Synthesis and Preliminary Screening of the Biological Activity of Sulindac Sulfoximine Derivatives
Abstract
1. Introduction
2. Materials and Methods
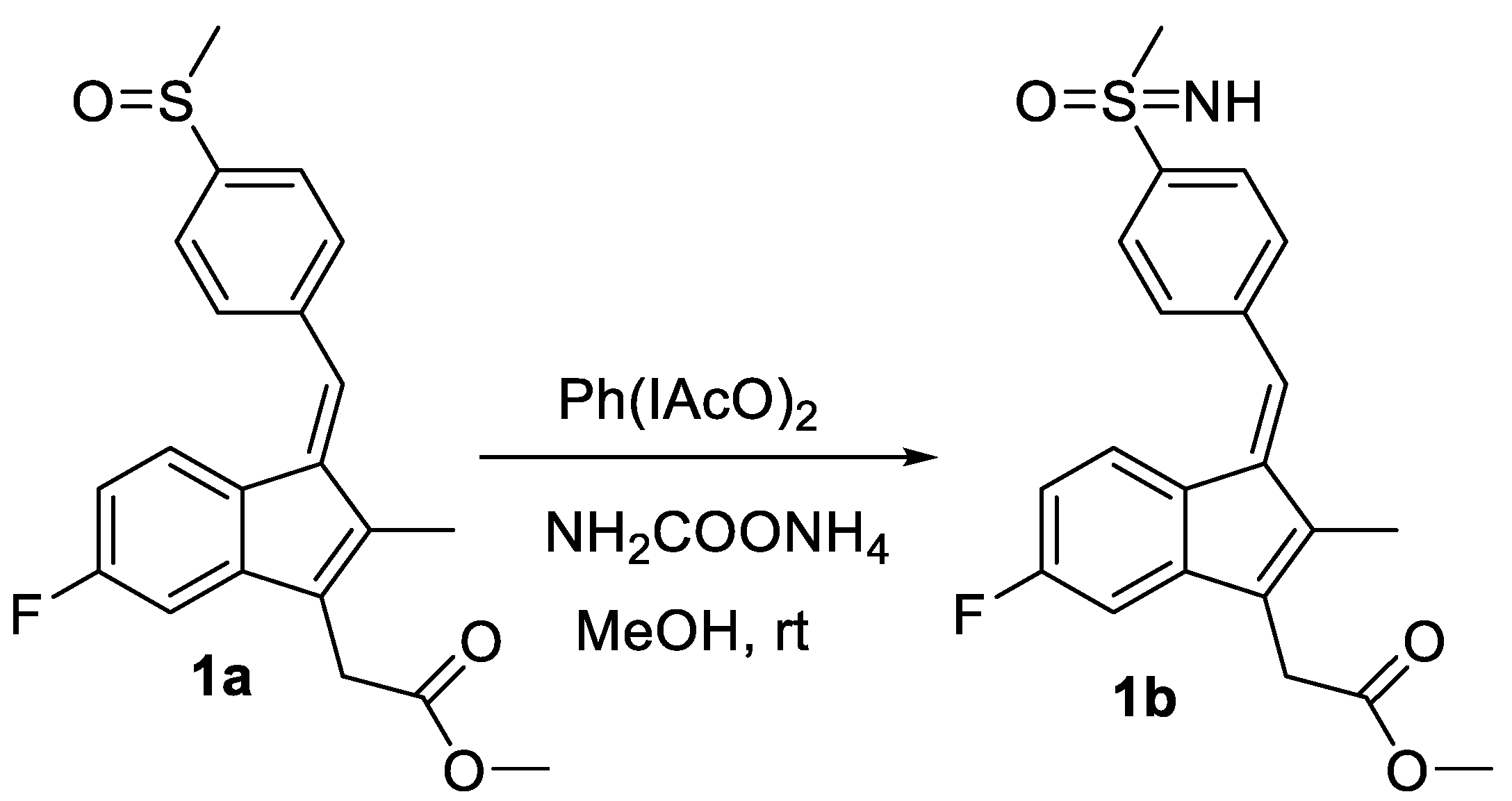
2.1. Synthesis
2.2. Biological Activity
2.2.1. Cell Cultures and Treatment
2.2.2. Cell Viability Assay
2.2.3. COX Inhibition Assay
2.2.4. Statistical Analysis
3. Results and Discussion
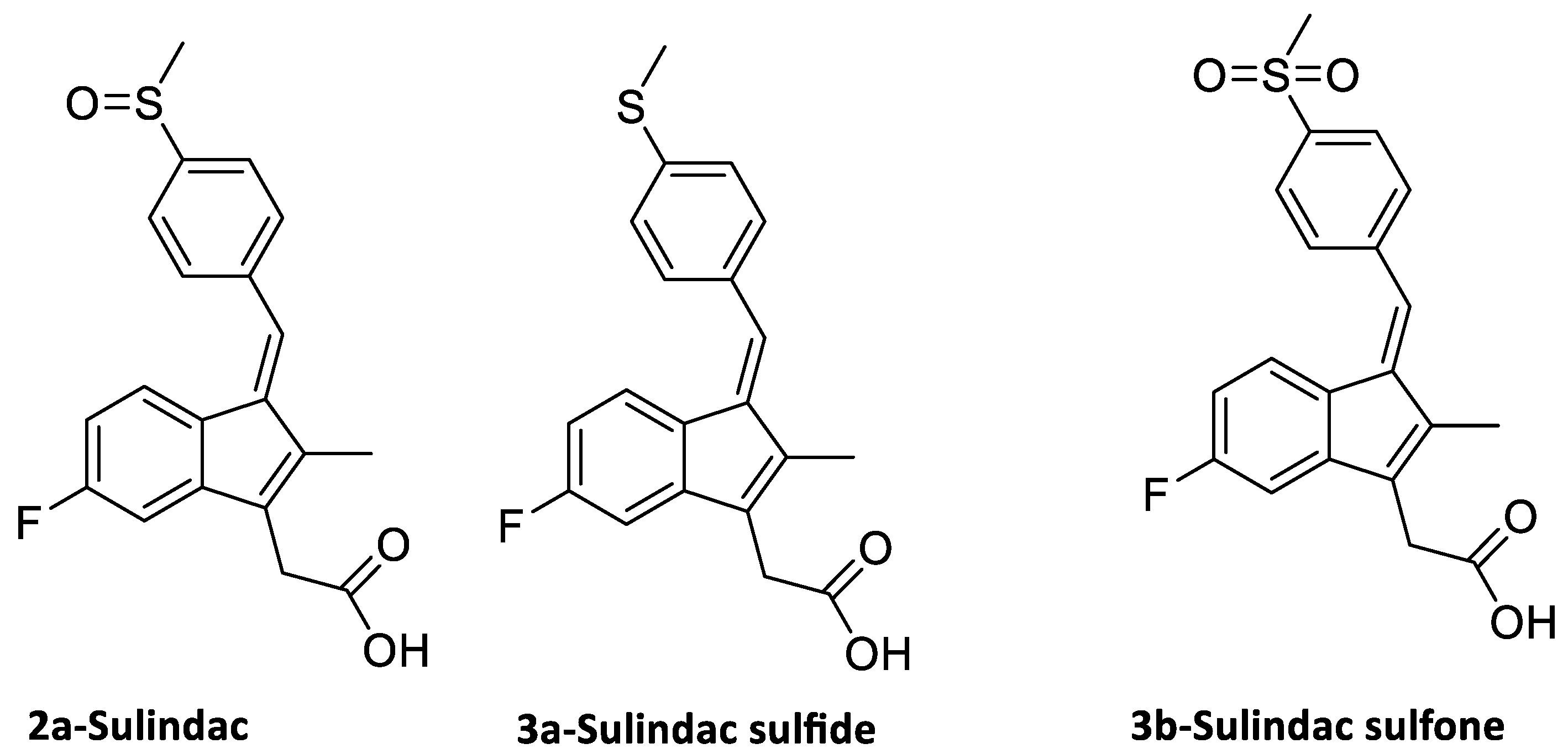
3.1. Synthesis of Sulindac Derivatives
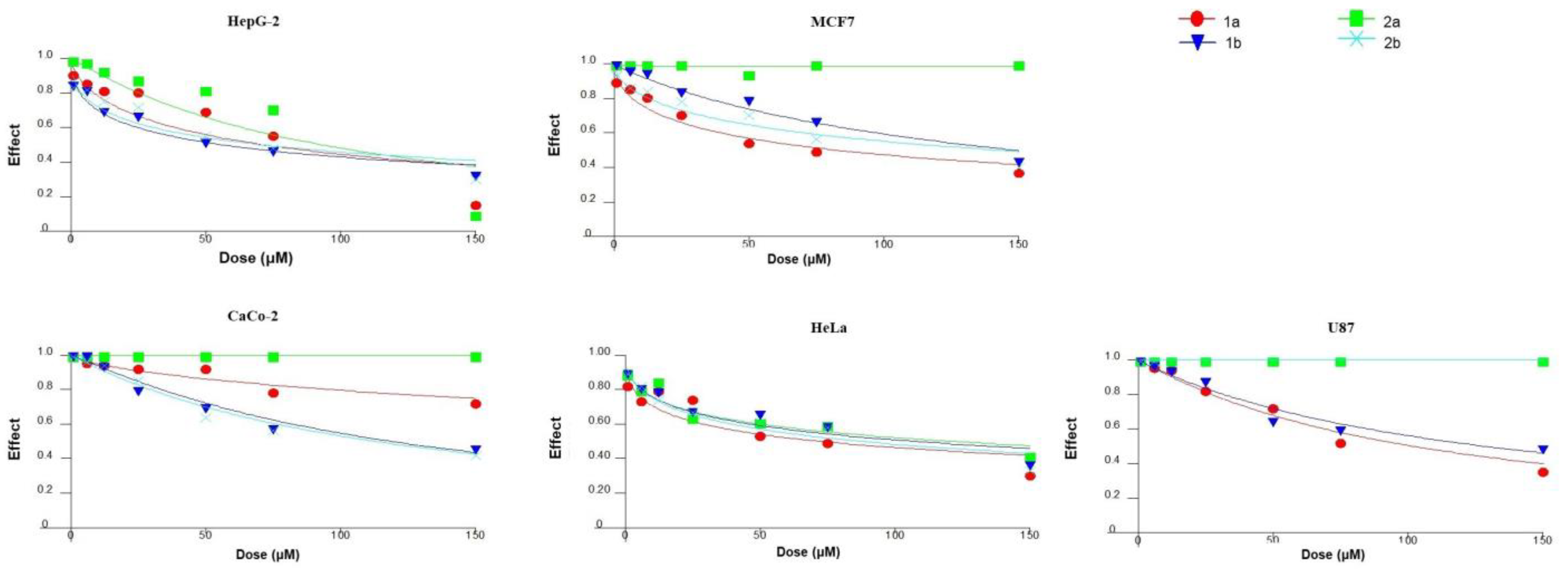
3.2. Cytotoxicity Studies
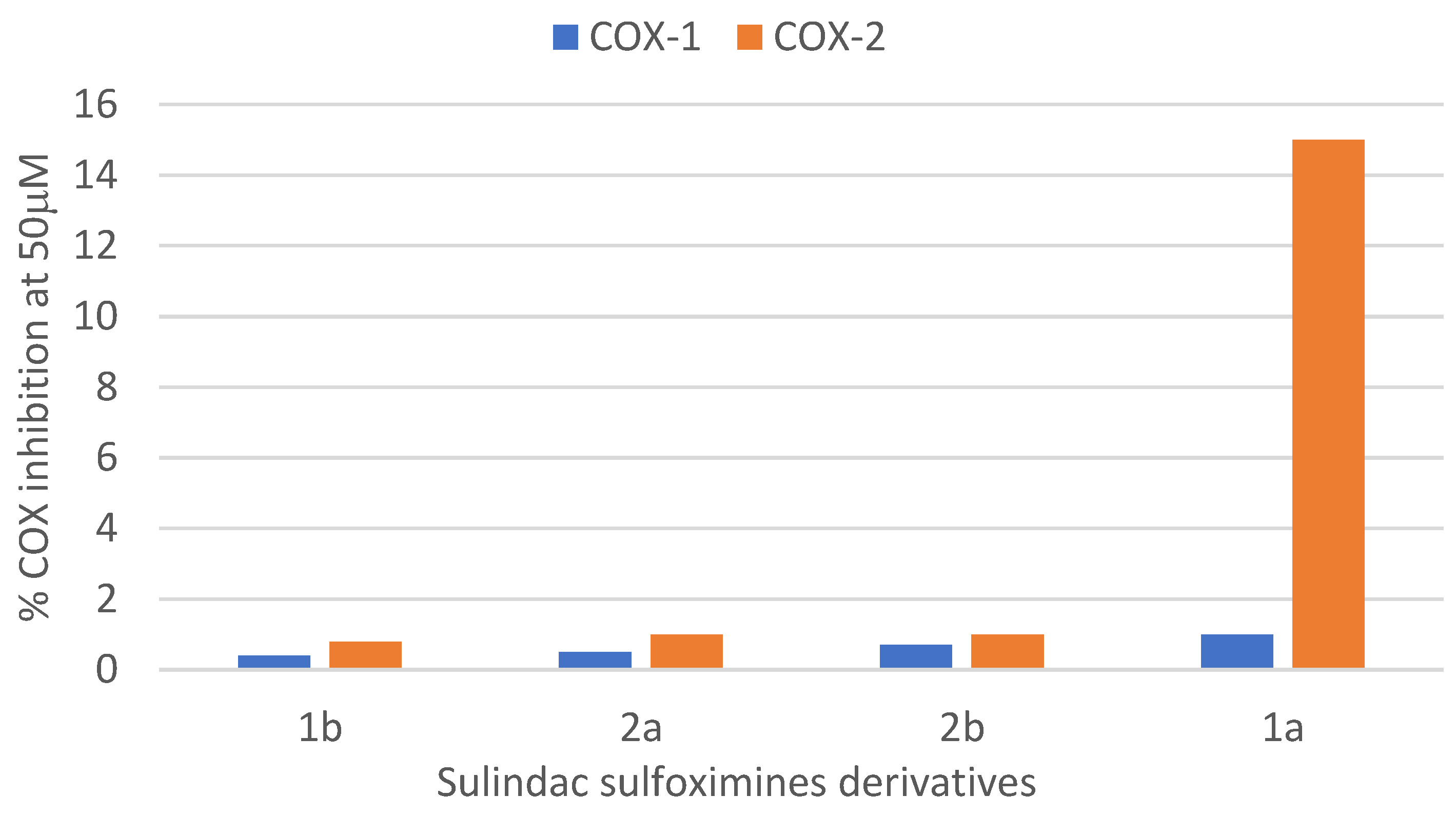
3.3. COX Inhibition Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thiruchenthooran, V.; Sanchez-Lopez, E.; Gliszczynska, A. Perspectives of the Application of Non-Steroidal Anti-Inflammatory Drugs in Cancer Therapy: Attempts to overcome their Unfavorable Side Effects. Cancers 2023, 13, 475. [Google Scholar] [CrossRef]
- Rashid, G.; Khan, N.A.; Elsori, D.; Rehman, A.; Tanzeelah, A.H.; Maryam, H.; Rais, A.; Usmani, M.S.; Babker, A.-M.; Kamal, M.A.; et al. Non-steroidal anti-inflammatory drugs and biomarkers: A new paradigm in colorectal cancer. Front. Med. 2023, 10, 1130710. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, A.; Yarla, N.S.; Madka, V.; Rao, C.V. Clinically Relevant Anti-Inflammatory Agents for Chemoprevention of Colorectal Cancer: New Perspectives. Int. J. Mol. Sci. 2018, 19, 2332. [Google Scholar] [CrossRef]
- Prasher, P.; Sharma, M. Medicinal Chemistry of Indane and Its Analogue: A Mini Review. Chem. Sel. 2021, 6, 2658–2677. [Google Scholar] [CrossRef]
- Suneela, D.; Astha, J.; Kunal, T. Design and Applications of Bioprecursors: A Retrometabolic Approach. Curr. Drug Metab. 2014, 15, 291–325. [Google Scholar] [CrossRef]
- Lim, J.T.; Piazza, G.A.; Han, E.K.-H.; Delohery, T.M.; Li, H.; Finn, T.S.; Buttyan, R.; Yamamoto, H.; Sperl, G.J.; Brendel, K.; et al. Sulindac derivatives inhibit growth and induce apoptosis in human prostate cancer cell lines. Biochem. Pharmacol. 1999, 58, 1097–1107. [Google Scholar] [CrossRef]
- Niitsu, Y.; Takayama, T.; Miyanishi, K.; Nobuoka, A.; Hayashi, T.; Kukitsu, T.; Takanashi, K.; Ishiwatari, H.; Abe, T.; Kogawa, T.; et al. Chemoprevention of colorectal cancer. Cancer Chemother Pharmacol. 2004, 54 (Suppl. S1), S40–S43. [Google Scholar] [CrossRef] [PubMed]
- Gerner, E.W.; Bruckheimer, E.; Cohen, A. Cancer pharmacoprevention: Targeting polyamine metabolism to manage risk factors for colon cancer. J. Biol. Chem. 2018, 293, 18770–18778. [Google Scholar] [CrossRef]
- Mathew, B.; Hobrath, J.V.; Connelly, M.C.; Guy, R.K.; Reynolds, R.C. Diverse amide analogs of sulindac for cancer treatment and prevention. Bioorganic Med. Chem. Lett. 2017, 27, 4614–4621. [Google Scholar] [CrossRef]
- Tinsley, H.N.; Mathew, B.; Chen, X.; Maxuitenko, Y.Y.; Li, N.; Lowe, W.M.; Whitt, J.D.; Zhang, W.; Gary, B.D.; Keeton, A.B. Novel Non-Cyclooxygenase Inhibitory Derivative of Sulindac Inhibits Breast Cancer Cell Growth In Vitro and Reduces Mammary Tumorigenesis in Rats. Cancers 2023, 15, 646. [Google Scholar] [CrossRef] [PubMed]
- Sahu, A.; Raza, K.; Pradhan, D.; Kumar Jain, A.; Verma, S. Cyclooxygenase-2 as a therapeutic target against human breast cancer: A comprehensive review. WIREs Mech Disease. 2023, 15, e1596. [Google Scholar] [CrossRef]
- Spector, D.; Krasnovskaya, O.; Pavlov, K.; Erofeev, A.; Gorelkin, P.; Bologlazkina, E.; Majouga, A. Pt(IV) Prodrugs with NSAID as Axial Ligands. Int. J. Mol. Sci. 2021, 22, 3817. [Google Scholar] [CrossRef]
- Bhata, M.A.; Al-Omara, M.A.; Alsaifa, N.A.; Almehiziaa, A.A.; Naglahb, A.M.; Razakd, S.; Khana, A.A.; Ashraf, N.M. Novel sulindac derivatives: Synthesis, characterisation, evaluation of antioxidant, analgesic, anti-inflammatory, ulcerogenic and COX-2 inhibition activity. J. Enzyme Inhib. Med. Chem. 2020, 35, 921–934. [Google Scholar] [CrossRef]
- Han, Y.; Xing, K.; Zhang, J.; Tong, T.; Shi, Y.; Cao, X.; Yu, H.; Zhang, Y.; Liu, D.; Zhao, L. Application of sulfoximines in medicinal chemistry from 2013 to 2020. Eur. J. Med. Chem. 2021, 209, 112885. [Google Scholar] [CrossRef] [PubMed]
- Capozzi, M.A.M.; Cardellicchio, C. Organosulfur Compounds as Chiral Building Blocks. In Chiral Building Blocks in Asymmetric Synthesis: Synthesis and Applications; Wojaczyńska, E., Wojaczyński, J., Eds.; John Wiley and Sons: Hoboken, NJ, USA, 2022; pp. 441–462. [Google Scholar] [CrossRef]
- Jones, B.C.; Markandu, R.; Gu, C.; Scarfe, G. CYP-Mediated Sulfoximine Deimination of AZD6738. Drug Metab. Dispos. 2017, 45, 1133–1138. [Google Scholar] [CrossRef]
- Sirvent, J.A.; Lücking, U. Novel Pieces for the Emerging Picture of Sulfoximines in Drug Discovery: Synthesis and Evaluation of Sulfoximine Analogues of Marketed Drugs and Advanced Clinical Candidates. Chem. Med. Chem. 2017, 12, 487–501. [Google Scholar] [CrossRef] [PubMed]
- Zenzola, M.; Doran, R.; Degennaro, L.; Luisi, R.; Bull, J.A. Transfer of Electrophilic NH Using Convenient Sources of Ammonia: Direct Synthesis of NH Sulfoximines from Sulfoxides. Angew. Chem. Int. Ed. Engl. 2016, 128, 7203–7207. [Google Scholar] [CrossRef] [PubMed]
- Degennaro, L.; Tota, A.; De Angelis, S.; Andresini, M.; Cardellicchio, C.; Capozzi, M.A.; Romanazzi, G.; Luisi, R. A Convenient, Mild and Green Synthesis of NH-Sulfoximines in Flow Reactors. Eur. J. Org. Chem. 2017, 2017, 6486–6490. [Google Scholar] [CrossRef]
- Naso, F.; Cardellicchio, C.; Affortunato, F.; Capozzi, M.A.M. Asymmetric synthesis of Sulindac esters by enantioselective sulfoxidation in the presence of chiral titanium complexes. Tetrahedron-Asymmetry 2006, 17, 3226–3229. [Google Scholar] [CrossRef]
- Fanizza, E.; Urso, C.; Iacobazzi, R.M.; Depalo, N.; Corricelli, M.; Panniello, A.; Agostiano, A.; Denora, N.; Laquintana, V.; Striccoli, M.; et al. Fabrication of photoactive heterostructures based on quantum dots decorated with Au nanoparticles. Sci. Technol. Adv. Mater. 2016, 17, 98–108. [Google Scholar] [CrossRef]
- Liedtke, A.J.; Crews, B.C.; Daniel, C.M.; Blobaum, A.L.; Kingsley, P.J.; Ghebreselasie, K.; Marnett, L.J. Cyclooxygenase-1-Selective Inhibitors Based on the (E)-2′-Desmethyl-sulindac Sulfide Scaffold. J. Med. Chem. 2012, 55, 2287–2300. [Google Scholar] [CrossRef] [PubMed]
- Vitale, P.; Scilimati, A.; Perrone, M.G. Update on SAR Studies Toward New COX-1 Selective Inhibitors. Curr. Med. Chem. 2015, 22, 4271–4292. [Google Scholar] [CrossRef] [PubMed]

| IC50 (µM) or % Cell Viability | |||||
|---|---|---|---|---|---|
| Compound | HepG-2 | MCF7 | U87 | CaCo-2 | HeLa |
| 1b | 58 ± 4 | 145 ± 9 | 130 ± 10 | 120 ± 9 | 105 ± 6 |
| 2a | 93 ± 4 | 1 107 ± 8% | 1 114 ± 7% | 1 115 ± 7% | 120 ± 5 |
| 2b | 71 ± 3 | 145 ± 6 | 1 99 ± 5% | 1 72 ± 6% | 83 ± 6 |
| 1a | 70 ± 2 | 80 ± 6 | 102 ± 8 | 100 ± 5 | 75 ± 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cardellicchio, C.; Laquintana, V.; Iacobazzi, R.M.; Denora, N.; Scilimati, A.; Perrone, M.G.; Capozzi, M.A.M. Synthesis and Preliminary Screening of the Biological Activity of Sulindac Sulfoximine Derivatives. Appl. Sci. 2023, 13, 12002. https://doi.org/10.3390/app132112002
Cardellicchio C, Laquintana V, Iacobazzi RM, Denora N, Scilimati A, Perrone MG, Capozzi MAM. Synthesis and Preliminary Screening of the Biological Activity of Sulindac Sulfoximine Derivatives. Applied Sciences. 2023; 13(21):12002. https://doi.org/10.3390/app132112002
Chicago/Turabian StyleCardellicchio, Cosimo, Valentino Laquintana, Rosa Maria Iacobazzi, Nunzio Denora, Antonio Scilimati, Maria Grazia Perrone, and Maria Annunziata M. Capozzi. 2023. "Synthesis and Preliminary Screening of the Biological Activity of Sulindac Sulfoximine Derivatives" Applied Sciences 13, no. 21: 12002. https://doi.org/10.3390/app132112002
APA StyleCardellicchio, C., Laquintana, V., Iacobazzi, R. M., Denora, N., Scilimati, A., Perrone, M. G., & Capozzi, M. A. M. (2023). Synthesis and Preliminary Screening of the Biological Activity of Sulindac Sulfoximine Derivatives. Applied Sciences, 13(21), 12002. https://doi.org/10.3390/app132112002










