1. Introduction
Clostridium perfringens is an opportunistic pathogen that is commonly found in the gastrointestinal tract of livestock and poultry. It is a Gram-positive, anaerobic, rod-shaped, and spore-forming bacterium that is the major causative agent of enteritis necroticans and enterotoxemia in animals. It can also cause food poisoning in humans [
1,
2,
3]. The main virulence factors of
C.
perfringens are its exotoxins including at least 12 toxins. Although some researchers recently proposed that
C. perfringens toxinotypes should be expanded to G (A to G seven toxinotypes) [
4], the traditional
C. perfringens toxinotyping scheme still recognizes that
C.
perfringens is classified into five toxinotypes (A, B, C, D, and E) according to the exotoxins α, β, ε, and ι [
5]. Among these exotoxins, α, β, and ε are the most important pathogenic factors of
C. perfringens. α-toxin is excreted by all
C. perfringens toxinotypes, and it is also the only exotoxin produced by toxinotype A. α-toxin has phospholipase and sphingomyelinase activities, and it can cause hemolysis, tissue necrosis, edema, and other effects [
6,
7]. β-toxin, also known as β1-toxin, is a key lethal virulence factor mainly produced by
C. perfringens toxinotype B and C, which has strong neurotoxicity, lethality, and necrotic effects. It can cause fatal hemorrhagic enterocolitis and enterotoxemia [
8]. β2-toxin, which has similar biological activities to β1-toxin, can cause gastrointestinal diseases [
9]. ε-toxin, which is produced by
C. perfringens toxinotypes B and D, is a potent pore-forming toxin that can cause central nervous system diseases in animals [
10]. Since
C. perfringens produces many exotoxins, particularly α, β, and ε exotoxins, which are the main virulence factors of the pathogenic bacterium, a multivalent vaccine would be more effective against
C. perfringens exotoxins.
Generally, the use of antibiotics like virginiamycin and tylosin is very common in livestock husbandry to prevent
C. perfringens infection. Although in-feed antibiotics effectively control bacterial diseases, the abuse of antibiotics has brought unavoidable negative effects on the environment and human health. In particular, antibiotic resistance and its persistence in the environment are causes of growing worldwide concern. Moreover, vaccines like injected killed vaccines can effectively prevent
C. perfringens infection [
11]. For example, in China,
C. perfringens (type A) inactivated vaccine for rabbits,
C. perfringens (types A and C) bivalent inactivated vaccine for piglets, and
C. perfringens (types A, B, C, D) tetravalent inactivated vaccine for sheep (Qilu Animal Health Products Co., LTD., Jinan, China) are available on the market. However, the vaccines often fail to resist the effects of exotoxins produced by
C. perfringens in the circulatory system and intestinal tracts.
C. perfringens is commonly found in intestinal tracts, and its exotoxins are mainly absorbed via the intestinal mucosa, causing disease development. Therefore, a vaccine that could induce efficacious protective immune responses against
C. perfringens toxins in the circulatory system and in the intestinal tracts is a promising approach.
Oral vaccination may be a better choice because oral vaccine stimulation can effectively induce secretory immunoglobulin A (sIgA)-based antigen-specific mucosal immune response and IgG-based systemic immune response, providing effective host protection [
12]. Furthermore, the antigen delivery carrier is essential for developing effective oral vaccines. An ideal delivery carrier can deliver antigens to intestinal mucosa to induce effective antigen-specific immune responses while also being safe and beneficial to the body.
Lactobacillus, commonly found in food and intestinal tracts, is an important probiotic. Many
Lactobacillus strains can promote adhesive interactions with intestinal epithelial cells [
13], prevent epithelial cell barrier injury [
14], ameliorate inflammation [
15], modulate innate immunity [
16], and regulate dendritic cell and T cell immunological functions [
17,
18]. Therefore, the use of
Lactobacillus as an antigen delivery carrier to express heterologous antigens for oral vaccine development has attracted much attention in this field, including using
Lactobacillus strains to express classical swine fever virus E2 protein [
19], dendritic cells, or microfold cells and dendritic cells-targeting peptide fused with porcine epidemic diarrhea virus COE antigen [
20,
21], bovine viral diarrhea virus E2 protein [
22], and α-toxoid of
C. perfringens [
23].
We previously constructed a genetically engineered
Lactobacillus strain pPG-E-α-β2-ε-β1/
L. casei constitutively expressing α, ε, β1, and β2 toxoids of
C. perfringens, obtaining good immunogenicity in mice orally administered with the recombinant
Lactobacillus strain [
24]. However, it is not clear if the recombinant probiotic oral vaccine can effectively induce mucosal and systemic immune responses and provide effective immune protection for livestock against
C. perfringens exotoxins. Here, we prepared a bait particle vaccine with the pPG-E-α-β2-ε-β1/
L. casei (
Lactobacillus casei was used as antigen delivery carrier to express α, ε, β1, and β2 toxoids of
C. perfringens. Subsequently, the recombinant probiotic expressing the tetravalent toxoids was made into bait particles with feed raw materials, which was used as oral vaccine for immunization). We further evaluated the immunogenicity and immune-protective effect of the bait particle vaccine in rabbits against α, β, and ε natural exotoxins via oral vaccination.
2. Materials and Methods
2.1. Animal and Bacterial Strains
Eight-week-old healthy New Zealand White (NZW) rabbits purchased from Harbin Fenying Farming Company, China were kept under clean conventional conditions with free access to standard chow diet and water.
Lactobacillus casei ATCC 393 (
L. casei) and recombinant lactobacillus pPG-E-α-β2-ε-β1/
L. casei constitutively expressing
C. perfringens α, ε, β1, and β2 toxoids were constructed by our laboratory [
24] and grown anaerobically in de Man, Rogosa, and Sharpe (MRS) broth (Sigma, St. Louis, MO, USA) at 37 °C without shaking.
C. perfringens toxinotype A (C57-1), toxinotype C (CACC-61), and toxinotype D (CCVC-81) were purchased from the China Institute of Veterinary Drug Control, (Beijing, China). Animal experiments were carried out in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The protocol (NEAU2018024) was approved by the Ethical Committee for Animal Experiments of Northeast Agricultural University, China.
2.2. Constitutive Protein Expression
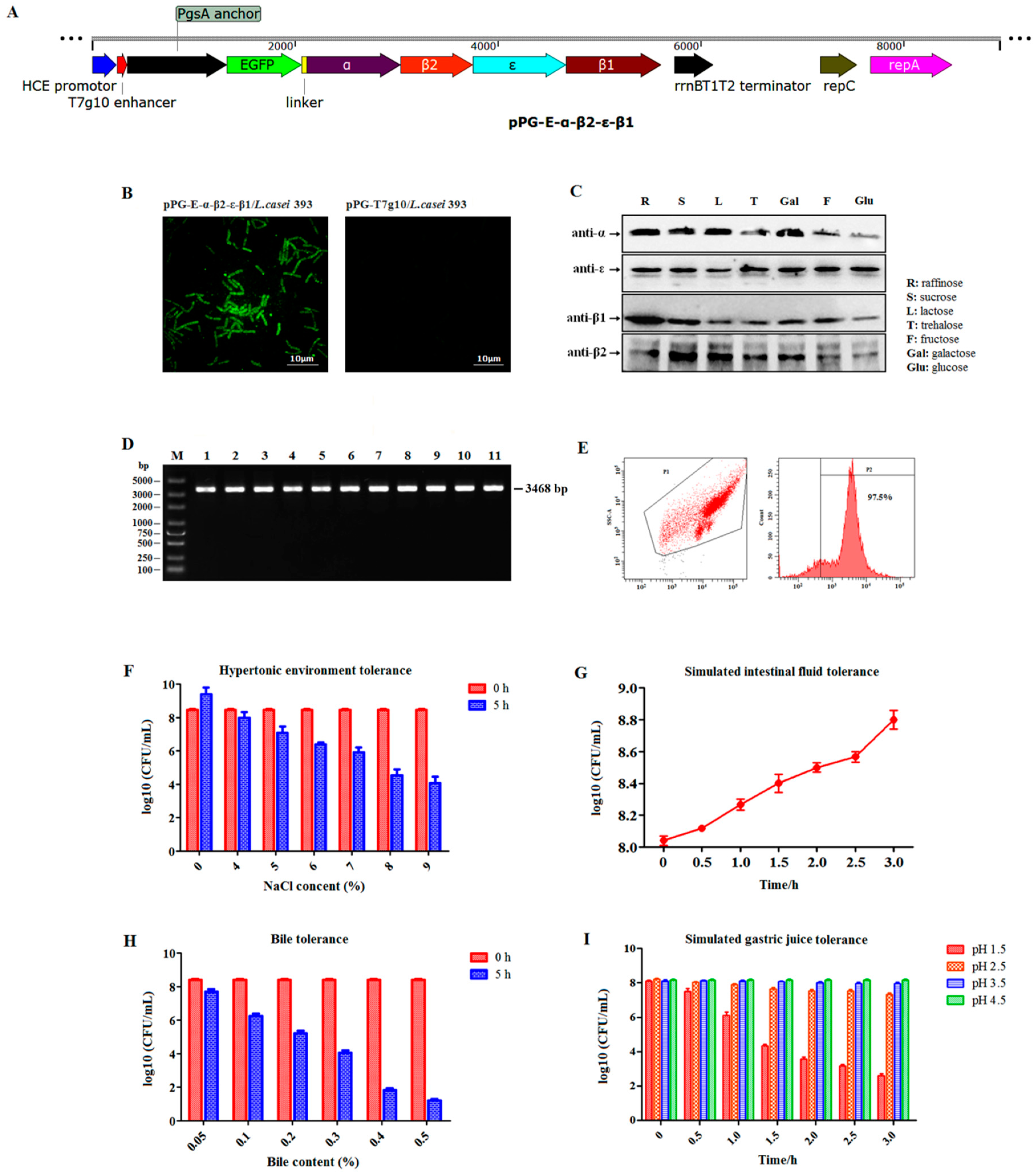
Here, enhanced green fluorescent protein (EGFP) was used as a marker to screen recombinant lactobacillus. So, we first identified the expression of EGFP without any specific inducers. Briefly, pPG-α-β2-ε-β1/L. casei was cultured in MRS broth at 37 °C for 16 h without shaking before centrifuging at 12,000× g for 10 min. After washing three times with sterile PBS, the cells were observed using ultra high-resolution microscopy. Subsequently, constitutive toxoid expression was identified using western blotting. Briefly, pPG-α-β2-ε-β1/L. casei was grown in basic MRS broth at 37 °C for 16 h using glucose, sucrose, galactose, lactose, raffinose, fructose, and trehalose as carbon sources, respectively. After centrifuging, the cell pellets were lysed with 2× sodium dodecyl sulfate (SDS) buffer before placing in a boiling water bath for 10 min and centrifuging. Next, the supernatant was analyzed using 10% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) combined with western blotting. For western blotting, the mouse anti-α, β2, ε, and β1 toxoid monoclonal antibody that was prepared in our laboratory (to prepare these monoclonal antibodies, α, β2, ε, and β1 toxoid protein was respectively expressed by E. coli followed by purification, immunization, fusion of immunized mice splenocytes and SP2/0. After that, the monoclonal antibodies targeting each toxoid protein were screened followed by evaluation of the specificity) was used as the primary antibody, and horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG (Sigma, Louis, MO, USA) was used as the secondary antibody.
2.3. Recombinant Plasmid pPG-α-β2-ε-β1 Segregational Stability
Segregational stability of recombinant plasmid pPG-α-β2-ε-β1 in
L. casei was evaluated as previously described [
12]. Briefly, pPG-α-β2-ε-β1/
L. casei was cultured in MRS broth at 37 °C until optical density at 600 nm (OD
600) of approximately 1.0. Subsequently, serial subcultures were performed by diluting the culture (1:100) in fresh broth and growing it until OD
600 ≈ 1.0. To determine the structural stability of recombinant plasmid pPG-α-β2-ε-β1 after 50 generations, 100 μL of diluted culture from every five generations was plated onto MRS agar plate and cultured at 37 °C for 12 h. Ten colonies were selected at random and analyzed using colony PCR assay (the primer sequences were that forward—5′-GATACAGATAATAATTTCTCAAAGGATAAT-3′ and reverse—5′-AATAGCTGTTACTTTGTGAGTAAGCC-3′ with annealing at 50 °C. The expected PCR result size is 3468 bp) combined with gene sequencing to detect the integrity of the α-β2-ε-β1 gene in the plasmid.
2.4. Digestive Tract Tolerance and Adhesion Ability of Recombinant Lactobacillus
One-milliliter of pPG-α-β2-ε-β1/L. casei cultured in MRS broth (OD600 ≈ 1.0) was centrifuged, and the following experiments were performed. (1) Bile tolerance test: the cells were re-inoculated in 1 mL MRS broth supplemented with 0.05%, 0.1%, 0.2%, 0.3%, 0.4%, or 0.5% of bile (wt/vol), and continually cultured at 37 °C for 5 h. (2) Hypertonic environment tolerance test: The cells were re-inoculated in 1 mL MRS broth containing 4%, 5%, 6%, 7%, 8%, or 9% of NaCl, and continually cultured at 37 °C for 5 h. (3) Gastric environment tolerance test: the cells were re-inoculated in 1 mL of simulated gastric juice (1 mol/L hydrochloric acid and 1 g pepsin per 100 mL) with pH 1.5, 2.5, 3.5, or 4.5 and continually cultured at 37 °C for 3 h. (4) Intestinal fluid environment tolerance test: the cells were re-inoculated in 1 mL of simulated intestinal fluid (0.68 g KH2PO4 and 1 g trypsase per 100 mL) and continually cultured at 37 °C for 3 h. In each experiment, the number of live bacteria was counted at 30-min intervals using the plate method.
Adhesion ability of recombinant lactobacillus in the intestinal tracts of NZW rabbits was evaluated as previously described [
12] with some modification. Briefly, eight-week-old NZW rabbits (
n = 18) were orally administered approximately 10
11 CFU/mL of cFDA-SE-labeled pPG-α-β2-ε-β1/
L. casei. Three NZW rabbits were randomly selected and sacrificed on days 1, 4, 7, 11, 15, and 20 after oral administration. Colon, jejunum, and ileum were extracted from each rabbit. Individual sections were cut longitudinally, and visible residual food particles or fecal material was removed from the intestinal tracts. Subsequently, the intestinal mucosa was scraped and placed in sterile PBS. After full oscillation, the microbes were dislodged from the mucosa tissue, fixed with formaldehyde (0.75%,
v/
v), and analyzed using flow cytometry. Normal rabbits (
n = 6) and rabbits treated with pPG-α-β2-ε-β1/
L. casei (unlabeled recombinant lactobacillus) (
n = 6) were used as negative control. The results were calculated as the following formula: the value of flow cytometry (experimental group)—the value of flow cytometry (negative control group).
2.5. Immunization
A bait particle vaccine was prepared with the pPG-α-β2-ε-β1/
L. casei strain. Briefly, the overnight cultures of the pPG-α-β2-ε-β1/
L. casei grown in MRS broth were thoroughly mixed with feed raw materials and cold-extruded into bait particles through an extruder type 210 with membrane hole of 6 mm (Jinquan Machinery Co., Ltd. Weihui, China) according to the manufacturer’s instructions. The bait particle vaccine contains, on average, approximately 10
10 CFU/g of the pPG-α-β2-ε-β1/
L. casei. The NZW rabbits in the probiotic vaccine group (
n = 31) were fed bait particles containing the recombinant lactobacillus (
Figure 1), and the control group (
n = 31) was fed bait particles containing PBS or pPG-T7g10/
L. casei. The immune protocol is shown in
Figure 1 [
23]. Animals were not fed for half a day before each immunization. In parallel, the group of NZW rabbits (
n = 20) was injected intramuscularly with commercial inactivated
C.
perfringens vaccine made by
C.
perfringens toxinotypes A, C, and D (China Animal Husbandry Industry Co., Ltd. Beijing, China) according to the manufacturer’s instructions. This group was used as the vaccine control group to evaluate the immune protection effect of the recombinant lactobacillus pPG-α-β2-ε-β1/
L. casei.
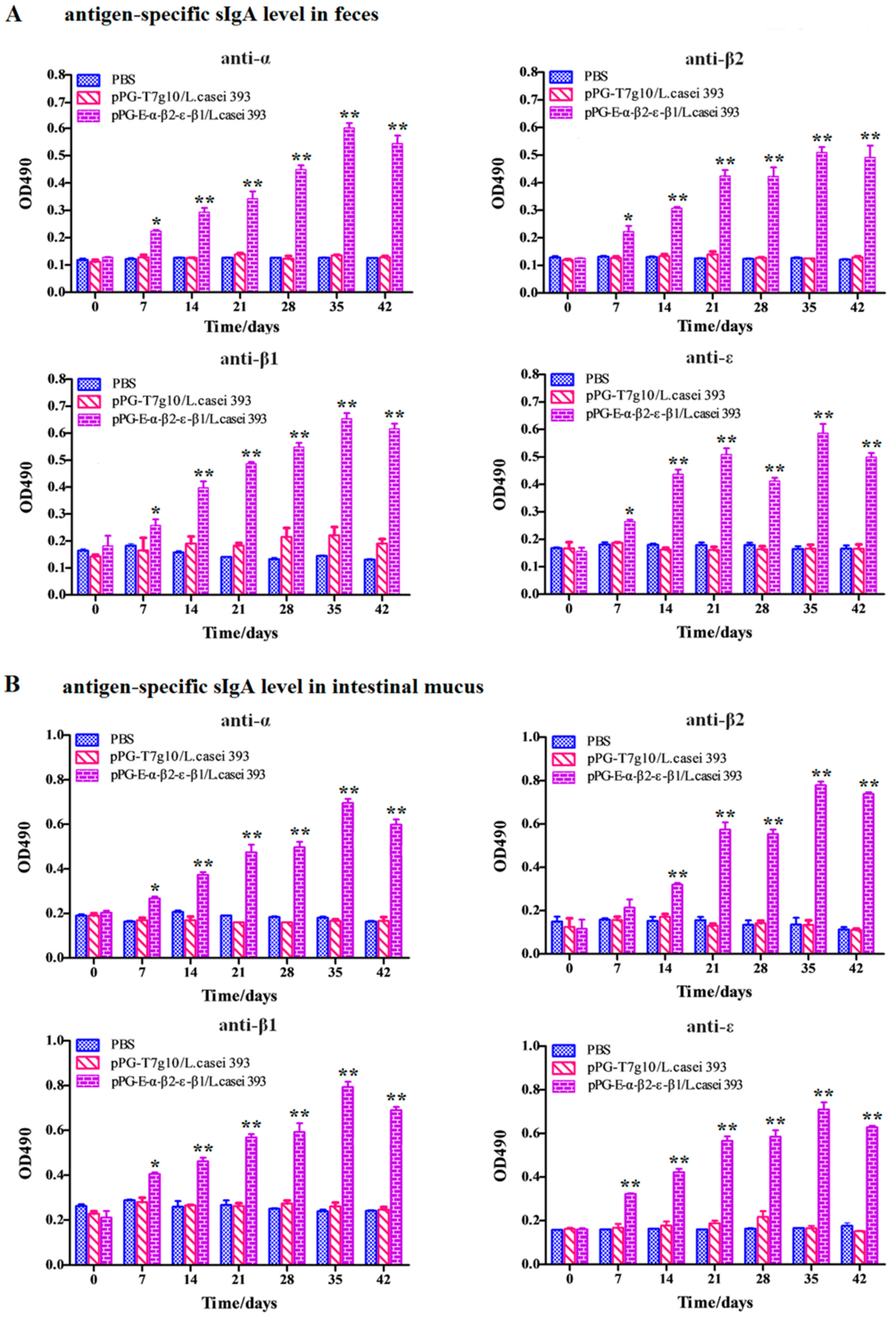
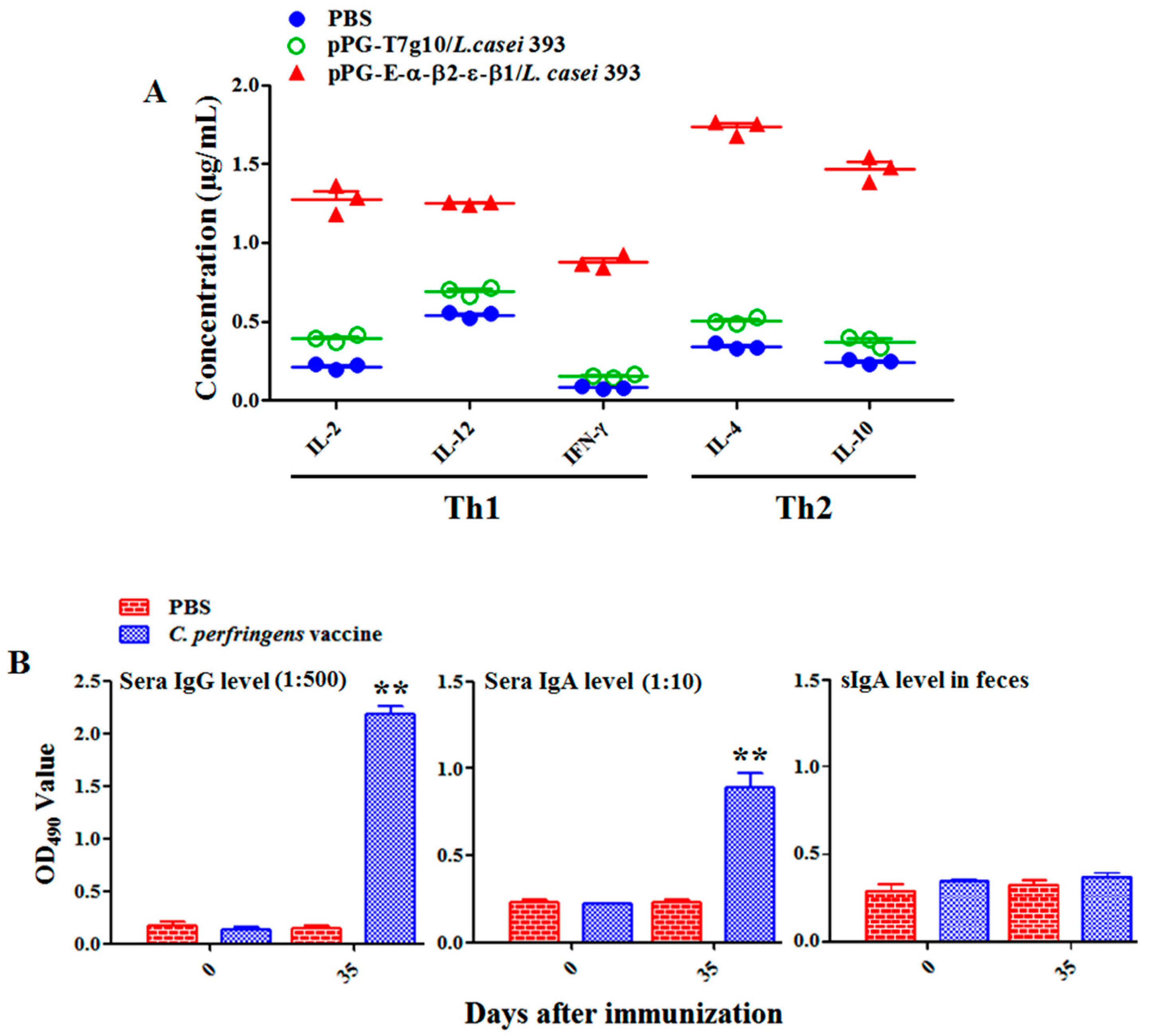
2.6. Detection of Antigen-Specific Antibodies and Cytokines
Serum, intestinal mucus, and fecal samples were collected from the NZW rabbits (
n = 3) in each group on days 0, 7, 14, 21, 28, 35, and 42 after primary immunization and prepared as previously described, respectively [
22]. Subsequently, antigen-specific sIgA levels in the intestinal mucus and fecal samples (diluted at 1:16) and antigen-specific IgG levels in serum samples (diluted at 1:32) were detected using ELISA against α, β1, β2, and ε toxoid proteins expressed by
E. coli in our laboratory as the coat antigen and using HRP-conjugated goat anti-mouse IgA or IgG (Abcam, Cambridge, MA, USA) as the secondary antibody. In addition, IL-2, IL-4, IL-10, IL-12, and IFN-γ cytokine levels in the serum samples obtained from the NZW rabbits in each group were detected using ELISA kits (Biosource International, Waltham, MA, USA). Moreover, to determine the antibody levels of vaccine control group, the serum samples diluted at 1:500/1:10 used for detecting specific IgG/IgA antibody level and fecal samples (diluted at 1:1) used for detecting specific sIgA level were collected on day 0 and 35 after primary immunization before performing ELISA assay using inactivated
C.
perfringens vaccine antigens as the coating antigen.
2.7. Ability of Antibodies to Neutralize Exotoxins of C. perfringens
The exotoxin-neutralizing ability of IgG and sIgA antibodies obtained from the NZW rabbits orally immunized with the pPG-α-β2-ε-β1/L. casei was evaluated using the antibodies obtained from group pPG-T7g10/L. casei and PBS as a control. Briefly, undiluted IgG and sIgA antibodies collected from each group on day 35 after primary immunization were mixed with equal volume (1× lethal dose, LD100) of natural exotoxin mixtures produced by C. perfringens toxinotype A, C, and D, and incubated at 37 °C for 1 h. Next, eight-week-old healthy NZW rabbits (five rabbits for each group) were challenged with the exotoxin mixtures pretreated with the IgG/sIgA antibody via injection. The survival rate of rabbits in each group was determined.
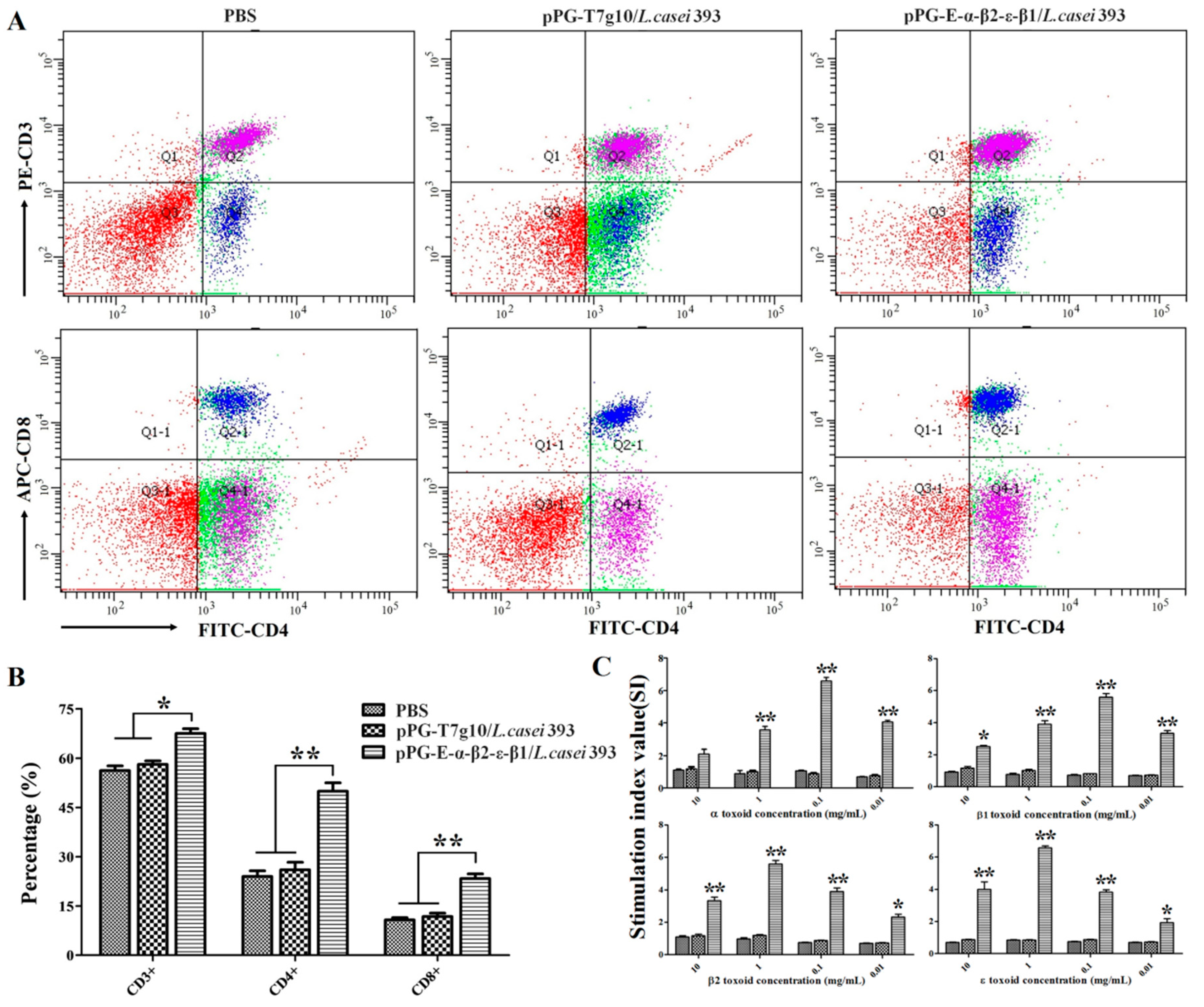
2.8. Detection of CD4+ and CD8+ T Cells
Spleen lymphocytes of the NZW rabbits (n = 3) in each group were collected and adjusted to 2 × 106/mL on day 35 after primary immunization. Subsequently, the lymphocytes were stained with FITC anti-rabbit CD4, APC anti-rabbit CD3, and PE anti-rabbit CD8 fluorescence antibody (Abcam, Cambridge, England) in dark conditions at 37 °C for 30 min, respectively. After washing three times with sterile PBS, flow cytometry analysis was performed using a FACS Caliber (Becton Dickinson, Franklin Lakes, NJ, USA).
2.9. Lymphoproliferation Activity Test
Briefly, the concentration of spleen lymphocytes obtained from the NZW rabbits (n = 3) in each group was adjusted to 5 × 106 cells/mL, added to a 96-well plate (100 μL per cell in eight duplicates) containing RPMI 1640 plus 10% fetal bovine serum, and placed in a 5% CO2 incubator at 37 °C for 8–12 h. Subsequently, the cells were re-stimulated using purified α, β1, β2, and ε toxoid proteins at 0.01 mg/mL, 0.1 mg/mL, 1.0 mg/mL, or 10 mg/mL for 60 h, respectively, using RPMI 1640 culture medium as a blank control. Next, 5 mg/mL of thiazolyl blue tetrazolium bromide (MTT) was added into the cell plate (10 μL per well) and incubated at 37 °C for another 4 h followed by lymphoproliferation activity detection with CellTiter 96 AQueous Non-Radioactive Cell Proliferation Assay according to the manufacturer’s instructions (Promega, USA). The stimulation index was calculated as follows: SI = OD600(sample)/OD600(blank control).
2.10. Challenge Tests
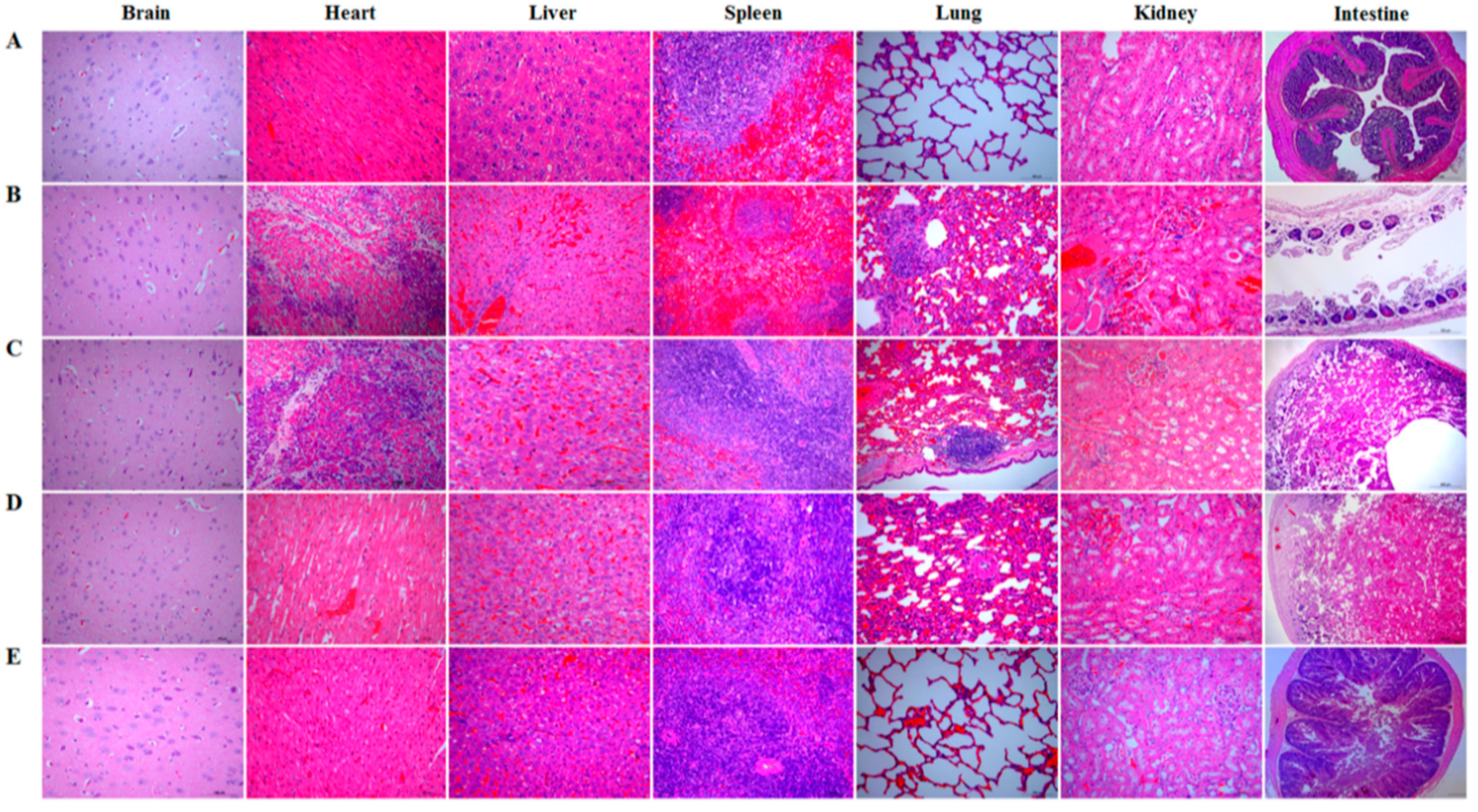
Briefly, on day 35 after the primary oral immunization, each rabbit (n = 10) in each group was intramuscularly injected with 1 × LD100 (90 mg of exotoxins mixture per kg body weight) of crude exotoxin mixture produced by C. perfringens toxinotype A, C, and D, and simultaneously orally challenged with 2 mL of C. perfringens toxinotype A, C, and D culture (the total concentration of culture mixture was 3 × 108 CFU/mL, and 1 × 108 CFU/mL for each toxinotype). The survival rate of rabbits in each group was recorded post-challenge, and histopathological changes in brain, heart, liver, spleen, lung, kidney, and intestine of rabbits in each group were observed.
2.11. Analysis of Intestinal Flora Diversity of Immunized Rabbits Challenged with C. perfringens
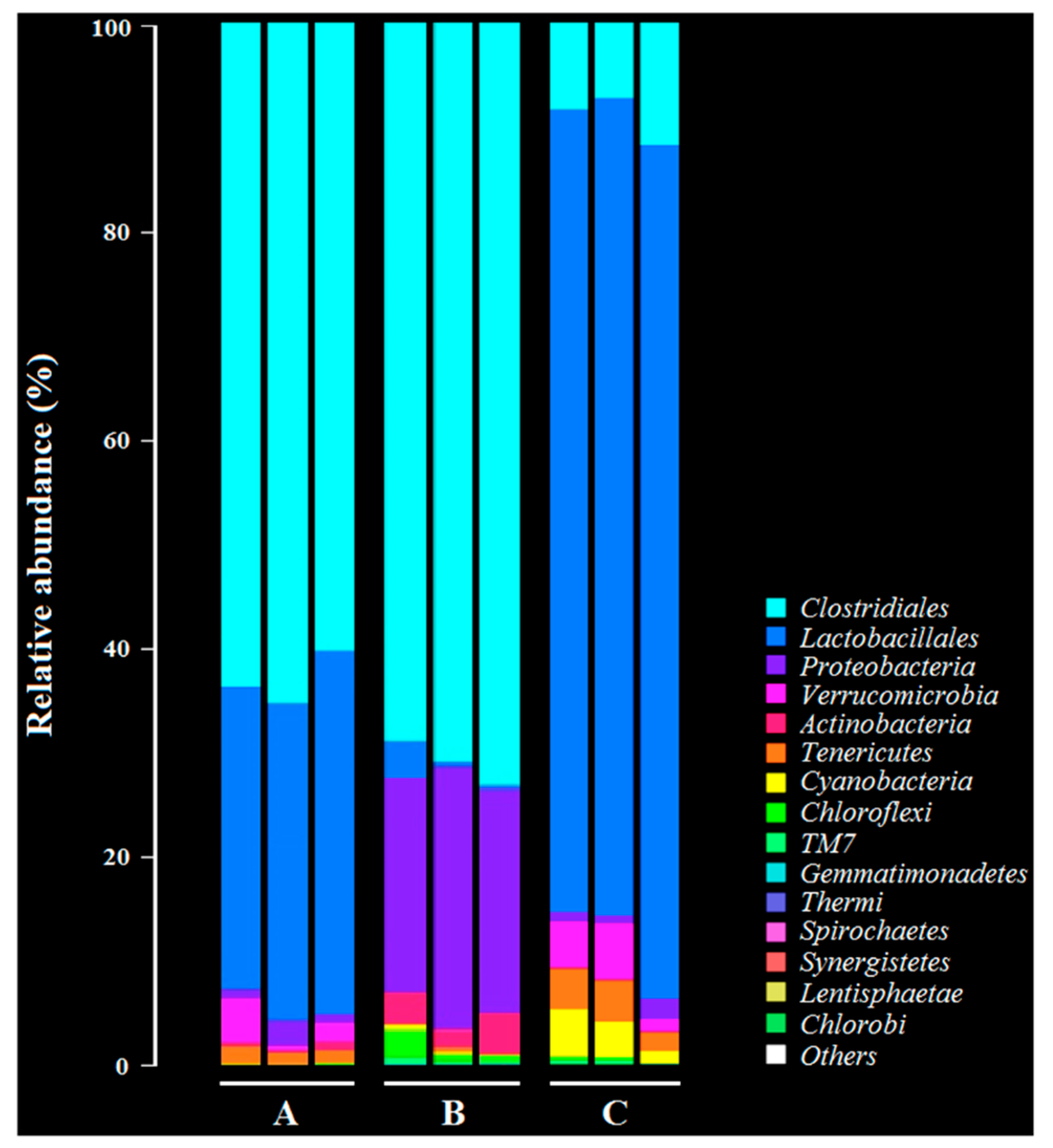
After challenging the rabbits with C. perfringens, we collected the fecal samples to analyze the changes in intestinal flora diversity of the immunized rabbits with the recombinant lactobacillus. Briefly, fecal samples were collected from the immunized rabbits (n = 3) on day 15 after oral challenge with C. perfringens. The Illumina MiSeq platform was used to perform 16S rRNA high throughout sequencing to analyze the intestinal flora diversity in fecal samples. In parallel, the fecal samples collected from rabbits in the PBS group and in the C. perfringens group were used as controls. Sequencing and data analysis was performed by Guangzhou Gene Denovo Biotechnology Co., Ltd., Guangzhou, China. The raw sequence reads were deposited into the NCBI database as a BioProject (accession number: PRJNA541228).
2.12. Statistical Analysis
GraphPad Prism V5.0 software (GraphPad Software, San Diego, CA, USA) was used to carry out statistical analyses for all data. The results are shown as means ± standard errors of five replicates per test in a single experiment repeated three times. Tukey’s multiple comparison tests and one-way analysis of variance (ANOVA) were used to analyze the significance of differences between groups. A p value < 0.05 (*) indicated statistical significance, and a p value < 0.01 (**) indicated highly significant differences.
4. Discussion
C. perfringens is an opportunistic pathogen commonly found in animal intestines. Exotoxins are the main virulence factors associated with
C. perfringens lethality, cytotoxicity, necrosis, and hemorrhagic activity. These exotoxins are mainly absorbed through the intestinal tracts of animals, resulting in enterotoxemia, dysentery, and hemorrhagic enteritis [
5,
25]. Although available commercial intramuscular injection vaccines (inactivated bacterins or subunit vaccines) have been used to prevent
C. perfringens-mediated diseases in livestock, these vaccines cannot effectively inhibit the invasion of pathogenic bacteria through intestinal tracts or toxin absorption through intestinal mucosa. Therefore, developing a vaccine that could effectively induce both antigen-specific intestinal mucosal and systemic immune responses against exotoxins is very important for preventing and controlling
C. perfringens-mediated diseases. An oral vaccine may be a promising strategy [
26].
In our previous study, we constructed a recombinant lactobacillus pPG-E-α-β2-ε-β1/
L. casei expressing tetravalent
C. perfringens toxoids. This showed a good immunogenicity in mice after oral administration [
24]. Here, a rabbit model was used to explore the immunogenicity and immune protection effect of the recombinant lactobacillus in livestock. Before immunization, we evaluated its segregational stability. Our data indicated that the recombinant lactobacillus had good stability properties with no target gene loss or structural rearrangement. Subsequently, we systemically evaluated the tolerance of the recombinant lactobacillus to digestive environments. Although there was an approximately 75% drop in recombinant lactobacillus in 0.5% bile salt conditions for 5 h and a gastric environment of pH 1.5 for 3 h, we found that the survival rate was efficient for the activity of the vaccine, which was confirmed by subsequent immunization experiment. In addition, in order to further enhance the immune effect of the recombinant lactobacillus, the oral immunization protocol was performed on three consecutive days (days 0, 1, and 2), and a booster immunization was given at days 14, 15, and 16, and a second booster was given at days 28, 29, and 30. The adhesion ability in the intestinal tracts is also important and desirable for live oral probiotic vaccine. After evaluating this characteristic, we found that the recombinant lactobacillus had good adhesion and colonization ability in the intestinal tracts of rabbits. In addition, we prepared the bait particle vaccine as a mixture of recombinant lactobacillus and feed raw materials to facilitate immunization via oral route; this reduced the workload and prevented injection stress.
As an oral vaccine, it is generally expected to efficiently induce both antigen-specific intestinal mucosal and systemic immune responses. Therefore, we evaluated the immunogenicity of the bait particle vaccine in rabbits via oral administration. Secretory IgA (sIgA) is the predominant antibody at the mucosal surface, and it plays a key role in host defense against mucosal infections [
27]. Here, we found that a significant level (
p < 0.05) of antigen-specific sIgA was elicited in the rabbits orally immunized with recombinant lactobacillus from the 7th day onwards, and an extremely significant level (
p < 0.01) was elicited from the 14th day onwards after the primary immunization. Moreover, a significant level (
p < 0.01) of antigen-specific IgG was elicited from the 21st day onward after the primary immunization. Along with the results obtained from our previous study using mice as animal model [
24], we can confirm that the recombinant lactobacillus can effectively induce antigen-specific intestinal mucosal and systemic immune responses after oral administration. Significantly, the antigen-specific sIgA and IgG antibodies that were induced by the bait vaccine showed effective
C. perfringens exotoxin-neutralizing activity that contributed to host immune defense by preventing the absorption of exotoxins in the gut and their entrance into the systemic circulation. Although modified lactobacillus has been used to express
C. perfringens α toxoid [
23] and ε toxoid [
28] for oral vaccine development, it cannot provide effective protection for the animals against other major exotoxins, especially against multiple α, β, and ε exotoxins at the same time.
Generally, lymphoproliferation positively correlates with cellular immunity. Our results indicated that the bait vaccine could induce cellular immunity. Significant levels (p < 0.01) of Th1-type (IL-2, IL-12, IFN-γ) and Th2-type (IL-4, IL-10) cytokines were detected in the sera of the rabbits immunized with the bait vaccine compared to control groups, indicating induction of mixed Th1- and Th2-type immune responses by the bait vaccine. We also determined the levels of CD4+ and CD8+ T cells, finding that the percentages of CD4+ and CD8+ T cells in splenocytes of the rabbits immunized with the bait vaccine increased significantly and indicating that vaccination with the bait vaccine could promote proliferation of CD4+ and CD8+ T lymphocytes.
A vaccine against C. perfringens-mediated diseases should provide effective immune protection against C. perfringens exotoxins in animals. Therefore, we evaluated the protective effect of the bait vaccine after a challenge model of exotoxins plus pathogens using a commercial inactivated C. perfringens vaccine as a vaccine control. The protection rates for the rabbits immunized with the bait vaccine and commercial vaccine were 80% and 60%, respectively, compared to group pPG-T7g10/L. casei or PBS (all rabbits died). Significantly, the protection effect provided by the bait vaccine was better than that provided by the commercial vaccine. Furthermore, the most significant histopathological changes between the groups were observed in the lung and intestine tissues. The lung and intestine tissues in the commercial vaccine group developed severe clinical symptoms, particularly the intestinal tissue which developed enteritis necroticans. This indicated that the commercial vaccine administered via injection could not provide effective protection for the intestine tissue against C. perfringens exotoxins, but the bait vaccine could. This possibly explains the lower protection rate of the commercial vaccine compared to the bait vaccine. Therefore, antigen-specific sIgA and IgG antibodies elicited by the bait vaccine contributed to the host immune defense by preventing diseases induced by exotoxin absorption in the gut and entrance into the systemic circulation. Moreover, we found that L. casei 393 had good probiotic properties, which could promote the elimination of pathogenic bacteria from the intestinal tracts, enhance animal feed intake and daily gain, increase feed utilization rate, and reduce the feed-to-weight ratio (data not shown).