Release of Microplastics from Reusable Kitchen Plasticware and Generation of Thermal Potential Toxic Degradation Products in the Oven
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Material
2.2. Sample Preparation
2.3. Microplastic Analysis (FT-IR Spectroscopy)
2.4. Analysis of Thermal Decomposition Products
3. Results and Discussion
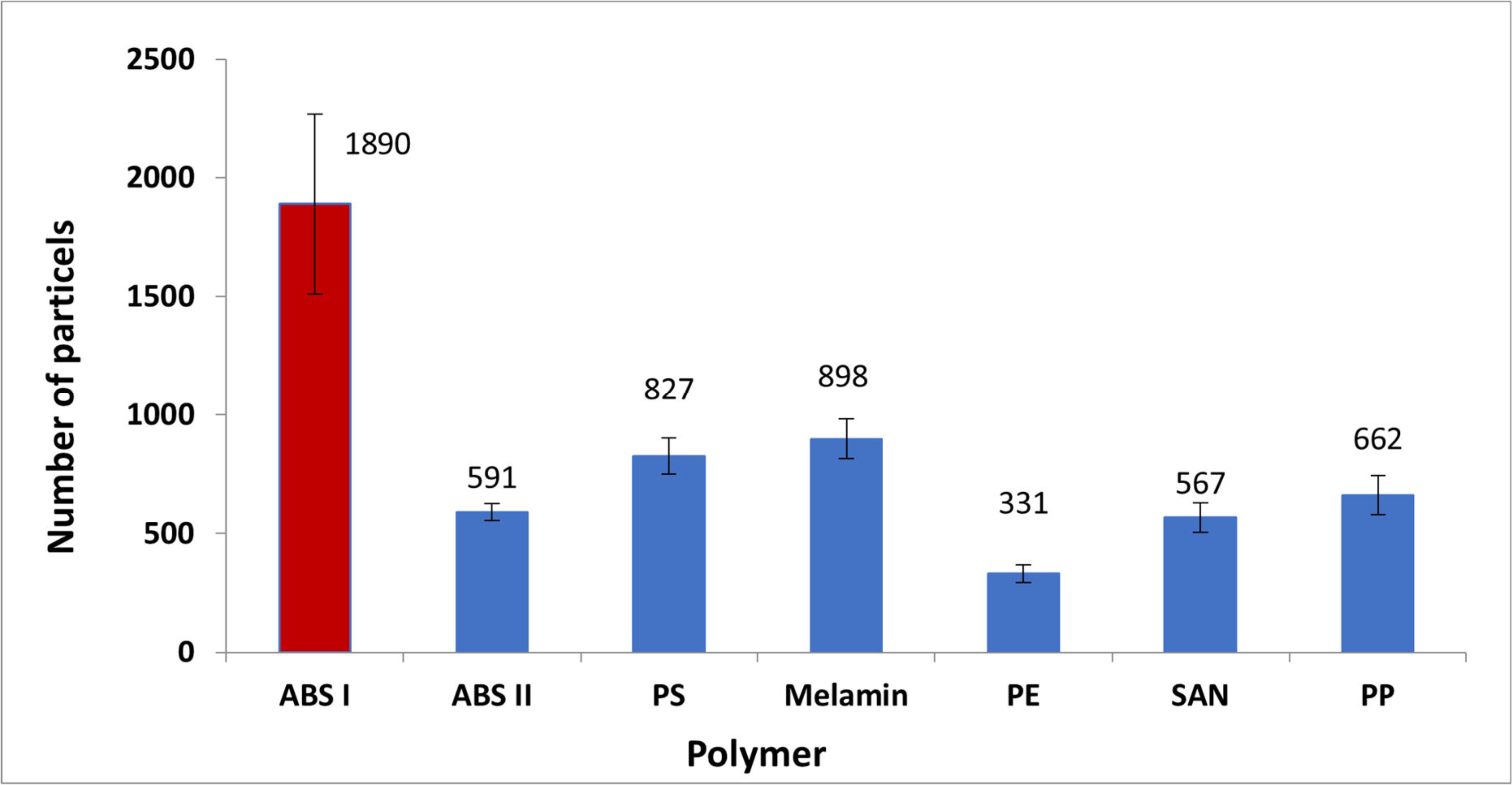
3.1. Microplastic Analysis (FTIR Spectroscopy)
3.2. Analysis of Thermal Decomposition Products (Pyrolysis and GC-MS)
4. Discussion/Conclusions
4.1. Microplastic Analysis (FTIR Spectroscopy)
4.2. Analysis of Thermal Decomposition Products (Pyrolysis and GC-MS)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sivan, A. New perspectives in plastic biodegradation. Curr. Opin. Biotechnol. 2011, 22, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chubarenko, I.; Efimova, I.; Bagaeva, M.; Bagaev, A.; Isachenko, I. On mechanical fragmentation of single-use plastics in the sea swash zone with different types of bottom sediments: Insights from laboratory experiments. Mar. Pollut. Bull. 2020, 150, 110726. [Google Scholar] [CrossRef] [PubMed]
- Andrady, A.L. Microplastics in the marine environment. Mar. Pollut. Bull. 2011, 6, 1596–1605. [Google Scholar] [CrossRef] [PubMed]
- Kooi, M.; van Nes, E.H.; Scheffer, M.; Koelmans, A.A. Ups and Downs in the Ocean: Effects of Biofouling on Vertical Transport of Microplastics. Environ. Sci. Technol. 2017, 51, 7963–7971. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hantoro, I.; Löhr, A.J.; van Belleghem, F.G.A.J.; Widianarko, B.; Ragas, A.M.J. Microplastics in coastal areas and seafood: Implications for food safety. Food Addit. Contam. Part A. Chem. Anal. Control. Expo Risk Assess. 2019, 36, 674–711. [Google Scholar] [CrossRef] [PubMed]
- Du, F.; Cai, H.; Zhang, Q.; Chen, Q.; Shi, H. Microplastics in take-out food containers. J. Hazard. Mater. 2020, 399, 122969. [Google Scholar] [CrossRef] [PubMed]
- Kwon, W.; Kim, D.; Kim, H.Y.; Jeong, S.W.; Lee, S.G.; Kim, H.C.; Lee, Y.J.; Kwon, M.K.; Hwang, J.S.; Han, J.E.; et al. Microglial phagocytosis of polystyrene microplastics results in immune alteration and apoptosis in vitro and in vivo. Sci. Total Environ. 2022, 807, 150817. [Google Scholar] [CrossRef] [PubMed]
- Kormos, J.L.; Schulz, M.; Ternes, T.A. Occurrence of iodinated X-ray contrast media and their biotransformation products in the urban water cycle. Environ. Sci. Technol. 2011, 45, 8723–8732. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.; Ngo, H.H.; Guo, W.; Chang, S.W.; Nguyen, D.D.; Liu, Y.; Wei, Q.; Wei, D. A critical review on antibiotics and hormones in swine wastewater: Water pollution problems and control approaches. J. Hazard. Mater. 2020, 387, 121682. [Google Scholar] [CrossRef] [PubMed]
- Binda, G.; Spanu, D.; Monticelli, D.; Pozzi, A.; Bellasi, A.; Bettinetti, R.; Carnati, S.; Nizzetto, L. Unfolding the interaction between microplastics and (trace) elements in water: A critical review. Water Res. 2021, 204, 117637. [Google Scholar] [CrossRef] [PubMed]
- Bussan, D.D.; Snaychuk, L.; Bartzas, G.; Douvris, C. Quantification of trace elements in surgical and KN95 face masks widely used during the SARS-COVID-19 pandemic. Sci. Total Environ. 2022, 814, 151924. [Google Scholar] [CrossRef] [PubMed]
- Cox, K.D.; Covernton, G.A.; Davies, H.L.; Dower, J.F.; Juanes, F.; Dudas, S.E. Human Consumption of Microplastics. Environ. Sci. Technol. 2019, 53, 7068–7074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwabl, P.; Koppel, S.; Konigshofer, P.; Bucsics, T.; Trauner, M.; Reiberger, T.; Liebmann, B. Detection of various microplastics in human stool: A prospective case series. Ann. Intern. Med. 2019, 171, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Basantes, M.F.; Conesa, J.A.; Fullana, A. Microplastics in Honey, Beer, Milk and Refreshments in Ecuador as Emerging Contaminants. Sustainability 2020, 12, 5514. [Google Scholar] [CrossRef]
- Karami, A.; Golieskardi, A.; Keong Choo, C.; Larat, V.; Galloway, T.S.; Salamatinia, B. The presence of microplastics in commercial salts from different countries. Sci. Rep. 2017, 7, 46173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jander, J. Mikroplastik in Flüssen und Lebensmitteln. Bachelor’s Thesis, Hochschule Furtwangen University, Villingen-Shwenningen, Germany, 2021. [Google Scholar]
- Institut für Arbeitsschutz der Deutschen Gesetzlichen Unfallversicherung. GESTIS-Stoffdatenbank. Available online: https://gestis.dguv.de/ (accessed on 11 January 2022).
- European Union, Eurpean Chemicals Agency. Available online: https://echa.europa.eu/de/home (accessed on 11 January 2022).
- Conti, B.; Maltoni, C.; Perino, G.; Ciliberti, A. Long-term carcinogenicity bioassays on styrene administered by inhalation, ingestion and injection and styrene oxide administered by ingestion in Sprague-Dawley rats, and para-methylstyrene administered by ingestion in Sprague-Dawley rats and Swiss mice. Ann. N. Y. Acad. Sci. 1988, 534, 203–234. [Google Scholar] [CrossRef] [PubMed]
- Linhart, I.; Šmejkal, J.; Mládková, I. Stereochemical aspects of styrene biotransformation. Toxicol. Lett. 1998, 94, 127–135. [Google Scholar] [CrossRef]
- Bundesinstitut für Risikobewertung. Freisetzung von Melamin und Formaldehyd aus Geschirr und Küchenutensilien; Bundesinstitut für Risikobewertung: Berlin, Germany, 2011; pp. 1–5.
- Committee on Acute Exposure Guideline Levels; Committee on Toxicology; Board on Environmental Studies and Toxicology; Division on Earth and Life Studies; National Research Council. Butane: Acute Exposure Guideline Levels for Selected Airbone Chem; National Academies Press: Washington, DC, USA, 2012; p. 12. [Google Scholar] [CrossRef]
| Polymeric Material | Minimum Rockwell Hardness | Maximum Rockwell Hardness |
|---|---|---|
| Acrylonitrile–butadiene styrene (ABS) | 2.00 | 70.00 |
| Low-density polyethylene (LDPE) | 1.00 | 20.00 |
| Melamine | 67.00 | 86.00 |
| Polypropylene (PP) | 1.00 | 20.00 |
| Polystyrene (PS) | 60.00 | 75.00 |
| Styrene–acrylonitrile copolymers (SAN) | 10.00 | 80.00 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jander, J.; Hummel, D.; Stürmer, S.; Monteleone, A.; Neumaier, T.; Broghammer, F.; Lewin-Kretzschmar, U.; Brock, T.; Knoll, M.; Fath, A.S. Release of Microplastics from Reusable Kitchen Plasticware and Generation of Thermal Potential Toxic Degradation Products in the Oven. Appl. Sci. 2022, 12, 2535. https://doi.org/10.3390/app12052535
Jander J, Hummel D, Stürmer S, Monteleone A, Neumaier T, Broghammer F, Lewin-Kretzschmar U, Brock T, Knoll M, Fath AS. Release of Microplastics from Reusable Kitchen Plasticware and Generation of Thermal Potential Toxic Degradation Products in the Oven. Applied Sciences. 2022; 12(5):2535. https://doi.org/10.3390/app12052535
Chicago/Turabian StyleJander, Juri, Darius Hummel, Sophie Stürmer, Adrian Monteleone, Tizian Neumaier, Felix Broghammer, Uta Lewin-Kretzschmar, Thomas Brock, Martin Knoll, and Andreas Siegbert Fath. 2022. "Release of Microplastics from Reusable Kitchen Plasticware and Generation of Thermal Potential Toxic Degradation Products in the Oven" Applied Sciences 12, no. 5: 2535. https://doi.org/10.3390/app12052535
APA StyleJander, J., Hummel, D., Stürmer, S., Monteleone, A., Neumaier, T., Broghammer, F., Lewin-Kretzschmar, U., Brock, T., Knoll, M., & Fath, A. S. (2022). Release of Microplastics from Reusable Kitchen Plasticware and Generation of Thermal Potential Toxic Degradation Products in the Oven. Applied Sciences, 12(5), 2535. https://doi.org/10.3390/app12052535






