Pathobiology of Type 2 Inflammation in Asthma and Nasal Polyposis
Abstract
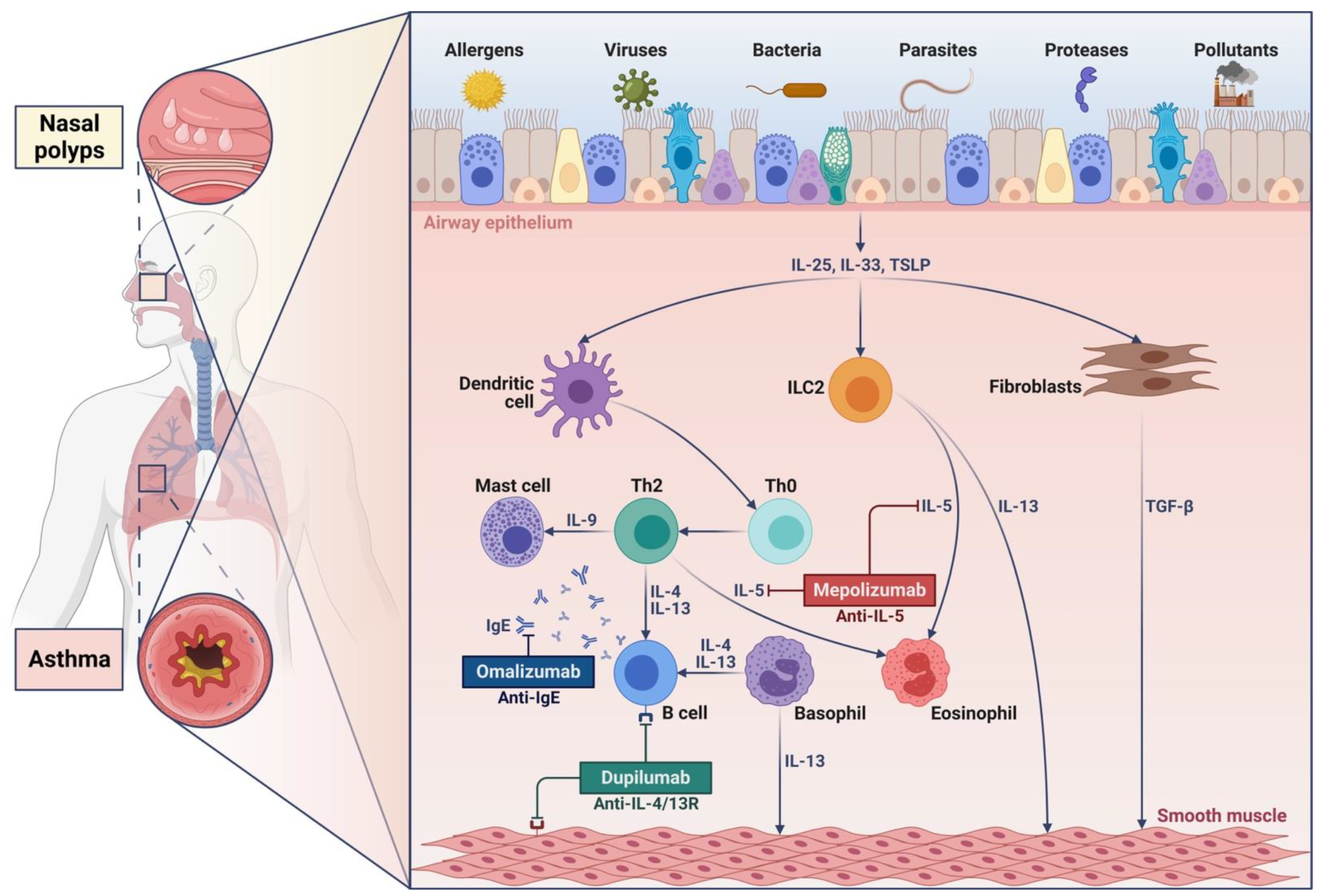
1. Introduction
2. Cellular and Molecular Mechanisms Underlying Type 2 Inflammation in Asthma and Nasal Polyposis
2.1. Interleukin-4 and Interleukin-13
2.2. Interleukin-5
3. Biological Therapies of Severe Asthma and Nasal Polyposis
3.1. Omalizumab
3.2. Mepolizumab
3.3. Reslizumab
3.4. Benralizumab
3.5. Dupilumab
4. Closing Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Laidlaw, T.M.; Mullol, J.; Woessner, K.M.; Amin, N.; Mannent, L.P. Chronic rhinosinusitis with nasal polyps and asthma. J. Allergy Clin. Immunol. Pract. 2021, 9, 1133–1141. [Google Scholar] [CrossRef]
- Matucci, A.; Bormioli, S.; Nencini, F.; Chiccoli, F.; Vivarelli, E.; Maggi, E.; Vultaggio, A. Asthma and chronic rhinosinusitis: How similar are they in pathogenesis and treatment responses? Int. J. Mol. Sci. 2021, 22, 3340. [Google Scholar] [CrossRef] [PubMed]
- Porsbjerg, C.; Melen, E.; Lehtimaki, L.; Shaw, D. Asthma. Lancet 2023, 401, 858–873. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.J.; Lund, V.J.; Hopkins, C.; Hellings, P.W.; Kern, R.; Reitsma, S.; Toppila-Salmi, S.; Bernal-Sprekelsen, M.; Mullol, J.; Alobid, I.; et al. European position paper on rhinosinusitis and nasal polyps 2020. Rhinology 2020, 58 (Suppl. S29), 1–464. [Google Scholar] [CrossRef]
- Hakansson, K.; Bachert, C.; Konge, L.; Thomsen, S.F.; Pedersen, A.E.; Poulsen, S.S.; Martin-Bertelsen, T.; Winther, O.; Backer, V.; von Buchwald, C. Airway inflammation in chronic rhinosinusitis with nasal polyps and asthma: The united airways concept further supported. PLoS ONE 2015, 10, e0127228. [Google Scholar] [CrossRef] [PubMed]
- Fokkens, W.; Reitsma, S. Unified airway disease: A contemporary review and introduction. Otolaryngol. Clin. N. Am. 2023, 56, 1–10. [Google Scholar] [CrossRef]
- Langdon, C.; Mullol, J. Nasal polyps in patients with asthma: Prevalence, impact and management challenges. J. Asthma Allergy 2016, 9, 45–53. [Google Scholar]
- Castillo, J.A.; Plaza, V.; Rodrigo, G.; Julia, B.; Mullol, J. Chronic rhinosinusitis with and without nasal polyps and rhinitis in adult asthma. Frequency distribution and relationship with asthma control and severity (the IRIS-ASMA study). Eur. Respir. J. 2013, 42 (Suppl. S57), P3448. [Google Scholar]
- Won, H.K.; Kim, Y.C.; Kang, M.G.; Park, H.K.; Lee, S.E.; Kim, M.H.; Yang, M.S.; Chang, Y.S.; Cho, S.H.; Song, W.J. Age-related prevalence of chronic rhinosinusitis and nasal polyps and their relationships with asthma onset. Ann. Allergy Asthma Immunol. 2018, 120, 389–394. [Google Scholar] [CrossRef]
- Maspero, J.; Adir, Y.; Al-Ahmad, M.; Celis-Preciado, C.A.; Colodenco, F.D.; Giavina-Bianchi, P.; Lababidi, H.; Ledanois, O.; Mahoub, B.; Perng, D.W.; et al. Type 2 inflammation in asthma and other airway diseases. ERJ Open Res. 2022, 8, 00576–02021. [Google Scholar] [CrossRef]
- Hammad, H.; Debeuf, N.; Aegerter, H.; Brown, A.S.; Lambrecht, B.N. Emerging paradigms in type 2 immunity. Annu. Rev. Immunol. 2022, 40, 443–467. [Google Scholar] [CrossRef]
- Carr, T.F.; Zeki, A.A.; Kraft, M. Eosinophilic and noneosinophilic asthma. Am. J. Respir. Crit. Care Med. 2018, 197, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Nelson, R.K.; Bush, A.; Stokes, J.; Nair, P.; Akuthota, P. Eosinophilic asthma. J. Allergy Clin. Immunol. Pract. 2020, 8, 465–473. [Google Scholar] [CrossRef]
- Kato, A.; Peters, A.T.; Stevens, W.W.; Schleimer, R.P.; Tan, B.K.; Kern, R.C. Endotypes of chronic rhinosinusitis: Relationships to disease phenotypes, pathogenesis, clinical findings, and treatment approaches. Allergy 2022, 77, 812–826. [Google Scholar] [CrossRef]
- Bachert, C.; Maurer, M.; Palomares, O.; Busse, W.W. What is the contribution of IgE to nasal polyposis? J. Allergy Clin. Immunol. 2021, 147, 1997–2008. [Google Scholar] [CrossRef]
- Ten Brinke, A.; Grootendorst, D.C.; Schmidt, J.T.; De Bruïne, F.T.; van Buchem, M.A.; Sterk, P.J.; Rabe, K.F.; Bel, E.H. Chronic sinusitis in severe asthma is related to sputum eosinophilia. J. Allergy Clin. Immunol. 2002, 109, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Flora, M.; Perna, F.; Abbadessa, S.; Garziano, F.; Maffucci, R.; Maniscalco, M.; Mollica, M.; Pelaia, C.; Tremante, E.; Maffei, M.; et al. Basophil activation test for Staphylococcus aureus enterotoxins in severe asthmatics. Clin. Exp. Allergy 2021, 51, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, C.; Seccia, V.; Pelaia, C.; Spinelli, F.; Morini, P.; Rizzi, A.; Detoraki, A. S. aureus and IgE-mediated diseases: Pilot or copilot? A narrative review. Exp. Rev. Clin. Immunol. 2022, 18, 639–647. [Google Scholar] [CrossRef]
- Mullol, J.; Picado, C. Rhinosinusitis and nasal polyps in aspirin-exacerbated respiratory disease. Immunol. Allergy Clin. N. Am. 2013, 33, 163–176. [Google Scholar] [CrossRef]
- Laidlaw, T.M.; Boyce, J.A. Aspirin-exacerbated respiratory disease—New prime suspects. N. Engl. J. Med. 2016, 374, 484–488. [Google Scholar] [CrossRef]
- Laidlaw, T.M.; Boyce, J.A. Updates on immune mechanisms in aspirin-exacerbated respiratory disease. J. Allergy Clin. Immunol. 2023, 151, 301–309. [Google Scholar] [CrossRef]
- Hizawa, N. The understanding of asthma pathogenesis in the era of precision medicine. Allergol. Int. 2023, 72, 3–10. [Google Scholar] [CrossRef]
- Hsieh, A.; Assadinia, N.; Hackett, T.L. Airway remodeling heterogeneity in asthma and its relationship to disease outcomes. Front. Physiol. 2023, 14, 1113100. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, A.G.; Peters, A.T.; Kato, A.; Stevens, W.W. Use of endotypes, phenotypes, and inflammatory markers to guide treatment decisions in chronic rhinosinusitis. Ann. Allergy Asthma Immunol. 2020, 124, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Stevens, W.W.; Kato, A. Group 2 innate lymphoid cells in nasal polyposis. Ann. Allergy Asthma Immunol. 2021, 126, 110–117. [Google Scholar] [CrossRef]
- Huise, K.E.; Stevens, W.W.; Tan, B.K.; Schleimer, R.P. Pathogenesis of nasal polyposis. Clin. Exp. Allergy 2015, 45, 328–346. [Google Scholar]
- Nagase, H.; Suzukawa, M.; Oishi, K.; Matsunaga, K. Biologics for severe asthma: The real-world evidence, effectiveness of switching, and prediction factors for the efficacy. Allergol. Int. 2023, 72, 11–23. [Google Scholar] [CrossRef]
- Shamil, E.; Hopkins, C. Unified airway disease: Medical management. Otolaryngol. Clin. N. Am. 2023, 56, 157–168. [Google Scholar] [CrossRef]
- Kim, D.H.; Lim, J.Y.; Jang, J.Y.; Gwak, J.; Joo, H.A.; Ryu, S.; Kim, J.H. Distinct subsets of innate lymphoid cells in nasal polyp. Allergol. Int. 2023, 72, 151–160. [Google Scholar] [CrossRef]
- Rodriguez-Rodriguez, N.; Gogoi, M.; McKenzie, A.N.J. Group 2 innate lymphoid cells: Team players in regulating asthma. Annu. Rev. Immunol. 2021, 39, 167–198. [Google Scholar] [CrossRef]
- Hammad, H.; Lambrecht, B.N. The basic immunology of asthma. Cell 2021, 184, 1469–1485. [Google Scholar] [CrossRef]
- Matucci, A.; Bormioli, S.; Nencini, F.; Maggi, E.; Vultaggio, A. The emerging role of type 2 inflammation in asthma. Expert Rev. Clin. Immunol. 2021, 17, 63–71. [Google Scholar] [CrossRef]
- Poto, R.; Criscuolo, G.; Marone, G.; Brightling, C.E.; Varricchi, G. Human lung mast cells: Therapeutic implications in asthma. Int. J. Mol. Sci. 2022, 23, 14466. [Google Scholar] [CrossRef] [PubMed]
- Iype, J.; Fux, M. Basophils orchestrating eosinophils’ chemotaxis and function in allergic inflammation. Cells 2021, 10, 895. [Google Scholar] [CrossRef]
- Vatrella, A.; Maglio, A.; Pelaia, C.; Ciampo, L.; Pelaia, G.; Vitale, C. Eosinophilic inflammation: An appealing target for pharmacologic treatments in severe asthma. Biomedicines 2022, 10, 2181. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Heffler, E.; Crimi, C.; Maglio, A.; Vatrella, A.; Pelaia, G.; Canonica, G.W. Interleukins 4 and 13 in asthma: Key pathophysiologic cytokines and druggable molecular targets. Front. Pharmacol. 2022, 13, 851940. [Google Scholar] [CrossRef]
- Hong, H.; Liao, S.; Chen, F.; Yang, Q.; Wang, D.Y. Role of IL-25, IL-33, and TSLP in triggering united airway diseases toward type 2 inflammation. Allergy 2020, 75, 2794–2804. [Google Scholar] [CrossRef] [PubMed]
- Calven, J.; Ax, E.; Radinger, M. The airway epithelium—A central player in asthma pathogenesis. Int. J. Mol. Sci. 2020, 21, 8907. [Google Scholar] [CrossRef]
- Lameire, S.; Hammad, H. Lung epithelial cells: Upstream targets in type 2-high asthma. Eur. J. Immunol. 2023, in press. [Google Scholar] [CrossRef]
- Komlosi, Z.; van de Veen, W.; Kovacs, N.; Szűcs, G.; Sokolowska, M.; O’Mahony, L.; Akdis, M.; Akdis, C.A. Cellular and molecular mechanisms of allergic asthma. Mol. Asp. Med. 2022, 85, 100995. [Google Scholar] [CrossRef]
- Kaiko, G.E.; Horvat, J.C.; Beagley, K.W.; Hansbro, P.M. Immunological decision making: How does the immune system decide to mount a helper T-cell response? Immunology 2008, 123, 326–338. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, N.A.; Bennett, B.L.; Graham, N.L.; Pirozzi, G.; Stahl, N.; Yancopoulos, G.D. Targeting key proximal drivers of type 2 inflammation in disease. Nat. Rev. Drug Discov. 2016, 15, 35–50. [Google Scholar] [CrossRef] [PubMed]
- Tu, L.; Chen, J.; Zhang, H.; Duan, L. Interleukin-4 inhibits regulatory T cell differentiation through regulating CD103+ dendritic cells. Front. Immunol. 2017, 8, 214. [Google Scholar] [CrossRef] [PubMed]
- Palomares, O.; Akdis, M.; Martin-Fontecha, M.; Akdis, C.A. Mechanisms of immune regulation in allergic diseases: The role of regulatory T and B cells. Immunol. Rev. 2017, 278, 219–236. [Google Scholar] [CrossRef]
- Geha, R.S.; Jabara, H.H.; Brodeur, S.R. The regulation of immunoglobulin E class-switch recombination. Nat. Rev. Immunol. 2003, 9, 721–732. [Google Scholar] [CrossRef]
- Froidure, A.; Mouthuy, J.; Durham, S.R.; Chanez, P.; Sibille, Y.; Pilette, C. Asthma phenotypes and IgE responses. Eur. Respir. J. 2016, 47, 304–319. [Google Scholar] [CrossRef] [PubMed]
- Stone, K.D.; Prussin, C.; Metcalfe, D.D. IgE, mast cells, basophils, and eosinophils. J. Allergy Clin. Immunol. 2010, 125, S73–S80. [Google Scholar] [CrossRef] [PubMed]
- Marone, G.; Granata, F.; Pucino, V.; Pecoraro, A.; Heffler, E.; Loffredo, S.; Scadding, G.W.; Varricchi, G. The intriguing role of interleukin 13 in the pathophysiology of asthma. Front. Pharmacol. 2019, 10, 1387. [Google Scholar] [CrossRef] [PubMed]
- Abdelaziz, M.H.; Abdelwahab, S.F.; Wan, J.; Cai, W.; Huixuan, W.; Jianjun, C.; Kumar, K.D.; Vasudevan, A.; Sadek, A.; Su, Z.; et al. Alternatively activated macrophages; a double-edged sword in allergic asthma. J. Transl. Med. 2020, 18, 58. [Google Scholar] [CrossRef] [PubMed]
- Becerra-Diaz, M.; Lerner, A.D.; Yu, D.H.; Thiboutot, J.P.; Liu, M.C.; Yarmus, L.B.; Bose, S.; Heller, N.M. Sex differences in M2 polarization, chemokine and IL-4 receptors in monocytes and macrophages from asthmatics. Cell. Immunol. 2021, 360, 104252. [Google Scholar] [CrossRef]
- Schleimer, R.P. Immunopathogenesis of chronic rhinosinusitis and nasal polyposis. Annu. Rev. Pathol. 2017, 12, 331–357. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, H.F.; Phipps, S.; Foster, P.S. Eosinophil trafficking in allergy and asthma. J. Allergy Clin. Immunol. 2007, 119, 1303–1310. [Google Scholar] [CrossRef]
- Nagata, M.; Nakagome, K.; Soma, T. Mechanisms of eosinophilic inflammation. Asia Pac. Allergy 2020, 10, e14. [Google Scholar] [CrossRef] [PubMed]
- Mehta, M.P.; Wise, S.K. Unified airway disease: Examining prevalence and treatment of upper airway eosinophilic disease with comorbid asthma. Otolaryngol. Clin. N. Am. 2023, 56, 65–81. [Google Scholar] [CrossRef] [PubMed]
- Sweerus, K.; Lachowicz-Scroggins, M.; Gordon, E.; LaFemina, M.; Huang, X.; Parikh, M.; Kanegai, C.; Fahy, J.V.; Frank, J.A. Claudin-18 deficiency is associated with airway epithelial barrier dysfunction and asthma. J. Allergy Clin. Immunol. 2017, 139, 72–81.e71. [Google Scholar] [CrossRef]
- Steelant, B.; Wawrzyniac, P.; Martens, K.; Jonckheere, A.C.; Pugin, B.; Schrijvers, R.; Bullens, D.M.; Vanoirbeek, J.A.; Krawczyk, K.; Dreher, A.; et al. Blocking histone deacetylase activity as a novel target for epithelial barrier defects in patients with allergic rhinitis. J. Allergy Clin. Immunol. 2019, 144, 1242–1253.e7. [Google Scholar] [CrossRef] [PubMed]
- Saatian, B.; Rezaee, F.; Desando, S.; Emo, J.; Chapman, T.; Knowlden, S.; Georas, S.N. Interleukin-4 and interleukin-13 cause barrier dysfunction in human airway epithelial cells. Tissue Barriers 2013, 1, e24333. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, J.D.; Alevy, Y.; Malvin, N.P.; Patel, K.K.; Gunsten, S.P.; Holtzman, M.J.; Stappenbeck, T.S.; Brody, S.L. IL-13 activates autophagy to regulate secretion in airway epithelial cells. Autophagy 2016, 12, 397–409. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Li, Y.Y.; Andiappan, A.K.; Yan, Y.; Tan, K.S.; Ong, H.H.; Thong, K.T.; Ong, Y.K.; Yu, F.G.; Low, H.B.; et al. Role of IL-13Rα2 in modulating IL-13-induced MUC5AC and ciliary changes in healthy and CRSwNP mucosa. Allergy 2018, 73, 1673–1685. [Google Scholar] [CrossRef]
- Dunican, E.M.; Elicker, B.M.; Gierada, D.S.; Nagle, S.K.; Schiebler, M.L.; Newell, J.D.; Raymond, W.W.; Lachowicz-Scroggins, M.E.; Di Maio, S.; Hoffman, E.A.; et al. Mucus plugs in patients with asthma linked to eosinophilia and airflow obstruction. J. Clin. Investig. 2018, 128, 997–1009. [Google Scholar] [CrossRef] [PubMed]
- Bonser, L.R.; Zlock, L.; Finkbeiner, W.; Erle, D.J. Epithelial tethering of MUC5AC-rich mucus impairs mucociliary transport in asthma. J. Clin. Investig. 2016, 126, 2367–2371. [Google Scholar] [CrossRef] [PubMed]
- Guida, G.; Carriero, V.; Bertolini, F.; Pizzimenti, S.; Heffler, E.; Paoletti, G.; Ricciardolo, F.L.M. Exhaled nitric oxide in asthma: From diagnosis to management. Curr. Opin. Allergy Clin. Immunol. 2023, 23, 29–35. [Google Scholar] [CrossRef]
- Corren, J. Role of interleukin-13 in asthma. Curr. Allergy Asthma Rep. 2013, 13, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.G.; Homer, R.J.; Zhu, Z.; Lanone, S.; Wang, X.; Koteliansky, V.; Shipley, J.M.; Gotwals, P.; Noble, P.; Chen, Q.; et al. Interleukin-13 induces tissue fibrosis by selectively stimulating and activating transforming growth factor-β1. J. Exp. Med. 2001, 194, 809–821. [Google Scholar] [CrossRef]
- Firszt, R.; Francisco, D.; Church, T.D.; Thomas, J.M.; Ingram, J.L.; Kraft, M. Interleukin-13 induces collagen type-1 expression through matrix metalloproteinase-2 and transforming growth factor-β1 in airway fibroblasts in asthma. Eur. Respir. J. 2014, 43, 464–473. [Google Scholar] [CrossRef]
- Takabayashi, T.; Kato, A.; Peters, A.T.; Hulse, K.E.; Suh, L.A.; Carter, R.; Norton, J.; Grammer, L.C.; Cho, S.H.; Tan, B.K.; et al. Excessive fibrin deposition in nasal polyps caused by fibrinolytic impairment through reduction of tissue plasminogen activator expression. Am. J. Respir. Crit. Care Med. 2013, 187, 49–57. [Google Scholar] [CrossRef]
- Lambrecht, B.N.; Hammad, H.; Fahy, J.V. The cytokines of asthma. Immunity 2019, 50, 975–991. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Paoletti, G.; Puggioni, F.; Racca, F.; Pelaia, G.; Canonica, G.W.; Heffler, E. Interleukin-5 in the pathophysiology of severe asthma. Front. Physiol. 2019, 10, 1514. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Pelaia, G.; Crimi, C.; Maglio, A.; Stanziola, A.A.; Calabrese, C.; Terracciano, R.; Longhini, F.; Vatrella, A. Novel Biological Therapies for Severe Asthma Endotypes. Biomedicines 2022, 10, 1064. [Google Scholar] [CrossRef]
- Xu, J.; Jiang, F.; Nayeri, F.; Zetterstrom, O. Apoptotic eosinophils in sputum from asthmatic patients correlate negatively with levels of IL-5 and eotaxin. Respir. Med. 2007, 101, 1447–1454. [Google Scholar] [CrossRef]
- Fulkerson, P.C.; Rothenberg, M.E. Targeting eosinophils in allergy, inflammation and beyond. Nat. Rev. Drug Discov. 2013, 12, 117–129. [Google Scholar] [CrossRef]
- Bhalla, A.; Mukherjee, M.; Nair, P. Airway eosinophilopoietic and autoimmune mechanisms of eosinophilia in severe asthma. Immunol. Allergy Clin. N. Am. 2018, 38, 639–654. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.V. Eosinophil activation status in separate compartments and association with asthma. Front. Med. 2017, 4, 75. [Google Scholar] [CrossRef] [PubMed]
- Mould, A.W.; Ramsay, A.J.; Matthaei, K.I.; Young, I.G.; Rothenberg, M.E.; Foster, P.S. The effect of IL-5 and eotaxin expression in the lung on eosinophil trafficking and degranulation and the induction of bronchial hyperreactivity. J. Immunol. 2000, 164, 2142–2150. [Google Scholar] [CrossRef]
- Vatrella, A.; Ponticiello, A.; Parrella, R.; Romano, L.; Zofra, S.; DiLeva, A.; Bariffi, F. Serum eosinophil cationic protein (ECP) as a marker of disease activity and treatment efficacy in seasonal asthma. Allergy 1996, 51, 547–555. [Google Scholar] [CrossRef]
- McBrien, C.N.; Menzies-Gow, A. The biology of eosinophils and their role in asthma. Front. Med. 2017, 4, 93. [Google Scholar] [CrossRef] [PubMed]
- Ohno, I.; Nitta, Y.; Yamauchi, K.; Hoshi, H.; Honma, M.; Woolley, K.; O’Byrne, P.; Tamura, G.; Jordana, M.; Shirato, K. Transforming growth factor β1 (TGF-β1) gene expression by eosinophils in asthmatic airway inflammation. Am. J. Respir. Cell Mol. Biol. 1996, 15, 404–409. [Google Scholar] [CrossRef]
- Choi, Y.; Kim, Y.M.; Lee, H.R.; Mun, J.; Sim, S.; Lee, D.H.; Pham, D.L.; Kim, S.H.; Shin, Y.S.; Lee, S.W.; et al. Eosinophil extracellular traps activate type 2 innate lymphoid cells through stimulating airway epithelium in severe asthma. Allergy 2020, 75, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Van Hulst, G.; Bureau, F.; Desmet, C.J. Eosinophils as drivers of severe eosinophilic asthma: Endotypes or plasticity? Int. J. Mol. Sci. 2021, 22, 10150. [Google Scholar] [CrossRef]
- Park, S.W.; Kim, D.; Chang, H.S.; Park, S.J.; Lee, Y.M.; Park, J.S.; Chung, I.Y.; Lee, J.H.; Park, C.S. Association of interleukin-5 and eotaxin with acute exacerbation of asthma. Int. Arch. Allergy Immunol. 2003, 131, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Ilmarinen, P.; Moilanen, E.; Kankaanranta, H. Regulation of spontaneous eosinophil apoptosis—A neglected area of importance. J. Cell Death 2014, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Bachert, C.; Wagenmann, M.; Hauser, U.; Rudack, C. IL-5 synthesis is upregulated in human nasal polyp tissue. J. Allergy Clin. Immunol. 1997, 99, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Tomassen, P.; Vandeplas, G.; Van Zele, T.; Cardell, L.O.; Arebro, J.; Olze, H.; Förster-Ruhrmann, U.; Kowalski, M.L.; Olszewska-Ziąber, A.; Holtappels, G.; et al. Inflammatory endotypes of chronic rhinosinusitis based on cluster analysis of biomarkers. J. Allergy Clin. Immunol. 2016, 137, 1449–1456. [Google Scholar] [CrossRef]
- Simon, H.-U.; Yousefi, S.; Schranz, C.; Schapowal, A.; Bachert, C.; Blaser, K. Direct demonstration of delayed eosinophil apoptosis as a mechanism causing tissue eosinophilia. J. Immunol. 1997, 158, 3902–3908. [Google Scholar] [CrossRef]
- Gitomer, S.A.; Fountain, C.R.; Kingdom, T.T.; Getz, A.E.; Sillau, S.H.; Katial, R.K.; Ramakrishnan, V.R. Clinical examination of tissue eosinophilia in patients with chronic rhinosinusitis and nasal polyposis. Otolaryngol. Head Neck Surg. 2016, 155, 173–178. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Deng, J.; Yang, M.; Chen, Y.; Chen, F.; Gao, W.X.; Lai, Y.; Shi, J.; Sun, Y. Concordant systemic and local eosinophilia relates to poorer disease control in patients with nasal polyps. World Allergy Organ. J. 2019, 12, 100052. [Google Scholar] [CrossRef] [PubMed]
- Gevaert, P.; Han, J.K.; Smith, S.G.; Sousa, A.R.; Howarth, P.H.; Yancey, S.W.; Chan, R.; Bachert, C. The roles of eosinophils and interleukin-5 in the pathophysiology of chronic rhinosinusitis with nasal polyps. Int. Forum Allergy 2022, 12, 1413–1423. [Google Scholar] [CrossRef]
- Buchheit, K.M.; Lewis, E.; Gakpo, D.; Hacker, J.; Sohail, A.; Taliaferro, F.; Berreondo Giron, E.; Asare, C.; Vukovic, M.; Bensko, J.C.; et al. Mepolizumab targets multiple immune cells in aspirin-exacerbated respiratory disease. J. Allergy Clin. Immunol. 2021, 148, 574–584. [Google Scholar] [CrossRef]
- Bakakos, A.; Schleich, F.; Bakakos, P. Biological therapy of severe asthma and nasal polyps. J. Pers. Med. 2022, 12, 976. [Google Scholar] [CrossRef]
- Pelaia, C.; Calabrese, C.; Terracciano, R.; de Blasio, F.; Vatrella, A.; Pelaia, G. Omalizumab, the first available antibody for biological treatment of asthma: More than a decade of real life effectiveness. Ther. Adv. Respir. Dis. 2018, 12, 1753466618810192. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, G.; Canonica, G.W.; Matucci, A.; Paolini, R.; Triggiani, M.; Paggiaro, P. Targeted therapy in severe asthma today: Focus on immunoglobulin E. Drug Des. Dev. Ther. 2017, 11, 1979–1987. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, G.; Vatrella, A.; Busceti, M.T.; Gallelli, L.; Terracciano, R.; Maselli, R. Anti-IgE therapy with omalizumab for severe asthma: Current concepts and potential developments. Curr. Drug Targets 2015, 16, 171–178. [Google Scholar] [CrossRef]
- Rodrigo, G.J.; Neffen, H.; Castro-Rodriguez, J.A. Efficacy and safety of subcutaneous omalizumab vs placebo as add-on therapy to corticosteroids for children and adults with asthma: A systematic review. Chest 2011, 139, 28–35. [Google Scholar] [CrossRef]
- Cabrejos, S.; Moreira, A.; Ramirez, A.; Quirce, S.; Soto Campos, G.; Dávila, I.; Campo, P. FENOMA study: Achieving full control in patients with severe allergic asthma. J. Asthma Allergy 2020, 13, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Calabrese, C.; Barbuto, S.; Busceti, M.T.; Preianò, M.; Gallelli, L.; Savino, R.; Vatrella, A.; Pelaia, G. Omalizumab lowers asthma exacerbations, oral corticosteroid intake and blood eosinophils: Results of a 5-year single centre observational study. Pulm. Pharmacol. Ther. 2019, 54, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Pace, E.; Ferraro, M.; Bruno, A.; Chiappara, G.; Bousquet, J.; Gjomarkaj, M. Clinical benefits of 7 years of treatment with omalizumab in severe uncontrolled asthmatics. J. Asthma 2011, 48, 387–392. [Google Scholar] [CrossRef]
- Menzella, F.; Galeone, C.; Formisano, D.; Castagnetti, C.; Ruggiero, P.; Simonazzi, A.; Zucchi, L. Real life efficacy of omalizumab after 9 years of follow-up. Allergy Asthma Immunol. Res. 2017, 9, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Di Bona, D.; Fiorino, I.; Taurino, M.; Frisenda, F.; Minenna, E.; Pasculli, C.; Kourtis, G.; Rucco, A.S.; Nico, A.; Albanesi, M.; et al. Long-term “real-life” safety of omalizumab in patients with severe uncontrolled asthma: A nine-year study. Respir. Med. 2017, 130, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Humbert, M.; Beasley, R.; Ayres, J.; Slavin, R.; Hébert, J.; Bousquet, J.; Beeh, K.M.; Ramos, S.; Canonica, G.W.; Hedgecock, S.; et al. Benefits of omalizumab as add-on therapy in patients with severe persistent asthma who are inadequately controlled despite best available therapy (GINA 2002 step 4 treatment): INNOVATE. Allergy 2005, 60, 309–316. [Google Scholar] [CrossRef]
- Kariyawasam, H.H.; James, L.K. Chronic rhinosinusitis with nasal polyps: Targeting IgE with anti-IgE omalizumab therapy. Drug Des. Devel. Ther. 2020, 14, 5483–5494. [Google Scholar] [CrossRef] [PubMed]
- Gevaert, P.; Omachi, T.A.; Corren, J.; Mullol, J.; Han, J.; Lee, S.E.; Kaufman, D.; Ligueros-Saylan, M.; Howard, M.; Zhu, R.; et al. Efficacy and safety of omalizumab in nasal polyposis: 2 randomized phase 3 trials. J. Allergy Clin. Immunol. 2020, 146, 595–605. [Google Scholar] [CrossRef] [PubMed]
- Gevaert, P.; Saenz, R.; Corren, J.; Han, J.K.; Mullol, J.; Lee, S.E.; Ow, R.A.; Zhao, R.; Howard, M.; Wong, K.; et al. Long-term efficacy and safety of omalizumab for nasal polyposis in an open-label extension study. J. Allergy Clin. Immunol. 2022, 149, 957–965.e3. [Google Scholar] [CrossRef]
- Tiotiu, A.; Oster, J.P.; Roux, P.R.; Nguyen Thi, P.L.; Peiffer, G.; Bonniaud, P.; Dalphin, J.C.; de Blay, F. Effectiveness of omalizumab in severe allergic asthma and nasal polyposis: A real-life study. J. Investig. Allergol. Clin. Immunol. 2020, 30, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Vatrella, A.; Busceti, M.T.; Gallelli, L.; Terracciano, R.; Savino, R.; Pelaia, G. Severe eosinophilic asthma: From the pathogenic role of interleukin-5 to the therapeutic action of mepolizumab. Drug Design Dev. Ther. 2017, 11, 3137–3144. [Google Scholar] [CrossRef]
- Pavord, I.D.; Bel, E.H.; Bourdin, A.; Chan, R.; Han, J.K.; Keene, O.N.; Liu, M.C.; Martin, N.; Papi, A.; Roufosse, F.; et al. From DREAM to REALITY-A and beyond: Mepolizumab for treatment of eosinophil-driven diseases. Allergy 2022, 77, 778–797. [Google Scholar] [CrossRef] [PubMed]
- Pavord, I.D.; Korn, S.; Howarth, P.; Bleecker, E.R.; Buhl, R.; Keene, O.N.; Ortega, H.; Chanez, P. Mepolizumab for severe eosinophilic asthma (DREAM): A multicentre, double-blind, placebo-controlled trial. Lancet 2012, 380, 651–659. [Google Scholar] [CrossRef] [PubMed]
- Ortega, H.G.; Liu, M.C.; Pavord, I.D.; Brusselle, G.G.; FitzGerald, J.M.; Chetta, A.; Humbert, M.; Katz, L.E.; Keene, O.N.; Yancey, S.W.; et al. Mepolizumab treatment in patients with severe eosinophilic asthma. N. Engl. J. Med. 2014, 371, 1198–1207. [Google Scholar] [CrossRef]
- Bel, E.H.; Wenzel, S.E.; Thompson, P.J.; Prazma, C.M.; Keene, O.N.; Yancey, S.W.; Ortega, H.G.; Pavord, I.D.; SIRIUS Investigators. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N. Engl. J. Med. 2014, 371, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Chupp, G.L.; Bradford, E.S.; Albers, F.C.; Bratton, D.J.; Wang-Jairaj, J.; Nelsen, L.M.; Trevor, J.L.; Magnan, A.; Ten Brinke, A. Efficacy of mepolizumab add-on therapy on health-related quality of life and markers of asthma control in severe eosinophilic asthma (MUSCA): A randomized, double-blind, placebo-controlled, parallel-group, multicenter, phase 3b trial. Lancet Respir. Med. 2017, 5, 390–400. [Google Scholar] [CrossRef] [PubMed]
- Lugogo, N.; Domingo, C.; Chanez, P.; Leigh, R.; Gilson, M.J.; Price, R.G.; Yancey, S.W.; Ortega, H.G. Long-term efficacy and safety of mepolizumab in patients with severe eosinophilic asthma: A multi-center, open-label, phase IIIb study. Clin. Ther. 2016, 38, 2058–2070. [Google Scholar] [CrossRef] [PubMed]
- Khatri, S.; Moore, W.; Gibson, P.G.; Leigh, R.; Bourdin, A.; Maspero, J.; Barros, M.; Buhl, R.; Howarth, P.; Albers, F.C.; et al. Assessment of the long-term safety of mepolizumab and durability of clinical response in patients with severe eosinophilic asthma. J. Allergy Clin. Immunol. 2019, 143, 1742–1751.e7. [Google Scholar] [CrossRef]
- Harrison, T.; Canonica, G.W.; Chupp, G.; Lee, J.; Schleich, F.; Welte, T.; Valero, A.; Gemzoe, K.; Maxwell, A.; Joksaite, S.; et al. Real-world mepolizumab in the prospective severe asthma REALITI-A study: Initial analysis. Eur. Respir. J. 2020, 56, 2000151. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Busceti, M.T.; Solinas, S.; Terracciano, R.; Pelaia, G. Real-life evaluation of the clinical, functional, and hematological effects of mepolizumab in patients with severe eosinophilic asthma: Results of a single-centre observational study. Pulm. Pharmacol. Ther. 2018, 53, 1–5. [Google Scholar] [CrossRef]
- Bagnasco, D.; Milanese, M.; Rolla, G.; Lombardi, C.; Bucca, C.; Heffler, E.; Canonica, G.W.; Passalacqua, G. The North-Western Italian experience with anti IL-5 therapy and comparison with regulatory trials. World Allergy Organ. J. 2018, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Crimi, C.; Pelaia, G.; Nolasco, S.; Campisi, R.; Heffler, E.; Valenti, G.; Crimi, N. Real-life evaluation of mepolizumab efficacy in patients with severe eosinophilic asthma, according to atopic trait and allergic phenotype. Clin. Exp. Allergy 2020, 50, 780–788. [Google Scholar] [CrossRef] [PubMed]
- Sposato, B.; Camiciottoli, G.; Bacci, E.; Scalese, M.; Carpagnano, G.E.; Pelaia, C.; Santus, P.; Maniscalco, M.; Masieri, S.; Corsico, A.; et al. Mepolizumab effectiveness on small airway obstruction, corticosteroid sparing and maintenance therapy step-down in real life. Pulm. Pharmacol. Ther. 2020, 61, 101899. [Google Scholar] [CrossRef]
- Maglio, A.; Vitale, C.; Pellegrino, S.; Calabrese, C.; D’Amato, M.; Molino, A.; Pelaia, C.; Triggiani, M.; Pelaia, G.; Stellato, C.; et al. Real-life effectiveness of mepolizumab on forced expiratory flow between 25% and 75% of forced vital capacity in patients with severe eosinophilic asthma. Biomedicines 2021, 9, 1550. [Google Scholar] [CrossRef] [PubMed]
- Maglio, A.; Vitale, C.; Pelaia, C.; D’Amato, M.; Ciampo, L.; Sferra, E.; Molino, A.; Pelaia, G.; Vatrella, A. Severe asthma remissions induced by biologics targeting IL-5/IL-5R: Results from a multicenter real-life study. Int. J. Mol. Sci. 2023, 24, 2455. [Google Scholar] [CrossRef]
- Thomas, D.; McDonald, V.M.; Pavord, I.D.; Gibson, P.G. Asthma remission: What is it and how can it be achieved? Eur. Respir. J. 2022, 60, 2102583. [Google Scholar] [CrossRef] [PubMed]
- Han, J.K.; Bachert, C.; Fokkens, W.; Desrosiers, M.; Wagenmann, M.; Lee, S.E.; Smith, S.G.; Martin, N.; Mayer, B.; Yancey, S.; et al. Mepolizumab for chronic rhinosinusitis with nasal polyps (SYNAPSE): A randomized, double-blind, placebo-controlled, phase 3 trial. Lancet Respir. Med. 2021, 9, 1141–1153. [Google Scholar] [CrossRef]
- Bachert, C.; Sousa, A.R.; Lund, V.J.; Scadding, G.K.; Gevaert, P.; Nasser, S.; Durham, S.R.; Cornet, M.E.; Kariyawasam, H.H.; Gilbert, J.; et al. Reduced need for surgery in severe nasal polyposis with mepolizumab: Randomized trial. J. Allergy Clin. Immunol. 2017, 140, 1024–1031.e14. [Google Scholar] [CrossRef] [PubMed]
- Detoraki, A.; Tremante, E.; D’Amato, M.; Calabrese, C.; Casella, C.; Maniscalco, M.; Poto, R.; Brancaccio, R.; Boccia, M.; Martino, M.; et al. Mepolizumab improves sino-nasal symptoms and asthma control in severe eosinophilic asthma patients with chronic rhinosinusitis and nasal polyps: A 12-month real-life study. Ther. Adv. Respir. Dis. 2021, 15, 17534666211009398. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, G.; Vatrella, A.; Busceti, M.T.; Gallelli, L.; Preianò, M.; Lombardo, N.; Terracciano, R.; Maselli, R. Role of biologics in severe eosinophilic asthma: Focus on reslizumab. Ther. Clin. Risk Manag. 2016, 12, 1075–1082. [Google Scholar] [CrossRef]
- Varricchi, G.; Senna, G.; Loffredo, S.; Bagnasco, D.; Ferrando, M.; Canonica, G.W. Reslizumab and eosinophilic asthma: One step closer to precision medicine? Front. Immunol. 2017, 8, 242. [Google Scholar] [CrossRef]
- Kips, J.C.; O’Connor, B.J.; Langley, S.J.; Woodcock, A.; Kerstjens, H.A.; Postma, D.S.; Danzig, M.; Cuss, F.; Pauwels, R.A. Effects of SCH55700, a humanized anti-human interleukin-5 antibody, in severe persistent asthma: A pilot study. Am. J. Respir. Crit. Care Med. 2003, 167, 1655–1659. [Google Scholar] [CrossRef]
- Castro, M.; Mathur, S.; Hargreave, F.; Boulet, L.P.; Xie, F.; Young, J.; Wilkins, H.J.; Henkel, T.; Nair, P.; Res-5-0010 Study Group. Reslizumab for poorly controlled, eosinophilic asthma: A randomized, placebo-controlled study. Am. J. Respir. Crit. Care Med. 2011, 184, 1125–1132. [Google Scholar] [CrossRef]
- Castro, M.; Zangrilli, J.; Wechsler, M.E.; Bateman, E.D.; Brusselle, G.G.; Bardin, P.; Murphy, K.; Maspero, J.F.; O’Brien, C.; Korn, S. Reslizumab for inadequately controlled asthma with elevated blood eosinophil counts: Results from two multicentre, parallel, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet Respir. Med. 2015, 3, 355–366. [Google Scholar] [CrossRef] [PubMed]
- Bjermer, L.; Lemiere, C.; Maspero, J.; Weiss, S.; Zangrilli, J.; Germinaro, M. Reslizumab for inadequately controlled asthma with elevated blood eosinophil levels: A randomized phase 3 study. Chest 2016, 150, 789–798. [Google Scholar] [CrossRef]
- Brusselle, G.; Germinaro, M.; Weiss, S.; Zangrilli, J. Reslizumab in patients with inadequately controlled late-onset asthma and elevated blood eosinophils. Pulm. Pharmacol. Ther. 2017, 43, 39–45. [Google Scholar] [CrossRef]
- Wechsler, M.E.; Peters, S.P.; Hill, T.D.; Ariely, R.; DePietro, M.R.; Driessen, M.T.; Terasawa, E.L.; Thomason, D.R.; Panettieri, R.A., Jr. Clinical outcomes and health-care resource use associated with reslizumab treatment in adults with severe eosinophilic asthma in real-world practice. Chest 2021, 159, 1734–1746. [Google Scholar] [CrossRef]
- Pelaia, C.; Calabrese, C.; Vatrella, A.; Busceti, M.T.; Garofalo, E.; Lombardo, N.; Terracciano, R.; Pelaia, G. Benralizumab: From the basic mechanism of action to the potential use in the biological therapy of severe eosinophilic asthma. BioMed Res. Int. 2018, 2018, 4839230. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Vatrella, A.; Bruni, A.; Terracciano, R.; Pelaia, G. Benralizumab in the treatment of severe asthma: Design, development and potential place in therapy. Drug Des. Dev. Ther. 2018, 12, 619–628. [Google Scholar] [CrossRef]
- Bleecker, E.R.; FitzGerald, J.M.; Chanez, P.; Papi, A.; Weinstein, S.F.; Barker, P.; Sproule, S.; Gilmartin, G.; Aurivillius, M.; Werkström, V.; et al. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting β2-agonists (SIROCCO): A randomized, multicentre, placebo-controlled phase 3 trial. Lancet 2016, 388, 2115–2127. [Google Scholar] [CrossRef]
- FitzGerald, J.M.; Bleecker, E.R.; Nair, P.; Korn, S.; Ohta, K.; Lommatzsch, M.; Ferguson, G.T.; Busse, W.W.; Barker, P.; Sproule, S.; et al. Benralizumab, an anti-interleukin-5 receptor α monoclonal antibody, as add-on treatment for patients with severe, uncontrolled eosinophilic asthma (CALIMA): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2016, 388, 2128–2141. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, G.T.; FitzGerald, J.M.; Bleecker, E.R.; Laviolette, M.; Bernstein, D.; LaForce, C.; Mansfield, L.; Barker, P.; Wu, Y.; Jison, M.; et al. Benralizumab for patients with mild to moderate persistent asthma (BISE): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet Respir. Med. 2017, 5, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Nair, P.; Wenzel, S.; Rabe, K.F.; Bourdin, A.; Lugogo, N.L.; Kuna, P.; Barker, P.; Sproule, S.; Ponnarambil, S.; Goldman, M.; et al. Oral glucocorticoid-sparing effect of benralizumab in severe asthma. N. Engl. J. Med. 2017, 376, 2448–2458. [Google Scholar] [CrossRef]
- Menzies-Gow, A.; Gurnell, M.; Heaney, L.G.; Corren, J.; Bel, E.H.; Maspero, J.; Harrison, T.; Jackson, D.J.; Price, D.; Lugogo, N.; et al. Oral corticosteroid elimination via a personalised reduction algorithm in adults with severe, eosinophilic asthma treated with benralizumab (PONENTE): A multicentre, open-label, single-arm study. Lancet Respir. Med. 2022, 10, 47–58. [Google Scholar] [CrossRef] [PubMed]
- Busse, W.W.; Bleecker, E.R.; FitzGerald, J.M.; Ferguson, G.T.; Barker, P.; Sproule, S.; Olsson, R.F.; Martin, U.J.; Goldman, M.; BORA study investigators. Long-term safety and efficacy of benralizumab in patients with severe, uncontrolled asthma: 1-year results from the BORA phase 3 extension trial. Lancet Respir. Med. 2019, 7, 46–59. [Google Scholar] [CrossRef]
- Pelaia, C.; Crimi, C.; Benfante, A.; Caiaffa, M.F.; Calabrese, C.; Carpagnano, G.E.; Ciotta, D.; D’Amato, M.; Macchia, L.; Nolasco, S.; et al. Therapeutic effects of benralizumab assessed in patients with severe eosinophilic asthma: Real-life evaluation correlated with allergic and non-allergic phenotype expression. J. Asthma Allergy 2021, 14, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Menzella, F.; Bargagli, E.; Aliani, M.; Bracciale, P.; Brussino, L.; Caiaffa, M.F.; Caruso, C.; Centanni, S.; D’Amato, M.; Del Giacco, S.; et al. ChAracterization of ItaliaN severe uncontrolled Asthmatic patients Key features when receiving Benralizumab in a real-life setting: The observational rEtrospective ANANKE study. Respir. Res. 2022, 23, 36. [Google Scholar] [CrossRef]
- Pelaia, C.; Crimi, C.; Nolasco, S.; Carpagnano, G.E.; Brancaccio, R.; Buonamico, E.; Campisi, R.; Gagliani, C.; Patella, V.; Pelaia, G.; et al. Switch from omalizumab to benralizumab in allergic patients with severe eosinophilic asthma: A real-life experience from Southern Italy. Biomedicines 2021, 9, 1822. [Google Scholar] [CrossRef]
- Pelaia, C.; Busceti, M.T.; Vatrella, A.; Rago, G.F.; Crimi, C.; Terracciano, R.; Pelaia, G. Real-life rapidity of benralizumab effects in patients with severe allergic asthma: Assessment of blood eosinophils, symptom control, lung function and oral corticosteroid intake after the first drug dose. Pulm. Pharmacol. Ther. 2019, 58, 101830. [Google Scholar] [CrossRef] [PubMed]
- Pelaia, C.; Busceti, M.T.; Crimi, C.; Carpagnano, G.E.; Lombardo, N.; Terracciano, R.; Vatrella, A.; Pelaia, G. Real-life effects of benralizumab on exacerbation number and lung hyperinflation in atopic patients with severe eosinophilic asthma. BioMed Pharmacother. 2020, 129, 110444. [Google Scholar] [CrossRef] [PubMed]
- Kavanagh, J.E.; Hearn, A.P.; Dhariwal, J.; d’Ancona, G.; Douiri, A.; Roxas, C.; Fernandes, M.; Green, L.; Thomson, L.; Nanzer, A.M.; et al. Real-world effectiveness of benralizumab in severe eosinophilic asthma. Chest 2021, 159, 496–506. [Google Scholar] [CrossRef]
- Di Bona, D.; Crimi, C.; D’Uggento, A.M.; Benfante, A.; Caiaffa, M.F.; Calabrese, C.; Campisi, R.; Carpagnano, G.E.; Ciotta, D.; D’Amato, M.; et al. Effectiveness of benralizumab in severe eosinophilic asthma: Distinct sub-phenotypes of response identified by cluster analysis. Clin. Exp. Allergy 2022, 52, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Harrison, T.W.; Chanez, P.; Menzella, F.; Canonica, G.W.; Louis, R.; Cosio, B.G.; Lugogo, N.L.; Mohan, A.; Burden, A.; McDermott, L.; et al. Onset of effect and impact on health-related quality of life, exacerbation rate, lung function, and nasal polyposis symptoms for patients with severe eosinophilic asthma treated with benralizumab (ANDHI): A randomised, controlled, phase 3b trial. Lancet Respir. Med. 2021, 9, 260–274. [Google Scholar] [CrossRef]
- Bagnasco, D.; Brussino, L.; Bonavia, M.; Calzolari, E.; Caminati, M.; Caruso, C.; D’Amato, M.; De Ferrari, L.; Di Marco, F.; Imeri, G.; et al. Efficacy of benralizumab in severe asthma in real life and focus on nasal polyposis. Respir. Med. 2020, 171, 106080. [Google Scholar] [CrossRef]
- Nolasco, S.; Crimi, C.; Pelaia, C.; Benfante, A.; Caiaffa, M.F.; Calabrese, C.; Carpagnano, G.E.; Ciotta, D.; D’Amato, M.; Macchia, L.; et al. Benralizumab effectiveness in severe eosinophilic asthma with and without chronic rhinosinusitis with nasal polyps: A real-world multicenter study. J. Allergy Clin. Immunol. Pract. 2021, 9, 4371–4380.e4. [Google Scholar] [CrossRef]
- Lombardo, N.; Pelaia, C.; Ciriolo, M.; Della Corte, M.; Piazzetta, G.; Lobello, N.; Viola, P.; Pelaia, G. Real-life effects of benralizumab on allergic chronic rhinosinusitis and nasal polyposis associated with severe asthma. Int. J. Immunopathol. Pharmacol. 2020, 34, 2058738420950851. [Google Scholar] [CrossRef]
- Harb, H.; Chatila, T. Mechanisms of dupilumab. Clin. Exp. Allergy 2020, 50, 5–14. [Google Scholar] [CrossRef]
- Pelaia, C.; Vatrella, A.; Gallelli, L.; Terracciano, R.; Navalesi, P.; Maselli, R.; Pelaia, G. Dupilumab for the treatment of asthma. Expert Opin. Biol. Ther. 2017, 17, 1565–1572. [Google Scholar] [CrossRef]
- Castro, M.; Corren, J.; Pavord, I.D.; Maspero, J.; Wenzel, S.; Rabe, K.F.; Busse, W.W.; Ford, L.; Sher, L.; FitzGerald, J.M.; et al. Dupilumab efficacy and safety in moderate-to-severe uncontrolled asthma. N. Engl. J. Med. 2018, 378, 2486–2496. [Google Scholar] [CrossRef]
- Rabe, K.F.; Nair, P.; Brusselle, G.; Maspero, J.F.; Castro, M.; Sher, L.; Zhu, H.; Hamilton, J.D.; Swanson, B.N.; Khan, A.; et al. Efficacy and safety of dupilumab in glucocorticoid-dependent severe asthma. N. Engl. J. Med. 2018, 378, 2475–2485. [Google Scholar] [CrossRef]
- Wechsler, M.E.; Ford, L.B.; Maspero, J.F.; Pavord, I.D.; Papi, A.; Bourdin, A.; Watz, H.; Castro, M.; Nenasheva, N.M.; Tohda, Y.; et al. Long-term safety and efficacy of dupilumab in patients with moderate-to-severe asthma (TRAVERSE): An open-label extension study. Lancet Respir. Med. 2022, 10, 11–25. [Google Scholar] [CrossRef]
- Bacharier, L.B.; Maspero, J.F.; Katelaris, C.H.; Fiocchi, A.G.; Gagnon, R.; de Mir, I.; Jain, N.; Sher, L.D.; Mao, X.; Liu, D.; et al. Dupilumab in children with uncontrolled moderate-to-severe asthma. N. Engl. J. Med. 2021, 385, 2230–2240. [Google Scholar] [CrossRef]
- Dupin, C.; Belhadi, D.; Guilleminault, L.; Gamez, A.S.; Berger, P.; De Blay, F.; Bonniaud, P.; Leroyer, C.; Mahay, G.; Girodet, P.O.; et al. Effectiveness and safety of dupilumab for the treatment of severe asthma in a real-life French multi-centre adult cohort. Clin. Exp. Allergy 2020, 50, 789–798. [Google Scholar] [CrossRef] [PubMed]
- Campisi, R.; Crimi, C.; Nolasco, S.; Beghè, B.; Antonicelli, L.; Guarnieri, G.; Scichilone, N.; Porto, M.; Macchia, L.; Scioscia, G.; et al. Real-world experience with dupilumab in severe asthma: One-year data from an Italian Named Patient Program. J. Asthma Allergy 2021, 14, 575–583. [Google Scholar] [CrossRef]
- Pelaia, C.; Lombardo, N.; Busceti, M.T.; Piazzetta, G.; Crimi, C.; Calabrese, C.; Vatrella, A.; Pelaia, G. Short-term evaluation of dupilumab effects in patients with severe asthma and nasal polyposis. J. Asthma Allergy 2021, 14, 1165–1172. [Google Scholar] [CrossRef]
- Pelaia, C.; Benfante, A.; Busceti, M.T.; Caiaffa, M.F.; Campisi, R.; Carpagnano, G.E.; Crimi, N.; D’Amato, M.; Foschino Barbaro, M.P.; Maglio, A.; et al. Real-life effects of dupilumab in patients with severe type 2 asthma, according to atopic trait and presence of chronic rhinosinusitis with nasal polyps. Front Immunol. 2023, 14, 1121237. [Google Scholar] [CrossRef]
- Bachert, C.; Mannent, L.; Naclerio, R.M.; Mullol, J.; Ferguson, B.J.; Gevaert, P.; Hellings, P.; Jiao, L.; Wang, L.; Evans, R.R.; et al. Effect of subcutaneous dupilumab on nasal polyp burden in patients with chronic sinusitis and nasal polyposis: A randomized clinical trial. JAMA 2016, 315, 469–479. [Google Scholar] [CrossRef]
- Bachert, C.; Han, J.K.; Desrosiers, M.; Hellings, P.W.; Amin, N.; Lee, S.E.; Mullol, J.; Greos, L.S.; Bosso, J.V.; Laidlaw, T.M.; et al. Efficacy and safety of dupilumab in patients with severe chronic rhinosinusitis with nasal polyps (LIBERTY NP SINUS-24 and LIBERTY NP SINUS-52): Results from two multicentre, randomised, double-blind, placebo-controlled, parallel-group phase 3 trials. Lancet 2019, 394, 1638–1650. [Google Scholar] [CrossRef] [PubMed]
- Desrosiers, M.; Mannent, L.P.; Amin, N.; Canonica, G.W.; Hellings, P.W.; Gevaert, P.; Mullol, J.; Lee, S.E.; Fujieda, S.; Han, J.K.; et al. Dupilumab reduces systemic corticosteroid use and sinonasal surgery rate in CRSwNP. Rhinology 2021, 59, 301–311. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelaia, C.; Pelaia, G.; Maglio, A.; Tinello, C.; Gallelli, L.; Lombardo, N.; Terracciano, R.; Vatrella, A. Pathobiology of Type 2 Inflammation in Asthma and Nasal Polyposis. J. Clin. Med. 2023, 12, 3371. https://doi.org/10.3390/jcm12103371
Pelaia C, Pelaia G, Maglio A, Tinello C, Gallelli L, Lombardo N, Terracciano R, Vatrella A. Pathobiology of Type 2 Inflammation in Asthma and Nasal Polyposis. Journal of Clinical Medicine. 2023; 12(10):3371. https://doi.org/10.3390/jcm12103371
Chicago/Turabian StylePelaia, Corrado, Giulia Pelaia, Angelantonio Maglio, Caterina Tinello, Luca Gallelli, Nicola Lombardo, Rosa Terracciano, and Alessandro Vatrella. 2023. "Pathobiology of Type 2 Inflammation in Asthma and Nasal Polyposis" Journal of Clinical Medicine 12, no. 10: 3371. https://doi.org/10.3390/jcm12103371
APA StylePelaia, C., Pelaia, G., Maglio, A., Tinello, C., Gallelli, L., Lombardo, N., Terracciano, R., & Vatrella, A. (2023). Pathobiology of Type 2 Inflammation in Asthma and Nasal Polyposis. Journal of Clinical Medicine, 12(10), 3371. https://doi.org/10.3390/jcm12103371









