Abstract
Currently, several biological activities are attributed to the acetogenins (ACGs) of Annonaceae (including Annona muricata); among these is antimicrobial activity. The main focus of this experiment was to evaluate the antimicrobial effect of the crude extract (CE) and purified acetogenins (P-ACGs) from the endosperm of A. muricata seeds using agar disk diffusion, lethality, sub-lethal, and potential damage membrane tests against Gram-positive and Gram-negative microorganisms. According to the results, P-ACGs present the highest antibacterial effect (12.5–4000 µg/mL) against Enterococcus faecalis (11–15.67 mm), Listeria monocytogenes (12–18 mm), Aeromonas hydrophila (10.33–11.67 mm), Bulkholderia cenocepacia (11–12 mm), and Salmonella paratiphy (11–15.67 mm), and a minimum inhibitory concentration ranging from 0.009 to 12.50 µg/mL. Measurement of the membrane potential shows that, in the presence of P-ACGs, the number of viable cells is reduced, with a significant logarithmic reduction observed (0.38, 1.27, and 1.81 CFU/mL) and a significant sub-lethal lesion (57.78, 96.14, and 98.42%) in Escherichia coli, E. faecalis, and L. monocytogenes, respectively. According to this study, the results demonstrate that P-ACGs from A. muricata seeds are potent and effective antibacterial compounds with potential pharmaceutical applications.
1. Introduction
Annona muricata, known as “guanabana” or soursop, is a highly valued fruit with a particular flavor, aroma, and nutritional composition [1]. Soursop is characterized by its pulp (which constitutes 67% of the edible fraction), mainly consumed in fresh or processed juices, nectars, purees, and ice cream, among others. The inedible fraction (33%) from fruit processing is composed of peel (20%), columella (4%), and seeds (9%), and, usually, they are discarded [2,3]. Several studies demonstrate bioactive compounds in A. muricata seeds, mainly acetogenins (ACGs) [4,5,6].
ACGs are natural compounds (polyketides) with a lengthy aliphatic chain with 35–37 carbon atoms. Its core contains either oxygenated tetrahydrofuran rings (-OH) or an γ-lactone α-β-saturated/unsaturated [7,8]. These compounds are subjected to several research projects of pharmaceutical importance due to their important biological activities (cytotoxic, antiproliferative, neuroprotective, and anxiolytic) in lower concentrations, including antimicrobial effects [7,9,10,11,12,13]. The extracts of crude, fractionated (phenolic compounds, alkaloids, and ACGs), and purified ACGs from A. muricata trees, such as roots, stems, leaves, and fruit pulp, present antifungal and antibacterial agents [11,14,15,16]. The ability of ACGs to inhibit NADH ubiquinone oxidoreductase (mitochondrial I complex) and NADH ubiquinone oxidase in the microbial plasma membrane consequently decreases ATP production, leading to microbial cell death [7,17].
Studies have evaluated the antimicrobial effect of crude extracts from A. muricata seeds [18,19,20]. However, purified ACG information is limited. One study evaluated the antibacterial properties of aqueous and methanolic extracts from A. muricata seeds against Salmonella enterica ser. Enteritidis, Staphylococcus aureus, and L. monocytogenes [18]. According to the authors, methanolic extract demonstrated concentration-dependent antibacterial activity. Recently, a study investigated the effectiveness of several ethanolic and aqueous extracts from A. muricata seeds in a concentration of 100 mg/mL against methicillin-resistant S. aureus (MRSA) and revealed a reduced effect (9.33–9.67 mm) [19]. However, no antimicrobial effect was observed in extracts (hexane, chloroform, and methanol) from A. muricata seeds against L. monocytogenes, Escherichia coli, Vibrio parahaemolylicus, and Pseudomonas aeuroginosa [20]. The studies based on extracts of A. muricata leaves evaluated the effect of crude methanolic extract from A. muricata leaves against strains of S. aureus, Bacillus cereus, E. faecalis, Enterobacter cloacae, Enterobacter aerogenes, Shigella dysenteriae, Salmonella typhimurium, Salmonella choleraesuis, Pseudomonas aeruginosa, and Escherichia coli, resulting in a bactericidal effect on the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) (MIC/MBC = 1) against S. typhimurium, S. aureus, and P. aeruginosa. However, a bacteriostatic effect is also observed in these microorganisms (MIC/MBC ≠ 1) 16]. Therefore, effectiveness is not always equated to bacterial cell death. The bacteriostatic activity, in the case of E. faecalis, presented a low MIC value (39 μg/mL); however, the bactericidal activity in P. aeruginosa obtained a MIC 16 times higher (625 μg/mL) [16].
Similarly, the analysis of the performance of the fractions obtained from the pulp extract of A. muricata [11] demonstrated (2 mg/mL) an inhibitory effect against all the bacterial strains evaluated (E. aerogenes (17.5–18.25 mm) > S. typhimurium (20 mm) > Enterococcus faecalis (14.25 mm) > B. subtilis (10.75 mm)). In evaluating A. muricata leaf extract against S. aureus and Escherichia coli with a MIC value of 1.25 mg/mL, the dichloromethane extracts demonstrated strong antibacterial activity on E. coli [14]. Nevertheless, an MIC > 5 mg/mL reduced the growing of S. aureus. All the bioactivity described previously in A. muricata extracts is mainly attributed to the presence of phenols, steroids, and alkaloids [11,14].
Due to the increase in the presentation of bacterial diseases and the presence of bacteria with multi-resistance to antibiotics, the search for new antimicrobial agents is an important affair for public health. The mortality associated with these infections worldwide is estimated at approximately 700,000 deaths per year in 2016, with an increase coming soon [15]. Therefore, this research aimed to assess the antibacterial activity of purified and crude acetogenin extract from A. muricata seeds against Gram-positive and Gram-negative bacteria.
2. Materials and Methods
2.1. Reagents
Silica gel, 3,5-hydroxybenzoic acid, dimethyl sulfoxide, ampicillin, and McFarland’s reagent 0.5 (Sigma-Aldrich, St. Louis, MO, USA) were used. Petroleum ether, methanol, potassium hydroxide, dichloromethane, ethanol, and sodium chloride with an analytical grade, as well as nutrient broth and agar, were used throughout the experiment (Jalmek Scientific S.A., Guadalajara, Jalisco, Mexico).
2.2. Bacterial Strains
E. faecalis (ATCC 51575), L. monocytogenes (ATCC 15313), S. aureus (ATCC 33862), Streptococcus salivaris (ATCC 13419), Streptococcus mutans (ATCC 35668), Streptococcus mitis (ATCC 13770), Aeromonas hydrophila (ATCC 7966), Klebsiella pneumoniae (ATCC 33095), E. coli (ATCC 8739), S. paratyphi (ATCC 9150), Salmonella abatetuba (ATCC 35640), Salmonella cholerasius (ATCC 7001), and Bulkholderia cenocepacia (J2315) were supplied by the University of Guadalajara and handled according to the manufacturer instructions (Microbiologics®, Saint Cloud, MN, US.A). Prior to use, all instruments and reagents for the various microbiological assays were sterilized for 15 min at 121 °C.
2.3. Obtaining Crude Extract and Purified Acetogenins
The crude extract (CE) and purified ACGs (P-ACGs) were previously obtained as previously reported [21]. Briefly, CE was obtained from defatted endosperm of A. muricata seeds using methanol and thermosonication (temperature (50 °C), time (50 min), amplitude (100%) and pulse cycles (0.5 s)) with an ultrasonic system UP400S (Hielscher Ultrasonic, Teltow, Germany). CE was dried with a rotary evaporator (Yamato RE300, Tokyo, Japan).
To obtain P-ACGs, a portion of the CE was purified using open column chromatography; according to López-Romero et al. [21] and Yang et al. [22]. These authors concluded that P-ACGs are only ACGs and they are composed of pseudoannonacin, desacetyluvaricin, annonacin, squamostatin-D, isodesacetyluvaricin, squamocin, bullatacin, and other non-identified ACGs.
2.4. Antibacterial Assays of Crude Extract and Purified Acetogenins
The antibacterial assays were performed using the agar disk diffusion method [23] with modifications. Gram-negative strains (A. hydrophila, Escherichia coli, Klebsiella pneumoniae, Salmonella paratyphi, Salmonella abatetuba, Salmonella cholerasius, and Burkholderia cenocepacia) and Gram-positive strains (Listeria monocytogenes, S. aureus, E. faecalis, S. salivaris, S. mutans, and Streptococcus mitis) were used during the experiment. The strains were aerobically cultivated for 24 h at 37 °C in a nourishing broth (8 g/L, pH 7.0 ± 0.1) until the bacterial suspensions reached 1 × 106 CFU/mL according to McFarland standard (0.5 OD). Sterile forceps were used to insert the discs of sterile filter paper (7 mm in diameter) into Petri dishes with nutrient agar (23 g/L, pH 6.8 ± 0.1). The filter paper discs were impregnated according to treatment: 200 µL sterile distilled water (negative control), ampicillin at 500 µg/mL and dimethyl sulfoxide (DMSO) (a positive control), and solutions of CE or P-ACGs at different concentrations (12.5, 25, 50, 100, 200, 400, 800, 1000, 2000, and 4000 μg/mL). The inoculated Petri dishes were incubated for 24 h at 37 °C, and the inhibition halo (IH) in mm2 formed around the discs was measured using a vernier.
2.5. Minimum Inhibitory Concentration (MIC) of the Crude Extract and Purified Acetogenins against Gram-Positive and Gram-Segative Bacteria
MIC was calculated according to Parhusip and Sitanggang [24]. CE and P-ACGs concentrations (12.5–4000 μg/mL) were converted to values of natural logarithm (Ln), and data of IH were converted to values of squared millimeters (mm2). Then, a plot was constructed (squared millimeters of the IH vs. Ln concentrations of CE or P-ACGs). A linear regression model (R2) was realized to obtain Equation (1).
Equation (1) was used to obtain Equation (2), where “X” is the Ln concentration, “y” is the minimum value of the IH in mm2, b is the intercept of the X-axis, and “m” is the slope. Subsequently, Equation (3) was used to calculate MIC.
2.6. Inhibition Percentage of the Most Prevalent Harmful Bacteria Found in Foods: Enterococcus faecalis, Salmonella paratyphi, Escherichia coli, and Listeria monocytogenes by the Crude Extract and Purified Acetogenins
According to results in the antibacterial activity, the inhibition percentage among harmful bacteria prevalent in food was calculated based on the procedure outlined by Bibi et al. [25] (Equation (4)).
Tested sample (TS), negative control (NC) and positive control (PC) were calculated in millimeters (mm). All procedures described above were performed in triplicate for each bacterial strain using both CE and P-ACGs.
2.7. Measurement of the Membrane Potential of Enterococcus faecalis, Listeria monocytogenes, Salmonella paratyphi, and Escherichia coli Treated with the Crude Extract and Purified Acetogenins
Foodborne pathogens were also selected to evaluate the membrane potential. The bacterial strains (10 times diluted in saline) were dyed using Molecular Probes LIVE/DEAD BacLight kit, distinguishing between bacteria with intact and damaged plasma membranes [26]. Briefly, pathogenic strains were cultivated for 24 h at 37 °C in nutrient broth, and the biomass was recovered by centrifugation (11,624× g, 1 min). Subsequently, the pellets were washed twice with sterile phosphate-buffered saline (PBS) solution and resuspended (1 × 106 CFU/mL) in a sterile PBS. Three aliquots (1 mL) of each strain were prepared and distributed as follows: untreated cells stained with 10 μL 3,3’-diethyloxacarbocyanine iodine (DiOC2(3)), cells treated with CE (400 µg/mL) and stained with 10 μL DiOC2(3), and cells treated with P-ACGs (400 µg/mL) and stained with 10 μL DiOC2(3). All cells were incubated at room temperature for 15 min. The membrane potential of the cell suspensions was evaluated utilizing a flow cytometer (AttuneTM, Applied Biosystems, Waltham, MA, USA) and BacLight™ Bacterial Membrane Potential Kit (B34950; Molecular Probes, Eugene, OR, USA), according to the supplier. Each staining was prepared in duplicate. The negative control consisted of an inactivated cell suspension depolarized with ten μL of 3,3’-carbonyl cyanide-m-chlorophenylhydrazone (CCCP). An unstained cell suspension represents a positive control. Each sample was placed in the cytometer, starting with the positive control, to adjust the detection threshold, followed by the depolarized solution and the samples. The samples were excited with a 488 nm laser, and the fluorescence emitted in the red and green channels was recorded. Forward and side scatter and fluorescence with logarithmic signal amplification were recorded. The membrane potential of the cells treated with CE and P-ACGs was assessed and compared with cells that were not treated (positive control) and depolarized cells. The magnitude of membrane potential was measured through the red: green fluorescence ratio using density plots obtained by flow cytometry.
2.8. Lethality and Sublethal Injury on Enterococcus faecalis, Listeria monocytogenes, and Escherichia coli Caused by the Crude Extract and Purified Acetogenins
The pour-plate method assessed bacterial lethality and sub-lethal injury by serial dilution [27]. Nutrient broth (200 mL) with 10 mL of cell suspension (1 × 106 CFU/mL) adjusted to the McFarland scale (0.5 OD) was added with 100 µg/mL of CE or P-ACGs. The positive control consisted of cell suspension without extract. All samples were incubated at 37 °C for 15 min. Subsequently, 1 mL of each treatment was mixed with 9 mL of sterile saline solution (0.85% w/v) and homogenized. Serial dilutions (up to 10−7) were prepared, and 1 mL was taken and plated into nutrient agar using a plate pouring. Each bacterial strain was subjected to this process with each treatment, and the results were expressed as log CFU/mL. Lethality was measured by the difference between log CFU/mL colony counts in the treated samples (N), and the colony counts in the control positive without the extracts (No) (Equation (5)). The difference between the CFU/mL of the treated samples and the control positive (No) samples was used to calculate the sub-lethal injury percentage (Equation (6)).
2.9. Statistical Analysis
Data were collected from three separate experiments, and each sample was developed in triplicate. Results were given as the mean ± standard deviation. Data were analyzed with an ANOVA and Fisher’s test (α = 0.05) using a factorial design (10 × 2) for each bacteria strain. Additionally, Student’s T-test (α = 0.05) was utilized to compare the two treatments. Statistical software (v. 10 Stafsoft®, Tulsa, OK, USA) was used to analyze all data.
3. Results and Discussion
3.1. Antibacterial Assays of Crude Extract and Purified Acetogenins
The susceptibility of bacteria-specific compounds was determined using the disk diffusion test, where IH ≤ 7 mm is considered not active against bacteria and IH ≥ 12 mm demonstrates an inhibitory effect [28]. The impact of different concentrations relating to the bacteria, extract, and concentration of CE and P-ACGs from the endosperm of A. muricata seeds on Gram-positive bacteria are described in Table 1.

Table 1.
Antibacterial activity of the crude extract (CE) and purified acetogenins (P-ACGs) from the endosperm of A. muricata seeds against several Gram-positive bacteria.
According to the results, the P-ACGs showed superior performance compared to the CE; the highest IH of the CE and P-ACGs was observed in L. monocytogenes, with 16 mm IH applying 4000 µg/mL of CE, and 18 mm IH using 800 µg/mL of P-ACGs. In contrast, at all concentrations, the lowest inhibition was observed in S. salivaris for both treatments (7 mm IH with CE and 7–8 mm IH with P-ACGs); in particular, in concentrations <400 µg/mL, no inhibition was observed with S. aureus. Similarly, no inhibition with the CE against S. mutans was observed, while the P-ACGs induced an inhibition dependent on their concentration.
Raybaudi-Massilia et al. [18] reported that the methanolic extract from A. muricata seeds showed an antimicrobial effect on S. aureus and L. monocytogenes in a concentration-dependent response (from 0.1 to 0.5% v/v); however, a lyophilized extract presented no antimicrobial effect. Reports described that a CE extraction with hexane, chloroform, and methanol of A. muricata seeds has no antimicrobial activity against L. monocytogenes, Vibrio parahaemolylicus, and Pseudomonas aeuroginosa [20]. Partial purification of methanolic/chloroform fractions from the A. muricata pulp exhibited an IH of 13.75 mm with E. faecalis [29]. According to the authors, a general conclusion is that the CE concentration, type, purity, and compounds present in these fractions (ACGs, terpenes, saponins, and alkaloids) affected their antimicrobial activity. Therefore, the differences between CE and P-ACGs affect the ability to exert their mechanism of action. Isolated ACGs impacted the complex I (NADH-ubiquinone oxidoreductase) of bacterial cells, modifying the transit of negatively charged ions through the respiratory chain, [30] inhibiting the proton cascades affecting cellular respiration [30]. Thus, ACGs can directly modify the bacterial growth cycle by inducing cell death [16].
The antibacterial effect of the CE and P-ACGs was also determined against several Gram-negative bacterial strains. A significant effect (p < 0.05) on the IH was found with CE and P-ACGs with the bacteria tested (Table 2). The highest antibacterial activity was observed with P-ACGs compared to CE; however, some differences were observed according to the type of bacteria and concentration. Particularly in Salmonella paratyphi, CE presented an IH of 14 mm (50 µg/mL), compared to the P-ACGs’ IH of 14.5 mm (200–400 µg/mL); this is comparable to the methanolic and aqueous extracts from A. muricata seeds’ previously informed antimicrobial effects against Salmonella enterica ser. Enteritidis in a concentration-dependent response [18].

Table 2.
Antibacterial activity of the crude extract (CE) and purified acetogenins (P-ACGs) from endosperm of A. muricata seeds against several Gram-negative bacteria.
On the other hand, the lowest inhibition was observed against E. coli, with an IH of 9.3 mm (P-ACGs 1000 µg/mL); however, this increased with the concentration (<1000 µg/mL). As previously reported, A. muricata seed extracts with hexane, chloroform, and methanol present no effect against E. coli, suggesting a natural resistance to A. muricata extracts [20] that might be related to several signaling pathways as a response to stress, inducing an adaptive response [31]. The antimicrobial activity of the CE and P-ACGs was more efficient with Gram-negative bacteria than with Gram-positive bacteria (Table 1 and Table 2), suggesting cell envelope variation, physiology, and morphology as factors of natural resistance [27].
The cell envelope of Gram-negative bacteria is a relatively thin wall composed of a single layer of peptidoglycan, lipoproteins, lipopolysaccharides, and the outer membrane, which is crucial as a molecular barrier for stopping the loss of intracellular proteins and restricting access to drugs, especially hydrophobic antibiotics [16,31]. Due to the chelation capacity of calcium, A.G.C.s induce the formation of pores, modifying the solubility of the membrane and changing the structure and functionality of the membrane phospholipid bilayer [14,32]. Therefore, the chelating activity of ACGs plays an important role by increasing the membrane permeability of these microorganisms. Gram-positive and Gram-negative bacteria treated (CE and P-ACGs) with an inhibition halo are shown in Figure 1; the P-ACGs and CE presented a substantial IH, depending on the concentration and the type of bacteria evaluated.

Figure 1.
Effect of P-ACGs (a–d) and CE (e–h) on Gram-positive ((a,e) Enterococcus faecalis (b,f) Listeria Monocytogenes) and Gram-negative ((c,g) Aeromonas hydrophila; (d,h) Burkholderia cenocepacia). Concentration of P-ACGs and CE = (1) 4000 µg/mL; (2) 2000 µg/mL; (3) 1000 µg/mL, and (4) 800 µg/mL.
3.2. Minimum Inhibitory Concentration (MIC) of the Crude Extract and Purified Acetogenins against Gram-Positive and Gram-Negative Bacteria
The MIC values of the CE and P-ACGs tested on Gram-positive and Gram-negative bacteria demonstrated significantly different antibacterial activity (p < 0.05) (Table 3). The MIC values were observed in a range of 0.009 to 12.50 µg/mL, except for the case of the CE on S. mutans, where no inhibition was observed, associated with the low concentration of ACGs in the crude extract, which is higher in the P-ACGs [25]. In previous reports of purified compounds, MIC values of 10 µg/mL to > 100 µg/mL of crude extracts are reported, which present a good antimicrobial effect [33]. The CE and P-ACGs evaluated in this study showed characteristics of antibacterial agents, except for the P-ACGs on S. aureus (2.40–4.01 µg/mL). The MIC values of the CE and P-ACGs against the tested Gram-positive and Gram-negative bacteria are completely described in Table 3.

Table 3.
Antibacterial activity of crude extract and purified acetogenins on several bacterial strains.
The MIC values reported in the present work are higher than for the methanolic, ethanolic, and aqueous extracts of A. muricata bark, leaf, and root (65.25, 125, and 250 mg/mL, respectively) of E. coli and S. paratyphi [34].
Reports describe that combining antibiotics with A. muricata seed extract may increase drug uptake, reducing the efflux and biofilm activity of biofilm-forming Methicillin-resistant S. aureus [19]. Additionally, the MIC values for E. coli applying A. muricata extracts (leaves, seeds, roots, pulp) were between 256 and 1024 µg/mL were previously reported [35]. Comparatively, the MIC values reported for E. coli in this work with the CE (0.24 µg/mL) and P-ACGs (0.009 µg/mL) are much lower, indicating its greater antibacterial effect.
3.3. Inhibition Percentage of the Most Prevalent Harmful Bacteria Found in Foods: Enterococcus faecalis, Salmonella paratyphi, Escherichia coli, and Listeria monocytogenes by the Crude Extract and Purified Acetogenins
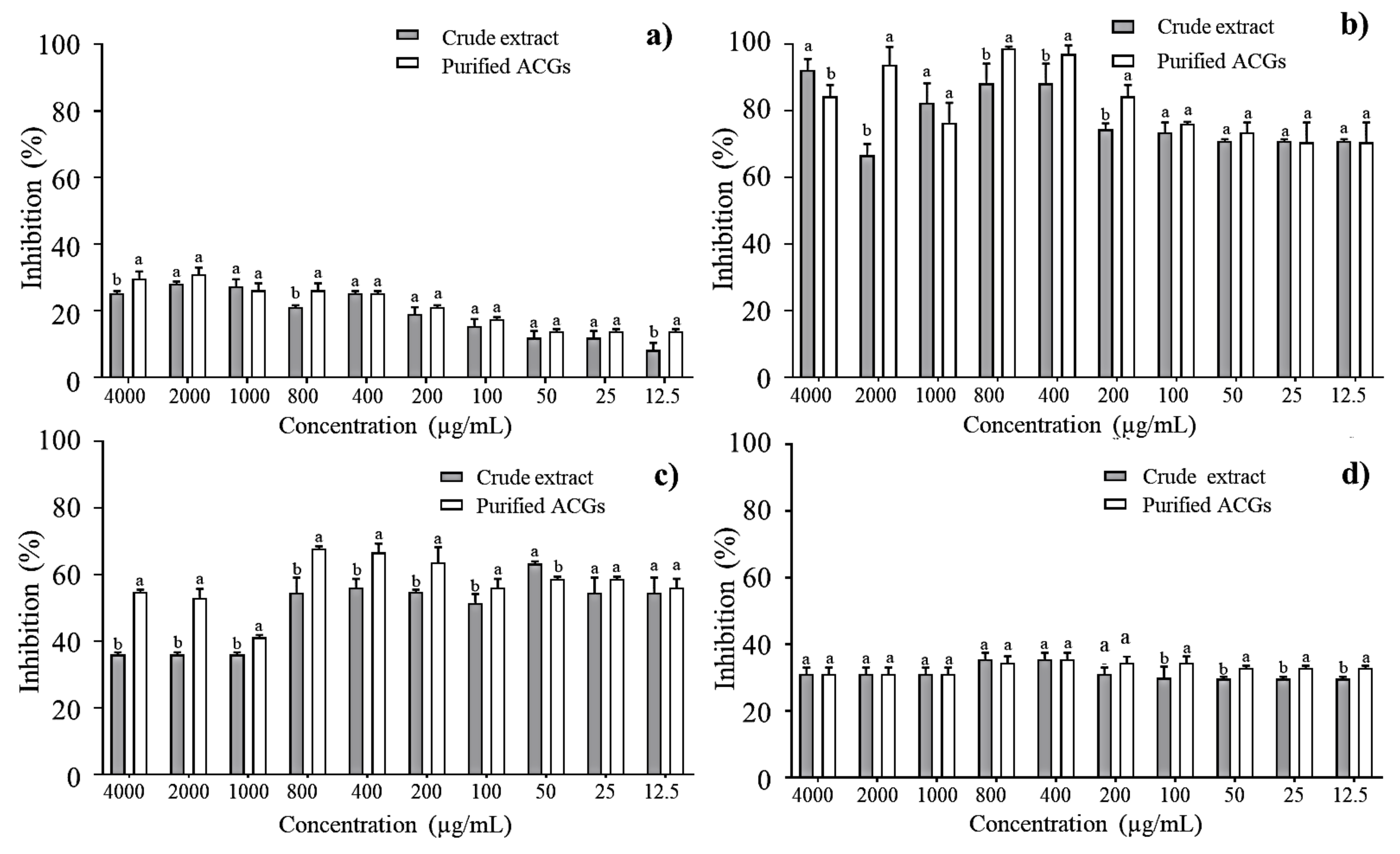
E. faecalis, S. paratyphi, E. coli, and L. monocytogenes were selected as the most prevalent harmful bacteria found in foods [36] to determine the percentage of bacterial inhibition of the CE and P-ACGs. Figure 2 shows statistically significant differences (p < 0.05) between the concentration-dependent CE and P-ACGs. The P-ACGs showed greater inhibition in L. monocytogenes (98.66%) when using 800 µg/mL. The number of unsaturations present in the aliphatic chain of ACGs is a structural feature that significantly influences antibacterial activity. ACGs insert into the cell membrane, affecting the cellular functionality and viability of the bacteria [37].
Figure 2.
Percentage inhibition of crude extract and purified acetogenins on the most common pathogenic bacteria in food: (a) Enterococcus faecalis; (b) Listeria monocytogenes; (c) Salmonella paratyphi and (d) Escherichia coli. Different letters (a, b) indicate significant statistical differences between treatments (α = 0.05) by each concentration.
Furthermore, at higher doses (4000 µg/mL), the CE exhibited a higher antibacterial effect (92.15%) against L. monocytogenes than P-ACGs (84.31%). This can be explained through previous reports. It has been reported that high concentrations of crude extracts can overcome antibacterial activity (depending on the individual bacteria) compared to some purified compounds. This is attributed to the fact that the crude extracts may contain different bioactive compounds with antimicrobial activity and that some bacteria are susceptible not only to a compound but to a number of them; in addition, a crude extract contains a complex mixture of secondary metabolites that could be exerting a synergistic effect against a certain type of bacteria [25].
In addition, at high doses (4000 µg/mL), the CE exhibited a greater antibacterial effect (92.15%) against L. monocytogenes than the P-ACGs (84.31%). This is consistent with previous reports, which demonstrate that CE may exceed the antibacterial activity of purified compounds. This is attributed to it containing different bioactive compounds with antimicrobial activity, and to the fact that some bacteria are susceptible not only to one compound but to several of them. Therefore, a CE contains a complex mixture of secondary metabolites that could exert a synergistic effect against certain types of bacteria [25]. Thus, different compounds working against L. monocytogenes could exist in greater quantities in the CE at that specific dose; however, in a minor concentration, the effectiveness of the CE decreases.
Regarding S. paratyphi and E. coli, the antibacterial activity was higher when P-ACGs was used (67.84% and 35.55%, respectively) at 800 µg/mL and 400 µg/mL compared to those observed with CE in the exact quantities. Congruent with the antibacterial effect reported, P-ACGs present an inhibition of 63% for S. paratyphi and 56.25% for E. coli [38].
Figure 2 shows that both the CE and P-ACGs present the lowest inhibitory effect against E. faecalis, having the highest inhibition percentage (30.95%) at 2000 µg/mL; this is similar to previously published results [39], informing a mortality rate of E. faecalis near 25% using a reconstituted solution of dried fractions from a leaf extract of A. muricata (1000 μg/mL). According to Neglo et al. [19], some bacteria may be resistant to some natural extracts, which is associated with their ability to produce biofilm.
3.4. Measurement of the Membrane Potential of E. faecalis, Listeria monocytogenes, Salmonella paratyphi, and Escherichia coli Treated with the Crude Extract and Purified Acetogenins
Flow cytometry is a fast and cost-effective technology for assessing the viability of microbial cells under the effect of natural extracts/bioactive compounds [40,41]. In this study, bacterial death was investigated using the fluorescent viability technique (Live/Dead® BacLight). This clearly shows the loss of the integrity of the plasma membrane. Damaged membranes render cells incapable of maintaining an electrical potential and these cells are considered dead. With the Live/Dead® BacLight technique, fluorochrome enters and stains dead cells with damaged membranes (Green fluorescence).
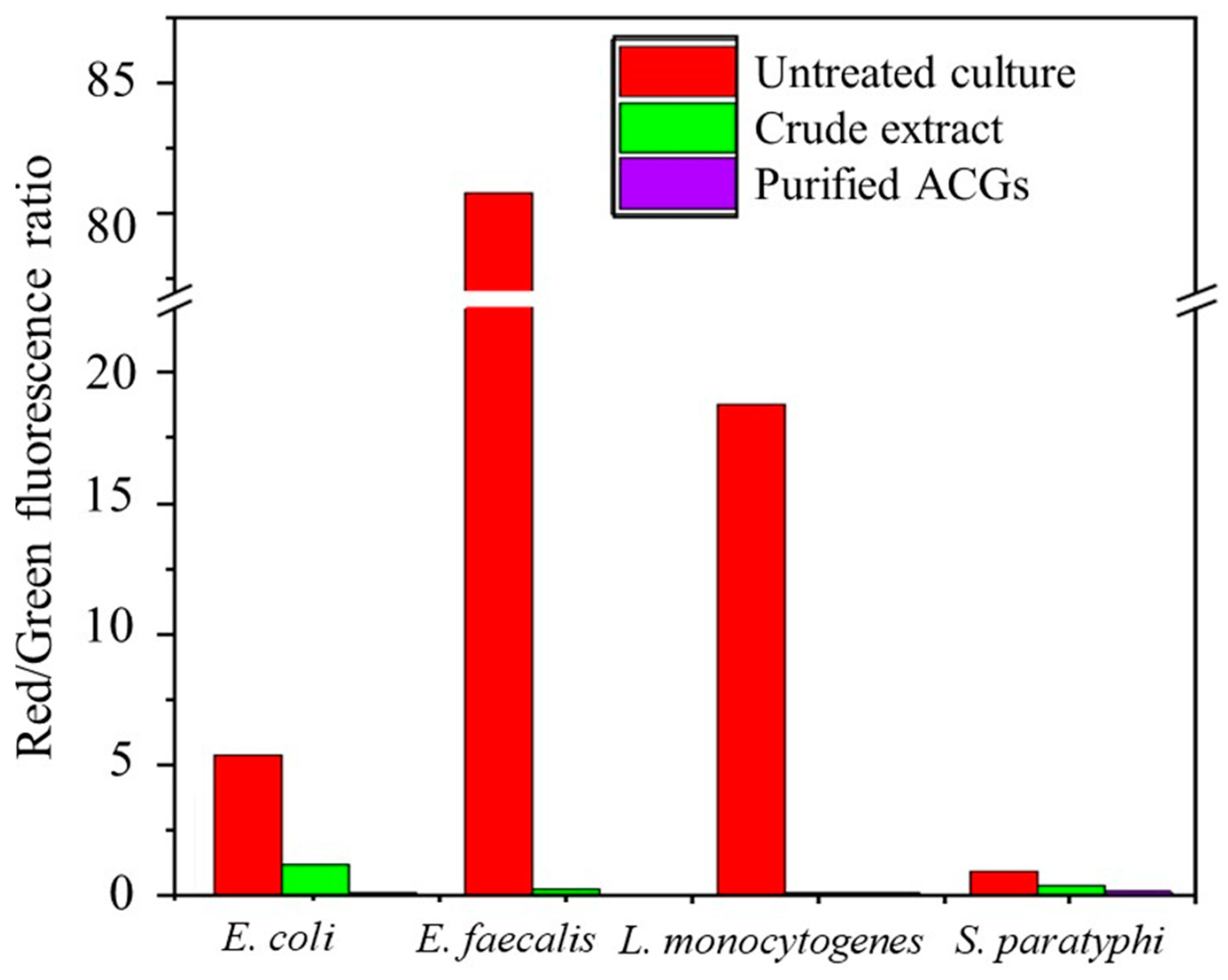
In contrast, living cells do not pick up fluorochrome because their membrane is intact and can be distinguished as stained or unstained (Red fluorescence) [16,41]. The cell viability ratio is expressed as follows: a high red/green ratio means that bacterial cells retain their membrane potential, whereas a low red/green ratio means that bacterial cells do not maintain their membrane potential because they are damaged. The cell viability measured by flow cytometry and expressed as a red/green fluorescence ratio is shown in Figure 3. Compared to traditional approaches, the BacLight membrane potential kit (Molecular Probes B34950, Carlsbad, CA, USA) allowed for a significantly more quantitative determination of the cell viability of CE and P-ACG-treated cells. This allowed for the calculation of the percentages of polarized and depolarized cells within a population of bacteria.
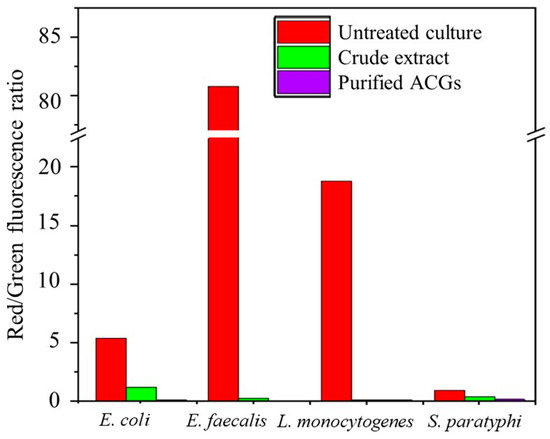
Figure 3.
The membrane potential of pathogenic bacterial strains treated and untreated with antibacterial extracts.
As expected, 10 M CCCP did not increase the potential of the treated cell membrane. However, the antibacterial effect was more significant in the cells exposed to P-ACGs (red/green ratio of 0.09 to 0.18) than in those exposed to CE (red/green ratio of 0.17 to 1.22), and this depended on the type of bacteria.
Figure 3 describes that bacteria treated with the P-ACGs induced a significant decrease in cell viability and increased bacterial cell death. Cell death is related to the effect of programmed cell death and lysis [16]. As previously mentioned, the CE and P-ACGs presented a more significant effect on Gram-positive bacteria than Gram-negative bacteria. Since Gram-negative bacteria have relatively thin cell walls composed of a single layer of peptidoglycan, lipoproteins, lipopolysaccharides, and the outer membrane, which acts as an efficient permeability barrier against hydrophobic antibacterial agents like ACGs, these results may be related to the structure of Gram-negative bacteria [16,42]. Therefore, the outer membrane reduces the access of ACGs, thus preventing these compounds from effectively infiltrating this intact membrane of Gram-negative bacteria. However, they may permeate the membrane of specific defective mutants of the outer membrane, which are damaged by chelators or polycations [16]. In this regard, Pinto et al. [16] used the LIVE/DEAD BacLight approach to assess the impact of A. muricata leaf extract on the cell viability of S. aureus, S. typhimurium, and E. faecalis. These authors showed the potential antibacterial impact of a leaf extract from A. muricata, increasing the number of dead cells in a concentration-dependent response. They reported that the leaf extract promoted changes at the cellular level in the tested bacteria that are indicative of cell death, mostly linked to the degradation of plasma membrane integrity. Furthermore, these authors attributed the antimicrobial effect to several alkaloids included in the extract. They suggested a synergy with compounds present in the extract, such as acetogenins.
3.5. Lethality and Sublethal Injury on Enterococcus faecalis, Listeria monocytogenes, and Escherichia coli Caused by the Crude Extract and Purified Acetogenins
According to the results, all treatments decreased the plate count (p < 0.05) for all tested bacteria (initial bacterial load ≈ 7.74–8.16 log CFU/mL) (Table 4).

Table 4.
Lethal effect of crude extract and purified acetogenins on Enterococcus faecalis, Listeria monocytogenes, and Escherichia coli.
The lowest log reduction was observed using CE (0.35–1.27 log CFU/mL), while the P-ACGs induced the highest log reduction (0.38–1.81 log CFU/mL). The P-ACGs had a high effect on L. monocytogenes (1.81 log CFU/mL) and E. faecalis (1.27 log CFU/mL) due to the presence of ACGs with unsaturations in the aliphatic chain that cause bacterial inhibition, [37] and the increase in purity [13]. The authors indicate that a final concentration of compounds is critical in antibacterial activity. However, the lowest bacterial reduction was obtained with CE and P-ACGs when evaluated against E. coli (0.35–0.38 log CFU/mL), where there was no significant difference (p < 0.05) between both samples.
According to the results, P-ACGs are more effective than CE in Gram-positive and Gram-negative bacteria, but their antibacterial activity varies according to the microorganisms. Gram-negative bacteria possess an effective permeability barrier against hydrophobic antibacterial agents in their outer cell wall membrane, which do not effectively infiltrate the outer membrane of such bacteria. However, they can penetrate the outer membrane of specific mutants with a defective outer membrane or outer membrane damaged by chelators or polycationic compounds [16].
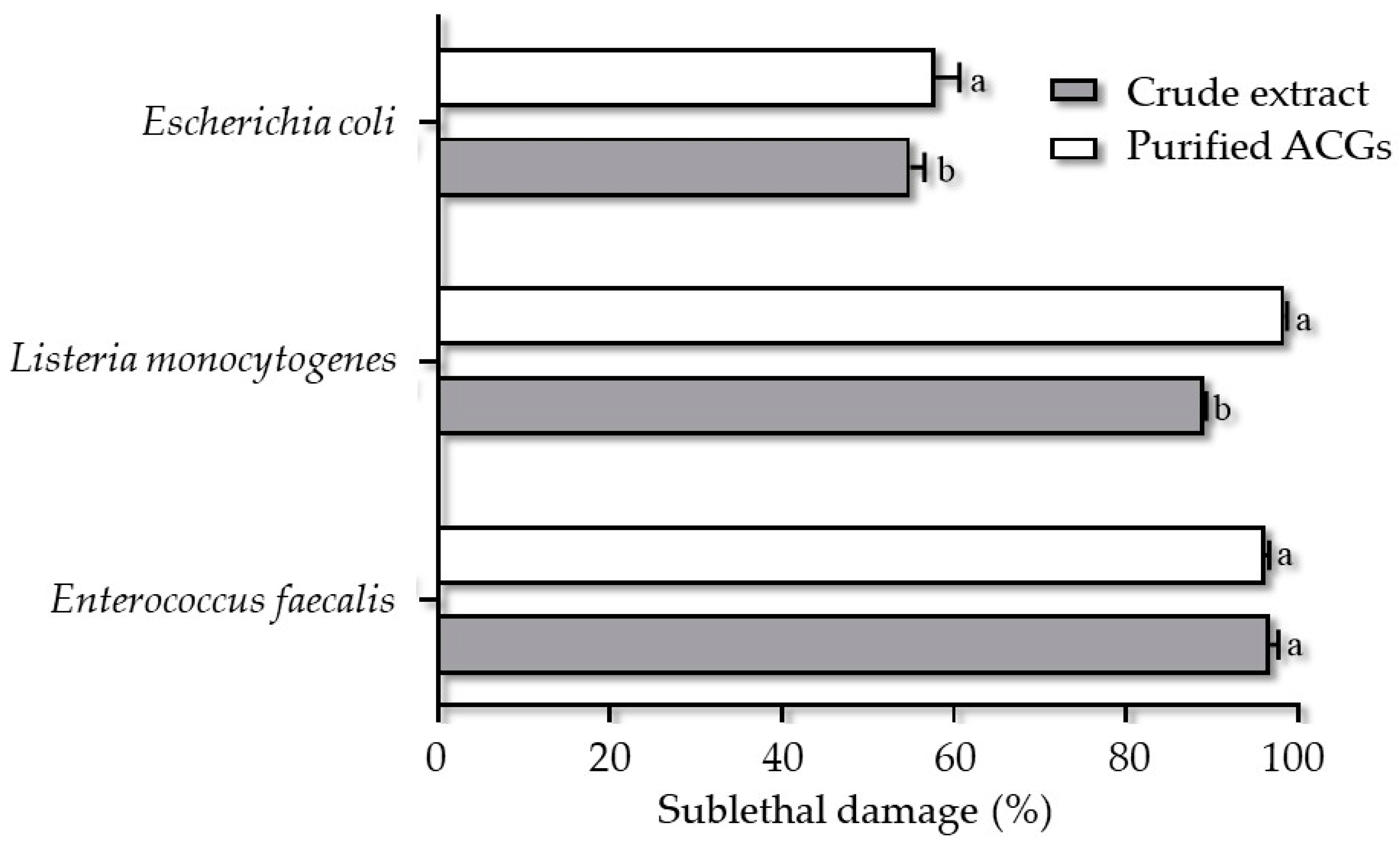
Sub-lethal injury is damage that leads to the inability of a microorganism to survive under stressful conditions and inhospitable environments without causing death. This lesion can be caused by structural (membrane permeability, cell wall disruption) or metabolic (functional components) factors [23,43]. The most significant sublethal lesion occurred when strains of L. monocytogenes (98.42%), E. faecalis (96.14%), and E. coli (57.78%) were treated with P-ACGs (Figure 4).
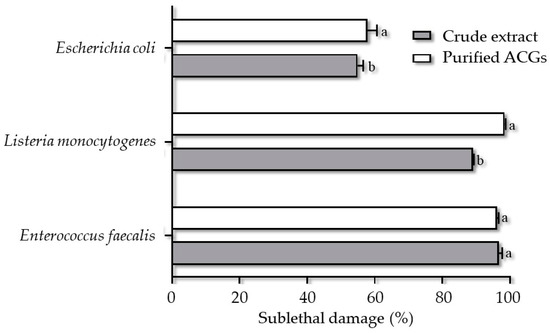
Figure 4.
Sublethal injury of crude extract acetogenins and purified acetogenins on Enterococcus faecalis, Listeria monocytogenes, and Escherichia coli. Different letters (a, b) indicate significant statistical differences between treatments (α = 0.05) by each concentration.
A complete inhibition of L. monocytogenes is described using P-ACGs [44], which agrees with the results obtained in this study. However, the CE showed an analogous activity to the P-ACGs on the E. faecalis strain. This behavior is not unusual, since there are reports of the sensitivity of this microorganism to the components of A. muricata. In this regard, Oyedeji et al. [39] showed that a fraction of an extract from A. muricata leaves had a 25% mortality rate on E. faecalis after 30 min of exposure using a volume of 4.5 mL of the extract. Likewise, Pinto et al. [16] found a bacteriostatic effect of a methanol extract of A. muricata leaves on the same strain.
4. Conclusions
In this study, it was observed that the CE, as well as the P-ACGs, showed a significant antibacterial effect against Gram-positive and Gram-negative bacteria. However, this effect was concentration and strain-dependent. Likewise, the most substantial effect was observed when P-ACGs were used against E. faecalis, L. monocytogenes, S. mutans, Klebsiella pneumoniae, A. hydrophila, B. cenocepacia, E. coli, and S. paratiphy. It was possible to observe that the MIC for all the evaluated strains ranged from 0.009 to 12.50 µg/mL. Flow cytometry further showed that the P-ACGs reduced bacterial abundance by affecting membrane integrity. In addition, a logarithmic reduction (0.38–1.81 CFU/mL) and a significant sub-lethal lesion (57.78–98.42%) were observed against E. coli, E. faecalis, and L. monocytogenes. P-ACGs from A. muricata seeds are an excellent alternative with strong antibacterial potential for future pharmaceutical applications.
Author Contributions
Conceptualization, G.A.-H., B.A.L.-R. and E.M.-G.; Methodology, G.A.-H., B.A.L.-R., L.M.A.-E., J.M.R.-G., A.P.-L. and E.M.-G., Validation, G.A.-H., V.M.P.-G., A.P.-L. and E.M.-G., Formal analysis, G.A.-H., I.C.-H., G.T.-I. and E.M.-G., Writing—original draft preparation, G.A.-H. and E.M.-G., Writing—review and editing, L.M.A.-E., I.C.-H., G.T.-I. and V.M.P.-G. Project administration, V.M.P.-G., A.P.-L. and E.M.-G., Funding acquisition, I.C.-H., G.T.-I., V.M.P.-G. and E.M.-G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported in part by funds provided by USDA-NIFA Sustainable Agriculture Systems with the project “Empowering U.S. Broiler Production for Transformation and Sustainability”. Grant No. 2019-69012-29905.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The dataset used and/or analyzed in the current study is available from the corresponding author upon reasonable request.
Acknowledgments
The authors thank CONACYT-Mexico for the postdoctoral granted to Gabriela Aguilar-Hernández (No. 590664) and the scholarship (No. 794251) awarded to Brandon A. López- Romero to carry out this project. This research is part of the activities of the National and International Networks “Anonáceas” and “Reduce and Valorize Food Losses and Waste Towards Sustainable Food Systems (294768)”.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Aguilar-Hernández, G.; Zepeda-Vallejo, L.G.; García-Magaña, M.L.; López-García, U.M.; Aguilera-Aguirre, S.; Montalvo-González, E. Acetogenin content from soursop fruit (Annona muricata L.) seeds: Evaluation of different extraction conditions. Biotecnia 2022, 24, 12–19. [Google Scholar] [CrossRef]
- Pinto, A.C.Q.; Cordeiro, M.C.R.; de Andrade, F.R.; Ferreira, H.A.; de Filgueiras, C.; Alves, D.E.; Kinpara, D.I. Annona Species; International Centre for Underutilized Crops, University of Southampton: Southampton, UK, 2005; pp. 1–284. [Google Scholar]
- Aguilar-Hernández, G.; García-Magaña, M.L.; Vivar-Vera, M.A.; Sáyago-Ayerdi, S.G.; Sánchez-Burgos, J.A.; Morales-Castro, J.; Montalvo-González, E. Optimization of ultrasound-assisted extraction of phenolic compounds from Annona muricata by-products and pulp. Molecules 2019, 24, 904. [Google Scholar] [CrossRef] [PubMed]
- Duran-Ruíz, C.A.; Cruz-Ortega, R.; Zaldívar-Riverón, A.; Zavaleta-Mancera, H.A.; de la Cruz-Chacón, I.; González-Esquinca, A.R. Ontogenic synchronization of Bephratelloides cubensis, Annona macroprophyllata seeds and acetogenins from Annonaceae. J. Med. Plant Res. 2019, 132, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Hernández, G.; Vivar-Vera, M.A.; García-Magaña, M.L.; González-Silva, N.; Pérez-Larios, A.; Montalvo-González, E. Ultrasound-assisted extraction of total acetogenins from the soursop fruit by response surface methodology. Molecules 2020, 25, 1139. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Fuentes, L.M.; Montalvo-González, E.; García-Magaña, M.L.; Anaya-Esparza, L.M.; Nolasco-González, Y.; Villagrán, Z.; González-Torres, S.; Velázquez-Monreal, J.J.; Morelos-Flores, D.A. Current situation and perspectives of fruit Annonaceae in Mexico: Biological and agronomic importance and bioactive properties. Plants 2022, 11, 7. [Google Scholar] [CrossRef]
- Abe, M.; Murai, M.; Ichimaru, N.; Kenmochi, A.; Yoshida, T.; Kubo, A.; Miyoshi, H. Dynamic function of the alkyl spacer of acetogenins in their inhibitory action with mitochondrial complex I (NADH-ubiquinone oxidoreductase). Biochemistry 2005, 44, 14898–14906. [Google Scholar] [CrossRef]
- Bermejo, A.; Figadére, B.; Zafra-Polo, M.C.; Barrachina, I.; Estornell, E.; Cortes, D. Acetogenins from Annonaceae: Recent progress in isolation, synthesis and mechanisms of action. Nat. Prod. Res. 2005, 22, 269–303. [Google Scholar] [CrossRef]
- Coria-Téllez, A.V.; Montalvo-González, E.; Yahia, E.M.; Obledo-Vázquez, E.N. Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity. Arab. J. Chem 2016, 11, 662–691. [Google Scholar] [CrossRef]
- Agu, K.C.; Okolie, N.P.; Falodun, A.; Engel-Lutz, N. In vitro anticancer assessments of Annona muricata fractions and in vitro antioxidant profile of fractions and isolated acetogenin (15-acetyl guanacone). J. Cancer Res. Pract. 2018, 5, 53–66. [Google Scholar] [CrossRef]
- León-Fernández, A.E.; Martínez, L.; Zepeda-Vallejo, L.G.; Arteaga-Garibay, R.I.; Gutiérrez-Martínez, P.; Montalvo-González, E. Antibacterial, antifungal, antioxidant and toxic effect of fractioned extracts from Soursop pulp. Bio Ciencias 2019, 6, e400. [Google Scholar] [CrossRef]
- Terezan, A.P.; Joáo, G.M.; Junqueira, V.G.; Wakui, L.K.; Oliveira, A.; Martins, C.E.; Santiago, M.B.; Severino, V.P. Qualitative analysis of acetogenins from Annona coriaceae leaves (Annonaceae) by HPLC-Q-Orbitrap and their antibacterial potential against oral pathogens. Nat. Prod. Res. 2020, 36, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Villalva, R.; Molina, G.A.; España-Sánchez, B.L.; Díaz-Peña, L.F.; Elizalde-Mata, A.; Valerio, E.; Estevez, M. Antioxidant capacity and antibacterial activity from Annona cherimola phytochemicals by ultrasound-assisted extraction and its comparison to conventional methods. Arab. J. Chem. 2021, 14, 103239. [Google Scholar] [CrossRef]
- Indrianingsih, A.W.; Wulanjati, M.P.; Windarsih, A.; Bhattacharjya, D.K.; Suzuki, T.; Katayama, T. In vitro studies of antioxidant, antidiabetic, and antibacterial activities of Theobroma cacao, Anonna muricata and Clitoria ternatea. Biocatal. Agric. Biotechnol. 2021, 33, 101995. [Google Scholar] [CrossRef]
- Falagas, M.E.; Kyriakidou, M.; Voulgaris, G.L.; Vokos, F.; Politi, S.; Kechagias, K.S. Clinical use of intravenous polymyxin B for the treatment of patients with multidrug-resistant Gram-negative bacterial infections: An evaluation of the current evidence. J. Glob. Antimicrob. Resist. 2021, 24, 342–359. [Google Scholar] [CrossRef] [PubMed]
- Pinto, N.C.C.; Campos, L.M.; Evangelista, A.C.S.; Lemos, A.S.O.; Silva, T.P.; Melo, R.C.N.; Fabri, R.L. Antimicrobial Annona muricata L. (soursop) extract targets the cell membranes of Gram-positive and Gram-negative bacteria. Ind. Crops Prod. 2017, 107, 332–340. [Google Scholar] [CrossRef]
- Zafra-Polo, M.C.; González, M.C.; Estornell, E.; Sahpaz, S.; Cortes, D. Acetogenins from annonaceae, inhibitors of mitochondrial complex I. Phytochemistry 1996, 42, 253–271. [Google Scholar] [CrossRef]
- Raybaudi-Massilia, R.; Suárez, A.I.; Arvelo, F.; Sojo, F.; Mosqueda-Melgar, J.; Zambrano, A.; Calderón-Gabaldón, M.I. An analysis in vitro of the cytotoxic, antioxidant and antimicrobial activity of aqueous and alcoholic extracts of Annona muricata L. seed and pulp. Br. J. Appl. Sci. Technol. 2015, 5, 333. [Google Scholar] [CrossRef]
- Neglo, D.; Tettey, C.O.; Essuman, E.K.; Amenu, J.D.; Mills-Robertson, F.C.; Sedohia, D.; Adjoa, A.B.; Abaye, D.A. Evaluation of the modulatory effect of Annona muricata extracts on the activity of some selected antibiotics against biofilm-forming MRSA. Evid. Based Complement. Altern. Med. 2021, 2021, 9342110. [Google Scholar] [CrossRef]
- Hoe, P.K.; Yiu, P.H.; Ee, G.C.L.; Wong, S.C.; Rajan, A.; Bong, C.F. Biological activity of Annona muricata seed extracts. Malays. J. Sci. 2010, 29, 153–159. [Google Scholar] [CrossRef]
- López-Romero, B.A.; Luna-Bárcenas, G.; García-Magaña, M.L.; Anaya-Esparza, L.M.; Zepeda-Vallejo, L.G.; López-García, U.M.; Ortiz-Basurto, R.I.; Aguilar-Hernández, G.; Pérez-Larios, A.; Montalvo-González, E. Extraction of Acetogenins Using Thermosonication-Assisted Extraction from Annona muricata Seeds and Their Antifungal Activity. Molecules 2022, 27, 6045. [Google Scholar] [CrossRef]
- Yang, H.J.; Li, X.; Zhang, N.; Chen, J.W.; Wang, M.Y. Two new cytotoxic acetogenins from Annona squamosa. J. Asian Nat. Prod. Res. 2009, 11, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Anaya-Esparza, L.M.; Montalvo-González, E.; González-Silva, N.; Méndez-Robles, M.; Romero-Toledo, R.; Yahia, E.; Pérez-Larios, A. Synthesis and characterization of TiO2-ZnO-MgO mixed oxide and their antibacterial activity. Materials 2019, 12, 698. [Google Scholar] [CrossRef]
- Parhusip, A.J.N.; Sitanggang, A.B. Antimicrobial activity of melinjo (Gnetum gnemon) seed and peel extract against selected pathogenic bacteria. Microbiol. Indones. 2011, 5, 103–112. [Google Scholar] [CrossRef]
- Bibi, Y.; Nisa, S.; Chaudhary, F.M.; Zia, M. Antibacterial activity of some selected medicinal plants from Pakistan. BMC Complement. Altern. Med. 2011, 11, 52. [Google Scholar] [CrossRef] [PubMed]
- Khater, M.; Khater, S.S.; Gholap, H.; Patil, R.; Kulkarni, G. Comparative studies on measurement of membrane potential of bacterial cells treated with ZnO nanoparticles by Spectrofluorometry, fluorescence microscopy and flowcytometry. J. Microbiol. Methods 2020, 173, 105920. [Google Scholar] [CrossRef] [PubMed]
- Anaya-Esparza, L.M.; González-Silva, N.; Yahia, E.M.; González-Vargas, O.A.; Montalvo-González, E.; Pérez-Larios, A. Effect of TiO2-ZnO-MgO mixed oxide on microbial growth and toxicity against Artemia salina. Nanomaterials 2019, 9, 992. [Google Scholar] [CrossRef]
- Trindade, M.L.; Martins, D.A.; Radünz, M.; Ramos, A.; Silveira, C.; Silva, G.; Avila, E.; Helbig, E. Chemical characterization, antimicrobial and antioxidant activity of Annona (Annona squamosa L.) pulp extract. Chil. J. Nutr. 2022, 47, 281–285. [Google Scholar] [CrossRef]
- Rodriguez-Pérez, J.L.; Millones-Gómez, P.A. Antibacterial effect of Annona muricata L. leaves on Streptococcus mutans ATCC 25175 strains. J. Clin. Diagn. Res. 2019, 13, 1–16. [Google Scholar] [CrossRef]
- Masato, A.; Kubo, A.; Yamamoto, S.; Hatoh, Y.; Murai, M.; Hattori, Y. Dynamic function of the spacer region of acetogenins in the inhibition of bovine mitocondrial NADH-ubiquinone oxidoreductase (complex I). Biochemistry 2008, 47, 6260–6266. [Google Scholar] [CrossRef]
- Rowlett, V.W.; Mallampalli, V.K.P.S.; Karlstaedt, A.; Dowhan, W.; Taegtmeyer, H.; Margolin, W.; Vitrac, H. Impact of membrane phospholipid alterations in Escherichia coli on cellular function and bacterial stress adaptation. J. Bacteriol. 2017, 199, 1–50. [Google Scholar] [CrossRef]
- Campos, L.M.; Lemos, A.S.O.; Silva, T.P.; Melo, R.M.N.; Apolônio, A.C.M.; Fabri, R.L. Antibacterial activity of ethanolic extract from Annona squamosa leaves against Gram-positive and Gram-negative bacteria. Glob. J. Pathol. Microbiol. 2021, 9, 1–9. [Google Scholar] [CrossRef]
- Ríos, J.L.; Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef] [PubMed]
- Obum-Nnadi, C.N.; Ezenwa, C.M.; Amaechi, D.; Ohabughiro, N.B.; Nnagbo, P.A.; Nwokorie, K.S.; Okoli, C.S. Evaluation of the Antimicrobial and Phytochemical Properties of Annona muricata L. (Soursop). J. Biomed. Biosens. 2022, 2, 43–59. Available online: https://www.gpub.org/ (accessed on 29 October 2022).
- Da Silva, B.D.; Patricia Bernardes, P.; Pinheiro, P.F.; Giorgio, J.D.; Domenici, G.C. Plectranthus amboinicus (Lour.) Spreng. essential oil as a natural alternative for the conservation of beef patties stored under refrigeration. Food Biosci. 2022, 49, 101896. [Google Scholar] [CrossRef]
- Siderakou, D.; Zilelidou, E.; Poimenidou, S.; Tsipra, I.; Ouranou, E.; Papadimitriou, K.; Skandamis, P. Assessing the survival and sublethal injury kinetics of Listeria monocytogenes under different food processing-related stresses. Int. J. Food Microbiol. 2021, 346, 109–159. [Google Scholar] [CrossRef] [PubMed]
- Salinas-Salazar, C.; Hernández-Brenes, C.; Rodríguez-Sánchez, D.G.; Castillo, E.C.; Navarro-Silva, J.M.; Pacheco, A. Inhibitory activity of avocado seed fatty acid derivatives (acetogenins) against Listeria monocytogenes. J. Food Sci. 2016, 82, 134–144. [Google Scholar] [CrossRef]
- Vairappan, C.S.; Tan, K.L. C15 halogenated acetogenin with antibacterial activity against food pathogens. Malays. J. Sci. 2009, 28, 263–268. [Google Scholar] [CrossRef]
- Oyedeji, O.; Taiwo, F.O.; Ajayi, O.S.; Ayinde, F.; Oziegbe, M.; Oseghare, C.O. Biocidal and Phytochemical analysis of leaf extracts of Annona muricata (Linn.). Int. J. Sci. Basic Appl. Res. 2015, 24, 76–87. [Google Scholar]
- Rottenberg, H.; Wu, S. Quantitative assay by flow cytometry of the mitochondrial membrane potential in intact cells. Biochim. Biophys. Acta Mol. Cell Res. 1998, 1404, 393–404. [Google Scholar] [CrossRef]
- Sánchez-Torres, L.E.; Espinosa-Bonilla, A.; Diosdado-Vargas, F. Flow cytometry, a universe of possibilities in the veterinary field. Review. J. Livest. Sci. 2022, 13, 763–786. [Google Scholar]
- Gutiérrez-del-Río, I.; Fernández, J.; Lombó, F. Plant nutraceuticals as antimicrobial agents in food preservation: Terpenoids, polyphenols and thiols. Int. J. Antimicrob. Agents 2018, 52, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Kethireddy, V.; Oey, I.; Jowett, T.; Bremer, P. Critical analysis of the maximum non-inhibitory concentration (MNIC) method for quantifying sublethal lesions in Saccharomyces cerevisiae cells exposed to heat or pulsed electric field treatments. Int. J. Food Microbiol. 2016, 233, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Villarreal-Lara, R.; Rodríguez-Sánchez, D.G.; Díaz De La Garza, R.I.; García-Cruz, M.I.; Castillo, A.; Pacheco, A.; Hernández-Brenes, C. Purified avocado seed acetogenins: Antimicrobial spectrum and complete inhibition of Listeria monocytogenes in a refrigerated food matrix. CyTA J. Food 2019, 17, 228–239. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).