Abstract
Biliary tract cancer, and intrahepatic cholangiocarcinoma (iCC) in particular, represents a rather uncommon, highly aggressive malignancy with unfavorable prognosis. Therapeutic options remain scarce, with platinum-based chemotherapy is being considered as the gold standard for the management of advanced disease. Comprehensive molecular profiling of tumor tissue biopsies, utilizing multi-omics approaches, enabled the identification of iCC’s intratumor heterogeneity and paved the way for the introduction of novel targeted therapies under the scope of precision medicine. Yet, the unmet need for optimal care of patients with chemo-refractory disease or without targetable mutations still exists. Immunotherapy has provided a paradigm shift in cancer care over the past decade. Currently, immunotherapeutic strategies for the management of iCC are under intense research. Intrinsic factors of the tumor, including programmed death-ligand 1 (PD-L1) expression and mismatch repair (MMR) status, are simply the tip of the proverbial iceberg with regard to resistance to immunotherapy. Acknowledging the significance of the tumor microenvironment (TME) in both cancer growth and drug response, we broadly discuss about its diverse immune components. We further review the emerging role of immunotherapy in this rare disease, summarizing the results of completed and ongoing phase I–III clinical trials, expounding current challenges and future directions.
1. Introduction
Primary hepatic malignancies, which represent the fourth cause of cancer-related death worldwide, can be mainly classified as hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (iCC) [1,2]. HCC and iCC are two closely related yet distinct entities that share common molecular and phenotypic characteristics [3]. Interestingly, the combination of HCC and iCC has been also described as a rare type of primary liver cancer, which contains pathologic features of both HCC and iCC cells [4]. Within this context, several clinical trials evaluating novel immunotherapies for patients with primary liver cancer include both HCC and iCC.
The incidence of iCC has been consistently rising in high-income countries, from 0.1 to 0.6 cases per 100,000 over the last three decades [2,5,6], whereas the mortality rates also follow the incidence pattern [7,8]. iCC is defined as a desmoplastic stroma-rich adenocarcinoma of cholangiocyte origin, arising proximal to the secondary biliary ducts [9,10]. In general, iCC is more prevalent in elderly male patients [11], whilst several risk factors have been identified, including obesity, diabetes, primary sclerosing cholangitis, Caroli’s disease, hepatolithiasis, viral hepatitis, liver fluke infections, and cirrhosis [12,13]. Noteworthily, almost 50% of iCC cases have no identifiable risk factor [14].
Patients with iCC usually present with non-specific symptoms (fatigue, weight loss, abdominal pain, obstructive jaundice) when the disease has been ultimately disseminated [15]. Less than 25% of patients are eligible to undergo surgery, which remains the only curative potential for early-stage disease [16]. Even after microscopically margin-negative (R0) resection, prognosis remains dismal due to the high risk of recurrence; a median overall survival (OS) of 40 months has been reported [17]. There is no clinical evidence at present to support the use of radiotherapy for locoregional cancer control, while the American Society of Clinical Oncology (ASCO) guidelines recommend the addition of adjuvant capecitabine [18], based on the reported benefit in both relapse-free survival (RFS) and OS of the randomized phase III BILCAP trial [19].
Current treatment options for advanced-stage iCC are limited and the median OS of these patients varies between 2.5 and 4.5 months [20,21]. Since 2010, the combination of gemcitabine and cisplatin represents the standard of care for patients with locally advanced or metastatic disease, based on the results of the landmark phase III ABC-02 study and the phase II BT22 trial [22,23]. Recently, gemcitabine plus tegafur/gimeracil/oteracil (TS-1) demonstrated non-inferiority to the aforementioned doublet regimen, with acceptable tolerability, in Japanese patients with advanced biliary tract cancer (BTC) [24]. Nonetheless, more than 50% of patients will inevitably develop disease progression [25]; second-line therapy with modified 5-fluorouracil/folinic acid/oxaliplatin (FOLFOX) and active symptom control resulted in a modest OS improvement, but improved six- and 12-month survival rates, compared to active symptom control alone, according to the ABC-06 trial [26].
The parallel development of omics-based studies and novel targeted therapies have unveiled potential molecular-based treatment options for chemo-refractory disease. Indeed, more than 60% of cholangiocarcinoma patients harbour unique gene aberrations including fibroblast growth factor receptor (FGFR) 2 gene translocations, isocitrate dehydrogenase-1 (IDH1) and KRAS proto-oncogene mutations, and receptor tyrosine-protein kinase erbB-2 (ERBB2) amplification [27,28,29]. Pemigatinib is a selective, oral FGFR1-3 inhibitor and the first United States Food and Drug Administration (FDA) and European Medicines Agency (EMA) approved targeted agent for the second-line treatment of iCC [30,31,32]. Even though neurotrophic tyrosine receptor kinase (NTRK) rearrangements are found in less than 5% of BTC, they represent targetable alterations; both larotrectinib and entrectinib are FDA approved tissue agnostic drugs for NTRK fusion-positive tumors [33]. Moreover, dabrafenib plus trametinib showed promising antitumor activity in BRAF V600E-mutated BTC in the phase II, basket trial, ROAR [34], while the combination of bevacizumab and erlotinib may constitute an alternative therapeutic option for patients with epidermal growth factor receptor (EGFR)-mutated advanced BTC [35]. The compelling results of the phase III ClarIDHy trial, regarding the use of ivosidenib in IDH-mutated cholangiocarcinoma, have been recently presented at the 2021 Gastrointestinal Cancers Symposium [36].
Despite these emerging advances towards an individualized treatment plan for iCC, there still exists a paucity of efficacious therapeutic options for this highly challenging and biological heterogeneous malignancy. In recent years, immune-oncology has revolutionized the therapeutic arsenal of various solid tumors, yet its efficacy in BTC merits further investigation [37,38]. Herein, we summarize and critically discuss current evidence, challenges, and future perspectives with regards to the emerging role of immunotherapy in iCC.
2. Immunological Characterisation of iCC
Immunotherapy, consisting of (a) immune checkpoint inhibitors (ICIs) targeting programmed death 1 (PD-1), programmed death-ligand 1 (PD-L1), and cytotoxic T-lymphocyte antigen-4 (CTLA-4), (b) cancer vaccines, and (c) adoptive cell transfer (ACT), has blossomed over the last decade, leading to a dramatic advancement of cancer therapeutics [39]. Thoroughly, immune-oncology is emerging as the fifth pillar of cancer treatment, alongside surgery, radiation therapy, chemotherapy, and targeted therapies; yet its efficacy varies and only a subset of patients (approximately 10–35%) achieves durable responses [40]. Taking into account that immunotherapy aims to enhance natural, anti-tumor immune responses, including both innate cells (neutrophils, macrophages, dendritic cells, natural killers, etc.) and adaptive cells (T- and B- lymphocytes) infiltrations into the tumor microenvironment (TME) [41], it becomes prominent that the improved cellular and functional characterization of the immune landscape within TME holds tremendous clinical potential, in both predicting responsiveness to immunotherapy and in identifying novel treatment strategies.
Hence, the TME is a highly dynamic, tridimensional, sophisticated interplay comprising of cancer, stromal, and endothelial cells, which encompasses as well as an abundance of immune components, proliferative factors, vasculature, nerve fibers, extracellular matrix, and acellular components [42]. According to their cytotoxic T cell infiltration, solid tumors are classified into four distinct phenotypes, namely hot, altered immunosuppressed, altered excluded, and cold, with the first exerting the greatest benefit from ICI-based therapy and the latter representing the most challenging to treat [43,44].
Immunotherapy approaches have emerged as viable therapeutic options for iCC, albeit available data are limited to sub-analyses of either small single-arm studies or basket trials; the conflicting results, so far, highlight the unmet need for discovery and validation of predictive biomarkers [45]. In a transcriptome study which categorized 566 cases of iCC, based on their cellular TME features, 11% displayed hot phenotype, whereas 45% showed an immune desert—cold phenotype [46]. Furthermore, PD-L1-expressing iCC cells, which derived from the preinvasive intraductal papillary neoplasms of the bile duct (IPNB), have been associated with PD-L1+ mononuclear cells, PD-1+ lymphocytes, and cytotoxic CD8+ lymphocytes infiltrations within the tumor, thus with PD-1/PD-L1 axis activation, suggesting ICI efficacy [47,48].
Given that the cross-talk between tumor cells and TME, along with genetic and epigenetic alterations, dictate cholangiocarcinoma’s phenotype [49], it is of paramount importance to facilitate an in-depth knowledge of iCC’s immune contexture in order to unravel potential pathophysiological mechanisms behind these controversial responses to immunotherapy. iCC’s prominent histological hallmark is its rich desmoplastic stroma containing primarily cancer-associated fibroblasts (CAFs) and an exceedingly reactive TME of (a) innate immune cells with mainly immunosuppressive and pro-tumorigenic function, like tumor-associated macrophages (TAMs), tumor-associated neutrophils (TANs), and dendritic cells (DCs), and (b) adaptive immune cells, including tumor infiltrating lymphocytes (TILs) and natural killers (NKs) [8,50,51,52]. This complex, multicellular compartment is orchestrated by various soluble signaling mediators (chemokines, extracellular vesicles) and growth factors like platelet-derived growth factor (PDGF) [52] and contributes to iCC’s pathogenesis [8,50,51] (Figure 1).
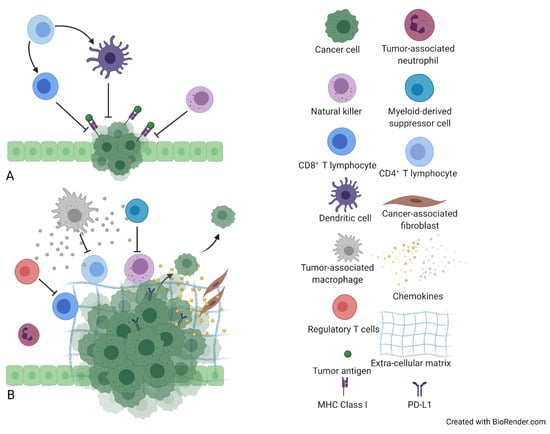
Figure 1.
Immunological features of intrahepatic cholangiocarcinoma (iCC) tumor microenvironment (TME). (A) The cross-talk between anti-tumorigenic immune cells, including tumor-infiltrating lymphocytes (TILs), dendritic cells (DCs), and natural killer cells (NKs) and tumor cells. (B) Cancer-associated fibroblasts (CAFs) and tumor-associated macrophages (TAMs) contribute to tumorigenesis, by secreting a plethora of growth factors, proteases, and cytokines. Responding to the activated chemokine axes, myeloid-derived suppressor cells (MDSCs) and regulatory T-cells (Tregs) are recruited into TME, leading to immune surveillance disruption. Moreover, extracellular matrix (ECM) has been correlated with tumor growth, whereas programmed death-ligand 1 (PD-L1) expression mediates tumor immune escape.
2.1. Cellular Components of TME
2.1.1. Cancer-Associated Fibroblasts (CAFs)
CAFs represent one of the most prominent and critical cellular components of iCC’s desmoplastic stroma [52], which originate from diverse cell populations. Potential sources of CAFs include hepatic stellate cells and portal fibroblasts, bone marrow-derived mesenchymal stem cells (MSCs), cholangiocyte-derived fibroblasts through epithelial-to-mesenchymal transition (EMT), and endothelial cells through endothelial-to-mesenchymal transition (EndMT) [52,53,54,55,56,57,58,59,60,61].
Apart from the direct tumor-promoting activities (cancer cell proliferation, survival, and migration), CAFs also mediate angiogenesis, lymphangiogenesis, and tumor-promoting inflammation via secretion of growth factors like vascular endothelial growth factor (VEGF), fibroblast growth factor (FGF), and soluble chemokines such as CXCL2, CXCL12, CXCL14, interleukin 8 (IL-8), IL-13, stromal derived factor -1 alpha (SDF-1a), and monocyte chemotactic factor 1(MCP-1) [7,56]. Moreover, CAFs elicit structural remodeling of the extracellular matrix (ECM), by the release of protein-lysine 6-oxidase (LOX), matrix metalloproteases (MMPs) like MMP1, MMP2, MMP3, MMP9, and matricellular proteins including periostin, tenascin-C, and osteopontin, thus promoting tumor growth and local invasiveness [7]. The modified, stiffer ECM serves as a barrier itself to immune cells’ invasion. It has been demonstrated that the abundant a-smooth muscle actin (a-SMA) + CAFs correlate with decreased cytotoxic T cell infiltration, improved tumor-associated macrophages (TAMs) infiltration, and poor clinical outcome [7,53,62,63]. Moreover, in a transcriptomic analysis of 10 iCCs, high VEGF expression was associated with TME polarization, restricted T cell infiltration, and poor responsiveness to immunotherapy [64].
Lastly, CAFs display immunosuppressive properties; CAFs’ pro-inflammatory role, when overexpressing fibroblast activation protein (FAP), could be sustained by CCL2-STAT3 signaling, resulting in inhibited myeloid-derived suppressor cells (MDSCs) infiltration in murine models of cholangiocarcinoma [65], whereas the interaction between CAFs and dendritic cells (DCs) dampens the activation of tumor-infiltrating lymphocytes (TILs) [66].
2.1.2. Tumor-Associated Macrophages (TAMs)
TAMs, and especially macrophages with M2 phenotype and high phagocytosis capacity, constitute the key immune cell population infiltrating TME [67,68]. Ontogenetically, hepatic macrophages can be classified as resident Kupffer cells and as recruited bone marrow-derived (or monocyte-derived) hepatic macrophages, yet their exact origin remains to be elucidated [69,70]. In iCC, the increased levels of tumor necrosis factor- alpha (TNF-α) expressed by Kupffer liver cells promotes abnormal cell proliferation and carcinogenesis, through the c-Jun N-terminal kinase (JNK) pathways activation [70,71]. The recruitment of bone marrow-derived monocytes, which differentiate into TAMs, is driven by circulating mediators released by both tumor and stromal cells, including cytokines (IL-1β, IL-10, IL-13, IL-34), osteoactivin, VEGF-A, and colony stimulating factor-1 (CSF-1) [68,72,73].
The close cross-talk between TAMs and cholangiocarcinoma cells generates a plethora of tumor-promoting phenomena. First, TAMs can exert angiogenesis via secreting pro-angiogenic factors (VEGF-A, IL-8, angiopoietin) and pro-inflammatory mediators like cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS) [74]. Second, TAMs play a pivotal role in cholangiocarcinoma cell proliferation via the activation of the Wnt/β-caterin signaling pathway [75,76]. Third, TAMs contribute to T cell immunosuppression, promoting tumor growth and metastasis through the expression of hypoxia-inducible factor 1-alpha (HIF-1α) [77].
Despite, CD68+ TAMs have been correlated with a reduced risk of iCC recurrence [78], yet high density of TAMs in cholangiocarcinoma tissues associates with poor prognosis [79].
2.1.3. Tumor-Associated Neutrophils (TANs)
Intratumoral neutrophils (TANs) are engaged into the TME through the angiogenic chemokine CXCL5, which is secreted by tumor and stroma cells via the activated phosphatidylinositol 3-kinase (PI3K)/protein kinase B (AKT) and extracellular signal-regulated kinase 1/2 (ERK1/2) pathway [80]. The presence of increased circulating neutrophil-to lymphocyte ratio (NLR) has been associated with poor clinical outcome in iCC patients [81,82], whilst CD66+ or CD15+ TANs in cholangiocarcinoma have also been correlated with worse survival [83,84,85]. TANs’ role in cholangiocarcinoma remains indefinite and merits further investigation, given their recently apparent importance in cancer progression. Indeed, neutrophil-based therapeutics is likely to become a rapidly growing field of research [86].
2.1.4. Natural Killers (NKs)
NKs, a CD3-CD56+ subset of innate lymphoid cells, account for 30–40% of liver lymphocytes [87] and are characterized by their natural cytotoxic capacity to recognize and destroy malignant cells, in an antigen nonspecific way, via cell lysis [88,89]. Besides inhibiting cell proliferation and metastasis, NKs produce cytokines (mainly interferon-γ, INF-γ), resulting in adaptive immunity modulation [90,91] and decreasing off-target effects through specific antitumor cytotoxic activity [92].
In iCC, endogenous CXCL9 expression has been closely correlated with high NKs infiltrations [93], which in turn inhibit tumor growth and reduce chemoresistance [89,94]. Jung et al., demonstrated that the infusions of human NKs in cholangiocarcinoma cell line HuCCT-1-derived xenograft mouse models induced significant tumor growth inhibition, thus highlighting the potential of NKs-based immunotherapy [89].
2.1.5. Tumor-Infiltrating Lymphocytes (TILs)
TILs comprise of highly heterogeneous immune cells, including CD8+ T cells (cytotoxic T lymphocytes, CTLs), CD4+ T cells (T helper lymphocytes, Th cells), CD20+ B lymphocytes, and CD4+CD25+ regulatory T cells (Tregs) [95]. Immunohistochemical staining of cholangiocarcinoma specimens reveals variable localization of distinct subsets of TILs within the tumor; CD8+ TILs prevail in the intratumoral region, whereas CD4+ T cells are prevalent across the peritumoral area [15,96]. CD8+ TILs preferentially accumulate in loose fibronectin and collagen peritumoral stroma and migrate poorly through dense tumor tissue, guided by chemokines, since they are not capable of producing ECM-degrading proteases [97,98]. Thus, the distribution of CD8+ lymphocytes in TME could be dictated by both density and orientation of a stroma’s structural components.
TILs confer prognostic value in BTC, as well as in other malignancies [99,100]; high numbers of CD8+ and CD4+ TILs within TME correlate with better OS, while low frequencies of CD8+ lymphocytes are associated with unfavorable prognosis [15,83,85,94,99,100,101,102,103]. As aforementioned, an elevated NRL has been related to poor outcomes in iCC [81,82] and has been correlated with higher percentage of IFN-γ+ TILs, CD8+ TILs, and PD-1+CD4+ TILs [104]. Notably, the expression of PD-L1 is more prominent on immune cells rather than on tumor cells [105,106,107,108]. In fact, PD-L1 expression on cancer cells associates with lower responses to immunotherapy [109].
Furthermore, various tumor-associated antigens (TAAs) have been identified in cholangiocarcinoma and represent attractive candidates for cancer vaccines [110]. In a recent transcriptomic study of 10 iCC cases, which revealed the variability among T cell populations within TME, CD8+ TILs from highly heterogeneous tumors exerted decreased cytotoxic capacity [64].
With regards to B TILs, their pathogenic relevance in cholangiocarcinoma has not yet been clarified, yet higher CD20+ B lymphocytes populations have been observed in a rare type of Epstein-Barr virus (EBV) infection-related iCC [111]. Moreover, elevated numbers of CD20+ B TILs have been reported in low-grade tumors and are associated with favorable prognosis [15,96].
Forkhead box P3 (FOXP3), also known as scurfin, is a transcription factor overexpressed by Tregs and associates with the up-regulation of CTLA-4 expression, which in case of iCC is predictive of tumor recurrence and chemoresistance [112]. Additionally, in the presence of CCL2 produced by CAFs, TAMs, and tumor cells, cytotoxic TILs acquire a CD4+CD25+ Tregs phenotype, which leads to secretion of the immunosuppressive transforming growth factor-beta (TGF-β) and IL-10 [113].
2.1.6. Dendritic Cells (DCs)
Along with macrophages and B cells, DCs are among the most important antigen-presenting cells (APCs), capable of providing the necessary signals for effective T cell activation into peripheral lymphoid organs [114]. Whereas immature CD1a+ DCs are found into the tumor, mature CD83+ DCs reside mainly into the invasive front [103]. In cholangiocarcinoma, the suppression of IL-10 and TGF-β receptors on self-differentiated DCs generated intensified activation of effector T cells against cancer cells [115]. In another study, the decreased number of TNF-α producing DCs in cholangiocarcinoma patients could affect DC-mediated immunity [116].
2.1.7. Myeloid-Derived Suppressor Cells (MDSCs)
MDSCs represent a major obstacle for natural antitumor immunity, as a heterogeneous group of immature myeloid progenitors which suppresses both the innate and adaptive immune responses [117]. Recruited mainly by CAFs to the TME, MDSCs exhibit suppressor activity, especially upon CD8+ T cells, by synthesizing reactive oxygen species (ROS) and by affecting the metabolism of L-arginine [65,118,119]. Extensive research regarding their role in iCC pathogenesis may permit the future development of novel immunotherapeutic agents.
2.2. Non-Cellular Components of TME
2.2.1. Extracellular Matrix (ECM)
Intense structural remodeling of the ECM through the release of MMPs, periostin, tenascin-C, and osteopontin is crucial in cholangiocarcinogenesis, as described above [7]. Indeed, the overexpression of matricellular proteins has been related to tumor growth, lymph node metastatic potential, and poor OS [120,121]. Elevated expression of osteopontin in stromal cells, in particular, has been significantly correlated with tumor size, local and distant invasion, and advanced stage, through the activation of RAS-RAF-MEK-ERK and Wnt/β-caterin signaling pathways, in BTC patients [122,123]. Moreover, osteopontin promotes NKs’ development and T cells’ survival [124,125]. Further studies will focus on the ECM therapeutic potentials in cholangiocarcinoma, including iCC.
2.2.2. Extracellular Vesicles (EVs)
EVs represent small membranous structures secreted from cells, serving as intercellular messengers [126]. They can be classified as (a) exosomes with 30–100 nm diameter, (b) microvesicles with 0.1–1 μm diameter, and (c) apoptotic bodies with >1 μm diameter which are more commonly eliminated by phagocytes immediately after their release from apoptotic cells [127]. Their components include proteins, lipids, messenger ribonucleic acids (mRNAs), microRNAs (miRNAs), long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs), which are protected from enzymatic degradation by their lipid bilayer [128].
Several miRNAs ad lncRNAs have been identified as candidates in cholangiocarcinoma tissues [129,130]. Previous study demonstrated that miRNA-15a downregulation in cholangiocarcinoma-associated CAFs promotes increased secretion of plasminogen activator inhibitor-2 (PAI-2), which in turn leads to the migration of cholangiocarcinoma cells [131]. The therapeutic significance of miRNAs has been indicated in rat models, whereas the administration of miRNA-195- enriched EVs decreased tumor size and led to increased survival rates [132].
2.2.3. Chemokines
Chemokines are a class of cytokines with approximately 8–12 kilodaltons mass and the ability to induce chemotaxis of nearby leukocytes (monocytes, macrophages, T lymphocytes, etc.) [133]. Together with their receptors, they represent emerging key factors of a well-orchestrated network of tumor-promoting events, including cancer cell migration, invasion, and immune evasion [133].
CXCL1–3, CXCL5–6, CXCL8, CXCL12, CCL2, CCL11, and CCL16 are among the various chemokines which contribute to angiogenesis, either in a direct or indirect way [134]. In cholangiocarcinoma, CXCL12 is positively modulated by angiotensin II and negatively by TGF-β, while CXCR4 expression is promoted by TNF-a-secreting TAMs [133]. Interactions between CXCR4 and CXCL12 have been demonstrated to activate the PI3K/AKT and ERK1/2 pathway, and Wnt/β-caterin signaling pathway [133]. Via the activation of the PI3K/AKT and ERK1/2 pathway, CXCL5 acts as chemoattractant for neutrophils, while FAP-induced CCL2 secretion mediates the migration of both macrophages and MDSCs [133]. Ongoing trials aim to assess the clinical utility of chemokines in patients with hematological malignancies and breast cancer [135,136].
2.2.4. PD-L1, PD-1, and CTLA-4 Expression
Manipulations of the immune checkpoints PD-1 and CTLA-4 are among the immune escape mechanisms of cancer cells and the PD-1/PD-L1 pathway seems to play a pivotal role in the development of a tumor-tolerant TME in BTC [137,138]. Indeed, high tumor cells expression of PD-L1 and PD-1, accompanied by decreased CD3+ and/or CD8+ infiltrates, associates with worse clinical outcome [99,112,139]. Nakamura et al. demonstrated an upregulation of the expression of immune checkpoint molecules in approximately 45% of BTC, which was indicative of worst prognosis [140]. Similarly, Gani et al., showed that 72% of iCC samples expressed PD-L1, which was associated with a 60%-reduction in OS, compared with negative tumor tissue samples [108]. In another study, PD-1 and PD-L1 were expressed in a total of surgically resected iCC, with PD-1 expressed only on TILs and PD-L1 expressed on 30% of tumor cells, TILS, and macrophages [141].
Moreover, FOXP3 overexpression by cholangiocarcinoma cells correlates with lymph node metastasis and poor survival [83,142]. Alongside, FOXP3 overexpression often occurs in association with CTLA-4 overexpression, which holds unfavorable prognostic value in cholangiocarcinoma [112]. Downregulation of the major histocompatibility complex-I (MHC-I) might also play a crucial role in tumor escape from immunosurveillance, and has been related to decreased TILs infiltration and poor prognosis in BTC [15].
3. Immunotherapeutic Strategies
Since 2010, immunotherapy has heralded a new era in cancer treatment. On the basis of limited therapeutic options in patients with BTC, including iCC, immunotherapeutic strategies with checkpoint inhibitors, peptide- and dendritic- based vaccines, and adoptive cell therapy, alone or in combination with targeted therapy and/or chemotherapy, have been in progress. Table 1 summarizes results from recently completed immunotherapy trials in BTC patients, including iCC, whereas Table 2 and Table 3 present ongoing trials of immunotherapeutic strategies for the treatment of BTC.

Table 1.
Reported outcomes of immunotherapy for biliary tract cancer including intrahepatic cholangiocarcinoma.

Table 2.
Ongoing trials of ICC for HCC and biliary tract cancer including intrahepatic cholangiocarcinoma.

Table 3.
Ongoing trials of TME targeted therapy and ACT for HCC and biliary tract cancer including intrahepatic cholangiocarcinoma.
3.1. Immune Checkpoint Inhibitors
CTLA-4 and PD-1 are both immune checkpoints expressed by activated T cells, which could dampen the immune response by downregulating the function of activated T cells, in order to maintain immunologic balance. Tumor cells enhance their survival potential tolerance by stimulating this negative signaling. ICIs aim to block this inhibitory signaling, thus restore immune system’s function to destroy cancer cells [143]. Ipilimumab, an anti-CTLA-4 monoclonal antibody (MAb), was the first ICI to gain FDA approval, for the treatment of melanoma, in 2011 [144]. Since then, several ICIs have been approved for cancer therapy, including the anti-PD-1 MAbs nivolumab, pembrolizumab, and cemiplimab, and the anti-PD-L1 MAbs atezolizumab, avelumab, and durvalumab [145], whereas the discovery of CTLA-4 and PD-1 blockade by James P. Allison and Tasuku Honjo, respectively, led them to the Nobel-Prize in 2018 [146].
In the case of approved ICIs for BTC, the phase II basket trial of pembrolizumab monotherapy, which demonstrated significant OS benefit and improved objective response rate (ORR) in mismatch repair (MMR)-deficient patients and led to the FDA approval of pembrolizumab for patients with inoperable or metastatic solid tumors with MMR deficiency or high microsatellite instability (MSI), included four patients with ampullary cancer or cholangiocarcinoma [147,148]. Notably, Lynch syndrome, known as hereditary non-polyposis colorectal cancer (HNPCC), is characterized by MMR deficiency and represents, despite uncommon (almost 2% of cases), a risk factor for iCC [149].
As aforementioned, PD-1 and PD-L1 are expressed in iCC tissue samples, underlying the therapeutic potential of targeting the PD-1/PD-L1 pathway [107,150,151]. Furthermore, there is growing evidence that ICIs demonstrate promising clinical efficacy in virus-associated cancers, like Hodgkin lymphoma, merkel cell carcinoma, head and neck-, and hepatocellular- cancer, probably due to the increased neo-antigen presentation [152]. Noteworthy, various oncogenic viruses have been related to iCC’s development [12,13]. Currently, several phase I-III clinical trials are underway, using ICIs alone or in combination with other chemotherapeutic modalities or targeted therapies.
The phase Ib KEYNOTE-028 and phase II KEYNOTE-158 trials, whereas pembrolizumab resulted in durable, high response rates of 40% in previously treated, advanced non-colorectal, MSI-high/MMR-deficient cancer patients, included 22 (9.4%) cholangiocarcinoma cases and retrospectively evaluated PD-L1 status [153]. Indeed, PD-L1 expression, which was defined as a combined positive score of at least 1% of tumor and inflammatory (lymphocytes, macrophages) cells, was noted in all cholangiocarcinoma tissue samples [154]. In the majority of cases, treatment-related adverse events were mild to moderate regarding severity and occurred in almost 65% of patients, while 6% of patients experienced grade 3 immune-mediated adverse events or infusion reactions [153].
Pembrolizumab demonstrated promising antitumor activity when combined with lenvatinib, an oral multiple kinase inhibitor against the VEGFR1, VEGFR2, and VEGFR3 kinases, in the ongoing phase II LEAP-005 study, which evaluated both the efficacy and safety of this regimen, among others, in 31 previously treated, advanced BTC patients [155].
A multicenter phase II trial demonstrated durable partial response in MMR-proficient, refractory BTC patients treated with nivolumab as second-line therapy. 32 (59%) patients had iCC, while PD-L1 expression predicted response to immunotherapy and correlated with prolonged progression-free survival (PFS) [156]. In the case of a 40-year-old female patient with recurrent iCC, the combination of nivolumab and lenvatinib showed promising efficacy, with the complete response in liver metastases and the stabilization of lung lesions within nine months [157]. Nivolumab has also been examined in the first-line setting, whereas its combination with gemcitabine and cisplatin resulted in a RR of 37% and a median OS of 15.4 months [158]. Durable responses have also been reported with the combination of nivolumab and ipilimumab in patients with advanced iCC, irrespective of biomarkers status, according to a subgroup analysis of the phase II CA209-538 trial [159].
The combination of chemotherapy and durvalumab, alone or in addition to tremelimumab (anti-CTLA-4 antibody), apart from being tolerable, resulted in high response- and disease control- rates in the majority of chemotherapy-naïve advanced BTC patients, in a phase II study [160]. Currently, the combination of gemcitabine and cisplatin with durvalumab is being under further investigation in the phase III TOPAZ-1 trial (NCT03875235).
M7824, also known as bintrafusp alfa, is a first-in-class bifunctional fusion protein immunotherapy, which combines a TGF-β ‘trap’ with an anti-PD-L1 immunoglobulin G (IgG) monoclonal antibody, thus simultaneously targeting two immune-suppressive pathways in the TME [161]. Data from an ongoing expansion cohort from a phase I trial (NCT02699515), with 30 patients with BTC who progressed after platinum-based first-line therapy, highlight its therapeutic potential [162], which is at present under evaluation either as monotherapy (NCT03833661) or in combination with platinum-based chemotherapy (NCT04066491).
Poly adenosine diphosphate-ribose polymerase inhibitors (PARPi) represent a novel class of targeted cancer therapy, by exploiting synthetic lethality in tumours which harbour germline and somatic alterations in DNA damage repair (DDR) genes; whereas their combination with immune-oncology appears to be a promising treatment approach for solid tumors [163]. The biological rationale behind their synergistic effects has been thoroughly determined; DDR gene aberrations have been associated with genomic instability, immunomodulation of TME, and increased tumor mutational burden (TMB) [164,165,166,167,168,169]. DDR mutations have been observed in 28.9–63.5% of BTC, with BRCA mutations occur-ring in less than 7% of these patients [170,171,172]. Currently, two early-phase trials (NCT03639935, NCT03991832) are underway to evaluate the efficacy from the combination of PARPi and ICI treatment and uncover the patient population which will potentially derive the greatest benefit from this combo.
3.2. Cancer Vaccines
TAAs, normally expressed in tumors, represent theoretically ideal targets for cancer vaccines, since they allow specific T cell responses. However, loss or downregulation of major histocompatibility complex class I molecules (MHC-I or HLA-I), by which tumor antigens are presented to CTLs, are among the T cell immunosurveillance mechanisms [173]. In iCC, derangements in HLA-I have been associated with more advanced stages, despite the underlying mechanism has not yet been elucidated [141]. Various TAAs, like Wilms’ tumor gene 1 (WT1) and mucin 1 (MUC1), have been studied as potential targets in BTC patients [174,175,176,177,178]. Both of them have been reported in approximately 80–85% of iCC cases and are of negative prognostic value [177,178].
In a Japanese open-label, dose-escalation, phase I trial, which enrolled 4 patients with iCC, the combination of WT1 peptide vaccine and gemcitabine resulted in a median OS of 9.5 months, with good tolerability [179]. The researchers now investigate in a randomized phase II study the combination of WT1 vaccine with gemcitabine and cisplatin as first-line treatment in patients with unresectable or recurrent BTC [180].
Yamamoto et al., had accrued patients with mixed gastrointestinal tract cancers (pancreatic and BTC) in a phase I trial of vaccination with MUC1 peptides and incomplete Freund’s adjuvant (Montanide ISA51), which failed to provide survival benefit [181]. Similarly, Lepisto et al. conducted a phase I/II trial of vaccination with DCs loaded with MUC1, as adjuvant therapy in 10 patients with early-stage pancreatic cancer and two with stage II iCC. One of the two iCC patients remained free of recurrence [182]. These small-scale trials proved the satisfactory profile of the vaccines, even if the survival data were rather inconclusive.
Promising results have been obtained by a case report of a female patient with advanced iCC, repeatedly operated for disease recurrences, whereas her immunization with a personalized multi-peptide vaccine, targeting tumor mutations presented by HLA-I, conferred durable response [183]. Another phase II trial of personalized peptide vaccine evaluated the feasibility of HLA-matched vaccine peptides in six iCC patients and proved that low levels of IL-6 were significantly associated with improved OS (hazard ratio = 1.123; 95% CI 1.008–1.252; p = 0.035) [184]. Based on this result, the same group had started an early phase trial to examine whether blockade of IL-6-mediated inflammation with tocilizumab could enhance the immune responses after personalized peptide vaccination in advanced BTC patients [185,186].
In a single-arm phase II trial, combination of gemcitabine plus elpamotide (an HLA-A* 24:02-restricted epitope peptide of VEGFR-2, which induces CTL responses) in 54 patients with unresectable or recurrent BTC demonstrated moderate antitumor effect, response rate of 18.5%, and median OS of 10.1 months in comparison with the 7.6 months of the historical control [187].
Tumor lysate-based DCs cancer vaccines remain under clinical investigation, yet early efficacy has been demonstrated in in-vitro studies [188]. In an early phase trial, the autologous tumor lysate-pulsed DCs injection and the following transfer of CD3-activated T cells in postoperative patients with iCC led to improved PFS and OS, compared to the control group who received surgery alone (18.3 versus 7.7 months; p = 0.005 and 31.9 versus 17.4 months; p = 0.022, respectively) [189]. We still await the results of two recently completed phase I clinical trials for DC-based immunotherapy in BTC (NCT00027534, NCT00004604).
3.3. Adoptive Cell Therapy (ACT)
Adoptive cell therapy with (a) autologous TILs infusion or (b) genetically modified T cells to express either T cell receptor (TCR) or chimeric antigen receptor (CAR) represent another therapeutic strategy to manipulate the immune system to recognize and destroy tumor cells, with undeniable results seen in melanoma patients [190]. In the case of BTC, ACT has been limited to small case-series, case reports, and single-arm early phase studies.
Back in 2006, a patient with operable, node-positive iCC received adjuvant CD3-activated T cells and tumor lysate- or peptide-pulsed DCs, and survived 3.5 years [191]. ACT of TILs containing CD4+ erbb2 interacting protein (ERBB2IP) mutation-reactive T cells generated promising results in a patient with metastatic cholangiocarcinoma; the patient achieved a 30% decrease in tumor lesions, with prolonged disease stabilization of 13 months, while the rechallenge with a second T cell infusion resulted in further response [192]. Recently, a phase I trial demonstrated encouraging results of clinical activity of CAR-T cell immunotherapy targeting human epidermal growth factor receptor 2 (HER2) in 11 patients with advanced BTC and pancreatic cancers [193].
4. Conclusions
The role of immunotherapy in iCC is currently investigational and the results of ongoing studies are eagerly anticipated. Based on promising results from several solid tumors clinical trials, it will hopefully increment patient outcomes in cases of advanced disease. This rapidly transformational landscape of immune-oncology in cholangiocarcinoma poses several challenges. In order to consider this advancing therapeutic strategy in the exquisitely heterogeneous iCC, a deeper, rudimentary, understanding of the cellular diversity of its constantly evolving TME is fairly crucial. The milieu of tumor and immune cells, along with vasculature, extracellular matrix, and signaling molecules regulate immune responses and influence immunotherapy’s efficacy. The modulation of the crosstalk between iCC and TME, by targeting the activation of these cells, in combination with ICIs represents an attractive therapeutic prospective [194,195].
The next frontier of this emerging field is the development of combinational therapeutic approaches. Such approaches, constituting of ICIs along with chemotherapy and/or targeted therapy, have garnered clinical research interest, as they represent appealing strategies for enhanced efficacy, by overcoming both primary and acquired resistance. The rational design of preclinical research to benchmark the drug combinations, side-by-side, against monotherapies and/or other combinations will confer important insights into the degree of synergy [196]. Last but not least, the lack of validated predictive biomarkers, in order to determine the patients’ sub-cohorts which will derive benefit from immunotherapy, represents a crucial challenge and should be underscored. To date, no single biomarker, including TME, MMR/MSI status, TMB, and PD-L1, has proved efficient to predict response to immune-targeted therapies and guide treatment decisions in iCC clinical settings [45].
Though representing a relatively young field, immunotherapy in iCC is burgeoning, with several gaps that need to be filled. It is of the utmost importance to fully decipher the underpinnings of the optimal antitumor immune response, at both cellular and molecular levels, in the effort to optimize its efficacy against this challenging malignancy.
Author Contributions
Conceptualization, M.L. and F.Z.; writing—original draft preparation, O.F. and I.N.-S.; writing—review and editing, M.G., M.L., K.K., M.A.D. and F.Z. All authors have read and agreed to the published version of the manuscript.
Funding
This review article received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Komuta, M.; Yeh, M.M. A Review on the Update of Combined Hepatocellular Cholangiocarcinoma. Semin. Liver Dis. 2020, 40, 124–130. [Google Scholar] [CrossRef]
- Patel, T. Increasing incidence and mortality of primary intrahepatic cholangiocarcinoma in the United States. Hepatology 2001, 33, 1353–1357. [Google Scholar] [CrossRef]
- Stavraka, C.; Rush, H.; Ross, P. Combined hepatocellular cholangiocarcinoma (cHCC-CC): An update of genetics, molecular biology, and therapeutic interventions. J. Hepatocell. Carcinoma 2018, 6, 11–21. [Google Scholar] [CrossRef]
- Leoni, S.; Sansone, V.; Lorenzo, S.; Ielasi, L.; Tovoli, F.; Renzulli, M.; Golfieri, R.; Spinelli, D.; Piscaglia, F. Treatment of Combined Hepatocellular and Cholangiocarcinoma. Cancers 2020, 12, 794. [Google Scholar] [CrossRef]
- Shaib, Y.H.; A Davila, J.; McGlynn, K.; El-Serag, H.B. Rising incidence of intrahepatic cholangiocarcinoma in the United States: A true increase? J. Hepatol. 2004, 40, 472–477. [Google Scholar] [CrossRef]
- West, J.; Wood, H.; A Logan, R.F.; Quinn, M.; Aithal, G.P. Trends in the incidence of primary liver and biliary tract cancers in England and Wales 1971–2001. Br. J. Cancer 2006, 94, 1751–1758. [Google Scholar] [CrossRef]
- Sirica, A.E. The role of cancer-associated myofibroblasts in intrahepatic cholangiocarcinoma. Nat. Rev. Gastroenterol. Hepatol. 2011, 9, 44–54. [Google Scholar] [CrossRef]
- Sirica, A.E.; Gores, G.J.; Groopman, J.D.; Selaru, F.M.; Strazzabosco, M.; Wang, X.W.; Zhu, A.X. Intrahepatic Cholangiocarcinoma: Continuing Challenges and Translational Advances. Hepatology 2019, 69, 1803–1815. [Google Scholar] [CrossRef] [PubMed]
- Brivio, S.; Cadamuro, M.; Strazzabosco, M.; Fabris, L. Tumor reactive stroma in cholangiocarcinoma: The fuel behind cancer aggressiveness. World J. Hepatol. 2017, 9, 455–468. [Google Scholar] [CrossRef]
- Gentilini, A.; Pastore, M.; Marra, F.; Raggi, C. The Role of Stroma in Cholangiocarcinoma: The Intriguing Interplay between Fibroblastic Component, Immune Cell Subsets and Tumor Epithelium. Int. J. Mol. Sci. 2018, 19, 2885. [Google Scholar] [CrossRef]
- Minicozzi, P.; Cassetti, T.; Vener, C.; Sant, M. Analysis of incidence, mortality and survival for pancreatic and biliary tract cancers across Europe, with assessment of influence of revised European age standardisation on estimates. Cancer Epidemiol. 2018, 55, 52–60. [Google Scholar] [CrossRef]
- Massarweh, N.N.; El-Serag, H.B. Epidemiology of Hepatocellular Carcinoma and Intrahepatic Cholangiocarcinoma. Cancer Control 2017, 24, 1073274817729245. [Google Scholar] [CrossRef]
- Clements, O.; Eliahoo, J.; Kim, J.U.; Taylor-Robinson, S.D.; Khan, S.A. Risk factors for intrahepatic and extrahepatic cholan-giocarcinoma: A systematic review and meta-analysis. J. Hepatol. 2020, 72, 95–103. [Google Scholar] [CrossRef]
- Personeni, N.; Lleo, A.; Pressiani, T.; Colapietro, F.; Openshaw, M.R.; Stavraka, C.; Pouptsis, A.; Pinato, D.J.; Rimassa, L. Biliary Tract Cancers: Molecular Heterogeneity and New Treatment Options. Cancers 2020, 12, 3370. [Google Scholar] [CrossRef]
- Goeppert, B.; Frauenschuh, L.; Zucknick, M.; Stenzinger, A.; Andrulis, M.; Klauschen, F.; Joehrens, K.; Warth, A.; Renner, M.; Mehrabi, A.; et al. Prognostic impact of tumour-infiltrating immune cells on biliary tract cancer. Br. J. Cancer 2013, 109, 2665–2674. [Google Scholar] [CrossRef]
- Wu, L.; Tsilimigras, D.I.; Paredes, A.Z.; Mehta, R.; Hyer, J.M.; Merath, K.; Sahara, K.; Bagante, F.; Beal, E.W.; Shen, F.; et al. Trends in the Incidence, Treatment and Outcomes of Patients with Intrahepatic Cholangiocarcinoma in the USA: Facility Type is Associated with Margin Status, Use of Lymphadenectomy and Overall Survival. World J. Surg. 2019, 43, 1777–1787. [Google Scholar] [CrossRef]
- Mazzaferro, V.; Gorgen, A.; Roayaie, S.; dit Busset, M.D.; Sapisochin, G. Liver resection and transplantation for intrahepatic cholangiocarcinoma. J. Hepatol. 2020, 72, 364–377. [Google Scholar] [CrossRef]
- Shroff, R.T.; Kennedy, E.B.; Bachini, M.; Bekaii-Saab, T.; Crane, C.; Edeline, J.; El-Khoueiry, A.; Feng, M.; Katz, M.H.; Primrose, J.; et al. Adjuvant Therapy for Resected Biliary Tract Cancer: ASCO Clinical Practice Guideline. J. Clin. Oncol. 2019, 37, 1015–1027. [Google Scholar] [CrossRef]
- Primrose, J.N.; Fox, R.P.; Palmer, D.H.; Malik, H.Z.; Prasad, R.; Mirza, D.; Anthony, A.; Corrie, P.; Falk, S.; Finch-Jones, M.; et al. Capecitabine compared with observation in resected biliary tract cancer (BILCAP): A randomised, controlled, multicentre, phase 3 study. Lancet Oncol. 2019, 20, 663–673. [Google Scholar] [CrossRef]
- Ntanasis-Stathopoulos, I.; Tsilimigras, D.I.; Gavriatopoulou, M.; Schizas, D.; Pawlik, T.M. Cholangiocarcinoma: Investigations into pathway-targeted therapies. Expert Rev. Anticancer. Ther. 2020, 20, 765–773. [Google Scholar] [CrossRef]
- Sharma, A.; Dwary, A.D.; Mohanti, B.K.; Deo, S.V.; Pal, S.; Sreenivas, V.; Raina, V.; Shukla, N.K.; Thulkar, S.; Garg, P.; et al. Best supportive care compared with chemotherapy for unresectable gall bladder cancer: A randomized controlled study. J. Clin. Oncol. 2010, 28, 4581–4586. [Google Scholar] [CrossRef]
- Valle, J.; Wasan, H.; Palmer, D.H.; Cunningham, D.; Anthoney, A.; Maraveyas, A.; Madhusudan, S.; Iveson, T.; Hughes, S.; Pereira, S.P.; et al. Cisplatin plus Gemcitabine versus Gemcitabine for Biliary Tract Cancer. N. Engl. J. Med. 2010, 362, 1273–1281. [Google Scholar] [CrossRef] [PubMed]
- Okusaka, T.; Nakachi, K.; Fukutomi, A.; Mizuno, N.; Ohkawa, S.; Funakoshi, A.; Nagino, M.; Kondo, S.; Nagaoka, S.; Funai, J.; et al. Gemcitabine alone or in combination with cisplatin in patients with biliary tract cancer: A comparative multicentre study in Japan. Br. J. Cancer 2010, 103, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Morizane, C.; Okusaka, T.; Mizusawa, J.; Katayama, H.; Ueno, M.; Ikeda, M.; Ozaka, M.; Okano, N.; Sugimori, K.; Fukutomi, A.; et al. Combination gemcitabine plus S-1 versus gemcitabine plus cisplatin for advanced/recurrent biliary tract cancer: The FUGA-BT (JCOG1113) randomized phase III clinical trial. Ann. Oncol. 2019, 30, 1950–1958. [Google Scholar] [CrossRef] [PubMed]
- Malenica, I.; Donadon, M.; Lleo, A. Molecular and Immunological Characterization of Biliary Tract Cancers: A Paradigm Shift towards a Personalized Medicine. Cancers 2020, 12, 2190. [Google Scholar] [CrossRef]
- Lamarca, A.; Palmer, D.H.; Wasan, H.S.; Ross, P.J.; Ma, Y.T.; Arora, A.; Falk, S.; Gillmore, R.; Wadsley, J.; Patel, K.; et al. ABC-06. A randomised phase III, multi-centre, open-label study of active symptom control (ASC) alone or ASC with oxaliplatin/5-FU chemotherapy (ASC + mFOLFOX) for patients (pts) with locally advanced/metastatic biliary tract cancers (ABC) previously-treated with cisplatin/gemcitabine (CisGem) chemotherapy. J. Clin. Oncol. 2019, 37 (Suppl. 15), 4003. [Google Scholar]
- Jusakul, A.; Cutcutache, I.; Yong, C.H.; Lim, J.Q.; Ni Huang, M.; Padmanabhan, N.; Nellore, V.; Kongpetch, S.; Ng, A.W.T.; Ng, L.M.; et al. Whole-Genome and Epigenomic Landscapes of Etiologically Distinct Subtypes of Cholangiocarcinoma. Cancer Discov. 2017, 7, 1116–1135. [Google Scholar] [CrossRef]
- Farshidfar, F.; Zheng, S.; Gingras, M.-C.; Newton, Y.; Shih, J.; Robertson, A.G.; Hinoue, T.; Hoadley, K.A.; Gibb, E.A.; Roszik, J.; et al. Integrative Genomic Analysis of Cholangiocarcinoma Identifies Distinct IDH-Mutant Molecular Profiles. Cell Rep. 2017, 18, 2780–2794. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Research Network. Electronic address: wheeler@bcm.edu; Cancer Genome Atlas Research Network. Comprehensive and Integrative Genomic Characterization of Hepatocellular Carcinoma. Cell 2017, 169, 1327–1341. [Google Scholar] [CrossRef]
- Available online: www.fda.gov/drugs/resources-information-approved-drugs/fda-grants-accelerated-approval-pemigatinib-cholangiocarcinoma-fgfr2-rearrangement-or-fusion (accessed on 17 April 2020).
- Available online: www.ema.europa.eu/en/medicines/human/summaries-opinion/pemazyre (accessed on 28 January 2021).
- Abou-Alfa, G.K.; Sahai, V.; Hollebecque, A.; Vaccaro, F.; Melisi, D.; Al-Rajabi, R.; Paulson, A.S.; Borad, M.J.; Gallinson, D.; Murphu, A.G.; et al. Pemigatinib for previously treated, locally advanced or metastatic cholangiocarcinoma: A multicentre, open-label, phase 2 study. Lancet Oncol. 2020, 21, 671–684. [Google Scholar] [CrossRef]
- Drilon, A.; Laetsch, T.W.; Kummar, S.; DuBois, S.G.; Lassen, U.N.; Demetri, G.D.; Nathenson, M.; Doebele, R.C.; Farago, A.F.; Pappo, A.S.; et al. Efficacy of Larotrectinib in TRK fusion-positive cancers in adults and children. N. Engl. J. Med. 2018, 378, 731–739. [Google Scholar] [CrossRef]
- Subbiah, V.; Lassen, U.; Élez, E.; Italiano, A.; Curigliano, G.; Javle, M.; de Braud, F.; Prager, G.W.; Greil, R.; Stein, A.; et al. Dabrafenib plus trametinib in patients with BRAFV600E-mutated biliary tract cancer (ROAR): A phase 2, open-label, single-arm, multicentre basket trial. Lancet Oncol. 2020, 21, 1234–1243. [Google Scholar] [CrossRef]
- Lubner, S.J.; Mahoney, M.R.; Kolesar, J.L.; LoConte, N.K.; Kim, G.P.; Pitot, H.C.; Philip, P.A.; Picus, J.; Yong, W.-P.; Horvath, L.; et al. Report of a Multicenter Phase II Trial Testing a Combination of Biweekly Bevacizumab and Daily Erlotinib in Patients with Unresectable Biliary Cancer: A Phase II Consortium Study. J. Clin. Oncol. 2010, 28, 3491–3497. [Google Scholar] [CrossRef] [PubMed]
- Zhu, A.X.; Macarulla, T.; Javle, M.M.; Kelley, R.K.; Lubner, S.J.; Adeva, J.; Cleary, J.M.; Catenacci, D.V.; Borad, M.J.; Bridgewater, J.A.; et al. Final results from ClarIDHy, a global, phase III, randomized, double-blind study of ivosidenib (IVO) versus placebo (PBO) in patients (pts) with previously treated cholangiocarcinoma (CCA) and an isocitrate dehydrogenase 1 (IDH1) mutation. J. Clin. Oncol. 2021, 39, 266. [Google Scholar] [CrossRef]
- Vogel, A.; Bathon, M.; Saborowski, A. Immunotherapies in clinical development for biliary tract cancer. Expert Opin. Investig. Drugs 2020, 31, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, A.; Ricci, A.D.; Brandi, G. Recent advances of immunotherapy for biliary tract cancer. Expert Rev. Gastroenterol. Hepatol. 2021, 8, 1–10. [Google Scholar] [CrossRef]
- Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 2020, 11, 3801. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Friedman, C.F.; Wolchok, J.D. Combinatorial Cancer Immunotherapies. Adv. Immunol. 2016, 130, 251–277. [Google Scholar] [CrossRef]
- Koury, J.; Lucero, M.; Cato, C.; Chang, L.; Geiger, J.; Henry, D.; Hernandez, J.; Hung, F.; Kaur, P.; Teskey, G.; et al. Immunotherapies: Exploiting the Immune System for Cancer Treatment. J. Immunol. Res. 2018, 2018, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.-Z.; Jin, W.-L. The updated landscape of tumor microenvironment and drug repurposing. Signal Transduct. Target. Ther. 2020, 5, 1–16. [Google Scholar] [CrossRef]
- Li, J.; Byrne, K.T.; Yan, F.; Yamazoe, T.; Chen, Z.; Baslan, T.; Richman, L.P.; Lin, J.H.; Sun, Y.H.; Rech, A.J.; et al. Tumor Cell-Intrinsic Factors Underlie Heterogeneity of Immune Cell Infiltration and Response to Immunotherapy. Immunity 2018, 49, 178–193.e7. [Google Scholar] [CrossRef]
- Galon, J.; Bruni, D. Approaches to treat immune hot, altered and cold tumours with combination immunotherapies. Nat. Rev. Drug Discov. 2019, 18, 197–218. [Google Scholar] [CrossRef]
- Rizzo, A.; Ricci, A.D.; Brandi, G. PD-L1, TMB, MSI, and Other Predictors of Response toImmune Checkpoint Inhibitors in Biliary Tract Cancer. Cancers 2021, 13, 558. [Google Scholar] [CrossRef] [PubMed]
- Job, S.; Rapoud, D.; Dos Santos, A.; Gonzalez, P.; Desterke, C.; Pascal, G.; Elarouci, N.; Ayadi, M.; Adam, R.; Azoulay, D.; et al. Identification of Four Immune Subtypes Characterized by Distinct Composition and Functions of Tumor Microenvironment in Intrahepatic Cholangiocarcinoma. Hepatology 2020, 72, 965–981. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Kinoshita, M.; Takemura, S.; Tanaka, S.; Hamano, G.; Nakamori, S.; Fujikawa, M.; Sugawara, Y.; Yamamoto, T.; Arimoto, A.; et al. The PD-1/PD-L1 axis may be aberrantly activated in occupational cholangiocarcinoma. Pathol. Int. 2017, 67, 163–170. [Google Scholar] [CrossRef]
- Sato, Y.; Tanaka, S.; Kinoshita, M.; Takemura, S.; Shinkawa, H.; Kokudo, T.; Hasegawa, K.; Tanaka, H.; Yoshimoto, H.; Mori, A.; et al. Immunosuppressive tumor microenvironment in occupational cholangiocarcinoma: Supportive evidence for the efficacy of immune checkpoint inhibitor therapy. J. Hepato-Biliary-Pancreatic Sci. 2020, 27, 860–869. [Google Scholar] [CrossRef] [PubMed]
- Banales, J.M.; Marin, J.J.G.; Lamarca, A.; Rodrigues, P.M.; Khan, S.A.; Roberts, L.R.; Cardinale, V.; Carpino, G.; Andersen, J.B.; Braconi, C.; et al. Cholangiocarcinoma 2020: The next horizon in mechanisms and management. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Fabris, L.; Perugorria, M.J.; Mertens, J.; Björkström, N.K.; Cramer, T.; Lleo, A.; Solinas, A.; Sänger, H.; Lukacs-Kornek, V.; Moncsek, A.; et al. The tumour microenvironment and immune milieu of cholangiocarcinoma. Liver Int. 2019, 39 (Suppl. 1), 63–78. [Google Scholar] [CrossRef] [PubMed]
- Cadamuro, M.; Brivio, S.; Spirli, C.; Joplin, R.E.; Strazzabosco, M.; Fabris, L. Autocrine and Paracrine Mechanisms Promoting Chemoresistance in Cholangiocarcinoma. Int. J. Mol. Sci. 2017, 18, 149. [Google Scholar] [CrossRef]
- Sirica, A.E.; Campbell, D.J.; Dumur, C.I. Cancer-associated fibroblasts in intrahepatic cholangiocarcinoma. Curr. Opin. Gastroenterol. 2011, 27, 276–284. [Google Scholar] [CrossRef]
- Chuaysri, C.; Thuwajit, P.; Paupairoj, A.; Chau-In, S.; Suthiphongchai, T.; Thuwajit, C. Alpha-smooth muscle actin-positive fibroblasts promote biliary cell proliferation and correlate with poor survival in cholangiocarcinoma. Oncol. Rep. 2009, 21, 957–969. [Google Scholar] [PubMed]
- Quail, D.F.; Joyce, J.A. Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 2013, 19, 1423–1437. [Google Scholar] [CrossRef] [PubMed]
- Cadamuro, M.; Stecca, T.; Brivio, S.; Mariotti, V.; Fiorotto, R.; Spirli, C.; Strazzabosco, M.; Fabris, L. The deleterious interplay between tumor epithelia and stroma in cholangiocarcinoma. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2018, 1864, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Rimassa, L.; Personeni, N.; Aghemo, A.; Lleo, A. The immune milieu of cholangiocarcinoma: From molecular pathogenesis to precision medicine. J. Autoimmun. 2019, 100, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Vaquero, J.; Aoudjehane, L.; Fouassier, L. Cancer-associated fibroblasts in cholangiocarcinoma. Curr. Opin. Gastroenterol. 2020, 36, 63–69. [Google Scholar] [CrossRef]
- Vaquero, J.; Guedj, N.; Clapéron, A.; Ho-Bouldoires, T.H.N.; Paradis, V.; Fouassier, L. Epithelial-mesenchymal transition in cholangiocarcinoma: From clinical evidence to regulatory networks. J. Hepatol. 2017, 66, 424–441. [Google Scholar] [CrossRef] [PubMed]
- Okabe, H.; Beppu, T.; Hayashi, H.; Horino, K.; Masuda, T.; Komori, H.; Ishikawa, S.; Watanabe, M.; Takamori, H.; Iyama, K.; et al. Hepatic stellate cells may relate to progression of intrahepatic cholangiocarcinoma. Ann. Surg. Oncol. 2009, 16, 2555–2564. [Google Scholar] [CrossRef] [PubMed]
- Scholten, D.; Österreicher, C.H.; Scholten, A.; Iwaisako, K.; Gu, G.; Brenner, D.A.; Kisseleva, T. Genetic Labeling Does Not Detect Epithelial-to-Mesenchymal Transition of Cholangiocytes in Liver Fibrosis in Mice. Gastroenterology 2010, 139, 987–998. [Google Scholar] [CrossRef]
- Chu, A.S.; Diaz, R.; Hui, J.J.; Yanger, K.; Zong, Y.; Alpini, G.; Stanger, B.Z.; Wells, R.G. Lineage tracing demonstrates no evidence of cholangiocyte epithelial-to-mesenchymal transition in murine models of hepatic fibrosis. Hepatology 2011, 53, 1685–1695. [Google Scholar] [CrossRef]
- Høgdall, D.; Lewinska, M.; Andersen, J.B. Desmoplastic Tumor Microenvironment and Immunotherapy in Cholangiocarcinoma. Trends Cancer 2018, 4, 239–255. [Google Scholar] [CrossRef]
- Acerbi, I.; Cassereau, L.; Dean, I.; Shi, Q.; Au, A.; Park, C.; Chen, Y.Y.; Liphardt, J.; Hwang, E.S.; Weaver, V.M. Human breast cancer invasion and aggression correlates with ECM stiffening and immune cell infiltration. Integr. Biol. 2015, 7, 1120–1134. [Google Scholar] [CrossRef]
- Ma, L.; Hernandez, M.O.; Zhao, Y.; Mehta, M.; Tran, B.; Kelly, M.; Rae, Z.; Hernandez, J.M.; Davis, J.L.; Martin, S.P.; et al. Tumor Cell Biodiversity Drives Microenvironmental Reprogramming in Liver Cancer. Cancer Cell 2019, 36, 418–430. [Google Scholar] [CrossRef]
- Yang, X.; Lin, Y.; Shi, Y.; Li, B.; Liu, W.; Dang, Y.; Chu, Y.; Fan, J.; He, R. FAP promotes immunosuppression by cancer-associated fibroblasts in the tumor microenvironment via STAT3-CCL2 signaling. Cancer Res. 2016, 76, 4124–4135. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.-T.; Deng, Y.-N.; Yi, H.-M.; Wang, G.-Y.; Fu, B.-S.; Chen, W.-J.; Liu, W.; Tai, Y.; Peng, Y.-W.; Zhang, Q. Hepatic carcinoma-associated fibroblasts induce IDO-producing regulatory dendritic cells through IL-6-mediated STAT3 activation. Oncogenesis 2016, 5, e198. [Google Scholar] [CrossRef]
- Thanee, M.; Loilome, W.; Techasen, A.; Namwat, N.; Boonmars, T.; Pairojkul, C.; Yonhvanit, P. Quantitative changes in tumor-associated M2 macrophages characterize cholangiocarcinoma and their association with metastasis. Asian Pac. J. Cancer Prev. 2015, 16, 3043–3050. [Google Scholar] [CrossRef] [PubMed]
- Hasita, H.; Komohara, Y.; Okabe, H.; Masuda, T.; Ohnishi, K.; Lei, X.F.; Beppu, T.; Baba, H.; Takeya, M. Significance of alternatively activated macrophages in patients with intrahepatic cholangiocarcinoma. Cancer Sci. 2010, 101, 1913–1919. [Google Scholar] [CrossRef]
- Sato, K.; Hall, C.; Glaser, S.; Francis, H.; Meng, F.; Alpini, G. Pathogenesis of Kupffer Cells in Cholestatic Liver Injury. Am. J. Pathol. 2016, 186, 2238–2247. [Google Scholar] [CrossRef]
- Yuan, D.; Huang, S.; Berger, E.; Liu, L.; Gross, N.; Heinzmann, F.; Ringelhan, M.; Connor, T.O.; Stadler, M.; Meister, M.; et al. Kupffer Cell-Derived Tnf Triggers Cholangiocellular Tumorigenesis through JNK due to Chronic Mitochondrial Dysfunction and ROS. Cancer Cell 2017, 31, 771–789.e6. [Google Scholar] [CrossRef]
- Sato, K.; Meng, F.; Giang, T.; Glaser, S.; Alpini, G. Mechanisms of cholangiocyte responses to injury. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2018, 1864, 1262–1269. [Google Scholar] [CrossRef]
- Mantovani, A.; Germano, G.; Marchesi, F.; Locatelli, M.; Biswas, S.K. Cancer-promoting tumor-associated macrophages: New vistas and open questions. Eur. J. Immunol. 2011, 41, 2522–2525. [Google Scholar] [CrossRef] [PubMed]
- Raggi, C.; Correnti, M.; Sica, A.; Andersen, J.B.; Cardinale, V.; Alvaro, D.; Chiorino, G.; Forti, E.; Glaser, S.; Alpini, G.; et al. Cholangiocarcinoma stem-like subset shapes tumor-initiating niche by educating associated macrophages. J. Hepatol. 2017, 66, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Henze, A.-T.; Mazzone, M. The impact of hypoxia on tumor-associated macrophages. J. Clin. Investig. 2016, 126, 3672–3679. [Google Scholar] [CrossRef]
- Boulter, L.; Guest, R.V.; Kendall, T.J.; Wilson, D.H.; Wojtacha, D.; Robson, A.J.; Ridgway, R.A.; Samuel, K.; Van Rooijen, N.; Barry, S.T.; et al. WNT signaling drives cholangiocarcinoma growth and can be pharmacologically inhibited. J. Clin. Investig. 2015, 125, 1269–1285. [Google Scholar] [CrossRef]
- Loilome, W.; Bungkanjana, P.; Techasen, A.; Namwat, N.; Yongvanit, P.; Puapairoj, A.; Khuntikeo, N.; Riggins, G.J. Activated macrophages promote Wnt/beta-catenin signaling in cholangiocarcinoma cells. Tumour Biol. 2014, 35, 5357–5367. [Google Scholar] [CrossRef]
- Thongchot, S.; Yongvanit, P.; Loilome, W.; Seubwai, W.; Phunicom, K.; Tassaneeyakul, W.; Pairojkul, C.; Promkotra, W.; Techasen, A.; Namwat, N. High Expression of HIF-1α, BNIP3 and PI3KC3: Hypoxia-Induced Autophagy Predicts Cholangiocarcinoma Survival and Metastasis. Asian Pac. J. Cancer Prev. 2014, 15, 5873–5878. [Google Scholar] [CrossRef]
- Atanasov, G.; Dietel, C.; Feldbrügge, L.; Benzing, C.; Krenzien, F.; Brandl, A.; Mann, E.; Englisch, J.P.; Schierle, K.; Robson, S.C.; et al. Tumor necrosis and infiltrating macrophages predict survival after curative resection for cholangiocarcinoma. OncoImmunology 2017, 6, e1331806. [Google Scholar] [CrossRef]
- Wongkham, S.; Subimerb, C.; Pinlaor, S.; Khuntikeo, N.; Leelayuwat, C.; Morris, A.; McGrath, M.S. Tissue invasive macrophage density is correlated with prognosis in cholangiocarcinoma. Mol. Med. Rep. 2010, 3, 597–605. [Google Scholar] [CrossRef]
- Zhou, S.L.; Dai, Z.; Zhou, Z.J.; Chen, Q.; Wang, Z.; Xiao, Y.S.; Hu, Z.Q.; Huang, X.Y.; Yang, G.H.; Shi, Y.H.; et al. CXCL5 contributes to tumor metastasis and recurrence of intrahepatic cholangiocarcinoma by recruiting infiltrative intratumoral neutrophils. Carcinogenesis 2014, 35, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Buettner, S.; Spolverato, G.; Kimbrough, C.W.; Alexandrescu, S.; Marques, H.P.; Lamelas, J.; Aldrighetti, L.; Gamblin, T.C.; Maithel, S.K.; Pulitano, C.; et al. The impact of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio among patients with intrahepatic cholangiocarcinoma. Surgery 2018, 164, 411–418. [Google Scholar] [CrossRef] [PubMed]
- Kitano, Y.; Yamashita, Y.-I.; Yamamura, K.; Arima, K.; Kaida, T.; Miyata, T.; Nakagawa, S.; Mima, K.; Imai, K.; Hashimoto, D.; et al. Effects of Preoperative Neutrophil-to-Lymphocyte and Platelet-to-Lymphocyte Ratios on Survival in Patients with Extrahepatic Cholangiocarcinoma. Anticancer Res. 2017, 37, 3229–3237. [Google Scholar] [CrossRef]
- Kitano, Y.; Okabe, H.; Yamashita, Y.-I.; Nakagawa, S.; Saito, Y.; Umezaki, N.; Tsukamoto, M.; Yamao, T.; Yamamura, K.; Arima, K.; et al. Tumour-infiltrating inflammatory and immune cells in patients with extrahepatic cholangiocarcinoma. Br. J. Cancer 2018, 118, 171–180. [Google Scholar] [CrossRef]
- Gu, F.-M.; Gao, Q.; Shi, G.-M.; Zhang, X.; Wang, J.; Jiang, J.-H.; Wang, X.-Y.; Shi, Y.-H.; Ding, Z.-B.; Fangming, G.; et al. Intratumoral IL-17+ Cells and Neutrophils show Strong Prognostic Significance in Intrahepatic Cholangiocarcinoma. Ann. Surg. Oncol. 2012, 19, 2506–2514. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.Y.; Zhu, G.Q.; Xiong, M.; Ren, L.; Bai, L. Prognostic value of neutrophil distribution in cholangiocarcinoma. World J. Gastroenterol. 2015, 21, 4961–4968. [Google Scholar] [CrossRef] [PubMed]
- Shaul, M.E.; Fridlender, Z.G. Tumour-associated neutrophils in patients with cancer. Nat. Rev. Clin. Oncol. 2019, 16, 601–620. [Google Scholar] [CrossRef] [PubMed]
- Björkström, N.K.; Ljunggren, H.-G.; Michaëlsson, J. Emerging insights into natural killer cells in human peripheral tissues. Nat. Rev. Immunol. 2016, 16, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Morvan, M.G.; Lanier, L.L. NK cells and cancer: You can teach innate cells new tricks. Nat. Rev. Cancer 2016, 16, 7–19. [Google Scholar] [CrossRef]
- Jung, I.H.; Kim, D.H.; Yoo, D.K.; Baek, S.Y.; Jeong, S.H.; Jung, D.E.; Park, S.W.; Chung, Y.Y. In vivo study of natural killer (NK) cell cytotoxicity against cholangiocarcinoma in a nude mouse model. Vivo 2018, 32, 771–781. [Google Scholar] [CrossRef]
- Luetke-Eversloh, M.; Cicek, B.B.; Siracusa, F.; Thom, J.T.; Hamann, A.; Frischbutter, S.; Baumgrass, R.; Chang, H.D.; Thiel, A.; Dong, J.; et al. NK cells gain higher IFN-gamma competence during terminal differentiation. Eur. J. Immunol. 2014, 44, 2074–2084. [Google Scholar] [CrossRef]
- Vivier, E.; Raulet, D.H.; Moretta, A.; Caligiuri, M.A.; Zitvogel, L.; Lanier, L.L.; Yokoyama, W.M.; Ugolini, S. Innate or Adaptive Immunity? The Example of Natural Killer Cells. Science 2011, 331, 44–49. [Google Scholar] [CrossRef]
- Guillerey, C.; Huntington, N.D.; Smyth, C.G.M.J. Targeting natural killer cells in cancer immunotherapy. Nat. Immunol. 2016, 17, 1025–1036. [Google Scholar] [CrossRef]
- Fukuda, Y.; Asaoka, T.; Eguchi, H.; Yokota, Y.; Kubo, M.; Kinoshita, M.; Urakawa, S.; Iwagami, Y.; Tomimaru, Y.; Akita, H.; et al. Endogenous CXCL9 affects prognosis by regulating tumor-infiltrating natural killer cells in intrahepatic cholangiocarcinoma. Cancer Sci. 2020, 111, 323–333. [Google Scholar] [CrossRef]
- Morisaki, T.; Umebayashi, M.; Kiyota, A.; Koya, N.; Tanaka, H.; Onishi, H.; Katano, M. Combining cetuximab with killer lymphocytes synergis-tically inhibits human cholangiocarcinoma cells in vitro. Anticancer Res. 2012, 32, 2249–2256. [Google Scholar] [PubMed]
- Fridman, W.H.; Pages, F.; Sautes-Fridman, C.; Galon, J. The immune contexture in human tumours: Impact on clinical outcome. Nat. Rev. Cancer 2012, 12, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Kasper, H.-U.; Drebber, U.; Stippel, D.L.; Dienes, H.P.; Gillessen, A. Liver tumor infiltrating lymphocytes: Comparison of hepatocellular and cholangiolar carcinoma. World J. Gastroenterol. 2009, 15, 5053–5057. [Google Scholar] [CrossRef] [PubMed]
- Salmon, H.; Franciszkiewicz, K.; Damotte, D.; Dieu-Nosjean, M.C.; Validire, P.; Trautmann, A.; Mami-Chouaib, F.; Donnadieu, E. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J. Clin. Investig. 2012, 122, 899–910. [Google Scholar] [CrossRef] [PubMed]
- Krummel, M.F.; Bartumeus, F.; Gérard, A. T cell migration, search strategies and mechanisms. Nat. Rev. Immunol. 2016, 16, 193–201. [Google Scholar] [CrossRef]
- Lim, Y.J.; Koh, J.; Kim, K.; Chie, E.K.; Kim, B.; Lee, K.B.; Jang, J.Y.; Kim, S.W.; Oh, D.Y.; Bang, Y.J.; et al. High ratio of programmed cell death protein 1 (PD-1)+/CD8+ tumor-infiltrating lymphocytes identifies a poor prognostic subset of extrahepatic bile duct cancer undergoing surgery plus adjuvant chemoradiotherapy. Radiother. Oncol. 2015, 117, 165–170. [Google Scholar] [CrossRef]
- Miura, T.; Yoshizawa, T.; Hirai, H.; Seino, H.; Morohashi, S.; Wu, Y.; Wakiya, T.; Kimura, N.; Kudo, D.; Ishido, K.; et al. Prognostic Impact of CD163+ Macrophages in Tumor Stroma and CD8+ T-Cells in Cancer Cell Nests in Invasive Extrahepatic Bile Duct Cancer. Anticancer Res. 2017, 37, 183–190. [Google Scholar] [CrossRef]
- Jonuleit, H.; Schmitt, E.; Schuler, G.; Knop, J.; Enk, A.H. Induction of Interleukin 10–Producing, Nonproliferating Cd4+ T Cells with Regulatory Properties by Repetitive Stimulation with Allogeneic Immature Human Dendritic Cells. J. Exp. Med. 2000, 192, 1213–1222. [Google Scholar] [CrossRef]
- Oshikiri, T.; Miyamoto, M.; Shichinohe, T.; Suzuoki, M.; Hiraoka, K.; Nakakubo, Y.; Shinohara, T.; Itoh, T.; Kondo, S.; Katoh, H. Prognostic value of intratumoral CD8+ T lymphocyte in extrahepatic bile duct carcinoma as essential immune response. J. Surg. Oncol. 2003, 84, 224–228. [Google Scholar] [CrossRef]
- Takagi, S.; Miyagawa, S.-I.; Ichikawa, E.; Soeda, J.; Miwa, S.; Miyagawa, Y.; Iijima, S.; Noike, T.; Kobayashi, A.; Kawasaki, S. Dendritic cells, T-cell infiltration, and grp94 expression in cholangiocellular carcinoma. Hum. Pathol. 2004, 35, 881–886. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.; Liu, Y.; Li, S.; Mao, Y.; Wang, J.; Shuang, Z.; Chen, J.; Li, S. Elevated neutrophil-to lymphocyte ratio is an independent poor prognostic factor in patients with intrahepatic cholangiocarcinoma. Oncotarget 2016, 7, 50963–50971. [Google Scholar] [CrossRef] [PubMed]
- Montal, R.; Sia, D.; Montironi, C.; Leow, W.Q.; Esteban-Fabró, R.; Pinyol, R.; Torres-Martin, M.; Bassaganyas, L.; Moeini, A.; Peix, J.; et al. Molecular classification and therapeutic targets in extrahepatic cholangiocarcinoma. J. Hepatol. 2020. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, X.Y.; Zhang, Y.; Xu, D.; Dong, J.; Zhang, Z.; Yi, C.H.; Jia, H.L.; Yang, X. Programmed death ligand 1 expression in human intrahepatic cholangiocarcinoma and its association with prognosis and CD8(+) T-cell immune responses. Cancer Manag. Res. 2018, 10, 4113–4123. [Google Scholar] [CrossRef]
- Fontugne, J.; Augustin, J.; Pujals, A.; Compagnon, P.; Rousseau, B.; Luciani, A.; Tournigand, C.; Cherqui, D.; Azoulay, D.; Pawlotsky, J.-M.; et al. PD-L1 expression in perihilar and intrahepatic cholangiocarcinoma. Oncotarget 2017, 8, 24644–24651. [Google Scholar] [CrossRef] [PubMed]
- Gani, F.; Nagarajan, N.; Kim, Y.; Zhu, Q.; Luan, L.; Bhaijjee, F.; Anders, R.A.; Pawlik, T.M. Program Death 1 Immune Checkpoint and Tumor Microenvironment: Implications for Patients with Intrahepatic Cholangiocarcinoma. Ann. Surg. Oncol. 2016, 23, 2610–2617. [Google Scholar] [CrossRef]
- Jing, C.Y.; Fu, Y.P.; Yi, Y.; Zhang, M.X.; Zheng, S.S.; Huang, J.L.; Gan, W.; Xu, X.; Lin, J.J.; Zhang, J.; et al. HHLA2 in intrahepatic cholangiocarcinoma: An immune checkpoint with prognostic significance and wider expression compared with PD-L1. J. Immunother. Cancer 2019, 7, 77. [Google Scholar] [CrossRef]
- Kida, A.; Mizukoshi, E.; Tamai, T.; Terashima, T.; Kitahara, M.; Arai, K.; Yamashita, T.; Fushimi, K.; Honda, M.; Kaneko, S. Immune responses against tumour-associated antigen-derived cytotoxic T lymphocyte epitopes in cholangiocarcinoma patients. Liver Int. 2018, 38, 2040–2050. [Google Scholar] [CrossRef]
- Jeng, Y.M.; Chen, C.L.; Hsu, H.C. Lymphoepithelioma-like cholangiocarcinoma: An Epstein-Barr virus-associated tumor. Am. J. Surg. Pathol. 2001, 25, 516–520. [Google Scholar] [CrossRef]
- Ghidini, M.; Cascione, L.; Carotenuto, P.; Lampis, A.; Trevisani, F.; Previdi, M.C.; Hahne, J.C.; Said-Huntingford, I.; Raj, M.; Zerbi, A.; et al. Characterisation of the immune-related transcriptome in resected biliary tract cancers. Eur. J. Cancer 2017, 86, 158–165. [Google Scholar] [CrossRef]
- Whiteside, T.L. What are regulatory T cells (Treg) regulating in cancer and why? Semin. Canc. Biol. 2012, 22, 327–334. [Google Scholar] [CrossRef]
- von Andrian, U.H.; Mempel, T.R. Homing and cellular traffic in lymph nodes. Nat. Rev. Immunol. 2003, 3, 867–878. [Google Scholar] [CrossRef]
- Thepmalee, C.; Panya, A.; Sujjitjoon, J.; Sawasdee, N.; Poungvarin, N.; Junking, M.; Yenchitsomanus, P.T. Suppression of TGF-beta and IL-10 receptors on self-differentiated dendritic cells by short-hairpin RNAs enhanced activation of effector T-cells against cholangiocarcinoma cells. Hum. Vaccin. Immunother. 2020, 16, 1–10. [Google Scholar] [CrossRef]
- Martin-Sierra, C.; Martins, R.; Laranjeira, P.; Abrantes, A.M.; Oliveira, R.C.; Tralhão, J.G.; Botelho, M.F.; Furtado, E.; Domingues, R.; Paiva, A. Functional Impairment of Circulating FcepsilonRI(+) Monocytes and Myeloid Dendritic Cells in Hepatocellular Carcinoma and Cholangiocarcinoma Patients. Cytom. B Clin. Cytom. 2019, 96, 490–495. [Google Scholar] [CrossRef]
- Veglia, F.; Perego, M.; Gabrilovich, D. Myeloid-derived suppressor cells coming of age. Nat. Immunol. 2018, 19, 108–119. [Google Scholar] [CrossRef]
- Lin, Y.; Li, B.; Yang, X.; Cai, Q.; Liu, W.; Tian, M.; Luo, H.; Yin, W.; Song, Y.; Shi, Y.; et al. Fibroblastic FAP promotes intrahepatic cholangiocarcinoma growth via MDSCs recruitment. Neoplasia 2019, 21, 1133–1142. [Google Scholar] [CrossRef] [PubMed]
- Dunne, R.F.; Figueroa, N.; Belt, B.; Findeis-Hosey, J.; Lunt, M.; McMahon, J.Y.; Baran, A.; Shubin, A.; Noel, M.S.; Tejani, M.A.; et al. The role of myeloid derived suppressor cells in cholangiocarcinoma: A potential target for therapy. J. Clin. Oncol. 2016, 34, 273. [Google Scholar] [CrossRef]
- Tsilimigras, D.I.; Brodt, P.; Clavien, P.A.; Muschel, R.J.; D’Angelica, M.I.; Endo, I.; Parks, R.W.; Doyle, M.; de Santibañes, E.; Pawlik, T.M. Liver metastases. Nat. Rev. Dis. Primers 2021, 7, 27. [Google Scholar] [CrossRef] [PubMed]
- Sirica, A.E.; Almenara, J.A.; Li, C. Periostin in intrahepatic cholangiocarcinoma: Pathobiological insights and clinical implications. Exp. Mol. Pathol. 2014, 97, 515–524. [Google Scholar] [CrossRef]
- Zheng, Y.; Zhou, C.; Yu, X.X.; Wu, C.; Jia, H.L.; Gao, X.M.; Yang, J.M.; Wang, C.Q.; Luo, Q.; Zhu, Y.; et al. Osteopontin promotes metastasis of intrahepatic cholangiocarcinoma through recruiting MAPK1 and mediating Ser675 phosphorylation of beta-Catenin. Cell Death Dis. 2018, 9, 179. [Google Scholar] [CrossRef] [PubMed]
- Laohaviroj, M.; Chamgramol, Y.; Pairojkul, C.; Mulvenna, J.; Sripa, B. Clinicopathological Significance of Osteopontin in Cholangiocarcinoma Cases. Asian Pac. J. Cancer Prev. 2016, 17, 201–205. [Google Scholar] [CrossRef]
- Chung, J.W.; Kim, M.S.; Piao, Z.-H.; Jeong, M.; Yoon, S.R.; Shin, N.; Kim, S.Y.; Hwang, E.S.; Yang, Y.; Lee, Y.H.; et al. Osteopontin Promotes the Development of Natural Killer Cells from Hematopoietic Stem Cells. Stem Cells 2008, 26, 2114–2123. [Google Scholar] [CrossRef]
- Stromnes, I.M.; Goverman, J.M. Osteopontin-induced survival of T cells. Nat. Immunol. 2007, 8, 19–20. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Meng, F.; Glaser, S.; Alpini, G. Exosomes in liver pathology. J. Hepatol. 2016, 65, 213–221. [Google Scholar] [CrossRef]
- Tao, S.C.; Guo, S.C. Extracellular vesicles in bone: “dogrobbers” in the “eternal battle field”. Cell Commun. Signal. 2019, 17, 6. [Google Scholar] [CrossRef] [PubMed]
- Braicu, C.; Tomuleasa, C.; Monroig, P.D.C.; Cucuianu, A.; Berindanneagoe, I.; Calin, G.A. Exosomes as divine messengers: Are they the Hermes of modern molecular oncology? Cell Death Differ. 2015, 22, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Miao, D.-L.; Chen, L. Comprehensive analysis of long noncoding RNA-associated competing endogenous RNA network in cholangiocarcinoma. Biochem. Biophys. Res. Commun. 2018, 506, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Shen, J.; Chan, M.T.; Wu, W.K.K. The role of microRNAs in intrahepatic cholangiocarcinoma. J. Cell. Mol. Med. 2017, 21, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Utaijaratrasmi, P.; Vaeteewoottacharn, K.; Tsunematsu, T.; Jamjantra, P.; Wongkham, S.; Pairojkul, C.; Khuntikeo, N.; Ishimaru, N.; Sirivatanauksorn, Y.; Pongpaibul, A.; et al. The microRNA-15a-PAI-2 axis in cholangiocarcinoma-associated fibroblasts promotes migration of cancer cells. Mol. Cancer 2018, 17, 10. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Piontek, K.; Ishida, M.; Fausther, M.; Dranoff, J.A.; Fu, R.; Mezey, E.; Gould, S.J.; Fordjour, F.K.; Meltzer, S.J.; et al. Extracellular vesicles carry microRNA-195 to intrahepatic cholangiocarcinoma and improve survival in a rat model. Hepatology 2017, 65, 501–514. [Google Scholar] [CrossRef]
- Caligiuri, A.; Pastore, M.; Lori, G.; Raggi, C.; Di Maira, G.; Marra, F.; Gentilini, A. Role of Chemokines in the Biology of Cholangiocarcinoma. Cancers 2020, 12, 2215. [Google Scholar] [CrossRef]
- Kiefer, F.; Siekmann, A.F. The role of chemokines and their receptors in angiogenesis. Cell. Mol. Life Sci. 2011, 68, 2811–2830. [Google Scholar] [CrossRef]
- Steinberg, M.; Silva, M. Plerixafor: A chemokine receptor-4 antagonist for mobilization of hematopoietic stem cells for transplantation after high-dose chemotherapy for non-hodgkin’s lymphoma or multiple myeloma. Clin. Ther. 2010, 32, 821–843. [Google Scholar] [CrossRef]
- Schott, A.F.; Goldstein, L.J.; Cristofanilli, M.; Ruffini, P.A.; McCanna, S.; Reuben, J.M.; Perez, R.P.; Kato, G.; Wicha, M. Phase Ib Pilot Study to Evaluate Reparixin in Combination with Weekly Paclitaxel in Patients with HER-2–Negative Metastatic Breast Cancer. Clin. Cancer Res. 2017, 23, 5358–5365. [Google Scholar] [CrossRef]
- Melero, I.; Berman, D.M.; Aznar, M.A.; Korman, A.J.; Gracia, J.L.P.; Haanen, J.B.A.G. Evolving synergistic combinations of targeted immunotherapies to combat cancer. Nat. Rev. Cancer 2015, 15, 457–472. [Google Scholar] [CrossRef]
- Moris, D.; Rahnemai-Azar, A.A.; Zhang, X.; Ntanasis-Stathopoulos, I.; Tsilimigras, D.I.; Chakedis, J.; Argyrou, C.; Fung, J.J.; Pawlik, T.M. Program death-1 immune checkpoint and tumor microenvironment in malignant liver tumors. Surg. Oncol. 2017, 26, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Goeppert, B.; Frauenschuh, L.; Zucknick, M.; Roessler, S.; Mehrabi, A.; Hafezi, M.; Stenzinger, A.; Warth, A.; Pathil, A.; Renner, M.; et al. Major histocompatibility complex class I expression impacts on patient survival and type and density of immune cells in biliary tract cancer. Br. J. Cancer 2015, 113, 1343–1349. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Arai, Y.; Totoki, Y.; Shirota, T.; ElZawahry, A.; Kato, M.; Hama, N.; Hosoda, F.; Urushidate, T.; Ohashi, S.; et al. Genomic spectra of biliary tract cancer. Nat. Genet. 2015, 47, 1003–1010. [Google Scholar] [CrossRef] [PubMed]
- Sabbatino, F.; Villani, V.; Yearley, J.H.; Deshpande, V.; Cai, L.; Konstantinidis, I.T.; Moon, C.; Nota, S.P.F.T.; Wang, Y.; Al-Sukaini, A.; et al. PD-L1 and HLA Class I Antigen Expression and Clinical Course of the Disease in Intrahepatic Cholangiocarcinoma. Clin. Cancer Res. 2016, 22, 470–478. [Google Scholar] [CrossRef]
- Ma, C.; Peng, C.; Lu, X.; Ding, X.; Zhang, S.; Zou, X.; Zhang, X. Downregulation of FOXP3 inhibits invasion and immune escape in cholangiocarcinoma Biochem. Biochem. Biophys. Res. Commun. 2015, 458, 234–239. [Google Scholar] [CrossRef]
- Pardoll, D.M. The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 2012, 12, 252–264. [Google Scholar] [CrossRef]
- Cameron, F.; Whiteside, G.; Perry, C. Ipilimumab: First global approval. Drugs 2011, 71, 1093–1104. [Google Scholar] [CrossRef]
- Shih, K.; Arkenau, H.-T.; Infante, J.R. Clinical Impact of Checkpoint Inhibitors as Novel Cancer Therapies. Drugs 2014, 74, 1993–2013. [Google Scholar] [CrossRef]
- The Nobel Prize in Physiology or Medicine. 2018. Available online: https://www.nobelprize.org/prizes/medicine/2018/press-release/ (accessed on 1 October 2018).
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T.; Uram, J.N.; Wang, H.; Bartlett, B.R.; Kemberling, H.; Eyring, A.D.; Skora, A.D.; Luber, B.S.; Azad, N.S.; Laheru, D.; et al. PD-1 Blockade in Tumors with Mismatch-Repair Deficiency. N. Engl. J. Med. 2015, 372, 2509–2520. [Google Scholar] [CrossRef]
- Shigeyasu, K.; Tanakaya, K.; Nagasaka, T.; Aoki, H.; Fujiwara, T.; Sugano, K.; Ishikawa, H.; Yoshida, T.; Moriya, Y.; Furukawa, Y.; et al. Early detection of metachronous bile duct cancer in Lynch syndrome: Report of a case. Surg. Today 2014, 44, 1975–1981. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, R.; Coppola, D.; Wang, E.; Chang, Y.D.; Kim, Y.; Anaya, D.; Kim, D.W. Prognostic value of CD8CD45RO tumor infiltrating lymphocytes in patients with extrahepatic cholangiocarcinoma. Oncotarget 2018, 9, 23366–23372. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Liu, F.; Zhang, F.; Zhou, W.; Jiang, X.; Yang, Y.; Qu, K.; Wang, Y.; Ma, Q.; Wang, T.; et al. Genomic ERBB2/ERBB3 mutations promote PD-L1-mediated immune escape in gallbladder cancer: A whole-exome sequencing analysis. Gut 2019, 68, 1024–1033. [Google Scholar] [CrossRef]
- Santana-Davila, R.; Bhatia, S.; Chow, L.Q.M. Harnessing the Immune System as a Therapeutic Tool in Virus-Associated Cancers. JAMA Oncol. 2017, 3, 106–112. [Google Scholar] [CrossRef]
- Ueno, M.; Chung, H.; Nagrial, A.; Marabelle, A.; Kelley, R.; Xu, L.; Mahoney, J.; Pruitt, S.; Oh, D.-Y. Pembrolizumab for advanced biliary adenocarcinoma: Results from the multicohort, phase II KEYNOTE-158 study. Ann. Oncol. 2018, 29, viii210. [Google Scholar] [CrossRef]
- Bang, Y.-J.; Ueno, M.; Malka, D.; Chung, H.C.; Nagrial, A.; Kelley, R.K.; Piha-Paul, S.A.; Ros, W.; Italiano, A.; Nakagawa, K.; et al. Pembrolizumab (pembro) for advanced biliary adenocarcinoma: Results from the KEYNOTE-028 (KN028) and KEYNOTE-158 (KN158) basket studies. J. Clin. Oncol. 2019, 37, 4079. [Google Scholar] [CrossRef]
- Lwin, Z.; Gomez-Roca, C.; Saada-Bouzid, E.; Yanez, E.; Longo Muñoz, F.; Im, S.; Castanon, E.; Senellart, H.; Graham, D.; Voss, M.; et al. LEAP-005: Phase II study of Lenvatinib (len) plus pembrolizumab (pembro) in patients (pts) with previously treated advanced solid tumors. Ann. Oncol. 2020, 31, S1142–S1215. [Google Scholar] [CrossRef]
- Kim, R.D.; Chung, V.; Alese, O.B.; El-Rayes, B.F.; Li, D.; Al-Toubah, T.E.; Schell, M.J.; Zhou, J.-M.; Mahipal, A.; Kim, B.H.; et al. A Phase 2 Multi-institutional Study of Nivolumab for Patients with Advanced Refractory Biliary Tract Cancer. JAMA Oncol. 2020, 6, 888. [Google Scholar] [CrossRef]
- Chen, W.X.; Li, G.X.; Hu, Z.N.; Zhu, P.; Zhang, B.X.; Ding, Z.Y. Significant response to anti-PD-1 based immunotherapy plus lenvatinib for recurrent intrahepatic cholangiocarcinoma with bone metastasis: A case report and literature review. Medicine 2019, 98, e17832. [Google Scholar] [CrossRef] [PubMed]
- Ueno, M.; Ikeda, M.; Morizane, C.; Kobayashi, S.; Ohno, I.; Kondo, S.; Okano, N.; Kimura, K.; Asada, S.; Namba, Y.; et al. Nivolumab alone or in combination with cisplatin plus gemcitabine in Japanese patients with unresectable or recurrent biliary tract cancer: A non-randomised, multicentre, open-label, phase 1 study. Lancet Gastroenterol. Hepatol. 2019, 4, 611–621. [Google Scholar] [CrossRef]
- Klein, O.; Kee, D.; Nagrial, A.; Markman, B.; Underhill, C.; Michael, M.; Jackett, L.; Lum, C.; Behren, A.; Palmer, J.; et al. Evaluation of Combination Nivolumab and Ipilimumab Im-munotherapy in Patients with Advanced Biliary Tract Cancers: Subgroup Analysis of a Phase 2 Nonrandomized Clinical Trial. JAMA Oncol. 2020, 6, 1405–1409. [Google Scholar] [CrossRef]
- Oh, D.Y.; Lee, K.H.; Lee, D.W.; Yong Kim, T.; Bang, J.H.; Nam, A.R.; Lee, Y.; Zhang, Q.; Rebelatto, M.; Li, W.; et al. Phase II study assessing tolerability, efficacy, and biomarkers for durvalumab, tremelimumab and gemcitabine/cisplatin in chemo-naïve advanced biliary tract cancer. J. Clin. Oncol. 2020, 38 (Suppl. 15), 4520. [Google Scholar] [CrossRef]
- Lind, H.; Gameiro, S.R.; Jochems, C.; Donahue, R.N.; Strauss, J.; Gulley, J.L.M.D.; Palena, C.; Schlom, J. Dual targeting of TGF-β and PD-L1 via a bifunctional anti-PD-L1/TGF-βRII agent: Status of preclinical and clinical advances. J. Immunother. Cancer 2020, 8, e000433. [Google Scholar] [CrossRef]
- Yoo, C.; Oh, D.; Choi, H.J.; Kudo, M.; Ueno, M.; Kondo, S.; Chen, L.T.; Osada, M.; Helwig, C.; Dussault, I.; et al. M7824 (MSB0011359C), a bifunctional fusion protein targeting PD-L1 AND TGF-ß, in Asian patients with pretreated biliry tract cancer: Preliminary results from a phase 1 trial. Ann. Oncol. 2018, 29 (Suppl. 8), viii258–viii259. [Google Scholar] [CrossRef]
- Li, A.; Yi, M.; Qin, S.; Chu, Q.; Luo, S.; Wu, K. Prospects for combining immune checkpoint blockade with PARP inhibition. J. Hematol. Oncol. 2019, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Teo, M.Y.; Seier, K.; Ostrovnaya, I.; Regazzi, A.M.; Kania, B.E.; Moran, M.M.; Cipolla, C.K.; Bluth, M.J.; Chaim, J.; Al-Ahmadie, H.; et al. Alterations in DNA damage response and repair genes as potential marker of clinical benefit from PD-1/PD-L1 blockade in advanced urothelial cancers. J. Clin. Oncol. 2018, 36, 1685–1694. [Google Scholar] [CrossRef] [PubMed]
- Chabanon, R.M.; Muirhead, G.; Krastev, D.B.; Adam, J.; Morel, D.; Garrido, M.; Lamb, A.; Hénon, C.; Dorvault, N.; Rouanne, M.; et al. PARP inhibition enhances tumor cell–intrinsic immunity in ERCC1-deficient non–small cell lung cancer. J. Clin. Investig. 2019, 129, 1211–1228. [Google Scholar] [CrossRef] [PubMed]
- Mouw, K.W.; Goldberg, M.S.; Konstantinopoulos, P.A.; D’Andrea, A.D. DNA damage and repair biomarkers of immunotherapy response. Cancer Discov. 2017, 7, 675–693. [Google Scholar] [CrossRef] [PubMed]
- Strickland, K.C.; Howitt, B.E.; Shukla, S.A.; Rodig, S.; Ritterhouse, L.L.; Liu, J.F.; Garber, J.E.; Chowdhury, D.; Wu, C.J.; D’Andrea, A.D.; et al. Association and prognostic significance of BRCA1/2-mutation status with neoantigen load, number of tumor-infiltrating lymphocytes and expression of PD-1/PD-L1 in high grade serous ovarian cancer. Oncotarget 2016, 7, 13587–13598. [Google Scholar] [CrossRef] [PubMed]
- Chae, Y.K.; Davis, A.A.; Raparia, K.; Agte, S.; Pan, A.; Mohindra, N.; Villaflor, V.; Giles, F. Association of tumor mutational burden with DNA repair mutations and response to anti–PD-1/PD-L1 therapy in non–small-cell lung cancer. Clin. Lung Cancer 2019, 20, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, N.A.; Hellmann, M.D.; Snyder, A.; Kvistborg, P.; Makarov, V.; Havel, J.J.; Lee, W.; Yuan, J.; Wong, P.; Ho, T.S.; et al. Mutational landscape determines sensitivity to PD-1 blockade in non–small cell lung cancer. Science 2015, 348, 124–128. [Google Scholar] [CrossRef]
- Spizzo, G.; Puccini, A.; Xiu, J.; Goldberg, R.M.; Grothey, A.; Shields, A.F.; Arora, S.P.; Khushmann, M.; Salem, M.E.; Battaglin, F.; et al. Molecular profile of BRCA-mutated biliary tract cancers. ESMO Open 2020, 5, e000682. [Google Scholar] [CrossRef]
- Saeed, A.; Park, R.; Al-Jumayli, M.; Al-Rajabi, R.; Sun, W. Biologics, Immunotherapy, and Future Directions in the Treatment of Advanced Cholangiocarcinoma. Clin. Color. Cancer 2019, 18, 81–90. [Google Scholar] [CrossRef]
- Rizzo, A.; Frega, G.; Ricci, A.D.; Palloni, A.; Abbati, F.; De Lorenzo, S.; Deserti, M.; Tavolari, S.; Brandi, G. Anti-EGFR Monoclonal Antibodies in Advanced Biliary Tract Cancer: A Systematic Review and Meta-analysis. In Vivo 2020, 34, 479–488. [Google Scholar] [CrossRef]
- Garrido, F.; Perea, F.; Bernal, M.; Sánchez-Palencia, A.; Aptsiauri, N.; Ruiz-Cabello, F. The Escape of Cancer from T Cell-Mediated Immune Surveillance: HLA Class I Loss and Tumor Tissue Architecture. Vaccines 2017, 5, 7. [Google Scholar] [CrossRef]
- Mall, A.S.; Tyler, M.G.; Ho, S.B.; Krige, J.E.; Kahn, D.; Spearman, W.; Myer, L.; Govender, D.; Spearman, C.W. The expression of MUC mucin in cholangiocarcinoma. Pathol. Res. Pr. 2010, 206, 805–809. [Google Scholar] [CrossRef]
- Nakatsuka, S.-I.; Oji, Y.; Horiuchi, T.; Kanda, T.; Kitagawa, M.; Takeuchi, T.; Kawano, K.; Kuwae, Y.; Yamauchi, A.; Okumura, M.; et al. Immunohistochemical detection of WT1 protein in a variety of cancer cells. Mod. Pathol. 2006, 19, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, N.; Yamamoto, M.; Aruga, A.; Takasaki, K.; Nakano, M. Correlation between expression of MUC1 core protein and outcome after surgery in mass-forming intrahepatic cholangiocarcinoma. Cancer 2002, 94, 1770–1776. [Google Scholar] [CrossRef]
- Higashi, M.; Yonezawa, S.; Ho, J.J.; Tanaka, S.; Irimura, T.; Kim, Y.S.; Sato, E. Expression of MUC1 and MUC2 mucin antigens in intrahepatic bile duct tumors: Its relationship with a new morphological classification of cholangiocarcinoma. Hepatology 1999, 30, 1347–1355. [Google Scholar] [CrossRef] [PubMed]
- I Marks, E.; Yee, N.S. Immunotherapeutic approaches in biliary tract carcinoma: Current status and emerging strategies. World J. Gastrointest. Oncol. 2015, 7, 338–346. [Google Scholar] [CrossRef] [PubMed]
- Kaida, M.; Morita-Hoshi, Y.; Soeda, A.; Wakeda, T.; Yamaki, Y.; Kojima, Y.; Ueno, H.; Kondo, S.; Morizane, C.; Ikeda, M.; et al. Phase 1 Trial of Wilms Tumor 1 (WT1) Peptide Vaccine and Gemcitabine Combination Therapy in Patients with Advanced Pancreatic or Biliary Tract Cancer. J. Immunother. 2011, 34, 92–99. [Google Scholar] [CrossRef]
- Okusaka, T.; Ueno, M.; Sato, T.; Heike, Y. Possibility of immunotherapy for biliary tract cancer: How do we prove efficacy? Introduction to a current ongoing phase I and randomized phase II study to evaluate the efficacy and safety of adding Wilms tumor 1 peptide vaccine to gemcitabine and cisplatin for the treatment of advanced biliary tract cancer (WT-BT trial). J. Hepato-Biliary-Pancreatic Sci. 2012, 19, 314–318. [Google Scholar]
- Yamamoto, K.; Ueno, T.; Kawaoka, T.; Hazama, S.; Fukui, M.; Suehiro, Y.; Hamanaka, Y.; Ikematsu, Y.; Imai, K.; Oka, M.; et al. MUC1 peptide vaccination in patients with advanced pancreas or biliary tract cancer. Anticancer Res. 2005, 25, 3575–3579. [Google Scholar]
- Lepisto, A.J.; Moser, A.J.; Zeh, H.; Lee, K.; Bartlett, D.; McKolanis, J.R.; Geller, B.A.; Schmotzer, A.; Potter, D.P.; Whiteside, T.; et al. A phase I/II study of a MUC1 peptide pulsed autologous dendritic cell vaccine as adjuvant therapy in patients with resected pancreatic and biliary tumors. Cancer Ther. 2008, 6, 955–964. [Google Scholar]
- Löffler, M.W.; Chandran, P.A.; Laske, K.; Schroeder, C.; Bonzheim, I.; Walzer, M.; Hilke, F.J.; Trautwein, N.; Kowalewski, D.J.; Schuster, H.; et al. Personalized peptide vaccine-induced immune response associated with long-term survival of a metastatic cholangiocarcinoma patient. J. Hepatol. 2016, 65, 849–855. [Google Scholar] [CrossRef]
- Yoshitomi, M.; Yutani, S.; Matsueda, S.; Ioji, T.; Komatsu, N.; Shichijo, S.; Yamada, A.; Itoh, K.; Sasada, T.; Kinoshita, H. Personalized peptide vaccination for advanced biliary tract cancer: IL-6, nutritional status and pre-existing antigen-specific immunity as possible biomarkers for patient prognosis. Exp. Ther. Med. 2012, 3, 463–469. [Google Scholar] [CrossRef]
- Sansone, P.; Bromberg, J. Targeting the Interleukin-6/Jak/Stat Pathway in Human Malignancies. J. Clin. Oncol. 2012, 30, 1005–1014. [Google Scholar] [CrossRef]
- Nishimoto, N.; Kishimoto, T. Interleukin 6: From bench to bedside. Nat. Clin. Pr. Rheumatol. 2006, 2, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Matsuyama, M.; Ishii, H.; Furuse, J.; Ohkawa, S.; Maguchi, H.; Mizuno, N.; Yamaguchi, T.; Ioka, T.; Ajiki, T.; Ikeda, M.; et al. Phase II trial of combination therapy of gemcitabine plus anti-angiogenic vaccination of elpamotide in patients with advanced or recurrent biliary tract cancer. Investig. New Drugs 2015, 33, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Sepúlveda, D.; Tittarelli, A.; Gleisner, M.A.; Ávalos, I.; Pereda, C.; Gallegos, I.; González, F.E.; López, M.N.; Butte, J.M.; Roa, J.C.; et al. Tumor lysate-based vaccines: On the road to immunotherapy for gallbladder cancer. Cancer Immunol. Immunother. 2018, 67, 1897–1910. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Kotera, Y.; Aruga, A.; Takeshita, N.; Takasaki, K.; Yamamoto, M. Clinical utilization of postoperative dendritic cell vaccine plus activated T-cell transfer in patients with intrahepatic cholangiocarcinoma. J. Hepato-Biliary-Pancreatic Sci. 2012, 19, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Dudley, M.E.; Gross, C.A.; Langhan, M.M.; Garcia, M.R.; Sherry, R.M.; Yang, J.C.; Phan, G.Q.; Kammula, U.S.; Hughes, M.S.; Citrin, D.E.; et al. CD8+ enriched “young” tumor infiltrating lymphocytes can mediate regression of metastatic melanoma. Clin. Cancer Res. 2010, 16, 6122–6131. [Google Scholar] [CrossRef]
- Higuchi, R.; Yamamoto, M.; Hatori, T.; Shimizu, K.; Imai, K.; Takasaki, K. Intrahepatic cholangiocarcinoma with lymph node metastasis successfully treated by immunotherapy with CD3-activated T cells and dendritic cells after surgery: Report of a case. Surg. Today 2006, 36, 559–562. [Google Scholar] [CrossRef]
- Tran, E.; Turcotte, S.; Gros, A.; Robbins, P.F.; Lu, Y.-C.; Dudley, M.E.; Wunderlich, J.R.; Somerville, R.P.; Hogan, K.; Hinrichs, C.S.; et al. Cancer Immunotherapy Based on Mutation-Specific CD4+ T Cells in a Patient with Epithelial Cancer. Science 2014, 344, 641–645. [Google Scholar] [CrossRef]
- Feng, K.; Liu, Y.; Guo, Y.; Qiu, J.; Wu, Z.; Dai, H.; Yang, Q.; Wang, Y.; Han, W. Phase I study of chimeric antigen receptor modified T cells in treating HER2-positive advanced biliary tract cancers and pancreatic cancers. Protein Cell 2018, 9, 838–847. [Google Scholar] [CrossRef]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 2017, 14, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Mertens, J.C.; Fingas, C.D.; Christensen, J.D.; Smoot, R.L.; Bronk, S.F.; Werneburg, N.W.; Gustafson, M.P.; Dietz, A.B.; Roberts, L.R.; Sirica, A.E.; et al. Therapeutic effects of deleting cancer-associated fibroblasts in cholangiocarcinoma. Cancer Res. 2013, 73, 897–907. [Google Scholar] [CrossRef] [PubMed]
- Meric-Bernstam, F.; Larkin, J.; Tabernero, J.; Bonini, C. Enhancing anti-tumour efficacy with immunotherapy combinations. Lancet 2021, 397, 1010–1022. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).