Anti-Hypertensive Activity of Some Selected Unani Formulations: An Evidence-Based Approach for Verification of Traditional Unani Claims Using LC-MS/MS for the Evaluation of Clinically Relevant Blood Parameters in Laboratory Rats
Abstract
:1. Introduction
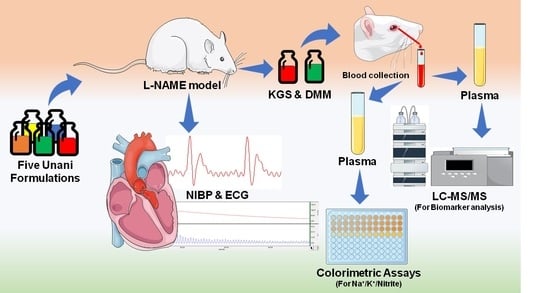
2. Materials and Methods
2.1. Chemicals
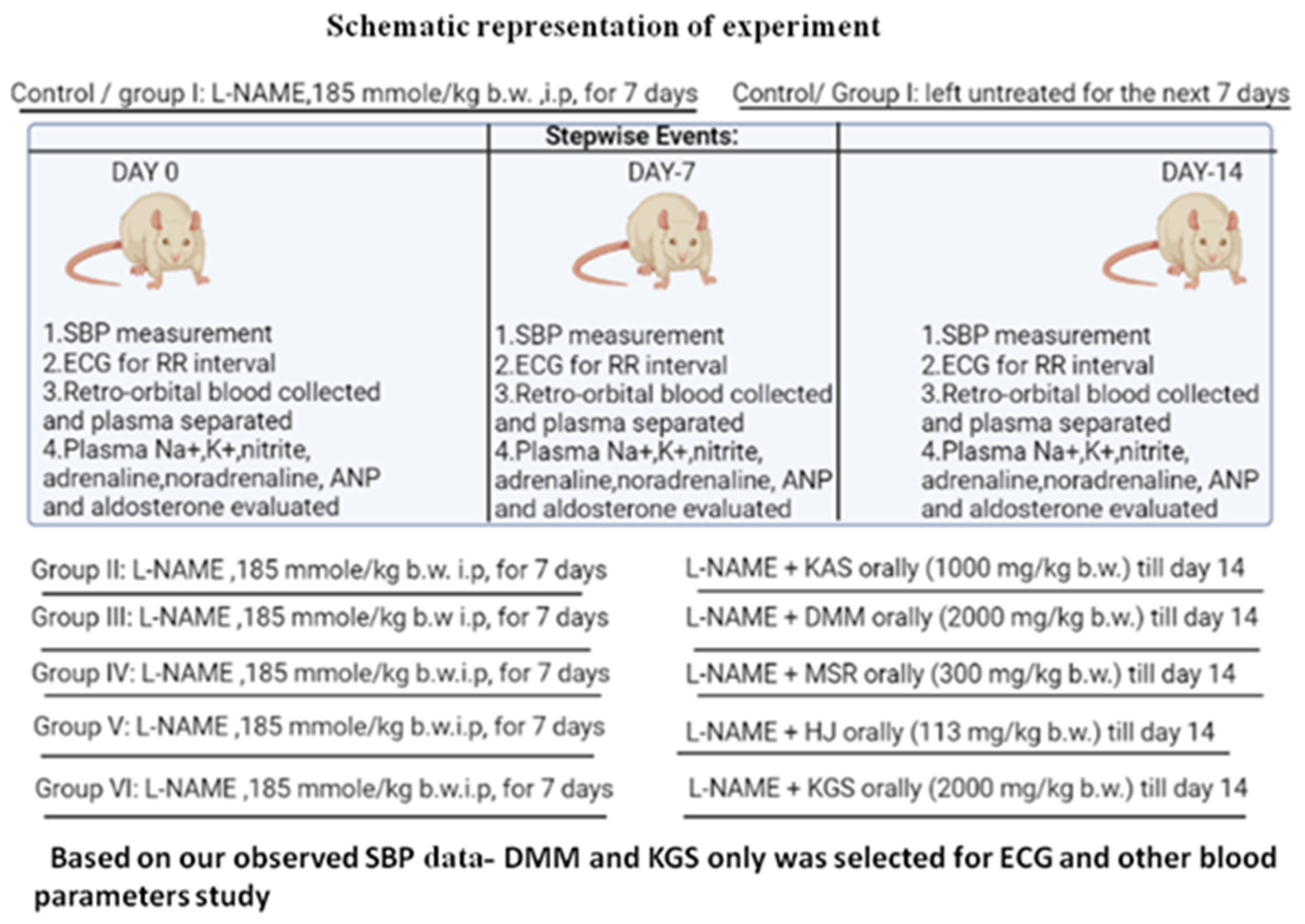
2.2. Animals and Experimental Protocol
2.3. Development of Hypertensive Model in Rats
2.4. Non-Invasive Blood-Pressure Measurement
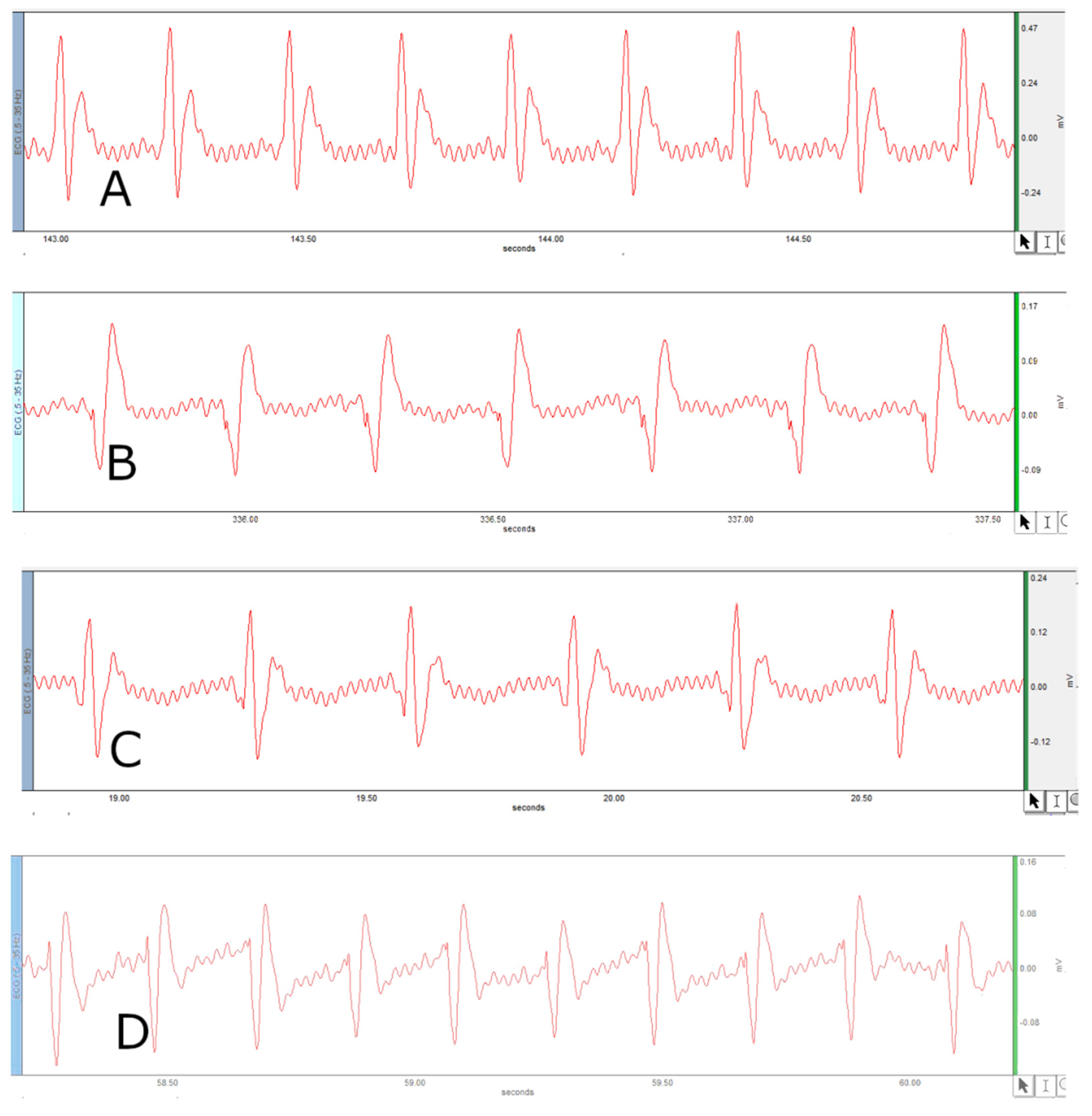
2.5. Surface Electrocardiogram (ECG) Measurement in Anesthetized Rats
2.6. Allocation of Groups: Animals Were Divided into Six Groups (n = 6)
2.7. Preparation of 0.2M Phosphate Buffer Solution pH 7.5
2.8. Collection of Plasma for Quantification of Biomarkers
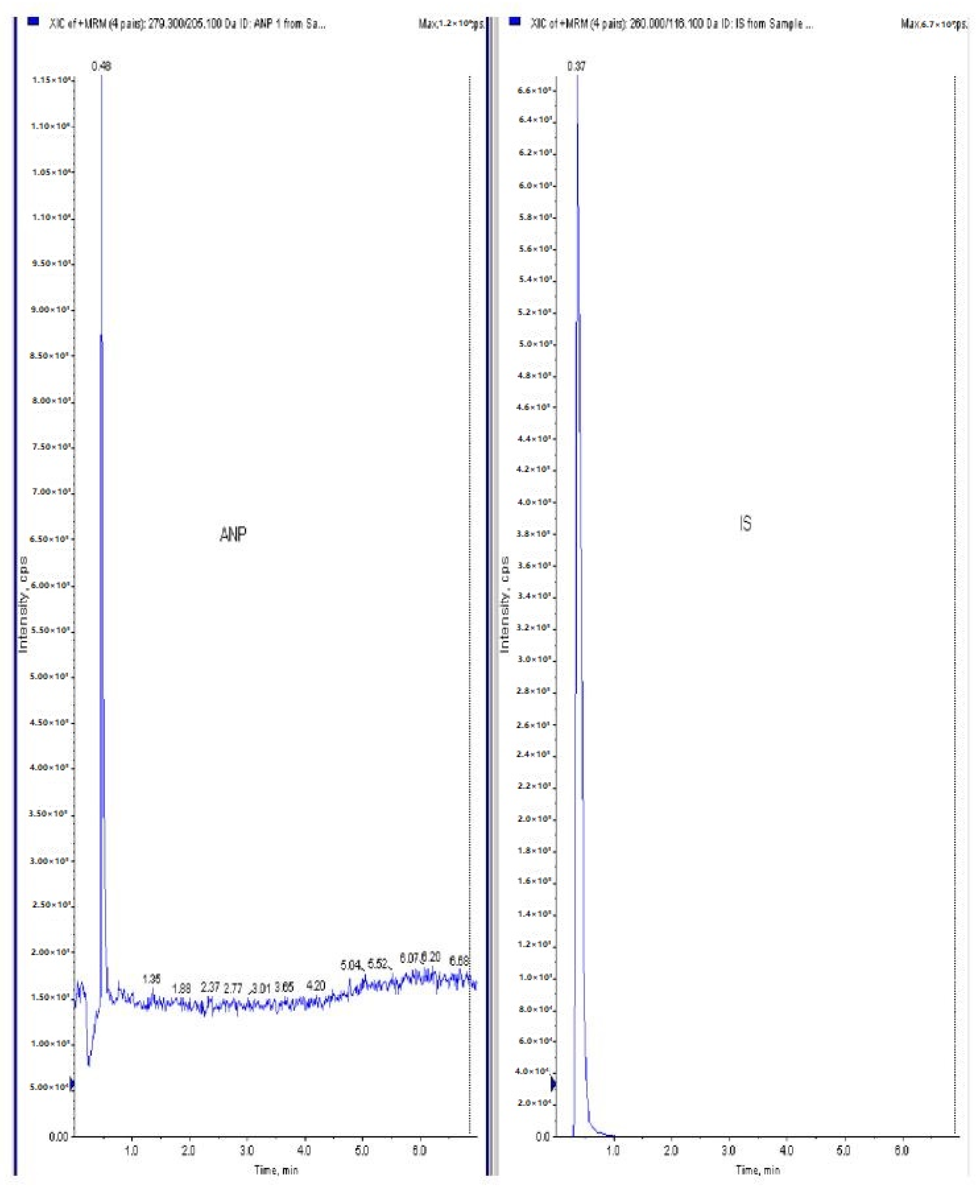
2.9. LC-MS/MS Methodology
Chromatographic Conditions
3. Statistical Analysis
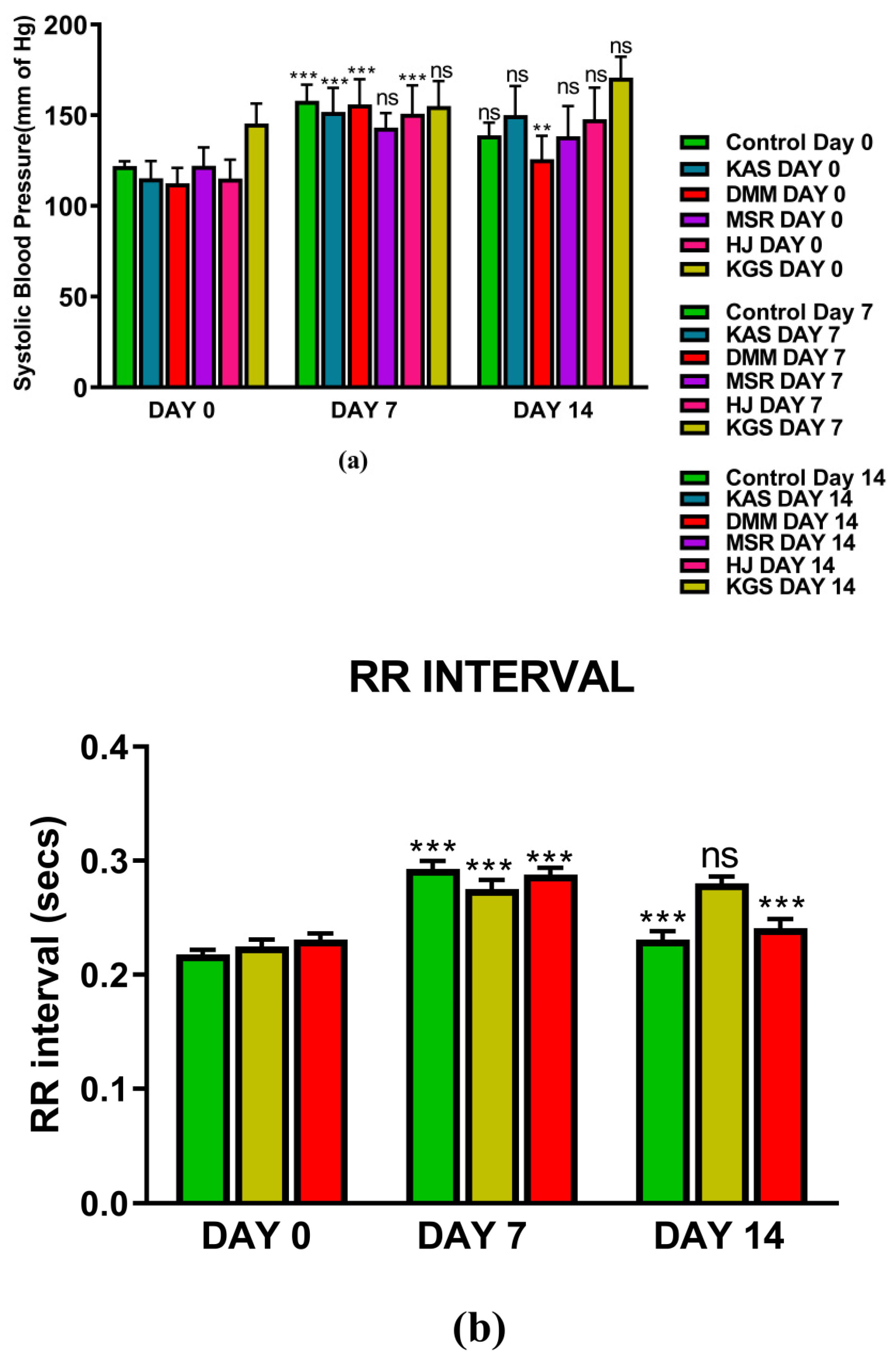
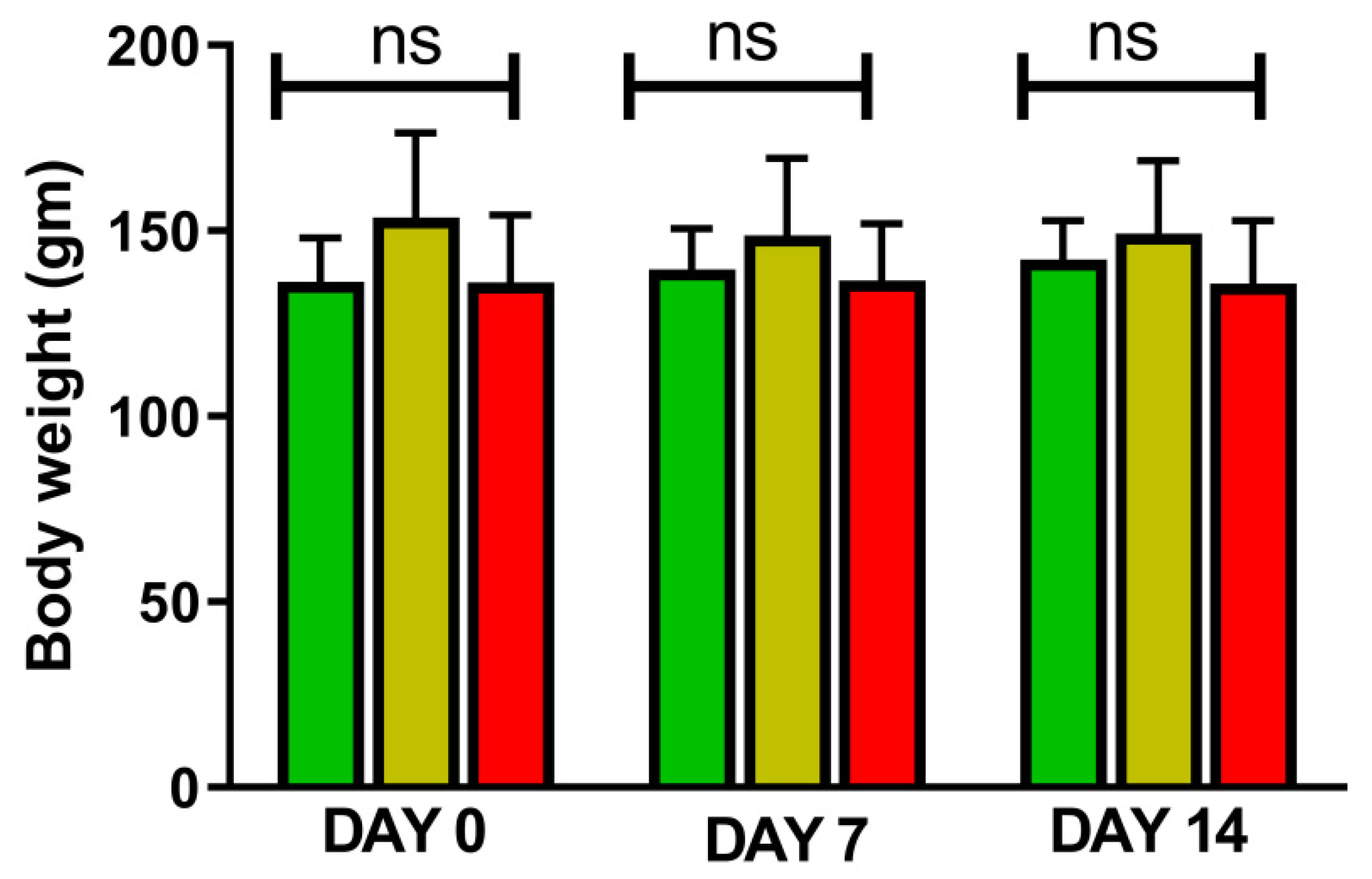
4. Results
5. Discussion
5.1. Summary of Observation on Day 7
5.1.1. DMM
5.1.2. KGS
5.2. Summary of Observations on Day 14
5.2.1. DMM
5.2.2. KGS
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Zhou, B.; Perel, P.; Mensah, G.A.; Ezzati, M. Global epidemiology, health burden and effective interventions for elevated blood pressure and hypertension. Nat. Rev. Cardiol. 2021, 18, 785–802. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Seto, S.W.; Chang, D.; Kiat, H.; Razmovski-Naumovski, V.; Chan, K.; Bensoussan, A. Synergistic Effects of Chinese Herbal Medicine: A Comprehensive Review of Methodology and Current Research. Front. Pharmacol. 2016, 7, 201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The Traditional Medicine and Modern Medicine from Natural Products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmad, S.; Rehman, S.; Ahmad, A.M.; Siddiqui, K.M.; Shaukat, S.; Khan, M.S.; Kamal, Y.T.; Jahangir, T. Khamiras, a natural cardiac tonic: An overview. J. Pharm. Bioallied Sci. 2010, 2, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health and Family Affairs. Part V. In National Formulary of Unani Medicine (NFUM); Central Council for Research in Unani Medicine, Government of India, Ministry of Health and Family Welfare, Department of Health: New Delhi, India, 2006; pp. 76, 79, 110. [Google Scholar]
- Ministry of Health and Family Affairs. Part I. In National Formulary of Unani Medicine (NFUM); Central Council for Research in Unani Medicine: New Delhi, India, 2006; pp. 19, 90. [Google Scholar]
- Ravishankar, B.; Shukla, V.J. Indian systems of medicine: A brief profile. Afr. J. Tradit. Complement. Altern. Med. 2007, 3, 319–337. [Google Scholar] [CrossRef] [Green Version]
- Gorain, B.; Choudhury, H.; Kundu, A.; Sarkar, L.; Karmakar, S.; Jaisankar, P.; Pal, T.K. Nanoemulsion strategy for olmesartan medoxomil improves oral absorption and extended antihypertensive activity in hypertensive rats. Colloids Surf. B Biointerfaces 2014, 115, 286–294. [Google Scholar] [CrossRef]
- Karmakar, S.; Padman, A.; Mane, N.S.; Sen, T. Hypokalemia: A potent risk for QTc prolongation in clarithromycin treated rats. Eur. J. Pharmacol. 2013, 709, 80–84. [Google Scholar] [CrossRef]
- Council of Europe; European Pharmacopoeia Commission; European Directorate for the Quality of Medicines & Healthcare. European Pharmacopoeia, 7th ed.; Council Of Europe: Strasbourg, Germany, 2010; p. 492. [Google Scholar]
- Maruna, R.F. Serum sodium determination; critical study on colorimetric determination and method. Clin. Chim. Acta 1957, 2, 581–585. [Google Scholar] [CrossRef]
- Trinder, P. Determination of serum sodium. Analyst 1951, 76, 596. [Google Scholar] [CrossRef]
- Suderman, H.J.; Delory, G.E. A rapid method for the determination of sodium in serum. Can. J. Med. Sci. 1952, 30, 302–307. [Google Scholar] [CrossRef]
- Lee, A.Y.; Choi, J.W.; Yokozawa, T.; Cho, E.J. Preventive effect of oligonol on nitric oxide and reactive oxygen species production through regulation of nuclear factor kappa B signaling pathway in RAW 264.7 macrophage cells against sodium nitroprusside. RSC Adv. 2019, 9, 3987–3993. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shishkova, E.; Coon, J.J. Rapid preparation of human blood plasma for bottom-up proteomics analysis. STAR Protoc. 2021, 2, 100856. [Google Scholar] [CrossRef] [PubMed]
- Pechanova, O.; Vrankova, S.; Cebova, M. Chronic L-Name-Treatment Produces Hypertension by Different Mechanisms in Peripheral Tissues and Brain: Role of Central eNOS. Pathophysiology 2020, 27, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Kvetnanský, R.; Pacák, K.; Tokarev, D.; Jeloková, J.; Jezová, D.; Rusnák, M. Chronic blockade of nitric oxide synthesis elevates plasma levels of catecholamines and their metabolites at rest and during stress in rats. Neurochem. Res. 1997, 22, 995–1001. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, S.; Ibrahim, M.; Schlitzer, A.; Maegdefessel, L.; Röll, W.; Pfeifer, A. PDGF regulates guanylate cyclase expression and cGMP signaling in vascular smooth muscle. Commun. Biol. 2022, 5, 298. [Google Scholar] [CrossRef] [PubMed]
- Chowdhary, S.; Ng, G.A.; Nuttall, S.L.; Coote, J.H.; Ross, H.F.; Townend, J.N. Nitric oxide and cardiac parasympathetic control in human heart failure. Clin. Sci. 2002, 102, 397–402. [Google Scholar] [CrossRef]
- Sung, J.H.; Jo, Y.S.; Kim, S.J.; Ryu, J.S.; Kim, M.C.; Ko, H.J.; Sim, S.S. Effect of Lutein on L-NAME-Induced Hypertensive Rats. Korean J. Physiol. Pharmacol. 2013, 17, 339–345. [Google Scholar] [CrossRef] [Green Version]
- Stauss, H.M. Heart rate variability: Just a surrogate for mean heart rate? Hypertension 2014, 64, 1184–1186. [Google Scholar] [CrossRef] [Green Version]
- Xu, H.; Li, J.; Zhong, G.; Li, L.; Huang, C.; Guo, P.; Chen, Y.; He, T. Characteristics of the Dynamic Electrocardiogram in the Elderly with Nonvalvular Atrial Fibrillation Combined with Long R-R Intervals. Evid. Based Complement. Alternat. Med. 2021, 2021, 4485618. [Google Scholar] [CrossRef]
- Ganguly, A. Atrial natriuretic peptide-induced inhibition of aldosterone secretion: A quest for mediator(s). Am. J. Physiol. 1992, 263 Pt 1, E181–E194. [Google Scholar] [CrossRef]
- Leskinen, H.; Vuolteenaho, O.; Leppäluoto, J.; Ruskoaho, H. Role of nitric oxide on cardiac hormone secretion: Effect of NG-nitro-L-arginine methyl ester on atrial natriuretic peptide and brain natriuretic peptide release. Endocrinology 1995, 136, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Kotchen, T.A. Hypertensive vascular disease. In Harrison’s Principles of Internal Medicine, 17th ed.; Tinsley, R.H., Anthony, S.F., Eds.; McGraw-Hill: New York, NY, USA, 1998; Volume 2, p. 1549, ISBN-10 1259834808. [Google Scholar]
- Martinez, D.V.; Rocha, R.; Matsumura, M.; Oestreicher, E.; Ochoa-Maya, M.; Roubsanthisuk, W.; Williams, G.H.; Adler, G.K. Cardiac damage prevention by eplerenone: Comparison with low sodium diet or potassium loading. Hypertension 2002, 39, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Essentials of Medical Pharmacology; Tripathi, K.D. (Ed.) Jaypee Brothers Medical: New Delhi, India, 2013; ISBN 13 978-9350259375. [Google Scholar]
- Walsh, C.R.; Larson, M.G.; Vasan, R.S.; Levy, D. Serum potassium is not associated with blood pressure tracking in the Framingham Heart Study. Am. J. Hypertens. 2002, 15 Pt 1, 130–136. [Google Scholar] [CrossRef] [Green Version]





| SL.No | Composition | Part Used | Quantity |
|---|---|---|---|
| 1 | Abresham muqarraz | - | 21.93 mg |
| 2 | Badranjboya (Nepeta hindostana (B.Heyne ex Roth) Haines) | Flower | 153.51 mg |
| 3 | Berg e Gawzaban (Borago officinalis L.) | Leaf | 131.58 mg |
| 4 | Burada Sandal Safaid (Santalum album L.) | Heart wood | 109.65 mg |
| 5 | Behman Surkh (Salvia haematodes L.) | Root | 87.72 |
| 6 | Tukhm Balangu (Lallemantia royleana (Benth)Benth. | Seed | 109.65 mg |
| 7 | Tudri Surkh (Cheiranthus cheiri L.) | Seed | 43.86 mg |
| 8 | Kishneez Khushk (Coriandrum sativum L.) | Fruit | 131.58 mg |
| 9 | Gule Khatmi (Althaea officinalis L.) | Flower | 43.86 mg |
| 10 | Gule Gawzaban (Borago officinalis L.) | Flower | 43.86 mg |
| 11 | Shakar safaid (Saccharum officinarum) | Crystal | 8.722 g |
| 12 | Sat Leemun (Citrus aurantium L.) | Crystal | 17.54 mg |
| 13 | Natroon Banjawi | - | 5.26 mg |
| SL.No | Composition | Part Used | Quantity |
|---|---|---|---|
| 1 | Zarishk (Berberis aristata DC) | Fruit | 132.0 mg |
| 2 | Tabasheer Safaid (Bambusa arundinace willd.) | Silicacious concretion | 88 mg |
| 3 | Sandal safaid (Santalum album L.) | Heart wood | 88 mg |
| 4 | Sandal Surkh (Pterocarpus santalinus L.f.) | Heart wood | 88 mg |
| 5 | Kishneez Muqashshar (Coriandrum sativum L.) | Dried seed | 88 mg |
| 6 | Gule Gawzaban (Borago officinalis L.) | Flower | 88 mg |
| 7 | Amla (Embelica officinalis) | Dried fruit | 88 mg |
| 8 | Tukhm Khurfa (Portulaca oleracea L.) | Dried seed | 88 mg |
| 9 | Gule Surkh (Rosa damascene Herrm.) | Flower | 51 mg |
| 10 | Abresham Muqarraz (Bombyx mori cocoon) | Cocoon | 51 mg |
| 11 | Darchini (Cinnamomum zeylanicum Blume) | Stem, Bark | 51 mg |
| 12 | Behman Safaid (Centaurea behen L.) | Root | 51 mg |
| 13 | Behan Surkh (Salvia haematodes L.) | Root | 51 mg |
| 14 | Darunaj Aqrabi (Doronicum hookeri C.B.Clarke ex Hook.f) | Rhizome | 51 mg |
| 15 | Ood Hindi (Aquilaria agallocha Roxb.) | Heart wood | 36 mg |
| 16 | Badranjboya (Nepeta hindostana (B.Heyne ex Roth) Haines) | Whole plant | 36 mg |
| 17 | Mastagi (Pistacia lentiscus L.) | 30 mg | |
| 18 | Ushana (Usnea longissima Ach.) | Thallus | 30 mg |
| 19 | Dana Elaichi Khurd (Elettaria cardamomum L.) | Dried seed | 30 mg |
| 20 | Qand Safaid (Saccharum officinarum) | Crystal | 2.44 g |
| 21 | Shahad (Honey) | -- | 1.22 g |
| 22 | Aabe Seb Shinn (Pyrus malus L.) | Fruit | 1.22 g |
| 23 | Zafran (Crocus sativa L.) | Stigma | 51 mg |
| 24 | Amber (resin) without musk | - | 14 mg |
| SL.No | Composition | Part Used | Quantity |
|---|---|---|---|
| 1 | Abresham Muqarraz | Cocoon | 603.3 mg |
| 2 | Berg Badranjboys ((Nepeta hindostana (B.Heyne ex Roth)) Haines) | Leaf | 452.4 mg |
| 3 | Berg Gawzaban (Borago officinalis L.) | Leaf | 262 mg |
| 4 | Gule Khatmi (Althaea officinalis L.) | Flower | 111.6 mg |
| 5 | Shakar Safaid (Saccharum officinarum) | Crystal | 9.049 g |
| 6 | Sat Leemun (Citrus aurantium L.) | Crystal | 36.20 mg |
| 7 | Natroon Banjawi | 9.049 mg | |
| 8 | Zafran (Crocus sativus L.) | Stigma | 5.279 mg |
| 9 |
| SL.No | Composition | Part of the Plant | Quantity |
|---|---|---|---|
| 1 | Afyun (Papaver somniferum L.) | Fruit | 25.68 mg |
| 2 | Jadwar (Delphinium denudatum Wall. ex Hook.f. and Thomson) | Tuber | 5.136 mg |
| 3 | Zafran (Crocus sativa L.) | Style and Stigma | 2.568 mg |
| 4 | Sheer Gao (Cow Milk) | 6.420 mL | |
| 5 | Narjeel (Cocos nucifera L.) | Kernel | 102.72 mg |
| 6 | Bisbasa (Myristica fragrans Houtt) | fruit coat | |
| 7 | Behman safed (Centaurea behen L.) | Root | 6.629 mg |
| 8 | Behman surkh (Salvia heamatodes L.) | Root | 6.629 mg |
| 9 | Maghz Badam Shirin (Prunus amygdalus L.) | Stem oil | 9.946 mg |
| 10 | Maghz Chilghoza (Pinus gerardiana Wall.) | Kernel | 9.946 mg |
| 11 | Tukhme Khurfa (Portulaca oleracea L.) | Seed | 9.946 mg |
| 12 | Roghan Balsan (Commiphora opobalsamum L.) | Oil and Stem | 13.268 mg |
| 13 | Jawitri (Myristica fragrans Houtt.) | Aril | 6.629 mg |
| 14 | Badranjboya (Nepeta hindostana (B.Heyne ex Roth) Haines) | Whole plant | 6.629 mg |
| 15 | Banslochan (Bambusa arundinacea willd.) | Silicacious concretion | 2.94 mg |
| 16 | Gond Keekar (Acacia arabica) | Gum | 2.94 |
| 17 | Misri (Saccharum officinarum) | Cubes | 13.268 mg |
| 18 | Kaleera (Cochlospermum religiosum (L.) Alston) | Gum | 2.94 mg |
| 19 | Ajwain Khurasani (Hyoscyamus niger L.) | Fruit | 2.94 |
| 20 | Beikh Luffah (Atropa Belladona L.) | Root | 2.94 |
| 21 | Jaiphal (Myristica fragrans Houtt.) | Seed | 2.94 |
| 22 | Warq Nuqra (silver) | 7.210 mg | |
| 23 | Ghee | q.s. |
| SL.No | Composition | Part of the Plant | Quantity |
|---|---|---|---|
| 1 | Agar (Aquilaria agallocha Roxb.) | Heart wood | 37 mg |
| 2 | Elaichi Khurd (Elettaria cardamomum L.) | Fruit | 55.5 mg |
| 3 | Berg Gawzaban (Borago officinalis L.) | Leaf | 92.5 mg. |
| 4 | Burada Sandal Surkh (Pterocarpus santalinus L.f.) | HeartWood | 27.75 mg |
| 5 | Burada Sandal Safaid (Santalum album L.) | HeartWood | 55.5 mg |
| 6 | Banslochan (Bambusa arundinacea willd) | Silicaceous concretion | 55.5 mg |
| 7 | Behman Surkh (Salvia haematodes L.) | Root | 37 mg |
| 8 | Tukhm Khurfa Siyah (Portulaca oleracea L.) | Seed | 83.25 mg |
| 9 | Tukhm Kahu (Lactuca sativa L.) | Seed | 83.25 mg |
| 10 | Qust Shireen (Saussurea lappa (Falc.) Lipsch.) | Root | 37 mg |
| 11 | Zarambad (Curcuma zedoaria Roxb.) | Root Trunk | 37 mg |
| 12 | Sartan Sokhta (Scylla serrata (Forskål, 1775) ash) | - | 27.75 mg |
| 13 | Gule Surkh (Rosa damascene Herrm.) | Flower | 138.75 mg |
| 14 | Maghz Tukhm Kharbooza (Cucumis melo L.) | - | 83.25 mg |
| 15 | Maghz Tukhm Khayarain (Cucumis sativus L.) | - | 83.25 mg |
| 16 | Maghz Kadu Shireen (Cucurbita maxima Duch ex.Lam) | - | 83.25 mg |
| 17 | Kafoor (Camphor) | - | 27.75 mg |
| 18 | Busad Sokhta Mehlool | - | 27.75 mg, |
| 19 | Marwareed Mehlool | - | 27.75 mg |
| 20 | Abresham Muqarraz | Cocoon | 27.75 mg |
| 21 | Qwam Shakar Safaid (Saccharum officinarum) | Crystal | 2.222 g |
| 22 | Rub Anar Shireen (Punica granatum L.) | Fruit | 555 mg |
| 23 | Rub Behi (Cydonia oblonga Mill) | Fruit | 555 mg |
| 24 | Rub Seb (Malus sylvestris (L.) Mill.) | Fruit | 555 mg |
| 25 | Natroon Banjawi. | - | 5.55 mg |
| 26 | Zafran (Crocus sativa L.) | Stigma | 6.48 mg |
| 27 | Arq Gawzaban (Borago officinalis L.) | Distilled | 0.07 mL |
| 28 | Warq Nuqra | - | 9.25 mg |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shaharyar, M.A.; Bhowmik, R.; Afzal, O.; Altamimi, A.S.A.; Alzarea, S.I.; Almalki, W.H.; Ali, S.Z.; Mandal, P.; Mandal, A.; Ayoob, M.; et al. Anti-Hypertensive Activity of Some Selected Unani Formulations: An Evidence-Based Approach for Verification of Traditional Unani Claims Using LC-MS/MS for the Evaluation of Clinically Relevant Blood Parameters in Laboratory Rats. J. Clin. Med. 2022, 11, 4628. https://doi.org/10.3390/jcm11154628
Shaharyar MA, Bhowmik R, Afzal O, Altamimi ASA, Alzarea SI, Almalki WH, Ali SZ, Mandal P, Mandal A, Ayoob M, et al. Anti-Hypertensive Activity of Some Selected Unani Formulations: An Evidence-Based Approach for Verification of Traditional Unani Claims Using LC-MS/MS for the Evaluation of Clinically Relevant Blood Parameters in Laboratory Rats. Journal of Clinical Medicine. 2022; 11(15):4628. https://doi.org/10.3390/jcm11154628
Chicago/Turabian StyleShaharyar, Md. Adil, Rudranil Bhowmik, Obaid Afzal, Abdulmalik S. A. Altamimi, Sami I. Alzarea, Waleed Hassan Almalki, Sk Zeeshan Ali, Pallab Mandal, Avishek Mandal, Mohd Ayoob, and et al. 2022. "Anti-Hypertensive Activity of Some Selected Unani Formulations: An Evidence-Based Approach for Verification of Traditional Unani Claims Using LC-MS/MS for the Evaluation of Clinically Relevant Blood Parameters in Laboratory Rats" Journal of Clinical Medicine 11, no. 15: 4628. https://doi.org/10.3390/jcm11154628
APA StyleShaharyar, M. A., Bhowmik, R., Afzal, O., Altamimi, A. S. A., Alzarea, S. I., Almalki, W. H., Ali, S. Z., Mandal, P., Mandal, A., Ayoob, M., Kazmi, I., & Karmakar, S. (2022). Anti-Hypertensive Activity of Some Selected Unani Formulations: An Evidence-Based Approach for Verification of Traditional Unani Claims Using LC-MS/MS for the Evaluation of Clinically Relevant Blood Parameters in Laboratory Rats. Journal of Clinical Medicine, 11(15), 4628. https://doi.org/10.3390/jcm11154628