Abstract
As a beneficial fungus, Trichoderma harzianum (T. harzianum) has been widely applied for growth promotion and biocontrol. Recently, it has attracted much attention with regard to improving stress tolerance in plants under abiotic stress. In this paper, the multiple mechanisms of T. harzianum for alleviating abiotic stress damage in plants are reviewed. T. harzianum can regulate the synthesis of key phytohormones, such as abscisic acid (ABA), indole-3-acetic acid (IAA), etc., thereby enhancing the physiological response ability of plants under stress conditions such as drought, salt stress, and high temperature. These are associated with antioxidant system regulation in plants, which reduces levels of reactive oxygen species (ROS) and oxidative damage and maintains intracellular redox balance. T. harzianum can also improve plant nutrient uptake and root development, secondary metabolism, soil environment and structure, and expression of related genes. In addition, in this paper, the characteristics of T. harzianum application in field and horticultural crops are summarized and compared, revealing differences in the methods, concentrations, time, and effects of applying T. harzianum to various crops. We further explore the synergistic regulation effect of T. harzianum and plant–microbiome interaction on the stress microenvironment. Future perspectives on the molecular mechanism of T. harzianum and its field application potential are discussed. This review provides a theoretic and practical reference for the application of T. harzianum in agricultural production.
1. Introduction
By 2030, nearly 600 million people are projected to be chronically undernourished and struggling to stay healthy [1]. Therefore, there is an urgent need to significantly increase agricultural productivity. However, with increases in extreme weather, abiotic stresses such as heat and drought can adversely affect the growth and development of plants, thereby reducing crop yield and quality [2].
The most important feature of drought stress is that ABA accumulation triggers a protective mechanism, leading to stomatal closure [3,4]. Stomatal closure reduces carbon dioxide absorption, which decreases photosynthesis and thereby inhibits plant growth and development [5]. Drought can also lead to the accumulation of peroxides, which can cause damage to cells [6]. Salt stress usually leads to disturbed ion absorption and hinders water transport, thus limiting plant growth and development [7]. High temperatures can cause plant pollen sterility, reduce stigma vitality, and significantly increase the levels of reactive oxygen species (ROS) in buckwheat flowers [8]. Furthermore, high temperatures lead to Rubisco inactivation and a decrease in the chloroplast electron transport rate, which reduces plant photosynthesis and inhibits growth [9]. These stressors not only interfere with the normal growth and metabolism of plants but also lead to a significant decline in crop yields, which seriously threatens global food security. Therefore, the development of effective strategies to improve the adaptability and productivity of crops under abiotic stress has become a top priority in agricultural science research and practice.
In recent years, biomicrobial agents have received a lot of attention as an innovative and sustainable solution to abiotic stresses. T. harzianum is a filamentous fungus belonging to the family Carno-bacteriaceae and Xylaria, which was first classified separately from Trichoderma by Rifai in 1969 and officially named T. harzianum [10]. This microorganism demonstrates the ability to colonize plant surfaces and form hyphal sheaths, effectively suppressing pathogenic bacteria while modulating the composition of fungal communities in the rhizosphere [11]. Its broad-spectrum resistance against various pests and diseases [12,13] has established it as a significant subject for research into plant biocontrol and growth promotion.
Recent research has consistently demonstrated the remarkable capacity of T. harzianum to enhance abiotic stress tolerance and improve agricultural productivity across a wide spectrum of economically important crops. Regarding vegetable crops, numerous controlled studies have validated its efficacy in tomato, where T. harzianum inoculation has been shown to significantly improve drought tolerance while simultaneously enhancing key growth parameters including plant height, leaf area, and root biomass [14,15,16]. Comparable beneficial effects have been well documented in other solanaceous crops such as cucumber [17] and eggplant [18], as well as perennial crops like black pepper [19]. Beyond horticultural species, this beneficial fungus has been proven equally effective in major cereal crops, with multiple independent studies confirming its growth-promoting effects in rice [20,21,22] and maize [23] under various stress conditions. More recent investigations have expanded the scope of T. harzianum’s applications to include important forage crops like Sudan grass [24] and oilseed crops such as Indian mustard [25], demonstrating its versatility across diverse agricultural systems. These consistent findings across multiple crops underscore the broad-spectrum potential of T. harzianum as a bio-enhancer in modern agriculture.
This review examines T. harzianum application methods in horticultural and field crops, along with its mechanisms for enhancing crop stress tolerance under abiotic stress conditions. The fungus can be applied through various methods including seed treatment, soil inoculation, foliar spraying, and root dipping, all of which have shown significant potential for improving plant growth and stress resilience. The mechanism of action is described in detail from six aspects: root development and nutrient absorption, secondary metabolism, antioxidant defense systems, plant hormone balance, soil structure, and expression of related genes. These multifaceted mechanisms collectively contribute to improved stress adaptation, significantly advancing our understanding of microbial-mediated plant stress tolerance and providing valuable insights for developing sustainable agricultural strategies under challenging environmental conditions. The integration of these findings with emerging multi-omics approaches promises to further elucidate the complex interactions between T. harzianum and crop plants, optimizing its application for enhanced agricultural productivity.
2. Methods of Applying T. harzianum
The effects of T. harzianum are wide-ranging; it can boosts resistance [26,27], control pathogens like Fusarium acuminatum [12] and Fusarium wilt [28], inhibit root-knot nematodes [13], improve aphid resistance (through RNA-mediated gene silencing) [29], and increase fruit antioxidant capacity and anthocyanin content [30]. As shown in Table 1, T. harzianum is applied to horticultural crops in a variety of ways that aim to enhance the symbiotic relationship between plants and microorganisms through early colonization. There are six main methods of applying T. harzianum to plants, including soil inoculation, seed treatment, root dipping, foliar spraying, addition to the irrigation system, and compost or organic fertilizer inoculation. The three most commonly used application methods for horticultural crops are seed treatment (tomato, eggplant) [15,16,18], soil application (cucumber, melon, black pepper) [17,19,27,28], and root treatments (strawberry, chrysanthemum) [30,31] (Table 1). Additionally, application times are diverse, in order to activate the plant defense mechanism in advance or to promote the development of the root system (Table 1).

Table 1.
Method, amount, time and effect of application of T. harzianum to horticultural crops.
Recently, research trends have tended to favor complex stress management and precise application strategies (e.g., regular irrigation for chrysanthemum [31]). In the future, the synergistic effect of T. harzianum and other probiotics can be further explored in combination with molecular means to analyze crop-specific response mechanisms, to optimize the effects of field application.
As shown in Table 2, the application of T. harzianum to field crops is mainly carried out through soil application (maize, Indian Mustard) [23,25], seed treatment (rice) [20,21,22], or root irrigation (Sorghum sudangrass, Hordeum vulgare L.) [24,32] with the optimal period being pre-sowing or at the transplanting period. These treatments significantly improve crop stress tolerance, modulate inter-root microbial communities, enhance antioxidant enzyme activities, and increase yield (Table 2).

Table 2.
Method, amount, time, and effect of application of T. harzianum to field crops.
Horticultural plants and field crops have both commonalities and differences with regard to the way T. harzianum is applied (Table 1 and Table 2). In terms of application methods, both types of crops may receive seed treatment, soil application, or root irrigation. However, horticultural plants are more inclined to receive seedling-stage treatment (Table 1). Application to field crops focuses more on seed treatment or soil application before sowing (Table 2). In terms of application effects, both types of crops showed significant effects relating to stress and growth promotion. Treatment of horticultural plants focuses more on quality enhancement and disease prevention and control. By comparison, those working with field crops have been more concerned with yield enhancement and stress tolerance enhancement. In addition, horticultural plants tend to receive multiple applications at critical growth periods, while field crops are mainly treated with a single treatment prior to planting or at transplanting (Table 1 and Table 2). These differences mainly stem from the differences in growth cycles, cultivation patterns, and types of stress between the two types of crops, and need to be optimized and adjusted to specific crop needs in practical applications.
The actual effect of T. harzianum can vary greatly depending on strain-specific characteristics, application methods, timing, concentration, and crop species. Since these factors together affect efficacy, follow-up studies need to combine these factors to select the most effective regimen. In addition, it is advisable to develop region-specific application guidelines to integrate local climate, soil conditions, and crop growth stages. This approach enables individualized biological control strategies to maximize the growth promotion and stress resistance potential of T. harzianum.
3. Mechanisms of T. harzianum Promotes Stress Tolerance of Plants Under Abiotic Stresses
3.1. Improving Plant Root Development and Nutrient Uptake
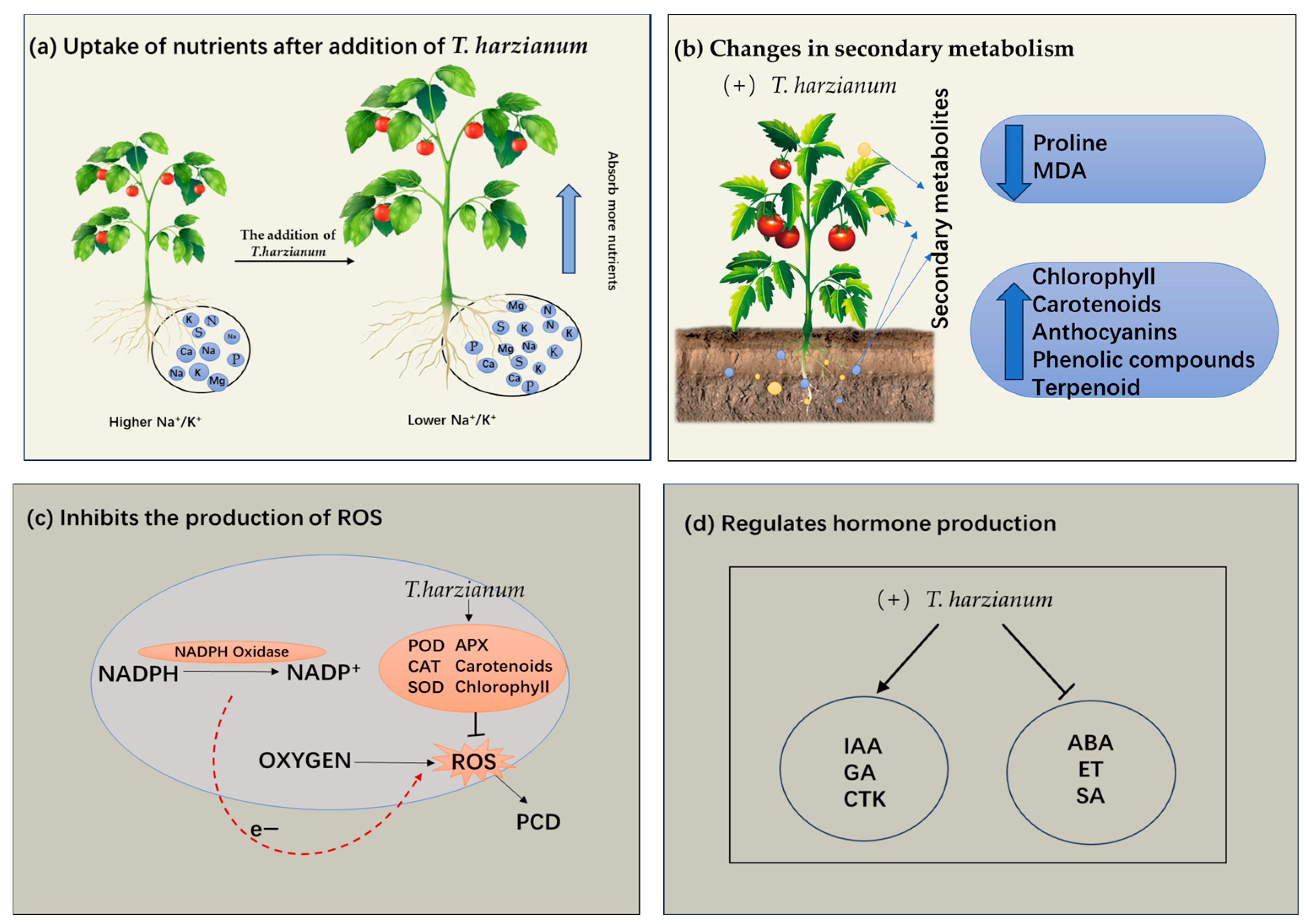
The effects of T. harzianum on plant roots have been a key research focus [33]. Negative effects of abiotic stresses on roots are characterized by reductions in root length and weight, inhibition of root growth, and reductions in root surface area and nutrient uptake. T. harzianum ameliorates injuries from abiotic stresses (Figure 1a). Under salt stress, the total root length of barley cultivars Gairdner and Vlamingh cultivars inoculated with T. harzianum increased [32]. Under drought stress, T. harzianum promoted lateral root branching and increased total root length from 32.5% to 42.7% [26]. In addition, peptaibol secreted by T. harzianum increased the fresh weight of pepper roots by enhancing membrane stability and osmotic regulation [19]. T. harzianum treatment increased proline accumulation by 50%, upregulated CsAPX expression, and improved salt tolerance in cucumber via promoting lateral root development [17].
Figure 1.
Four key mechanisms of T. harzianum to improve plant stress tolerance. (a) Promoting plant root growth and nutrient absorption. (b) Effect of T. harzianum on secondary metabolites in plants. (c) Enhancement of the plant’s antioxidant defense system. (d) Regulating the phytohormones. MDA: Malondialdehyde; ROS: Reactive oxygen species; PCD: Programmed cell death; POD: Peroxidase; APX: Ascorbate peroxidase; SOD: Superoxide dismutase; GA: Gibberellin; IAA: Indole acetic acid; ET: Endothelin; CTK: cytokinin; ABA: Abscisic acid; SA: Salicylic acid; CAT: Catalase.
Studies have shown that organic acids (such as oxalic acid, citric acid) secreted by T. harzianum can reduce soil pH and dissolve insoluble phosphorus and potassium, thereby releasing available nutrients such as phosphorus and potassium [34]. The concentrations of nutrients (N, P, S, Ca, Mg, and K) were significantly increased under different Trichoderma treatments, and the Na/K ratio was significantly reduced, mainly due to increased K absorption [25]. In addition, T. harzianum, as a species of Trichoderma, can produce various substances to promote plant growth, such as ethylene (Ethylene), and IAA to stimulate root development, and the hydrophobins and swollenin produced by Trichoderma assist in its colonization of plant roots [35]. When plants encounter salt stress via their roots, T. harzianum initiates a sophisticated protective response by modulating ion homeostasis, which facilitates the selective accumulation of beneficial ions and promotes the synthesis of compatible osmolytes, thereby effectively reducing ionic toxicity and maintaining cellular turgor pressure [17].
T. harzianum has shown great potential for use in promoting plant root absorption and development, enhancing stress resistance, and improving soil microecology. It has enormous potential for the future development of sustainable agriculture, ecological restoration, and smart agriculture.
3.2. Enhancement of Plant Secondary Metabolism
Recent studies have confirmed that levels of plant secondary metabolite accumulation significantly correlate with abiotic stress tolerance [36,37]. T. harzianum can enhance plant tolerance by modulating the levels of plant secondary metabolites (Figure 1b). In black pepper, T. harzianum inoculation under drought stress reduced proline accumulation and improved biomass yield compared with T. asperellum [19]. This interaction reduces lipid peroxidation, as evidenced by 40% lower malondialdehyde (MDA) levels in Trichoderma-colonized cucumber under salt stress compared with untreated plants [17]. T. harzianum also enhances stress resistance through broader metabolic reprogramming, including increased other synthesis and secondary metabolites such as phenol and flavonoid complexes [26]. T. harzianum also induces changes in terpenoid content in plants [38]. By modulating these metabolites, T. harzianum enhances plant resilience to multiple abiotic stressors.
The contents of chlorophyll a, chlorophyll b, carotenoids, and anthocyanins increased after inoculation with T. harzianum, and the maintained photosynthetic efficiency to varying degrees compared to the salt-stressed plants that were not inoculated with T. harzianum under 150 mM NaCl treatment [39]. This suggests that T. harzianum helps maintain photosynthesis in plants and supports normal growth and development under salt stress. Collectively, these findings demonstrate that T. harzianum enhances plant abiotic stress tolerance through multifaceted metabolic and physiological adjustments. This metabolic reprogramming not only improves plant survival under adverse conditions but also supports sustained growth and productivity, highlighting its potential as a powerful bioinoculant for stress-resilient agriculture.
3.3. Enhancement of Plant Antioxidant Defense System
Under abiotic stress, ROS in plants are altered [40]. ROS are reactive oxygen molecules in cells, and an excess of ROS leads to oxidative stress, which in turn triggers programmed cell death (PCD). NADPH oxidases play a pivotal role in plant growth and development by catalyzing the production of reactive oxygen species (ROS). These ROS act as signaling molecules that regulate various cellular processes essential for growth, including cell expansion. For instance, in Arabidopsis roots, the NADPH oxidase RHD2 generates ROS that are required to sustain root hair elongation [41]. Genetic mutation of RHD2 or pharmacological inhibition of NADPH oxidase activity with diphenylene iodonium (DPI) reduces ROS levels, resulting in a short-root-hair phenotype, demonstrating the essential role of NADPH oxidase-derived ROS in root hair growth regulation [41].
T. harzianum induces increased expression of antioxidant enzyme genes and elevated activity of antioxidant enzymes such as superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), etc., in plant cells, which reduces the level of ROS [17] (Figure 1c). Similar increases in POD and APX activity were observed in Trichoderma-treated Indian mustard under saline conditions and were associated with improved yield and stress tolerance [25]. As further evidence, inoculation of T. harzianum reduced H2O2 levels in cucurbits by 10% under salt stress, effectively reducing oxidative damage [39]. These results consistently suggest that T. harzianum plays a central protective role in plants in response to abiotic stresses by regulating the enzyme antioxidant defense system.
Moreover, T. harzianum increased chlorophyll and carotenoid levels by 30% in Satureja hortensis under salt stress, thereby maintaining photosynthetic efficiency and ROS homeostasis [38]. Preservation of chlorophyll ensures continued light harvesting under stress, while carotenoids act as antioxidants and reduce lipid peroxidation [26]. Thereby, T. harzianum can improve plant stress tolerance via both enzyme and non-enzyme antioxidant defense systems.
3.4. Regulation of Plant Hormone Balance
Phytohormones are important for various aspects of plant growth and development, such as cell division, elongation, and differentiation, as well as plant budding, rooting, and flowering [42]. Plants are subjected to a variety of abiotic stresses that elicit different responses to defend against external abiotic stresses, and phytohormones are the most important endogenous subunits that regulate physiological and molecular responses [43] which can enhance resistance under abiotic stresses. For example, auxin can help plants maintain growth and metabolic homeostasis by regulating the expression of recombinant genes under drought and salt stress [44].
Studies have shown that plant hormones regulate abiotic stress response through complex interaction networks and molecular mechanisms, while microorganisms (such as Trichoderma) can enhance plant stress resistance through multitarget intervention (Figure 1d). This versatile fungus enhances plant performance through multiple physiological pathways. It stimulates the production of IAA, a crucial phytohormone [22,26,27]. As an important phytohormone, ethylene is widely involved in regulating plant growth, development, and senescence [45]. Lower concentrations of ethylene in the root zone significantly promote bud growth and biomass accumulation [46]. T. harzianum reduces ethylene biosynthesis by secreting ACC deaminase, thereby alleviating the inhibitory effect of ethylene on plant growth [47]. Research also revealed that T. harzianum can promote the production of salicylic acid (SA) and cytokinin (CTK). When tomato is subjected to abiotic stress, T. harzianum enhances the synthesis of growth hormones such as IAA, gibberellin (GA), and indolebutyric acid (IBA) [26]. T. harzianum protects cell membranes from ROS damage by regulating the secondary metabolism and promotes root nutrient uptake, thereby synergistically enhancing plant stress resistance and growth [26]. Similarly, Contreras-Cornejo et al. found that T. harzianum could enhance the antioxidant defense system of plants by regulating endogenous hormones and gene expression to alleviate adversity stress, by investigating the mechanism of T. harzianum action under different abiotic stresses [48]. When the Thkel1 (transketolase-like 1) in T. harzianum was overexpressed in Arabidopsis thaliana, ABA levels in transgenic Arabidopsis were significantly lower than those in wild plants, which was more pronounced under salt stress [49]. Expression of the Thkel1 in Arabidopsis thaliana enhances plant tolerance to salt and osmotic stress, accompanied by increased glucosidase activity and decreased abscisic acid (ABA) levels compared to those observed in wild-type plants [49].
3.5. Improvement of Soil Environment and Structure
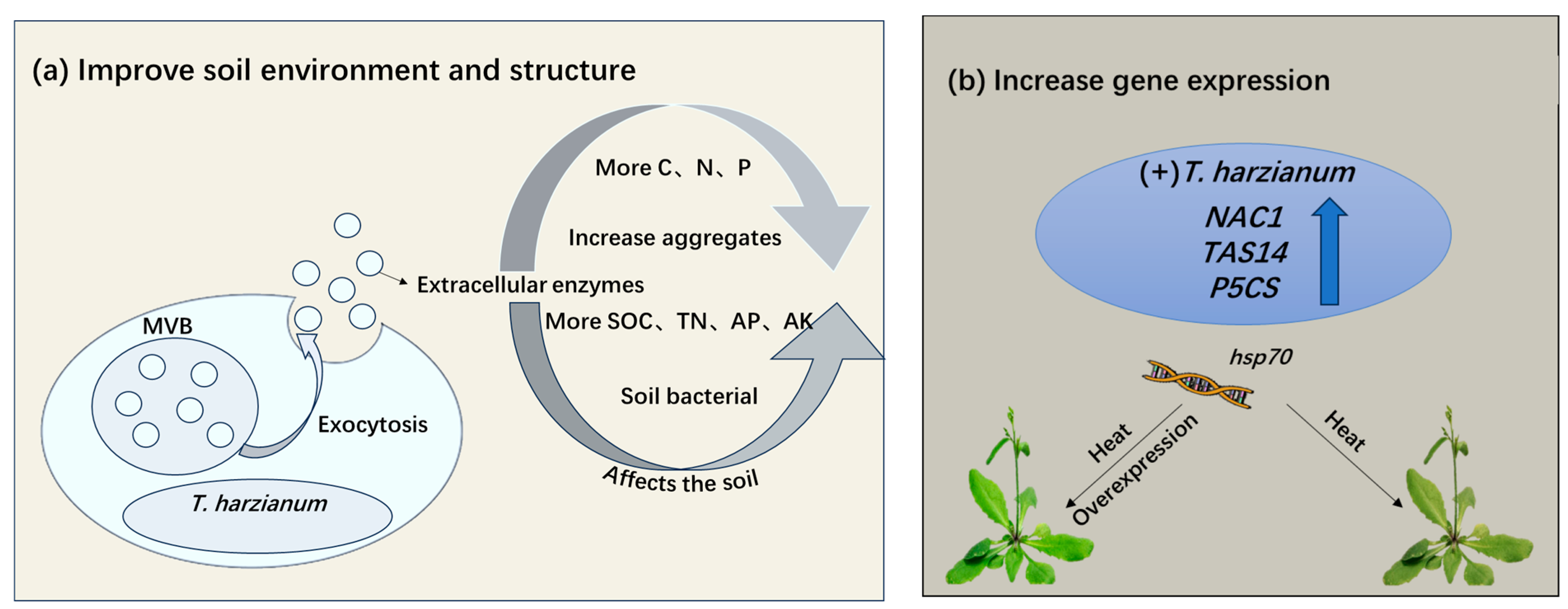
Abiotic stress is a stress condition that not only causes direct harm to plants but also alters the biochemical and structural properties of soils [50]. T. harzianum can alleviate the negative effects of abiotic stress on soil through multiple mechanisms to improve plant stress tolerance (Figure 2a). T. harzianum can secrete a variety of extracellular enzymes, such as protease, cellulase, ligninase, hemicellulase, etc., and gradually decompose complex or organic macromolecules into simple organic small molecules [51]. This releases carbon, nitrogen, phosphorus, and other elements in the soil for plants to absorb and utilize, and enhances soil fertility. T. harzianum can also significantly increase the content of large aggregates (>2 mm) and small aggregates (0.25–2 mm) in soil and improve the stability of soil aggregates. In one reported study, the contents of soil organic carbon, total nitrogen, available phosphorus, and available potassium increased [52]. The soil bacterial community structure was significantly changed by T. harzianum application, with increased relative abundance of Proteobacteria and Actinobacteria and decreased relative abundance of Acidobacteria and Chloroflexi, providing evidence for plant-Trichoderma interactions [52,53].

Figure 2.
T. harzianum enhances plant adaptability to abiotic stress by improving soil environment and regulating stress resistance genes. (a) Improvement of soil environment and crop structure. (b) Altering the expression of related genes that enhance plant resistance under abiotic stresses by T. harzianum. MVB: Multivesicular body; SOC: Soil organic carbon; TN: Total nitrogen; AP: Available phosphorus; AK: Available potassium; hsp70 (heat-shock protein 70), TAS14 inducible by ABA (abscisic acid) and environmental stress; NAC1 (NAM, ATAF1/2, and CUC2); P5CS (delta 1-Pyrroline-5-Carboxylate Synthetase).
T. harzianum effectively alleviates the adverse effects of abiotic stress on the soil–plant system by secreting various extracellular enzymes to decompose soil organic matter, improve aggregate structure, and regulate microbial communities. Future research should focus on elucidating the mechanisms by which its extracellular polysaccharides mediate soil structure restoration, developing synergistic microbial agents with functional microorganisms, optimizing application techniques for different soil–crop systems, and assessing its long-term ecological effects under extreme climatic conditions. These efforts aim to advance the large-scale application of this green biotechnology in soil remediation and sustainable agriculture.
3.6. Expression Analysis of the Hsp70 (Heat-Shock Protein 70), TAS14 Inducible by ABA and Environmental Stress, NAC1 (NAM, ATAF1/2, and CUC2) and P5CS (Delta 1-Pyrroline-5-Carboxylate Synthetase) Genes
Overexpression of the hsp70 gene in T. harzianum has emerged as a promising strategy to increase plant tolerance to various abiotic stresses [54,55]. In addition, hsp70 overexpression confers cross-tolerance to drought and salinity in plants by stabilizing cellular proteins and regulating ion homeostasis through interactions with stress-responsive pathways, such as SOS1-mediated Na+/H+ exchange [56].
The role of Trichoderma-derived genes in plant stress acclimation is not limited to hsp70. For example, inoculation of tomato plants with T. harzianum induced the expression of the NAC1, TAS14, and P5CS genes under cold stress conditions, with a 2.1-to-2.3-fold increase in the transcriptional level of P5CS [15] (Figure 2b). As important transcriptional regulators, NAC family genes are involved in plant development regulation and mediate adaptive responses to abiotic stresses [57]. Arabidopsis thaliana lines overexpressing NAC1 gene showed significantly improved plant survival under cold stress [58]. Overexpression of TAS14 enhanced plant tolerance to abiotic stresses such as drought and salt stress [59]. P5CS is a key gene for proline biosynthesis, which is accumulated in excess to enhance plant stress tolerance [60]. Research has demonstrated that T. harzianum treatment enhances drought tolerance of rice by modulating the expression of key genes involved in photosynthesis, antioxidant activity, osmotic regulation, and phytohormone signaling under drought stress [21]. These findings indicate that T. harzianum improves plants’ resilience to multiple abiotic stresses through transcriptional regulation.
In the future, it will be possible to validate the function of stress-resistant genes in major crops through cross-species verification and develop molecular marker-assisted breeding technologies. Synthetic biology methods can be used to construct multi-gene co-expression systems, which can be combined with gene editing technologies to optimize plant stress resistance. Finally, the agronomic stability of gene regulation can be assessed through multi-environment stress field trials, providing new targets and multi-dimensional breeding strategies for crop stress resistance and genetic improvement.
4. Conclusions
In conclusion, this review provides a robust theoretical foundation for T. harzianum application and insights into the molecular mechanisms of T. harzianum-mediated plant abiotic stress responses. T. harzianum, as an important promoting plant growth fungus, has shown remarkable effects in enhancing plant resistance to abiotic stresses such as drought, salinity, and extreme temperature. Its mechanism of action mainly includes improving the root environment, activating the plant antioxidant defense system, reducing the accumulation of reactive oxygen species, coordinating the plant hormone signaling network, etc., and optimizing the stress response. These synergistic effects significantly mitigate the adverse effects of stress on plant growth and development. Its multifaceted mechanisms make T. harzianum an exceptionally valuable biological tool for enhancing crop performance under challenging environmental conditions.
Although T. harzianum has made important progress in the application of abiotic stress, there are still some key scientific problems in this field that need to be solved urgently. First, there are significant differences in the stress response mechanisms of different genotypes of T. harzianum, and the molecular basis of its interaction with plants remain unclear. Secondly, the functional stability and regulatory network of strains under compound stress still need to be systematically analyzed. In view of these problems, future research should focus on the integration of multiomics technologies (genomics, transcriptomics, proteomics, etc.) to reveal strain-specific stress response mechanisms. In addition, it is necessary to develop efficient gene editing tools to improve key functional genes (such as antioxidant enzyme genes and osmoregulation-related genes). It is also necessary to optimize the formulation and application technology of the fungus agent to improve the colonization efficiency and application effect in the complex environment of the field. The study provides a robust theoretical foundation for T. harzianum application and insights into the molecular mechanisms of T. harzianaum-mediated plant stress responses.
Author Contributions
Y.G. and R.Z. wrote the original draft and conceived the study; S.C., P.L., Y.L., J.L., F.J., Z.W. and Q.S. revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
We acknowledge the supports from the funding from Anhui Higher Education Research Institute-Enterprise Joint Scientific Research and Talent Cultivation Project, Fundamental Research Funds for the Central Universities (KJYQ2025027), the Natural Science Foundation of Jiangsu Province (BK2022148), earmarked fund for China Agriculture Research System (CARS-23), and the Bioinformatics Center of Nanjing Agricultural University.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
There are no conflicts of interest.
References
- Rosegrant, M.W.; Sulser, T.B.; Dunston, S.; Mishra, A.; Cenacchi, N.; Gebretsadik, Y.; Robertson, R.; Thomas, T.; Wiebe, K. Food and nutrition security under changing climate and socioeconomic conditions. Glob. Food Secur. 2024, 41, 100755. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, J.; Zhao, L.; Wu, H.; Zhu, Y.; Ahmad, I.; Zhou, G. Abiotic stress responses in crop plants: A multi-scale approach. J. Integr. Agric. 2024, 23, 1456–1472. [Google Scholar] [CrossRef]
- Gupta, A.; Rico-Medina, A.; Caño-Delgado, A.I. The Physiology of Plant Responses to Drought. Science 2020, 368, 266–269. [Google Scholar] [CrossRef]
- Xie, G.; Xu, R.; Chong, L.; Zhu, Y. Understanding drought stress response mechanisms in tomato. Veg. Res. 2024, 4, e001. [Google Scholar] [CrossRef]
- Haghpanah, M.; Hashemipetroudi, S.; Arzani, A.; Araniti, F. Drought Tolerance in Plants: Physiological and Molecular Responses. Plants 2024, 13, 2962. [Google Scholar] [CrossRef] [PubMed]
- Hayat, S.; Hayat, Q.; Alyemeni, M.N.; Wani, A.S.; Pichtel, J.; Ahmad, A. Role of proline under changing environments: A review. Plant Signal. Behav. 2012, 7, 1456–1466. [Google Scholar] [CrossRef]
- Isayenkov, S.V.; Maathuis, F.J.M. Plant Salinity Stress: Many Unanswered Questions Remain. Front. Plant Sci. 2019, 10, 80. [Google Scholar] [CrossRef]
- Qian, D.; Wang, M.; Niu, Y.; Yang, Y.; Xiang, Y. Sexual reproduction in plants under high temperature and drought stress. Cell Rep. 2025, 44, 115390. [Google Scholar] [CrossRef]
- Scafaro, A.P.; Posch, B.C.; Evans, J.R.; Atkin, O.K. Rubisco Deactivation and Chloroplast Electron Transport Rates Co-Limit Photosynthesis Above Optimal Leaf Temperature in Terrestrial Plants. Nat. Commun. 2023, 14, 2820. [Google Scholar] [CrossRef]
- Rifai, M.A.; Webster, J. A Revision of the Genus Trichoderma. Mycol. Pap. 1969, 116, 1–56. [Google Scholar]
- Yan, S.H.; Wu, S.P.; Lu, D.Q.; Chen, X.J. Effects of Biocontrol Strain of Trichoderma harzianum on Microflora in Rhizosphere and Its Interactions with Microbe. Southwest China J. Agric. Sci. 2005, 18, 40–46. [Google Scholar]
- Mohammad, A.; Khan, R.M.; Khan, S.H. T. harzianum (Th. azad) as a Mycoparasite of Fusarium and Growth Enhancer of Tomato in Glasshouse Conditions. J. Pure Appl. Microbiol. 2016, 10, 1463–1468. [Google Scholar]
- Woo, S.L.; Ruocco, M.; Vinale, F.; Nigro, M.; Marra, R.; Lombardi, N.; Pascale, A.; Lanzuise, S.; Manganiello, G.; Lorito, M. Trichoderma: A multipurpose, plant-beneficial microorganism for eco-sustainable agriculture. Nat. Rev. Microbiol. 2023, 21, 312–326. [Google Scholar] [CrossRef]
- Ahmad, S.A.; Muhammad, T. Effects of different inoculum densities of T. harzianum and Trichoderma viride against Meloidogyne javanica on tomato. Saudi J. Biol. Sci. 2016, 23, 2. [Google Scholar]
- Ghorbanpour, A.; Salimi, A.; Ghanbary, M.A.T.; Pirdashti, H.; Dehestani, A. The effect of T. harzianum in mitigating low temperature stress in tomato (Solanum lycopersicum L.) plants. Sci. Hortic. 2017, 230, 134–141. [Google Scholar] [CrossRef]
- Khoshmanzar, E.; Aliasgharzad, N.; Rezaei, R.; Neyshabouri, M.R. Effects of Trichoderma Isolates on Tomato Growth and Inducing Its Tolerance to Water-Deficit Stress. Int. J. Environ. Sci. Technol. 2020, 17, 869–878. [Google Scholar] [CrossRef]
- Zhang, F.; Wang, Y.; Liu, C.; Chen, F.; Ge, H.; Tian, F.; Yang, T.; Ma, K.; Zhang, Y. T. harzianum mitigates salt stress in cucumber via multiple responses. Ecotoxicol. Environ. Saf. 2019, 170, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Chen, X.; Wang, Y.; Zhang, H.; Li, J. Effects of Solid Matrix Priming with T. harzianum on Seed Germination, Seedling Emergence and Photosynthetic Capacity of Eggplant. Afr. J. Biotechnol. 2017, 16, 699–703. [Google Scholar]
- Valiyambath, V.K.; Thomas, T.A.; George, P.; Nair, S.; Kumar, A. Characterization and Quantification of Peptaibol Produced by Novel Trichoderma spp.: Harnessing Their Potential to Mitigate Moisture Stress Through Enhanced Biochemical and Physiological Responses in Black Pepper (Piper nigrum L.). World J. Microb. Biot. 2024, 40, 330. [Google Scholar] [CrossRef]
- Bashyal, B.M.; Zaidi, N.W.; Singh, S.A.R. Effect of fungal biocontrol agents on enhancement of drought stress tolerance in rice (Oryza sativa L.). Indian J. Biochem. Biophys. 2020, 57, 101–108. [Google Scholar]
- Bashyal, B.M.; Gupta, R.; Pareek, N.; Rai, S. Molecular Programming of Drought-Challenged T. harzianum-Bioprimed Rice (Oryza sativa L.). Front. Microbiol. 2021, 12, 655165. [Google Scholar] [CrossRef]
- Pandey, V.; Ansari, M.W.; Tula, S.; Yadav, S.; Sahoo, R.K.; Shukla, N.; Bains, G.; Badal, S.; Chandra, S.; Gaur, A.K.; et al. Dose-dependent response of T. harzianum in improving drought tolerance in rice genotypes. Planta 2016, 243, 1251–1264. [Google Scholar] [CrossRef] [PubMed]
- Saravanakumar, K.; Li, Y.; Yu, C.; Wang, Q.; Wang, M. Effect of T. harzianum on Maize Rhizosphere Microbiome and Biocontrol of Fusarium Stalk Rot. Sci. Rep. 2017, 7, 1771. [Google Scholar] [CrossRef] [PubMed]
- Han, Y. Metabolic and Antioxidant Responses of Different Control Methods to the Interaction of Sorghum sudangrass hy-brids-Colletotrichum boninense. Int. J. Mol. Sci. 2024, 25, 9505. [Google Scholar]
- Saha, K.C.; Uddin, M.K.; Shaha, P.K.; Hossain Chowdhury, M.A.; Hassan, L.; Saha, B.K. Application of T. harzianum enhances salt tolerance and yield of Indian mustard through increasing antioxidant enzyme activity. Heliyon 2024, 11, e41114. [Google Scholar] [CrossRef]
- Alwhibi, M.S.; Alqarawi, A.A.; Wirth, S.; Egamberdieva, D. Increased Resistance of Drought by T. harzianum Fungal Treatment Correlates with Increased Secondary Metabolites and Proline Content. J. Integr. Agric. 2017, 16, 1751–1760. [Google Scholar]
- Lian, H.; Li, R.; Ma, G.; Zhang, X.; Wang, Y.; Liu, Y. The Effect of T. harzianum Agents on Physiological-Biochemical Characteristics of Cucumber and the Control Effect Against Fusarium Wilt. Sci. Rep. 2023, 13, 17606. [Google Scholar] [CrossRef]
- Martínez-Medina, A.; Roldán, A.; Pascual, J.A. Interaction between arbuscular mycorrhizal fungi and T. harzianum under conventional and low input fertilization field condition in melon crops: Growth response and Fusarium wilt biocontrol. Appl. Soil Ecol. 2011, 47, 98–105. [Google Scholar] [CrossRef]
- Coppola, M.; Diretto, G.; Digilio, M.C.; Woo, S.L.; Giuliano, G.; Molisso, D.; Pennacchio, F.; Lorito, M.; Rao, R. Transcriptome and Metabolome Reprogramming in Tomato Plants by T. harzianum strain T22 Primes and Enhances Defense Responses Against Aphids. Front. Physiol. 2019, 10, 745. [Google Scholar] [CrossRef]
- Lombardi, N.; Caira, S.; Troise, A.D.; Scaloni, A.; Vitaglione, P.; Vinale, F.; Marra, R.; Salzano, A.M.; Lorito, M.; Woo, S.L. Trichoderma applications on strawberry plants modulate the physiological processes positively affecting fruit production and quality. Front. Microbiol. 2020, 11, 1364. [Google Scholar] [CrossRef]
- Wu, Y.-J.; Chen, Y.-L.; Li, J.-Y.; Zhang, H.; Wang, X.-M. T. harzianum Promoting Chrysanthemum Cutting Rooting and Reshaping Microbial Communities in Endophytic and Rhizosphere Environments. Appl. Soil Ecol. 2024, 203, 105636. [Google Scholar] [CrossRef]
- Gupta, S.; Chakraborty, D.; Mishra, A.; Singh, R.P. Inoculation of Barley with T. harzianum T-22 Modifies Lipids and Metabolites to Improve Salt Tolerance. J. Exp. Bot. 2021, 72, 7229–7246. [Google Scholar] [CrossRef] [PubMed]
- Dhanyalakshmi, K.H.; Pawar, R.K.; Aishwarya, G.; Kumar, P. Shaping Root System Architecture: Holobiont-Based Phenotyping for Root Traits. Plant Physiol. Rep. 2024, 29, 716–725. [Google Scholar] [CrossRef]
- Altomare, C.; Norvell, W.A.; Björkman, T.; Harman, G.E. Solubilization of phosphates and micronutrients by the plant-growth-promoting and biocontrol fungus Trichoderma harzianum Rifai 1295-22. Appl. Environ. Microbiol. 1999, 65, 2926–2933. [Google Scholar] [CrossRef] [PubMed]
- Druzhinina, I.S.; Seidl-Seiboth, V.; Herrera-Estrella, A.; Horwitz, B.A.; Kenerley, C.M.; Monte, E.; Mukherjee, P.K.; Zeilinger, S.; Grigoriev, I.V.; Kubicek, C.P. Trichoderma: The Genomics of Opportunistic Success. Nat. Rev. Microbiol. 2011, 9, 749–759. [Google Scholar] [CrossRef]
- Chávez-Arias, C.C.; Ramírez-Godoy, A.; Restrepo-Díaz, H. Influence of drought, high temperatures, and/or defense against arthropod herbivory on the production of secondary metabolites in maize plants. A review. Curr. Plant Biol. 2022, 32, 100268. [Google Scholar] [CrossRef]
- Kaur, S.; Samota, M.K.; Choudhary, M.; Choudhary, M.; Pandey, A.K.; Sharma, A.; Thakur, J. How do plants defend themselves against pathogens-biochemical mechanisms and genetic interventions. Physiol. Mol. Biol. Plants 2022, 28, 485–504. [Google Scholar] [CrossRef]
- Sabzi-Nojadeh, A.M.; Pouresmaeil, M.; Amani, M.; Younessi-Hamzekhanlu, M.; Maggi, F. Colonization of Satureja hortensis L. (Summer savory) with T. harzianum alleviates salinity stress via improving physio-biochemical traits and biosynthesis of secondary metabolites. Ind. Crops Prod. 2024, 208, 117831. [Google Scholar] [CrossRef]
- Soliman, M.H.; Alnusaire, T.S.; Abdelbaky, N.F.; Alayafi, A.A.M.; Hasanuzzaman, M.; Rowezak, M.M.; El-Esawi, M.; Elkelish, A. Trichoderma-Induced Improvement in Growth, Photosynthetic Pigments, Proline, and Glutathione Levels in Cucurbita pepo Seedlings under Salt Stress. Phyton-Int. J. Exp. Bot. 2020, 89, 473–486. [Google Scholar] [CrossRef]
- Choudhary, A.; Sharma, S.; Kaur, H.; Sharma, N.; Gadewar, M.M.; Mehta, S.; Husen, A. Plant system, abiotic stress resilience, reactive oxygen species, and coordination of engineered nanomaterials: A review. S. Afr. J. Bot. 2024, 171, 45–59. [Google Scholar] [CrossRef]
- Foreman, J.; Demidchik, V.; Bothwell, J.; Mylona, P.; Miedema, H.; Torres, M.A.; Linstead, P.; Costa, S.; Brownlee, C.; Jones, J.D.G.; et al. Reactive oxygen species produced by NADPH oxidase regulate plant cell growth. Nature 2003, 422, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Bouzayen, M. Plant Hormones: A broader understanding of plant development processes for better adapted agriculture. Plant Horm. 2025, 1, e001. [Google Scholar] [CrossRef]
- Fahad, S.; Hussain, S.; Bano, A.; Saud, S.; Hassan, S.; Shan, D.; Khan, F.A.; Khan, F.; Chen, Y.; Wu, C. Potential role of phytohormones and plant growth-promoting rhizobacteria in abiotic stresses: Consequences for changing environment. Environ. Sci. Pollut. Res. 2015, 22, 4907–4921. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zhuang, S.; Zhang, W. Advances in plant auxin biology: Synthesis, metabolism, signaling, interaction with other hormones, and roles under abiotic stress. Plants 2024, 13, 2523. [Google Scholar] [CrossRef]
- Khan, N.A.; Khan, M.I.R.; Ferrante, A.; Poor, P. Editorial: Ethylene: A key regulatory molecule in plants. Front. Plant Sci. 2017, 8, 1782. [Google Scholar] [CrossRef]
- Peres, L.E.P.; Amar, S.; Kerbauy, G.B.; Salatino, A.; Zaffar, G.R.; Mercier, H. Effects of Auxin, Cytokinin and Ethylene Treatments on the Endogenous Ethylene and Auxin-to-Cytokinin Ratio Related to Direct Root Tip Conversion of Catasetum fimbriatum Lindl. (Orchidaceae) into Buds. J. Plant Physiol. 1999, 155, 551–555. [Google Scholar] [CrossRef]
- Illescas, M.; Pedrero-Méndez, A.; Pitorini-Bovolini, M.; Hermosa, R.; Monte, E. Phytohormone Production Profiles in Trichoderma Species and Their Relationship to Wheat Plant Responses to Water Stress. Pathogens 2021, 10, 991. [Google Scholar] [CrossRef]
- Contreras-Cornejo, H.A.; Schmoll, M.; Esquivel-Ayala, B.A.; López-Bucio, J. Abiotic Plant Stress Mitigation by Trichoderma Species. Soil Ecol. Lett. 2024, 6, 240240. [Google Scholar] [CrossRef]
- Hermosa, R.; Botella, L.; Keck, E.; Jiménez, J.Á.; Montero-Barrientos, M.; Arbona, V.; Gómez-Cadenas, A.; Monte, E.; Nicolás, C. The overexpression in Arabidopsis thaliana of a T. harzianum gene that modulates glucosidase activity, and enhances tolerance to salt and osmotic stresses. J. Plant Physiol. 2011, 168, 1295–1302. [Google Scholar] [CrossRef]
- Dervash, M.A.; Yousuf, A.; Bhat, M.A.; Ozturk, M. Soil stress ecology: Concept, impacts, and management strategies. In Soil Organisms; SpringerBriefs in Microbiology; Springer: Cham, Switzerland, 2024; p. 3. [Google Scholar]
- Singh, S.; Singh, A.K.; Pradhan, B.; Kumar, A.; Pandey, A. Harnessing Trichoderma Mycoparasitism as a Tool in the Management of Soil Dwelling Plant Pathogens. Microb. Ecol. 2024, 87, 158. [Google Scholar] [CrossRef]
- Zhua, L.; Wang, Y.; Wang, G.; Xiao, M.; Luo, H.; Zhang, F.; Li, L. Manure and T. harzianum Increase Cotton Yield via Regulating Soil Bacterial Community and Physicochemical Properties. ScienceAsia 2024, 50, 1–10. [Google Scholar] [CrossRef]
- De Palma, M.; Salzano, M.; Villano, C.; Aversano, R.; Lorito, M.; Ruocco, M.; Docimo, T.; Piccinelli, A.L.; D’Agostino, N.; Tucci, M. Transcriptome reprogramming, epigenetic modifications and alternative splicing orchestrate the tomato root response to the beneficial fungus Trichoderma harzianum. Hortic. Res. 2019, 6, 5. [Google Scholar] [CrossRef]
- Usman, M.G.; Rafii, M.Y.; Martini, M.Y.; Yusuff, O.A.; Ismail, M.R.; Miah, G. Molecular analysis of Hsp70 mechanisms in plants and their function in response to stress. Biotechnol. Genet. Eng. Rev. 2017, 33, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Alam, P.; Al Balawi, T.; Manzoor, M.A.; Al-Malki, A.L.; Alansi, S. Genome-Wide Analysis of HSP70 Gene Family in Beta vulgaris and In-Silico Expression Under Environmental Stress. BMC Plant Biol. 2025, 25, 214. [Google Scholar] [CrossRef] [PubMed]
- Montero-Barrientos, M.; Hermosa, R.; Nicolás, C.; Cardoza, R.E.; Gutiérrez, S. Overexpression of a Trichoderma HSP70 Gene Increases Fungal Resistance to Heat and Other Abiotic Stresses. Fungal Genet. Biol. 2008, 45, 1506–1513. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; You, J.; Fang, Y.; Zhu, X.; Qi, Z.; Xiong, L. Characterization of Transcription Factor Gene SNAC2 Conferring Cold and Salt Tolerance in Rice. Plant Mol. Biol. 2008, 67, 169–181. [Google Scholar] [CrossRef]
- Li, X.-L.; Yang, X.; Hu, Y.-X.; Yu, X.-D.; Chen, X.-Y. A Novel NAC Transcription Factor from Suaeda liaotungensis K. Enhanced Transgenic Arabidopsis Drought, Salt, and Cold Stress Tolerance. Plant Cell Rep. 2014, 33, 767–778. [Google Scholar] [CrossRef]
- Muñoz-Mayor, A.; Pineda, B.; Garcia-Abellán, J.O.; Antón, T.; Garcia-Sogo, B.; Sanchez-Bel, P.; Flores, F.B.; Atarés, A.; Angosto, T.; Pintor-Toro, J.A.; et al. Overexpression of Dehydrin tas14 Gene Improves the Osmotic Stress Imposed by Drought and Salinity in Tomato. J. Plant Physiol. 2012, 169, 459–468. [Google Scholar] [CrossRef]
- Rai, A.N.; Penna, S. Molecular Evolution of Plant P5CS Gene Involved in Proline Biosynthesis. Mol. Biol. Rep. 2013, 40, 6429–6435. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).