Oral and Maxillofacial Infections—A Bacterial and Clinical Cross-Section
Abstract
:1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
3.1. Comparison of Clinical Profile between Diabetic and Non-Diabetic Patients
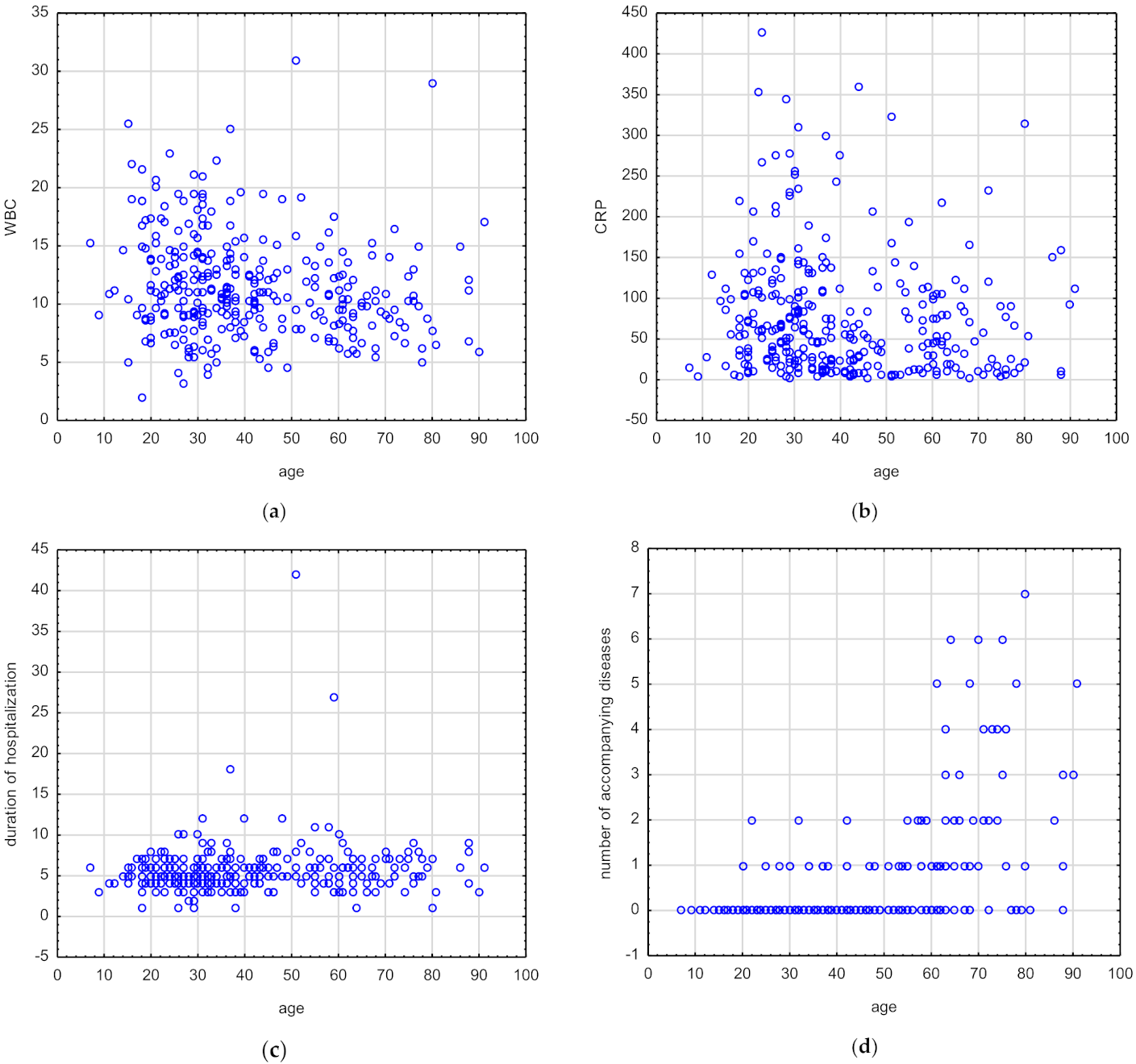
3.2. Correlations between Age and Selected Laboratory and Clinical Parameters in Patients with Oral and Maxillofacial Infections
3.3. Correlations between Accompanying Diseases and Selected Laboratory and Clinical Parameters in Patients with Oral and Maxillofacial Infections
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jevon, P.; Abdelrahman, A.; Pigadas, N. Management of odontogenic infections and sepsis: An update. Br. Dent. J. 2020, 34, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Rastenienė, R.; Pūrienė, A.; Aleksejūnienė, J.; Pečiulienė, V.; Zaleckas, L. Odontogenic Maxillofacial Infections: A Ten-Year Retrospective Analysis. Surg. Infect. 2015, 16, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Błochowiak, K.J.; Kamiński, B.; Sokalski, J. Deep neck infections of non-odontogenic origin: Clinical manifestation and treatment. Med. Stud. 2018, 34, 98–102. [Google Scholar] [CrossRef] [Green Version]
- Blankson, P.K.; Parkins, G.; Boamah, M.O.; Abdulai, A.E.; Ahmed, A.M.; Bondorin, S.; Nuamah, I. Severe odontogenic infections: A 5-year review of a major referral hospital in Ghana. Pan Afr. Med. J. 2019, 32, 71. [Google Scholar] [CrossRef]
- Sánchez, R.; Mirada, E.; Arias, J.; Paño, J.R.; Burgueño, M. Severe odontogenic infections: Epidemiological, microbiological and therapeutic factors. Med. Oral Patol. Oral. Cir. Bucal. 2011, 16, e670–e676. [Google Scholar] [CrossRef]
- Zawiślak, E.; Nowak, R. Odontogenic Head and Neck Region Infections Requiring Hospitalization: An 18-Month Retrospective Analysis. BioMed Res. Int. 2021, 2021, 7086763. [Google Scholar] [CrossRef]
- Keswani, E.S.; Venkateshwa, G. Odontogenic Maxillofacial Space Infections: A 5-Year Retrospective Review in Navi Mumbai. J. Maxillofac. Oral Surg. 2019, 18, 345–353. [Google Scholar] [CrossRef]
- Yuvaraj, V. Maxillofacial Infections of Odontogenic Origin: Epidemiological, Microbiological and Therapeutic Factors in an Indian Population. Indian J. Otolaryngol. Head Neck Surg. 2016, 68, 396–399. [Google Scholar] [CrossRef] [Green Version]
- Katoumas, K.; Anterriotis, D.; Fyrgiola, M.; Lianou, V.; Triantafylou, D.; Dimopoulos, I. Epidemiological Analysis of Management Of Severe Odontogenic Infections Before Referral To The Emergency Department. J. Craniomaxillofac. Surg. 2019, 47, 1292–1299. [Google Scholar] [CrossRef]
- Kauffmann, P.; Cordesmeyer, R.; Tröltzsch, M.; Sömmer, C.; Laskawi, R. Deep neck infections: A single-center analysis of 63 cases. Med. Oral Patol. Oral Cir. Bucal. 2017, 22, e536–e541. [Google Scholar] [CrossRef]
- Martínez Pascual, P.; Pinacho Martinez, P.; Friedlander, E.; Martin Oviedo, C.; Scola Yurrita, B. Peritonsillar and deep neck infections: A review of 330 cases. Braz. J. Otorhinolaryngol. 2018, 84, 305–310. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.W.; Liu, Y.H.; Su, H.H. Bacteriology of peritonsillar abscess: The changing trend and predisposing factors. Braz. J. Otorhinolaryngol. 2018, 84, 532–539. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Hu, L.; Wang, Z.; Nie, G.; Li, X.; Lin, D.; Luo, J.; Qin, H.; Wu, J.; Wen, W.; et al. Deep Neck Infection: A Review of 130 Cases in Southern China. Medicine 2015, 94, e994. [Google Scholar] [CrossRef] [PubMed]
- Weise, H.; Naros, A.; Weise, C.; Reinert, S.; Hoefert, S. Severe odontogenic infections with septic progress—A constant and increasing challenge: A retrospective analysis. BMC Oral Health 2019, 19, 173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiaojie, L.; Hui, L.; Zhongcheng, G.; Chenggang, W.; Yaqi, N. The Predictive Value of Interleukin-6 and Neutrophil-Lymphocyte Ratio in Patients with Severe and Extremely Severe Oral and Maxillofacial Space Infections. Biomed Res. Int. 2021, 2021, 2615059. [Google Scholar] [CrossRef] [PubMed]
- Stathopoulos, P.; Igoumenakis, D.; Shuttleworth, J.; Smith, W.; Ameerally, P. Predictive factors of hospital stay in patients with odontogenic maxillofacial infections: The role of C-reactive protein. Br. J. Oral Maxillofac. Surg. 2017, 55, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Rao, D.D.; Desai, A.; Kulkarni, R.D.; Gopalkrishnan, K.; Rao, C.B. Comparison of maxillofacial space infection in diabetic and nondiabetic patients. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2010, 110, e7–e12. [Google Scholar] [CrossRef]
- Zheng, L.; Yang, C.; Zhang, W.; Cai, X.; Kim, E.; Jiang, B.; Wang, B.; Pu, Y.; Wang, J.; Zhang, Z.; et al. Is there association between severe multispace infections of the oral maxillofacial region and diabetes mellitus? J. Oral Maxillofac. Surg. 2012, 70, 1565–1572. [Google Scholar] [CrossRef]
- Seppänen, L.; Rautemaa, R.; Lindqvist, C.; Lauhio, A. Changing clinical features of odontogenic maxillofacial infections. Clin. Oral Investig. 2010, 14, 459–465. [Google Scholar] [CrossRef]
- Meurman, J.H.; Hämäläinen, P. Oral health and morbidity—Implications of oral infections on the elderly. Gerodontology 2006, 23, 3–16. [Google Scholar] [CrossRef]
- Celakovsky, P.; Kalfert, D.; Smatanova, K.; Tucek, L.; Cermakova, E.; Mejzlik, J.; Kotulek, M.; Vrbacky, A.; Matousek, P.; Stanikova, L.; et al. Bacteriology of deep neck infections: Analysis of 634 patients. Aust. Dent. J. 2015, 60, 212–215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Farmahan, S.; Tuopar, D.; Ameerally, P.J. The clinical relevance of microbiology specimens in head and neck space infections of odontogenic origin. Br. J. Oral Maxillofac. Surg. 2014, 52, 629–631. [Google Scholar] [CrossRef] [PubMed]
- Rega, A.J.; Aziz, S.R.; Ziccardi, V.B. Microbiology and antibiotic sensitivities of head and neck space infections of odontogenic origin. J. Oral Maxillofac. Surg. 2006, 64, 1377–1380. [Google Scholar] [CrossRef] [PubMed]
- Galioto, N.J. Peritonsillar Abscess. Am Fam Physician 2017, 95, 501–506. [Google Scholar] [PubMed]
- Johnston, J.; Stretton, M.; Mahadevan, M.; Douglas, R.G. Peritonsillar abscess: A retrospective case series of 1773 patients. Clin. Otolaryngol. 2018, 43, 940–944. [Google Scholar] [CrossRef]
- Klug, T.; Greve, T.; Hentze, M. Complications of peritonsillar abscess. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 32. [Google Scholar] [CrossRef] [PubMed]
- Mazur, E.; Czerwińska, E.; Korona-Głowniak, I.; Grochowalska, A.; Kozioł-Montewka, M. Epidemiology, clinical history and microbiology of peritonsillar abscess. Eur. J. Clin. Microbiol. Infect. Dis. 2015, 34, 549–554. [Google Scholar] [CrossRef] [Green Version]
- Stefanopoulos, P.K.; Kolokotronis, A.E. The clinical significance of anaerobic bacteria in acute orofacial odontogenic infections. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2004, 98, 398–408. [Google Scholar] [CrossRef]
- Mejzlik, J.; Celakovsky, P.; Tucek, L.; Kotulek, M.; Vrbacky, A.; Matousek, P.; Stanikova, L.; Hoskova, T.; Pazs, A.; Mittu, P.; et al. Univariate and multivariate models for the prediction of life-threatening complications in 586 cases of deep neck space infections: Retrospective multi-institutional study. J. Laryngol. Otol. 2017, 131, 779–784. [Google Scholar] [CrossRef]
- Han, X.; An, J.; Zhang, Y.; Gong, X.; He, Y. Risk Factors for Life-Threatening Complications of Maxillofacial Space Infection. J. Craniofac. Surg. 2016, 27, 385–390. [Google Scholar] [CrossRef] [Green Version]
- Wei, J.; Luo, J.; Wang, X.; Chen, G.; Wang, X.; Wang, J. Clinical features of patients of different ages with postoperative multi-space maxillofacial infection. Cell Biochem. Biophys. 2014, 70, 1779–1782. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.Y.; Selvaraju, K.; Audimulam, H.; Yong, Z.C.; Adnan, T.H.; Balasundram, S. Length of hospital stay among oral and maxillofacial patients: A retrospective study. J. Korean Assoc. Oral Maxillofac. Surg. 2021, 47, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Kozakiewicz, M.; Trzcińska-Kubik, M.; Wlazeł, R.N. Index of Body Inflammation for Maxillofacial Surgery Purpose-to Make the Soluble Urokinase-Type Plasminogen Activator Receptor Serum Level Independent on Patient Age. Appl. Sci. 2021, 11, 1345. [Google Scholar] [CrossRef]
- Kozakiewicz, M.; Wlazeł, R.N. Does Soluble Urokinase-Type Plasminogen Activator Receptor Level Predicts the Occurrence of Inflammatory Complications in Maxillofacial Surgery? Appl. Sci. 2021, 11, 2192. [Google Scholar] [CrossRef]
- Slouka, D.; Hanakova, J.; Kostlivy, T.; Skopek, P.; Kubec, V.; Babuska, V.; Pecen, L.; Topolcan, O.; Kucera, R. Epidemiological and Microbiological Aspects of the Peritonsillar Abscess. Int. J. Environ. Res. Public Health 2020, 17, 4020. [Google Scholar] [CrossRef] [PubMed]
- Trybek, G.; Chruściel-Nogalska, M.; Machnio, M.; Smektała, T.; Malinowski, J.; Tutak, M.; Sporniak-Tutak, K. Surgical extraction of impacted teeth in elderly patients. A retrospective analysis of perioperative complications—The experience of a single institution. Gerodontology 2016, 33, 410–415. [Google Scholar] [CrossRef] [PubMed]
| Parameters | Values |
|---|---|
| Number of individuals, n | 329 |
| Gender, Female/Male, n | 137/192 |
| Age, mean ± SD years | 40.88 ± 18.15 |
| Age, median (range), years | 36.00 (7.00–91.00) |
| BMI (kg/m2), median | 24.02 |
| Symptoms, n (%): | |
| Pain | 302 (91.8) |
| Trismus | 129 (39.2) |
| Dysphagia | 160 (48.6) |
| Otalgia | 12 (3.6) |
| Fever | 47 (14.2) |
| Dyspnea | 8 (2.4) |
| Neck swelling | 138 (41.9) |
| Sialorrhea | 1 (0.3) |
| Hoarseness | 4 (1.2) |
| Other symptoms | 106 (32.2) |
| Number of symptoms, n: | |
| ≤2 symptoms | 147 |
| 3 symptoms | 107 |
| ≥4 symptoms | 75 |
| Space involvement, n (%) | |
| Submandibular | 92 (27.96) |
| Parapharyngeal | 8 (2.43) |
| Peritonsillar | 152 (46.2) |
| Buccal | 54 (16.41) |
| Parotid | 5 (1.52) |
| Temporal | 3 (0.60) |
| Infratemporal | 3 (0.60) |
| Submental | 5 (1.52) |
| Orbital | 5 (1.52) |
| Cervical | 20 (6.07) |
| Lacrimal sac | 1 (0.30) |
| Other space involvement (tongue, lip, epiglottis, lower pharynx) | 7 (2.12) |
| Laboratory values | |
| WBC (K/μL) | 10.94 |
| ESR (mm/h) | 44.00 |
| CRP (mg/L) | 55.48 |
| Causes, n (%) | |
| Odontogenic | 106 (32.2) |
| Tonsilitis | 155 (47.1) |
| Sialoadenitis | 6 (1.8) |
| Sinusitis | 1 (0.3) |
| Posttraumatic | 13 (3.9) |
| Postoperative | 10 (3.0) |
| Other/Undetermined | 33 (10.0) |
| Number of accompanying diseases, n: | |
| One disease | 41 |
| ≥Two diseases | 26 |
| Accompanying systemic diseases, n: | |
| Peripheral vascular diseases | 42 |
| Circulatory failure | 32 |
| Diabetes without complications | 24 |
| Diabetes with complications | 7 |
| Cerebrovascular diseases | 13 |
| Connective tissue diseases | 7 |
| Chronic obstructive pulmonary disease | 6 |
| Liver failure | 6 |
| Renal failure | 5 |
| Dementia | 5 |
| Other: Solid tumor, Peptic ulcer disease, Paresis, Stroke, Myocardial infarction, Disseminated tumor | 10 |
| Parameters | Values |
|---|---|
| Duration of hospitalization, median (range), days | 5.00 (1.00–42.00) |
| Antibiotics n | |
| No use of antibiotics | 2 |
| Use of single antibiotic | 327 |
| First-choice antibiotics: cefuroxime, lincomycin, penicillin, gentamicin, amoxicillin + clavulanic acid, meropenemum, clindamycin, azithromycin, vancomycin | |
| Multi-antibiotic therapy: | |
| Use of two antibiotics | |
| Second choice antibiotics: metronidazole, cefazolin, cefuroxime, clindamycin, gentamycin | |
| Third choice antibiotics: gentamycin, cefuroxime, metronidazole, penicillin, clindamycin, lincomycin, | 252 |
| Use of three antibiotics | |
| Microbiology n | 16 |
| Gram-positive bacteria: | |
| Staphylococcus: | |
| Aureus | 35 |
| Epidermidis | 25 |
| Capitis | 4 |
| Hominis | 4 |
| Other: Warneri, Auricularis, Xylosus | 3, 1, 1 |
| Streptococcus: | |
| Mitis, | 40 |
| Anginosus, | 20 |
| Haemolyticus, | 12 |
| Sanguinis, | 8 |
| Mutaris, | 8 |
| Oralis, | 4 |
| Identified as gr C | 4 |
| Other: Salivarius, Parasanguinis, Constellatus, Pluranimalium, | 3, 3, 3, 3 |
| Intermedius Pseudoporcinus, Agalactiae, | 2, 2, 2 |
| Ovis, Gordonii, Liquefaciens, Vestibularis | 1, 1, 1, 1 |
| Enterococcus: faecalis, casseliflavus | 6, 1 |
| Leuconostoc mesenteroides | 2 |
| Eggerthella lenta | 1 |
| Gram-negative bacteria: | |
| Pseudomonas aerigunosa | 11 |
| Acinetobacter baumannii | 2 |
| Escherichia coli | 6 |
| Klebsiella pneumoniae | 5 |
| Moraxella catarrhalis | 1 |
| Citrobacter: freundii, brakii | 5 |
| Enterobacter cloacae | 5 |
| Serratia marcescens | 2 |
| Anaerobic bacteria:Leuconostoc mesenteroides, Eggerthella lenta, | |
| Escherichia coli | 9 |
| Candida albicans | 246 |
| Biopsy | |
| Type of anesthesia n, (%) | |
| Local anesthesia | 221 (67.1) |
| General intravenous anesthesia | 5 (1.51) |
| Complications n, (%) | |
| Tracheostomy | 3 (0.91) |
| Intubation | 10 (3.03) |
| Parameters | Non-Diabetic Patients | Diabetic Patients | p Value |
|---|---|---|---|
| Number of individuals, n | 297 | 31 | |
| Age, years, mean (±SD) | 39.12 (±17.37) | 57.77 (±16.98) | |
| Age, years, median (confidence interval) | 35.00 (27.00–48.00) | 61.00 (51.00–71.00) | ˂0.0001 a |
| BMI (kg/m2), median (confidence interval) | 23.79 (21.46–27.27) | 27.48 (22.83–31.25) | 0.0198 a |
| WBC (K/μL) median (confidence interval) | 11.00 (8.68–13.87) | 10.72 (8.90–14.00) | 0.5491 a |
| ESR (mm/h) median (confidence interval) | 41.00 (26.00–66.00) | 72.50 (32.00–116.50) | 0.0292 a |
| CRP (mg/L) median (confidence interval) | 55.61 (23.06–105.44) | 44.01 (12.50–114.00) | 0.5552 a |
| Duration of hospitalization (days) | 5.00 (4.00–6.00) | 6.00 (4.00–7.00) | 0.0631 a |
| Number of accompanying diseases | 0.00 (0.00–0.00) | 1.00 (0.00–3.00) | 0.0000 a |
| Symptoms | |||
| Number of symptoms | 3.00 (2.00–3.00) | 2.00 (2.00–5.00) | 0.8470 a |
| Pain n, yes/no | 274/24 | 28/3 | 0.7299 c |
| Trismus, n, (%) yes/no | 120 (40.27)/178 (59.73) | 9 (29.03)/22 (70.97) | 0.2226 b |
| Dysphagia, n, (%) yes/no | 144 (48.32)/154 (51.68) | 16 (51.61)/15 (48.39) | 0.7271 b |
| Otalgia n, (%) yes/no | 8 (2.68)/290 (97.32) | 4 (12.90)/27 (87.10) | 0.0187 c |
| Fever n, (%) yes/no | 41 (13.76)/257 (86.24) | 6 (19.35)/25 (80.65) | 0.4174 |
| Dyspnea n, (%) yes/no | 3 (1.01)/294 (98.99) | 5 (16.13)/26 (83.87) | 0.0002 |
| Hoarseness n, (%) yes/no | 1 (0.34)/297 (99.66) | 3 (9.68)/28 (90.32) | 0.0028 c |
| Sialorrhea n, (%) yes/no | 1 (0.34)/297 (99.66) | 0 (0.00)/31 (100.00) | 1.0000 c |
| Cervical swelling n, (%) yes/no | 125 (41.95)/173 (58.05) | 13 (41.94)/18 (58.06) | 0.9990 b |
| Space involvement | |||
| Submandibular n, (%) | 82 (27.52) | 10 (32.26) | 0.5756 c |
| Buccal n, (%) | 48 (16.11) | 6 (19.35) | 0.6422 b |
| Parotid n, (%) | 4 (1.34) | 1 (3.23) | 0.3922 c |
| Temporal n, (%) | 1 (0.34) | 1 (3.23) | 0.1798 c |
| Infratemporal n, (%) | 1 (0.34) | 1 (3.23) | 0.1798 c |
| Submental n, (%) | 3 (1.01) | 2 (6.45) | 0.0718 c |
| Orbital n, (%) | 4 (1.34) | 1 (3.23) | 0.3922 c |
| Lacrimal sac n, (%) | 1 (0.34) | 0 (0.00) | 1.0000 c |
| Cervical n, (%) | 14 (4.70) | 6 (19.35) | 0.0064 c |
| Causes | |||
| Odontogenic n, (%) | 93 (31.21) | 13 (41.94) | 0.2238 b |
| Tonsillitis n, (%) | 147 (49.33) | 8 (25.81) | 0.0125 b |
| Sialoadenitis n, (%) | 5 (1.68) | 1 (3.23) | 0.4504 c |
| Sinusitis n, (%) | 1 (0.34) | 0 (0.00) | 1.0000 c |
| Posttraumatic n, (%) | 13 (4.36) | 0 (0.00) | 0.6205 c |
| Postoperative n, (%) | 9 (3.02) | 1 (3.23) | 1.0000 c |
| Iatrogenic n, (%) | 2 (0.67) | 0 (0.00) | 1.0000 c |
| Unidentified n, (%) | 4 (1.34) | 1 (3.23) | 0.3922 c |
| Complications n, (%) | 0 (0.00) | 1 (3.23) | 0.0942 c |
| Parameters | rs | p Value | |
|---|---|---|---|
| Age | BMI | 0.46 | 0.00 |
| WBC | −0.21 | ˂0.001 | |
| CRP | −0.16 | ˂0.001 | |
| ESR | 0.07 | 0.39 | |
| Duration of hospitalization | 0.13 | 0.01 | |
| Number of symptoms | −0.10 | 0.06 | |
| Number of accompanying diseases | 0.51 | 0.00 |
| Symptoms | Mean ± SD | Median | Min | Max | Q1 | Q3 | p-Value |
|---|---|---|---|---|---|---|---|
| Pain | |||||||
| Yes, n = 301 | 40.94 ± 18.04 | 36.00 | 7.00 | 91.00 | 27.00 | 55.00 | 0.7013 |
| No, n = 27 | 40.22 ± 19.69 | 34.00 | 14.00 | 81.00 | 27.00 | 57.00 | |
| Trismus | |||||||
| Yes, n = 128 | 36.70 ± 16.41 | 31.00 | 12.00 | 91.00 | 26.00 | 44.00 | 0.0006 |
| No, n = 200 | 43.56 ± 18.74 | 40.00 | 7.00 | 90.00 | 29.00 | 59.00 | |
| Dysphagia | |||||||
| Yes, n = 159 | 38.60 ± 17.89 | 33.00 | 7.00 | 91.00 | 26.00 | 49.00 | 0.0178 |
| No, n = 169 | 43.02 ± 18.19 | 38.00 | 9.00 | 90.00 | 29.00 | 58.00 | |
| Otalgia | |||||||
| Yes, n = 12 | 38.00 ± 17.92 | 30.00 | 18.00 | 80.00 | 27.50 | 45.50 | 0.4959 |
| No, n = 316 | 40.99 ± 18.18 | 36.00 | 7.00 | 91.00 | 27.00 | 55.00 | |
| Fever | |||||||
| Yes, n = 47 | 38.44 ± 19.23 | 31.00 | 15.00 | 88.00 | 25.00 | 42.00 | 0.1621 |
| No, n = 281 | 41.29 ± 17.97 | 36.00 | 7.00 | 91.00 | 28.00 | 55.00 | |
| Neck swelling | |||||||
| Yes, n = 137 | 40.42 ± 16.76 | 36.00 | 18.00 | 88.00 | 27.00 | 52.00 | 0.8478 |
| No, n = 191 | 41.21 ± 19.12 | 36.00 | 7.00 | 91.00 | 27.00 | 58.00 | |
| Dyspnea | |||||||
| Yes, n = 8 | 60.12 ± 19.32 | 58.50 | 33.00 | 86.00 | 45.00 | 77.50 | 0.0070 |
| No, n = 319 | 40.34 ± 17.88 | 36.00 | 7.00 | 91.00 | 27.00 | 54.00 |
| Parameters | rs | p Value | |
|---|---|---|---|
| Accompanying diseases | BMI | 0.23 | 0.000 |
| WBC | −0.14 | 0.008 | |
| CRP | −0.06 | 0.25 | |
| ESR | 0.14 | 0.07 | |
| Duration of hospitalization | 0.07 | 0.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamiński, B.; Błochowiak, K.; Kołomański, K.; Sikora, M.; Karwan, S.; Chlubek, D. Oral and Maxillofacial Infections—A Bacterial and Clinical Cross-Section. J. Clin. Med. 2022, 11, 2731. https://doi.org/10.3390/jcm11102731
Kamiński B, Błochowiak K, Kołomański K, Sikora M, Karwan S, Chlubek D. Oral and Maxillofacial Infections—A Bacterial and Clinical Cross-Section. Journal of Clinical Medicine. 2022; 11(10):2731. https://doi.org/10.3390/jcm11102731
Chicago/Turabian StyleKamiński, Bartłomiej, Katarzyna Błochowiak, Konrad Kołomański, Maciej Sikora, Sławomir Karwan, and Dariusz Chlubek. 2022. "Oral and Maxillofacial Infections—A Bacterial and Clinical Cross-Section" Journal of Clinical Medicine 11, no. 10: 2731. https://doi.org/10.3390/jcm11102731
APA StyleKamiński, B., Błochowiak, K., Kołomański, K., Sikora, M., Karwan, S., & Chlubek, D. (2022). Oral and Maxillofacial Infections—A Bacterial and Clinical Cross-Section. Journal of Clinical Medicine, 11(10), 2731. https://doi.org/10.3390/jcm11102731







