Automatic Breast Tumor Screening of Mammographic Images with Optimal Convolutional Neural Network
Abstract
:1. Introduction
2. Materials and Methods
2.1. Design of the Multilayer Deep-Learning-Based CNN
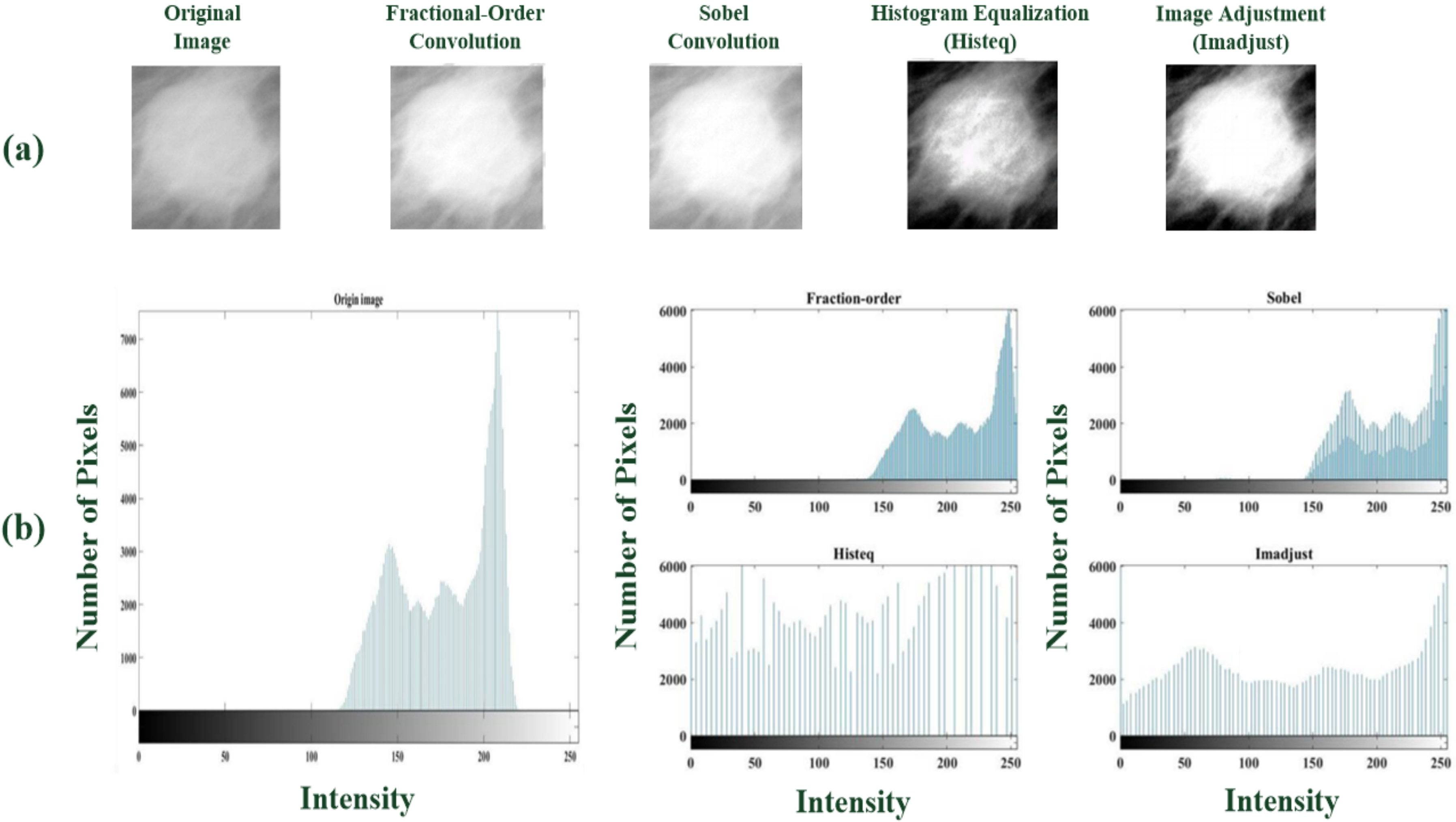
- Feature enhancement and extraction: A multilayer 2D convolution operation is used to magnify the texture of what might be tumor tissue and edge information (usually two or more layers are used), as shown in Figure 2a. Each layer uses a 3 × 3 sliding window to perform the operation of the convolutional weight. First, a 2D fractional convolution operation is performed to magnify the tumor characteristics. Then, by combining multilayer convolutional weight calculations, the contour of the tumor is gradually strengthened, noise is removed, and the image is sharpened. These effects help strengthen the target area and retain non-characteristic information. This study applies the 2D spatial fractional-order convolutional processes in the fractional convolutional layer, selects the appropriate fractional order parameters, and performs convolution in the x and y directions, thus yielding a combination of 2D weight values in space, the general formula being [35,36,37,38]:where h = 3 is the dimension of the convolution window, v is a fractional parameter and v ∈ (0, 1), and I(x, y) ∈ [0, 255] is the pixel value at point (x, y) in a 2D image. Each fractional-order convolutional mask multiplies each element, M(i, j) or M(j, i), by the corresponding input pixel values, I(x, y), and then obtains an enhanced feature pattern containing spatial features in the x-axis and y-axis directions. These 2D spatial convolutional processes act as two low-pass frequency filters [39] and then remove the high-spatial-frequency components from a breast mammogram. In this study, the image dimension is n × n, x = 1, 2, 3, …, n, and y = 1, 2, 3, …, n. Mx and My are 3 × 3 convolutional windows that can be written as follows [35,36,37,38]:where v ∈ (0, 1) is the fractional-order parameter. A sliding stride = 1 is selected for spatial domain-based convolution operations in the horizontal and vertical directions. The results of the convolution operation of (1) and (2) are combined and normalized, and the approximate formula is written below:
- Rapid screening of breast tumors: Breast tumors can be identified at the image classification layer, which includes the flattening process (FP) and a multilayer classifier, as seen in Figure 1. The FP can convert a 2D feature matrix into a 1D feature vector, which is then fed as the input vector of the classifier for further pattern recognition. After two MP treatments, the FP treatment may be written as shown in the general formula (7):where X is the 1D feature vector of the multilayer classifier used as input. In this study, the multilayer classifier includes an input layer, two hidden layers (i.e., the first and second hidden layers), and an output layer.
2.2. Adaptive Moment Estimation Method
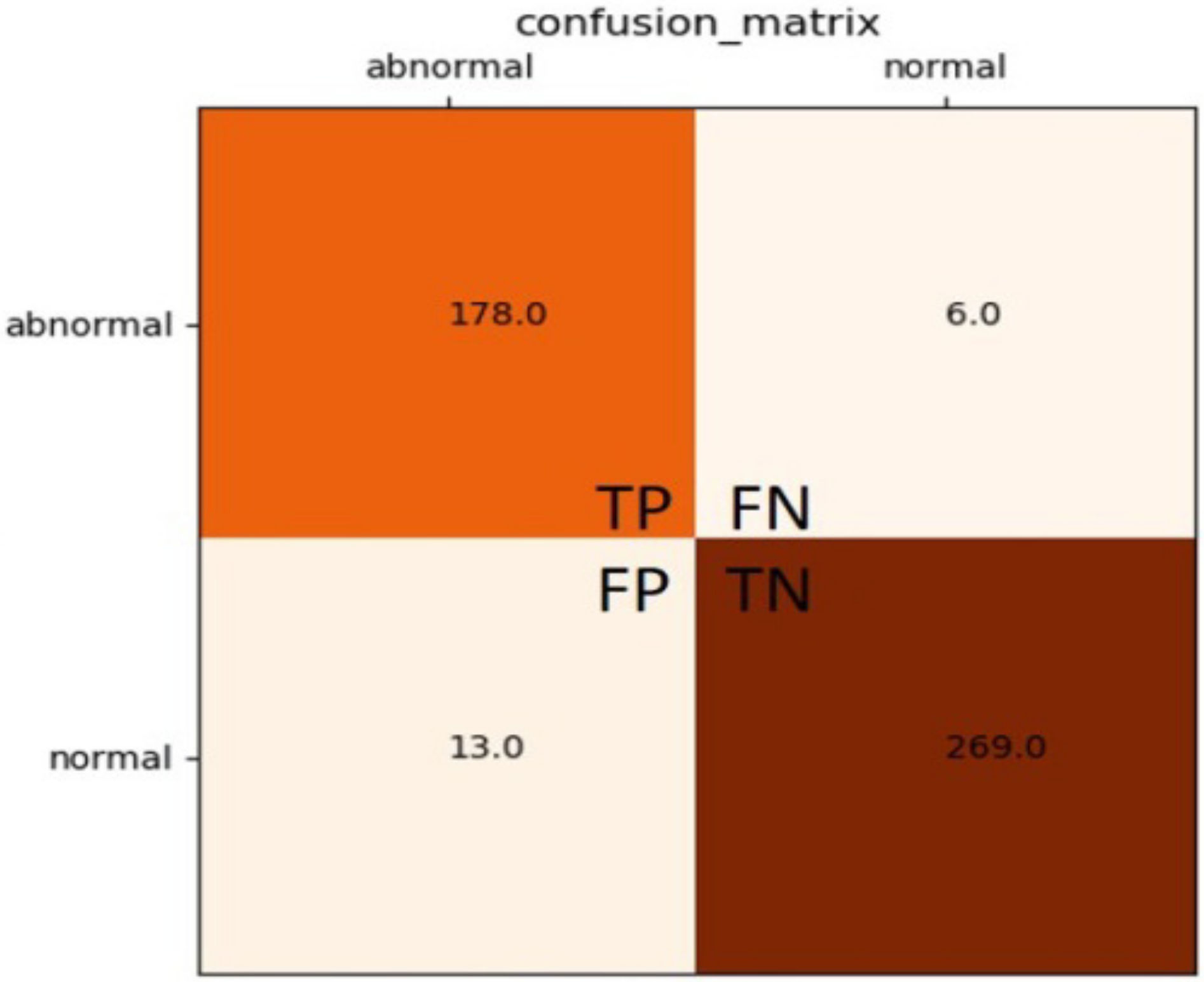
2.3. Classifier’s Performance Evaluations
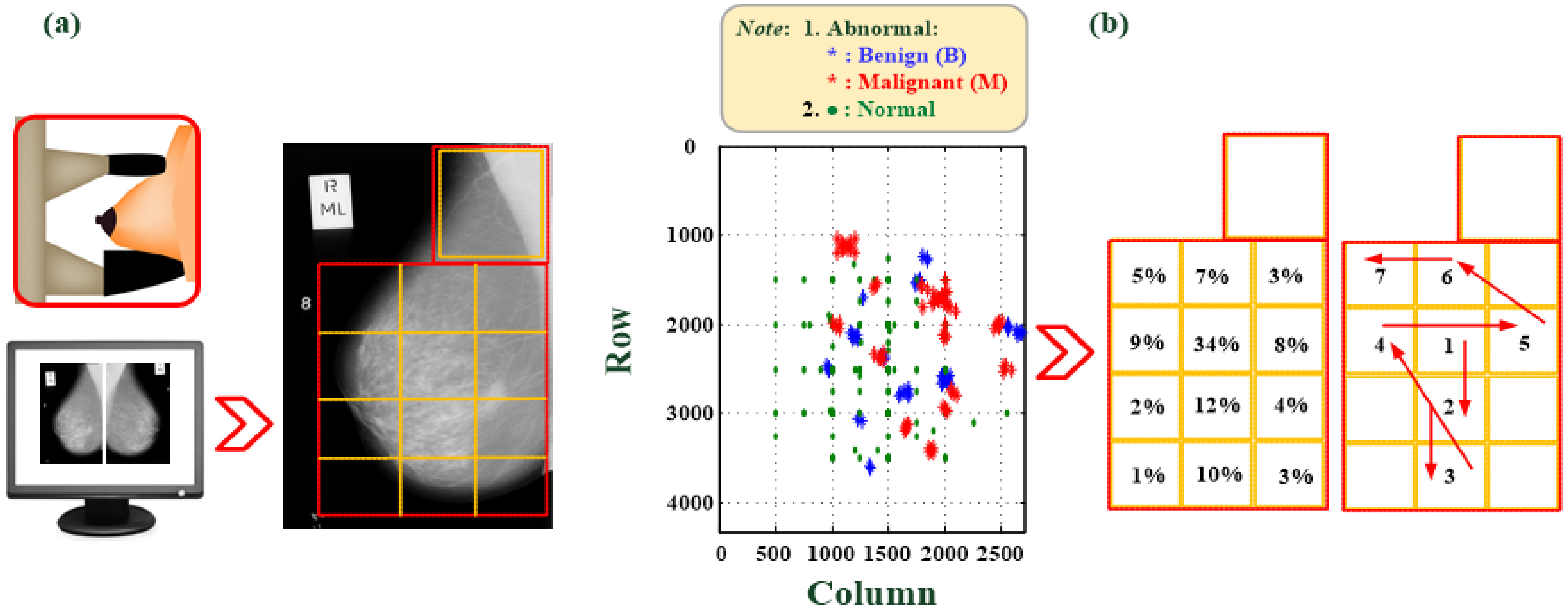
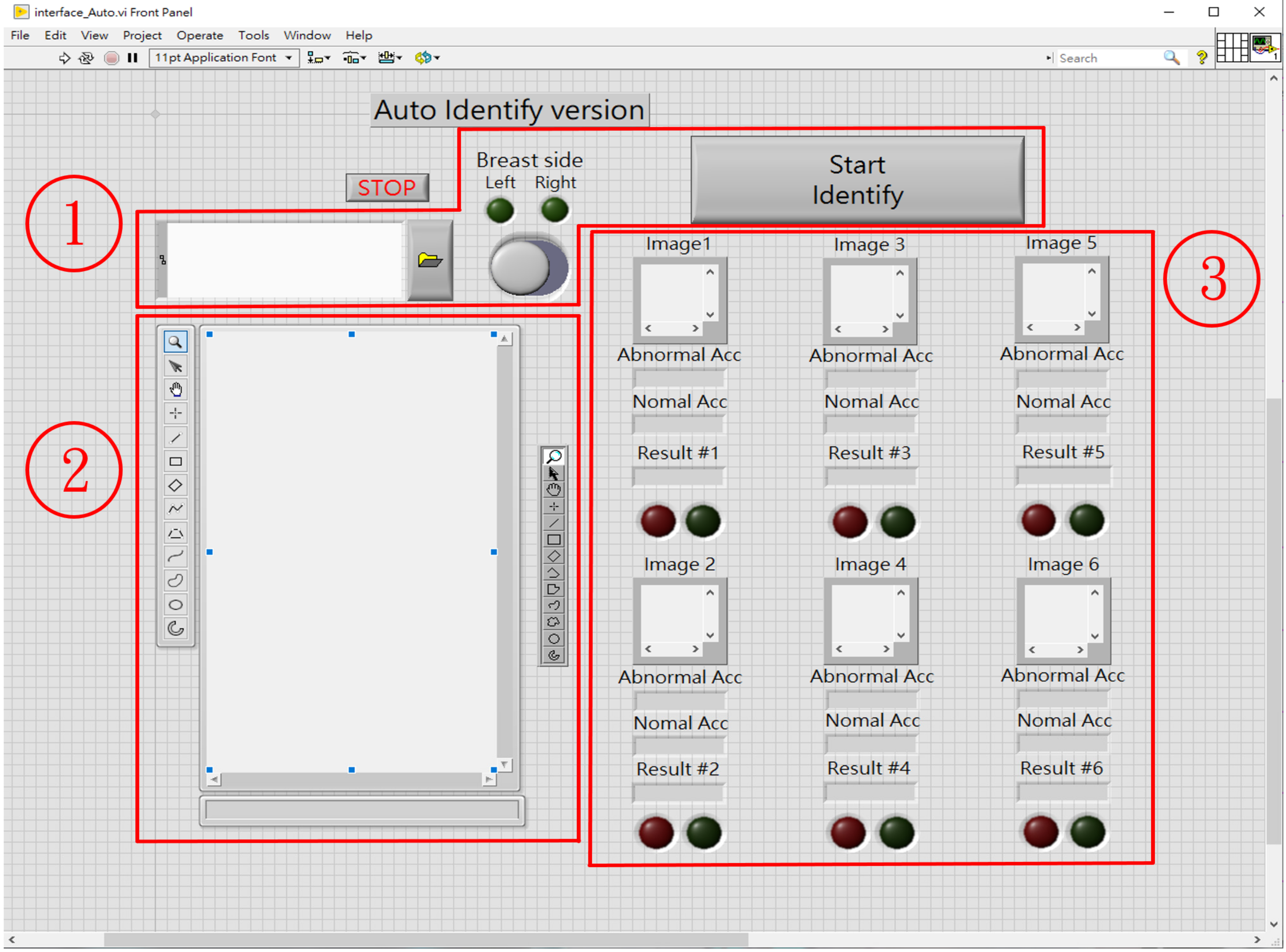
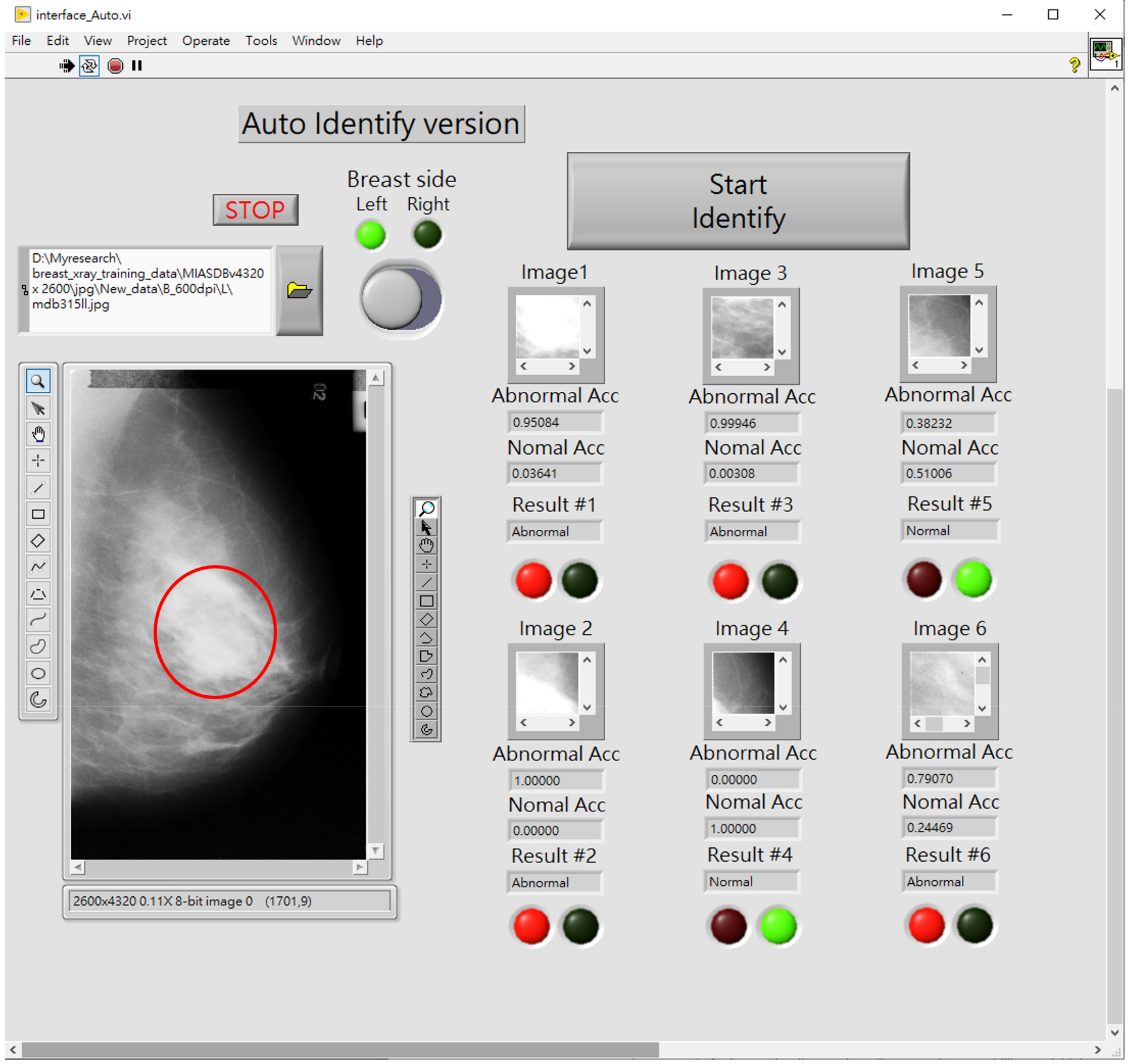
2.4. Computer Assistive System for Automatic Breast Tumor Screening
- Zone ①: Sets the source path of breast mammography images;
- Zone ②: Loads and displays the selected mammography images;
- Zone ③: As per the priority order, extract ROI images and perform automatic tumor screening. In this study, six areas at which tumors are most possibly identified are designated. The CAS automatically prioritizes the ROI cutting feature patterns (100 pixels × 100 pixels), as seen in Figure 2, and then screens those areas. The block marked ③ can show the output of the classifier, the identification result, and the classification information. The red and green circles show the normality and abnormality. The output value of the classifier must be >0.5 to have a high degree of confidence that there is a suspected breast tumor.
2.5. Experimental Setup
3. Experimental Results
- The number of convolutional layers and pooling layers: This study increases the number of convolutional layers and pooling layers from 1 to 5 and the sizes of convolution windows from 3 × 3 to 11 × 11. The processing windows for the pooling layers are set to 2 × 2, and the second to fifth convolutional layers have 16 kernel convolution windows to perform feature enhancement and extraction.
3.1. Testing of Different Multilayer CNN Models and Determination of the Most Suitable Architecture
3.2. Testing of the First Convolutional Layer and Determination of the Window Type
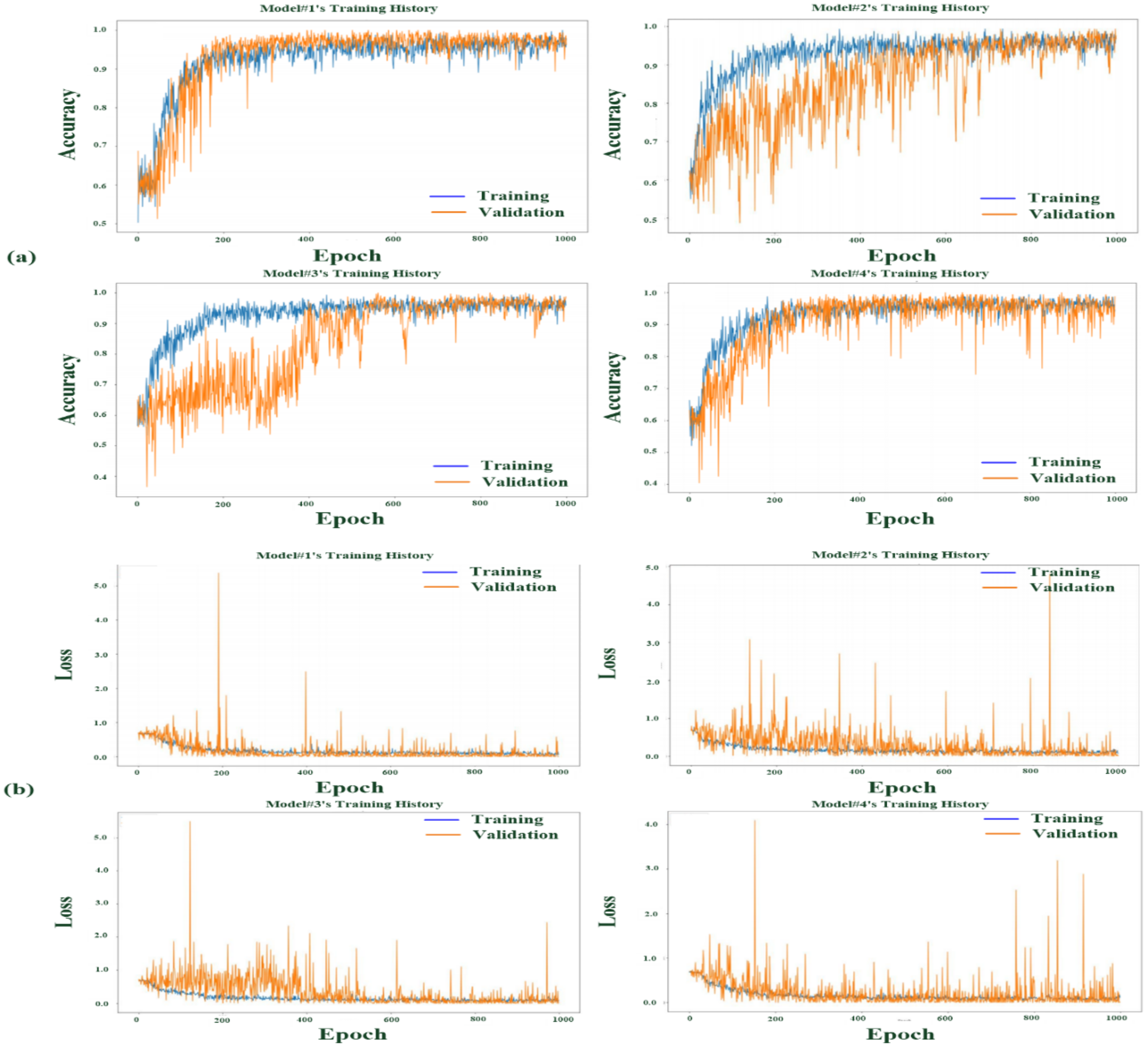
3.3. Multilayer CNN-Based Classifier Testing and Validation
3.4. Discussion
- The ROI extraction, image enhancement, and feature classification tasks are integrated into one learning model;
- The fractional-order convolutional process with fractional-order parameter, v = 0.30–0.40, is used to extract the tumor edges in the first convolutional layer; subsequently, two kernel convolution processes are used to extract the tumor shapes;
- The ADAM algorithm is easy to implement and operate with large datasets and parameter adjustment;
- The proposed CNN-based classifier has better classification accuracy than the CNN architecture with Sobel and Histeq convolutional windows in the first convolutional layer.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CNN | Convolutional Neural Network |
| CBIS-DDSM | Curated Breast Imaging Subset of a Digital Database for Screening Mammography |
| BC | Breast Cancer |
| AI | Artificial Intelligence |
| BD | Big Data |
| MRI | Magnetic Resonance Imaging |
| CT | Computer Tomography |
| ROI | Region of Interest |
| 2D | Two-Dimensional |
| 1D | One-Dimensional |
| ML | Machine Learning |
| MP | Maximum-Pooling |
| FP | Flattening Process |
| MLP | Multilayer Perceptron |
| GPU | Graphics Processing Unit |
| ADAM | Adaptive Moment Estimation Method |
| GeLU | GeLU |
| MIAS | Mammographic Image Analysis Society |
| SuReMapp | Suspicious Region Detection on Mammogram from PP |
| TP | True Positive |
| FP | False Positive |
| TN | True Negative |
| FN | False Negative |
| PPV | Positive Predictive Value |
| YI | Youden’s Index |
| Sens | Sensitivity |
| Spec | Specificity |
| B | Benign |
| M | Malignant |
References
- Ministry Health and Welfare, Taiwan. 2020 Cause of Death Statistics. 2021. Available online: https://dep.mohw.gov.tw/dos/lp-1800-113.html (accessed on 1 January 2022).
- Tsui, P.-H.; Liao, Y.-Y.; Chang, C.-C.; Kuo, W.-H.; Chang, K.-J.; Yeh, C.-K. Classification of benign and malignant breast tumors by 2-D analysis based on contour description and scatterer characterization. IEEE Trans. Med. Imaging 2010, 29, 513–522. [Google Scholar] [CrossRef] [PubMed]
- Kallenberg, M.; Petersen, K.; Nielsen, M.; Ng, A.Y.; Diao, P.; Igel, C.; Vachon, C.M.; Holland, K.; Winkel, R.R.; Karssemeijer, N.; et al. Unsupervised deep learning applied to breast density segmentation and mammographic risk scoring. IEEE Trans. Med. Imaging 2016, 35, 1322–1331. [Google Scholar] [CrossRef] [PubMed]
- Samala, R.K.; Chan, H.; Hadjiiski, L.; Helvie, M.A.; Richter, C.D.; Cha, K.H. Breast cancer diagnosis in digital breast tomosynthesis: Effects of training sample size on multi-stage transfer learning using deep neuralnets. IEEE Trans. Med. Imaging 2019, 38, 686–696. [Google Scholar] [CrossRef] [PubMed]
- Valkonen, M.; Isola, J.; Ylinen, O.; Muhonen, V.; Saxlin, A.; Tolonen, T.; Nykter, M.; Ruusuvuori, P. Cytokeratin-supervised deep learning for automatic recognition of epithelial cells in breast cancers stained for ER, PR, and Ki-67. IEEE Trans. Med. Imaging 2020, 39, 534–542. [Google Scholar] [CrossRef]
- Lee, S.; Kim, H.; Higuchi, H.; Ishikawa, M. Classification of metastatic breast cancer cell using deep learning approach. In Proceedings of the 2021 International Conference on Artificial Intelligence in Information and Communication (ICAIIC), Jeju Island, Korea, 13–16 April 2021; pp. 425–428. [Google Scholar]
- Chougrad, H.; Zouaki, H.; Alheyane, O. Deep convolutional neural networks for breast cancer screening. Comput. Methods Programs Biomed. 2018, 157, 19–30. [Google Scholar] [CrossRef]
- Jia, G.; Lam, H.-K.; Althoefer, K. Variable weight algorithm for convolutional neural networks and its applications to classification of seizure phases and types. Pattern Recognit. 2021, 121, 108226. [Google Scholar] [CrossRef]
- Li, X.; Zhai, M.; Sun, J. DDCNNC: Dilated and depthwise separable convolutional neural network for diagnosis COVID-19 via chest X-ray images. Int. J. Cogn. Comput. Eng. 2021, 2, 71–82. [Google Scholar] [CrossRef]
- University of South Florida. DDSM: Digital Database for Screening Mammography, Version 1 (Updated 2017/09/14); University of South Florida: Tampa, FL, USA; Available online: http://www.eng.usf.edu/cvprg/Mammography/Database.html (accessed on 1 January 2022).
- McGarthy, N.; Dahlan, A.; Gook, T.S.; Hare, N.O.; Ryan, M.; John, B.S.; Lawlor, A.; Gurran, K.M. Enterprise imaging and big data: A review from a medical physics perspective. Phys. Med. 2021, 83, 206–220. [Google Scholar] [CrossRef]
- Yaffe, M.J. Emergence of big data and its potential and current limitations in medical imaging. Semin. Nucl. Med. 2019, 49, 94–104. [Google Scholar] [CrossRef]
- The European Federation of Organisations for Medical Physics (EFOMP). White Paper: Big data and deep learning in medical imaging and in relation to medical physics profession. Phys. Med. 2018, 56, 90–93. [Google Scholar] [CrossRef] [Green Version]
- Diaz, O.; Kushibar, K.; Osuala, R.; Linardos, A.; Garrucho, L.; Igual, L.; Radeva, P.; Prior, F.; Gkontra, P.; Lekadir, K. Data preparation for artificial intelligence in medical imaging: A comprehensive guide to open-access platforms and tools. Phys. Med. 2021, 83, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Y.; Lu, J. A visualization algorithm for medical big data based on deep learning. Measurement 2021, 183, 109808. [Google Scholar] [CrossRef]
- Saranya, N.; Priya, S.K. Deep convolutional neural network feed-Forward and back propagation (DCNN-FBP) algorithm for predicting heart disease using internet of things. Int. J. Eng. Adv. Technol. 2021, 11, 283–287. [Google Scholar]
- Zhang, J.; Qu, S. Optimization of backpropagation neural network under the adaptive genetic algorithm. Complexity 2021, 2021, 1718234. [Google Scholar] [CrossRef]
- Sadad, T.; Munir, A.; Saba, T.; Hussain, A. Fuzzy C-means and region growing based classification of tumor from mammograms using hybrid texture feature. J. Comput. Sci. 2018, 29, 34–45. [Google Scholar] [CrossRef]
- Comelli, A.; Bruno, A.; Di Vittorio, M.L.; Ienzi, F.; Legalla, R.; Vitabile, S.; Ardizzone, E. Automatic multi-seed detection for MR breast image segmentation. Int. Conf. Image Anal. Process. 2017, 10484, 706–717. [Google Scholar]
- Lindquist, E.M.; Gosnell, J.M.; Khan, S.K.; Byl, J.L.; Zhou, W.; Jiang, J.; Vettukattilb, J.J. 3D printing in cardiology: A review of applications and roles for advanced cardiac imaging. Ann. 3D Print. Med. 2021, 4, 100034. [Google Scholar] [CrossRef]
- Drozdzal, M.; Chartrand, G.; Vorontsov, E.; Shakeri, M.; Di Jorio, L.; Tang, A.; Romero, A.; Bengio, Y.; Pal, C.; Kadoury, S. Learning normalized inputs for iterative estimation in medical image segmentation. Med. Image Anal. 2018, 44, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Racz, A.; Bajusz, D.; Heberger, K. Multi-level compaeison of machine learning classifier and thrir performance metrics. Molecules 2019, 24, 2811. [Google Scholar] [CrossRef] [Green Version]
- Allen, J.; Liu, H.; Iqbal, S.; Zheng, D.; Stansby, G. Deep learning-based photoplethysmography classification for peripheral arterial disease detection: A proof-of-concept study. Physiol. Meas. 2021, 42, 054002. [Google Scholar] [CrossRef]
- Panwar, M.; Gautam, A.; Dutt, R.; Acharyya, A. CardioNet: Deep learning framework for prediction of CVD risk factors. In Proceedings of the 2020 IEEE International Symposium on Circuits and Systems (ISCAS), Seville, Spain, 12–14 October 2020; pp. 1–5. [Google Scholar]
- Krizhevsky, A.; Sutskever, I.; Hinton, G.E. ImageNet Classi-fication with Deep Convolutional Neural Networks. Commun. ACM 2017, 60, 84–90. [Google Scholar] [CrossRef]
- Zeiler, M.D.; Fergus, R. Visualizing and understanding convolutional networks. In Lecture Notes in Computer Science; 8689 LNCS; Springer: Cham, Switzerland, 2014; pp. 818–833. [Google Scholar]
- Deng, J.; Dong, W.; Socher, R.; Li, L.-J.; Li, K.; Li, F.-F. ImageNet: A large-scale hierarchical image database. In Proceedings of the 2009 IEEE Conference on Computer Vision and Pattern Recognition, Miami, FL, USA, 20–25 June 2009; pp. 248–255. [Google Scholar]
- Li, Y.-C.; Shen, T.-Y.; Chen, C.-C.; Chang, W.-T.; Lee, P.-Y.; Huang, C.-C. Automatic detection of atherosclerotic plaque and calcification from intravascular ultrasound Images by using deep convolutional neural networks. IEEE Trans. Ultrason. Ferroelectr. Freq. Control 2021, 68, 1762–1772. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Wang, Z.; Kuen, J.; Ma, L.; Shahroudy, A.; Shuai, B.; Liu, T.; Wang, X.; Wang, G.; Cai, J.; et al. Recent advances in convolutional neural network. Pattern Recognit. 2018, 77, 354–377. [Google Scholar] [CrossRef] [Green Version]
- Kiranyaz, S.; Avci, O.; Abdeljaber, O.; Ince, T.; Gabbouj, M.; Inman, D.J. 1D convolutional neural networks and applications: A survey. Mech. Syst. Signal Process. 2021, 151, 107s398. [Google Scholar] [CrossRef]
- Kingma, D.P.; Ba, J. Adam: A method for stochastic optimization. In Proceedings of the 3rd International Conference for Learning Representations, San Diego, CA, USA, 7–9 May 2015. [Google Scholar]
- Ma, J.; Yarats, D. Quasi-hyperbolic momentum and Adam for deep learning. In Proceedings of the International Conference on Learning Representations, New Orleans, LA, USA, 6–9 May 2019. [Google Scholar] [CrossRef]
- Pilot European Image Processing Archive. The Mini-MIAS Database of Mammograms. 2012. Available online: http://peipa.essex.ac.uk/pix/mias/ (accessed on 1 January 2022).
- Mammographic Image Analysis Society (MIAS). Database v1.21. 2019. Available online: https://www.repository.cam.ac.uk/handle/1810/250394 (accessed on 1 January 2022).
- Wu, J.-X.; Chen, P.-Y.; Li, C.-M.; Kuo, Y.-C.; Pai, N.-S.; Lin, C.-H. Multilayer fractional-order machine vision classifier for rapid typical lung diseases screening on digital chest X-ray images. IEEE Access 2020, 8, 105886–105902. [Google Scholar] [CrossRef]
- Lin, C.-H.; Wu, J.-X.; Li, C.-M.; Chen, P.-Y.; Pai, N.-S.; Kuo, Y.-C. Enhancement of chest X-ray images to improve screening accuracy rate using iterated function system and multilayer fractional-order machine learning classifier. IEEE Photonics J. 2020, 12, 1–19. [Google Scholar] [CrossRef]
- Pu, Y.-F.; Zhou, J.-L.; Yuan, X. Fractional differential mask: A fractional differential-based approach for multiscale texture enhancement. IEEE Trans. Image Process. 2010, 19, 491–511. [Google Scholar]
- Zhang, Y.; Pu, Y.-F.; Zhou, J.-L. Construction of fractional differential masks based on Riemann-Liouville definition. J. Comput. Inf. Syst. 2010, 6, 3191–3199. [Google Scholar]
- Thanh, D.N.H.; Kalavathi, P.; Thanh, L.T.; Prasath, V.B.S. Chest X-ray image denoising using Nesterov optimization method with total variation regularization. Procedia Comput. Sci. 2020, 171, 1961–1969. [Google Scholar] [CrossRef]
- Ba, L.J.; Frey, B. Adaptive dropout for training deep neural networks. Adv. Neural Inf. Process. Syst. 2013, 26, 1–9. [Google Scholar]
- Clevert, D.; Unterthiner, T.; Hochreite, S. Fast and accurate deep network learning by exponential linear units (ELUs). In Proceedings of the 4th International Conference on Learning Representations, San Juan, Puerto Rico, 2–4 May 2016. [Google Scholar]
- Hendrycks, D.; Gimpel, K. Gaussian Error Linear Units (GELUs). arXiv 2016, arXiv:1606.08415. [Google Scholar]
- Boer, P.; Kroese, D.P.; Mannor, S.; Rubinstein, R.Y. A tutorial on the cross-entropy method. Ann. Oper. Res. 2005, 134, 19–67. [Google Scholar] [CrossRef]
- Rubinstein, R.Y.; Kroese, D.P. The Cross-Entropy Method: A Unified Approach to Combinatorial Optimization, Monte-Carlo Simulation, and Machine Learning; Information Science and Statistics; Springer: New York, NY, USA, 2004; pp. 1–47. [Google Scholar]
- Ho, Y.; Wookey, S. The real-world-weight cross- entropy loss function: Modeling the costs of mislabeling. IEEE Access 2019, 8, 4806–4813. [Google Scholar] [CrossRef]
- Chen, X.; Liu, S.; Sun, R.; Hong, M. On the convergence of a class of ADAM-type algorithms for non-convex optimization. arXiv 2018, arXiv:1808.02941. [Google Scholar]
- Syntax: Edge, 1994–2021 Years. Available online: https://www.mathworks.com/help/images/ref/edge.html (accessed on 1 January 2022).
- Chicco, D.; Jurman, G. The advantages of the Matthews correlation coefficient (MCC) over F1 score and accuracy in binary classification evaluation. BMC Genom. 2020, 21, 6. [Google Scholar] [CrossRef] [Green Version]
- Syntax: Histeq, 1994–2021 Years. Available online: https://www.mathworks.com/help/images/ref/histeq.html (accessed on 1 January 2022).
- Syntax: Imadjust, 1994–2021 Years. Available online: https://www.mathworks.com/help/images/ref/imadjust.html (accessed on 1 January 2022).
- Wu, J.-X.; Liu, H.-C.; Chen, P.-Y.; Lin, C.-H.; Chou, Y.-H.; Shung, K.K. Enhancement of ARFI-VTI elastography images in order to preliminary rapid screening of benign and malignant breast tumors using multilayer fractional-order machine vision classifier. IEEE Access 2020, 8, 164222–164237. [Google Scholar] [CrossRef]
- Valenzuela, G.; Laimes, R.; Chavez, I.; Salazar, C.; Bellido, E.G.; Tirado, I.; Pinto, J.; Guerrero, J.; Lavarello, R.J. In vivo diagnosis of metastasis in cervical lymph nodes using backscatter coefficient. In Proceedings of the 2018 IEEE International Ultrasonics Symposium (IUS), Kobe, Japan, 22–25 October 2018. [Google Scholar]
- Chansong, D.; Supratid, S. Impacts of Kernel size on different resized images in object recognition based on convolutional neural network. In Proceedings of the 2021 9th International Electrical Engineering Congress (iEECON), Pattaya, Thailand, 10–12 March 2021. [Google Scholar]
- Sidek, K.A.; Khalil, I.; Jelinek, H.F. ECG biometric with abnormal cardiac conditions in remote monitoring system. IEEE Trans. Syst. Man Cybern. Syst. 2014, 44, 1498–1509. [Google Scholar] [CrossRef]
- Zhang, X.-H. A Convolutional Neural Network Assisted Fast Tumor Screening System Based on Fractional-Order Image Enhancement: The Case of Breast X-ray Medical Imaging. Master’s Thesis, Department of Electrical Engineering, National Chin-Yi University of Technology, Taichung City, Taiwan, July 2021. [Google Scholar]
- Bruno, A.; Ardizzone, E.; Vitabile, S.; Midiri, M. A novel solution based on scale invariant feature transform descriptors and deep learning for the detection of suspicious regions in mammogram images. J. Med. Signals Sens. 2020, 10, 158–173. [Google Scholar]
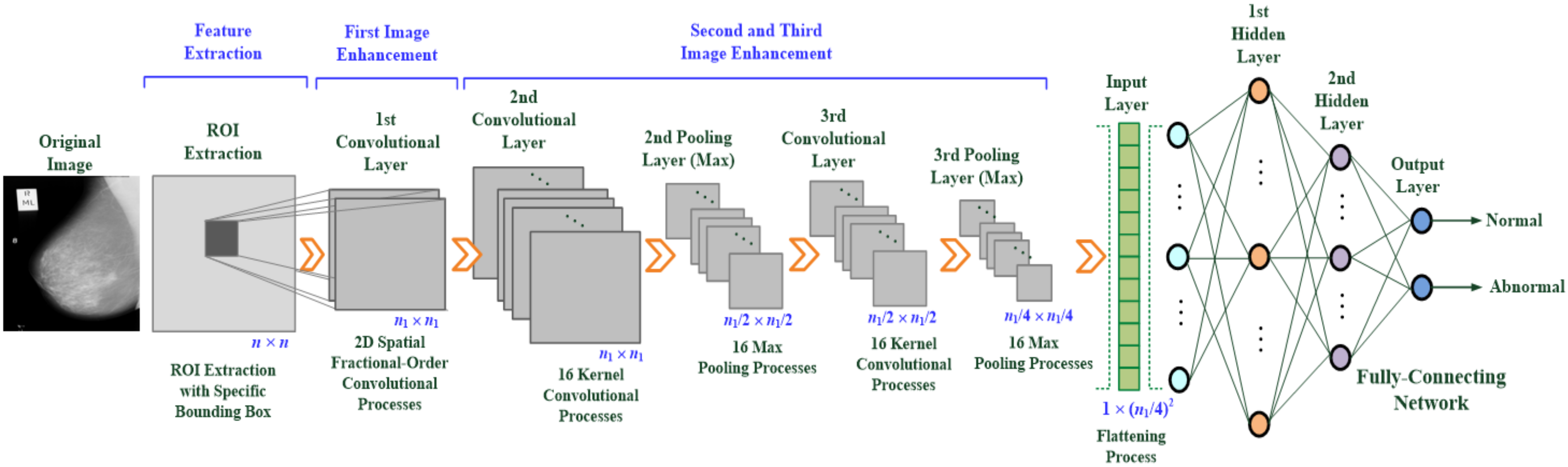
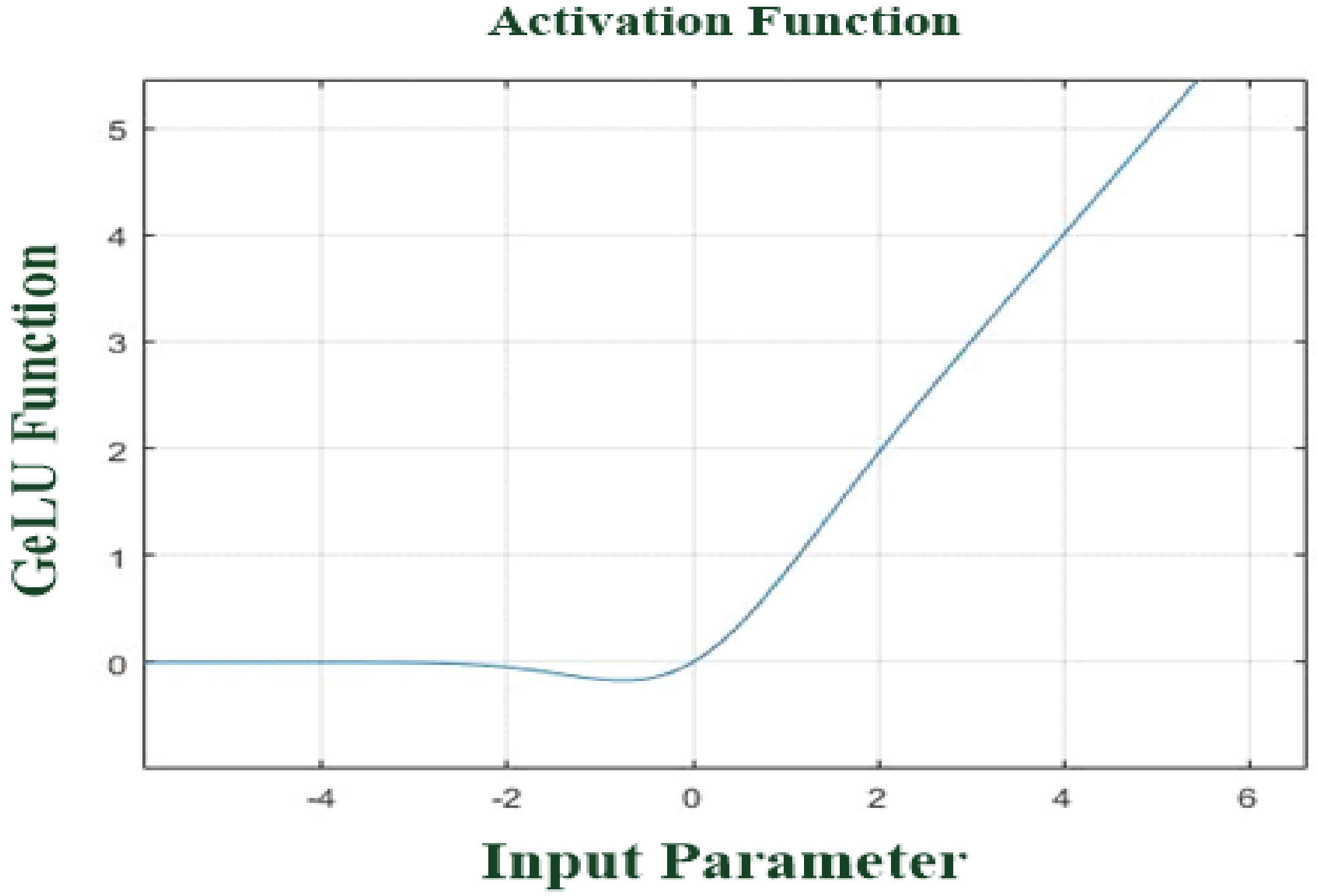
| Layer Function | Manner | Feature Pattern |
|---|---|---|
| Input Feature Pattern | ROI Extraction with 100 × 100 Bounding Box | 2D, 100 pixels × 100 pixels |
| 1st Convolutional Layer | 3 × 3 Fractional-Order Convolutional Window, Stride = 1 | 2D, 100 pixels × 100 pixels |
| 2nd Convolutional Layer | 3 × 3 Kernel Convolution Window, Stride = 1 | 2D, 100 pixels × 100 pixels |
| 2nd Pooling Layer | 2 × 2 Maximum Pooling Layer, Stride = 2 | 2D, 50 pixels × 50 pixels |
| 3rd Convolutional Layer | 3 × 3 Kernel Convolution Window, Stride = 1 | 2D, 50 pixels × 50 pixels |
| 3rd Pooling Layer | 2 × 2 Maximum Pooling Layer, Stride = 2 | 2D, 25 pixels × 25 pixels |
| Flattening Layer | Flattening Process | 1D, 1 × 625 feature vector |
| Classification Layer | Multilayer Classifier: 625 input nodes, 168 hidden nodes 64 hidden nodes, 2 output nodes (for normality and abnormality) | 1 × 625 Feature Vector Feeding into Multi-Layer Classifier |
| Algorithm: ADAM Algorithm |
| Actual | Total | Precision (%) | ||
|---|---|---|---|---|
| Predicted | TP | FP | TP + FP | (TP)/(TP + FP) |
| FN | TN | FN + TN | ||
| Total | TP + FN | FP + TN | Accuracy (%): (TP + TN)/(TP + FP + TN + FN) | |
| Recall (%) | (TP)/(TP + FN) | |||
| F1 Score | (2TP)/(2TP + FP + FN) | |||
| Model | 1st Convolution Window | 2nd Convolution Window | 3rd Convolution Window | 4th Convolution Window | 5th Convolution Window | Stride | Padding |
|---|---|---|---|---|---|---|---|
| 1 | 3 × 3, 2 | - | - | - | - | 1 | 1 |
| 2 | 3 × 3, 2 | 5 × 5, 16 | - | - | - | 1 | 1 |
| 3 | 3 × 3, 2 | 5 × 5, 16 | 7 × 7, 16 | - | - | 1 | 1 |
| 4 | 3 × 3, 2 | 5 × 5, 16 | 7 × 7, 16 | 9 × 9, 16 | - | 1 | 1 |
| 5 | 3 × 3, 2 | 5 × 5, 16 | 7 × 7, 16 | 9 × 9, 16 | 11 × 11, 16 | 1 | 1 |
| Model | 1 | 2 | 3 | 4 | 5 |
|---|---|---|---|---|---|
| Training CPU Time (min) | <30 | <240 | <7 | <10 | <180 |
| Average Accuracy (%) | 90.99% | 90.34% | 95.92% | 95.28% | 95.71% |
| Model | 1st Convolutional Window and Window Size | 2nd Convolutional Window and Window Size | 3rd Convolutional Window and Window Size | Stride/ Padding | Maximum Pooling Window | Stride |
|---|---|---|---|---|---|---|
| 1 | Fractional Order, 3 × 3, 2 | 3 × 3or 5 × 5, 16 | 3 × 3 or 5 × 5, 16 | 1/1 | 2 × 2, 16 | 2 |
| 2 | Sobel (First Order), 3 × 3, 2 | 3 × 3 or 5 × 5, 16 | 3 × 3 or 5 × 5, 16 | 1/1 | 2 × 2, 16 | 2 |
| 3 | Histeq, 3 × 3, 2 | 3 × 3 or 5 × 5, 16 | 3 × 3 or 5 × 5, 16 | 1/1 | 2 × 2, 16 | 2 |
| Model | 1st Convolutional Layer | 2nd Convolutional Layer | 2nd Pooling Layer | 3rd Convolutional Layer | 3rd Pooling Layer | Classification Layer (Fully Connecting Network) | Average Training Time (s) |
|---|---|---|---|---|---|---|---|
| 1 | 3 × 3, 2 | 3 × 3, 16 | 2 × 2, 16 | 3 × 3, 16 | 2 × 2, 16 | 625 input nodes, 168 1st hidden nodes, 64 2nd hidden nodes, 2 output nodes | <280 |
| 2 | 3 × 3, 16 | 5 × 5, 16 | <220 | ||||
| 3 | 5 × 5, 16 | 3 × 3, 16 | <240 | ||||
| 4 | 5 × 5, 16 | 5 × 5, 16 | <330 |
| Test Fold | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | Average Accuracy | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Model | ||||||||||||
| 1 | 96.14 | 97.43 | 98.07 | 97.96 | 98.93 | 98.07 | 96.35 | 95.60 | 96.89 | 98.28 | 97.37 | |
| 2 | 97.42 | 98.93 | 98.28 | 97.64 | 99.14 | 97.21 | 97.85 | 98.28 | 99.14 | 97.42 | 98.13 | |
| 3 | 96.14 | 97.64 | 98.50 | 98.71 | 99.14 | 97.85 | 97.64 | 95.06 | 97.21 | 99.57 | 97.75 | |
| 4 | 98.93 | 98.71 | 96.14 | 97.32 | 99.36 | 98.18 | 91.74 | 90.34 | 97.64 | 90.02 | 95.84 | |
| Test Fold | Precision (%) | Recall (%) | Accuracy (%) | F1 Score |
|---|---|---|---|---|
| 1 | 95.00 | 96.48 | 95.60 | 0.9574 |
| 2 | 94.38 | 95.92 | 95.20 | 0.9514 |
| 3 | 94.82 | 91.80 | 93.80 | 0.9389 |
| 4 | 95.02 | 96.50 | 95.60 | 0.9575 |
| 5 | 96.51 | 91.68 | 95.40 | 0.9577 |
| 6 | 94.09 | 94.84 | 94.40 | 0.9447 |
| 7 | 92.80 | 97.61 | 95.00 | 0.9515 |
| 8 | 92.77 | 95.85 | 94.40 | 0.9429 |
| 9 | 96.46 | 95.70 | 96.00 | 0.9608 |
| 10 | 95.19 | 95.54 | 95.00 | 0.9536 |
| Average | 95.19 | 95.19 | 95.04 | 0.9516 |
| Model | 1st Convolutional Layer | 2nd Convolutional Layer | 2nd Pooling Layer | 3rd Convolutional Layer | 3rd Pooling Layer | Classification Layer (Fully Connecting Network) | Average Training Time (s) |
|---|---|---|---|---|---|---|---|
| 1–1 | 3 × 3, 2 | 3 × 3, 4 | 2 × 2, 4 | 3 × 3, 4 | 2 × 2, 4 | 625 input nodes, 168 1st hidden nodes, 64 2nd hidden nodes, 2 output nodes | <150 |
| 1–2 | 3 × 3, 8 | 2 × 2, 8 | 3 × 3, 8 | 2 × 2, 8 | <240 | ||
| 1–3 | 3 × 3, 16 | 2 × 2, 16 | 3 × 3, 16 | 2 × 2, 16 | <280 | ||
| 1–4 | 3 × 3, 32 | 2 × 2, 32 | 3 × 3, 32 | 2 × 2, 32 | <330 |
| Test Fold | Precision (%) | Recall (%) | Accuracy (%) | F1 Score |
|---|---|---|---|---|
| 1 | 85.30 | 84.70 | 87.80 | 0.8640 |
| 2 | 87.10 | 93.00 | 91.20 | 0.9000 |
| 3 | 82.80 | 93.40 | 89.00 | 0.8760 |
| 4 | 86.10 | 91.50 | 93.20 | 0.9200 |
| 5 | 84.00 | 92.00 | 89.20 | 0.8780 |
| 6 | 93.20 | 84.80 | 91.08 | 0.8880 |
| 7 | 91.20 | 95.70 | 95.40 | 0.9480 |
| 8 | 90.10 | 94.80 | 93.60 | 0.9260 |
| 9 | 92.40 | 86.70 | 91.40 | 0.8950 |
| 10 | 97.20 | 99.10 | 98.40 | 0.9810 |
| Average | 88.94 | 91.57 | 92.30 | 0.9076 |
| Test Fold | Precision (%) | Recall (%) | Accuracy (%) | F1 Score |
|---|---|---|---|---|
| 1 | 96.60 | 95.70 | 96.80 | 0.9620 |
| 2 | 94.80 | 95.60 | 96.00 | 0.9530 |
| 3 | 96.10 | 93.00 | 95.40 | 0.9450 |
| 4 | 88.30 | 96.20 | 93.00 | 0.9210 |
| 5 | 90.10 | 95.30 | 93.60 | 0.9260 |
| 6 | 92.10 | 93.40 | 93.80 | 0.9270 |
| 7 | 93.40 | 93.80 | 94.70 | 0.9360 |
| 8 | 92.30 | 93.00 | 93.80 | 0.9270 |
| 9 | 94.10 | 97.60 | 96.40 | 0.9580 |
| 10 | 90.30 | 96.70 | 94.20 | 0.9340 |
| Average | 92.81 | 95.03 | 94.77 | 0.9389 |
| Test Fold | Precision (%) | Recall (%) | Accuracy (%) | F1 Score |
|---|---|---|---|---|
| 1 | 96.70 | 96.20 | 97.00 | 0.9640 |
| 2 | 97.60 | 95.30 | 96.60 | 0.9570 |
| 3 | 95.10 | 93.40 | 95.40 | 0.9420 |
| 4 | 97.10 | 94.00 | 96.20 | 0.9540 |
| 5 | 97.20 | 97.10 | 97.60 | 0.9680 |
| 6 | 93.00 | 94.00 | 94.40 | 0.9340 |
| 7 | 95.60 | 92.40 | 95.00 | 0.9400 |
| 8 | 98.00 | 97.20 | 98.10 | 0.9760 |
| 9 | 96.50 | 95.70 | 96.00 | 0.9610 |
| 10 | 95.20 | 95.50 | 95.00 | 0.9540 |
| Average | 96.30 | 95.04 | 95.93 | 0.9553 |
| Test Fold | Precision (%) | Recall (%) | Accuracy (%) | F1 Score |
|---|---|---|---|---|
| 1 | 96.70 | 96.20 | 97.00 | 0.9640 |
| 2 | 99.00 | 96.20 | 99.00 | 0.9760 |
| 3 | 90.80 | 93.40 | 93.20 | 0.9210 |
| 4 | 95.60 | 93.40 | 95.40 | 0.9230 |
| 5 | 99.50 | 97.60 | 98.80 | 0.9860 |
| 6 | 95.30 | 95.70 | 96.20 | 0.9550 |
| 7 | 66.00 | 95.60 | 77.20 | 0.7800 |
| 8 | 96.70 | 98.60 | 98.00 | 0.9770 |
| 9 | 97.40 | 91.50 | 95.40 | 0.9440 |
| 10 | 99.00 | 94.80 | 97.40 | 0.9690 |
| Average | 93.60 | 95.30 | 94.60 | 0.9395 |
| Model | First Convolutional Window | Second and Third Convolutional Window | Second and Third Pooling Window | Classification Layer | Average Accuracy |
|---|---|---|---|---|---|
| 1 | Fractional-Order Convolutional Window (2) | Kernel convolution Window (16) | Maximum Pooling Window (16) | One Input Layer, Two Hidden Layers, One Output Layer | >95% |
| 2 | Sobel (First-Order) Convolutional Window (2) | Kernel convolution Window (16) | Maximum Pooling Window (16) | >85% | |
| 3 | Histeq Convolutional Window (2) | Kernel convolution Window (16) | Maximum Pooling Window (16) | >90% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, P.-Y.; Zhang, X.-H.; Wu, J.-X.; Pai, C.-C.; Hsu, J.-C.; Lin, C.-H.; Pai, N.-S. Automatic Breast Tumor Screening of Mammographic Images with Optimal Convolutional Neural Network. Appl. Sci. 2022, 12, 4079. https://doi.org/10.3390/app12084079
Chen P-Y, Zhang X-H, Wu J-X, Pai C-C, Hsu J-C, Lin C-H, Pai N-S. Automatic Breast Tumor Screening of Mammographic Images with Optimal Convolutional Neural Network. Applied Sciences. 2022; 12(8):4079. https://doi.org/10.3390/app12084079
Chicago/Turabian StyleChen, Pi-Yun, Xuan-Hao Zhang, Jian-Xing Wu, Ching-Chou Pai, Jin-Chyr Hsu, Chia-Hung Lin, and Neng-Sheng Pai. 2022. "Automatic Breast Tumor Screening of Mammographic Images with Optimal Convolutional Neural Network" Applied Sciences 12, no. 8: 4079. https://doi.org/10.3390/app12084079
APA StyleChen, P.-Y., Zhang, X.-H., Wu, J.-X., Pai, C.-C., Hsu, J.-C., Lin, C.-H., & Pai, N.-S. (2022). Automatic Breast Tumor Screening of Mammographic Images with Optimal Convolutional Neural Network. Applied Sciences, 12(8), 4079. https://doi.org/10.3390/app12084079










