Death from COVID-19 in a Fully Vaccinated Subject: A Complete Autopsy Report
Abstract
:1. Introduction
2. Materials and Methods
2.1. Case Description
2.2. Laboratory Investigation
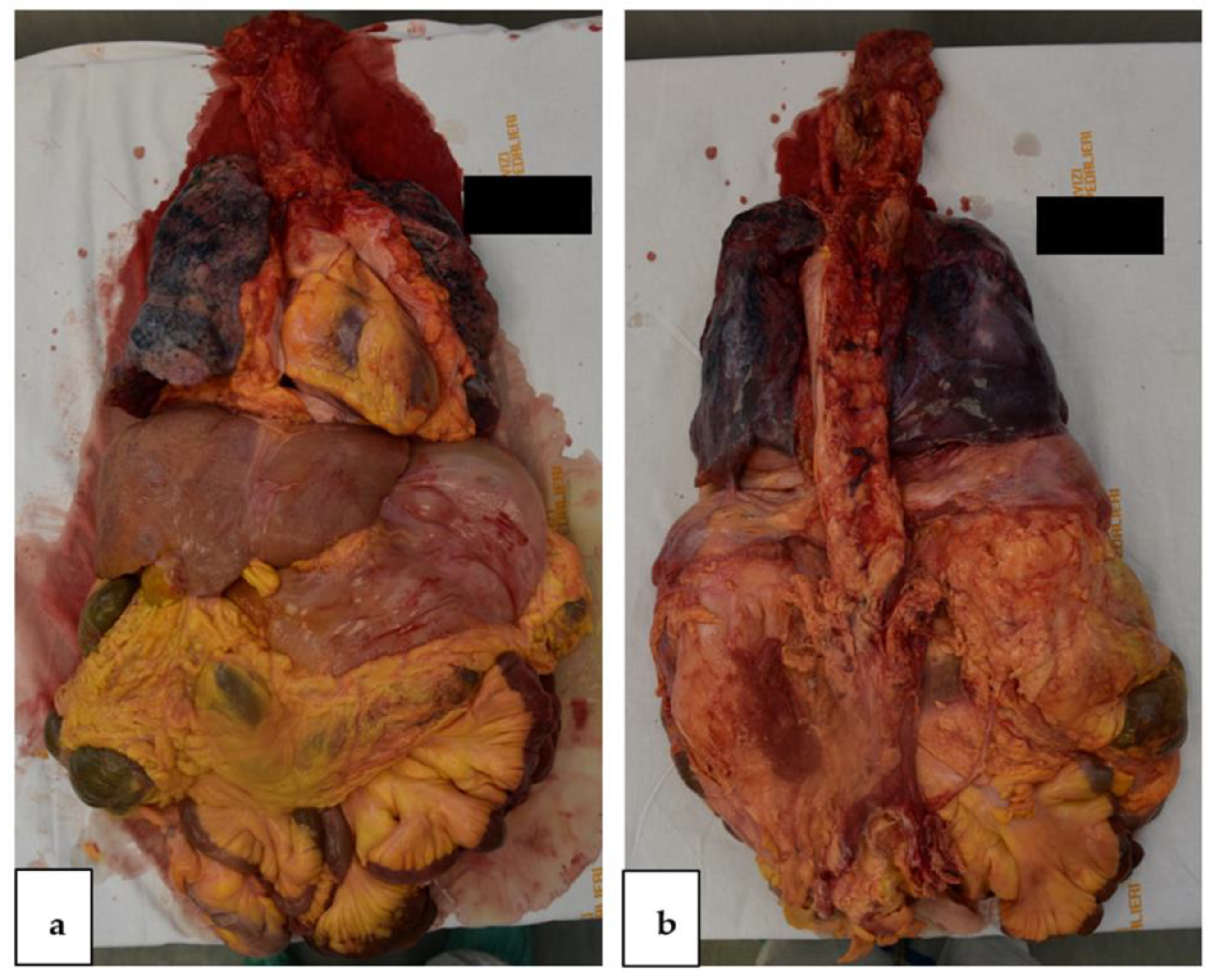
2.3. Autopsy Findings
2.4. Histological Analysis
2.5. Immunohistochemical Analysis
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leung, K.; Wu, J.T. Managing Waning Vaccine Protection against SARS-CoV-2 Variants. Lancet 2022, 399, 2–3. [Google Scholar] [CrossRef] [PubMed]
- Dejnirattisai, W.; Shaw, R.H.; Supasa, P.; Liu, C.; Stuart, A.S.; Pollard, A.J.; Liu, X.; Lambe, T.; Crook, D.; Stuart, D.I.; et al. Reduced Neutralisation of SARS-CoV-2 Omicron B.1.1.529 Variant by Post-Immunisation Serum. Lancet 2022, 399, 234–236. [Google Scholar] [CrossRef] [PubMed]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Robertson, C.; Stowe, J.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M.; et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca Vaccines on COVID-19 Related Symptoms, Hospital Admissions, and Mortality in Older Adults in England: Test Negative Case-Control Study. BMJ 2021, 373, n1088. [Google Scholar] [CrossRef]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Gallagher, E.; Simmons, R.; Thelwall, S.; Stowe, J.; Tessier, E.; Groves, N.; Dabrera, G.; et al. Effectiveness of COVID-19 Vaccines against the B.1.617.2 (Delta) Variant. N. Engl. J. Med. 2021, 385, 585–594. [Google Scholar] [CrossRef]
- Chalkias, S.; Harper, C.; Vrbicky, K.; Walsh, S.R.; Essink, B.; Brosz, A.; McGhee, N.; Tomassini, J.E.; Chen, X.; Chang, Y.; et al. A Bivalent Omicron-Containing Booster Vaccine against COVID-19. N. Engl. J. Med. 2022, 387, 1279–1291. [Google Scholar] [CrossRef]
- Dagan, N.; Barda, N.; Kepten, E.; Miron, O.; Perchik, S.; Katz, M.A.; Hernán, M.A.; Lipsitch, M.; Reis, B.; Balicer, R.D. BNT162b2 MRNA COVID-19 Vaccine in a Nationwide Mass Vaccination Setting. N. Engl. J. Med. 2021, 384, 1412–1423. [Google Scholar] [CrossRef]
- Harris, R.J.; Hall, J.A.; Zaidi, A.; Andrews, N.J.; Dunbar, J.K.; Dabrera, G. Effect of Vaccination on Household Transmission of SARS-CoV-2 in England. N. Engl. J. Med. 2021, 385, 759–760. [Google Scholar] [CrossRef]
- Jara, A.; Undurraga, E.A.; González, C.; Paredes, F.; Fontecilla, T.; Jara, G.; Pizarro, A.; Acevedo, J.; Leo, K.; Leon, F.; et al. Effectiveness of an Inactivated SARS-CoV-2 Vaccine in Chile. N. Engl. J. Med. 2021, 385, 875–884. [Google Scholar] [CrossRef]
- Andrews, N.; Stowe, J.; Kirsebom, F.; Toffa, S.; Rickeard, T.; Gallagher, E.; Gower, C.; Kall, M.; Groves, N.; O’Connell, A.-M.; et al. COVID-19 Vaccine Effectiveness against the Omicron (B.1.1.529) Variant. N. Engl. J. Med. 2022, 386, 1532–1546. [Google Scholar] [CrossRef]
- Hammerman, A.; Sergienko, R.; Friger, M.; Beckenstein, T.; Peretz, A.; Netzer, D.; Yaron, S.; Arbel, R. Effectiveness of the BNT162b2 Vaccine after Recovery from COVID-19. N. Engl. J. Med. 2022, 386, 1221–1229. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Requirement for Proof of COVID-19 Vaccination for Air Passengers. Available online: https://www.cdc.gov/coronavirus/2019-ncov/travelers/proof-of-vaccination.html#faq (accessed on 18 August 2022).
- Sessa, F.; Salerno, M.; Esposito, M.; di Nunno, N.; Zamboni, P.; Pomara, C. Autopsy Findings and Causality Relationship between Death and COVID-19 Vaccination: A Systematic Review. J. Clin. Med. 2021, 10, 5876. [Google Scholar] [CrossRef]
- Kwok, H.F. Review of COVID-19 Vaccine Clinical Trials - A Puzzle with Missing Pieces. Int. J. Biol. Sci. 2021, 17, 1461–1468. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2021 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Berlin, D.A.; Gulick, R.M.; Martinez, F.J. Severe COVID-19. N. Engl. J. Med. 2020, 383, 2451–2460. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, R.T.; Lynch, J.B.; del Rio, C. Mild or Moderate COVID-19. N. Engl. J. Med. 2020, 383, 1757–1766. [Google Scholar] [CrossRef]
- Grasselli, G.; Scaravilli, V.; Mangioni, D.; Scudeller, L.; Alagna, L.; Bartoletti, M.; Bellani, G.; Biagioni, E.; Bonfanti, P.; Bottino, N.; et al. Hospital-Acquired Infections in Critically Ill Patients With COVID-19. Chest 2021, 160, 454–465. [Google Scholar] [CrossRef]
- Pomara, C.; Salerno, M.; Sessa, F.; Esposito, M.; Barchitta, M.; Ledda, C.; Grassi, P.; Liberto, A.; Mattaliano, A.R.; Rapisarda, V.; et al. Safe Management Strategies in Clinical Forensic Autopsies of Confirmed COVID-19 Cases. Diagnostics 2021, 11, 457. [Google Scholar] [CrossRef]
- Musso, N.; Falzone, L.; Stracquadanio, S.; Bongiorno, D.; Salerno, M.; Esposito, M.; Sessa, F.; Libra, M.; Stefani, S.; Pomara, C. Post-Mortem Detection of SARS-CoV-2 RNA in Long-Buried Lung Samples. Diagnostics 2021, 11, 1158. [Google Scholar] [CrossRef]
- Pomara, C.; Sessa, F.; Galante, D.; Pace, L.; Fasanella, A.; di Nunno, N.; Esposito, M.; Salerno, M. Do We Really Need Hazard Prevention at the Expense of Safeguarding Death Dignity in COVID-19? Diagnostics 2021, 11, 1913. [Google Scholar] [CrossRef]
- Cipolloni, L.; Sessa, F.; Bertozzi, G.; Baldari, B.; Cantatore, S.; Testi, R.; D’Errico, S.; di Mizio, G.; Asmundo, A.; Castorina, S.; et al. Preliminary Post-Mortem COVID-19 Evidence of Endothelial Injury and Factor VIII Hyperexpression. Diagnostics 2020, 10, 575. [Google Scholar] [CrossRef]
- Zimmermann, A.K.; Camenisch, U.; Rechsteiner, M.P.; Bode-Lesniewska, B.; Rössle, M. Value of Immunohistochemistry in the Detection of BRAFV600E Mutations in Fine-Needle Aspiration Biopsies of Papillary Thyroid Carcinoma. Cancer Cytopathol. 2014. [Google Scholar] [CrossRef] [PubMed]
- Kapoor, P.; Deshmukh, R.S. VEGF: A Critical Driver for Angiogenesis and Subsequent Tumor Growth: An IHC Study. J. Oral Maxillofac. Pathol. 2012, 16, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Kukreja, I.; Kapoor, P.; Deshmukh, R.; Kulkarni, V. VEGF and CD 34: A Correlation between Tumor Angiogenesis and Microvessel Density-an Immunohistochemical Study. J. Oral Maxillofac. Pathol. 2013, 17, 367–373. [Google Scholar] [CrossRef] [PubMed]
- D’Errico, S.D.; Zanon, M.; Montanaro, M.; Radaelli, D.; Sessa, F.; di Mizio, G.; Montana, A.; Corrao, S.; Salerno, M.; Pomara, C.; et al. More than Pneumonia: Distinctive Features of SARS-CoV-2 Infection. from Autopsy Findings to Clinical Implications: A Systematic Review. Microorganisms 2020, 8, 1642. [Google Scholar] [CrossRef]
- Hippisley-Cox, J.; Coupland, C.A.C.; Mehta, N.; Keogh, R.H.; Diaz-Ordaz, K.; Khunti, K.; Lyons, R.A.; Kee, F.; Sheikh, A.; Rahman, S.; et al. Risk Prediction of COVID-19 Related Death and Hospital Admission in Adults after COVID-19 Vaccination: National Prospective Cohort Study. BMJ 2021, 374, n2244. [Google Scholar] [CrossRef]
- Heudel, P.; Favier, B.; Solodky, M.L.; Assaad, S.; Chaumard, N.; Tredan, O.; Bachelot, T.; Ray-Coquard, I.; Russias, B.; Fournier, M.L.; et al. Survival and Risk of COVID-19 after SARS-COV-2 Vaccination in a Series of 2391 Cancer Patients. Eur. J. Cancer 2022, 165, 174–183. [Google Scholar] [CrossRef]
- Esposito, M.; Salerno, M.; Scoto, E.; di Nunno, N.; Sessa, F. The Impact of the COVID-19 Pandemic on the Practice of Forensic Medicine: An Overview. Healthcare (Switzerland) 2022, 10, 319. [Google Scholar] [CrossRef]
- Putra, S.P.; Hidayat, T.; Zhuhra, R.T. SARS-CoV-2 persistence and infectivity in COVID-19 corpses: A systematic review. Forensic. Sci. Med. Pathol. 2022, 1–9. [Google Scholar] [CrossRef]
- Schneider, J.; Sottmann, L.; Greinacher, A.; Hagen, M.; Kasper, H.U.; Kuhnen, C.; Schlepper, S.; Schmidt, S.; Schulz, R.; Thiele, T.; et al. Postmortem Investigation of Fatalities Following Vaccination with COVID-19 Vaccines. Int. J. Leg. Med. 2021, 135, 2335. [Google Scholar] [CrossRef]
- Edler, C.; Klein, A.; Schröder, A.S.; Sperhake, J.P.; Ondruschka, B. Deaths Associated with Newly Launched SARS-CoV-2 Vaccination (Comirnaty®). Leg. Med. (Tokyo) 2021, 51, 101895. [Google Scholar] [CrossRef]
- Wiedmann, M.; Skattør, T.; Stray-Pedersen, A.; Romundstad, L.; Antal, E.A.; Marthinsen, P.B.; Sørvoll, I.H.; Leiknes Ernstsen, S.; Lund, C.G.; Holme, P.A.; et al. Vaccine Induced Immune Thrombotic Thrombocytopenia Causing a Severe Form of Cerebral Venous Thrombosis With High Fatality Rate: A Case Series. Front Neurol. 2021, 12, 721146. [Google Scholar] [CrossRef]
- Sessa, F.; Bertozzi, G.; Cipolloni, L.; Baldari, B.; Cantatore, S.; D’Errico, S.; di Mizio, G.; Asmundo, A.; Castorina, S.; Salerno, M.; et al. Clinical-Forensic Autopsy Findings to Defeat COVID-19 Disease: A Literature Review. J. Clin. Med. 2020, 9, 26. [Google Scholar] [CrossRef]
- Schaller, T.; Hirschbühl, K.; Burkhardt, K.; Braun, G.; Trepel, M.; Märkl, B.; Claus, R. Postmortem Examination of Patients with COVID-19. JAMA J. Am. Med. Assoc. 2020, 323, 2518–2520. [Google Scholar] [CrossRef]
- Hansen, T.; Titze, U.; Kulamadayil-Heidenreich, N.S.A.; Glombitza, S.; Tebbe, J.J.; Röcken, C.; Schulz, B.; Weise, M.; Wilkens, L. First Case of Postmortem Study in a Patient Vaccinated against SARS-CoV-2. Int. J. Infect. Dis. 2021, 107, 172–175. [Google Scholar] [CrossRef]
- Krause, P.R.; Fleming, T.R.; Peto, R.; Longini, I.M.; Figueroa, J.P.; Sterne, J.A.C.; Cravioto, A.; Rees, H.; Higgins, J.P.T.; Boutron, I.; et al. Considerations in Boosting COVID-19 Vaccine Immune Responses. Lancet 2021, 398, 1377–1380. [Google Scholar] [CrossRef]
- Pomara, C.; Li Volti, G.; Cappello, F. The Post-Lockdown Era: What Is next in Italy? Front Pharm. 2020, 11, 1074. [Google Scholar] [CrossRef]
- Albano, G.D.; Rifiorito, A.; Malta, G.; Sorrentino, E.S.; Falco, V.; Firenze, A.; Argo, A.; Zerbo, S. The Impact on Healthcare Workers of Italian Law n. 24/2017 “Gelli–Bianco” on Patient Safety and Medical Liability: A National Survey. Int. J. Environ. Res. Public Health 2022, 19, 8448. [Google Scholar] [CrossRef]
- Verma, M.; Sharma, S.; Kumar, A.; Hakim, A.; Bhansali, S.; Meena, R. Comorbidities and Vaccination Status of COVID-19 All-Cause Mortality at a Tertiary Care Center of Western India. Cureus 2022, 14, e21721. [Google Scholar] [CrossRef]
- Rodriguez, M.; López-Cepero, A.; Ortiz-Martínez, A.P.; Fernández-Repollet, E.; Pérez, C.M. Influence of Health Beliefs on COVID-19 Vaccination among Individuals with Cancer and Other Comorbidities in Puerto Rico. Vaccines 2021, 9, 994. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, E.S.; Dorabawila, V.; Easton, D.; Bauer, U.E.; Kumar, J.; Hoen, R.; Hoefer, D.; Wu, M.; Lutterloh, E.; Conroy, M.B.; et al. COVID-19 Vaccine Effectiveness in New York State. N. Engl. J. Med. 2022, 386, 116–127. [Google Scholar] [CrossRef]
- Piernas, C.; Patone, M.; Astbury, N.M.; Gao, M.; Sheikh, A.; Khunti, K.; Shankar-Hari, M.; Dixon, S.; Coupland, C.; Aveyard, P.; et al. Associations of BMI with COVID-19 Vaccine Uptake, Vaccine Effectiveness, and Risk of Severe COVID-19 Outcomes after Vaccination in England: A Population-Based Cohort Study. Lancet Diabetes Endocrinol. 2022, 10, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Andrews, N.; Tessier, E.; Stowe, J.; Gower, C.; Kirsebom, F.; Simmons, R.; Gallagher, E.; Thelwall, S.; Groves, N.; Dabrera, G.; et al. Duration of Protection against Mild and Severe Disease by COVID-19 Vaccines. N. Engl. J. Med. 2022, 386, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Andrews, N.; Stowe, J.; Kirsebom, F.; Toffa, S.; Sachdeva, R.; Gower, C.; Ramsay, M.; Lopez Bernal, J. Effectiveness of COVID-19 Booster Vaccines against COVID-19-Related Symptoms, Hospitalization and Death in England. Nat. Med. 2022, 28, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Arbel, R.; Sergienko, R.; Friger, M.; Peretz, A.; Beckenstein, T.; Yaron, S.; Netzer, D.; Hammerman, A. Effectiveness of a Second BNT162b2 Booster Vaccine against Hospitalization and Death from COVID-19 in Adults Aged over 60 Years. Nat. Med. 2022, 28, 1486–1490. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, P.; Scerrati, A.; Sessa, F.; Pomara, C.; Mannucci, P.M. Vaccine-Induced Immune Thrombotic Thrombocytopenia with Atypical Vein Thrombosis: Implications for Clinical Practice. Phlebology 2022. [Google Scholar] [CrossRef]
- Barouch, D.H. COVID-19 Vaccines — Immunity, Variants, Boosters. N. Engl. J. Med. 2022, 387, 1011–1020. [Google Scholar] [CrossRef]
- Pomara, C.; Sessa, F.; Ciaccio, M.; Dieli, F.; Esposito, M.; Giammanco, G.M.; Garozzo, S.F.; Giarratano, A.; Prati, D.; Rappa, F.; et al. COVID-19 Vaccine and Death: Causality Algorithm According to the WHO Eligibility Diagnosis. Diagnostics 2021, 11, 955. [Google Scholar] [CrossRef]
- Pomara, C.; Salerno, M.; Esposito, M.; Sessa, F.; Certo, F.; Tripodo, C.; Rappa, F.; Barbagallo, G.M. Histological and Immunohistochemical Findings in a Fatal Case of Thrombotic Thrombocytopenia after ChAdOx1 NCov-19 Vaccination. Pathol. Res. Pr. 2022, 231, 153796. [Google Scholar] [CrossRef]
- Pomara, C.; Sessa, F.; Ciaccio, M.; Dieli, F.; Esposito, M.; Garozzo, S.F.; Giarratano, A.; Prati, D.; Rappa, F.; Salerno, M.; et al. Post-Mortem Findings in Vaccine-Induced Thrombotic Thombocytopenia. Haematologica 2021, 106, 2291–2293. [Google Scholar] [CrossRef]
- Pomara, C.; Salerno, M.; Miani, A.; Piscitelli, P. Autopsies in COVID-Related Deaths: The Need for Following the Model of German COVID-19 Autopsy Registry. Lancet Reg. Health. Eur. 2022, 17, 100392. [Google Scholar] [CrossRef]
- François Watkins, L.K.; Mitruka, K.; Dorough, L.; Bressler, S.S.; Kugeler, K.J.; Sadigh, K.S.; Birhane, M.G.; Nolen, L.D.; Fischer, M. Characteristics of Reported Deaths Among Fully Vaccinated Persons With Coronavirus Disease 2019—United States, January–April 2021. Clin. Infect. Dis. 2022, 75, e645–e652. [Google Scholar] [CrossRef] [PubMed]
- Nohynek, H.; Wilder-Smith, A. Does the World Still Need New COVID-19 Vaccines? N. Engl. J. Med. 2022, 386, 2140–2142. [Google Scholar] [CrossRef] [PubMed]
- Larson, H.J.; Gakidou, E.; Murray, C.J.L. The Vaccine-Hesitant Moment. N. Engl. J. Med. 2022, 387, 58–65. [Google Scholar] [CrossRef]
- Morens, D.M.; Taubenberger, J.K.; Fauci, A.S. Universal Coronavirus Vaccines—An Urgent Need. N. Engl. J. Med. 2022, 386, 297–299. [Google Scholar] [CrossRef] [PubMed]


| Organ | Weight Grams (g) and Measurements Centimeters (cm) | Histological Findings |
|---|---|---|
| Brain | g 1470 cm 18 × 17 × 7 | Perivasal edema and perineuronal edema |
| Heart | g 433 cm 12 × 9 × 5 | Myofiber breakup and colliquative myocytolysis |
| Right lung | g 925 cm 23 × 11 × 9 | Interstitial lympho-plasma cell infiltrate, interstitial edema, and interstitial fibrosis |
| Left lung | g 601 cm 22 × 10 × 7 | |
| Liver | g 1085 cm 17 × 15 × 6 | Intraparenchymal polymorphonuclear granulocytes |
| Spleen | g 95 cm 12 × 3 × 3 | Stasis |
| Right kidney | g 45 cm 9 × 4 × 2 | Large areas of connective tissue replacement of the parenchyma |
| Left kidney | g 35 cm 8 × 5 × 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esposito, M.; Cocimano, G.; Vanaria, F.; Sessa, F.; Salerno, M. Death from COVID-19 in a Fully Vaccinated Subject: A Complete Autopsy Report. Vaccines 2023, 11, 142. https://doi.org/10.3390/vaccines11010142
Esposito M, Cocimano G, Vanaria F, Sessa F, Salerno M. Death from COVID-19 in a Fully Vaccinated Subject: A Complete Autopsy Report. Vaccines. 2023; 11(1):142. https://doi.org/10.3390/vaccines11010142
Chicago/Turabian StyleEsposito, Massimiliano, Giuseppe Cocimano, Fabrizio Vanaria, Francesco Sessa, and Monica Salerno. 2023. "Death from COVID-19 in a Fully Vaccinated Subject: A Complete Autopsy Report" Vaccines 11, no. 1: 142. https://doi.org/10.3390/vaccines11010142
APA StyleEsposito, M., Cocimano, G., Vanaria, F., Sessa, F., & Salerno, M. (2023). Death from COVID-19 in a Fully Vaccinated Subject: A Complete Autopsy Report. Vaccines, 11(1), 142. https://doi.org/10.3390/vaccines11010142









