A Nation-Wide, Multi-Center Study on the Quality of Life of ALS Patients in Germany
Abstract
1. Introduction
- Perform a descriptive analysis of HRQoL in a large, nation-wide ALS cohort, with an emphasis on the differences between lALS and bALS;
- Perform an analysis of some medical interventions that may influence QoL in ALS patients treated according to current standards of care; and
- Perform a comparison of two QoL questionnaires, namely the EQ-5D-5L and the ALSAQ-5.
2. Methods
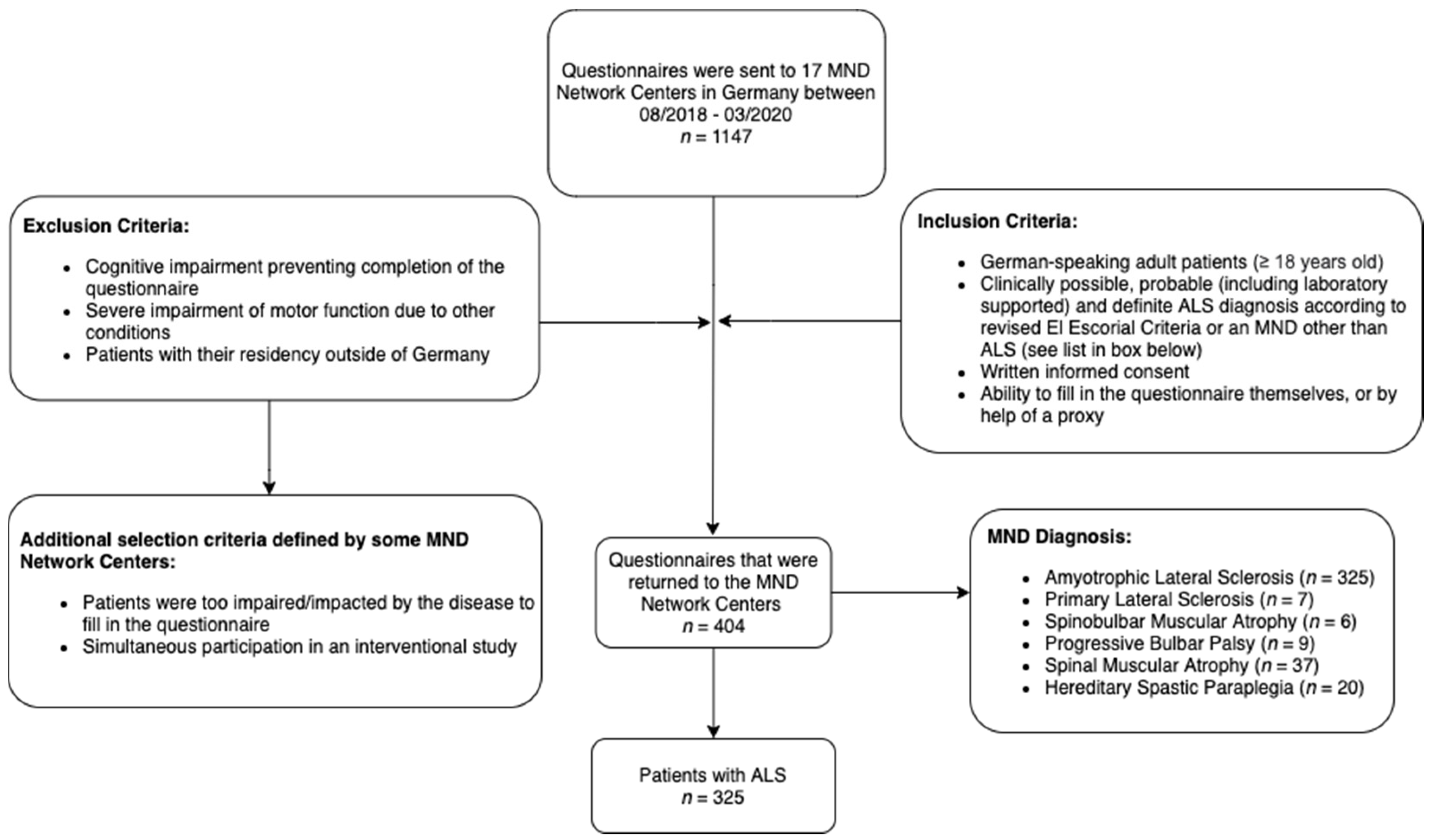
2.1. Study Design, Setting and Participants
2.2. Study Questionnaire
2.3. Health-Related Quality of Life Measures
2.4. Statistical Analysis
3. Results
3.1. Participants
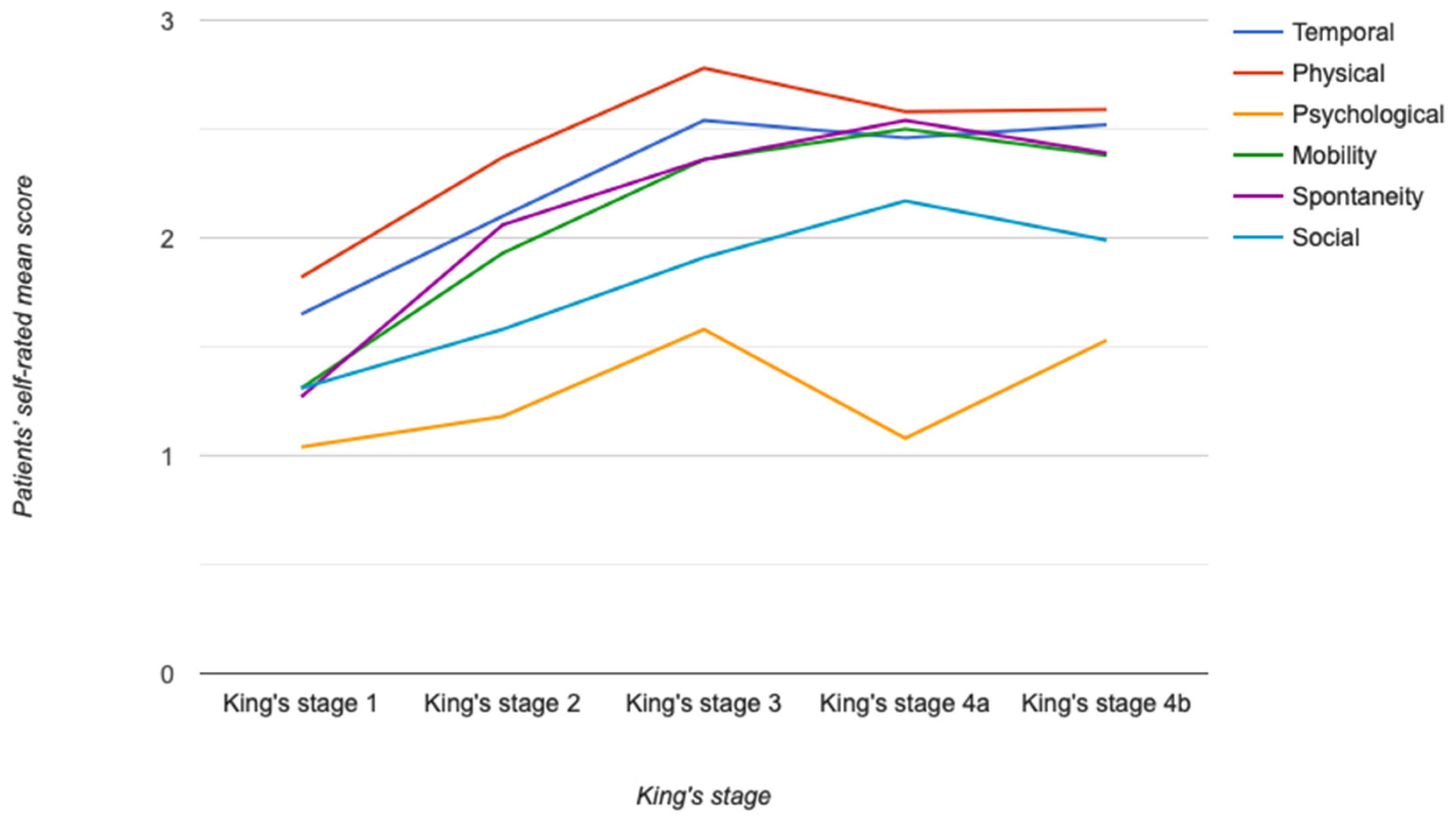
3.2. Descriptive Analysis of HRQoL in the Total Cohort
3.3. Positive Influence of Mobility Aids and Ventilator Support on HRQoL
3.4. Effect of Bulbar Dsyfunction on HRQoL
3.5. Comparison of the EQ-5D-5L and the ALSAQ-5 Questionnaires
4. Discussion
Limitations and Strengths
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | n (Percentage) |
|---|---|
| Feeding tube | 325 |
| Yes | 44 (13.5%) |
| No | 281 (86.5%) |
| Respiratory aid | 317 |
| Yes | 101 (31.9%) |
| No | 216 (68.1%) |
| Tracheostomy | 325 |
| Yes | 14 (4.3%) |
| No | 311 (95.7%) |
| Non-invasive ventilation | 325 |
| Yes | 66 (20.3%) |
| No | 259 (79.7%) |
| Other respiratory aid (incl. portable oxygen device, inhalation device and suction device) | 317 |
| Yes | 21 (6.6%) |
| No | 296 (93.4%) |
| Mobility aid | 321 |
| Yes | 219 (68.2%) |
| No | 102 (31.8%) |
| Communication aid | 314 |
| Yes | 100 (31.8%) |
| No | 214 (68.2%) |
| Caring aid | 317 |
| Yes | 186 (58.7%) |
| No | 131 (41.3%) |
| Speech therapy | 325 |
| Yes | 169 (52.0%) |
| No | 156 (48.0%) |
| Respiratory therapy | 325 |
| Yes | 31 (9.5%) |
| No | 294 (90.5) |
| Physiotherapy | 325 |
| Yes | 261 (80.3%) |
| No | 64 (19.7%) |
| Ergotherapy | 325 |
| Yes | 178 (54.8%) |
| No | 147 (45.2%) |
| Lymphatic drainage | 325 |
| Yes | 14 (4.3%) |
| No | 311 (95.7%) |
| Psychological intervention | 299 |
| Yes | 29 (9.7%) |
| No | 270 (90.3%) |
| Riluzole | 321 |
| Yes | 275 (85.7%) |
| No | 46 (14.3%) |
| Antidepressants | 321 |
| Yes | 64 (19.9%) |
| No | 257 (80.1%) |
| Opioid analgesic drugs | 321 |
| Yes | 16 (5.0%) |
| No | 305 (95.0%) |
| Non-opioid analgesic drugs | 321 |
| Yes | 27 (8.4%) |
| No | 294 (91.6%) |
| Benzodiazepines | 321 |
| Yes | 10 (3.1%) |
| No | 311 (96.9%) |
| Antispasmodics | 321 |
| Yes | 33 (10.3%) |
| No | 288 (89.7%) |
| Anticholinergics | 325 |
| Yes | 29 (8.9%) |
| No | 296 (91.1%) |
| Cannabis 1 | 321 |
| Yes | 11 (3.4%) |
| No | 310 (96.6%) |
| Edaravone | 321 |
| Yes | 20 (6.2%) |
| No | 301 (93.8%) |
| State | n Included in Study (Percentage) | Total Population of the State, 2019 [in Thousands] (Percentage) [84] |
|---|---|---|
| Lower Saxony | 133 (40.9%) | 7994 (9.7%) |
| North Rhine-Westphalia | 74 (22.8%) | 17,947 (21.6%) |
| Bavaria | 52 (16.0%) | 13,125 (15.8%) |
| Saxony | 15 (4.6%) | 4072 (4.9%) |
| Baden-Wuerttemberg | 10 (3.1%) | 11,100 (13.3%) |
| Hesse | 8 (2.5%) | 6288 (7.6%) |
| Schleswig-Holstein | 8 (2.5%) | 2904 (3.5%) |
| Saxony-Anhalt | 8 (2.5%) | 2195 (2.6%) |
| Rhineland-Palatinate | 4 (1.2%) | 4094 (4.9%) |
| Bremen | 4 (1.2%) | 681 (0.8%) |
| Mecklenburg-Western Pomerania | 3 (0.9%) | 1608 (1.9%) |
| Brandenburg | 2 (0.6%) | 2522 (3.0%) |
| Hamburg | 2 (0.6%) | 1847 (2.2%) |
| Thuringia | 1 (0.3%) | 2133 (2.6%) |
| Berlin | 0 (0.0%) | 3669 (4.4%) |
| Saarland | 0 (0.0%) | 987 (1.2%) |
| (Did not answer) | 1 (0.3%) | N/A |
References
- Zarei, S.; Carr, K.; Reiley, L.; Diaz, K.; Guerra, O.; Altamirano, P.F.; Pagani, W.; Lodin, D.; Orozco, G.; Chinea, A. A comprehensive review of amyotrophic lateral sclerosis. Surg. Neurol. Int. 2015, 6, 171. [Google Scholar] [CrossRef]
- Rosenbohm, A.; The ALS Registry Study Group; Peter, R.S.; Erhardt, S.; Lulé, D.; Rothenbacher, D.; Ludolph, A.C.; Nagel, G. Epidemiology of amyotrophic lateral sclerosis in Southern Germany. J. Neurol. 2017, 264, 749–757. [Google Scholar] [CrossRef] [PubMed]
- Yunusova, Y.; Plowman, E.K.; Green, J.R.; Barnett, C.; Bede, P. Clinical Measures of Bulbar Dysfunction in ALS. Front. Neurol. 2019, 10, 106. [Google Scholar] [CrossRef]
- Kiernan, M.C.; Vucic, S.; Cheah, B.C.; Turner, M.R.; Eisen, A.; Hardiman, O.; Burrell, J.R.; Zoing, M.C. Amyotrophic lateral sclerosis. Lancet 2011, 377, 942–955. [Google Scholar] [CrossRef]
- Schönfelder, E.; Osmanovic, A.; Müschen, L.H.; Petri, S.; Schreiber-Katz, O. Costs of illness in amyotrophic lateral sclerosis (ALS): A cross-sectional survey in Germany. Orphanet J. Rare Dis. 2020, 15, 149. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHOQOL: Measuring Quality of Life; World Health Organization: Geneva, Switzerland, 1997. [Google Scholar]
- Van Groenestijn, A.C.; Reenen, E.T.K.-V.; Visser-Meily, J.M.A.; Berg, L.H.V.D.; Schröder, C.D. Associations between psychological factors and health-related quality of life and global quality of life in patients with ALS: A systematic review. Health Qual. Life Outcomes 2016, 14, 107. [Google Scholar] [CrossRef] [PubMed]
- Burns, T.M.; Graham, C.D.; Rose, M.R.; Simmons, Z. Quality of life and measures of quality of life in patients with neuromuscular disorders. Muscle Nerve 2011, 46, 9–25. [Google Scholar] [CrossRef] [PubMed]
- Winter, Y.; Schepelmann, K.; Spottke, A.E.; Claus, D.; Grothe, C.; Schröder, R.; Heuss, D.; Vielhaber, S.; Tackenberg, B.; Mylius, V.; et al. Health-related quality of life in ALS, myasthenia gravis and facioscapulohumeral muscular dystrophy. J. Neurol. 2010, 257, 1473–1481. [Google Scholar] [CrossRef] [PubMed]
- Sandstedt, P.; Johansson, S.; Ytterberg, C.; Ingre, C.; Holmqvist, L.W.; Kierkegaard, M. Predictors of health-related quality of life in people with amyotrophic lateral sclerosis. J. Neurol. Sci. 2016, 370, 269–273. [Google Scholar] [CrossRef]
- Kiebert, G.M.; Green, C.; Murphy, C.; Mitchell, J.; O’Brien, M.; Burrell, A.; Leigh, P. Patients’ health-related quality of life and utilities associated with different stages of amyotrophic lateral sclerosis. J. Neurol. Sci. 2001, 191, 87–93. [Google Scholar] [CrossRef]
- Thakore, N.J.; Pioro, E.P.; Udeh, B.L.; Lapin, B.R.; Katzan, I.L. A Cost-Effectiveness Framework for Amyotrophic Lateral Sclerosis, Applied to Riluzole. Value Health 2020, 23, 1543–1551. [Google Scholar] [CrossRef]
- Miller, R.G.; Mitchell, J.D.; Moore, D.H. Riluzole for amyotrophic lateral sclerosis (ALS)/motor neuron disease (MND). Cochrane Database Syst. Rev. 2012, 2012, CD001447. [Google Scholar] [CrossRef]
- Andersen, P.M.; Abrahams, S.; Borasio, G.D.; De Carvalho, M.; Chio, A.; Van Damme, P.; Hardiman, O.; Kollewe, K.; Morrison, K.E.; Petri, S.; et al. EFNS guidelines on the Clinical Management of Amyotrophic Lateral Sclerosis (MALS), revised report of an EFNS task force. Eur. J. Neurol. 2011, 19, 360–375. [Google Scholar] [CrossRef]
- Bourke, S.C.; Tomlinson, M.; Williams, T.L.; Bullock, R.E.; Shaw, P.J.; Gibson, G.J. Effects of non-invasive ventilation on survival and quality of life in patients with amyotrophic lateral sclerosis: A randomised controlled trial. Lancet Neurol. 2006, 5, 140–147. [Google Scholar] [CrossRef]
- Vergonjeanne, M.; Fayemendy, P.; Marin, B.; Penoty, M.; Lautrette, G.; Sourisseau, H.; Preux, P.-M.; Desport, J.-C.; Couratier, P.; Jésus, P. Predictive factors for gastrostomy at time of diagnosis and impact on survival in patients with amyotrophic lateral sclerosis. Clin. Nutr. 2020, 39, 3112–3118. [Google Scholar] [CrossRef]
- Linse, K.; Rüger, W.; Joos, M.; Schmitz-Peiffer, H.; Storch, A.; Hermann, A. Eye-tracking-based assessment suggests preserved well-being in locked-in patients. Ann. Neurol. 2017, 81, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Linse, K.; Rüger, W.; Joos, M.; Schmitz-Peiffer, H.; Storch, A.; Hermann, A. Usability of eyetracking computer systems and impact on psychological wellbeing in patients with advanced amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Front. Degener. 2017, 19, 212–219. [Google Scholar] [CrossRef]
- Körner, S.; Siniawski, M.; Kollewe, K.; Rath, K.J.; Krampfl, K.; Zapf, A.; Dengler, R.; Petri, S. Speech therapy and communication device: Impact on quality of life and mood in patients with amyotrophic lateral sclerosis. Amyotroph. Lateral Scler. Front. Degener. 2012, 14, 20–25. [Google Scholar] [CrossRef]
- Hogden, A.; Foley, G.; Henderson, R.D.; James, N.; Aoun, S.M. Amyotrophic lateral sclerosis: Improving care with a multidisciplinary approach. J. Multidiscip. Healthc. 2017, 10, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Garuti, G.; Rao, F.; Ribuffo, V.; Sansone, V.A. Sialorrhea in patients with ALS: Current treatment options. Degener. Neurol. Neuromuscul. Dis. 2019, 9, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Pioro, E.P. Review of Dextromethorphan 20 mg/Quinidine 10 mg (NUEDEXTA®) for Pseudobulbar Affect. Neurol. Ther. 2014, 3, 15–28. [Google Scholar] [CrossRef] [PubMed]
- Bond, L.; Bowen, G.; Mertens, B.; Denson, K.; Jordan, K.; Vidakovic, B.; Mitchell, C.S. Associations of Patient Mood, Modulators of Quality of Life, and Pharmaceuticals with Amyotrophic Lateral Sclerosis Survival Duration. Behav. Sci. 2020, 10, 33. [Google Scholar] [CrossRef] [PubMed]
- Duffy, E.I.; Garry, J.; Talbot, L.; Pasternak, D.; Flinn, A.; Minardi, C.; Dookram, M.; Grant, K.; Fitzgerald, D.; Rubano, J.; et al. A pilot study assessing the spiritual, emotional, physical/environmental, and physiological needs of mechanically ventilated surgical intensive care unit patients via eye tracking devices, head nodding, and communication boards. Trauma Surg. Acute Care Open 2018, 3, e000180. [Google Scholar] [CrossRef] [PubMed]
- Pagnini, F.; Lunetta, C.; Rossi, G.; Banfi, P.; Gorni, K.; Cellotto, N.; Castelnuovo, G.; Molinari, E.; Corbo, M. Existential well-being and spirituality of individuals with amyotrophic lateral sclerosis is related to psychological well-being of their caregivers. Amyotroph. Lateral Scler. 2010, 12, 105–108. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, M.R.; Clark, D. Spirituality and/or religious faith: A means for coping with the effects of amyotrophic lateral sclerosis/motor neuron disease? Palliat. Support. Care 2015, 13, 1603–1614. [Google Scholar] [CrossRef] [PubMed]
- Shellikeri, S.; Karthikeyan, V.; Martino, R.; Black, S.; Zinman, L.; Keith, J.; Yunusova, Y. The neuropathological signature of bulbar-onset ALS: A systematic review. Neurosci. Biobehav. Rev. 2017, 75, 378–392. [Google Scholar] [CrossRef]
- Moura, M.C.; Novaes, M.R.C.G.; Eduardo, E.J.; Zago, Y.S.S.P.; Freitas, R.D.N.B.; Casulari, L.A. Prognostic Factors in Amyotrophic Lateral Sclerosis: A Population-Based Study. PLoS ONE 2015, 10, e0141500. [Google Scholar] [CrossRef] [PubMed]
- Elamin, M.; Bede, P.; Montuschi, A.; Pender, N.; Chio, A.; Hardiman, O. Predicting prognosis in amyotrophic lateral sclerosis: A simple algorithm. J. Neurol. 2015, 262, 1447–1454. [Google Scholar] [CrossRef]
- Chiò, A.; Logroscino, G.; Hardiman, O.; Swingler, R.; Mitchell, D.; Beghi, E.; Traynor, B.G.; On Behalf of the Eurals Consortium. Prognostic factors in ALS: A critical review. Amyotroph. Lateral Scler. 2009, 10, 310–323. [Google Scholar] [CrossRef]
- Waito, A.A.; Valenzano, T.J.; Peladeau-Pigeon, M.; Steele, C.M. Trends in Research Literature Describing Dysphagia in Motor Neuron Diseases (MND): A Scoping Review. Dysphagia 2017, 32, 734–747. [Google Scholar] [CrossRef]
- Herdman, M.; Gudex, C.; Lloyd, A.; Janssen, M.F.; Kind, P.; Parkin, D.; Bonsel, G.; Badia, X. Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Qual. Life Res. 2011, 20, 1727–1736. [Google Scholar] [CrossRef] [PubMed]
- Dodel, R.; Peter, H.; Spottke, A.; Noelker, C.; Althaus, A.; Siebert, U.; Walbert, T.; Kesper, K.; Becker, H.F.; Mayer, G. Health-related quality of life in patients with narcolepsy. Sleep Med. 2007, 8, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Reuther, M.; Spottke, E.; Klotsche, J.; Riedel, O.; Peter, H.; Berger, K.; Athen, O.; Kohne-Volland, R.; Dodel, R. Assessing health-related quality of life in patients with Parkinson’s disease in a prospective longitudinal study. Park. Relat. Disord. 2007, 13, 108–114. [Google Scholar] [CrossRef]
- Michalowsky, B.; Xie, F.; Kohlmann, T.; Gräske, J.; Wübbeler, M.; Thyrian, J.R.; Hoffmann, W. Acceptability and Validity of the EQ-5D in Patients Living with Dementia. Value Health 2020, 23, 760–767. [Google Scholar] [CrossRef]
- Jenkinson, C.; Fitzpatrick, R.; Swash, M.; Jones, G. Comparison of the 40-item Amyotrophic Lateral Sclerosis Assessment Questionnaire (ALSAQ-40) with a short-form five-item version (ALSAQ-5) in a longitudinal survey. Clin. Rehabil. 2007, 21, 266–272. [Google Scholar] [CrossRef] [PubMed]
- German Network for Motor Neuron Diseases (MND-NET). MND Net, German Network for Motor Neuron Diseases. 2021. Available online: http://www.mnd-als.de/html/home?set-language-to=en (accessed on 11 March 2021).
- Ludolph, A.C.; Drory, V.; Hardiman, O.; Nakano, I.; Ravits, J.; Robberecht, W.; Shefner, J.M.; for The WFN Research Group On ALS/MND. A revision of the El Escorial criteria 2015. Amyotroph. Lateral Scler. Front. Degener. 2015, 16, 291–292. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef]
- Klug, C.; Schreiber-Katz, O.; Thiele, S.; Schorling, E.; Zowe, J.; Reilich, P.; Walter, M.C.; Nagels, K.H. Disease burden of spinal muscular atrophy in Germany. Orphanet. J. Rare Dis. 2016, 11, 1–9. [Google Scholar] [CrossRef]
- Miano, B.; Stoddard, G.J.; Davis, S.; Bromberg, M.B. Inter-evaluator reliability of the ALS Functional Rating Scale. Amyotroph. Lateral Scler. 2004, 5, 235–239. [Google Scholar] [CrossRef]
- Abdulla, S.; Vielhaber, S.; Körner, S.; Machts, J.; Heinze, H.-J.; Dengler, R.; Petri, S. Validation of the German version of the extended ALS functional rating scale as a patient-reported outcome measure. J. Neurol. 2013, 260, 2242–2255. [Google Scholar] [CrossRef]
- Cedarbaum, J.M.; Stambler, N.; Malta, E.; Fuller, C.; Hilt, D.; Thurmond, B.; Nakanishi, A. The ALSFRS-R: A revised ALS functional rating scale that incorporates assessments of respiratory function. J. Neurol. Sci. 1999, 169, 13–21. [Google Scholar] [CrossRef]
- Leigh, P.N.; Swash, M.; Iwasaki, Y.; Ludolph, A.; Meininger, V.; Miller, R.G.; Mitsumoto, H.; Shaw, P.; Tashiro, K.; Berg, L.V.D. Amyotrophic lateral sclerosis: A consensus viewpoint on designing and implementing a clinical trial. Amyotroph. Lateral Scler. 2004, 5, 84–98. [Google Scholar] [CrossRef] [PubMed]
- Balendra, R.; Jones, A.; Jivraj, N.; Knights, C.; Ellis, C.M.; Burman, R.; Turner, M.R.; Leigh, P.N.; Shaw, C.E.; Al-Chalabi, A. Estimating clinical stage of amyotrophic lateral sclerosis from the ALS Functional Rating Scale. Amyotroph. Lateral Scler. Front. Degener. 2014, 15, 279–284. [Google Scholar] [CrossRef]
- Corcia, P.; Beltran, S.; Lautrette, G.; Bakkouche, S.; Couratier, P. Staging amyotrophic lateral sclerosis: A new focus on progression. Rev. Neurol. 2019, 175, 277–282. [Google Scholar] [CrossRef]
- Al-Chalabi, A.; Hardiman, O.; Kiernan, M.C.; Chiò, A.; Rix-Brooks, B.; Berg, L.H.V.D. Amyotrophic lateral sclerosis: Moving towards a new classification system. Lancet Neurol. 2016, 15, 1182–1194. [Google Scholar] [CrossRef]
- Balendra, R.; Jones, A.; Jivraj, N.; Steen, I.N.; Young, C.A.; Shaw, P.J.; Turner, M.R.; Leigh, P.N.; Al-Chalabi, A. Use of clinical staging in amyotrophic lateral sclerosis for phase 3 clinical trials. J. Neurol. Neurosurg. Psychiatry 2014, 86, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Van Hout, B.; Janssen, M.; Feng, Y.-S.; Kohlmann, T.; Busschbach, J.; Golicki, D.; Lloyd, A.; Scalone, L.; Kind, P.; Pickard, A.S. Interim Scoring for the EQ-5D-5L: Mapping the EQ-5D-5L to EQ-5D-3L Value Sets. Value Health 2012, 15, 708–715. [Google Scholar] [CrossRef]
- EuroQol Research Foundation. EQ-5D-5L User Guide. 2019. Available online: https://euroqol.org/publications/user-guides (accessed on 11 March 2021).
- Jenkinson, C.; Fitzpatrick, R.; Swash, M.; Levvy, G. ALSAQ User Manual: Amyotrophic Lateral Sclerosis Assessment Questionnaire; Health Services Research Unit, Oxford University: Oxford, UK, 2001. [Google Scholar]
- Grochtdreis, T.; Dams, J.; König, H.-H.; Konnopka, A. Health-related quality of life measured with the EQ-5D-5L: Estimation of normative index values based on a representative German population sample and value set. Eur. J. Health Econ. 2019, 20, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Maessen, M.; Post, M.W.; Maillé, R.; Lindeman, E.; Mooij, R.; Veldink, J.H.; Berg, L.H.V.D. Validity of the Dutch version of the Amyotrophic Lateral Sclerosis Assessment Questionnaire, ALSAQ-40, ALSAQ-5. Amyotroph. Lateral Scler. 2007, 8, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, A.; Soraru, G.; Lombardi, L.; D’Ascenzo, C.; Baggio, L.; Ermani, M.; Pegoraro, E.; Angelini, C. Quality of life and motor impairment in ALS: Italian validation of ALSAQ. Neurol. Res. 2010, 32, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Sawilowsky, S.S.; Blair, R.C. A more realistic look at the robustness and Type II error properties of the t test to departures from population normality. Psychol. Bull. 1992, 111, 352–360. [Google Scholar] [CrossRef]
- Schmidt, A.F.; Finan, C. Linear regression and the normality assumption. J. Clin. Epidemiol. 2018, 98, 146–151. [Google Scholar] [CrossRef]
- Tavakol, M.; Dennick, R. Making sense of Cronbach’s alpha. Int. J. Med. Educ. 2011, 2, 53–55. [Google Scholar] [CrossRef] [PubMed]
- Wijesekera, L.L.; Leigh, P.N. Amyotrophic lateral sclerosis. Orphanet J. Rare Dis. 2009, 4, 3–22. [Google Scholar] [CrossRef]
- Bundesgesundheitsministerium. Pflegegrade. 2018. Available online: https://www.bundesgesundheitsministerium.de/pflegegrade.html (accessed on 11 March 2021).
- Medrano, C.R.V.; Aho-Özhan, H.E.A.; Weiland, U.; Uttner, I.; Ludolph, A.C.; Lulé, D. Disease progression but not physical state per se determines mental wellbeing in ALS. J. Neurol. 2020, 267, 3593–3601. [Google Scholar] [CrossRef] [PubMed]
- Kaub-Wittemer, D.; Von Steinbüchel, N.; Wasner, M.; Laier-Groeneveld, G.; Borasio, G.D. Quality of life and psychosocial issues in ventilated patients with amyotrophic lateral sclerosis and their caregivers. J. Pain Symptom Manag. 2003, 26, 890–896. [Google Scholar] [CrossRef]
- Ang, K.; Lim, M.Y.; Srinivasan, S. Ethical and legal issues of tracheostomy ventilation in patients with amyotrophic lateral sclerosis. Proc. Singap. Healthc. 2019, 28, 193–202. [Google Scholar] [CrossRef]
- Vianello, A.; Arcaro, G.; Palmieri, A.; Ermani, M.; Braccioni, F.; Gallan, F.; Soraru’, G.; Pegoraro, E. Survival and quality of life after tracheostomy for acute respiratory failure in patients with amyotrophic lateral sclerosis. J. Crit. Care 2011, 26, 329.e7–329.e14. [Google Scholar] [CrossRef] [PubMed]
- Barras, A.-C.H.; Adler, D.; Ferfoglia, R.I.; Ricou, B.; Gasche, Y.; Leuchter, I.; Hurst, S.; Escher, M.; Pollak, P.; Janssens, J.-P. Is tracheostomy still an option in amyotrophic lateral sclerosis? Reflections of a multidisciplinary work group. Swiss Med. Wkly. 2013, 143, w13830. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Routledge: New York, NY, USA, 1988. [Google Scholar]
- Gotqb-Janowska, M.; Honczarenko, K.; Stankiewicz, J. Usefulness of the ALSAQ-5 scale in evaluation of quality of life in amyotrophic lateral sclerosis. Neurol. Neurochir. Pol. 2010, 44, 560–566. [Google Scholar] [CrossRef]
- Rabkin, J.; Ogino, M.; Goetz, R.; McElhiney, M.; Marziliano, A.; Imai, T.; Atsuta, N.; Morita, M.; Tateishi, T.; Matsumura, T.; et al. Tracheostomy with invasive ventilation for ALS patients: Neurologists’ roles in the US and Japan. Amyotroph. Lateral Scler. Front. Degener. 2012, 14, 116–123. [Google Scholar] [CrossRef]
- Radunovic, A.; Annane, D.; Rafiq, M.K.; Brassington, R.; Mustfa, N. Mechanical ventilation for amyotrophic lateral sclerosis/motor neuron disease. Cochrane Database Syst. Rev. 2017, 2017, CD004427. [Google Scholar] [CrossRef] [PubMed]
- Piepers, S.; Berg, J.V.D.; Kalmijn, S.; Van Der Pol, W.; Wokke, J.H.J.; Lindeman, E.; Berg, L.H.V.D. Effect of non-invasive ventilation on survival, quality of life, respiratory function and cognition: A review of the literature. Amyotroph. Lateral Scler. 2006, 7, 195–200. [Google Scholar] [CrossRef]
- Bourke, S.C.; Bullock, R.E.; Williams, T.L.; Shaw, P.J.; Gibson, G.J. Noninvasive ventilation in ALS: Indications and effect on quality of life. Neurology 2003, 61, 171–177. [Google Scholar] [CrossRef]
- Calzada, N.G.; Soro, E.P.; Gomez, L.M.; Bulta, E.G.; Izquierdo, A.C.; Panades, M.P.; Sargatal, J.D.; Muñoz, E.F. Factors predicting survival in amyotrophic lateral sclerosis patients on non-invasive ventilation. Amyotroph. Lateral Scler. Front. Degener. 2016, 17, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Sancho, J.; Servera, E.; Morelot-Panzini, C.; Salachas, F.; Similowski, T.; Gonzalez-Bermejo, J. Non-invasive ventilation effectiveness and the effect of ventilatory mode on survival in ALS patients. Amyotroph. Lateral Scler. Front. Degener. 2013, 15, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Spittel, S.; Maier, A.; Kettemann, D.; Walter, B.; Koch, B.; Krause, K.; Norden, J.; Münch, C.; Meyer, T. Non-invasive and tracheostomy invasive ventilation in amyotrophic lateral sclerosis: Utilization and survival rates in a cohort study over 12 years in Germany. Eur. J. Neurol. 2021, 28, 1160–1171. [Google Scholar] [CrossRef] [PubMed]
- Lulé, D.; Häcker, S.; Ludolph, A.; Birbaumer, N.; Kübler, A. Depression and Quality of Life in Patients with Amyotrophic Lateral Sclerosis. Dtsch. Aerzteblatt Online 2008, 105, 397–403. [Google Scholar] [CrossRef]
- Jenkinson, C.; Levvy, G.; Fitzpatrick, R.; Garratt, A. The amyotrophic lateral sclerosis assessment questionnaire (ALSAQ-40): Tests of data quality, score reliability and response rate in a survey of patients. J. Neurol. Sci. 2000, 180, 94–100. [Google Scholar] [CrossRef]
- Abe, K.; Itoyama, Y.; Sobue, G.; Tsuji, S.; Aoki, M.; Doyu, M.; Hamada, C.; Kondo, K.; Yoneoka, T.; Akimoto, M.; et al. Confirmatory double-blind, parallel-group, placebo-controlled study of efficacy and safety of edaravone (MCI-186) in amyotrophic lateral sclerosis patients. Amyotroph. Lateral Scler. Front. Degener. 2014, 15, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Pagnini, F. Psychological wellbeing and quality of life in amyotrophic lateral sclerosis: A review. Int. J. Psychol. 2013, 48, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Pizzimenti, A. Depression, pain and quality of life in patients with amyotrophic lateral sclerosis: A cross-sectional study. Funct. Neurol. 2013, 28, 115–119. [Google Scholar] [CrossRef]
- Simmons, Z. Patient-Perceived Outcomes and Quality of Life in ALS. Neurotherapeutics 2014, 12, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Körner, S.; Kollewe, K.; Abdulla, S.; Zapf, A.; Dengler, R.; Petri, S. Interaction of physical function, quality of life and depression in Amyotrophic lateral sclerosis: Characterization of a large patient cohort. BMC Neurol. 2015, 15, 84. [Google Scholar] [CrossRef]
- Meyer, T.; Kettemann, D.; Maier, A.; Grehl, T.; Weyen, U.; Grosskreutz, J.; Steinbach, R.; Norden, J.; George, A.; Hermann, A.; et al. Symptomatic pharmacotherapy in ALS: Data analysis from a platform-based medication management programme. J. Neurol. Neurosurg. Psychiatry 2020, 91, 783–785. [Google Scholar] [CrossRef] [PubMed]
- Roos, E.; Mariosa, D.; Ingre, C.; Lundholm, C.; Wirdefeldt, K.; Roos, P.M.; Fang, F. Depression in amyotrophic lateral sclerosis. Neurology 2016, 86, 2271–2277. [Google Scholar] [CrossRef]
- Thakore, N.J.; Pioro, E.P. Laughter, crying and sadness in ALS. J. Neurol. Neurosurg. Psychiatry 2017, 88, 825–831. [Google Scholar] [CrossRef]
- Statista. Population in Germany as of December 2019, by Federal State 2019. Available online: https://www.statista.com/statistics/1127686/population-by-federal-state-germany/ (accessed on 11 March 2021).

| Parameter | n (Percentage) | Mean (SD) | Median (Range) |
|---|---|---|---|
| Age | 325 | 63.94 (11.42) | 64 (27–88) |
| Gender | 325 | ||
| male | 203 (62.5%) | ||
| female | 122 (37.5%) | ||
| BMI (kg/m2) | 323 | 24.15 (4.40) | 24.21 (10.57–39.18) |
| Marital status | 324 | ||
| single | 40 (12.3%) | ||
| with partner | 284 (87.7%) | ||
| Currently working | 306 | ||
| yes | 51 (16.7%) | ||
| no | 255 (83.3%) | ||
| Genetics | 312 | ||
| familial ALS | 14 (4.5%) | ||
| sporadic ALS | 298 (95.5%) | ||
| First symptoms | 2871 | ||
| bulbar (bALS) | 85 (29.6%) | ||
| limb (lALS) 2 | 202 (70.4%) | ||
| ALSFRS-R total score (max./best = 48) | 325 | 30.92 (10.29) | 33 (1–48) |
| King’s stages | 325 | ||
| stage 1 | 57 (17.5%) | ||
| stage 2 | 70 (21.5%) | ||
| stage 3 | 83 (25.5%) | ||
| stage 4a | 24 (7.4%) | ||
| stage 4b | 91 (28.0%) | ||
| Ambulatory | 324 | ||
| Yes 3 | 239 (73.8%) | ||
| no | 85 (26.2%) | ||
| Care level4 | 268 | ||
| none | 40 (14.9%) | ||
| level 1 | 7 (2.6%) | ||
| level 2 | 45 (16.8%) | ||
| level 3 | 79 (29.5%) | ||
| level 4 | 56 (20.9%) | ||
| level 5 | 41 (15.3%) | ||
| 24 h carer | 303 | ||
| yes | 122 (40.3%) | ||
| no | 181 (59.7%) | ||
| ALSAQ-5 total score (max. = 100, best = 0) | 288 | 56.93 (19.46) | 56 (20–100) |
| Q1: physical mobility (max. = 100, best = 0) | 294 | 60.41 (28.19) | 60 (20–100) |
| Q2: activities of daily living and independence (max. = 100, best = 0) | 296 | 63.85 (27.61) | 80 (20–100) |
| Q3: eating and drinking (max. = 100, best = 0) | 299 | 44.82 (29.19) | 40 (20–100) |
| Q4: communication (max. = 100, best = 0) | 300 | 55.60 (31.50) | 60 (20–100) |
| Q5: emotional functioning (max. = 100, best = 0) | 298 | 59.80 (25.24) | 60 (20–100) |
| EQ-5D-5L index value (max./best = 1) | 304 | 0.48 (0.34) | 0.52 (−0.205–1) |
| Q1: mobility (max. = 100, best = 0) 5 | 307 | 67.30 (29.26) | 80 (20–100) |
| Q2: self-care (max. = 100, best = 0) | 308 | 64.35 (30.08) | 60 (20–100) |
| Q3: usual activities (max. = 100, best = 0) | 307 | 71.01 (26.02) | 80 (20–100) |
| Q4: pain/discomfort (max. = 100, best = 0) | 306 | 46.93 (22.24) | 40 (20–100) |
| Q5: anxiety/depression (max. = 100, best = 0) | 307 | 45.02 (21.80) | 40 (20–100) |
| EQ VAS total score (max./best = 100) | 304 | 42.58 (24.36) | 40.00 (0–95) |
| Parameter | Beta Coefficient (β) | Std. Error | t | p-Value | 95% Confidence Interval | |
|---|---|---|---|---|---|---|
| Lower Margin | Upper Margin | |||||
| ALSFRS-R total score | −1.96 | 0.157 | −12.461 | <0.001 | −2.271 | −1.649 |
| Mobility aid = yes | −7.60 | 2.219 | −3.424 | 0.001 | −11.985 | −3.211 |
| Tracheostomy = yes | −14.80 | 5.046 | −2.932 | 0.004 | −24.770 | −4.820 |
| Antidepressants = yes | 5.95 | 2.162 | 2.754 | 0.007 | 1.679 | 10.227 |
| Age | 0.18 | 0.080 | 2.294 | 0.023 | 0.025 | 0.343 |
| Non-invasive ventilation = yes | −5.71 | 2.614 | −2.186 | 0.030 | −10.880 | −0.546 |
| House helper = yes | −3.37 | 1.841 | −1.831 | 0.069 | −7.012 | 0.268 |
| Rehabilitation = yes | 3.51 | 2.008 | 1.747 | 0.083 | −0.460 | 7.477 |
| Opioid analgesic drugs = yes | 6.87 | 4.002 | 1.716 | 0.088 | −1.042 | 14.782 |
| Ergotherapy = yes | −3.08 | 1.859 | −1.659 | 0.099 | −6.758 | 0.591 |
| Currently working = yes | 3.89 | 2.635 | 1.475 | 0.142 | −1.322 | 9.095 |
| Ambulatory = yes | 3.99 | 2.809 | 1.420 | 0.158 | −1.563 | 9.542 |
| Non-opioid analgesic drugs = yes | −4.12 | 2.993 | −1.375 | 0.171 | −10.033 | 1.801 |
| Parameter | lALS | bALS | lALS vs. bALS |
|---|---|---|---|
| ALSAQ-5 total score (max. 100, best = 0) | |||
| n | 179 | 77 | |
| mean (SD) | 55.46 (19.76) | 60.99 (19.23) | t = −2.07; df = 254; p = 0.040; r = 0.13 |
| median (range) | 56 (20–100) | 60 (20–100) | |
| Q1: Physical mobility (max. 100, best = 0) | |||
| n | 183 | 78 | |
| mean (SD) | 63.39 (27.41) | 50.26 (28.96) | t = 3.48; df = 259; p = 0.001; r = 0.21 |
| median (range) | 60 (20–100) | 40 (20–100) | |
| Q2: Activities of daily living and independence (max. 100, best = 0) | |||
| n | 186 | 77 | |
| mean (SD) | 69.78 (25.40) | 48.05 (27.20) | t = 6.18; df = 261; p < 0.001; r = 0.36 |
| median (range) | 80 (20–100) | 40 (20–100) | |
| Q3: Eating and drinking (max. 100, best = 0) | |||
| n | 186 | 80 | |
| mean (SD) | 39.57 (26.79) | 60.50 (31.50) | t = −5.19; df = 130.41; p < 0.001; r = 0.41 |
| median (range) | 20 (20–100) | 60 (20–100) | |
| Q4: Communication (max. 100, best = 0) | |||
| n | 187 | 80 | |
| mean (SD) | 45.45 (28.25) | 84.75 (19.16) | t = −13.20; df = 215.23; p < 0.001; r = 0.67 |
| median (range) | 40 (20–100) | 100 (20–100) | |
| Q5: Emotional functioning (max. 100, best = 0) | |||
| n | 186 | 80 | |
| mean (SD) | 57.96 (25.43) | 62 (25.77) | t = −1.18; df = 264; p = 0.237; r = 0.07 |
| median (range) | 60 (20–100) | 60 (20–100) | |
| EQ-5D-5L index value (max./best = 1) | |||
| n | 191 | 79 | |
| mean (SD) | 0.42 (0.32) | 0.66 (0.33) | t = −5.43; df = 268; p < 0.001; r = 0.31 |
| median (range) | 0.43 (−0.21–1) | 0.81 (−0.14–1) | |
| Q1: Mobility (max. 100, best = 0) | |||
| n | 192 | 81 | |
| mean (SD) | 72.92 (26.60) | 52.35 (30.10) | t = 5.34; df = 135.28; p < 0.001; r = 0.42 |
| median (range) | 80 (20–100) | 40 (20–100) | |
| Q2: Self-care (max. 100, best = 0) | |||
| n | 191 | 82 | |
| mean (SD) | 71.20 (27.42) | 47.07 (30.69) | t = 6.43; df = 271; p < 0.001; r = 0.36 |
| median (range) | 80 (20–100) | 40 (20–100) | |
| Q3: Usual activities (max. 100, best = 0) | |||
| n | 192 | 81 | |
| mean (SD) | 76.04 (23.36) | 57.04 (28.30) | t = 5.33; df = 128.14; p < 0.001; r = 0.43 |
| median (range) | 80 (20–100) | 60 (20–100) | |
| Q4: Pain/Discomfort (max. 100, best = 0) | |||
| n | 192 | 80 | |
| mean (SD) | 49.27 (22.78) | 40.50 (19.87) | t = 3.17; df = 168.35; p = 0.002; r = 0.24 |
| median (range) | 40 (20–100) | 40 (20–100) | |
| Q5: Anxiety/Depression (max. 100, best = 0) | |||
| n | 192 | 81 | |
| mean (SD) | 46.25 (22.29) | 40.74 (21.32) | t = 1.89; df = 271; p = 0.060; r = 0.11 |
| median (range) | 40 (20–100) | 40 (20–100) | |
| EQ VAS total score (max./best 100) | |||
| n | 190 | 80 | |
| mean (SD) | 39.72 (24.18) | 50.99 (24.40) | t = −3.49; df = 268; p = 0.001; r = 0.21 |
| median (range) | 40 (0–95) | 50 (0–90) |
| Comparison | ρ (rho) | p-Value | n |
|---|---|---|---|
| AQ1 vs. EQ1 | 0.73 | <0.001 | 290 |
| AQ2 vs. EQ2 | 0.71 | <0.001 | 292 |
| AQ3 vs. EQ3 | 0.27 | <0.001 | 295 |
| AQ4 vs. EQ4 | −0.01 | 0.878 | 295 |
| AQ5 vs. EQ5 | 0.55 | <0.001 | 294 |
| ALSAQ-5 total score vs. reversed 1 EQ VAS score | 0.62 | <0.001 | 283 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peseschkian, T.; Cordts, I.; Günther, R.; Stolte, B.; Zeller, D.; Schröter, C.; Weyen, U.; Regensburger, M.; Wolf, J.; Schneider, I.; et al. A Nation-Wide, Multi-Center Study on the Quality of Life of ALS Patients in Germany. Brain Sci. 2021, 11, 372. https://doi.org/10.3390/brainsci11030372
Peseschkian T, Cordts I, Günther R, Stolte B, Zeller D, Schröter C, Weyen U, Regensburger M, Wolf J, Schneider I, et al. A Nation-Wide, Multi-Center Study on the Quality of Life of ALS Patients in Germany. Brain Sciences. 2021; 11(3):372. https://doi.org/10.3390/brainsci11030372
Chicago/Turabian StylePeseschkian, Tara, Isabell Cordts, René Günther, Benjamin Stolte, Daniel Zeller, Carsten Schröter, Ute Weyen, Martin Regensburger, Joachim Wolf, Ilka Schneider, and et al. 2021. "A Nation-Wide, Multi-Center Study on the Quality of Life of ALS Patients in Germany" Brain Sciences 11, no. 3: 372. https://doi.org/10.3390/brainsci11030372
APA StylePeseschkian, T., Cordts, I., Günther, R., Stolte, B., Zeller, D., Schröter, C., Weyen, U., Regensburger, M., Wolf, J., Schneider, I., Hermann, A., Metelmann, M., Kohl, Z., Linker, R. A., Koch, J. C., Büchner, B., Weiland, U., Schönfelder, E., Heinrich, F., ... Schreiber-Katz, O. (2021). A Nation-Wide, Multi-Center Study on the Quality of Life of ALS Patients in Germany. Brain Sciences, 11(3), 372. https://doi.org/10.3390/brainsci11030372










