Therapeutic Potential of Allicin-Rich Garlic Preparations: Emphasis on Clinical Evidence toward Upcoming Drugs Formulation
Abstract
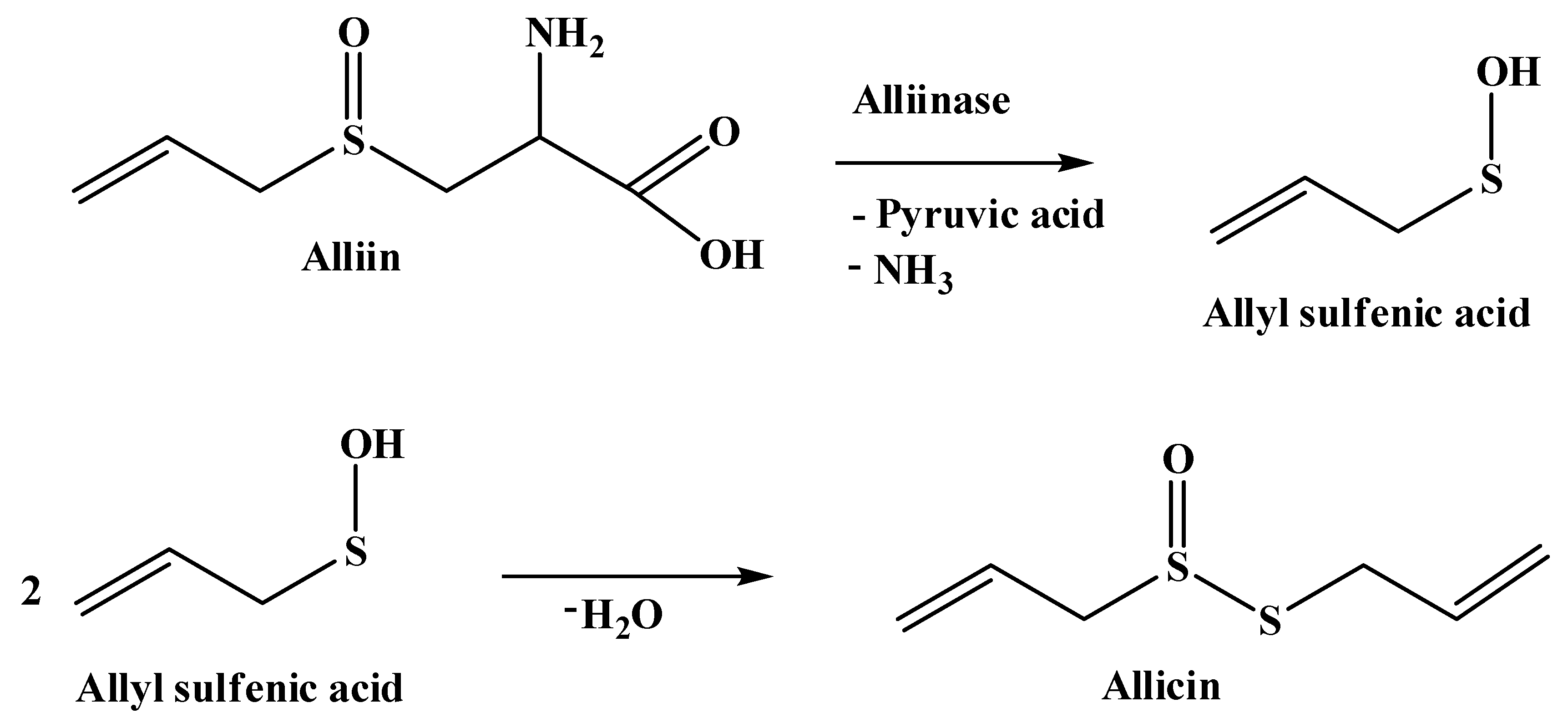
1. Introduction
2. Drifting from Molecular to Clinical Evidence
2.1. Cardioprotective Activity
2.1.1. Atherosclerosis
2.1.2. Hyperlipidemia and Hypertension
2.1.3. Bodyweight Management
2.2. Anti-Carcinogenic Activity
2.3. Antidiabetic Activity
2.4. Cytochrome Activity
2.5. Oxidative Stress Status
2.6. Preeclampsia
2.7. Antimicrobial Effects
2.8. Ulcer Healing
2.9. Others
3. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Fabricant, D.S.; Farnsworth, N.R. The value of plants used in traditional medicine for drug discovery. Environ. Health Perspect. 2001, 109, 69–75. [Google Scholar] [PubMed]
- Hechtman, L. Clinical Naturopathic Medicine; Elsevier Health Sciences: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Sharifi-Rad, M.; Fokou, P.; Sharopov, F.; Martorell, M.; Ademiluyi, A.; Rajkovic, J.; Salehi, B.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Antiulcer agents: From plant extracts to phytochemicals in healing promotion. Molecules 2018, 23, 1751. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.P.; Saklani, S.; Salehi, B.; Parcha, V.; Sharifi-Rad, M.; Milella, L.; Iriti, M.; Sharifi-Rad, J.; Srivastava, M. Satyrium nepalense, a high altitude medicinal orchid of Indian Himalayan region: Chemical profile and biological activities of tuber extracts. Cell. Mol. Biol. 2018, 64, 35. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; Sharifi-Rad, M.; Salehi, B.; Iriti, M.; Roointan, A.; Mnayer, D.; Soltani-Nejad, A.; Afshari, A. In vitro and in vivo assessment of free radical scavenging and antioxidant activities of Veronica persica Poir. Cell. Mol. Biol. 2018, 64, 57. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, M.; Roberts, T.H.; Matthews, K.R.; Bezerra, C.F.; Morais-Braga, M.F.B.; Coutinho, H.D.M.; Sharopov, F.; Salehi, B.; Yousaf, Z.; Sharifi-Rad, M.; et al. Ethnobotany of the genus Taraxacum—Phytochemicals and antimicrobial activity. Phytotherapy Res. 2018, 32, 2131–2145. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Fokou, P.V.T.; Sharifi-Rad, M.; Zucca, P.; Pezzani, R.; Martins, N.; Sharifi-Rad, J. The therapeutic potential of naringenin: A review of clinical trials. Pharmaceuticals 2019, 12, 11. [Google Scholar] [CrossRef]
- Salehi, B.; Venditti, A.; Sharifi-Rad, M.; Kręgiel, D.; Sharifi-Rad, J.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.B.; Novellino, E.; et al. The therapeutic potential of Apigenin. Int. J. Mol. Sci. 2019, 20, 1305. [Google Scholar] [CrossRef]
- Mattioli, L.; Funari, C.; Perfumi, M. Effects of Rhodiola rosea L. extract on behavioural and physiological alterations induced by chronic mild stress in female rats. J. Psychopharmacol. 2009, 23, 130–142. [Google Scholar] [CrossRef]
- Chan, J.Y.Y.; Yuen, A.C.; Chan, R.Y.; Chan, S. A review of the cardiovascular benefits and antioxidant properties of allicin. Phytother. Res. 2013, 27, 637–646. [Google Scholar] [CrossRef]
- Khalighi-Sigaroodi, F.; Jeddi-Tehrani, M.; Ahvazi, M.; Shahnazi, S.; Bayat, A.A.; Mohajer, N.; Zarei, S. Cytotoxicity evaluation of Taverniera spartea on human cancer cell lines. J. Med. Plants 2014, 2, 114–128. [Google Scholar]
- Forouzandeh, F.; Salimi, S.; Naghsh, N.; Zamani, N.; Jahani, S. Evaluation of anti-cancer effect of Peganum harmala L hydroalcholic extract on human cervical carcinoma epithelial cell line. J. Shahrekord Univ. Med. Sci. 2014, 16, 1–8. [Google Scholar]
- Salehi, B.; Sharopov, F.; Martorell, M.; Rajkovic, J.; Ademiluyi, A.O.; Sharifi-Rad, M.; Fokou, P.V.T.; Martins, N.; Iriti, M.; Sharifi-Rad, J. Phytochemicals in Helicobacter pylori infections: What are we doing now? Int. J. Mol. Sci. 2018, 19, 2361. [Google Scholar] [CrossRef] [PubMed]
- Marasini, B.P.; Baral, P.; Aryal, P.; Ghimire, K.R.; Neupane, S.; Dahal, N.; Singh, A.; Ghimire, L.; Shrestha, K. Evaluation of antibacterial activity of some traditionally used medicinal plants against human pathogenic bacteria. BioMed Res. Int. 2015. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.H.; Kang, D. Physicochemical properties, biological activity, health benefits, and general limitations of aged black garlic: a review. Molecules 2017, 22, 919. [Google Scholar] [CrossRef]
- Peng, Y.; Hu, K. Effect of garlic on rats with chronic intermittent hypoxia combined with diabetes mellitus. Mol. Med. Rep. 2018, 17, 6174–6184. [Google Scholar] [CrossRef]
- Mikaili, P.; Maadirad, S.; Moloudizargari, M.; Aghajanshakeri, S.; Sarahroodi, S. Therapeutic uses and pharmacological properties of garlic, shallot, and their biologically active compounds. Iran. J. Basic Med. Sci. 2013, 16, 1031–1048. [Google Scholar]
- Okada, Y.; Tanaka, K.; Fujita, I.; Sato, E.; Okajima, H. Antioxidant activity of thiosulfinates derived from garlic. Redox Rep. 2005, 10, 96–102. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, Y.; Li, F.; Zhu, Y.; Chen, Y.; Yang, S.; Sun, G. Allicin as a possible adjunctive therapeutic drug for stage II oral submucous fibrosis: A preliminary clinical trial in a Chinese cohort. Int. J. Oral Maxillofac. Surg. 2015, 44, 1540–1546. [Google Scholar] [CrossRef]
- D’Argenio, G.; Mazzone, G.; Ribecco, M.T.; Lembo, V.; Vitaglione, P.; Guarino, M. Garlic extract attenuating rat liver fibrosis by inhibiting TGF-β1. Clin. Nutr. 2013, 32, 252–258. [Google Scholar] [CrossRef]
- Jiang, X.W.; Zhang, Y.; Song, G.D.; Li, F.F.; Peng, H.Y.; Yang, S.K.; Sun, G.L. Clinical evaluation of allicin oral adhesive tablets in the treatment of recurrent aphthous ulceration. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 113, 500–504. [Google Scholar] [CrossRef]
- Gardner, C.D.; Lawson, L.D.; Block, E.; Chatterjee, L.M.; Kiazand, A.; Balise, R.R.; Kraemer, H.C. Effect of raw garlic vs. commercial garlic supplements on plasma lipid concentrations in adults with moderate hypercholesterolemia. Arch. Intern. Med. 2007, 167, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Koscielny, J.; Klussendorf, D.; Latza, R.; Schmitt, R.; Radtke, H.; Siegel, G.; Kiesewetter, H. The antiatherosclerotic effect of Allium sativum. Atherosclerosis 1999, 144, 237–249. [Google Scholar] [CrossRef]
- Budoff, M.J.; Ahmadi, N.; Gul, K.M.; Liu, S.T.; Flores, F.R.; Tiano, J.; Takasu, J.; Miller, E.; Tsimikas, S. Aged garlic extract supplemented with B vitamins, folic acid and L-arginine retards the progression of subclinical atherosclerosis: A randomized clinical trial. Prev. Med. 2009, 49, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Orekhov, A.N.; Sobenin, I.A.; Korneev, N.V.; Kirichenko, T.V.; Myasoedova, V.A.; Melnichenko, A.A.; Balcells, M.; Edelman, E.R.; Bobryshev, Y.V. Anti-atherosclerotic therapy based on botanicals. Recent Pat. Cardiovasc. Drug Discov. 2013, 8, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Larijani, V.N.; Ahmadi, N.; Zeb, I.; Khan, F.; Flores, F.; Budoff, M. Beneficial effects of aged garlic extract and coenzyme Q10 on vascular elasticity and endothelial function: The FAITH randomized clinical trial. Nutrition 2013, 29, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Ried, K.; Frank, O.R.; Stocks, N.P. Aged garlic extract lowers blood pressure in patients with treated but uncontrolled hypertension: A randomised controlled trial. Maturitas 2010, 67, 144–150. [Google Scholar] [CrossRef]
- Sobenin, I.A.; Andrianova, I.V.; Fomchenkov, I.V.; Gorchakova, T.V.; Orekhov, A.N. Time-released garlic powder tablets lower systolic and diastolic blood pressure in men with mild and moderate arterial hypertension. Hypertens Res. 2009, 32, 433–437. [Google Scholar] [CrossRef]
- Kiesewetter, H.; Jung, F.; Jung, E.M.; Blume, J.; Mrowietz, C.; Birk, A.; Koscielny, J.; Wenzel, E. Effects of garlic coated tablets in peripheral arterial occlusive disease. Clin. Investig. 1993, 71, 383–386. [Google Scholar] [CrossRef]
- Jain, A.K.; Vargas, R.; Gotzkowsky, S.; McMahon, F.G. Can garlic reduce levels of serum lipids? A controlled clinical study. Am. J. Med. 1993, 94, 632–635. [Google Scholar] [CrossRef]
- Simons, L.A.; Balasubramaniam, S.; Konigsmark, M.; Parfitt, A.; Simons, J.; Peters, W. On the effect of garlic on plasma lipids and lipoproteins in mild hypercholesterolaemia. Atherosclerosis 1995, 113, 219–225. [Google Scholar] [CrossRef]
- Sahebkar, A.; Serban, C.; Ursoniu, S.; Banach, M. Effect of garlic on plasma lipoprotein(a) concentrations: A systematic review and meta-analysis of randomized controlled clinical trials. Nutrition 2016, 32, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Vorberg, G.; Schneider, B. Therapy with garlic: Results of a placebocontrolled, double blind study. J. Clin. Pract. 1990, 44, 7–11. [Google Scholar]
- Superko, H.R.; Krauss, R.M. Garlic powder, effect on plasma lipids, postprandial lipemia, low-density lipoprotein particle size, high-density lipoprotein subclass distribution and lipoprotein(a). J. Am. Coll. Cardiol. 2000, 35, 321–326. [Google Scholar] [CrossRef]
- McCrindle, B.; Helden, E.; Conne, W. Garlic extract therapy in children with hypercholesterolemia. Arch. Pediatrics Adolesc. Med. 1998, 152, 1089–1094. [Google Scholar] [CrossRef]
- Isaacsohn, J.L.; Moser, M.; Stein, E.A.; Dudley, K.; Davey, J.A.; Liskov, E.; Black, H.R. Garlic powder and plasma lipids and lipoproteins: A multicenter, randomized, placebo-controlled trial. Arch. Intern. Med. 1998, 158, 1189–1194. [Google Scholar] [CrossRef]
- De Santos, A.O.S.; Grunwald, J. Effect of garlic powder tablets on blood lipids and blood pressure—A six month placebo controlled, double blind study. Br. J. Clin. Res. 1993, 4, 37–44. [Google Scholar]
- Saradeth, T.; Seidl, S.; Resch, K.; Ernst, E. Does garlic alter the lipid pattern in normal volunteers? Phytomedicine 1994, 1, 183–185. [Google Scholar] [CrossRef]
- Lachhiramka, P.; Patil, S. Cholesterol lowering property of garlic (Allium sativum) on patients with hypercholesterolemia. Int. J. Med. Sci. Public Health 2016, 5, 2249–2251. [Google Scholar] [CrossRef]
- Gardner, C.D.; Chatterjee, L.M.; Carlson, J.J. The effect of a garlic preparation on plasma lipid levels in moderately hypercholesterolemic adults. Atherosclerosis 2001, 154, 213–220. [Google Scholar] [CrossRef]
- Byrne, D.J.; Neil, H.A.W.; Vallance, D.T.; Winder, A.F. A pilot study of garlic consumption shows no significant effect on markers of oxidation or sub-fraction composition of low-density lipoprotein including lipoprotein(a) after allowance for non-compliance and the placebo effect. Clin. Chim. Acta 1999, 285, 21–33. [Google Scholar] [CrossRef]
- Holzgartner, H.; Schmidt, U.; Kuhn, U. Comparison of the efficacy and tolerance of a garlic preparation vs. bezafibrate. Arzneim. Frosch. 1992, 42, 1473–1477. [Google Scholar]
- Harenberg, J.; Giese, C.; Zimmermann, R. Effect of dried garlic on blood coagulation, fibrinolysis, platelet aggregation and serum cholesterol levels in patients with hyperlipoproteinemia. Atherosclerosis 1988, 74, 247–249. [Google Scholar] [CrossRef]
- Soleimani, D.; Paknahad, Z.; Askari, G.; Iraj, B.; Feizi, A. Effect of garlic powder consumption on body composition in patients with nonalcoholic fatty liver disease: A randomized, double-blind, placebo-controlled trial. Adv. Biomed. Res. 2016, 5. [Google Scholar] [CrossRef]
- Turner, B.; Molgaard, C.; Marckmann, P. Effect of garlic (Allium sativum) powder tablets on serum lipids, blood pressure and arterial stiffness in normo-lipidaemic volunteers: A randomized, double-blind, placebo-controlled trial. Br. J. Nutr. 2004, 92, 701–706. [Google Scholar] [CrossRef] [PubMed]
- Lawson, L.D.; Wang, Z.J.; Papadimitriou, D. Allicin release under simulated gastrointestinal conditions from garlic powder tablets employed in clinical trials on serum cholesterol. Planta Med. 2001, 67, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Haruma, K.; Yoshihara, M.; Kajiyama, G.; Kira, K.; Amagase, H.; Chayama, K. Aged garlic extract has potential suppressive effect on colorectal adenomas in humans. J. Nutr. 2006, 136, 821S–826S. [Google Scholar] [CrossRef]
- You, W.C.; Brown, L.M.; Zhang, L.; Li, J.Y.; Jin, M.L.; Chang, Y.S.; Ma, J.L.; Pan, K.F.; Liu, W.D.; Hu, Y.; et al. Randomized double-blind factorial trial of three treatments to reduce the prevalence of precancerous gastric lesions. J. Natl. Cancer Inst. 2006, 98, 974–983. [Google Scholar] [CrossRef]
- Li, H.; Li, H.Q.; Wang, Y. An intervention study to prevent gastric cancer by micro-selenium and large dose of allitridum. Chin. Med. J. 2004, 117, 1155–1160. [Google Scholar]
- Ishikawa, H.; Saeki, T.; Otani, T.; Suzuki, T.; Shimozuma, K.; Nishino, H.; Fukuda, S.; Morimoto, K. Aged garlic extract prevents a decline of NK cell number and activity in patients with advanced cancer. J. Nutr. 2006, 136, 816S–820S. [Google Scholar] [CrossRef]
- Shabani, E.; Sayemiri, K.; Mohammadpour, M. The effect of garlic on lipid profile and glucoseparameters in diabetic patients: A systematicreview and meta-analysis. Prim. Care Diabetes 2019, 13, 28–42. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, X.; Lan, H.; Wang, W. Effect of garlic supplement in the management of type 2 diabetes mellitus (T2DM): A meta-analysis of randomized controlled trials. Food Nutr. Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Ebadi, S.A.; Lenji, E.R.; Taghadosi, M.; Khorshidi, A.; Akbari, H. Effect of garlic on blood sugar in patients with type 2 diabetes mellitus. FEYZ 2007, 11, 20–25. [Google Scholar]
- Ashraf, R.; Khan, R.A.; Ashraf, I. Effects of garlic on blood glucose levels and HbA1c in patients with type 2 diabetes mellitus. J. Med. Plants Res. 2011, 5, 2922–2928. [Google Scholar]
- Sobenin, I.A.; Nedosugova, L.V.; Filatova, L.V.; Balabolkin, M.I.; Gorchakova, T.V.; Orekhov, A.N. Metabolic effects of time-released garlic powder tablets in type 2 diabetes mellitus: The results of double-blinded placebo-controlled study. Acta Diabetol. 2008, 45, 1–6. [Google Scholar] [CrossRef]
- Sukandar, E.Y.; Permana, H.; Adnyana, I.K.; Sigit, J.I.; Ilyas, R.A.; Hasimun, P.; Mardiyah, D. Clinical study of turmeric (Curcuma longa L.) and garlic (Allium sativum L.) extracts as antihyperglycemic and antihyperlipidemic agent in type-2 diabetes-dyslipidemia patients. Int. J. Pharmacol. 2010, 6, 456–463. [Google Scholar] [CrossRef]
- Mansouri, A.; Vahed, A.S.; Shahdadi, H.; Dashtban, F.; Arbabisarjou, A. The effect of garlic and cumin on blood pressure and glycosylated hemoglobin in patients with type 2 diabetes. Bali Med. J. 2018, 7, 156–160. [Google Scholar] [CrossRef]
- Emami, S.; Rouhani, M.H.; Azadbakht, L. The effect of garlic intake on glycemic control in humans: A systematic review and meta-analysis. Prog. Nutr. 2017, 19, 10–18. [Google Scholar]
- Gurley, B.J.; Gardner, S.F.; Hubbard, M.A.; Williams, D.K.; Gentry, W.B.; Cui, Y.; Ang, C.Y. Cytochrome P450 phenotypic ratios for predicting herb-drug interactions in humans. Clin. Pharmcol. 2002, 72, 276–287. [Google Scholar] [CrossRef]
- Gallicano, K.; Foster, B.; Choudhri, S. Effect of short-term administration of garlic supplements on single-dose ritonavir pharmacokinetics in healthy volunteers. Br. J. Clin. Pharmcol. 2003, 55, 199–202. [Google Scholar] [CrossRef]
- Piscitelli, S.C.; Burstein, A.H.; Welden, N.; Gallicano, K.D.; Falloon, J. The effect of garlic supplements on the pharmacokinetics of saquinavir. Clin. Infect. Dis. 2002, 34, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Markowitz, J.S.; DeVane, C.L.; Chavin, K.D.; Taylor, R.M.; Ruan, Y.; Donovan, J.L. Effect of garlic (Allium sativum L.) supplementation on cytochrome P450 2D6 and 3A4 activity in healthy volunteers. Clin. Pharmcol. 2003, 74, 170–177. [Google Scholar] [CrossRef]
- Katja, B.; Albin, K. The Mechanisms Responsible for Garlic—Drug Interactions and their In Vivo Relevance. Curr. Drug Metab. 2013, 14, 90–101. [Google Scholar] [CrossRef]
- Haghi Aminjan, H.; Abtahi, S.R.; Hazrati, E.; Chamanara, M.; Jalili, M.; Paknejad, B. Targeting of oxidative stress and inflammation through ROS/NF-kappaB pathway in phosphine-induced hepatotoxicity mitigation. Life Sci. 2019, 232, 116607. [Google Scholar] [CrossRef]
- Phelps, S.; Harris, W.S. Garlic supplementation and lipoprotein oxidation susceptibility. Lipids 1993, 28, 475–477. [Google Scholar] [CrossRef]
- Grune, T.; Scherat, T.; Behrend, H.; Conradi, E.; Brenke, R.; Siems, W. Influence of Allium sativum on oxidative stress status a clinical investigation. Phytomedicine 1996, 2, 205–207. [Google Scholar] [CrossRef]
- Aalami-Harandi, R.; Karamali, M.; Asemi, Z. The favorable effects of garlic intake on metabolic profiles, hs-CRP, biomarkers of oxidative stress and pregnancy outcomes in pregnant women at risk for pre-eclampsia: Randomized, double-blind, placebo-controlled trial. J. Matern. Fetal Neonatal Med. 2015, 28, 2020–2027. [Google Scholar] [CrossRef]
- Ziaei, S.; Hantoshzadeh, S.; Rezasoltani, P.; Lamyian, M. The effect of garlic tablet on plasma lipids and platelet aggregation in nulliparous pregnants at high risk of preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001, 99, 201–206. [Google Scholar] [CrossRef]
- Marchese, A.; Barbieri, R.; Sanches-Silva, A.; Daglia, M.; Nabavi, S.F.; Jafari, N.J.; Izadi, M.; Ajami, M.; Nabavi, S.M. Antifungal and antibacterial activities of allicin: A review. Trends Food Sci. Technol. 2016, 52, 49–56. [Google Scholar] [CrossRef]
- Aydin, A.; Ersoez, G.; Tekesin, O.; Akçiçek, E.; Tuncyürek, M. Garlic oil and Helicobacter pylori infection. Am. J. Gastroenterol. 2000, 95, 563–564. [Google Scholar] [CrossRef]
- Koçkar, C.; Oztürk, M.; Bavbek, N. Helicobacter pylori eradication with beta carotene, ascorbic acid and allicin. Acta Med. 2001, 44, 97–100. [Google Scholar] [CrossRef]
- Zhang, X.; Yang, Y.; Gang, S.; Yang, C.; Lu, M.; Zhi, J. Berberine-, allicin- or clarithromycin- based triple therapy for the first-line treatment of Helicobacter pylori infection: An open-label, randomized trial. Gastroenterology 2014, 146, 398S. [Google Scholar] [CrossRef]
- Mousavi, Z.B.; Mehrabian, A.; Golfakhrabadi, F.; Namjoyan, F. A clinical study of efficacy of garlic extract versus cryotherapy in the treatment of male genital wart. Dermatol. Sin. 2018, 36, 196–199. [Google Scholar] [CrossRef]
- Josling, P. Preventing the common cold with a garlic supplement: A double-blind, placebo-controlled survey. Adv. Ther. 2001, 18, 189–193. [Google Scholar] [CrossRef]
- Zhang, R.S. A clinical study on allicin in the prevention of thrush in newborn infants. Chin. J. Integr. Tradit. West. Med. 1992, 12, 28–29. [Google Scholar]
- Martin, K.W.; Ernst, E. Herbal medicines for treatment of fungal infections: A systematic review of controlled clinical trials. Mycoses 2003, 47, 87–92. [Google Scholar] [CrossRef]
- Berge, A.C.B.; Lindeque, P.; Moore, D.A.; Sischo, W.M. A clinical trial evaluating prophylactic and therapeutic antibiotic use on health and performance of preweaned calves. J. Dairy Sci. 2005, 88, 321–326. [Google Scholar] [CrossRef]
- Donovan, D.C.; Franklin, S.T.; Chase, C.C.; Hippen, A.R. Growth and health of Holstein calves fed milk replacers supplemented with antibiotics or Enteroguard. J. Dairy Sci. 2002, 85, 947–950. [Google Scholar] [CrossRef]
| Clinical Trial | Participants | Duration | Dose | Summary | References |
|---|---|---|---|---|---|
| Atherosclerosis | |||||
| R, DB, PC | 152 individuals | 48 months | High-dose garlic powder | Plaque volume in the whole collective remained practically constant within the age-span of 50–80 years, substantiating not only a garlic preventive but possibly a curative role in arteriosclerosis (plaque regression) | [23] |
| R, DB, PC | 65 individuals | 12 months | Capsule containing aged garlic extract (250 mg) plus Vitamins B12 (100 μg), B9 (300 μg), B6 (12.5 mg) and L-arginine (100 mg) | Aged garlic extract supplemented with sulphur was associated with a favorable improvement in oxidative biomarkers, vascular function, and reduced atherosclerosis progression | [24] |
| R, DB, PC | 196 asymptomatic men | Garlic-based drug Allicor (INAT-Pharma, Moscow, Russia) (300 mg/day) | 2-year treatment with Allicor had a direct anti-atherosclerotic effect on carotid atherosclerosis in asymptomatic men | [25] | |
| R | 65 firefighters | Aged garlic extract (300 mg/tablet) plus CoQ10 (30 mg/tablet) | This combination was independently associated with significant beneficial effects on vascular elasticity and endothelial function in firefighters with high occupational stress, highlighting the important role of these substances in atherosclerotic prevention | [26] | |
| Hypertension | |||||
| R | 50 individuals | 12 weeks | Aged garlic extract (960 mg containing 2.4 mg S-Allyl cysteine) | Aged garlic extract effect was superior to placebo in lowering systolic blood pressure (similar to current first line medications) in patients with treated but uncontrolled hypertension | [27] |
| 84 men | 8 weeks | 900 mg Kwai (garlic pills) | It was effective in the treatment of mild and moderate arterial hypertension | [28] | |
| R, DB, PC | 80 individuals | 8 weeks | 800 mg garlic powder | Increased walking distance and decreased diastolic blood pressure, spontaneous thrombocyte aggregation, plasma viscosity, and cholesterol levels | [29] |
| DB | 42 healthy adults | 12 weeks | 900 mg standardized garlic powder | No significant changes in serum HDL-c, triglycerides, blood pressure and glucose levels. Garlic seemed to be mostly effective in reducing LDL-c levels | [30] |
| Hyperlipidemia | |||||
| R, DB, PC | 30 individuals | 12 weeks | Kwai@ garlic powder tablets 900 mg/daily | There was no demonstrable effect of garlic on LDL oxidation, plasma lathosterol-I-cholestero1 ratio, nor on LDL receptor expression in lymphocytes | [31] |
| Meta-analysis | - | - | No significant effect of garlic supplementation on Lp (a) levels reduction | [32] | |
| R, DB, PC | 40 individuals | 4 months | 900 mg garlic powder | Significantly lowered total cholesterol, triglycerides and blood pressure | [33] |
| R, DB, PC | 50 individuals | 12 weeks | 900 mg tablet garlic extract | Garlic had no effect on major plasma lipoproteins, and it has no impact on HDL-c subclasses, Lp (a), apolipoprotein B, postprandial triglycerides or LDL-c subclass distribution | [34] |
| R, DB, PC | 30 pediatric patients, aged 8–18 years | 8 weeks | 900 mg garlic extract | No significant effect on cardiovascular risk factors in pediatric patients with familial hyperlipidemia | [35] |
| R, DB, PC | 2 out-patient lipid clinics | 12 weeks | 900 mg garlic powder | Ineffective in lowering cholesterol levels in patients | [36] |
| R, DB, PC | 52 out-patients | 6 months | 900 mg garlic powder tablet (Kwai) (standardized to 1.3% alliin) | Mean values for LDL-C were reduced by nearly 10% by garlic and 6% by placebo | [37] |
| R, DB, PC | 68 normal volunteers | 15 weeks | 600 mg dried garlic powder (standardized on 1.3% allicin) | Total cholesterol dropped from 223 to 214 mg/dl, triglycerides decreased from 124 to 118 mg/dl, and blood pressure remained constant | [38] |
| R, DB, PC | 42 healthy adults | 900 mg standardized garlic powder | Greater reduction in serum total cholesterol LDL-c | [30] | |
| open-label study | 50 individuals | 90 days | 3 g of raw garlic daily | Garlic has a role in cholesterol management as adjunctive therapy in most cases of significant hypercholesterolemia, reducing the risk of atherosclerosis and cardiovascular events | [39] |
| R, DB, PC | 51 adults | 12 weeks | 500 or 1000 mg dehydrated garlic powder | No significant effect on plasma lipids levels and therefore, there was no indication of a graded effect by garlic dose over 0, 500 and 1000 mg/day | [40] |
| DB, R, PC | 31 individuals | 26 weeks | 900 mg Kwai garlic (standardized to 0.6% allicin, 1.3% alliin) | Antibody levels to oxidized LDL were unchanged, and lipoprotein(a) levels in plasma also were not changed | [41] |
| 98 individuals | 900 mg of garlic powder (standardized as to 1.3% alliin) | Significant reduction in total cholesterol, LDL-c, and triglycerides, and an increase in HDL-c | [42] | ||
| 20 individuals | 4 weeks | 600 mg dried garlic | Significant decrease in fibrinogen and fibrinopeptide A and increase in streptokinase-activated plasminogen and fibrinopeptide B beta 15–42 coagulation factor. The decrease in Serum cholesterol levels, Systolic, and diastolic blood pressure | [43] | |
| Obesity | |||||
| DB, R, PC | 110 individuals | 800 mg garlic powder | Significant reductions were observed in body weight and body fat mass (P < 0.05) | [44] | |
| R, DB, PC | 75 individuals | 12 weeks | Dried garlic powder tablets (10.8 mg alliin (3-(2-propenylsulfinyl)-L-alanine) | No clinically relevant lipid-lowering and blood pressure-lowering effects in middle-aged, normolipidemic individuals. The putative anti-atherosclerotic effect of garlic may be linked to risk markers other than blood lipids | [45] |
| Clinical Trial | Participants | Duration | Dose | Summary | References |
|---|---|---|---|---|---|
| R | 52 patients | 16 weeks | Allicin (1 mg) was injected intralesionally | Allicin improved the signs and symptoms associated with stage II oral submucous fibrosis (OSF) without notable side effects | [19] |
| 51 patients | AGE (0.16–2.4 mL) | AGE had a potential suppressive effect on colorectal adenomas in humans | [47] | ||
| R, DB | 3365 individuals | 7.3 years | AGE and steam-distilled garlic oil | No beneficial effects on precancerous gastric lesion prevalence or gastric cancer incidence | [48] |
| >5000 participants | 200 mg synthetic allitridum and 100 μg selenium | Large doses of allitridum and microdoses of selenium may effectively prevent gastric cancer, especially in men | [49] | ||
| 50 patients (42 with liver cancer, 7 with pancreatic cancer and 1 with colon cancer) | AGE | A marked improvement on natural killer (NK) cell activity, but no improvement in quality of life | [50] |
| Clinical Trial | Participants | Duration | Dose | Summary | References |
|---|---|---|---|---|---|
| R, DB | 30 individuals | 3 months | 2 garlic tablets, 3 times/day (each tablet containing 400 mg dry garlic powder and over 1000 mg allicin) | Garlic significantly reduced fasting blood sugar (FBS) and HbA1c in T2DM | [53] |
| PC | 210 individuals | 24 weeks | Garlic tablets at 300, 600, 900, 1200 and 1500 mg/day | Significant reduction in FBS and improvement in HbA1C were observed at higher garlic doses | [54] |
| DB, PC | 60 individuals | 4 weeks | Garlic powder tablet Allicor | Better metabolic control due to the lowering of FBS, serum fructosamine and triglyceride levels | [55] |
| DB, R | 32 individuals | 12 weeks | Combination of turmeric and garlic extracts: 1.2, 1.6 and 2.4 g/day | 2.4 g/day decreased FBS, 2 h postprandial glucose level, HbA1C, total cholesterol, LDL-c, triglyceride, and body mass index more than the two other dosages (1.2 and 1.6 g/day). No side effects were observed on kidney and liver functions | [56] |
| R, PC | 25 individuals | 2 months | 900 mg of garlic capsules | Improvement in blood pressure in diabetic patients. Garlic supplementation can be useful in controlling complications and treating diabetes in the future | [57] |
| Meta-analysis | 768 individuals | Daily garlic (allicin) supplement ranged from 0.05–1.5 g | Garlic supplement played a positive and sustained role in blood glucose, total cholesterol, and high/low-density lipoprotein regulation in T2DM | [52] | |
| Meta-analysis | Garlic reduced lipid profiles and glucose parameters, being therapeutically effective in patients suffering from cardiovascular diseases and diabetes | [51] | |||
| Meta-analysis | Garlic consumption could significantly decrease FBS especially in patients suffering from both diabetes and hypercholesterolemia | [58] |
| Clinical Trial | Participants | Duration | Dose | Summary | Reference |
|---|---|---|---|---|---|
| Cytochrome 450 | |||||
| R | 12 healthy volunteers | 28 days | Probe-drug cocktails (caffeine 100 mg, midazolam 8mg, chlorzoxazone 500 mg and debrisoquin 5 mg) were daily administered before supplementation (baseline) and at the end of supplementation (1500 mg of garlic oil) | Garlic oil significantly decreased 6-hydroxychlorzoxazone/chlorzoxazone serum ratios by 39% (p = 0.03), suggesting CYP2E1 inhibition | [59] |
| R | 10 healthy volunteers | 4 days | 20 mg of garlic extract, with a single dose of ritonavir (a substrate of CYP3A4 and P-gp) co-administered on the last day | Garlic supplement decreased the peak plasma concentration of ritonavir by 1%, and the area under the plasma concentration-time curve by 17%. Garlic showed a large inter-subject variation on ritonavir pharmacokinetics, suggesting that combination of ritonavir with garlic may be both an inducer and inhibitor of CYP3A4 and P-gp | [60] |
| R | 10 healthy volunteers | 21 days | Garlic tablet (containing allicin 4.64 mg and alliin 11.2 mg) twice daily for 21 days, with a 1200 mg dose of saquinavir, thrice daily on the last 3 days of garlic supplementation. After a 10-day garlic washout period, saquinavir was administered again for 3 days at the same dosage | Authors suggested that the effect of garlic supplements on the pharmacokinetics of saquinavir may be related to differences in CYP3A4 content, differences in the metabolism or absorption of garlic | [61] |
| Cytochrome P450 2D6 and 3A4 | |||||
| 40 healthy volunteers | 14days | 1800 mg tablets of a garlic extract containing allicin, alliin and alliinase twice daily | No significant differences on pharmacokinetic parameters were found (maximum concentration in plasma, 27.3 ± 2.6 ng/mL versus 27.3 ± 4.8 ng/mL; time to reach maximum concentration in plasma, 1.9 ± 1.4 h versus 2.4 ± 1.8 h; area under the time-versus-concentration curve, 537 ± 94 h ng mL−1 versus 548 ± 159 h ng mL−1; half-life of elimination, 13.7 ± 4.4 h versus 14.5 ± 4.3 h) | [62] | |
| Oxidative stress status | |||||
| DB, R, PC | 10 individuals | 2 weeks | 600 mg/daily | Reduced the susceptibility of apolipoprotein B-containing lipoproteins to oxidation | [65] |
| 25 individuals | 2 months | Coated garlic powder tablets (900 mg with an alliin content of 1.3% and an allicin content of 0.6%) | Significant increase in reduced glutathione (GSH) concentration in circulating human erythrocytes after the 2-month period | [66] | |
| Preeclampsia | |||||
| R, DB, PC | 44 individuals | 9 weeks | Garlic tablet (400 mg garlic and 1 mg allicin) once daily | Decreased hs-CRP (the high-sensitivity C-reactive protein) and increased GSH, but did not affect lipid profiles, total antioxidant capacity, and pregnancy outcomes | [67] |
| R, DB, PC | 300 individuals | 8 weeks | Garlic tablet (800 mg) | Reduced the occurrence of hypertension alone, but it was not effective in prevention of preeclampsia in the third trimester of pregnancy | [68] |
| Anti-ulcer (H. pylori infection) | |||||
| 20 individuals | 14 days | Allicin (800 mg/daily) | After allicin-treatment, H. pylori infection was still found in all patients | [69,70] | |
| 210 individuals | 14 days | Allicin (1200–4200 μg/daily) | Allicin was potentially effective on H. pylori eradication | [69,71] | |
| Open-Label, Randomized | 112 patients | 14 days | Allicin (24 mg/daily) and omeprazole 20 mg, amoxicillin 2000 mg/daily | Eradication rate was 73% in allicin-based therapy | [72] |
| R, DB, PC | 96 individuals | 5 days | Allicin (10 mg/daily) | Allicin adhesive tablets were effective in reducing ulcer size and alleviating ulcer pain without significant side effects | [21] |
| Male genital wart | |||||
| DB, R, PC | 35 male patients | 2 months | 10% garlic extract | Garlic extract had a similar effect of cryotherapy. The majority of patients (69.7%) showed complete clearing of the lesions in the garlic group while complete clearing was observed in 78.8% of patients in the cryotherapy group. | [73] |
| Common cold prevention | |||||
| DB, PC | 146 volunteers | 12 weeks | Allicin-containing garlic supplement, one capsule daily | Allicin-containing supplement seemed to prevent attack by the common cold virus | [74] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sharifi-Rad, J.; Cristina Cirone Silva, N.; Jantwal, A.; D. Bhatt, I.; Sharopov, F.; C. Cho, W.; Taheri, Y.; Martins, N. Therapeutic Potential of Allicin-Rich Garlic Preparations: Emphasis on Clinical Evidence toward Upcoming Drugs Formulation. Appl. Sci. 2019, 9, 5555. https://doi.org/10.3390/app9245555
Sharifi-Rad J, Cristina Cirone Silva N, Jantwal A, D. Bhatt I, Sharopov F, C. Cho W, Taheri Y, Martins N. Therapeutic Potential of Allicin-Rich Garlic Preparations: Emphasis on Clinical Evidence toward Upcoming Drugs Formulation. Applied Sciences. 2019; 9(24):5555. https://doi.org/10.3390/app9245555
Chicago/Turabian StyleSharifi-Rad, Javad, Nathália Cristina Cirone Silva, Arvind Jantwal, Indra D. Bhatt, Farukh Sharopov, William C. Cho, Yasaman Taheri, and Natália Martins. 2019. "Therapeutic Potential of Allicin-Rich Garlic Preparations: Emphasis on Clinical Evidence toward Upcoming Drugs Formulation" Applied Sciences 9, no. 24: 5555. https://doi.org/10.3390/app9245555
APA StyleSharifi-Rad, J., Cristina Cirone Silva, N., Jantwal, A., D. Bhatt, I., Sharopov, F., C. Cho, W., Taheri, Y., & Martins, N. (2019). Therapeutic Potential of Allicin-Rich Garlic Preparations: Emphasis on Clinical Evidence toward Upcoming Drugs Formulation. Applied Sciences, 9(24), 5555. https://doi.org/10.3390/app9245555









