Antitumor Effects of Natural Compounds Derived from Allium sativum on Neuroblastoma: An Overview
Abstract
1. Neuroblastoma
2. Garlic
3. Types of Garlic Preparations
4. Antioxidant Activity of Garlic and Garlic Compounds
5. Allicin and NB
6. Ajoene and NB
7. SAC and NB
8. DAS, DADS, DATS, and NB
9. Effects of Garlic on Neurodegenerative Diseases
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Maris, J.M. Recent Advances in Neuroblastoma. N. Engl. J. Med. 2010, 362, 2202–2211. [Google Scholar] [CrossRef]
- PDQ Pediatric Treatment Editorial Board. Neuroblastoma Treatment (PDQ®): Health Professional Version. In PDQ Cancer Information Summaries; Bethesda: Rockville, MD, USA; National Cancer Institute: Rockville, MD, USA, 2002. [Google Scholar]
- London, W.B.; Castleberry, R.P.; Matthay, K.K.; Look, A.; Seeger, R.C.; Shimada, H.; Thorner, P.S.; Brodeur, G.M.; Maris, J.M.; Reynolds, C.P.; et al. Evidence for an Age Cutoff Greater Than 365 Days for Neuroblastoma Risk Group Stratification in the Children’s Oncology Group. J. Clin. Oncol. 2005, 23, 6459–6465. [Google Scholar] [CrossRef]
- Zafar, A.; Wang, W.; Liu, G.; Wang, X.; Xian, W.; McKeon, F.; Foster, J.; Zhou, J.; Zhang, R. Molecular targeting therapies for neuroblastoma: Progress and challenges. Med. Res. Rev. 2020, 41, 961–1021. [Google Scholar] [CrossRef]
- Cohn, S.L.; Pearson, A.D.J.; London, W.B.; Monclair, T.; Ambros, P.F.; Brodeur, G.M.; Faldum, A.; Hero, B.; Iehara, T.; Machin, D.; et al. The international neuroblastoma risk group (INRG) classification system: An INRG task force report. J. Clin. Oncol. 2009, 27, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Kholodenko, I.V.; Kalinovsky, D.V.; Doronin, I.I.; Deyev, S.M.; Kholodenko, R.V. Neuroblastoma Origin and Therapeutic Targets for Immunotherapy. J. Immunol. Res. 2018, 2018, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Twist, C.J.; Schmidt, M.L.; Naranjo, A.; London, W.B.; Tenney, S.C.; Marachelian, A.; Shimada, H.; Collins, M.H.; Esiashvili, N.; Adkins, E.S.; et al. Maintaining Outstanding Outcomes Using Response- and Biology-Based Therapy for Intermediate-Risk Neuroblastoma: A Report from the Children’s Oncology Group Study ANBL0531. J. Clin. Oncol. 2019, 37, 3243–3255. [Google Scholar] [CrossRef] [PubMed]
- Tolbert, V.P.; Matthay, K.K. Neuroblastoma: Clinical and biological approach to risk stratification and treatment. Cell Tissue Res. 2018, 372, 195–209. [Google Scholar] [CrossRef]
- Berlanga, P.; Cañete, A.; Castel, V. Advances in emerging drugs for the treatment of neuroblastoma. Expert Opin. Emerg. Drugs 2016, 22, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Matthay, K.K.; Reynolds, C.P.; Seeger, R.C.; Shimada, H.; Adkins, E.S.; Haas-Kogan, D.; Gerbing, R.B.; London, W.B.; Villablanca, J.G. Long-Term Results for Children with High-Risk Neuroblastoma Treated on a Randomized Trial of Myeloablative Therapy Followed by 13-cis-Retinoic Acid: A Children’s Oncology Group Study. J. Clin. Oncol. 2009, 27, 1007–1013. [Google Scholar] [CrossRef] [PubMed]
- Castel, V.; Cañete, A.; Navarro, S.; García-Miguel, P.; Melero, C.; Acha, T.; Navajas, A.; Badal, M. Outcome of high-risk neuroblastoma using a dose intensity approach: Improvement in initial but not in long-term results. Med. Pediatr. Oncol. 2001, 37, 537–542. [Google Scholar] [CrossRef] [PubMed]
- Luksch, R.; Castellani, M.R.; Collini, P.; De Bernardi, B.; Conte, M.; Gambini, C.; Gandola, L.; Garaventa, A.; Biasoni, D.; Podda, M.; et al. Neuroblastoma (Peripheral neuroblastic tumours). Crit. Rev. Oncol. 2016, 107, 163–181. [Google Scholar] [CrossRef]
- Schultz, C.R.; Gruhlke, M.C.H.; Slusarenko, A.J.; Bachmann, A.S. Allicin, a Potent New Ornithine Decarboxylase Inhibitor in Neuroblastoma Cells. J. Nat. Prod. 2020, 83, 2518–2527. [Google Scholar] [CrossRef]
- Zhuang, J.; Li, Y.; Chi, Y. Role of p38 MAPK activation and mitochondrial cytochrome-c release in allicin-induced apoptosis in SK-N-SH cells. Anti-Cancer Drugs 2016, 27, 312–317. [Google Scholar] [CrossRef]
- Gao, X.-Y.; Geng, X.-J.; Zhai, W.-L.; Zhang, X.-W.; Wei, Y.; Hou, G.-J. Effect of combined treatment with cyclophosphamidum and allicin on neuroblastoma–bearing mice. Asian Pac. J. Trop. Med. 2015, 8, 137–141. [Google Scholar] [CrossRef]
- Terrasson, J.; Xu, B.; Li, M.; Allart, S.; Davignon, J.-L.; Zhang, L.-H.; Wang, K.; Davrinche, C. Activities of Z-ajoene against tumour and viral spreading in vitro. Fundam. Clin. Pharmacol. 2007, 21, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Welch, C.; Wuarin, L.; Sidell, N. Antiproliferative effect of the garlic compound S-allyl cysteine on human neuroblastoma cells in vitro. Cancer Lett. 1992, 63, 211–219. [Google Scholar] [CrossRef]
- Kanamori, Y.; Via, L.D.; Macone, A.; Canettieri, G.; Greco, A.; Toninello, A.; Agostinelli, E. Aged garlic extract and its constituent, S-allyl-L-cysteine, induce the apoptosis of neuroblastoma cancer cells due to mitochondrial membrane depolarization. Exp. Ther. Med. 2019, 19, 1511–1521. [Google Scholar] [CrossRef]
- Filomeni, G.; Aquilano, K.; Rotilio, G.; Ciriolo, M.R. Reactive oxygen species-dependent c-Jun NH2-terminal kinase/c-Jun signaling cascade mediates neuroblastoma cell death induced by diallyl disulfide. Cancer Res. 2003, 63, 5940–5949. [Google Scholar] [PubMed]
- Karmakar, S.; Banik, N.L.; Patel, S.J.; Ray, S.K. Garlic compounds induced calpain and intrinsic caspase cascade for apoptosis in human malignant neuroblastoma SH-SY5Y cells. Apoptosis 2007, 12, 671–684. [Google Scholar] [CrossRef]
- Aquilano, K.; Vigilanza, P.; Filomeni, G.; Rotilio, G.; Ciriolo, M.R. Tau dephosphorylation and microfilaments disruption are upstream events of the anti-proliferative effects of dads in sh-sy5y cells. J. Cell. Mol. Med. 2008, 14, 564–577. [Google Scholar] [CrossRef]
- Pagliei, B.; Aquilano, K.; Baldelli, S.; Ciriolo, M.R. Garlic-derived diallyl disulfide modulates peroxisome proliferator activated receptor gamma co-activator 1 alpha in neuroblastoma cells. Biochem. Pharmacol. 2013, 85, 335–344. [Google Scholar] [CrossRef]
- Jurkowska, H.; Wróbel, M.; Kaczor-Kamińska, M.; Jasek-Gajda, E. A possible mechanism of inhibition of U87MG and SH-SY5Y cancer cell proliferation by diallyl trisulfide and other aspects of its activity. Amino Acids 2017, 49, 1855–1866. [Google Scholar] [CrossRef] [PubMed]
- White, D. Healthy Uses for Garlic. Nurs. Clin. N. Am. 2021, 56, 153–156. [Google Scholar] [CrossRef]
- Moyers, S. Garlic in Health, History and World Cuisine; Suncoast Press: St Petersburg, FL, USA, 1996; pp. 1–36. [Google Scholar]
- Woodward, P.W. Garlic and Friends: The History, Growth and Use of Edible Alliums; Hyland House: Melbourne, Australia, 1996; pp. 2–22. [Google Scholar]
- Gebhardt, R.; Beck, H.; Wagner, K.G. Inhibition of cholesterol biosynthesis by allicin and ajoene in rat hepatocytes and HepG2 cells. Biochim. Biophys. Acta (BBA) Lipids Lipid Metab. 1994, 1213, 57–62. [Google Scholar] [CrossRef]
- Eilat, S.; Oestraicher, Y.; Rabinkov, A.; Ohad, D.; Mirelman, D.; Battler, A.; Eldar, M.; Vered, Z. Alteration of lipid profile in hyperlipidemic rabbits by allicin, an active constituent of garlic. Coron. Arter. Dis. 1995, 6, 985–990. [Google Scholar]
- Shadkchan, Y.; Shemesh, E.; Mirelman, D.; Miron, T.; Rabinkov, A.; Wilchek, M.; Osherov, N. Efficacy of allicin, the reactive molecule of garlic, in inhibiting Aspergillus spp. in vitro, and in a murine model of disseminated aspergillosis. J. Antimicrob. Chemother. 2004, 53, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Benavides, G.A.; Squadrito, G.L.; Mills, R.W.; Patel, H.D.; Isbell, T.S.; Patel, R.P.; Darley-Usmar, V.M.; Doeller, J.E.; Kraus, D.W. Hydrogen sulfide mediates the vasoactivity of garlic. Proc. Natl. Acad. Sci. USA 2007, 104, 17977–17982. [Google Scholar] [CrossRef]
- Zoccali, C.; Catalano, C.; Rastelli, S. Blood pressure control: Hydrogen sulfide, a new gasotransmitter, takes stage. Nephrol. Dial. Transplant. 2009, 24, 1394–1396. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Ruan, J.; Zhuang, X.; Zhang, X.; Li, Z. Phytochemicals of garlic: Promising candidates for cancer therapy. Biomed. Pharmacother. 2019, 123, 109730. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Huang, G.; Huang, H. The antioxidant activities of garlic polysaccharide and its derivatives. Int. J. Biol. Macromol. 2019, 145, 819–826. [Google Scholar] [CrossRef]
- Furhad, S.; Bokhari, A.A. Herbal Supplements; StatPearls Publishing: St. Petersburg, FL, USA, 2021. [Google Scholar]
- Varshney, R.; Budoff, M.J. Garlic and Heart Disease. J. Nutr. 2016, 146, 416S–421S. [Google Scholar] [CrossRef]
- Mathew, B.; Biju, R. Neuroprotective Effects of Garlic: A Review. Libyan J. Med. 2008, 3, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Lawson, L.D. Garlic: A review of its medicinal effects and indicated active compounds. Phytomedicines of Europe. Chem. Biol. Act. 1998, 691, 176–209. [Google Scholar]
- Iberl, B.; Winkler, G.; Müller, B.; Knobloch, K. Quantitative Determination of Allicin and Alliin from Garlic by HPLC. Planta Med. 1990, 56, 320–326. [Google Scholar] [CrossRef]
- Iberl, B.; Winkler, G.; Knobloch, K. Products of Allicin Transformation: Ajoenes and Dithiins, Characterization and their Determination by HPLC. Planta Med. 1990, 56, 202–211. [Google Scholar] [CrossRef]
- Lawson, L.D.; Hughes, B.G. Characterization of the formation of allicin and other thiosulfinates from garlic. Planta Med. 1992, 58, 345–350. [Google Scholar] [CrossRef]
- Borek, C. Antioxidant Health Effects of Aged Garlic Extract. J. Nutr. 2001, 131, 1010S–1015S. [Google Scholar] [CrossRef]
- Czepukojc, B.; Baltes, A.-K.; Cerella, C.; Kelkel, M.; Viswanathan, U.M.; Salm, F.; Burkholz, T.; Schneider, C.; Dicato, M.; Montenarh, M.; et al. Synthetic polysulfane derivatives induce cell cycle arrest and apoptotic cell death in human hematopoietic cancer cells. Food Chem. Toxicol. 2014, 64, 249–257. [Google Scholar] [CrossRef]
- Nicastro, H.L.; Ross, S.A.; Milner, J.A. Garlic and Onions: Their Cancer Prevention Properties. Cancer Prev. Res. 2015, 8, 181–189. [Google Scholar] [CrossRef]
- Steinmetz, K.A.; Kushi, L.H.; Bostick, R.M.; Folsom, A.R.; Potter, J.D. Vegetables, Fruit, and Colon Cancer in the lowa Women’s Health Study. Am. J. Epidemiol. 1994, 139, 1–15. [Google Scholar] [CrossRef]
- Dorant, E.; Brandt, P.V.D.; Goldbohm, R.; Hermus, R.; Sturmans, F. Garlic and its significance for the prevention of cancer in humans: A critical view. Br. J. Cancer 1993, 67, 424–429. [Google Scholar] [CrossRef]
- Trio, P.Z.; You, S.; He, X.; He, J.; Sakao, K.; Hou, D.-X. Chemopreventive functions and molecular mechanisms of garlic organosulfur compounds. Food Funct. 2014, 5, 833–844. [Google Scholar] [CrossRef]
- Farhat, Z.; Hershberger, P.A.; Freudenheim, J.L.; Mammen, M.J.; Blair, R.H.; Aga, D.S.; Mu, L. Types of garlic and their anticancer and antioxidant activity: A review of the epidemiologic and experimental evidence. Eur. J. Nutr. 2021, 60, 3585–3609. [Google Scholar] [CrossRef]
- Munday, R.; Munday, C.M. Low Doses of Diallyl Disulfide, a Compound Derived from Garlic, Increase Tissue Activities of Quinone Reductase and Glutathione Transferase in the Gastrointestinal Tract of the Rat. Nutr. Cancer 1999, 34, 42–48. [Google Scholar] [CrossRef]
- Sheen, L.-Y.; Chen, H.-W.; Kung, Y.-L.; Liu, C.-T.; Lii, C.-K. Effects of Garlic Oil and Its Organosulfur Compounds on the Activities of Hepatic Drug-Metabolizing and Antioxidant Enzymes in Rats Fed High- and Low-Fat Diets. Nutr. Cancer 1999, 35, 160–166. [Google Scholar] [CrossRef]
- Balasenthil, S.; Arivazhagan, S.; Ramachandran, C.R.; Nagini, S. Effects of garlic on 7,12-Dimethylbenz[a]anthracene-induced hamster buccal pouch carcinogenesis. Cancer Detect. Prev. 1999, 23, 534–538. [Google Scholar] [CrossRef]
- Wei, Z.; Lau, B.H. Garlic inhibits free radical generation and augments antioxidant enzyme activity in vascular endothelial cells. Nutr. Res. 1998, 18, 61–70. [Google Scholar] [CrossRef]
- Mukherjee, S.; Banerjee, S.K.; Maulik, M.; Dinda, A.K.; Talwar, K.K.; Maulik, S.K. Protection against acute adriamycin-induced cardiotoxicity by garlic: Role of endogenous antioxidants and inhibition of TNF-α expression. BMC Pharmacol. 2003, 3, 16. [Google Scholar] [CrossRef][Green Version]
- Horev-Azaria, L.; Eliav, S.; Izigov, N.; Pri-Chen, S.; Mirelman, D.; Miron, T.; Rabinkov, A.; Wilchek, M.; Jacob-Hirsch, J.; Amariglio, N.; et al. Allicin up-regulates cellular glutathione level in vascular endothelial cells. Eur. J. Nutr. 2008, 48, 67–74. [Google Scholar] [CrossRef]
- Thomson, M.; Al-Qattan, K.K.; Js, D.; Ali, M. Anti-diabetic and anti-oxidant potential of aged garlic extract (AGE) in streptozotocin-induced diabetic rats. BMC Complement. Altern. Med. 2015, 16, 17. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, J.; Dou, C.; Li, N.; Kang, F.; Wang, Y.; Cao, Z.; Yang, X.; Dong, S. Alliin Attenuated RANKL-Induced Osteoclastogenesis by Scavenging Reactive Oxygen Species through Inhibiting Nox1. Int. J. Mol. Sci. 2016, 17, 1516. [Google Scholar] [CrossRef]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.C.H.; Nwachukwu, I.; Slusarenko, A.J. Allicin: Chemistry and Biological Properties. Molecules 2014, 19, 12591–12618. [Google Scholar] [CrossRef]
- Gruhlke, M.C.H.; Slusarenko, A.J. The Chemistry of Alliums. Molecules 2018, 23, 143. [Google Scholar] [CrossRef] [PubMed]
- Bat-Chen, W.; Golan, T.; Peri, I.; Ludmer, Z.; Schwartz, B. Allicin Purified from Fresh Garlic Cloves Induces Apoptosis in Colon Cancer Cells Via Nrf2. Nutr. Cancer 2010, 62, 947–957. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhu, B.; Zhao, L.; Liu, Y.; Zhao, F.; Feng, J.; Jin, Y.; Sun, J.; Geng, R.; Wei, Y. Allicin Inhibits Proliferation and Invasion in Vitro and in Vivo via SHP-1-Mediated STAT3 Signaling in Cholangiocarcinoma. Cell. Physiol. Biochem. 2018, 47, 641–653. [Google Scholar] [CrossRef]
- Li, X.; Ni, J.; Tang, Y.; Wang, X.; Tang, H.; Li, H.; Zhang, S.; Shen, X. Allicin inhibits mouse colorectal tumorigenesis through suppressing the activation of STAT3 signaling pathway. Nat. Prod. Res. 2018, 33, 2722–2725. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Liang, J.; Sun, J.; Hu, X.; Lei, L.; Wu, D.; Liu, L. Allicin sensitizes hepatocellular cancer cells to anti-tumor activity of 5-fluorouracil through ROS-mediated mitochondrial pathway. J. Pharmacol. Sci. 2016, 131, 233–240. [Google Scholar] [CrossRef]
- Jiang, W.; Huang, Y.; Wang, J.-P.; Yu, X.-Y.; Zhang, L.-Y. The synergistic anticancer effect of Artesunate COMBINED with allicin in osteosarcoma cell line in vitro and in vivo. Asian Pac. J. Cancer Prev. 2013, 14, 4615–4619. [Google Scholar] [CrossRef]
- Jobani, B.M.; Najafzadeh, N.; Mazani, M.; Arzanlou, M.; Vardin, M.M. Molecular mechanism and cytotoxicity of allicin and all-trans retinoic acid against CD44+ versus CD117+ melanoma cells. Phytomedicine 2018, 48, 161–169. [Google Scholar] [CrossRef]
- Auvinen, M.; Paasinen, A.; Andersson, L.C.; Hölttä, E. Ornithine decarboxylase activity is critical for cell transformation. Nature 1992, 360, 355–358. [Google Scholar] [CrossRef]
- Bachmann, A.S.; Geerts, D. Polyamine synthesis as a target of MYC oncogenes. J. Biol. Chem. 2018, 293, 18757–18769. [Google Scholar] [CrossRef]
- Ribatti, D. Anti-angiogenesis in neuroblastoma. Crit. Rev. Oncol. 2013, 86, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Ciu, J.-R.; Ye, Y.; Min, J.-M.; Zhang, L.-H.; Wang, K.; Gares, M.; Cros, J.; Wright, M.; Leung-Tack, J. Antitumor activity of Z-ajoene, a natural compound purified from garlic: Antimitotic and microtubule-interaction properties. Carcinogenesis 2002, 23, 573–579. [Google Scholar] [CrossRef]
- Chauhan, N.B. Effect of aged garlic extract on APP processing and tau phosphorylation in Alzheimer’s transgenic model Tg2576. J. Ethnopharmacol. 2006, 108, 385–394. [Google Scholar] [CrossRef]
- Jeong, J.H.; Jeong, H.R.; Na Jo, Y.; Kim, H.J.; Shin, J.H.; Heo, H.J. Ameliorating effects of aged garlic extracts against Aβ-induced neurotoxicity and cognitive impairment. BMC Complement. Altern. Med. 2013, 13, 268. [Google Scholar] [CrossRef]
- Thorajak, P.; Pannangrong, W.; Welbat, J.U.; Chaijaroonkhanarak, W.; Sripanidkulchai, K.; Sripanidkulchai, B. Effects of Aged Garlic Extract on Cholinergic, Glutamatergic and GABAergic Systems with Regard to Cognitive Impairment in Aβ-Induced Rats. Nutrients 2017, 9, 686. [Google Scholar] [CrossRef] [PubMed]
- Pannangrong, W.; Welbat, J.U.; Chaichun, A.; Sripanidkulchai, B. Effect of combined extracts of aged garlic, ginger, and chili peppers on cognitive performance and brain antioxidant markers in Aβ-induced rats. Exp. Anim. 2020, 69, 269–278. [Google Scholar] [CrossRef]
- Rojas, P.; Serrano-García, N.; Medina-Campos, O.N.; Pedraza-Chaverri, J.; Maldonado, P.D.; Ruiz-Sánchez, E. S-Allylcysteine, a garlic compound, protects against oxidative stress in 1-methyl-4-phenylpyridinium-induced parkinsonism in mice. J. Nutr. Biochem. 2011, 22, 937–944. [Google Scholar] [CrossRef]
- Bigham, M.; Mohammadipour, A.; Hosseini, M.; Malvandi, A.M.; Ebrahimzadeh-Bideskan, A. Neuroprotective effects of garlic extract on dopaminergic neurons of substantia nigra in a rat model of Parkinson’s disease: Motor and non-motor outcomes. Metab. Brain Dis. 2021, 36, 927–937. [Google Scholar] [CrossRef] [PubMed]
- La Cruz, V.P.-D.; González-Cortés, C.; Pedraza-Chaverri, J.; Maldonado, P.D.; Andrés-Martínez, L.; Santamaría, A. Protective effect of S-allylcysteine on 3-nitropropionic acid-induced lipid peroxidation and mitochondrial dysfunction in rat brain synaptosomes. Brain Res. Bull. 2006, 68, 379–383. [Google Scholar] [CrossRef]
- Herrera-Mundo, M.N.; Adaya, I.D.S.; Maldonado, P.D.; Galván-Arzate, S.; Andrés-Martínez, L.; La Cruz, V.P.-D.; Pedraza-Chaverrí, J.; Santamaría, A. S-Allylcysteine prevents the rat from 3-nitropropionic acid-induced hyperactivity, early markers of oxidative stress and mitochondrial dysfunction. Neurosci. Res. 2006, 56, 39–44. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, K.; Wang, Q.; Li, Z.; Yin, Y.; Xu, Q.; Duan, W.; Li, C. Neuroprotective effects of diallyl trisulfide in SOD1-G93A transgenic mouse model of amyotrophic lateral sclerosis. Brain Res. 2011, 1374, 110–115. [Google Scholar] [CrossRef]
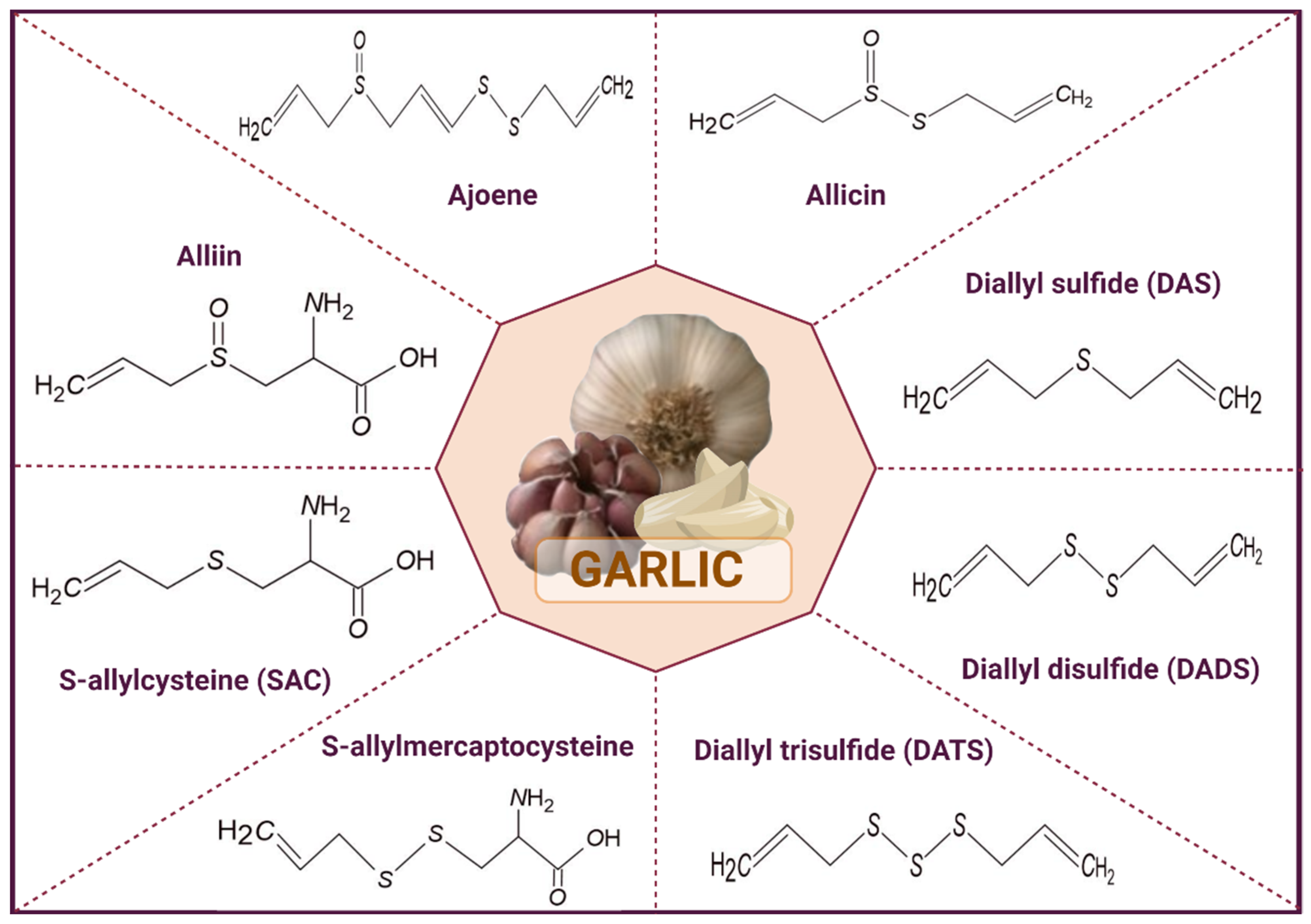
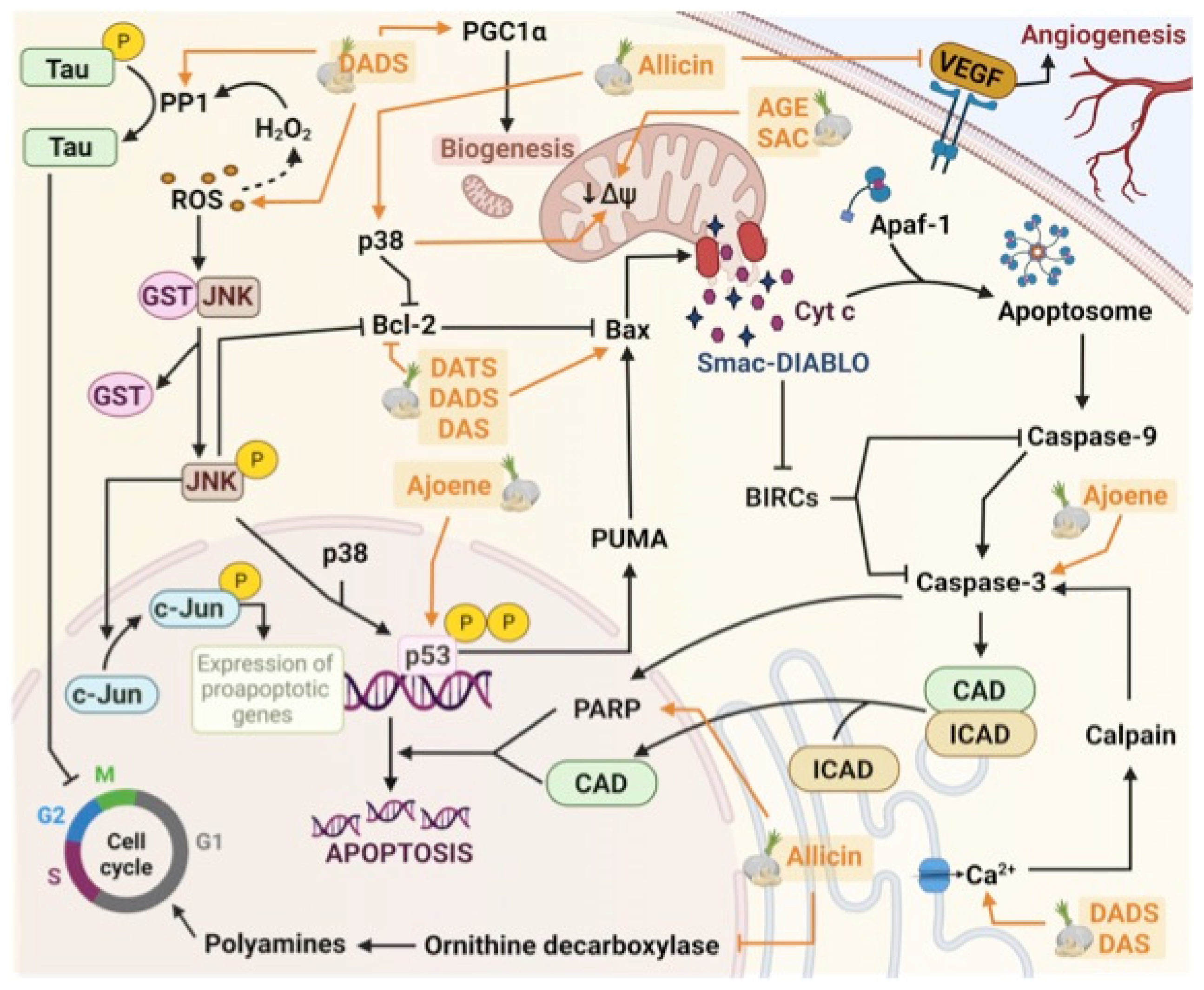
| Compound Used | Experimental Model | Main Findings | Reference |
|---|---|---|---|
| Allicin | SK-NFI, SK-N-AS, SK-N-Be, and Kelly cell lines | Induction of programmed cell death by the increase of poly (ADP-ribose) polymerase (PARP). | [13] |
| Allicin | SK-N-SH cell line | Induction of apoptosis by activating the p38 MAPK pathway and the release of cytochrome c. | [14] |
| Allicin | BALB/c-nu/nu mice | Tumour cell proliferation decreased; increase the number of CD4+, CD8+, and NK cells and IFN-γ levels in the serum; and decrease in mRNA and protein levels of VEGF. | [15] |
| Z-ajoene | SK-N-AS cell line | Increase active caspase-3 and p53. | [16] |
| SAC | LA-N-5 human NB cell line | Decrease cell proliferation. | [17] |
| SAC | SJ-NK-P and IMR5 cell lines | Induction of apoptosis and cell cycle arrest in the G1 phase. | [18] |
| AGE | SJ-NK-P and IMR5 cell lines | Decrease the mitochondrial membrane potential and increased glutathione oxidation. | [18] |
| DADS | SH-SY5Y cell line | Release of cytochrome c, and activation of caspase-9 in association with the activation of the JNK/c-Jun pathway. | [19] |
| DAS and DADS | SH-SY5Y cell line | Both compounds increase the intracellular Ca2+ and induce cell death through the release of cytochrome c. | [20] |
| DADS | SH-SY5Y cell line | Disruption of the cytoskeleton by Tau protein dephosphorylation. | [21] |
| DADS | SH-SY5Y cell line | Activation of PGC1α. | [22] |
| DATS | U87MG and SH-SY5Y cell line | Decrease of Bcl-2, and increased hydrogen sulfide production and ROS production. | [23] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patiño-Morales, C.C.; Jaime-Cruz, R.; Sánchez-Gómez, C.; Corona, J.C.; Hernández-Cruz, E.Y.; Kalinova-Jelezova, I.; Pedraza-Chaverri, J.; Maldonado, P.D.; Silva-Islas, C.A.; Salazar-García, M. Antitumor Effects of Natural Compounds Derived from Allium sativum on Neuroblastoma: An Overview. Antioxidants 2022, 11, 48. https://doi.org/10.3390/antiox11010048
Patiño-Morales CC, Jaime-Cruz R, Sánchez-Gómez C, Corona JC, Hernández-Cruz EY, Kalinova-Jelezova I, Pedraza-Chaverri J, Maldonado PD, Silva-Islas CA, Salazar-García M. Antitumor Effects of Natural Compounds Derived from Allium sativum on Neuroblastoma: An Overview. Antioxidants. 2022; 11(1):48. https://doi.org/10.3390/antiox11010048
Chicago/Turabian StylePatiño-Morales, Carlos César, Ricardo Jaime-Cruz, Concepción Sánchez-Gómez, Juan Carlos Corona, Estefani Yaquelin Hernández-Cruz, Ivia Kalinova-Jelezova, José Pedraza-Chaverri, Perla D. Maldonado, Carlos Alfredo Silva-Islas, and Marcela Salazar-García. 2022. "Antitumor Effects of Natural Compounds Derived from Allium sativum on Neuroblastoma: An Overview" Antioxidants 11, no. 1: 48. https://doi.org/10.3390/antiox11010048
APA StylePatiño-Morales, C. C., Jaime-Cruz, R., Sánchez-Gómez, C., Corona, J. C., Hernández-Cruz, E. Y., Kalinova-Jelezova, I., Pedraza-Chaverri, J., Maldonado, P. D., Silva-Islas, C. A., & Salazar-García, M. (2022). Antitumor Effects of Natural Compounds Derived from Allium sativum on Neuroblastoma: An Overview. Antioxidants, 11(1), 48. https://doi.org/10.3390/antiox11010048










