Abstract
The aim of the study was to determine the aroma profiles of four kinds of Slovak honey (sunflower, honeydew, acacia, and linden) by a qualitative and quantitative screening of their volatile compounds and by gas chromatography for the potential use in the aromachology and the business sphere. The results showed that several unique volatiles were identified in one kind of honey, while they were not identified in the remaining ones. The acacia honey had the unique volatile linalool oxide (1.13–3.9%); linden honey had the unique volatiles nerol oxide (0.6–1.6%), ethyl esters (0.41–8.78%), lilac aldehyde D (6.6%), and acetophenone (0.37%). The honeydew honey had the unique volatiles santene (0.28%) and cyclofenchene (0.59–1.39%), whereas 2-bornene (0.43–0.81%) was typical for sunflower honey. While linden honey was characterized by fruity ethyl esters, honeydew honey had more monoterpenoid compounds. In the principal component analysis model, the four kinds of honey could not be differentiated by aroma volatiles. However, it was possible to classify the linden and sunflower honey using the LDA. In conclusion, the current study provided experimental evidence that the marker compounds from different kinds of honey might be promising candidates for production of inhaling aromas.
1. Introduction
An aroma represents an effective marketing tool and is considered to be a new way of in-store communication with customers [1]. According to Horská et al. [2], nowadays the usage of fragrance compounds and essential oils in many business sectors is increasing. Many companies are using their own branded aromas in their shops, offices, or even during some marketing events or campaigns. More and more companies are creating aroma logos or corporate scents to be used indoors. Even retailers apply specific aromas to create comfortable environment where customers would spend more time and money by purchasing more products [3].
In general, these odors and aromas are distributed by installing aromatizers or diffusers in certain areas [4]. Moreover, aromatization may attract new customers; however, at first it is necessary to conduct aroma testing in order to select appropriate aromas prior to their implementation. For example, application of cappuccino aroma at store in Slovakia increased the volume of sales in confectionery category (desserts, chocolate bars, chocolates, and waffles) [5]. Furthermore; Berčík et al. [6] indicates that implementing aromatization in business spaces has positive impact on economic indicators, including the sector of services.
Aroma, as a marketing stimulus plays, an important role even in the food industry and gastronomy [7]. Honey, as a food product, has (besides health benefits and nutritional values) very specific characteristics from an organoleptic point of view. It is used not only in the food sector, but also in the cosmetic and pharmaceutical industries [8,9]. Healing properties of honey are used for massages, therapy wraps, or for production of scented candles. Honey contains compounds with antioxidant [10], antibacterial, antifungal, anticancer [11], and anti-inflammatory effects [8,9,11]. Compounds with such properties are mainly polyphenols [8,11]. However, these effects were confirmed also for some volatile organic compounds (VOCs), particularly terpenes [12,13]. VOCs from honey were previously determined by GC-MC analysis of simultaneous extraction and distillation extracts [14,15], HS-SPME/GC-MS [16,17,18,19,20,21], HS-GC-MS [22], and HS-SPME/GC×GC-TOF-MS [23,24] analyses. Even though honey diffusers are available on the market, there are no studies evaluating their impact on the business sphere.
The aim of this study was to characterize the aroma profiles of Slovak honey samples, in order to (i) find differences between four kinds of monofloral honey, and (ii) to determine promising volatile candidates for production of inhaling aromas.
2. Materials and Methods
2.1. Samples
In this study, a set of four kinds of honey (sunflower n = 5, acacia n = 5, honeydew n = 5, and linden n = 5) were analyzed (Table 1). The honey samples (300 g) were sourced from local Slovak small-scale beekeepers and were harvested in 2020. All the honey samples were stored at 20 °C and analyzed within six months after being harvested. Content of sugar was determined by refractometer designed to make corrections based on temperature using automatic temperature compensation (HR901, Krüss GmbH, Hamburg, Germany). A total of 20 samples (4 × 5) were analyzed in triplicate.

Table 1.
Basic characterization of Slovak honey samples, including kind of honey, location, and sugar content.
2.2. Determination of Volatile Organic Compounds
Sample preparation and isolation of VOCs were chosen based on a previous study by Kružík et al. [16]. VOCs were extracted from the honey samples using SPME fiber (DVB/CAR/PDMS 50/30 µm; Supelco, Bellefonte, PA, USA) with CombiPAL automated sample injector 120 (CTC Analytics AG, Zwingen, Switzerland). Each of the honey samples (2 g) was dissolved in a 20 mL glass vial using 2 mL NaCl solution in distilled water (200 g/L). The gas chromatography (Agilent GC7890B) with mass spectrometry (Agilent MSD 5977A) (Agilent Technologies Inc., Santa Clara, CA, USA) method equipped with the column HP-5ms (30 m × 0.25 mm × 0.25 µm; Agilent Technologies) previously described by Kružík et al. [16] was used for determination of VOCs in a modified version. A modified temperature program 40 °C (1 min), 5 °C/min, 250 °C (1 min) was used. Individual compounds were identified based on the comparison of the mass spectra with the commercial database of the National Institute of Standards and Technology (NIST, Gaithersburg, MD, USA) mass spectral library (NIST17), and on the assessment of retention times with the literature [16,17]. The relative content (expressed in percentage) of determined compounds was calculated by dividing individual peak area by the total area of all peaks. Each sample was measured in triplicate.
2.3. Statistical Analysis
All of the data obtained were analyzed by descriptive statistics arithmetic average and standard deviation. To determine the aroma differences (>5% content) between the honey samples, descriptive statistics, normality tests, LDA (Linear Discriminant Analysis) and the PCA (Principal Component Analysis) were performed using the MS Excel and XLSTAT package program [25].
3. Results
By analyzing the saline solution of honey by GC-MS analysis, in total, ninety-four VOCs were identified, including alkanes, alcohols, alkenes, nitriles, acids, esters, monoterpenes, monoterpenoids, aldehydes, and ketones (Table S1). Several volatiles, that were present in one kind of honey and not in the other ones, can be marked as markers. The values shown in the Table 2 for each unique VOC are means of triplicate determinations with the standard deviation, retention time, and previous identification by the literature. As an example, the chromatograms of each kind of honey sample are shown in Figures S1–S4.

Table 2.
Identified unique VOCs in four kinds of honey with percentage content 1 ± SD 2 and previous identification in literature 3.
The PCA for four kinds of honey (Figure 1) revealed that 46.42% of the total variation embodied in 15 variables could be effectively condensed into, and explained, by the first two principal components (PCs), with eigenvalues of 4.3 and 2.7, respectively. The results show that the various kinds of honey contain characteristic volatile substances. On the other hand, there were no differences between the tested kinds of honey. The compounds as 3-methyl-1-pentanol, dimethyl ether, p-cymen-8-ol, lilac aldehyde D, and dimethyl sulfide are characteristic for linden honey. The acacia, sunflower, and honeydew honey samples were characterized by lilac aldehyde B, benzaldehyde, furfural, trans-linalool oxide, linalool, acetic acid, and hotrienol.
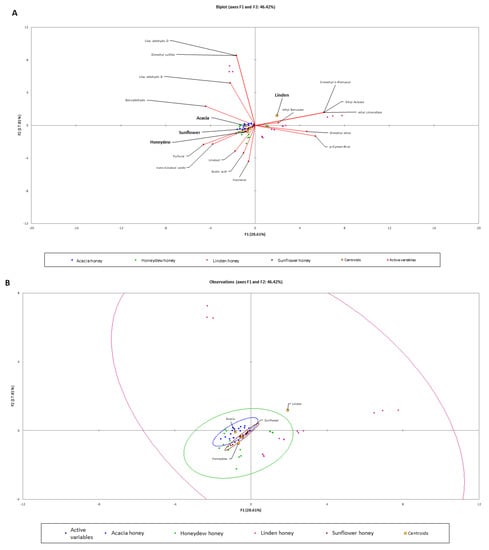
Figure 1.
PCA evaluation of four kinds of honey (honeydew, acacia, linden, and sunflower); (A)—significant (>5%) aroma profile of samples with corresponding aromas; (B)—categorization of samples.
Discriminant analysis was used to classify the tested kinds of honey samples. The Wilks’ Lambda test showed that the difference between the means vectors of the samples were significant (p < 0.0001). The first two eigen vectors explains 97.26% of variance (Figure 2). The confusion matrix calculated for the four tested samples was equal to 94.74% (data not shown). The Cross-validation: prior and posterior classification was performed to calculate the membership probabilities for unknown samples. The total accuracy of the cross-validation model was 85.96% (Table 3). Discriminant analysis can be used to classify the linden and sunflower kinds of honey but the acacia and honeydew kinds of honey were misclassified.
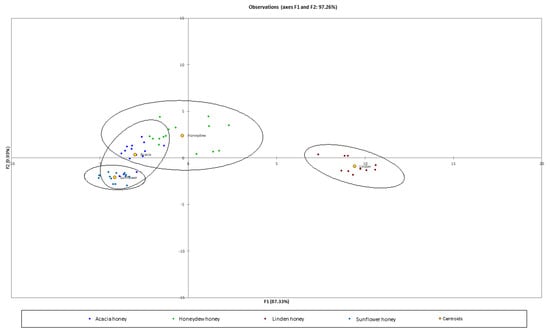
Figure 2.
LDA map of kind of honey based on VOCs content.

Table 3.
Confusion matrix for the cross-validation results.
4. Discussion
In total, 94 VOCs were detected in sunflower, acacia, honeydew, and linden honey samples. Some of these compounds have been previously reported by Plutowska et al. [26] in Polish linden, honeydew, and acacia honey. Sunflower and acacia honey samples from different countries were characterized by Radovic et al. [31]. The linalool oxide, as marker for acacia honey, is consistent with our results. The sunflower honey markers were α-pinene and 3-methyl-2-butanol according to Radovic et al. [31].
In this study, sunflower honey was characterized by β-calarene, cyclofenchene, and 2-bornene. Linden and honeydew had the most marker compounds. While tested linden honey was characterized by fruity ethyl esters [33], honeydew honey samples had more monoterpenoid compounds originated from aromatic plants [38]. The identified compounds in the tested honey samples were previously reported also in different kinds of honey [14,15,16,17,18,19,20,21,22,23,24,26,27,28,29,30,31,32,33,34,35,36,37], except linalyl acetate, which was previously tested for the fumigation of beehives to control the honey bee parasites [39]. Altogether, 16 volatiles have not been previously identified in honey samples.
According to The International Fragrance Association [40], 53 volatiles are recommended as honey fragrances. On the other hand, we identified honey volatile markers, which are not recommended as honey fragrances in the Fragrance Ingredient Glossary [40], but they are used in, for example, perfumery. The linden and honeydew samples contained anisyl alcohol (2-(para-anisyl) alcohol) (0.73%, and 0.80%, respectively), characterized as mild-floral, very sweet odor, reminiscent of lilac and vanilla with a faint, delicate, balsamic background used in perfumery [40].
The acetophenone is formed during the phenylpropane metabolism [37], and it was previously reported in chestnut honey [36,37]. However, this volatile was not previously considered by the research teams as a specific marker for the floral honey. It is characterized as a “floral” aroma fragrance described as: almond, orange, cherry, honeysuckle, jasmine, and strawberry [40]. In the present work, the acetophenone was found in linden honey (0.38%).
Each tested sample had hotrienol which is known as probably being formed during the honey’s ripening [41]. It was previously found in rapeseed honey from Czech beekeepers [16], leatherwood honey from Tasmania [41], and Greek citrus honey [42]. It could be the next honey fragrance, which is missing in the Fragrance Ingredient Glossary [40]. It can be used in many flavors, such as elderflower, grape, berry, and honey [43].
5. Conclusions
We confirmed that each kind of honey had its own unique volatiles, but the aroma differences by PCA in the tested monofloral honey samples were not confirmed. It is possible to classify linden and sunflower kinds of honey by using LDA. On the other hand, the LDA cannot clearly classify acacia and honeydew honey based on the selected VOCs. The reason may also be that the linden and honeydew honey samples have rich aromatic profiles and cover less aromatic honey samples. Studies evaluating their impact in the marketing or aromachology respectively were not published yet. According to our results, the anisyl alcohol, hotrienol, and acetophenone may be used as the honey fragrance.
In conclusion, the question arises as to whether customers would be able to recognize individual kinds of honey scents, and how this would affect their behavior.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/app11178177/s1. Table S1: Content of VOCs in four kinds of honey samples determined by HS-SPME GC-MS; Figure S1: Chromatogram of sunflower honey; Figure S2: Chromatogram of acacia honey; Figure S3: Chromatogram of honeydew honey; Figure S4: Chromatogram of linden honey.
Author Contributions
Conceptualization, J.Š., P.M. and P.Š.; methodology, J.Š. and P.M.; formal analysis, J.Š. and P.M.; resources, P.Š. and P.M.; data curation, M.Š.; writing—original draft preparation, J.Š., P.M., M.Š., P.Š. and V.V.; writing—review and editing, J.Š., P.M., M.Š., P.Š. and V.V.; visualization, J.Š., P.M. and M.Š.; project administration, V.V.; funding acquisition, V.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Slovak Research and Development Agency based on Contract no. APVV-17-0564 “The Use of Consumer Neuroscience and Innovative Research Solutions in Aromachology and its Application in Production, Business and Services” and the Grant Agency of The Slovak University of Agriculture in Nitra, grant number 14-GASPU-2021 “Analysis of consumer behavior towards honeys enriched with health-promoting substances”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Acknowledgments
We thank to the Operational Program Integrated Infrastructure within the project: Demand-driven research for the sustainable and innovative food, Drive4SIFood 313011V336, co-financed by the European Regional Development Fund (0.9) and the Operational Program Research and Innovation: “Support of research activities in the ABT RC”, 313011T465, co-financed by the European Regional Development Fund (0.1).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Rimkute, J.; Moraes, C.; Ferreira, C. The Effects of Scent on Consumer Behaviour. Int. J. Consum. Stud. 2016, 40, 24–34. [Google Scholar] [CrossRef]
- Horská, E.; Šedík, P.; Berčík, J.; Krasnodębski, A.; Witczak, M.; Filipiak-Florkiewicz, A. Aromachology in food sector-aspects of consumer food products choice. Żywność Nauka Technologia Jakość 2018, 25, 33–41. Available online: https://wydawnictwo.pttz.org/wp-content/uploads/2019/02/03_Horska.pdf (accessed on 28 July 2021).
- American Marketing Association. Good Smells Are Good Marketing: How to Use Scent to Your Advantage. 2020. Available online: https://www.ama.org/marketing-news-home (accessed on 15 June 2021).
- Jurášková, O.; Horňák, P. Large Dictionary of Marketing Communications; Grada Publishing: Praha, Czech Republic, 2012; 272p. [Google Scholar]
- Berčík, J.; Virágh, R.; Kádeková, Z.; Duchoňová, T. Aroma marketing as a tool to increase turnover in a chosen business entity. Potravin. Slovak J. Food Sci. 2020, 14, 1161–1175. [Google Scholar] [CrossRef]
- Berčík, J.; Mravcová, A.; Gálová, J.; Mikláš, M. The use of consumer neuroscience in aroma marketing of a service company. Potravin. Slovak J. Food Sci. 2020, 14, 1200–1210. [Google Scholar] [CrossRef]
- Berčík, J.; Paluchová, J.; Neomániová, K. Neurogastronomy as a Tool for Evaluating Emotions and Visual Preferences of Selected Food Served in Different Ways. Foods 2021, 10, 354. [Google Scholar] [CrossRef]
- Machado De-Melo, A.A.; de Almeida-Muradian, L.B.; Sancho, M.T.; Pascual-Maté, A. Composition and properties of Apis mellifera honey: A review. J. Apic. Res. 2017, 57, 5–37. [Google Scholar] [CrossRef]
- Bogdanov, S.; Jurendic, T.; Sieber, R.; Gallmann, P. Honey for Nutrition and Health: A Review. J. Am. Coll. Nutr. 2008, 27, 677–689. [Google Scholar] [CrossRef]
- Grassmann, J. Terpenoids as plant antioxidants. In Vitamins and Hormones, 1st ed.; Litwack, G., Ed.; Academic Press: Cambridge, MA, USA, 2005; Volume 72, pp. 505–535. [Google Scholar] [CrossRef]
- Mărgăoan, R.; Topal, E.; Balkanska, R.; Yücel, B.; Oravecz, T.; Cornea-Cipcigan, M.; Vodnar, D.C. Monofloral Honeys as a Potential Source of Natural Antioxidants, Minerals and Medicine. Antioxidants 2021, 10, 1023. [Google Scholar] [CrossRef]
- Kopaczyk, J.M.; Warguła, J.; Jelonek, T. The variability of terpenes in conifers under developmental and environmental stimuli. Environ. Exp. Bot. 2020, 180, 1–11. [Google Scholar] [CrossRef]
- Porres-Martínez, M.; Gonzáles-Burgos, E.; Carretero, M.E.; Gómez-Serranillos, M.P. In vitro neuroprotective potential of the monoterpenes α-pinene and 1,8-cineole against H2O2-induced oxidative stress in PC12 cells. Z. Naturforsch. 2016, 71, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Castro-Vázquez, L.; Diáz-Maroto, M.C.; Pérez-Coello, M.S. Aroma composition and new chemical markers of Spanish citrus honeys. Food Chem. 2007, 103, 601–606. [Google Scholar] [CrossRef]
- Serra Bonvehí, J.; Ventura Coll, F. Flavour index and aroma profiles of fresh and processed honeys. J. Sci. Food Agric. 2003, 83, 275–282. [Google Scholar] [CrossRef]
- Kružík, V.; Grégrová, A.; Ziková, A.; Čižková, H. Rape honey: Determination of botanical origin based on volatile compound profiles. J. Food Nutr. Res. 2019, 58, 339–348. [Google Scholar]
- Kružík, V.; Grégrová, A.; Rajchl, A.; Čížková, H. Study on honey quality evaluation and detection of adulteration by analysis of volatile compounds. J. Apic. Sci. 2017, 16, 17–27. [Google Scholar] [CrossRef] [Green Version]
- Acevedo, F.; Torres, P.; Oomah, B.D.; de Alencar, S.M.; Massarioli, A.P.; Marín-Venegas, R.; Albarral-Ávila, V.; Burgos-Diáz, C.; Ferrer, R.; Rubilar, M. Volatile and non-volatile/semi-volatile compounds and in vitro bioactive properties of Chilean Ulmo (Eucryphia cordifolia Cav.) honey. Food Res. Int. 2017, 94, 20–28. [Google Scholar] [CrossRef] [PubMed]
- da Costa, A.C.V.; Sousa, J.M.B.; da Silva, M.A.A.P.; Garruti, D.D.S.; Madruga, M.S. Sensory and volatile profiles of monofloral honeys produced by native stingless bees of the Brazilian semiarid region. Food Res. Int. 2018, 105, 110–120. [Google Scholar] [CrossRef] [PubMed]
- da Costa, A.C.V.; Sousa, J.M.B.; Bezerra, T.K.A.; da Silva, F.L.H.; Pastore, G.M.; da Silva, M.A.A.P.; Madruga, M.S. Volatile profile of monofloral honeys produced in Brazilian semiarid region by stingless bees and key volatile compounds. LWT 2018, 94, 198–207. [Google Scholar] [CrossRef]
- Bianchi, F.; Mangia, A.; Mattarozzi, M.; Musci, M. Characterization of the volatile profile of thistle honey using headspace solid-phase microextraction and gas chromatography-mass spectrometry. Food Chem. 2011, 129, 1030–1036. [Google Scholar] [CrossRef]
- de Lima Morais da Silva, P.; de Lima, L.S.; Caetano, Í.K.; Torres, Y.R. Comparative analysis of the volatile composition of honeys from Brazilian stingless bees by static headspace GC-MS. Food Res. Int. 2017, 102, 536–543. [Google Scholar] [CrossRef]
- Špánik, I.; Janáčová, A.; Šusterová, Z.; Jakubík, T.; Jánošková, N.; Novák, P.; Chlebo, R. Characterization of VOC composition of Slovak monofloral honeys by GCxGC-TOF-MS. Chem. Pap. 2012, 67, 127–134. [Google Scholar] [CrossRef]
- Dymerski, T.; Chmiel, T.; Mostafa, A.; Sliwinska, M.; Wisniewska, P.; Wardencki, W.; Namiesnik, J.; Gorecki, T. Botanical and Geographical Origin Characterization of Polish Honeys by Headspace SPME-GC×GC-TOFMS. Curr. Org. Chem. 2013, 17, 1–19. [Google Scholar] [CrossRef]
- XLSTAT (Addinsoft). Analyse de Données et Statistique avec MS Excel; Addinsoft: New York, NY, USA, 2014. [Google Scholar]
- Plutowska, B.; Chmiel, T.; Dymerski, T.; Wardencki, W. A headspace solid-phase microextraction method development and its application in the determination of volatiles in honeys by gas chromatography. Food Chem. 2011, 126, 1288–1298. [Google Scholar] [CrossRef]
- Patrignami, M.; Fagúndez, G.A.; Tananaki, C.; Thrasyvoulou, A.; Lupano, C.E. Volatile compounds of Argentinean honeys: Correlation with floral and geographical origin. Food Chem. 2018, 246, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Seisonen, S.; Kivima, E.; Ven, K. Characterization of the aroma profiles of different honeys and corresponding flowers using solid-phase microextraction and gas chromatography-mass spectrometry/olfactometry. Food Chem. 2015, 169, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Tanleque-Alberto, F.; Juan-Borrás, M.; Escriche, I. Quality parameters, pollen and volatile profiles of honey from North and Central Mozambique. Food Chem. 2019, 277, 543–553. [Google Scholar] [CrossRef]
- Kaškoniené, V.; Venskutonis, P.R.; Čeksteryté, V. Composition of volatile compounds of honey of various floral origin and beebread collected in Lithuania. Food Chem. 2008, 111, 988–997. [Google Scholar] [CrossRef]
- Radovic, B.S.; Careri, M.; Mangia, A.; Musci, M.; Gerboles, M.; Anklam, E. Contribution of dynamic headspace GC-MS analysis of aroma compounds to authenticity testing of honey. Food Chem. 2001, 72, 511–520. [Google Scholar] [CrossRef]
- Kortesniemi, M.; Rosenvald, S.; Laaksonen, O.; Vanag, A.; Ollikka, T.; Vene, K.; Yang, B. Sensory and chemical profiles of Finnish honeys of different botanical origins and consumer preferences. Food Chem. 2018, 246, 351–359. [Google Scholar] [CrossRef]
- Leng, P.; Hu, H.-W.; Cui, A.-H.; Tang, H.-J.; Liu, Y.-G. HS-GC-IMS with PCA to analyze volatile flavor compounds of honey peach packaged with different preservation methods during storage. LWT 2021, 149, 1–9. [Google Scholar] [CrossRef]
- Escriche, I.; Sobrino-Gregorio, L.; Conchado, A.; Juan-Borrás, M. Volatile profile in the accurate labelling of monofloral honey. The case of lavender and thyme honey. Food Chem. 2017, 226, 61–68. [Google Scholar] [CrossRef]
- Ruisinger, B.; Schieberle, P. Characterization of the Key Aroma Compounds in Rape Honey by Means of the Molecular Sensory Science Concept. J. Agric. Food Chem. 2012, 60, 4186–4194. [Google Scholar] [CrossRef] [PubMed]
- Bonaga, G.; Giumanini, A.G. The Volatile Fraction of Chestnut Honey. J. Apic. Res. 1986, 25, 113–120. [Google Scholar] [CrossRef]
- Guyot, C.; Bouseta, A.; Scheirman, V.; Collin, S. Floral Origin Markers of Chestnut and Lime Tree Honeys. J. Agric. Food Chem. 1998, 46, 625–633. [Google Scholar] [CrossRef]
- Jerković, I.; Kuś, P.M. Terpenes in honey: Occurrence, origin and their role as chemical biomarkers. RSC Adv. 2014, 4, 31710–31728. [Google Scholar] [CrossRef]
- Gonzales-Coloma, A.; Reina, M.; Diaz, C.E.; Fraga, B.M.; Santana-Meridas, O. Natural Product-Based Biopesticides for Insect Control. Ref. Modul. Chem. Mol. Sci. Chem. Eng. 2013, 1–55. [Google Scholar] [CrossRef]
- The International Fragrance Association. Fragrance Ingredient Glossary. 2020. Available online: https://ifrafragrance.org/docs/default-source/glossary/ifra-fragrance-ingredient-glossary---april-2020.pdf?sfvrsn=dc0e87ff_2&fbclid=IwAR0zezEAAkz2xTt7G2w-DIpJg6-gjBkYn4sbM8jx7kO1uXc04rsvx5_gw1A (accessed on 20 June 2021).
- Rowland, C.Y.; Blackman, A.J.; D’Arcy, B.R.; Rintoul, G.B. Comparison of organic extractives found in leatherwood (Eucryphia lucida) honey and leatherwood flowers and leaves. J. Agric. Food Chem. 1995, 43, 753–763. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Tarantilis, P.A.; Harizanis, P.C.; Polissiou, M. Aroma investigation of unifloral Greek citrus honey using solid-phase microextraction coupled to gas chromatographic-mass spectrometric analysis. Food Chem. 2017, 100, 396–404. [Google Scholar] [CrossRef]
- Berger, R.G. (Ed.) Flavours and Fragrances. Chemistry, Bioprocessing and Sustainability; Springer: Hannover, Germany, 2007; Volume 648, ISBN 978-3-540-49338-9. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).