antibody |
| eGFR | estimated glomerular filtration rate |
| ELISA | enzyme-linked immunosorbent assay |
| FSGS | focal and segmental glomerulosclerosis |
| GPCR | G-protein-coupled receptor |
| HCT | hematocrit |
| HLA | human leukocyte antigens |
| IgA | immunoglobulin A |
| IL-6 | interleukin 6 |
| IU | international unit |
| LN | lupus nephritis |
| MCP 1 | monocyte chemoattractant protein 1 |
| MDRD | Modification Diet in Renal Disease |
| m-RNA | mitochondrial ribonucleic acid |
| pANCA | perinuclear anti-neutrophil cytoplasmic antibody |
| POD | horseradish peroxidase |
| Th1 | T helper lymphocytes type 1 |
| TMB | tetramethylbenzidine |
| U | unit |
References
- Lohman, R.J.; Hamidon, J.K.; Reid, R.C.; Rowley, J.A.; Yau, M.K.; Halili, M.A.; Nielsen, D.S.; Lim, J.; Wu, K.C.; Loh, Z.; et al. Exploiting a novel conformational switch to control innate immunity mediated by complement protein C3a. Nat. Commun. 2017, 8, 351. [Google Scholar] [CrossRef]
- Strainic, M.G.; Shevach, E.M.; An, F.; Lin, F.; Medof, M.E. Absence of signaling into CD4(+) cells via C3aR and C5aR enables autoinductive TGF-beta1 signaling and induction of Foxp3(+) regulatory T cells. Nat. Immunol. 2013, 14, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Kildsgaard, J.; Hollmann, T.J.; Matthews, K.W.; Bian, K.; Murad, F.; Wetsel, R.A. Cutting edge: Targeted disruption of the C3a receptor gene demonstrates a novel protective anti-inflammatory role for C3a in endotoxin-shock. J. Immunol. 2000, 165, 5406–5409. [Google Scholar] [CrossRef]
- Hutamekalin, P.; Takeda, K.; Tani, M.; Tsuga, Y.; Ogawa, N.; Mizutani, N.; Yoshino, S. Effect of the C3a-receptor antagonist SB 290157 on anti-OVA polyclonal antibody-induced arthritis. J. Pharmacol. Sci. 2010, 112, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Baelder, R.; Fuchs, B.; Bautsch, W.; Zwirner, J.; Köhl, J.; Hoymann, H.G.; Glaab, T.; Erpenbeck, V.; Krug, N.; Braun, A. Pharmacological targeting of anaphylatoxin receptors during the effector phase of allergic asthma suppresses airway hyperresponsiveness and airway inflammation. J. Immunol. 2005, 174, 783–789. [Google Scholar] [CrossRef]
- Lim, J.; Iyer, A.; Suen, J.Y.; Seow, V.; Reid, R.C.; Brown, L.; Fairlie, D.P. C5aR and C3aR antagonists each inhibit diet-induced obesity, metabolic dysfunction, and adipocyte and macrophage signaling. FASEB J. 2013, 27, 822–831. [Google Scholar] [CrossRef]
- Mamane, Y.; Chung Chan, C.; Lavallee, G.; Morin, N.; Xu, L.J.; Huang, J.; Gordon, R.; Thomas, W.; Lamb, J.; Schadt, E.E.; et al. The C3a anaphylatoxin receptor is a key mediator of insulin resistance and functions by modulating adipose tissue macrophage infiltration and activation. Diabetes 2009, 58, 2006–2017. [Google Scholar] [CrossRef] [PubMed]
- Proctor, L.M.; Arumugam, T.V.; Shiels, I.; Reid, R.C.; Fairlie, D.P.; Taylor, S.M. Comparative anti-inflammatory activities of antagonists to C3a and C5a receptors in a rat model of intestinal ischaemia/reperfusion injury. Br. J. Pharmacol. 2004, 142, 756–764. [Google Scholar] [CrossRef] [PubMed]
- Akhir, F.N.M.; Noor, M.H.M.; Leong, K.W.K.; Nabizadeh, J.A.; Manthey, H.D.; Sonderegger, S.E.; Fung, J.N.T.; CMcGirr, C.E.; Shiels, J.A.; Mills, P.C.; et al. An Immunoregulatory Role for Complement Receptors in Murine Models of Breast Cancer. Antibodies 2021, 10, 2. [Google Scholar] [CrossRef]
- Grajales-Esquivel, E.; Luz-Madrigal, A.; Bierly, J.; Haynes, T.; Reis, E.S.; Han, Z.; Gutierrez, C.; McKinney, Z.; Tzekou, A.; Lambris, J.D.; et al. Complement component C3aR constitutes a novel regulator for chick eye morphogenesis. Dev. Biol. 2017, 428, 88–100. [Google Scholar] [CrossRef]
- Zhang, Q.; Bin, S.; Budge, K.; Petrosyan, A.; Villani, V.; Aguiari, P.; Vink, C.; Wetzels, J.; Soloyan, H.; La Manna, G.; et al. C3aR-initiated signaling is a critical mechanism of podocyte injury in membranous nephropathy. JCI Insight 2024, 9, e172976. [Google Scholar] [CrossRef]
- Angeletti, A.; Cantarelli, C.; Petrosyan, A.; Andrighetto, S.; Budge, K.; D’Agati, V.D.; Hartzell, S.; Malvi, D.; Donadei, C.; Thurman, J.M.; et al. Loss of decay-accelerating factor triggers podocyte injury and glomerulosclerosis. J. Exp. Med. 2020, 217, e20191699. [Google Scholar] [CrossRef]
- Mizuno, M.; Blanchin, S.; Gasque, P.; Nishikawa, K.; Matsuo, S. High levels of complement C3a receptor in the glomeruli in lupus nephritis. Am. J. Kidney Dis. 2007, 49, 598–606. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Duan, X.; Peng, Q.; Liu, Q.; Zhou, Y.; Quan, S.; Xing, G. C3a, C5a renal expression and their receptors are correlated to severity of IgA nephropathy. J. Clin. Immunol. 2014, 34, 224–232. [Google Scholar] [CrossRef]
- Dick, J.; Gan, P.Y.; Kitching, A.R.; Holdsworth, S.R. The C3aR promotes macrophage infiltration and regulates ANCA production but does not affect glomerular injury in experimental anti-myeloperoxidase glomerulonephritis. PLoS ONE 2018, 13, e0190655. [Google Scholar] [CrossRef]
- Obata, S.; Vaz de Castro, P.A.S.; Riella, L.V.; Cravedi, P. Recurrent C3 glomerulopathy after kidney transplantation. Transplant. Rev. 2024, 38, 100839. [Google Scholar] [CrossRef] [PubMed]
- Roquigny, J.; Meuleman, M.S.; El Sissy, C.; Martins, P.V.; Meri, S.; Duval, A.; Le Quintrec, M.; Fakhouri, F.; Chauvet, S.; Frémeaux-Bacchi, V. Acquired and genetic drivers of C3 and C5 convertase dysregulation in C3 glomerulopathy and immunoglobulin-associated MPGN. Nephrol. Dial. Transplant. 2025, 40, 1081–1090. [Google Scholar] [CrossRef]
- Han, R.; Hu, S.; Qin, W.; Shi, J.; Hou, Q.; Wang, X.; Xu, X.; Zhang, M.; Zeng, C.; Liu, Z.; et al. C3a and suPAR drive versican V1 expression in tubular cells of focal segmental glomerulosclerosis. JCI Insight 2019, 4, e130986. [Google Scholar] [CrossRef]
- Jacob, A.; Bao, L.; Brorson, J.; Quigg, R.J.; Alexander, J.J. C3aR inhibition reduces neurodegeneration in experimental lupus. Lupus 2010, 19, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Skiba, M.A.; Kruse, A.C. Autoantibodies as endogenous modulators of GPCR signaling. Trends Pharmacol Sci. 2021, 42, 135–150. [Google Scholar] [CrossRef] [PubMed]
- Yue, X.; Deng, F.; Chen, J.; Yin, J.; Zheng, J.; Chen, Y.; Huang, Q.; Gao, X.; Liu, Z.; Luo, J.; et al. Autoantibodies against C5aR1, C3aR1, CXCR3, and CXCR4 are decreased in primary Sjogren’s syndrome. Mol. Immunol. 2021, 131, 112–120. [Google Scholar]
- Pandey, S.; Maharana, J.; Li, X.X.; Woodruff, T.M.; Shukla, A.K. Emerging Insights into the Structure and Function of Complement C5a Receptors. Trends Biochem. Sci. 2020, 45, 693–705. [Google Scholar] [CrossRef] [PubMed]
- Mollnes, T.E.; Brekke, O.L.; Fung, M.; Fure, H.; Christiansen, D.; Bergseth, G.; Videm, V.; Lappegård, K.T.; Köhl, J.; Lambris, J.D. Essential role of the C5a receptor in E coli-induced oxidative burst and phagocytosis revealed by a novel lepirudin-based human whole blood model of inflammation. Blood 2002, 100, 1869–1877. [Google Scholar] [PubMed]
- Polat, Ö.; Orhun, G.; Anakli, I.; Yilmaz, V.; Koral, G.; Ulusoy, C.; Canbaz, M.; Ozcan, P.E.; Tüzün, E.; Esen, F. Renal Expression Levels of C5a Receptor and Autophagy-related Beclin-1 and LC3A/B Are Simultaneously Enhanced Under Immunoglobulin Treatment in a Rat Model of Sepsis. In Vivo 2025, 39, 810–818. [Google Scholar] [CrossRef]
- Bao, L.; Osawe, I.; Puri, T.; Lambris, J.D.; Haas, M.; Quigg, R.J. C5a promotes development of experimental lupus nephritis which can be blocked with a specific receptor antagonist. Eur. J. Immunol. 2005, 35, 2496–2506. [Google Scholar] [CrossRef]
- Atkinson, S.M.; Nansen, A.; Usher, P.A.; Sondergaard, B.C.; Mackay, C.R.; Friedrichsen, B.; Chang, C.C.; Tang, R.; Skov, S.; Haase, C.; et al. Treatment with anti-C5aR mAb leads to early-onset clinical and mechanistic effects in the murine delayed-type hypersensitivity arthritis model. Autoimmunity 2015, 48, 460–470. [Google Scholar] [CrossRef]
- Suwa, T.; Lee, K.S.; Chai, I.J.; Clark, H.O.L.; MacLean, D.J.; Machado, N.; Rodriguez-Berriguete, G.; Singh, L.; Higgins, G.S.; Hammond, E.M.; et al. UPR-induced intracellular C5aR1 promotes adaptation to the hypoxic tumour microenvironment. Cell Death Dis. 2025, 1, 547. [Google Scholar] [CrossRef]
- Liu, G.; Wang, Q.; Shi, Y.; Peng, X.; Liu, H.; Peng, Y.; He, L. Resveratrol Attenuates Adriamycin-Induced Focal Segmental Glomerulosclerosis through C3aR/C5aR- Sphingosine Kinase 1 Pathway. Pharmacology 2017, 100, 253–260. [Google Scholar] [CrossRef]
- Gong, X.J.; Huang, J.; Shu, Y.; Wang, M.; Ji, J.; Yang, L.; Zhao, M.H.; Cui, Z. Complement C5a and C5a receptor 1 mediates glomerular damage in focal segmental glomerulosclerosis. Clin. Immunol. 2025, 273, 110459. [Google Scholar] [CrossRef]
- Ye, B.; Chen, B.; Guo, C.; Xiong, N.; Huang, Y.; Li, M.; Lai, Y.; Li, J.; Zhou, M.; Wang, S.; et al. C5a-C5aR1 axis controls mitochondrial fission to promote podocyte injury in lupus nephritis. Mol. Ther. 2024, 32, 1540–1560. [Google Scholar]
- Zhang, Y.; Yan, X.; Zhao, T.; Xu, Q.; Peng, Q.; Hu, R.; Quan, S.; Zhou, Y.; Xing, G. Targeting C3a/C5a receptors inhibits human mesangial cell proliferation and alleviates immunoglobulin A nephropathy in mice. Clin. Exp. Immunol. 2017, 189, 60–70. [Google Scholar] [CrossRef]
- Batista-Liz, J.C.; Calvo-Río, V.; Mora-Gil, M.S.; Leonardo, M.T.; Peñalba, A.C.; Martín-Penagos, L.; Narváez, J.; Sevilla-Pérez, B.; Callejas-Rubio, J.L.; Gabrie, L.; et al. Evaluating the usefulness of C5 and C5AR1 as genetic biomarkers of IgA-mediated vasculitis. Mol. Med. 2025, 31, 267. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Gou, S.J.; Huang, J.; Hao, J.; Chen, M.; Zhao, M.H. C5a and its receptors in human anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis. Arthritis Res. Ther. 2012, 14, R140. [Google Scholar] [CrossRef]
- Xiao, H.; Dairaghi, D.J.; Powers, J.P.; Ertl, L.S.; Baumgart, T.; Wang, Y.; Seitz, L.C.; Penfold, M.E.; Gan, L.; Hu, P.; et al. C5a receptor (CD88) blockade protects against MPO-ANCA GN. J. Am. Soc. Nephrol. 2014, 25, 225–231. [Google Scholar] [CrossRef]
- Schreiber, A.; Xiao, H.; Jennette, J.C.; Schneider, W.; Luft, F.C.; Kettritz, R. C5a receptor mediates neutrophil activation and ANCA-induced glomerulonephritis. J. Am. Soc. Nephrol. 2009, 20, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.L.; Gullipalli, D.; Ueda, Y.; Sato, S.; Zhou, L.; Miwa, T.; Tung, K.S.; Song, W.C. C5 inhibition prevents renal failure in a mouse model of lethal C3 glomerulopathy. Kidney Int. 2017, 91, 1386–1397. [Google Scholar] [CrossRef] [PubMed]
- Ueda, Y.; Miwa, T.; Ito, D.; Kim, H.; Sato, S.; Gullipalli, D.; Zhou, L.; Golla, M.; Song, D.; Dunaief, J.L.; et al. Differential contribution of C5aR and C5b-9 pathways to renal thrombic microangiopathy and macrovascular thrombosis in mice carrying an atypical hemolytic syndrome-related factor H mutation. Kidney Int. 2019, 96, 67–79. [Google Scholar] [CrossRef]
- Xie, R.C.; Zhang, J.C.; Huang, T.; Lin, X.M.; Wang, Y.T.; Zhang, L.F.; Hong, X.Y.; Lin, X.F.; Zheng, H.J.; Zhou, K.L.; et al. Complement C5aR blockade attenuates LPS-induced acute kidney injury by regulating ferroptosis via nuclear factor-erythroid 2-related factor 2 signaling in mice. Free Radic. Biol. Med. 2025, 239, 104–115. [Google Scholar] [CrossRef]
- Wenderfer, S.E.; Ke, B.; Hollmann, T.J.; Wetsel, R.A.; Lan, H.Y.; Braun, M.C. C5a receptor deficiency attenuates T cell function and renal disease in MRLlpr mice. J. Am. Soc. Nephrol. 2005, 16, 3572–3582. [Google Scholar] [CrossRef]
- Colley, C.S.; Popovic, B.; Sridharan, S.; Debreczeni, J.E.; Hargeaves, D.; Fung, M.; An, L.L.; Edwards, B.; Arnold, J.; England, E.; et al. Structure and characterization of a high affinity C5a monoclonal antibody that blocks binding to C5aR1 and C5aR2 receptors. MAbs 2018, 10, 104–117. [Google Scholar] [CrossRef]
- Klapa, S.; Müller, A.; Koch, A.; Kerstein-Stähle, A.; Kähler, W.; Heidecke, H.; Schinke, S.; Huber-Lang, M.; Nitschke, M.; Pitann, S.; et al. Low Concentrations of C5a Complement Receptor Antibodies Are Linked to Disease Activity and Relapse in Antineutrophil Cytoplasmic Autoantibody-Associated Vasculitis. Arthritis Rheumatol. 2023, 75, 760–767. [Google Scholar] [CrossRef] [PubMed]
- Cabral-Marques, O.; Marques, A.; Giil, L.M.; De Vito, R.; Rademacher, J.; Günther, J.; Lange, T.; Humrich, J.Y.; Klapa, S.; Schinke, S.; et al. GPCR-specific autoantibody signatures are associated with physiological and pathological immune homeostasis. Nat. Commun. 2018, 9, 5224. [Google Scholar] [CrossRef] [PubMed]
- Klapa, S.; Müller, A.; Arnold, S.; Koch, A.; Staehle, A.; Kaehler, W.; Heidecke, H.; Riemekasten, G.; Karsten, C.M.; Lamprecht, P. Decreased concentrations of C5a complement receptor antibodies are associated with relapse in eosinophilic granulomatosis with polyangiitis (EGPA). RMD Open 2025, 11, e005323. [Google Scholar] [CrossRef]
- Legler, D.F.; Loetscher, M.; Jones, S.A.; Dahinden, C.A.; Arock, M.; Moser, B. Expression of high- and low-affinity receptors for C3a on the human mast cell line, HMC-1. Eur. J. Immunol. 1996, 26, 753–758. [Google Scholar] [CrossRef]
- FDA Bioanalytical Method Validation Guidance. Available online: https://www.fda.gov/media/70858/download (accessed on 4 August 2025).
- Szymczak, M.; Heidecke, H.; Żabińska, M.; Janek, Ł.; Wronowicz, J.; Kujawa, K.; Schulze-Forster, K.; Marek-Bukowiec, K.; Gołębiowski, T.; Banasik, M. The Influence of Anti-PAR 1 and Anti-ACE 2 Antibody Levels on the Course of Specific Glomerulonephritis Types. J. Clin. Med. 2025, 14, 3178. [Google Scholar] [CrossRef]
- Suresh, V.; Stillman, I.E.; Campbell, K.N.; Meliambro, K. Focal Segmental Glomerulosclerosis. Adv. Kidney Dis. Health 2024, 31, 275–289. [Google Scholar] [CrossRef]
- Mathern, D.R.; Heeger, P.S. Molecules Great and Small: The Complement System. Clin. J. Am. Soc. Nephrol. 2015, 10, 1636–1650. [Google Scholar] [CrossRef]
- Szymczak, M.; Heidecke, H.; Żabińska, M.; Rukasz, D.; Wiśnicki, K.; Kujawa, K.; Kościelska-Kasprzak, K.; Krajewska, M.; Banasik, M. Angiotensin II Type 2 Receptor Antibodies in Glomerular Diseases. Arch. Immunol. Ther. Exp. 2024, 19, 72. [Google Scholar] [CrossRef]
- Szymczak, M.; Heidecke, H.; Zabińska, M.; Janek, Ł.; Wronowicz, J.; Kujawa, K.; Bukowiec-Marek, K.; Gołębiowski, T.; Skalec, K.; Schulze-Forster, K.; et al. The influence of anti-ETAR and anti-CXCR3 antibody levels on the course of specific glomerulonephritis types. J. Clin. Med. 2024, 13, 7752. [Google Scholar] [CrossRef]
- Woodruff, T.M.; Nandakumar, K.S.; Tedesco, F. Inhibiting the C5-C5a receptor axis. Mol. Immunol. 2011, 48, 1631–1642. [Google Scholar] [CrossRef] [PubMed]
- Gattu, R.; Demory Beckler, M.; Kesselman, M.M. Superiority of Avacopan and Mepolizumab to Glucocorticoid Tapering in the Treatment of Anti-neutrophil Cytoplasmic Antibody (ANCA)-Associated Vasculitis: A Systematic Review. Cureus 2024, 16, e67161. [Google Scholar] [CrossRef] [PubMed]

| Specific Glomerular Disorder | Basic Level of Creatinine in Serum (mg/dL) | Basic Estimated Glomerular Filtration Rate (mL/min/1.73m2) MDRD (Modification Diet in Renal Disease) | Basic Blood Urea Nitrogen (mg/dL) | Albumin-to- Creatinine Ratio | Proteinuria (mg/per Day) | Basic Level of Total Protein in Serum (g/dL) | Basic Level of Albumin in Serum (g/dL) |
|---|---|---|---|---|---|---|---|
| membranous glomerulonephritis (n = 18) | 1.25 (0.8–3.3) | 66 (15–106) | 12 (8–32) | 1.6 (0.3–7.1) | 2640 (100–15,800) | 4.8 (3.7–5.9) | 2.8 (1.7–3.9) |
| focal and segmental glomerulosclerosis (n = 25) | 1.21 (0.73–3.19) | 68 (26–126) | 12 (8–30) | 1.3 (0.3–7.5) | 2300 (70–13,990) | 5 (3–7.3) | 2.9 (1.3–4.8) |
| lupus nephritis (n = 17) | 1.06 (0.77–2.19) | 68 (31–116) | 9 (7–23) | 0.8 (0.3–3.1) | 1590 (180–5950) | 5.5 (3.8–7.3) | 3.1 (2–4) |
| IgA nephropathy (n = 14) | 1.06 (0.71–1.82) | 70 (35–131) | 9.5 (7–20) | 0.6 (0.3–2.2) | 940 (90–4540) | 5.65 (4.4–6.5) | 3.4 (2.2–4) |
| mesangial proliferative (non-IgA) glomerulonephritis (n = 6) | 0.93 (0.59–1.55) | 105 (40–131) | 8.5 (6–16) | 0.8 (0.4–2.9) | 2580 (620–7130) | 4.8 (3.9–5.2) | 2.8 (1.6–3.2) |
| control group (n = 22) | 1.2 (0.9–1.3) | 63 (60–78) | 12 (9–16) | 0 (0–0) | 0 (0–0) | 7.4 (6.6–8.2) | 4.4 (3.5–5.2) |
| c-ANCA vasculitis (n = 40) | 1.81 (0.69–7.78) | 45 (7–126) | 19.8 (7–75) | 0.4 (0.3–10.9) | 640 (60–19,000) | 6.3 (5.3–7.1) | 3.6 (2.4–4.6) |
| p-ANCA vasculitis (n = 16) | 3.13 (0.79–9.04) | 19 (5–93) | 30 (8–81) | 0.8 (0.3–5) | 1730 (140–12,300) | 5.95 (4.8–8.3) | 3.5 (2.8–4.3) |
| Specific Glomerular Disorder | Age (Years) | Sex (Ratio of Men/Women) | Hemoglobin (g/dL) | HCT (Hematocrit) (%) | Leukocytes (Per Microliter) |
|---|---|---|---|---|---|
| membranous glomerulonephritis (n = 18) | 51.5 (28–69) | 1.22 | 13.5 (11.3–16) | 44.5 (38–52.6) | 6715 (2500–10,780) |
| focal and segmental glomerulosclerosis (n = 25) | 48 (19–74) | 1.27 | 13.9 (9.3–18) | 45.7 (36.8–55.2) | 7690 (4130–10,500) |
| lupus nephritis (n = 17) | 34 (19–66) | 0.89 | 13.2 (10.5–17.3) | 43 (34.5–56.9) | 6600 (4090–10,900) |
| IgA nephropathy (n = 14) | 45.5 (20–60) | 1 | 14.8 (12.2–16.5) | 48.6 (46–54.3) | 8420 (5360–10,900) |
| Mesangial proliferative (non-IgA) glomerulonephritis (n = 6) | 28 (20–52) | 1 | 14.8 (10.1–18) | 45.2 (33.2–55) | 10,200 (6200–10,490) |
| control group (n = 22) | 44 (26–80) | 1 | 14.6 (12.3–16.9) | 45.7 (37.9–53) | 6270 (4280–7780) |
| c-ANCA vasculitis (n = 40) | 58 (21–81) | 0.82 | 12 (7.7–16.1) | 37.3 (27–49.7) | 6555 (3740–10,450) |
| p-ANCA vasculitis (n = 16) | 62 (37–87) | 1.27 | 9.6 (8.3–13.2) | 30.4 (28.4–42) | 4905 (3240–9620) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szymczak, M.; Heidecke, H.; Żabińska, M.; Janek, Ł.; Wronowicz, J.; Kujawa, K.; Schulze-Forster, K.; Marek-Bukowiec, K.; Gołębiowski, T.; Banasik, M. The Influence of Anti-C3aR and Anti-C5aR Antibody Levels on the Course of Specific Glomerulonephritis Types. J. Clin. Med. 2025, 14, 6082. https://doi.org/10.3390/jcm14176082
Szymczak M, Heidecke H, Żabińska M, Janek Ł, Wronowicz J, Kujawa K, Schulze-Forster K, Marek-Bukowiec K, Gołębiowski T, Banasik M. The Influence of Anti-C3aR and Anti-C5aR Antibody Levels on the Course of Specific Glomerulonephritis Types. Journal of Clinical Medicine. 2025; 14(17):6082. https://doi.org/10.3390/jcm14176082
Chicago/Turabian StyleSzymczak, Maciej, Harald Heidecke, Marcelina Żabińska, Łucja Janek, Jakub Wronowicz, Krzysztof Kujawa, Kai Schulze-Forster, Karolina Marek-Bukowiec, Tomasz Gołębiowski, and Mirosław Banasik. 2025. "The Influence of Anti-C3aR and Anti-C5aR Antibody Levels on the Course of Specific Glomerulonephritis Types" Journal of Clinical Medicine 14, no. 17: 6082. https://doi.org/10.3390/jcm14176082
APA StyleSzymczak, M., Heidecke, H., Żabińska, M., Janek, Ł., Wronowicz, J., Kujawa, K., Schulze-Forster, K., Marek-Bukowiec, K., Gołębiowski, T., & Banasik, M. (2025). The Influence of Anti-C3aR and Anti-C5aR Antibody Levels on the Course of Specific Glomerulonephritis Types. Journal of Clinical Medicine, 14(17), 6082. https://doi.org/10.3390/jcm14176082








