Regulation of Nrf2/ARE Pathway by Dietary Flavonoids: A Friend or Foe for Cancer Management?
Abstract
1. Introduction
1.1. Oxidative Stress and Antioxidant Defense System in Relation to Cancer
1.2. Mechanisms of Activation of the Antioxidant Defense System and Other Cytoprotective Genes
1.3. Hormetic Effects of Dietary Flavonoids
2. Role of the Nrf2/ARE Pathway in Cancer Chemoprevention
2.1. Activators of Nrf2/ARE in Non-Cancer Experimental Models
2.2. Flavonoids: Nrf2/ARE Activation in Non-Cancer Experimental Models
| Group | Compound | Effective Concentration | Experimental Model | Mode of Action | Reference |
|---|---|---|---|---|---|
| Phytochemicals–Polyphenols | |||||
| Phenolic acid | Ellagic acid | 25–50 µM | Human keratinocyte HaCaT cells | ↑ Nrf2 nuclear translocation. ↑ SOD enzyme activity. | [141] |
| Chlorogenic acid | 100 µM | Human retinal pigment epithelial ARPE-19 cells | ↑ mRNA expression of Nrf2 and SOD. | [142] | |
| 500 mg/kg of body weight orally | Sprague-Dawley rats | ↑ mRNA expression of Nrf2. ↑ SOD and GSH activities. | [143] | ||
| Proanthocyanidin | Procyanidin C1 | 5–10 µM | Mouse hippocampal neuronal HT22 cells | ↑ Nrf2 nuclear translocation. ↑ HO-1 protein. | [144] |
| Lignans | Sesamin | 100 mg/kg body weight intraperitoneally | C57BL/6 mice | ↑ SOD and CAT activities. ↑ GSH and Nrf2 protein. | [145] |
| 10 µM | Primary chondrocytes | ↑ Nrf2 and HO-1 proteins. | [146] | ||
| Coumarins | Fraxin | 50 mg/kg of body weight up to 5 days orally | Sprague-Dawley rats | ↑ cellular GSH levels. | [147] |
| Stilbene derivative | Resveratrol | 5 µM | Primary human coronary artery endothelial cells | ↑ mRNA expression of NQO1. | [148] |
| 500 mg in a tablet/day up to 30 days in the morning fasting | Phase 3 clinical trial on chronic subclinical inflammation and redox status | ↑ electrophilic modification of Keap1-Cys-151 | [47,85] | ||
| 215 mg in a tablet/day up to 52 weeks | Phase 2 clinical trials on Alzheimer’s disease | ↑ electrophilic modification of Keap1-Cys-151 | [47,149] | ||
| Curcumminoid | Curcumin | 5 µM | Human extravillous trophoblast HTR8/Sveo cells | ↑ CAT and GSH activities. | [150] |
| 400 mg/kg body weight/day orally up to 21 days | White Pekin ducklings | ↑ CAT, SOD, and GPx activities. | [151] | ||
| 800 mg/day in two capsules up to 7 days | Phase 3 clinical on diabetic nephropathy | ↑ electrophilic modifications of Keap1-Cys-151 | [47,82] | ||
| 15 µM | Human retinal pigment epithelial ARPE-19 cells | ↑ Nrf2 protein. ↑ HO-1 activity. | [152] | ||
| 15–30 µM | Porcine renal epithelial proximal tubule LLC PK1 cells | ↑ ARE binding activity. ↑ Nrf2 protein. ↑ HO-1 activity. | [153] | ||
| 10 µM | Rat kidney epithelial NRK-52E cells | ↑ ARE binding activity. | [153] | ||
| 200 mg/kg body weight twice a week for 6 weeks | Kunming (KM) mice | ↑ Nrf2 nuclear translocation. ↑ HO-1 and NQO-1 proteins. | [154] | ||
| Phytochemicals–Polyphenols–Flavonoids | |||||
| Flavone | Luteolin | 0.1 mg/kg body weight/day for 7 days at two time points orally. | ICR mice | ↑ Nrf2 nuclear translocation. ↑ HO-1 and NQO-1 proteins. | [51] |
| 10 mg/kg body weight intracerebrally injected | Sprague-Dawley rats | ↑ Nrf2 nuclear translocation. ↑ HO-1 and NQO-1 proteins. | [131] | ||
| 5–10 µM | Rat myoblast H9c2 cells | ↑ Nrf2 protein. ↑ mRNA expression of SOD, NQO-1 and HO-1. | [93] | ||
| 5 µM | Mouse testis sertoli TM4 cells | ↑ Nrf2 nuclear translocation. | [121] | ||
| 3,5-di-O-Methyl Gossypetin | 10–25 µg/mL | Human keratinocyte HaCaT cells | ↑ Nrf2 nuclear translocation. ↑ GSH, SOD and HO-1 proteins. | [55] | |
| Baicalein | 160 mg/kg/day for 8 weeks orally | T2DM Kunming mice | ↑ Nrf2 nuclear translocation. ↑ SOD, CAT, GSH proteins. | [120] | |
| 50 µM | Human liver L-02 cells | ↑ p62 protein. ↑ Nrf2 dissociation from Keap1. ↑ Nrf2 phosphorylation via phosphorylation of ERK1/2 and protein kinase C. | [117] | ||
| 20 µM | Human liver HL-7702 cells | ↑ Nrf2 nuclear translocation. ↑ SOD, CAT, GSH protein. | [120] | ||
| Baicalin | 50 mg/kg body weight twice after 2 and 12 h of subarachnoid hemorrhage intraperitoneally. | Sprague-Dawley rats | ↑ SOD, GSH, NQO-1, and Nrf2 proteins. ↑ mRNA expression of HO-1. | [94] | |
| 75 µM | Rat myoblast H9C2 cells | ↑ Nrf2 and HO-1 proteins. | [97] | ||
| 450 mg/kg body weight/day up to 7 days orally. | Chicken | ↑ Nrf2 and HO-1 proteins. ↑ mRNA expression of Nrf2 and HO-1. | [116] | ||
| 50 µM | Human liver L-02 cells | ↑ p62 protein. ↑ Nrf2 dissociation from Keap1. ↑ Nrf2 phosphorylation via phosphorylation of ERK1/2 and protein kinase C. | [117] | ||
| Chrysin | 10–25 µM | Rat hepatocytes | ↑ Nrf2 nuclear translocation via ERK2 signaling. ↑cellular GSH protein. ↑ ARE binding ability. | [124] | |
| Apigenin | 400 µM | Human retinal pigment epithelial ARPE-19 cells | ↑ mRNA expression of Nrf2. ↑ Nrf2 protein. ↑ Nrf2 nuclear translocation. ↑SOD, CAT, and GPx activities. | [95] | |
| 200 µM | Human renal tubular epithelial HK-2 cells | ↑ mRNA expression of Nrf2 and HO-1. ↑ Nrf2 protein. | [100] | ||
| Flavonol | Myricetin | 100 mg/kg/day for 6 weeks orally | Kungming mice | ↑ Nrf2 nuclear translocation. | [132] |
| Quercetin | 30 µM | Human keratinocyte HaCaT and BJ foreskin fibroblast cells | ↑ Nrf2 protein. | [102] | |
| 200 mg/kg body weight/day for 20 days orally | Broiler chicken | ↑ Nrf2 protein. ↑ Nrf2 nuclear translocation. ↑ SOD and CAT proteins. | [111] | ||
| 100 µM | Intestinal epithelial IEC-6 cells | ↑ Nrf2 nuclear translocation. | [155] | ||
| 10 µM | Human umbilical endothelial cells | ↑ Nrf2 protein. | [108] | ||
| Rutin | 44 µM | Human keratinocyte HaCaT cells | ↑ mRNA expression of HO-1 and NQO-1. | [140] | |
| Flavanone | Naringenin | 80 µM | Sprague-Dawley rat neuron cells | ↑ Nrf2, HO-1, and NQO-1 proteins. | [103] |
| 70 mg/kg body weight/day up to 4 days orally | C57BL/6 mice | ↑ Nrf2 protein. | [112] | ||
| Hesperidin | 50 mg/kg body weight for 28 days orally | Sprague-Dawley rat | ↑ Nrf2 and HO-1 proteins. ↑ mRNA expression of HO-1. | [104] | |
| Flavan-3-ol | Epicatechin | 10–100 µM | Primary astrocytes from WT and Nrf2 deficient KO mice | ↑ Nrf2 nuclear translocation. | [122] |
| Epigallocatechin-3-gallate (EGCG) | 40 mg/kg body weight/day for 3 days intraperitoneally or a single dose of 50 mg/kg body weight intraperitoneally | Sprague-Dawley rat | ↑ Nrf2 protein. | [105,156] | |
| Isoflavones | Genistein | 1 mg/kg body weight intraperitoneally | Sprague-Dawley rat | ↑ Nrf2 nuclear translocation. ↑ HO-1 protein. | [138] |
| Anthocyanin | Cyanidin-3-O-glucoside (C3G) | 20–40 µM | Human umbilical vein epithelial cells | ↑ Nrf2 nuclear translocation. ↑mRNA expression of NQO-1 and HO-1. | [128] |
| Chalcone | Butein | 20 µM | Human dental pulp cells | ↑ Nrf2 nuclear translocation. | [130] |
| Other Phytochemicals | |||||
| Sulfur-containing | Sulforaphane | 5 µM | Mouse skin JB6 P+ cells | ↑ Nrf2 nuclear translocation. ↑ HO-1 and NQO-1 proteins. | [157] |
| Diallyl sulfide | 15 µM | Human embryonic lung MRC-5 cells | Dissociates Nrf2 from Keap1 through phosphorylated ERK and p38 interactions. ↑ Nrf2 nuclear translocation. | [89] | |
| 150 mg/kg body weight/day intraperitoneally for 6 days | Wistar rats | ↑ Nrf2 protein. ↑SOD, CAT, GPx, GR, GST, and quinone reductase activities. | [158] | ||
| Alkaloids | Berberine | 200 mg/kg body weight/day orally for 16 weeks | Wistar rats | ↑ mRNA expression of Nrf2. | [159] |
| Vitamins | |||||
| Fat-soluble vitamins | Vitamin D | 40 000 U/kg/week of body weight intratracheally for 8 weeks | C57BL/6 mice | ↑ mRNA expression Aldo-keto reductase family 1 member C1 (AKR1C1) and GCLM. | [160] |
| Vitamin E | 100 mg/kg body weight/day intraperitoneally for 6 days | Balb/c mice | ↑ Nrf2 and HO-1 protein levels. | [161] | |
| Vitamin A | 100,000 U/kg body weight/day subcutaneously for 14 days | Wistar rats | ↑ Nrf2 nuclear translocation. ↑ HO-1 and NQO 1 proteins. | [162] | |
| Water-soluble vitamins | Vitamin C | 27–65 mg/kg body feed twice a day for 8 weeks | Juvenile Sillago sihama | ↑ mRNA expression of Nrf2, CAT, SOD, GPx, GR, and GST in intestine and liver cells. | [163] |
| Vitamin B2 | 30 mg/kg body weight/day intra-gastrically | APP/PS1 double transgenic mice | ↑ SOD, CAT, GSH, and GPx activities. ↑ Nrf2 expression. ↓ Keap1 expression. | [164,165] | |
| Endogenous Signaling Molecules | |||||
| Protein kinases | PI3K | N/A | Human retinal pigment epithelial RPE-19 cells | ↑ Nrf2 nuclear translocation. | [92] |
| JNK 1 & 2 | N/A | Human embryonic kidney HEK 293T cells | Phosphorylates Nrf2 at S212, S408, S558, S577, and T559. | [80] | |
| p38 | N/A | Human embryonic kidney HEK 293T cells | Phosphorylates Nrf2 at Ser212, Ser408, Ser558, Ser577, and Thre559. | [80] | |
| AMPK | N/A | Human embryonic kidney HEK 293T cells | Phosphorylates Nrf2 at the Ser558 residue. ↑ Nrf2 nuclear translocation. | [49] | |
| ERK2 | N/A | Human embryonic kidney HEK 293T cells | Phosphorylates Nrf2 at Ser212, Ser408, Ser558, Ser577, and Thre559. | [80] | |
| Casein kinase 2 | N/A | Human embryonic kidney HEK 293T cells | ↑ Nrf2 phosphorylation. ↑ Nrf2 nuclear translocation. | [90] | |
| PKC | N/A | New Zealand white rabbits | ↑ Nrf2 nuclear translocation. | [91] | |
| PERK | N/A | Mouse embryonic fibroblasts | Dissociates Nrf2 from Keap1 by phosphorylation of Nrf2. | [79] | |
| Autophagy-substrate proteins | Sequestosome 1 (p62) | N/A | Human aortic smooth muscle cells | Competes with Nrf2 to bind with Keap1. | [88] |
| Synthetic compounds | |||||
| Synthetic triterpenoids | Bradoxolone-methyl (CDDO-Me) | 25–50 mg/day for 52 weeks | Phase 2 clinical on diabetic nephropathy | ↑ electrophilic modification of Keap1-Cys-151. | [47,84] |
| Synthetic triterpenoids | RTA-408 (omaveloxolone) | 1% ophthalmic suspension for twice a day for 14 days | Phase 2 clinical trial on inflammation and pain following ocular surgery | ↑ electrophilic modification of Keap1-Cys-151. | [47,83] |
| Synthetic lignans | LGM2605 (Secoisolariciresinol diglucoside) | 50 µM | Murine peritoneal macrophages derived from C57BL/6J mice | ↑ mRNA expression of GST and redoxin reductase 1. | [166] |
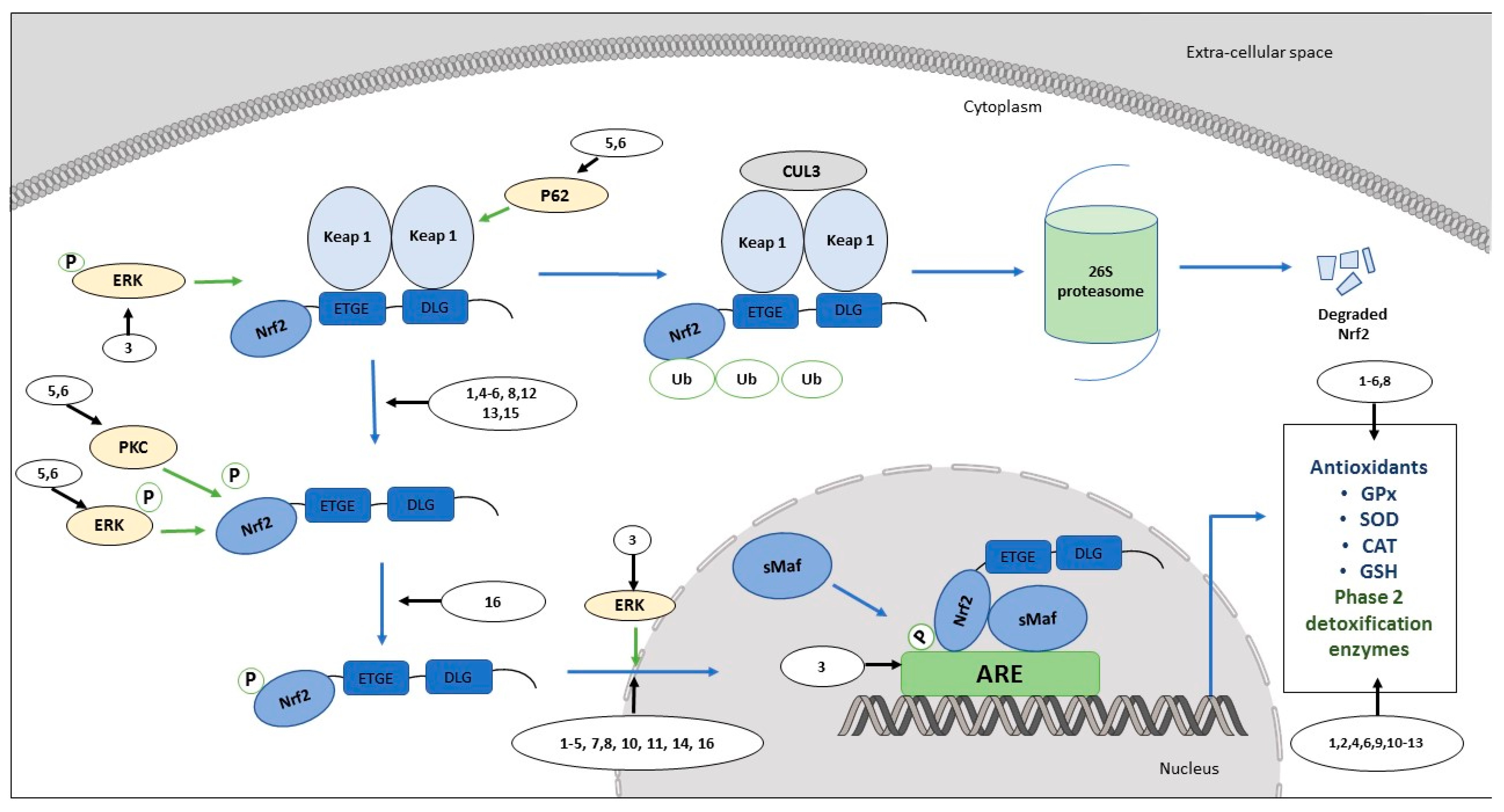
3. Promotion of Cancer Cell Proliferation by Activation of Nrf2/ARE: Nrf2-Associated Cell Signaling and Mechanisms
3.1. Flavonoids: Nrf2/ARE Activation in Cancer Cells
3.2. Flavonoids: Nrf2/ARE Inhibition in Cancer Cells
| Sub-Group | Compound | Effective Concentration/Concentration Range | Model | Mode of Action | Reference |
|---|---|---|---|---|---|
| Flavone | Luteolin | 30 µM | Human colorectal carcinoma HCT116 cells | ↑ mRNA expression of Nrf2. ↑ Nrf2 protein. | [211] |
| 10–15 µM | Human epithelial colorectal adenocarcinoma Caco-2 cells | ↑ Nrf2 nuclear translocation. ↑ GSH levels. | [72] | ||
| 1.2 mg/kg body weight/day intraperitoneal injection once a week for 3 weeks | Colorectal cancer in Balb/C mice | ↑ Nrf2 and GST proteins. | [200] | ||
| 1.5–6.25 µM | Human hepatocellular carcinoma HepG2 cells | ↑ mRNA and protein expression of Nrf2 and HO-1 via PI3K/Akt. | [65] | ||
| 1–10 µM | Human epithelial colorectal adenocarcinoma Caco-2 cells | ↓ mRNA expression of Nrf2, HO-1, NQO-1, aldo-keto reductases1C1 and C2 (AKR1C), glutamate-cysteine ligase catalytic subunit (GCLC), and multidrug resistance-associated protein (MRP) 2. | [61] | ||
| 5–10 µM | Human alveolar basal epithelial adenocarcinoma A549 cells | ↓ mRNA expression of HO-1, NQO-1, AKR1C, GCLC, and MRP2. ↓ Nrf2 protein. | [61] | ||
| 1–10 µM | Human breast cancer MCF7 cells | ↓ mRNA expression of Nrf2, HO-1, NQO-1, AKR1C, GCLC, and MRP2. | [61] | ||
| 25 µM | Opisthorchiasis-associated cholangiocarcinoma KKU-100 cells | ↓ Nrf2, gamma-glutamylcysteine ligase and HO-1 proteins | [60] | ||
| Chrysin | 10–20 µM | Hepatocellular carcinoma BEL-7402/ADM cells | ↓ mRNA and protein expression of Nrf2, HO-1, MRP5, and aldo-keto reductase family 1 member B10 (AKR1B10) | [63] | |
| 20 µM | Human breast cancer MCF7 cells | ↓ mRNA expression of Nrf2, MRP1, NQO-1, and HO-1. | [209] | ||
| Apigenin | 1.56–6.25 µM | Human hepatocellular carcinoma HepG2 cells | ↑ mRNA expression of Nrf2. ↑ Nrf2 and HO-1 proteins. ↑ mRNA expression of Nrf2 and HO-1. Activates PI3K/Akt and ERK1/2 signaling. | [65] | |
| 10–20 µM | Hepatocellular carcinoma BEL-7402/ADM cells | ↓ mRNA expression of Nrf2. ↓ mRNA and protein expression of HO-1, MRP5, and AKR1B10. | [62] | ||
| 100 µM | Human hepatocellular carcinoma HepG2 cells | ↓ mRNA expression of GSH and GPx. ↓ GSH and GPx activity. | [68] | ||
| Tangeretin | 20 µM | Human hepatocellular carcinoma HepG2 cells | ↑ Nrf2 nuclear translocation ↑mRNA and protein expression of HO-1 and NQO-1. | [212] | |
| Flavonol | Myricetin | 10–40 µM | Human hepatocellular carcinoma HepG2 cells | Activates Nrf2 by modifying Keap1 protein. ↓ Nrf2 ubiquitination. ↑ Nrf2 nuclear translocation. ↑ ARE binding ability. ↑ Nrf2 protein levels but not Keap1. ↑ protein expression of HO-1. | [203] |
| Quercetin | 10 µM | Human hepatocellular carcinoma HepG2 cells | ↑ Nrf2 nuclear translocation. ↑ARE binding activity of Nrf2. | [204] | |
| 10 µM | Human neuroblastoma SH-SY5Y cells | ↑ Nrf2 nuclear translocation. ↑ Nrf2 phosphorylation via PKC activation. ↑ protein and mRNA expression of glyoxalase-1. ↑ glyoxalase-1 activity | [123] | ||
| Rutin | 44 µM | Human epithelial colorectal adenocarcinoma Caco-2 cells | ↑ mRNA expression of Nrf2, HO-1 and NQO-1 without changing Keap1 mRNA levels. | [140] | |
| Flavanone | Hesperetin | 40 µM | Murine macrophage Raw 264.7 cells | ↑ Nrf2 nuclear translocation. ↑ degradation of Keap1. ↑ protein expression of HO-1 | [205] |
| Neohesperidin dihydrochalcone | 30 µM | Human hepatocellular carcinoma HepG2 cells | ↑ Keap1 modifications ↑ Nrf2 nuclear translocation. ↑ Nrf2 ARE binding ability. ↓ Nrf2 ubiquitination. ↑ phosphorylated JNK and p38 dependent protein expression of HO-1 and NQO-1. | [207] | |
| Naringenin | 20–80 µM | Human neuroblastoma SH-SY5Y cells | ↑ Nrf2 nuclear translocation. ↑ GSH protein ↑ protein expression of HO-1 | [112] | |
| Flavan-3-ol | Epicatechin | 10 µM | Human hepatocellular carcinoma HepG2 cells | ↑ Nrf2 phosphorylation. ↑ Nrf2 nuclear translocation. | [213] |
| Morin | 5–10 µM | Rat insulinoma INS-1E cells | ↑ Nrf2 phosphorylation. ↑ Nrf2 nuclear translocation. | [214] | |
| Isoflavones | Daidzein | 5 µM | Murine hepatoma Hepa-1c1c7 cells | ↑ mRNA expression of quinone reductase. ↑ quinone reductase activity. ↑ ARE binding ability | [208] |
| Genistein | 5 µM | Murine hepatoma Hepa-1c1c7 cells | ↑ mRNA expression of quinone reductase. ↑ quinone reductase activity. ↑ ARE binding ability. | [208] | |
| Chalcone | Phloretamide | 20 µM | Human hepatocellular carcinoma HepG2 cells | ↑ Nrf2 nuclear translocation. ↑mRNA expression of GST and NQO-1. | [215] |

4. Flavonoids and Nrf2/ARE: A Friend or Foe
5. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Akt | Protein kinase B |
| AMPK | AMP-activated protein kinase |
| Cul3 | Cullin 3 |
| ERK | Extracellular signal-regulated protein kinase |
| GPx | Glutathione peroxidase |
| GSH | Glutathione |
| GSK-3β | Glycogen synthase kinase 3 beta |
| HO-1 | Heme oxygenase 1 |
| HUVECs | Human umbilical vein epithelial cells |
| JNK | n- terminal kinase |
| Keap1 | Kelch-like ECH associated protein 1 |
| NADPH | Nicotine adenine dinucleotide phosphate |
| NQO1 | NAD(P)H quinone dehydrogenase 1 |
| Nrf2/ARE | Nuclear factor erythroid 2-related factor 2/Antioxidant response element |
| ROS | Reactive oxygen species |
| SOD | Superoxide dismutase |
| T2DM | Type 2 diabetes mellitus |
| t-BHP | tert-butyl hydroperoxide |
| p62 | Sequestosome 1 |
| PERK | Protein kinase-like endoplasmic reticulum-resident kinase |
| PI3K | Phosphatidylinositol 3-kinase |
| PKC | Protein kinase C |
| 6-OHDA | 6-hydroxydopamine |
References
- Hancock, J.T.; Desikan, R.; Neill, S.J. Role of reactive oxygen species in cell signalling pathways. Biochem. Soc. Trans. 2001, 29, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [PubMed]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Nimse, S.B.; Pal, D. Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv. 2015, 5, 27986–28006. [Google Scholar] [CrossRef]
- Bae, Y.S.; Oh, H.; Rhee, S.G.; Yoo, Y.D. Regulation of reactive oxygen species generation in cell signaling. Mol. Cells 2011, 32, 491–509. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.; Nguyen, L.N.; Akter, M.; Park, G.; Choi, E.H.; Kaushik, N.K. Impact of ROS generated by chemical, physical, and plasma techniques on cancer attenuation. Cancers 2019, 11, 1030. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Gorinstein, S.; Böhm, V.; Schaich, K.M.; Özyürek, M.; Güçlü, K. Methods of measurement and evaluation of natural antioxidant capacity/activity (IUPAC Technical Report). Pure Appl. Chem. 2013, 85, 957–998. [Google Scholar] [CrossRef]
- Lodish, H.; Berk, A.; Zipursky, S.L.; Matsudaira, P.; Baltimore, D.; Darnell, J. DNA damage and repair and their role in carcinogenesis. In Molecular Cell Biology, 4th ed.; W.H. Freeman and Company: New York, NY, USA, 2000. [Google Scholar]
- Sipowicz, M.A.; Amin, S.; Desai, D.; Kasprzak, K.S.; Anderson, L.M. Oxidative DNA damage in tissues of pregnant female mice and fetuses caused by the tobacco-specific nitrosamine, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK). Cancer Lett. 1997, 117, 87–91. [Google Scholar] [CrossRef]
- Wajed, S.A.; Laird, P.W.; DeMeester, T.R. DNA methylation: An alternative pathway to cancer. Ann. Surg. 2001, 234, 10–20. [Google Scholar] [CrossRef]
- Broustas, C.G.; Lieberman, H.B. DNA damage response genes and the development of cancer metastasis. Radiat. Res. 2014, 181, 111–130. [Google Scholar] [CrossRef]
- George, V.C.; Dellaire, G.; Rupasinghe, H.P.V. Plant flavonoids in cancer chemoprevention: Role in genome stability. J. Nutr. Biochem. 2017, 45, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Trachootham, D.; Lu, W.; Ogasawara, M.A.; Valle, N.R.-D.; Huang, P. Redox regulation of cell survival. Antioxid. Redox Signal. 2008, 10, 1343–1374. [Google Scholar] [CrossRef] [PubMed]
- Ighodaro, O.M.; Akinloye, O.A. First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): Their fundamental role in the entire antioxidant defence grid. Alex. J. Med. 2018, 54, 287–293. [Google Scholar] [CrossRef]
- Kalam, S.; Singh, R.; Mani, A.; Patel, J.; Khan, F.N.; Pandey, A. Antioxidants: Elixir of life. Int. Multidiscip. Res. J. 2012, 2, 18–34. [Google Scholar]
- Finkel, T. Redox-dependent signal transduction. FEBS Lett. 2000, 476, 52–54. [Google Scholar] [CrossRef]
- Vignais, P.V. The superoxide-generating NADPH oxidase: Structural aspects and activation mechanism. Cell. Mol. Life Sci. 2002, 59, 1428–1459. [Google Scholar] [CrossRef]
- Arteaga, O.; Álvarez, A.; Revuelta, M.; Santaolalla, F.; Urtasun, A.; Hilario, E. Role of antioxidants in neonatal hypoxic-ischemic brain injury: New therapeutic approaches. Int. J. Mol. Sci. 2017, 18, 265. [Google Scholar] [CrossRef] [PubMed]
- Sirokmány, G.; Geiszt, M. The relationship of NADPH oxidases and heme peroxidases: Fallin’ in and out. Front. Immunol. 2019, 10, 394. [Google Scholar] [CrossRef]
- Kelley, E.E.; Khoo, N.K.H.; Hundley, N.J.; Malik, U.Z.; Freeman, B.A.; Tarpey, M.M. Hydrogen peroxide is the major oxidant product of xanthine oxidase. Free Radic. Biol. Med. 2010, 48, 493–498. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, Q.; Sun, M.; Teng, M.; Niu, L. Hydrogen peroxide produced by two amino acid oxidases mediates antibacterial actions. J. Microbiol. 2004, 42, 336–339. [Google Scholar]
- Elsner, M.; Gehrmann, W.; Lenzen, S. Peroxisome-generated hydrogen Peroxide as Important Mediator of Lipotoxicity in Insulin-Producing Cells. Diabetes 2011, 60, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Hinkhouse, M.M.; Sun, W.; Weydert, C.J.; Ritchie, J.M.; Oberley, L.W.; Cullen, J.J. Redox regulation of pancreatic cancer cell growth: Role of glutathione peroxidase in the suppression of the malignant phenotype. Hum. Gene Ther. 2004, 15, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Lyngsie, G.; Krumina, L.; Tunlid, A.; Persson, P. Generation of hydroxyl radicals from reactions between a dimethoxyhydroquinone and iron oxide nanoparticles. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Kehrer, J.P. The Haber-Weiss reaction and mechanisms of toxicity. Toxicology 2000, 149, 43–50. [Google Scholar] [CrossRef]
- Jeeva, J.S.; Sunitha, J.; Ananthalakshmi, R.; Rajkumari, S.; Ramesh, M.; Krishnan, R. Enzymatic antioxidants and its role in oral diseases. J. Pharm. Bioallied. Sci. 2015, 7, S331–S333. [Google Scholar] [PubMed]
- Kim, S.J.; Kim, H.S.; Seo, Y.R. Understanding of ROS-inducing strategy in anticancer therapy. Oxid. Med. Cell. Longev. 2019, 2019, e5381692. [Google Scholar] [CrossRef] [PubMed]
- Storz, P. KRas, ROS and the initiation of pancreatic cancer. Small GTPases 2017, 8, 38–42. [Google Scholar] [CrossRef]
- Trachootham, D.; Alexandre, J.; Huang, P. Targeting cancer cells by ROS-mediated mechanisms: A radical therapeutic approach? Nat. Rev. Drug Discov. 2009, 8, 579–591. [Google Scholar] [CrossRef]
- Wang, J.; Yi, J. Cancer cell killing via ROS: To increase or decrease, that is the question. Cancer Biol. Ther. 2008, 7, 1875–1884. [Google Scholar] [CrossRef]
- Raza, M.H.; Siraj, S.; Arshad, A.; Waheed, U.; Aldakheel, F.; Alduraywish, S.; Arshad, M. ROS-modulated therapeutic approaches in cancer treatment. J. Cancer Res. Clin. Oncol. 2017, 143, 1789–1809. [Google Scholar] [CrossRef]
- Otsuki, A.; Yamamoto, M. Cis-element architecture of Nrf2–sMaf heterodimer binding sites and its relation to diseases. Arch. Pharm. Res. 2020, 43, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Lu, H.; Bai, Y. Nrf2 in cancers: A double-edged sword. Cancer Med. 2019, 8, 2252–2267. [Google Scholar] [CrossRef] [PubMed]
- Rojo de la Vega, M.; Chapman, E.; Zhang, D.D. NRF2 and the hallmarks of cancer. Cancer Cell 2018, 34, 21–43. [Google Scholar] [CrossRef] [PubMed]
- Krajka-Kuźniak, V.; Paluszczak, J.; Baer-Dubowska, W. The Nrf2-ARE signaling pathway: An update on its regulation and possible role in cancer prevention and treatment. Pharmacol. Rep. 2017, 69, 393–402. [Google Scholar] [CrossRef]
- Tsushima, M.; Liu, J.; Hirao, W.; Yamazaki, H.; Tomita, H.; Itoh, K. Emerging evidence for crosstalk between Nrf2 and mitochondria in physiological homeostasis and in heart disease. Arch. Pharm. Res. 2019, 43, 21–43. [Google Scholar] [CrossRef]
- Furfaro, A.L.; Traverso, N.; Domenicotti, C.; Piras, S.; Moretta, L.; Marinari, U.M.; Pronzato, M.A.; Nitti, M. The Nrf2/HO-1 axis in cancer cell growth and chemoresistance. Oxid. Med. Cell. Longev. 2016, 2016, 1958174. [Google Scholar] [CrossRef]
- Stefanson, A.L.; Bakovic, M. Dietary regulation of Keap1/Nrf2/ARE pathway: Focus on plant-derived compounds and trace minerals. Nutrients 2014, 6, 3777–3801. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, H.; Davies, K.J.A.; Sioutas, C.; Finch, C.E.; Morgan, T.E.; Forman, H.J. Nrf2-regulated phase II enzymes are induced by chronic ambient nanoparticle exposure in young mice with age-related impairments. Free Radic. Biol. Med. 2012, 52, 2038–2046. [Google Scholar] [CrossRef]
- Fernando, W.; Rupasinghe, H.P.V.; Hoskin, D.W. Dietary phytochemicals with anti-oxidant and pro-oxidant activities: A double-edged sword in relation to adjuvant chemotherapy and radiotherapy? Cancer Lett. 2019, 452, 168–177. [Google Scholar] [CrossRef]
- Cuadrado, A. Structural and functional characterization of Nrf2 degradation by glycogen synthase kinase 3/β-TrCP. Free Radic. Biol. Med. 2015, 88, 147–157. [Google Scholar] [CrossRef]
- Tong, K.I.; Katoh, Y.; Kusunoki, H.; Itoh, K.; Tanaka, T.; Yamamoto, M. Keap1 recruits Neh2 through binding to ETGE and DLG motifs: Characterization of the two-site molecular recognition model. Mol. Cell. Biol. 2006, 26, 2887–2900. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, A.; Kang, M.-I.; Okawa, H.; Ohtsuji, M.; Zenke, Y.; Chiba, T.; Igarashi, K.; Yamamoto, M. Oxidative stress sensor Keap1 functions as an adaptor for Cul3-based E3 ligase to regulate proteasomal degradation of Nrf2. Mol. Cell. Biol. 2004, 24, 7130–7139. [Google Scholar] [CrossRef] [PubMed]
- Cuadrado, A.; Manda, G.; Hassan, A.; Alcaraz, M.J.; Barbas, C.; Daiber, A.; Ghezzi, P.; León, R.; López, M.G.; Oliva, B.; et al. Transcription factor NRF2 as a therapeutic target for chronic diseases: A systems medicine approach. Pharmacol. Rev. 2018, 70, 348–383. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, M.; Kensler, T.W.; Motohashi, H. The KEAP1-NRF2 system: A thiol-based sensor-effector apparatus for maintaining redox homeostasis. Physiol. Rev. 2018, 98, 1169–1203. [Google Scholar] [CrossRef] [PubMed]
- Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Cole, R.N.; Itoh, K.; Wakabayashi, N.; Katoh, Y.; Yamamoto, M.; Talalay, P. Direct evidence that sulfhydryl groups of Keap1 are the sensors regulating induction of phase 2 enzymes that protect against carcinogens and oxidants. Proc. Natl. Acad. Sci. USA 2002, 99, 11908–11913. [Google Scholar] [CrossRef]
- Robledinos-Antón, N.; Fernández-Ginés, R.; Manda, G.; Cuadrado, A. Activators and inhibitors of NRF2: A review of their potential for clinical development. Oxid. Med. Cell. Longev. 2019, 2019, 9372182. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Dinkova-Kostova, A.T. The Nrf2 regulatory network provides an interface between redox and intermediary metabolism. Trends Biochem. Sci. 2014, 39, 199–218. [Google Scholar] [CrossRef]
- Joo, M.S.; Kim, W.D.; Lee, K.Y.; Kim, J.H.; Koo, J.H.; Kim, S.G. AMPK facilitates nuclear accumulation of Nrf2 by phosphorylating at Serine 550. Mol. Cell. Biol. 2016, 36, 1931–1942. [Google Scholar] [CrossRef]
- Zimmermann, K.; Baldinger, J.; Mayerhofer, B.; Atanasov, A.G.; Dirsch, V.M.; Heiss, E.H. Activated AMPK boosts the Nrf2/HO-1 signaling axis–A role for the unfolded protein response. Free Radic. Biol. Med. 2015, 88, 417–426. [Google Scholar] [CrossRef]
- Kitakaze, T.; Makiyama, A.; Yamashita, Y.; Ashida, H. Low dose of luteolin activates Nrf2 in the liver of mice at start of the active phase but not that of the inactive phase. PLoS ONE 2020, 15, e0231403. [Google Scholar] [CrossRef]
- Huang, Y.; Li, W.; Su, Z.; Kong, A.-N.T. The complexity of the Nrf2 pathway: Beyond the antioxidant response. J. Nutr. Biochem. 2015, 26, 1401–1413. [Google Scholar] [CrossRef] [PubMed]
- Abed, D.A.; Goldstein, M.; Albanyan, H.; Jin, H.; Hu, L. Discovery of direct inhibitors of Keap1–Nrf2 protein–protein interaction as potential therapeutic and preventive agents. Acta Pharm. Sin. B 2015, 5, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.-A.; Kwak, M.-K. The Nrf2 system as a potential target for the development of indirect antioxidants. Molecules 2010, 15, 7266–7291. [Google Scholar] [CrossRef] [PubMed]
- Sobeh, M.; Petruk, G.; Osman, S.; El Raey, M.A.; Imbimbo, P.; Monti, D.M.; Wink, M. Isolation of myricitrin and 3,5-di-O-methyl gossypetin from Syzygium samarangense and Evaluation of their involvement in protecting keratinocytes against oxidative stress via activation of the Nrf-2 pathway. Molecules 2019, 24, 1839. [Google Scholar] [CrossRef] [PubMed]
- Harris, I.S.; Treloar, A.E.; Inoue, S.; Sasaki, M.; Gorrini, C.; Lee, K.C.; Yung, K.Y.; Brenner, D.; Knobbe-Thomsen, C.B.; Cox, M.A.; et al. Glutathione and thioredoxin antioxidant pathways synergize to drive cancer initiation and progression. Cancer Cell 2015, 27, 211–222. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.-S.; Du, G.-Y.; Zhang, Z.-G.; Zhou, Z.; Sun, H.-L.; Yu, X.-Y.; Shi, Y.-T.; Xiong, D.-N.; Li, H.; Huang, Y.-H. NRF2 facilitates breast cancer cell growth via HIF1ɑ-mediated metabolic reprogramming. Int. J. Biochem. Cell Biol. 2018, 95, 85–92. [Google Scholar] [CrossRef]
- Lu, K.; Alcivar, A.L.; Ma, J.; Foo, T.K.; Zywea, S.; Mahdi, A.; Huo, Y.; Kensler, T.W.; Gatza, M.L.; Xia, B. NRF2 induction supporting breast cancer cell survival is enabled by oxidative stress–induced DPP3–KEAP1 interaction. Cancer Res. 2017, 77, 2881–2892. [Google Scholar] [CrossRef]
- Panieri, E.; Saso, L. Potential applications of NRF2 inhibitors in cancer therapy. Oxid. Med. Cell. Longev. 2019, 2019, 8592348. [Google Scholar] [CrossRef]
- Kittiratphatthana, N.; Kukongviriyapan, V.; Prawan, A.; Senggunprai, L. Luteolin induces cholangiocarcinoma cell apoptosis through the mitochondrial-dependent pathway mediated by reactive oxygen species. J. Pharm. Pharmacol. 2016, 68, 1184–1192. [Google Scholar] [CrossRef]
- Tang, X.; Wang, H.; Fan, L.; Wu, X.; Xin, A.; Ren, H.; Wang, X.J. Luteolin inhibits Nrf2 leading to negative regulation of the Nrf2/ARE pathway and sensitization of human lung carcinoma A549 cells to therapeutic drugs. Free Radic. Biol. Med. 2011, 50, 1599–1609. [Google Scholar] [CrossRef]
- Gao, A.-M.; Ke, Z.-P.; Wang, J.-N.; Yang, J.-Y.; Chen, S.-Y.; Chen, H. Apigenin sensitizes doxorubicin-resistant hepatocellular carcinoma BEL-7402/ADM cells to doxorubicin via inhibiting PI3K/Akt/Nrf2 pathway. Carcinogenesis 2013, 34, 1806–1814. [Google Scholar] [CrossRef] [PubMed]
- Gao, A.-M.; Ke, Z.-P.; Shi, F.; Sun, G.-C.; Chen, H. Chrysin enhances sensitivity of BEL-7402/ADM cells to doxorubicin by suppressing PI3K/Akt/Nrf2 and ERK/Nrf2 pathway. Chem. Biol. Interact. 2013, 206, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, V.; Cornelius, C.; Trovato, A.; Cavallaro, M.; Mancuso, C.; Di Rienzo, L.; Condorelli, D.; De Lorenzo, A.; Calabrese, E.J. The hormetic role of dietary antioxidants in free radical-related diseases. Curr. Pharm. Des. 2010, 16, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Paredes-Gonzalez, X.; Fuentes, F.; Jeffery, S.; Saw, C.L.-L.; Shu, L.; Su, Z.-Y.; Kong, A.-N.T. Induction of NRF2-mediated gene expression by dietary phytochemical flavones apigenin and luteolin. Biopharm. Drug Dispos. 2015, 36, 440–451. [Google Scholar] [CrossRef] [PubMed]
- Rattan, S.I.S. Hormesis in aging. Ageing Res. Rev. 2008, 7, 63–78. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Astrocytes: Adaptive responses to low doses of neurotoxins. Crit. Rev. Toxicol. 2008, 38, 463–471. [Google Scholar] [CrossRef]
- Valdameri, G.; Trombetta-Lima, M.; Worfel, P.R.; Pires, A.R.A.; Martinez, G.R.; Noleto, G.R.; Cadena, S.M.S.C.; Sogayar, M.C.; Winnischofer, S.M.B.; Rocha, M.E.M. Involvement of catalase in the apoptotic mechanism induced by apigenin in HepG2 human hepatoma cells. Chem. Biol. Interact. 2011, 193, 180–189. [Google Scholar] [CrossRef]
- Rahal, A.; Kumar, A.; Singh, V.; Yadav, B.; Tiwari, R.; Chakraborty, S.; Dhama, K. Oxidative stress, prooxidants, and antioxidants: The interplay. Biomed Res. Int. 2014, 2014, 761264. [Google Scholar] [CrossRef]
- Martins, L.A.M.; Coelho, B.P.; Behr, G.; Pettenuzzo, L.F.; Souza, I.C.C.; Moreira, J.C.F.; Borojevic, R.; Gottfried, C.; Guma, F.C.R. Resveratrol induces pro-oxidant effects and time-dependent resistance to cytotoxicity in activated hepatic stellate cells. Cell Biochem. Biophys. 2014, 68, 247–257. [Google Scholar] [CrossRef]
- Franco, R.; Navarro, G.; Martínez-Pinilla, E. Hormetic and mitochondria-related mechanisms of antioxidant action of phytochemicals. Antioxidants 2019, 8, 373. [Google Scholar] [CrossRef]
- Yang, Y.; Cai, X.; Yang, J.; Sun, X.; Hu, C.; Yan, Z.; Xu, X.; Lu, W.; Wang, X.; Cao, P. Chemoprevention of dietary digitoflavone on colitis-associated colon tumorigenesis through inducing Nrf2 signaling pathway and inhibition of inflammation. Mol. Cancer 2014, 13, 48. [Google Scholar] [CrossRef] [PubMed]
- Kwak, M.-K.; Kensler, T.W. Targeting NRF2 signaling for cancer chemoprevention. Toxicol. Appl. Pharmacol. 2010, 244, 66–76. [Google Scholar] [CrossRef] [PubMed]
- Basak, P.; Sadhukhan, P.; Sarkar, P.; Sil, P.C. Perspectives of the Nrf-2 signaling pathway in cancer progression and therapy. Toxicol. Rep. 2017, 4, 306–318. [Google Scholar] [CrossRef] [PubMed]
- Vogelstein, B.; Kinzler, K.W. Cancer genes and the pathways they control. Nat. Med. 2004, 10, 789–799. [Google Scholar] [CrossRef]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu. Rev. Pharmacol. Toxicol. 2007, 47, 89–116. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Shibata, T.; Takaya, K.; Shiraishi, K.; Kohno, T.; Kunitoh, H.; Tsuta, K.; Furuta, K.; Goto, K.; Hosoda, F.; et al. Regulatory nexus of synthesis and degradation deciphers cellular Nrf2 expression levels. Mol. Cell. Biol. 2013, 33, 2402–2412. [Google Scholar] [CrossRef]
- Xu, C.; Huang, M.-T.; Shen, G.; Yuan, X.; Lin, W.; Khor, T.O.; Conney, A.H.; Kong, A.-N.T. Inhibition of 7,12-dimethylbenz(a)anthracene-induced skin tumorigenesis in C57BL/6 mice by sulforaphane is mediated by nuclear factor E2–related factor 2. Cancer Res. 2006, 66, 8293–8296. [Google Scholar] [CrossRef]
- Cullinan, S.B.; Zhang, D.; Hannink, M.; Arvisais, E.; Kaufman, R.J.; Diehl, J.A. Nrf2 is a direct PERK substrate and effector of PERK-dependent cell survival. Mol. Cell. Biol 2003, 23, 7198–7209. [Google Scholar] [CrossRef]
- Sun, Z.; Huang, Z.; Zhang, D.D. Phosphorylation of Nrf2 at multiple sites by MAP kinases has a limited contribution in modulating the Nrf2-dependent antioxidant response. PLoS ONE 2009, 4, e6588. [Google Scholar] [CrossRef]
- Giudice, A.; Arra, C.; Turco, M.C. Review of molecular mechanisms involved in the activation of the Nrf2-ARE signaling pathway by chemopreventive agents. Methods Mol. Biol. 2010, 647, 37–74. [Google Scholar]
- Curcumin on NFE2L2 Gene Expression, Antioxidant Capacity and Renal Function According to rs35652124 in Diabetic Nephropathy. Available online: https://clinicaltrials.gov/ct2/show/NCT03262363 (accessed on 8 August 2020).
- RTA 408 Ophthalmic Suspension for the Treatment of Ocular Inflammation and Pain Following Ocular Surgery. Available online: https://clinicaltrials.gov/ct2/show/NCT02065375 (accessed on 8 August 2020).
- A 52-Week, Multi-Center, Double-Blind, Randomized, Placebo-Controlled Phase IIb Trial to Determine the Effects of Bardoxolone Methyl (RTA 402) on eGFR in Patients with Type 2 Diabetes and Chronic Kidney Disease with an eGFR of 20–45 mL/Min/1.73m2. Available online: https://clinicaltrials.gov/ct2/show/study/NCT00811889 (accessed on 8 August 2020).
- Anti-Inflammatory and Antioxidant Effects of Resveratrol on Healthy Adults. Available online: https://clinicaltrials.gov/ct2/show/record/NCT01492114 (accessed on 8 August 2020).
- Zhang, D.D.; Hannink, M. Distinct cysteine residues in Keap1 are required for Keap1-dependent ubiquitination of Nrf2 and for stabilization of Nrf2 by chemopreventive agents and oxidative stress. Mol. Cell. Biol. 2003, 23, 8137–8151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.D.; Lo, S.-C.; Cross, J.V.; Templeton, D.J.; Hannink, M. Keap1 is a redox-regulated substrate adaptor protein for a Cul3-dependent ubiquitin ligase complex. Mol. Cell. Biol. 2004, 24, 10941–10953. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Enaka, M.; Muragaki, Y. Activation of KEAP1/NRF2/P62 signaling alleviates high phosphate-induced calcification of vascular smooth muscle cells by suppressing reactive oxygen species production. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Ho, C.-Y.; Cheng, Y.-T.; Chau, C.-F.; Yen, G.-C. Effect of diallyl sulfide on in vitro and in vivo Nrf2-mediated pulmonic antioxidant enzyme expression via activation ERK/p38 signaling pathway. J. Agric. Food Chem. 2012, 60, 100–107. [Google Scholar] [CrossRef]
- Apopa, P.L.; He, X.; Ma, Q. Phosphorylation of Nrf2 in the transcription activation domain by casein kinase 2 (CK2) is critical for the nuclear translocation and transcription activation function of Nrf2 in IMR-32 neuroblastoma cells. J. Biochem. Mol. Toxicol. 2008, 22, 63–76. [Google Scholar] [CrossRef]
- Zhang, X.; Xiao, Z.; Yao, J.; Zhao, G.; Fa, X.; Niu, J. Participation of protein kinase C in the activation of Nrf2 signaling by ischemic preconditioning in the isolated rabbit heart. Mol. Cell. Biochem. 2013, 372, 169–179. [Google Scholar] [CrossRef]
- Wang, L.; Chen, Y.; Sternberg, P.; Cai, J. Essential roles of the PI3 kinase/Akt pathway in regulating Nrf2-dependent antioxidant functions in the RPE. Invest. Ophthalmol. Vis. Sci. 2008, 49, 1671–1678. [Google Scholar] [CrossRef]
- Li, L.; Luo, W.; Qian, Y.; Zhu, W.; Qian, J.; Li, J.; Jin, Y.; Xu, X.; Liang, G. Luteolin protects against diabetic cardiomyopathy by inhibiting NF-κB-mediated inflammation and activating the Nrf2-mediated antioxidant responses. Phytomedicine 2019, 59, 152774. [Google Scholar] [CrossRef]
- Zhang, H.; Tu, X.; Song, S.; Liang, R.; Shi, S. Baicalin reduces early brain injury after subarachnoid hemorrhage in rats. Chin. J. Integr. Med. 2020, 26, 510–518. [Google Scholar] [CrossRef]
- Xu, X.; Li, M.; Chen, W.; Yu, H.; Yang, Y.; Hang, L. Apigenin attenuates oxidative injury in ARPE-19 cells thorough activation of Nrf2 pathway. Oxid. Med. Cell. Longev. 2016, 2016, 4378461. [Google Scholar] [CrossRef]
- Sarawek, S.; Derendorf, H.; Butterweck, V. Pharmacokinetics of luteolin and metabolites in rats. Nat. Prod. Commun. 2008, 3, 2029–2036. [Google Scholar] [CrossRef]
- Yu, H.; Chen, B.; Ren, Q. Baicalin relieves hypoxia-aroused H9c2 cell apoptosis by activating Nrf2/HO-1-mediated HIF1α/BNIP3 pathway. Artif. Cells Nanomed. Biotechnol. 2019, 47, 3657–3663. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Wei, Y.; Huang, Y.; He, B.; Zhou, Y.; Fu, J. Nanoemulsion improves the oral bioavailability of baicalin in rats: In vitro and in vivo evaluation. Int. J. Nanomed. 2013, 8, 3769–3779. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.-M.; But, P.P.-H. Pharmacology and applications of Chinese materia medica. World Sci. 1986, 1, 304. [Google Scholar]
- Zhang, J.; Zhao, X.; Zhu, H.; Wang, J.; Ma, J.; Gu, M. Apigenin protects against renal tubular epithelial cell injury and oxidative stress by high glucose via regulation of NF-E2-related factor 2 (Nrf2) pathway. Med. Sci. Monit. 2019, 25, 5280–5288. [Google Scholar] [CrossRef]
- Meyer, H.; Bolarinwa, A.; Wolfram, G.; Linseisen, J. Bioavailability of apigenin from apiin-rich parsley in humans. Ann. Nutr. Metab. 2006, 50, 167–172. [Google Scholar] [CrossRef]
- Schadich, E.; Hlaváč, J.; Volná, T.; Varanasi, L.; Hajdúch, M.; Džubák, P. Effects of ginger phenylpropanoids and quercetin on Nrf2-ARE pathway in human BJ fibroblasts and HaCaT keratinocytes. BioMed Res. Int. 2016, 2016, 2173275. [Google Scholar] [CrossRef]
- Wang, K.; Chen, Z.; Huang, L.; Meng, B.; Zhou, X.; Wen, X.; Ren, D. Naringenin reduces oxidative stress and improves mitochondrial dysfunction via activation of the Nrf2/ARE signaling pathway in neurons. Int. J. Mol. Med. 2017, 40, 1582–1590. [Google Scholar] [CrossRef]
- Abdelaziz, R.M.; Abdelazem, A.Z.; Hashem, K.S.; Attia, Y.A. Protective effects of hesperidin against MTX-induced hepatotoxicity in male albino rats. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 393, 1405–1417. [Google Scholar] [CrossRef]
- Al-Maghrebi, M.; Alnajem, A.S.; Esmaeil, A. Epigallocatechin-3-gallate modulates germ cell apoptosis through the SAFE/Nrf2 signaling pathway. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2020, 393, 663–671. [Google Scholar] [CrossRef]
- Li, S.-Q.; Dong, S.; Su, Z.-H.; Zhang, H.-W.; Peng, J.-B.; Yu, C.-Y.; Zou, Z.-M. Comparative pharmacokinetics of naringin in rat after oral administration of Chaihu-Shu-Gan-San aqueous extract and naringin alone. Metabolites 2013, 3, 867–880. [Google Scholar] [CrossRef]
- Salehi, B.; Fokou, P.V.T.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of naringenin: A review of clinical trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef] [PubMed]
- Tian, R.; Yang, Z.; Lu, N.; Peng, Y.-Y. Quercetin, but not rutin, attenuated hydrogen peroxide-induced cell damage via heme oxygenase-1 induction in endothelial cells. Arch. Biochem. Biophys. 2019, 676, 108157. [Google Scholar] [CrossRef] [PubMed]
- Egert, S.; Wolffram, S.; Bosy-Westphal, A.; Boesch-Saadatmandi, C.; Wagner, A.E.; Frank, J.; Rimbach, G.; Mueller, M.J. Daily quercetin supplementation dose-dependently increases plasma quercetin concentrations in healthy humans. J. Nutr. 2008, 138, 1615–1621. [Google Scholar] [CrossRef] [PubMed]
- Erlund, I.; Kosonen, T.; Alfthan, G.; Mäenpää, J.; Perttunen, K.; Kenraali, J.; Parantainen, J.; Aro, A. Pharmacokinetics of quercetin from quercetin aglycone and rutin in healthy volunteers. Eur. J. Clin. Pharmacol. 2000, 56, 545–553. [Google Scholar] [CrossRef]
- Sun, L.; Xu, G.; Dong, Y.; Li, M.; Yang, L.; Lu, W. Quercetin protects against lipopolysaccharide-induced intestinal oxidative stress in broiler chickens through activation of Nrf2 pathway. Molecules 2020, 25, 1053. [Google Scholar] [CrossRef]
- Lou, H.; Jing, X.; Wei, X.; Shi, H.; Ren, D.; Zhang, X. Naringenin protects against 6-OHDA-induced neurotoxicity via activation of the Nrf2/ARE signaling pathway. Neuropharmacology 2014, 79, 380–388. [Google Scholar] [CrossRef]
- Ortiz-Andrade, R.R.; Sánchez-Salgado, J.C.; Navarrete-Vázquez, G.; Webster, S.P.; Binnie, M.; García-Jiménez, S.; León-Rivera, I.; Cigarroa-Vázquez, P.; Villalobos-Molina, R.; Estrada-Soto, S. Antidiabetic and toxicological evaluations of naringenin in normoglycaemic and NIDDM rat models and its implications on extra-pancreatic glucose regulation. Diabetes Obes. Metab. 2008, 10, 1097–1104. [Google Scholar] [CrossRef]
- Li, Y.; Kandhare, A.D.; Mukherjee, A.A.; Bodhankar, S.L. Acute and sub-chronic oral toxicity studies of hesperidin isolated from orange peel extract in Sprague Dawley rats. Regul. Toxicol. Pharmacol. 2019, 105, 77–85. [Google Scholar] [CrossRef]
- Chen, J.-H.; Tipoe, G.L.; Liong, E.C.; So, H.S.H.; Leung, K.-M.; Tom, W.-M.; Fung, P.C.W.; Nanji, A.A. Green tea polyphenols prevent toxin-induced hepatotoxicity in mice by down-regulating inducible nitric oxide-derived prooxidants. Am. J. Clin. Nutr. 2004, 80, 742–751. [Google Scholar] [CrossRef]
- Ishfaq, M.; Chen, C.; Bao, J.; Zhang, W.; Wu, Z.; Wang, J.; Liu, Y.; Tian, E.; Hamid, S.; Li, R.; et al. Baicalin ameliorates oxidative stress and apoptosis by restoring mitochondrial dynamics in the spleen of chickens via the opposite modulation of NF-κB and Nrf2/HO-1 signaling pathway during Mycoplasma gallisepticum infection. Poult. Sci. 2019, 98, 6296–6310. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Hao, Z.; Zhang, S.; Wei, M.; Lu, B.; Wang, Z.; Ji, L. Baicalein and baicalin alleviate acetaminophen-induced liver injury by activating Nrf2 antioxidative pathway: The involvement of ERK1/2 and PKC. Biochem. Pharmacol. 2018, 150, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Pang, C.; Zheng, Z.; Shi, L.; Sheng, Y.; Wei, H.; Wang, Z.; Ji, L. Caffeic acid prevents acetaminophen-induced liver injury by activating the Keap1-Nrf2 antioxidative defense system. Free Rad. Biol. Med. 2016, 91, 236–246. [Google Scholar] [CrossRef] [PubMed]
- Tian, S.; He, G.; Song, J.; Wang, S.; Xin, W.; Zhang, D.; Du, G. Pharmacokinetic study of baicalein after oral administration in monkeys. Fitoterapia 2012, 83, 532–540. [Google Scholar] [CrossRef]
- Dong, Y.; Xing, Y.; Sun, J.; Sun, W.; Xu, Y.; Quan, C. Baicalein alleviates liver oxidative stress and apoptosis induced by high-level glucose through the activation of the PERK/Nrf2 signaling pathway. Molecules 2020, 25, 599. [Google Scholar] [CrossRef]
- Ma, B.; Zhang, J.; Zhu, Z.; Zhao, A.; Zhou, Y.; Ying, H.; Zhang, Q. Luteolin ameliorates testis injury and blood–testis barrier disruption through the Nrf2 signaling pathway and by upregulating Cx43. Mol. Nutr. Food Res. 2019, 63, 1800843. [Google Scholar] [CrossRef]
- Lan, X.; Han, X.; Li, Q.; Wang, J. (−)-Epicatechin, a natural flavonoid compound, protects astrocytes against hemoglobin toxicity via Nrf2 and AP-1 signaling pathways. Mol. Neurobiol. 2017, 54, 7898–7907. [Google Scholar] [CrossRef]
- Liu, Y.-W.; Liu, X.-L.; Kong, L.; Zhang, M.-Y.; Chen, Y.-J.; Zhu, X.; Hao, Y.-C. Neuroprotection of quercetin on central neurons against chronic high glucose through enhancement of Nrf2/ARE/glyoxalase-1 pathway mediated by phosphorylation regulation. Biomed. Pharmacother. 2019, 109, 2145–2154. [Google Scholar] [CrossRef]
- Huang, C.-S.; Lii, C.-K.; Lin, A.-H.; Yeh, Y.-W.; Yao, H.-T.; Li, C.-C.; Wang, T.-S.; Chen, H.-W. Protection by chrysin, apigenin, and luteolin against oxidative stress is mediated by the Nrf2-dependent up-regulation of heme oxygenase 1 and glutamate cysteine ligase in rat primary hepatocytes. Arch. Toxicol. 2013, 87, 167–178. [Google Scholar] [CrossRef]
- Noh, K.; Oh, D.G.; Nepal, M.R.; Jeong, K.S.; Choi, Y.; Kang, M.J.; Kang, W.; Jeong, H.G.; Jeong, T.C. Pharmacokinetic interaction of chrysin with caffeine in rats. Biomol. Ther. 2016, 24, 446–452. [Google Scholar] [CrossRef]
- Manach, C.; Morand, C.; Demigné, C.; Texier, O.; Régérat, F.; Rémésy, C. Bioavailability of rutin and quercetin in rats. FEBS Lett. 1997, 409, 12–16. [Google Scholar] [CrossRef]
- Ishii, S.; Kitazawa, H.; Mori, T.; Kirino, A.; Nakamura, S.; Osaki, N.; Shimotoyodome, A.; Tamai, I. Identification of the catechin uptake transporter responsible for intestinal absorption of epigallocatechin gallate in mice. Sci. Rep. 2019, 9, 11014. [Google Scholar] [CrossRef] [PubMed]
- Speciale, A.; Anwar, S.; Canali, R.; Chirafisi, J.; Saija, A.; Virgili, F.; Cimino, F. Cyanidin-3-O-glucoside counters the response to TNF-alpha of endothelial cells by activating Nrf2 pathway. Mol. Nutr. Food Res. 2013, 57, 1979–1987. [Google Scholar] [CrossRef] [PubMed]
- Czank, C.; Cassidy, A.; Zhang, Q.; Morrison, D.J.; Preston, T.; Kroon, P.A.; Botting, N.P.; Kay, C.D. Human metabolism and elimination of the anthocyanin, cyanidin-3-glucoside: A 13C-tracer study. Am. J. Clin. Nutr. 2013, 97, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-S.; Li, B.; Kim, K.-S.; Jeong, G.-S.; Kim, E.-C.; Kim, Y.-C. Butein protects human dental pulp cells from hydrogen peroxide-induced oxidative toxicity via Nrf2 pathway-dependent heme oxygenase-1 expressions. Toxicol. Vitr. 2013, 27, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Yang, Y.; Xu, J.; Zhang, P.; Deng, R.; Mao, Y.; He, J.; Chen, Y.; Zhang, Y.; Ding, J.; et al. Luteolin exerts neuroprotection via modulation of the p62/Keap1/Nrf2 pathway in intracerebral hemorrhage. Front. Pharmacol. 2020, 10, 1551. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Li, Z.; Wu, S.; Li, X.; Sang, Y.; Li, J.; Niu, Y.; Ding, H. Myricetin alleviates cuprizone-induced behavioral dysfunction and demyelination in mice by Nrf2 pathway. Food Funct. 2016, 7, 4332–4342. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Tan, X.; Lv, Z.; Liu, B.; Baiyun, R.; Lu, J.; Zhang, Z. Regulation of Sirt1/Nrf2/TNF-α signaling pathway by luteolin is critical to attenuate acute mercuric chloride exposure induced hepatotoxicity. Sci. Rep. 2016, 6, 37157. [Google Scholar] [CrossRef]
- Zhang, H.; Tan, X.; Yang, D.; Lu, J.; Liu, B.; Baiyun, R.; Zhang, Z. Dietary luteolin attenuates chronic liver injury induced by mercuric chloride via the Nrf2/NF-κB/P53 signaling pathway in rats. Oncotarget 2017, 8, 40982–40993. [Google Scholar] [CrossRef]
- Dai, L.M.; Cheng, H.; Li, W.P.; Liu, S.Q.; Chen, M.Z.; Xu, S.Y. The influence of luteolin on experimental inflammatory models in rats. Acta Anhui Med. Univ. 1985, 20, 1–3. [Google Scholar]
- Chen, L.; Dou, J.; Su, Z.; Zhou, H.; Wang, H.; Zhou, W.; Guo, Q.; Zhou, C. Synergistic activity of baicalein with ribavirin against influenza A (H1N1) virus infections in cell culture and in mice. Antivir. Res. 2011, 91, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Choi, J.K.; Jung, C.H.; Koh, H.J.; Heo, P.; Shin, J.Y.; Kim, S.; Park, W.-S.; Shin, H.-J.; Kweon, D.-H. SNARE-wedging polyphenols as small molecular botox. Planta Med. 2012, 78, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Tu, J.; Zhang, Q.; Zhang, X.; Zhu, Y.; Ma, W.; Cheng, C.; Brann, D.W.; Yang, F. Genistein attenuates ischemic oxidative damage and behavioral deficits via eNOS/Nrf2/HO-1 signaling. Hippocampus 2013, 23, 634–647. [Google Scholar] [CrossRef]
- Singh, P.; Sharma, S.; Rath, S.K. Genistein induces deleterious effects during its acute exposure in Swiss mice. BioMed Res. Int. 2014, 2014, e619617. [Google Scholar] [CrossRef] [PubMed]
- Elle, R.E.; Rahmani, S.; Lauret, C.; Morena, M.; Bidel, L.P.R.; Boulahtouf, A.; Balaguer, P.; Cristol, J.-P.; Durand, J.-O.; Charnay, C.; et al. Functionalized mesoporous silica nanoparticle with antioxidants as a new carrier that generates lower oxidative stress impact on cells. Mol. Pharm. 2016, 13, 2647–2660. [Google Scholar] [CrossRef] [PubMed]
- Hseu, Y.-C.; Chou, C.-W.; Kumar, K.J.S.; Fu, K.-T.; Wang, H.-M.; Hsu, L.-S.; Kuo, Y.-S.; Wu, C.-R.; Chen, S.-C.; Yang, H.-L. Ellagic acid protects human keratinocyte (HaCaT) cells against UVA-induced oxidative stress and apoptosis through the upregulation of the HO-1 and Nrf-2 antioxidant genes. Food Chem. Toxicol. 2012, 50, 1245–1255. [Google Scholar] [CrossRef]
- Tate, P.S.; Marazita, M.C.; Marquioni-Ramella, M.D.; Suburo, A.M. Ilex paraguariensis extracts and its polyphenols prevent oxidative damage and senescence of human retinal pigment epithelium cells. J. Funct. Foods 2020, 67, 103833. [Google Scholar] [CrossRef]
- Liu, D.; Wang, H.; Zhang, Y.; Zhang, Z. Protective effects of chlorogenic acid on cerebral ischemia/reperfusion injury rats by regulating oxidative stress-related Nrf2 pathway. Drug Des. Dev. Ther. 2020, 14, 51–60. [Google Scholar] [CrossRef]
- Song, J.H.; Lee, H.-J.; Kang, K.S. Procyanidin C1 activates the Nrf2/HO-1 signaling pathway to prevent glutamate-induced apoptotic HT22 cell death. Int. J. Mol. Sci. 2019, 20, 142. [Google Scholar] [CrossRef]
- Rousta, A.-M.; Mirahmadi, S.-M.-S.; Shahmohammadi, A.; Nourabadi, D.; Khajevand-Khazaei, M.-R.; Baluchnejadmojarad, T.; Roghani, M. Protective effect of sesamin in lipopolysaccharide-induced mouse model of acute kidney injury via attenuation of oxidative stress, inflammation, and apoptosis. Immunopharmacol. Immunotoxicol. 2018, 40, 423–429. [Google Scholar] [CrossRef]
- Kong, P.; Chen, G.; Jiang, A.; Wang, Y.; Song, C.; Zhuang, J.; Xi, C.; Wang, G.; Ji, Y.; Yan, J. Sesamin inhibits IL-1β-stimulated inflammatory response in human osteoarthritis chondrocytes by activating Nrf2 signaling pathway. Oncotarget 2016, 7, 83720–83726. [Google Scholar] [CrossRef] [PubMed]
- Chang, B.Y.; Jung, Y.S.; Yoon, C.-S.; Oh, J.S.; Hong, J.H.; Kim, Y.-C.; Kim, S.Y. Fraxin prevents chemically induced hepatotoxicity by reducing oxidative stress. Molecules 2017, 22, 587. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Bagi, Z.; Feher, A.; Recchia, F.A.; Sonntag, W.E.; Pearson, K.; de Cabo, R.; Csiszar, A. Resveratrol confers endothelial protection via activation of the antioxidant transcription factor Nrf2. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H18–H24. [Google Scholar] [CrossRef] [PubMed]
- Pilot Study of the Effects of Resveratrol Supplement in Mild-To-Moderate Alzheimer’s Disease. Available online: https://clinicaltrials.gov/ct2/show/study/NCT00743743 (accessed on 8 August 2020).
- Qi, L.; Jiang, J.; Zhang, J.; Zhang, L.; Wang, T. Curcumin protects human trophoblast HTR8/SVneo cells from H2O2-induced oxidative stress by activating Nrf2 signaling pathway. Antioxidants 2020, 9, 121. [Google Scholar] [CrossRef] [PubMed]
- Zhai, S.S.; Ruan, D.; Zhu, Y.W.; Li, M.C.; Ye, H.; Wang, W.C.; Yang, L. Protective effect of curcumin on ochratoxin A–induced liver oxidative injury in duck is mediated by modulating lipid metabolism and the intestinal microbiota. Poult. Sci. 2020, 99, 1124–1134. [Google Scholar] [CrossRef]
- Bucolo, C.; Drago, F.; Maisto, R.; Romano, G.L.; D’Agata, V.; Maugeri, G.; Giunta, S. Curcumin prevents high glucose damage in retinal pigment epithelial cells through ERK1/2-mediated activation of the Nrf2/HO-1 pathway. J. Cell. Physiol. 2019, 234, 17295–17304. [Google Scholar] [CrossRef]
- Balogun, E.; Hoque, M.; Gong, P.; Killeen, E.; Green, C.J.; Foresti, R.; Alam, J.; Motterlini, R. Curcumin activates the haem oxygenase-1 gene via regulation of Nrf2 and the antioxidant-responsive element. Biochem. J. 2003, 371, 887–895. [Google Scholar] [CrossRef]
- Gao, S.; Duan, X.; Wang, X.; Dong, D.; Liu, D.; Li, X.; Sun, G.; Li, B. Curcumin attenuates arsenic-induced hepatic injuries and oxidative stress in experimental mice through activation of Nrf2 pathway, promotion of arsenic methylation and urinary excretion. Food Chem. Toxicol. 2013, 59, 739–747. [Google Scholar] [CrossRef]
- Barraza-Garza, G.; Pérez-León, J.A.; Castillo-Michel, H.; de la Rosa, L.A.; Martinez-Martinez, A.; Cotte, M.; Alvarez-Parrilla, E. Antioxidant effect of phenolic compounds (PC) at different concentrations in IEC-6 cells: A spectroscopic analysis. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2020, 227, 117570. [Google Scholar] [CrossRef]
- Han, J.; Wang, M.; Jing, X.; Shi, H.; Ren, M.; Lou, H. (−)-Epigallocatechin gallate protects against cerebral ischemia-induced oxidative stress via Nrf2/ARE signaling. Neurochem. Res. 2014, 39, 1292–1299. [Google Scholar] [CrossRef]
- Su, Z.-Y.; Zhang, C.; Lee, J.H.; Shu, L.; Wu, T.-Y.; Khor, T.O.; Conney, A.H.; Lu, Y.-P.; Kong, A.-N.T. Requirement and epigenetics reprogramming of Nrf2 in suppression of tumor promoter TPA-induced mouse skin cell transformation by sulforaphane. Cancer Prev. Res. 2014, 7, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Kalayarasan, S.; Prabhu, P.N.; Sriram, N.; Manikandan, R.; Arumugam, M.; Sudhandiran, G. Diallyl sulfide enhances antioxidants and inhibits inflammation through the activation of Nrf2 against gentamicin-induced nephrotoxicity in Wistar rats. Eur. J. Pharmacol. 2009, 606, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Wang, J.; Tang, X.; Li, Y.; Xia, P.; Gao, X. Berberine ameliorates non alcoholic fatty liver disease by a global modulation of hepatic mRNA and lncRNA expression profiles. J. Transl. Med. 2015, 13, 24. [Google Scholar] [CrossRef]
- Tao, S.; Zhang, H.; Xue, L.; Jiang, X.; Wang, H.; Li, B.; Tian, H.; Zhang, Z. Vitamin D protects against particles-caused lung injury through induction of autophagy in an Nrf2-dependent manner. Environ. Toxicol. 2019, 34, 594–609. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Li, J.; Ma, P.; Yang, X.; Xu, S. Vitamin E antagonizes ozone-induced asthma exacerbation in Balb/c mice through the Nrf2 pathway. Food Chem. Toxicol. 2017, 107, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Xiu, P.; Li, F.; Xin, C.; Li, K. Vitamin A Supplementation alleviates extrahepatic cholestasis liver injury through Nrf2 activation. Oxid. Med. Cell. Longev. 2014, 2014, 273692. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Zhang, S.; Du, T.; Yang, Q.; Chi, S.; Liu, H.; Yang, Y.; Dong, X.; Tan, B. Modulation of growth, immunity and antioxidant-related gene expressions in the liver and intestine of juvenile Sillago sihama by dietary vitamin C. Aquac. Nutr. 2020, 26, 338–350. [Google Scholar] [CrossRef]
- Zhao, R.; Wang, H.; Qiao, C.; Zhao, K. Vitamin B2 blocks development of Alzheimer’s disease in APP/PS1 transgenic mice via anti-oxidative mechanism. Trop. J. Pharm. Res. 2018, 17, 1049–1054. [Google Scholar] [CrossRef]
- Said, H.M. Intestinal absorption of water-soluble vitamins in health and disease. Biochem. J. 2011, 437, 357–372. [Google Scholar] [CrossRef]
- Pietrofesa, R.A.; Chatterjee, S.; Park, K.; Arguiri, E.; Albelda, S.M.; Christofidou-Solomidou, M. Synthetic lignan secoisolariciresinol diglucoside (LGM2605) reduces asbestos-induced cytotoxicity in an Nrf2-dependent and -independent manner. Antioxidants 2018, 7, 38. [Google Scholar] [CrossRef]
- Solis, L.M.; Behrens, C.; Dong, W.; Suraokar, M.; Ozburn, N.C.; Moran, C.A.; Corvalan, A.H.; Biswal, S.; Swisher, S.G.; Bekele, B.N.; et al. Nrf2 and Keap1 abnormalities in non-small cell lung carcinoma and association with clinicopathologic features. Clin. Cancer Res. 2010, 16, 3743–3753. [Google Scholar] [CrossRef] [PubMed]
- Yoo, N.J.; Kim, H.R.; Kim, Y.R.; An, C.H.; Lee, S.H. Somatic mutations of the KEAP1 gene in common solid cancers. Histopathology 2012, 60, 943–952. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.R.; Oh, J.E.; Kim, M.S.; Kang, M.R.; Park, S.W.; Han, J.Y.; Eom, H.S.; Yoo, N.J.; Lee, S.H. Oncogenic NRF2 mutations in squamous cell carcinomas of oesophagus and skin. J. Pathol. 2010, 220, 446–451. [Google Scholar] [CrossRef] [PubMed]
- Ooi, A.; Dykema, K.; Ansari, A.; Petillo, D.; Snider, J.; Kahnoski, R.; Anema, J.; Craig, D.; Carpten, J.; Teh, B.-T.; et al. CUL3 and NRF2 mutations confer an NRF2 activation phenotype in a sporadic form of papillary renal cell carcinoma. Cancer Res. 2013, 73, 2044–2051. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, L.D.; Lee, J.; Gnad, F.; Klijn, C.; Schaub, A.; Reeder, J.; Daemen, A.; Bakalarski, C.E.; Holcomb, T.; Shames, D.S.; et al. Recurrent loss of NFE2L2 exon 2 is a mechanism for Nrf2 pathway activation in human cancers. Cell Rep. 2016, 16, 2605–2617. [Google Scholar] [CrossRef]
- Chen, W.; Sun, Z.; Wang, X.-J.; Jiang, T.; Huang, Z.; Fang, D.; Zhang, D.D. Direct interaction between Nrf2 and p21Cip1/WAF1 upregulates the Nrf2-mediated antioxidant response. Mol. Cell 2009, 34, 663–673. [Google Scholar] [CrossRef]
- Ma, J.; Cai, H.; Wu, T.; Sobhian, B.; Huo, Y.; Alcivar, A.; Mehta, M.; Cheung, K.L.; Ganesan, S.; Kong, A.-N.T.; et al. PALB2 interacts with KEAP1 to promote NRF2 nuclear accumulation and function. Mol. Cell. Biol. 2012, 32, 1506–1517. [Google Scholar] [CrossRef]
- Komatsu, M.; Kurokawa, H.; Waguri, S.; Taguchi, K.; Kobayashi, A.; Ichimura, Y.; Sou, Y.-S.; Ueno, I.; Sakamoto, A.; Tong, K.I.; et al. The selective autophagy substrate p62 activates the stress responsive transcription factor Nrf2 through inactivation of Keap1. Nat. Cell Biol. 2010, 12, 213–223. [Google Scholar] [CrossRef]
- Hast, B.E.; Goldfarb, D.; Mulvaney, K.M.; Hast, M.A.; Siesser, P.F.; Yan, F.; Hayes, D.N.; Major, M.B. Proteomic analysis of ubiquitin ligase KEAP1 reveals associated proteins that inhibit NRF2 ubiquitination. Cancer Res. 2013, 73, 2199–2210. [Google Scholar] [CrossRef]
- Orthwein, A.; Noordermeer, S.M.; Wilson, M.D.; Landry, S.; Enchev, R.I.; Sherker, A.; Munro, M.; Pinder, J.; Salsman, J.; Dellaire, G.; et al. A mechanism for the suppression of homologous recombination in G1 cells. Nature 2015, 528, 422–426. [Google Scholar] [CrossRef]
- Adam, J.; Hatipoglu, E.; O’Flaherty, L.; Ternette, N.; Sahgal, N.; Lockstone, H.; Baban, D.; Nye, E.; Stamp, G.W.; Wolhuter, K.; et al. Renal cyst formation in Fh1-deficient mice is independent of the Hif/Phd pathway: Roles for fumarate in KEAP1 succination and Nrf2 signaling. Cancer Cell 2011, 20, 524–537. [Google Scholar] [CrossRef] [PubMed]
- Hanada, N.; Takahata, T.; Zhou, Q.; Ye, X.; Sun, R.; Itoh, J.; Ishiguro, A.; Kijima, H.; Mimura, J.; Itoh, K.; et al. Methylation of the KEAP1 gene promoter region in human colorectal cancer. BMC Cancer 2012, 12, 66. [Google Scholar] [CrossRef] [PubMed]
- DeNicola, G.M.; Karreth, F.A.; Humpton, T.J.; Gopinathan, A.; Wei, C.; Frese, K.; Mangal, D.; Yu, K.H.; Yeo, C.J.; Calhoun, E.S.; et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 2011, 475, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Mitsuishi, Y.; Taguchi, K.; Kawatani, Y.; Shibata, T.; Nukiwa, T.; Aburatani, H.; Yamamoto, M.; Motohashi, H. Nrf2 redirects glucose and glutamine into anabolic pathways in metabolic reprogramming. Cancer Cell 2012, 22, 66–79. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, K.; Yamamoto, M. The KEAP1–NRF2 system in cancer. Front. Oncol. 2017, 7, 85. [Google Scholar] [CrossRef]
- Wu, K.C.; Cui, J.Y.; Klaassen, C.D. Beneficial role of Nrf2 in regulating NADPH generation and consumption. Toxicol. Sci. J. Soc. Toxicol. 2011, 123, 590–600. [Google Scholar] [CrossRef]
- Kitteringham, N.R.; Abdullah, A.; Walsh, J.; Randle, L.; Jenkins, R.E.; Sison, R.; Goldring, C.E.P.; Powell, H.; Sanderson, C.; Williams, S.; et al. Proteomic analysis of Nrf2 deficient transgenic mice reveals cellular defence and lipid metabolism as primary Nrf2-dependent pathways in the liver. J. Proteom. 2010, 73, 1612–1631. [Google Scholar] [CrossRef]
- Lee, J.-M.; Calkins, M.J.; Chan, K.; Kan, Y.W.; Johnson, J.A. Identification of the NF-E2-related factor-2-dependent genes conferring protection against oxidative stress in primary cortical astrocytes using oligonucleotide microarray analysis. J. Biol. Chem. 2003, 278, 12029–12038. [Google Scholar] [CrossRef]
- Sánchez-Rodríguez, R.; Torres-Mena, J.E.; Quintanar-Jurado, V.; Chagoya-Hazas, V.; Rojas del Castillo, E.; del Pozo Yauner, L.; Villa-Treviño, S.; Pérez-Carreón, J.I. Ptgr1 expression is regulated by NRF2 in rat hepatocarcinogenesis and promotes cell proliferation and resistance to oxidative stress. Free Radic. Biol. Med. 2017, 102, 87–99. [Google Scholar] [CrossRef]
- Singh, A.; Happel, C.; Manna, S.K.; Acquaah-Mensah, G.; Carrerero, J.; Kumar, S.; Nasipuri, P.; Krausz, K.W.; Wakabayashi, N.; Dewi, R.; et al. Transcription factor NRF2 regulates miR-1 and miR-206 to drive tumorigenesis. J. Clin. Investig. 2013, 123, 2921–2934. [Google Scholar] [CrossRef]
- Reddy, N.M.; Kleeberger, S.R.; Cho, H.-Y.; Yamamoto, M.; Kensler, T.W.; Biswal, S.; Reddy, S.P. Deficiency in Nrf2-GSH signaling impairs type II cell growth and enhances sensitivity to oxidants. Am. J. Respir. Cell. Mol. Biol. 2007, 37, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Reddy, N.M.; Kleeberger, S.R.; Yamamoto, M.; Kensler, T.W.; Scollick, C.; Biswal, S.; Reddy, S.P. Genetic dissection of the Nrf2-dependent redox signaling-regulated transcriptional programs of cell proliferation and cytoprotection. Physiol. Genom. 2007, 32, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Ishimoto, T.; Nagano, O.; Yae, T.; Tamada, M.; Motohara, T.; Oshima, H.; Oshima, M.; Ikeda, T.; Asaba, R.; Yagi, H.; et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc− and thereby promotes tumor growth. Cancer Cell 2011, 19, 387–400. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Yang, J.W.; Kim, M.R.; Roh, S.H.; Kim, H.G.; Lee, K.Y.; Jeong, H.G.; Kang, K.W. Increased expression of Nrf2/ARE-dependent anti-oxidant proteins in tamoxifen-resistant breast cancer cells. Free Radic. Biol. Med. 2008, 45, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Murakami, S.; Motohashi, H. Roles of Nrf2 in cell proliferation and differentiation. Free Radic. Biol. Med. 2015, 88, 168–178. [Google Scholar] [CrossRef]
- Reddy, N.M.; Kleeberger, S.R.; Bream, J.H.; Fallon, P.G.; Kensler, T.W.; Yamamoto, M.; Reddy, S.P. Genetic disruption of the Nrf2 compromises cell-cycle progression by impairing GSH-induced redox signaling. Oncogene 2008, 27, 5821–5832. [Google Scholar] [CrossRef]
- Beyer, T.A.; Xu, W.; Teupser, D.; auf dem Keller, U.; Bugnon, P.; Hildt, E.; Thiery, J.; Kan, Y.W.; Werner, S. Impaired liver regeneration in Nrf2 knockout mice: Role of ROS-mediated insulin/IGF-1 resistance. EMBO J. 2008, 27, 212–223. [Google Scholar] [CrossRef]
- Homma, S.; Ishii, Y.; Morishima, Y.; Yamadori, T.; Matsuno, Y.; Haraguchi, N.; Kikuchi, N.; Satoh, H.; Sakamoto, T.; Hizawa, N.; et al. Nrf2 enhances cell proliferation and resistance to anticancer drugs in human lung cancer. Clin. Cancer Res. 2009, 15, 3423–3432. [Google Scholar] [CrossRef]
- Chio, I.I.C.; Jafarnejad, S.M.; Ponz-Sarvise, M.; Park, Y.; Rivera, K.; Palm, W.; Wilson, J.; Sangar, V.; Hao, Y.; Öhlund, D.; et al. NRF2 promotes tumor maintenance by modulating mRNA translation in pancreatic cancer. Cell 2016, 166, 963–976. [Google Scholar] [CrossRef]
- Holmström, K.M.; Baird, L.; Zhang, Y.; Hargreaves, I.; Chalasani, A.; Land, J.M.; Stanyer, L.; Yamamoto, M.; Dinkova-Kostova, A.T.; Abramov, A.Y. Nrf2 impacts cellular bioenergetics by controlling substrate availability for mitochondrial respiration. Biol. Open 2013, 2, 761–770. [Google Scholar] [CrossRef]
- Itoh, K.; Ye, P.; Matsumiya, T.; Tanji, K.; Ozaki, T. Emerging functional cross-talk between the Keap1-Nrf2 system and mitochondria. J. Clin. Biochem. Nutr. 2015, 56, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, H.; Motohashi, H. NRF2 addiction in cancer cells. Cancer Sci. 2018, 109, 900–911. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Zhang, Y.; Chen, W.; Zhao, X. The relationship between fasting plasma concentrations of selected flavonoids and their ordinary dietary intake. Br. J. Nutr. 2010, 103, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Pandurangan, A.K.; Sadagopan, S.K.A.; Dharmalingam, P.; Ganapasam, S. Luteolin, a bioflavonoid inhibits Azoxymethane-induced colorectal cancer through activation of Nrf2 signaling. Toxicol. Mech. Methods 2014, 24, 13–20. [Google Scholar] [CrossRef]
- Xiong, J.; Wang, K.; Yuan, C.; Xing, R.; Ni, J.; Hu, G.; Chen, F.; Wang, X. Luteolin protects mice from severe acute pancreatitis by exerting HO-1-mediated anti-inflammatory and antioxidant effects. Int. J. Mol. Med. 2017, 39, 113–125. [Google Scholar] [CrossRef]
- Ding, S.; Zhang, Z.; Song, J.; Cheng, X.; Jiang, J.; Jia, X. Enhanced bioavailability of apigenin via preparation of a carbon nanopowder solid dispersion. Int. J. Nanomed. 2014, 9, 2327–2333. [Google Scholar] [CrossRef]
- Qin, S.; Chen, J.; Tanigawa, S.; Hou, D.-X. Microarray and pathway analysis highlight Nrf2/ARE-mediated expression profiling by polyphenolic myricetin. Mol. Nutr. Food Res. 2013, 57, 435–546. [Google Scholar] [CrossRef]
- Weng, C.-J.; Chen, M.-J.; Yeh, C.-T.; Yen, G.-C. Hepatoprotection of quercetin against oxidative stress by induction of metallothionein expression through activating MAPK and PI3K pathways and enhancing Nrf2 DNA-binding activity. New Biotechnol. 2011, 28, 767–777. [Google Scholar] [CrossRef]
- Ren, H.; Hao, J.; Liu, T.; Zhang, D.; Lv, H.; Song, E.; Zhu, C. Hesperetin suppresses inflammatory responses in lipopolysaccharide-induced RAW 264.7 cells via the inhibition of NF-κB and activation of Nrf2/HO-1 pathways. Inflammation 2016, 39, 964–973. [Google Scholar] [CrossRef]
- Takumi, H.; Nakamura, H.; Simizu, T.; Harada, R.; Kometani, T.; Nadamoto, T.; Mukai, R.; Murota, K.; Kawai, Y.; Terao, J. Bioavailability of orally administered water-dispersible hesperetin and its effect on peripheral vasodilatation in human subjects: Implication of endothelial functions of plasma conjugated metabolites. Food Funct. 2012, 3, 389–398. [Google Scholar] [CrossRef]
- Su, C.; Xia, X.; Shi, Q.; Song, X.; Fu, J.; Xiao, C.; Chen, H.; Lu, B.; Sun, Z.; Wu, S.; et al. Neohesperidin dihydrochalcone versus CCl₄-induced hepatic injury through different mechanisms: The implication of free radical scavenging and Nrf2 activation. J. Agric. Food Chem. 2015, 63, 5468–5475. [Google Scholar] [CrossRef] [PubMed]
- Froyen, E.B.; Steinberg, F.M. Soy isoflavones increase quinone reductase in hepa-1c1c7 cells via estrogen receptor beta and nuclear factor erythroid 2-related factor 2 binding to the antioxidant response element. J. Nutr. Biochem. 2011, 22, 843–848. [Google Scholar] [CrossRef] [PubMed]
- Sabzichi, M.; Mohammadian, J.; Bazzaz, R.; Pirouzpanah, M.B.; Shaaker, M.; Hamishehkar, H.; Chavoshi, H.; Salehi, R.; Samadi, N. Chrysin loaded nanostructured lipid carriers (NLCs) triggers apoptosis in MCF-7 cancer cells by inhibiting the Nrf2 pathway. Process Biochem. 2017, 60, 84–91. [Google Scholar] [CrossRef]
- Walle, T.; Otake, Y.; Brubaker, J.A.; Walle, U.K.; Halushka, P.V. Disposition and metabolism of the flavonoid chrysin in normal volunteers. Br. J. Clin. Pharmacol. 2001, 51, 143–146. [Google Scholar] [PubMed]
- Zuo, Q.; Wu, R.; Xiao, X.; Yang, C.; Yang, Y.; Wang, C.; Lin, L.; Kong, A.-N. The dietary flavone luteolin epigenetically activates the Nrf2 pathway and blocks cell transformation in human colorectal cancer HCT116 cells. J. Cell Biochem. 2018, 119, 9573–9582. [Google Scholar] [CrossRef]
- Liang, F.; Fang, Y.; Cao, W.; Zhang, Z.; Pan, S.; Xu, X. Attenuation of tert-butyl hydroperoxide (t-BHP)-induced oxidative damage in HepG2 Cells by tangeretin: Relevance of the Nrf2–ARE and MAPK signaling pathways. J. Agric. Food Chem. 2018, 66, 6317–6325. [Google Scholar] [CrossRef]
- Granado-Serrano, A.B.; Martín, M.A.; Haegeman, G.; Goya, L.; Bravo, L.; Ramos, S. Epicatechin induces NF-κB, activator protein-1 (AP-1) and nuclear transcription factor erythroid 2p45-related factor-2 (Nrf2) via phosphatidylinositol-3-kinase/protein kinase B (PI3K/AKT) and extracellular regulated kinase (ERK) signalling in HepG2 cells. Br. J. Nutr. 2010, 103, 168–179. [Google Scholar] [CrossRef]
- Vanitha, P.; Senthilkumar, S.; Dornadula, S.; Anandhakumar, S.; Rajaguru, P.; Ramkumar, K.M. Morin activates the Nrf2-ARE pathway and reduces oxidative stress-induced DNA damage in pancreatic beta cells. Eur. J. Pharmacol. 2017, 801, 9–18. [Google Scholar] [CrossRef]
- Krajka-Kuźniak, V.; Paluszczak, J.; Celewicz, L.; Barciszewski, J.; Baer-Dubowska, W. Phloretamide, an apple phenolic compound, activates the Nrf2/ARE pathway in human hepatocytes. Food Chem. Toxicol. Int. J. 2013, 51, 202–209. [Google Scholar] [CrossRef]
- Hara, F.; Tatebe, J.; Watanabe, I.; Yamazaki, J.; Ikeda, T.; Morita, T. Molecular hydrogen alleviates cellular senescence in endothelial cells. Circ. J. 2016, 80, 2037–2046. [Google Scholar] [CrossRef]
- Juárez-Rebollar, D.; Rios, C.; Nava-Ruíz, C.; Méndez-Armenta, M. Metallothionein in brain disorders. Oxid. Med. Cell. Longev. 2017, 2017, 5828056. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Cheng, Y.; Wu, H.; Kong, L.; Wang, S.; Xu, Z.; Zhang, Z.; Tan, Y.; Keller, B.B.; Zhou, H.; et al. Metallothionein is downstream of Nrf2 and partially mediates sulforaphane prevention of diabetic cardiomyopathy. Diabetes 2017, 66, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Haldar, S.; Tripathi, A.K.; Horback, K.; Wong, J.; Sharma, D.; Beserra, A.; Suda, S.; Anbalagan, C.; Dev, S.; et al. Brain iron homeostasis: From molecular mechanisms to clinical significance and therapeutic opportunities. Antioxid. Redox Signal. 2013, 20, 1324–1363. [Google Scholar] [CrossRef] [PubMed]
- Thilakarathna, S.H.; Rupasinghe, H.P.V. Flavonoid bioavailability and attempts for bioavailability enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef]
- Mullen, W.; Edwards, C.A.; Crozier, A. Absorption, excretion and metabolite profiling of methyl-, glucuronyl-, glucosyl- and sulpho-conjugates of quercetin in human plasma and urine after ingestion of onions. Br. J. Nutr. 2006, 96, 107–116. [Google Scholar] [CrossRef]
- Rupasinghe, H.P.V.; Ronalds, C.M.; Rathgeber, B.; Robinson, R.A. Absorption and tissue distribution of dietary quercetin and quercetin glycosides of apple skin in broiler chickens. J. Sci. Food Agric. 2010, 90, 1172–1178. [Google Scholar] [CrossRef]
- Javan, N.; Ansari, M.H.K.; Dadashpour, M.; Khojastehfard, M.; Bastami, M.; Rahmati-Yamchi, M.; Zarghami, N. Synergistic antiproliferative effects of co-nanoencapsulated curcumin and chrysin on MDA-MB-231 breast cancer cells through upregulating miR-132 and miR-502c. Nutr. Cancer 2019, 71, 1201–1213. [Google Scholar] [CrossRef]
- Lotfi-Attari, J.; Pilehvar-Soltanahmadi, Y.; Dadashpour, M.; Alipour, S.; Farajzadeh, R.; Javidfar, S.; Zarghami, N. Co-Delivery of curcumin and chrysin by polymeric nanoparticles inhibit synergistically growth and hTERT gene expression in human colorectal cancer cells. Nutr. Cancer 2017, 69, 1290–1299. [Google Scholar] [CrossRef]
- Bondonno, N.P.; Dalgaard, F.; Kyrø, C.; Murray, K.; Bondonno, C.P.; Lewis, J.R.; Croft, K.D.; Gislason, G.; Scalbert, A.; Cassidy, A.; et al. Flavonoid intake is associated with lower mortality in the Danish diet cancer and health cohort. Nat. Commun. 2019, 10, 1–10. [Google Scholar] [CrossRef]
- Cutler, G.J.; Nettleton, J.A.; Ross, J.A.; Harnack, L.J.; Jacobs, D.R.; Scrafford, C.G.; Barraj, L.M.; Mink, P.J.; Robien, K. Dietary flavonoid intake and risk of cancer in postmenopausal women: The Iowa Women’s Health Study. Int. J. Cancer 2008, 123, 664–671. [Google Scholar] [CrossRef] [PubMed]
- Bo, Y.; Sun, J.; Wang, M.; Ding, J.; Lu, Q.; Yuan, L. Dietary flavonoid intake and the risk of digestive tract cancers: A systematic review and meta-analysis. Sci. Rep. 2016, 6, 24836. [Google Scholar] [CrossRef] [PubMed]
- Woo, H.D.; Kim, J. Dietary flavonoid intake and risk of stomach and colorectal cancer. World J. Gastroenterol. 2013, 19, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Lei, L.; Zhou, Y.; Ye, F.; Zhao, G. Dietary flavonoids and the risk of colorectal cancer: An updated meta-analysis of epidemiological studies. Nutrients 2018, 10, 950. [Google Scholar] [CrossRef]
- Gates, M.A.; Vitonis, A.F.; Tworoger, S.S.; Rosner, B.; Titus-Ernstoff, L.; Hankinson, S.E.; Cramer, D.W. Flavonoid intake and ovarian cancer risk in a population-based case-control study. Int. J. Cancer 2009, 124, 1918–1925. [Google Scholar] [CrossRef]
- Wang, L.; Lee, I.M.; Zhang, S.M.; Blumberg, J.B.; Buring, J.E.; Sesso, H.D. Dietary intake of selected flavonols, flavones, and flavonoid-rich foods and risk of cancer in middle-aged and older women. Am. J. Clin. Nutr. 2009, 89, 905–912. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, S.M.; Wu, K.; Willett, W.C.; Fuchs, C.S.; Giovannucci, E. Flavonoid intake and colorectal cancer risk in men and women. Am. J. Epidemiol. 2006, 164, 644–651. [Google Scholar] [CrossRef]
- Milkovic, L.; Zarkovic, N.; Saso, L. Controversy about pharmacological modulation of Nrf2 for cancer therapy. Redox Biol. 2017, 12, 727–732. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
L. Suraweera, T.; Rupasinghe, H.P.V.; Dellaire, G.; Xu, Z. Regulation of Nrf2/ARE Pathway by Dietary Flavonoids: A Friend or Foe for Cancer Management? Antioxidants 2020, 9, 973. https://doi.org/10.3390/antiox9100973
L. Suraweera T, Rupasinghe HPV, Dellaire G, Xu Z. Regulation of Nrf2/ARE Pathway by Dietary Flavonoids: A Friend or Foe for Cancer Management? Antioxidants. 2020; 9(10):973. https://doi.org/10.3390/antiox9100973
Chicago/Turabian StyleL. Suraweera, Tharindu, H. P. Vasantha Rupasinghe, Graham Dellaire, and Zhaolin Xu. 2020. "Regulation of Nrf2/ARE Pathway by Dietary Flavonoids: A Friend or Foe for Cancer Management?" Antioxidants 9, no. 10: 973. https://doi.org/10.3390/antiox9100973
APA StyleL. Suraweera, T., Rupasinghe, H. P. V., Dellaire, G., & Xu, Z. (2020). Regulation of Nrf2/ARE Pathway by Dietary Flavonoids: A Friend or Foe for Cancer Management? Antioxidants, 9(10), 973. https://doi.org/10.3390/antiox9100973






