Post-Vaccinal Encephalitis with Early Relapse after BNT162b2 (COMIRNATY) COVID-19 Vaccine: A Case Report
Abstract
:1. Introduction
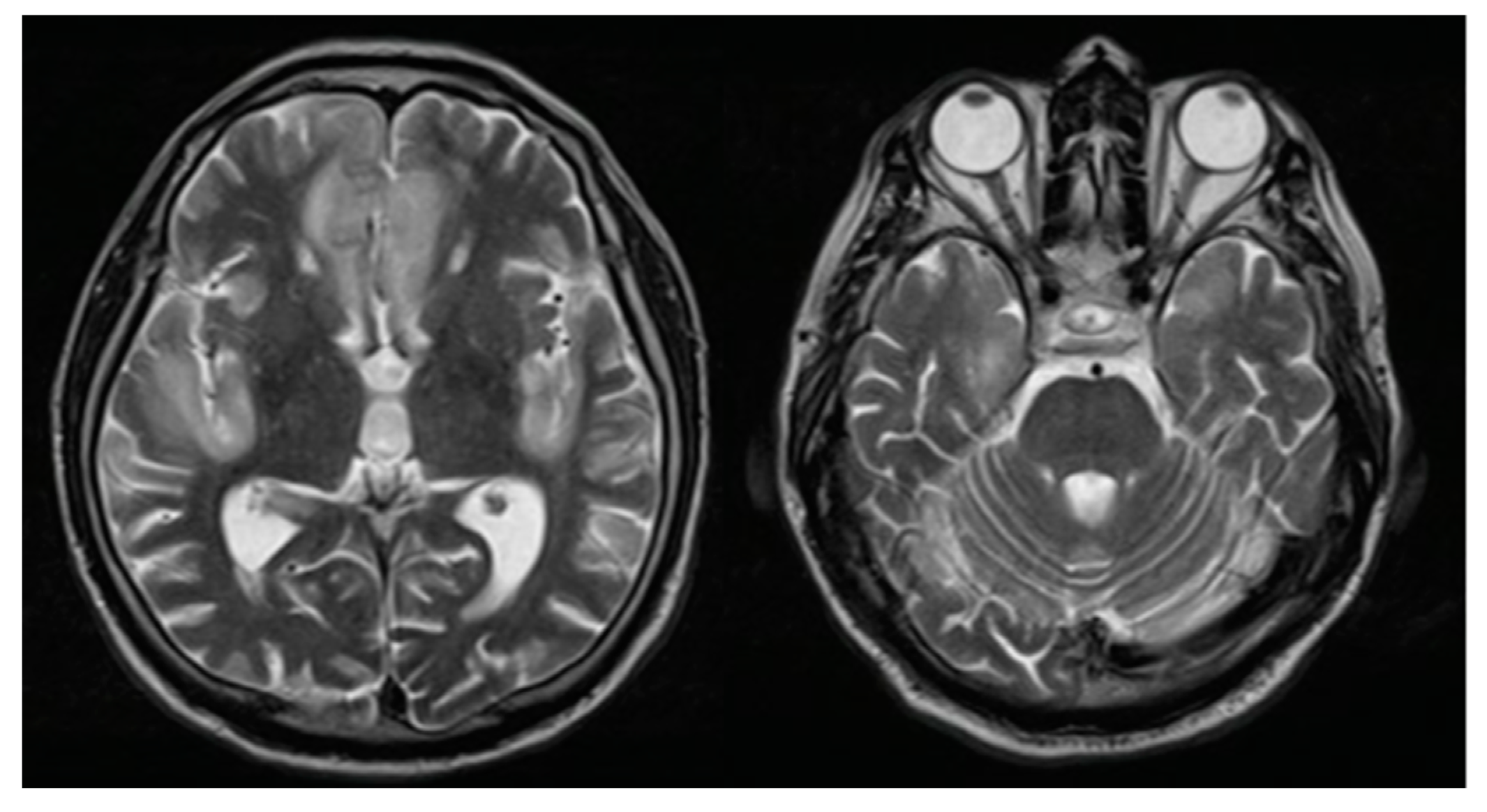
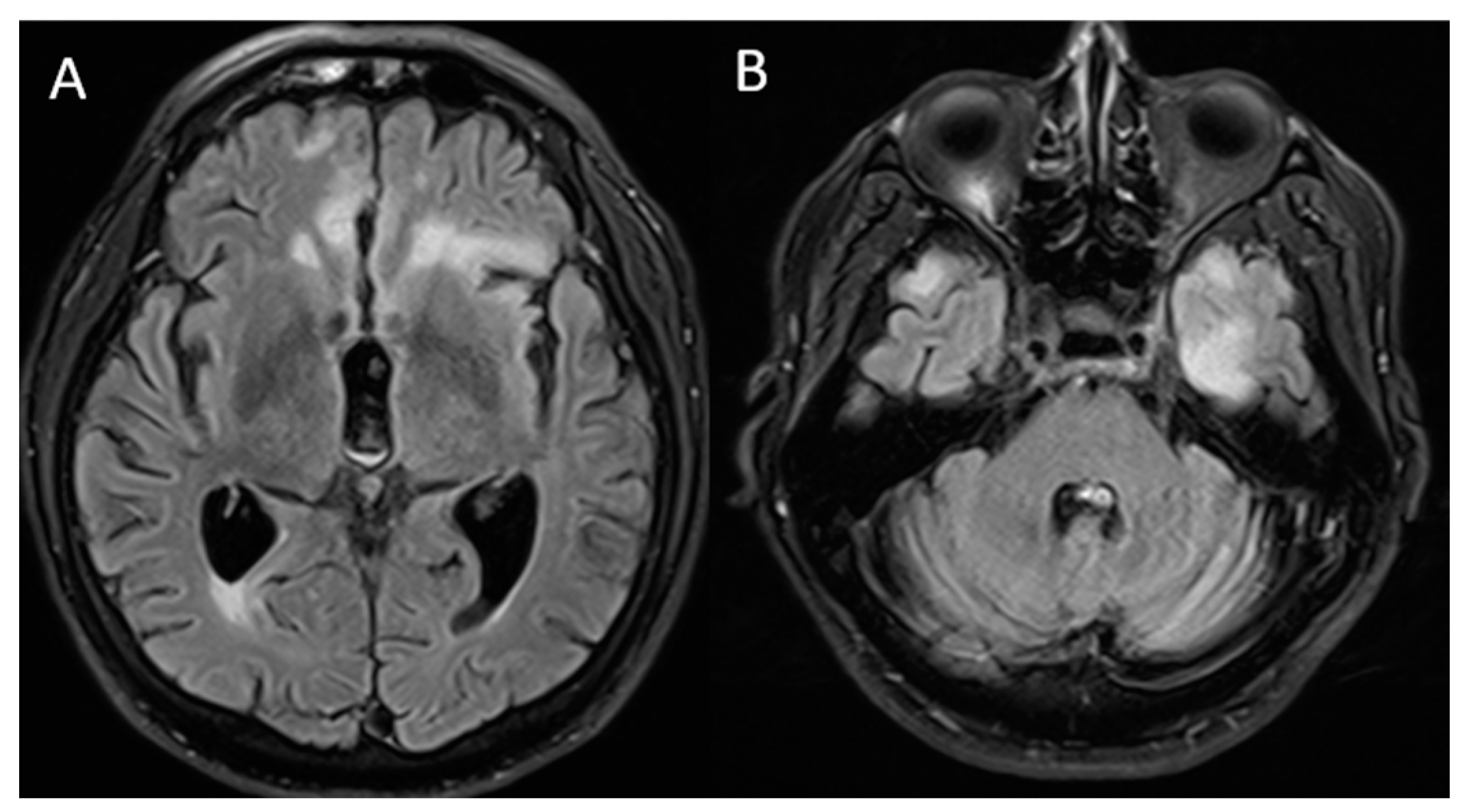
2. Case Report
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). WHO Coronavirus (COVID-19) Dashboard. WHO Coronavirus Disease (COVID-19). Available online: https://covid19.who.int/table (accessed on 23 February 2022).
- Marco, J.J.G.; Pasquín, M.J.Á.; Martín, S.M. Efectividad y seguridad de las vacunas para el SARS-CoV-2 actualmente disponibles. Form. Médica Contin. Atención Primaria 2021, 28, 442–451. [Google Scholar] [CrossRef] [PubMed]
- Zawbaa, H.M.; Osama, H.; El-Gendy, A.; Saeed, H.; Harb, H.S.; Madney, Y.M.; Abdelrahman, M.; Mohsen, M.; Ali, A.M.A.; Nicola, M.; et al. Effect of mutation and vaccination on spread, severity, and mortality of COVID-19 disease. J. Med. Virol. 2021, 94, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Patone, M.; Handunnetthi, L.; Saatci, D.; Pan, J.; Katikireddi, S.V.; Razvi, S.; Hunt, D.; Mei, X.W.; Dixon, S.; Zaccardi, F.; et al. Neurological complications after first dose of COVID-19 vaccines and SARS-CoV-2 infection. Nat. Med. 2021, 27, 2144–2153. [Google Scholar] [CrossRef] [PubMed]
- Finsterer, J.; Scorza, F.A.; Scorza, C.A. Post SARS-CoV-2 vaccination Guillain-Barre syndrome in 19 patients. Clinics 2021, 76, e3286. [Google Scholar] [CrossRef] [PubMed]
- Torrealba-Acosta, G.; Martin, J.C.; Huttenbach, Y.; Garcia, C.R.; Sohail, M.R.; Agarwal, S.K.; Wasko, C.; Bershad, E.M.; Hirzallah, M.I. Acute encephalitis, myoclonus and Sweet syndrome after mRNA-1273 vaccine. BMJ Case Rep. 2021, 14, e243173. [Google Scholar] [CrossRef] [PubMed]
- Raknuzzaman, M.; Jannaty, T.; Hossain, M.B.; Saha, B.; Dey, S.K.; Shahidullah, M. Post Covid19 vaccination acute disseminated encephalomyelitis: A case report in Bangladesh. Int. J. Med. Sci. Clin. Res. Stud. 2021, 1, 31–36. [Google Scholar]
- Kaulen, L.D.; Doubrovinskaia, S.; Mooshage, C.; Jordan, B.; Purrucker, J.; Haubner, C.; Seliger, C.; Lorenz, H.M.; Nagel, S.; Wildemann, B.; et al. Neurological autoimmune diseases following vaccinations against SARS-CoV-2: A case series. Eur. J. Neurol. 2021, 29, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Al-Mashdali, A.F.; Ata, Y.M.; Sadik, N. Post-COVID-19 vaccine acute hyperactive encephalopathy with dramatic response to methylprednisolone: A case report. Ann. Med. Surg. 2021, 69, 102803. [Google Scholar] [CrossRef] [PubMed]
- Zuliani, L.; Nosadini, M.; Gastaldi, M.; Spatola, M.; Iorio, R.; Zoccarato, M.; Mariotto, S.; De Gaspari, P.; Perini, F.; Ferrari, S.; et al. Management of antibody-mediated autoimmune encephalitis in adults and children: Literature review and consensus-based practical recommendations. Neurol. Sci. 2019, 40, 2017–2030. [Google Scholar] [CrossRef] [PubMed]
- Abu-Riash, A.; Tareef, A.B.; Aldwairy, A. Could Sinopharm COVID-19 Vaccine Cause Autoimmune Encephalitis? A Case Report. MAR Neurol. 2021, 3, 5. [Google Scholar]
- Flannery, P.; Yang, I.; Keyvani, M.; Sakoulas, G. Acute Psychosis Due to Anti-N-Methyl D-Aspartate Receptor Encephalitis Following COVID-19 Vaccination: A Case Report. Front. Neurol. 2021, 12, 764197. [Google Scholar] [CrossRef] [PubMed]
- Takata, J.; Durkin, S.M.; Wong, S.; Zandi, M.S.; Swanton, J.K.; Corrah, T.W. A case report of ChAdOx1 nCoV-19 vaccine–associated encephalitis. BMC Neurol. 2021, 21, 485. [Google Scholar] [CrossRef] [PubMed]
- Fan, H.T.; Lin, Y.Y.; Chiang, W.F.; Lin, C.Y.; Chen, M.H.; Wu, K.A.; Chan, J.S.; Kao, Y.H.; Shyu, H.Y.; Hsiao, P.J. COVID-19 vaccine-induced encephalitis and status epilepticus. QJM Int. J. Med. 2022, 115, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.-R.; Kim, B.-K.; Lee, S.-T.; Kim, A. Autoimmune Encephalitis as an Adverse Event of COVID-19 Vaccination. J. Clin. Neurol. 2022, 18, 114–116. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, G.D.A.; Barreira, R.M.; Antoniollo, K.E.N.T.; Pinheiro, A.M.N.; Maia, C.F.R.; Alves, D.M.B.S.; Nóbrega, P.R.; Braga-Neto, P. Autoimmune Encephalitis in Latin America: A Critical Review. Front. Neurol. 2021, 11, 1846. [Google Scholar] [CrossRef] [PubMed]
- Graus, F.; Titulaer, M.J.; Balu, R.; Benseler, S.; Bien, C.G.; Cellucci, T.; Cortese, I.; Dale, R.C.; Gelfand, J.M.; Geschwind, M.; et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016, 15, 391–404. [Google Scholar] [CrossRef] [Green Version]
- Abboud, H.; Probasco, J.C.; Irani, S.; Ances, B.; Benavides, D.R.; Bradshaw, M.; Christo, P.P.; Dale, R.C.; Fernandez-Fournier, M.; Flanagan, E.P.; et al. Autoimmune encephalitis: Proposed best practice recommendations for diagnosis and acute management. J. Neurol. Neurosurg. Psychiatry 2021, 92, 757–768. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Age (Sex) | Comorbidities | Onset Clinical Picture | Antineuronal Antibodies | Cerebrospinal Fluid | Immunization History: Type (Laboratory) and Number of Doses | Time between Vaccination and Symptoms | Imaging Findings | Relapse | Type of Treatment | Maintenance Treatment |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Vences M.A. (2022) In this study | 72 (Male) | High blood pressure and diabetes mellitus | Headache, confusion, aggressiveness, instability | Not performed | Elevated protein levels | mRNA (Pfizer) and two doses | 1st dose: 1 day and 2nd dose: 4 days | MRI: bilateral frontal and insular hyperintensity | Yes | Methylprednisolone, intravenous immunoglobulin | Six-monthly cycles of methylprednisolone |
| Abu-Riash A. (2021) [11] | 20 (Male) | None | Fatigue, tonic-clonic seizures, hallucinations, bilateral hand tremor and amnesia | Not described | Normal | Inactivated virus (Sinopharm) and two doses | 1 day | MRI: normal | No | Phenytoin, levetiracetam, teicoplanin, and methylprednisolone | Not described |
| Torrealba-Acosta G. (2021) [6] | 77 (Male) | Coronary artery disease, hyperlipidemia, and hypothyroidism | Confusion, fever and generalized rash | Normal | Normal | mRNA (Moderna) and one dose | 1 day | MRI: normal | No | Empiric broad-spectrum antibiotics, antiviral coverage, and methylprednisolone | Prednisone 60 mg daily for 3 weeks |
| Flannery P. (2021) [12] | 20 (Female) | None | Motor dysfunction, a transient aphasia, decreased mentally acuity, insomnia, somatization of bowel and kidney disease | 1:20 anti-NMDA | Mild lymphocyte pleocytosis with 12–14 nucleated cells/mm3 | mRNA (Pfizer) and one dose | 1 week | MRI and CT: normal | No | Intravenous immunoglobulin, methylprednisolone, rituximab | Not described |
| Takata J. (2021) [13] | 22 (Female) | Non-syndromic retinitis pigmentosa | Confusion, visual, and tactile hallucinations | Negative | Pleocytosis | Viral vector (AstraZeneca) and two doses | 3 weeks | MRI and CT: normal | No | Ceftriaxone, acyclovir, and olanzapine | Olanzapine 5 mg twice daily |
| H -T Fan (2022) [14] | 22 (Male) | None | Fever, blurred vision, and seizures | Negative | Elevated protein level | mRNA (Moderna) and two doses | 6 days | CT: normal, SPECT: mild hypoperfusion in the right temporal region | No | Levetiracetam, acyclovir, valproate sodium, and methylprednisolone | Not described |
| Hye-Rim Shin (2022) [15] | 35 (Female) | Intellectual disability | Dysarthria, anxiety, and reduced voluntary movements | Negative | Normal | Viral vector (AstraZeneca) and one dose | 5 days | MRI: mild swelling of the right hippocampus | No | Methylprednisolone, immunoglobulin, acyclovir, and rituximab | Not described |
| Al-Mashdali A. (2021) [9] | 32 (Male) | None | Agitation, disorientation to time, place, person, and memory disturbances | Negative | Elevated protein levels | mRNA (Moderna) and one dose | 2 days | MRI: normal | No | Ceftriaxone, acyclovir, and methylprednisolone | Not described |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vences, M.A.; Canales, D.; Albujar, M.F.; Barja, E.; Araujo-Chumacero, M.M.; Cardenas, E.; Alvarez, A.; Urrunaga-Pastor, D. Post-Vaccinal Encephalitis with Early Relapse after BNT162b2 (COMIRNATY) COVID-19 Vaccine: A Case Report. Vaccines 2022, 10, 1065. https://doi.org/10.3390/vaccines10071065
Vences MA, Canales D, Albujar MF, Barja E, Araujo-Chumacero MM, Cardenas E, Alvarez A, Urrunaga-Pastor D. Post-Vaccinal Encephalitis with Early Relapse after BNT162b2 (COMIRNATY) COVID-19 Vaccine: A Case Report. Vaccines. 2022; 10(7):1065. https://doi.org/10.3390/vaccines10071065
Chicago/Turabian StyleVences, Miguel A., Diego Canales, Maria Fe Albujar, Ebelin Barja, Mary M. Araujo-Chumacero, Edu Cardenas, Arturo Alvarez, and Diego Urrunaga-Pastor. 2022. "Post-Vaccinal Encephalitis with Early Relapse after BNT162b2 (COMIRNATY) COVID-19 Vaccine: A Case Report" Vaccines 10, no. 7: 1065. https://doi.org/10.3390/vaccines10071065
APA StyleVences, M. A., Canales, D., Albujar, M. F., Barja, E., Araujo-Chumacero, M. M., Cardenas, E., Alvarez, A., & Urrunaga-Pastor, D. (2022). Post-Vaccinal Encephalitis with Early Relapse after BNT162b2 (COMIRNATY) COVID-19 Vaccine: A Case Report. Vaccines, 10(7), 1065. https://doi.org/10.3390/vaccines10071065







