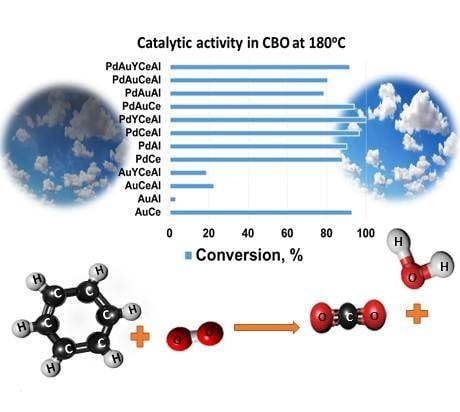
Complete Benzene Oxidation over Mono and Bimetallic Pd—Au Catalysts on Alumina-Supported Y-Doped Ceria
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Catalyst Synthesis
2.2. Catalyst Characterization
2.3. Catalytic Measurements
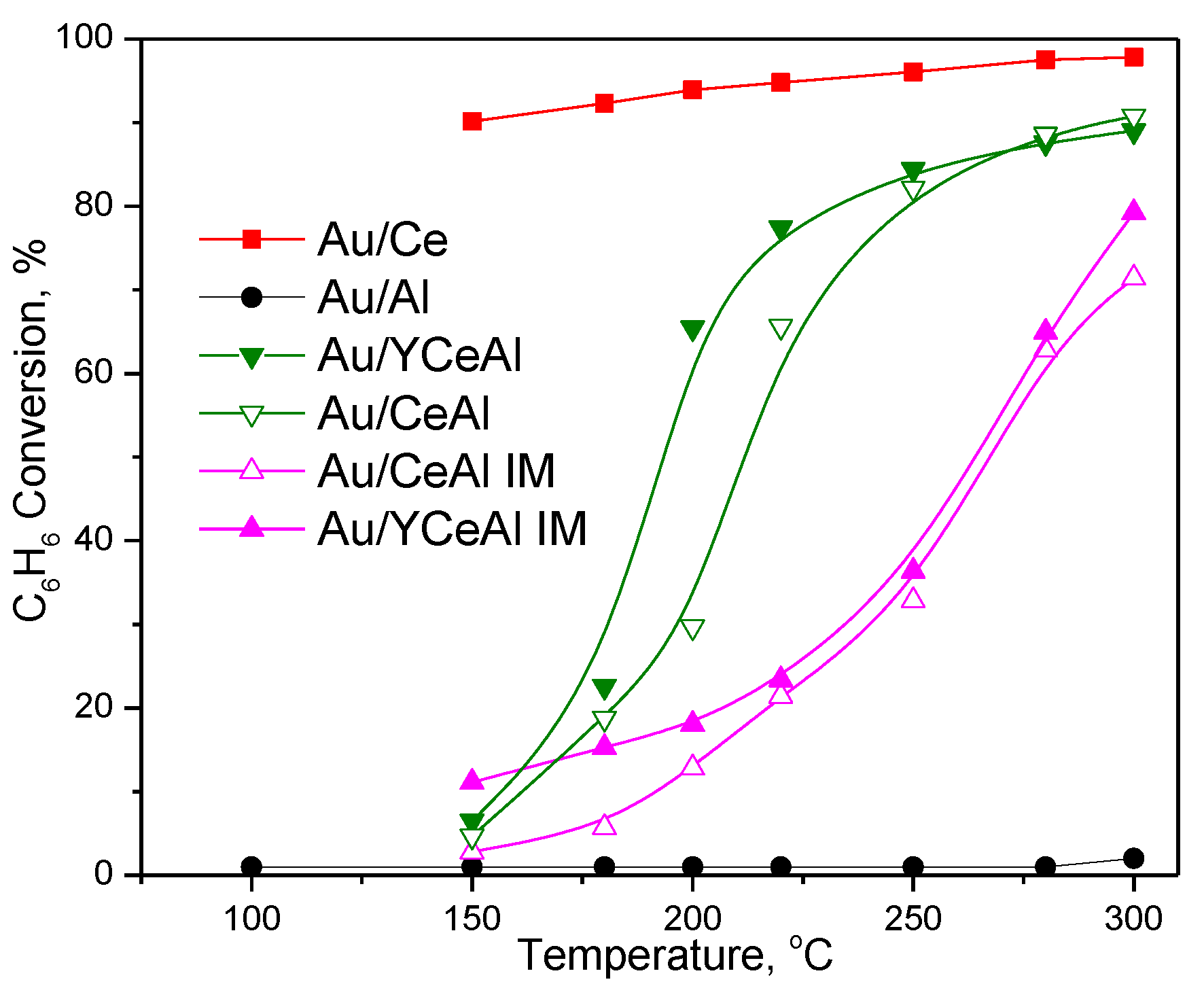
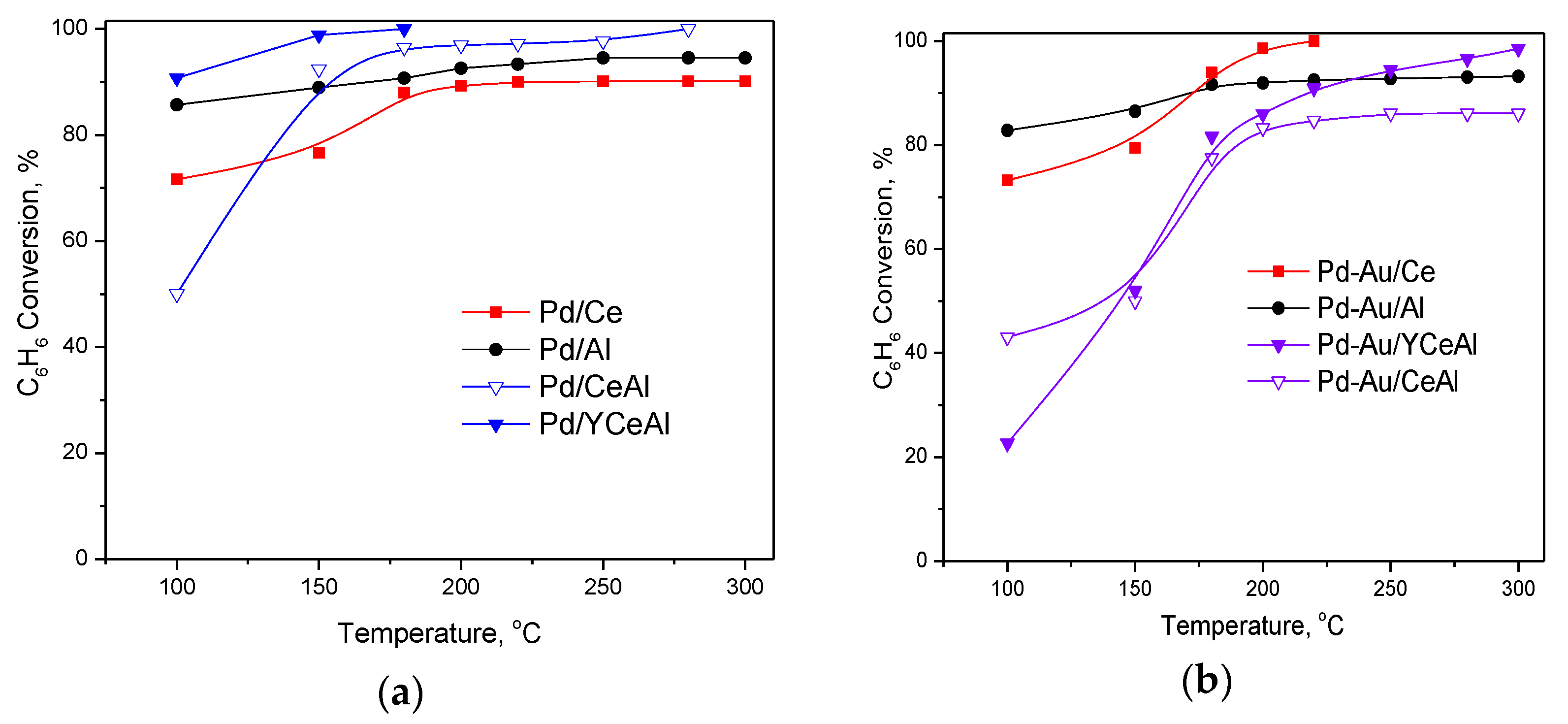
3. Results
3.1. Catalytic Activity Measurements
3.1.1. Effect of Preparation Method of CeO2/Al2O3 and Y-CeO2/Al2O3 on CBO Activity of Gold-Based Catalysts
3.1.2. Role of the Type of Noble Metal on CBO
3.1.3. Role of the Nature of Support
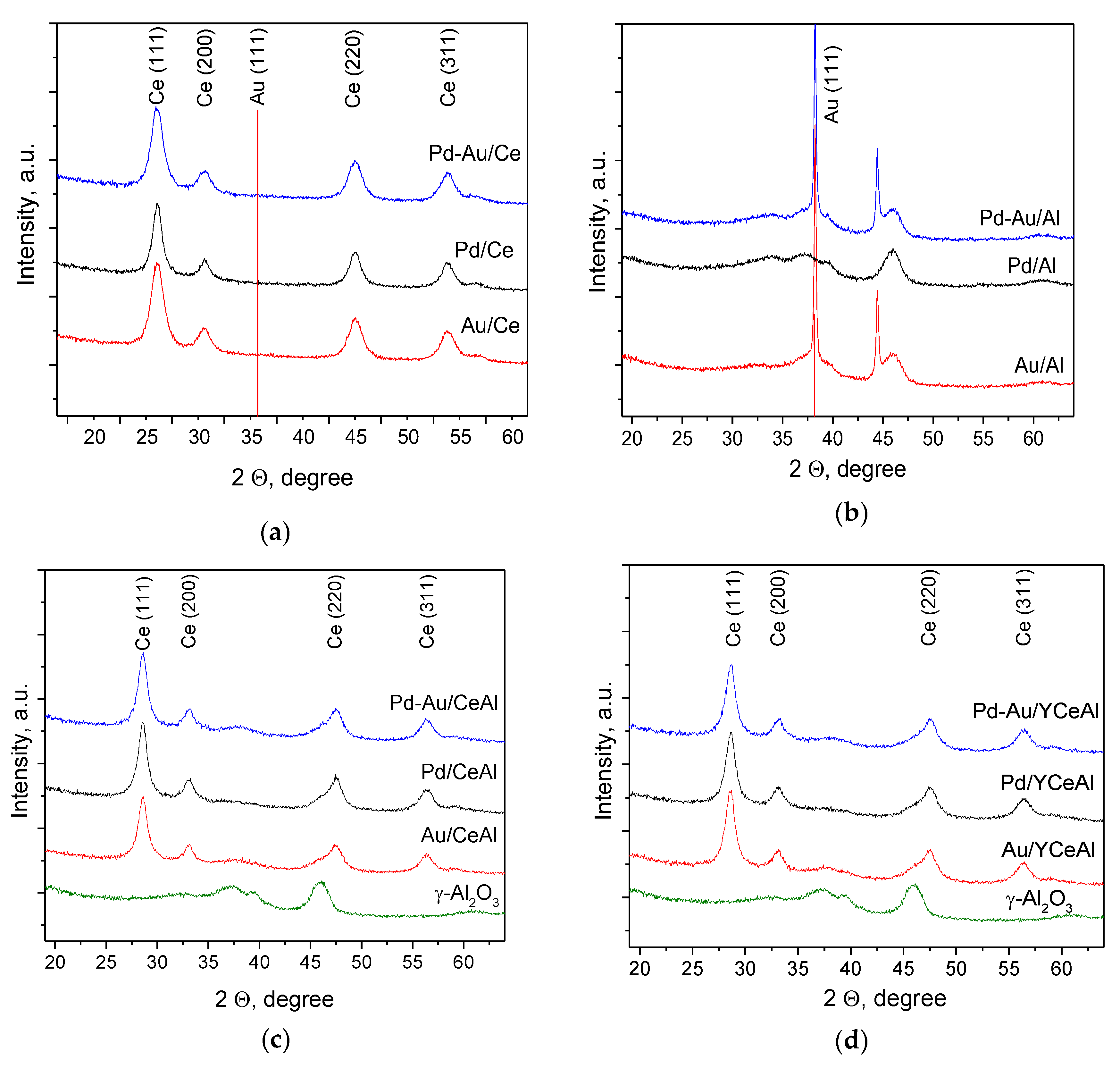
3.2. Catalyst Characterization
3.2.1. Textural Properties
3.2.2. PXRD measurements
3.2.3. XPS Measurements
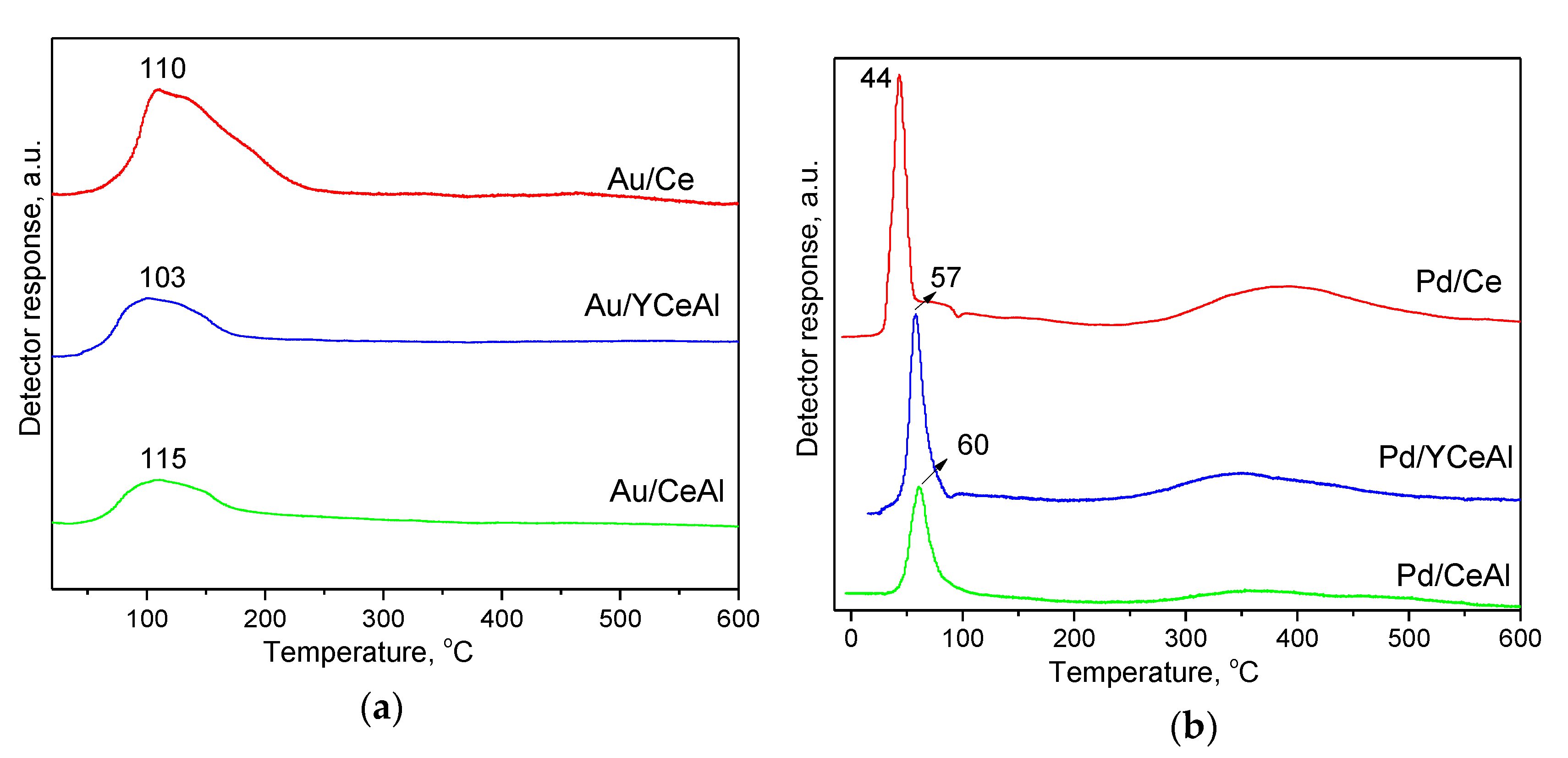
3.2.4. TPR
3.2.5. EPR Investigation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kamal, M.S.; Razzak, S.A.; Hossain, M.M. Catalytic oxidation of volatile organic compounds (VOCs)—A review. Atmos. Environ. 2016, 140, 117–134. [Google Scholar] [CrossRef]
- He, C.; Cheng, J.; Zhang, X.; Douthwaite, M.; Pattisson, S.; Hao, Z. Recent advances in the catalytic oxidation of volatile organic compounds: A review based on pollutant sorts and sources. Chem. Rev. 2019, 119, 4471–4568. [Google Scholar] [CrossRef]
- Zang, M.; Zhao, C.; Wang, Y.; Chen, S. A review of recent advances in catalytic combustion of VOCs on perovskite-type catalysts. J. Saudi Chem. Soc. 2019, 23, 645–654. [Google Scholar] [CrossRef]
- Gelles, T.; Krishnamurthy, A.; Adebayo, B.; Rownaghi, A.; Rezaei, F. Abatement of gaseous volatile organic compounds: A material perspective. Catal. Today 2019. [Google Scholar] [CrossRef]
- Ivanova, S.; Pérez, A.; Centeno, M.; Odriozola, J.A. Structured Catalysts for Volatile Organic Compound Removal. In New and Future Developments in Catalysis: Catalysis for Remediation and Environmental Concerns, 1st ed.; Suib, S., Ed.; Elsevier B.V.: Amsterdam, The Netherlands, 2013; pp. 233–256. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Deng, Y.; Liu, G.; Chen, Y.; Yang, J. Emerging nanostructured materials for the catalytic removal of volatile organic compounds. Nanotechnol. Rev. 2016, 5, 147–181. [Google Scholar] [CrossRef]
- Liotta, L.F. Catalytic oxidation of volatile organic compounds on supported noble metals. Appl. Catal. B Environ. 2010, 100, 403–412. [Google Scholar] [CrossRef]
- Barakat, T.; Rooke, J.C.; Tidahy, H.L.; Hosseini, M.; Cousin, R.; Lamonier, J.-F.; Giraudon, J.-M.; de Weireld, G.; Su, B.-L.; Siffert, S. Noble-metal-based catalysts supported on zeolites and macro-mesoporous metal oxide supports for the total oxidation of volatile organic compounds. ChemSusChem 2011, 4, 1420–1430. [Google Scholar] [CrossRef]
- Andreeva, D.; Tabakova, T.; Ilieva, L.; Naydenov, A.; Mehanjiev, D.; Abrashev, M.V. Nanosize gold catalysts promoted by vanadium oxide supported on titania and zirconia for complete benzene oxidation. Appl. Catal. A Gen. 2001, 209, 291–300. [Google Scholar] [CrossRef]
- Scire, S.; Liotta, L.F. Supported gold catalysts for the total oxidation of volatile organic compounds. Appl. Catal. B Environ. 2012, 125, 222–246. [Google Scholar] [CrossRef]
- Carabineiro, S.A.C.; Chen, X.; Martynyuk, O.; Bogdanchikova, N.; Avalos-Borja, M.; Pestryakov, A.; Tavares, P.B.; Órfão, J.J.M.; Pereira, M.F.R.; Figueiredo, J.L. Gold supported on metal oxides for volatile organic compounds total oxidation. Catal. Today 2015, 244, 103–114. [Google Scholar] [CrossRef]
- Wang, Q.; Yeung, K.L.; Bañares, M.A. Ceria and its related materials for VOC catalytic combustion: A review. Catal. Today 2019. [Google Scholar] [CrossRef]
- Chen, X.; Carabineiro, S.A.C.; Bastos, S.S.T.; Tavares, P.B.; Órfão, J.J.M.; Pereira, M.F.R.; Figueiredo, J.L. Catalytic oxidation of ethyl acetate on cerium-containing mixed oxides. Appl. Catal. A Gen. 2014, 472, 101–112. [Google Scholar] [CrossRef]
- Scirè, S.; Minicò, S.; Crisafulli, C.; Satriano, C.; Pistone, A. Catalytic combustion of volatile organic compounds on gold/cerium oxide catalysts. Appl. Catal. B Environ. 2003, 40, 43–49. [Google Scholar] [CrossRef]
- Scirè, S.; Riccobene, P.; Crisafulli, C. Ceria supported group IB metal catalysts for the combustion of volatile organic compounds and the preferential oxidation of CO. Appl. Catal. B Environ. 2010, 101, 109–117. [Google Scholar] [CrossRef]
- Ilieva, L.; Petrova, P.; Liotta, L.F.; Sobczak, J.W.; Lisowski, W.; Kaszkur, Z.; Munteanu, G.; Tabakova, T. Gold catalysts on Y-doped ceria supports for complete benzene oxidation. Catalysts 2016, 6, 99. [Google Scholar] [CrossRef]
- Xu, L.; Chen, D.; Qu, J.; Wang, L.; Tang, J.; Liu, H.; Yang, J. Replacement reaction-based synthesis of supported palladium catalysts with atomic dispersion for catalytic removal of benzene. J. Mater. Chem. A 2018, 6, 17032–17039. [Google Scholar] [CrossRef]
- Weng, X.; Shi, B.; Liu, A.; Sun, J.; Xiong, Y.; Wan, H.; Zheng, S.; Dong, L.; Chen, Y.-W. Highly dispersed Pd/modified-Al2O3 catalyst on complete oxidation of toluene: Role of basic sites and mechanism insight. Appl. Surf. Sci. 2019, 497, 143747. [Google Scholar] [CrossRef]
- Kim, S.C.; Shim, W.G. Properties and performance of Pd based catalysts for catalytic oxidation of volatile organic compounds. Appl. Catal. B Environ. 2009, 92, 429–436. [Google Scholar] [CrossRef]
- Hosseini, M.; Barakat, T.; Cousin, R.; Aboukaïs, A.; Su, B.-L.; De Weireld, G.; Siffert, S. Catalytic performance of core-shell and alloy Pd-Au nanoparticles for total oxidation of VOC: The effect of metal deposition. Appl. Catal. B Environ. 2012, 111, 218–224. [Google Scholar] [CrossRef]
- Sankar, M.; Dimitratos, N.; Miedziak, P.J.; Wells, P.P.; Kiely, C.J.; Hutchings, G.J. Designing bimetallic catalysts for a green and sustainable future. Chem. Soc. Rev. 2012, 41, 8099–8139. [Google Scholar] [CrossRef]
- Tabakova, T.; Ilieva, L.; Petrova, P.; Venezia, A.M.; Avdeev, G.; Zanella, R.; Karakirova, Y. Complete benzene oxidation over mono and bimetallic Au-Pd catalysts supported on Fe-modified ceria. Chem. Eng. J. 2015, 260, 133–141. [Google Scholar] [CrossRef]
- Ilieva, L.; Venezia, A.M.; Petrova, P.; Pantaleo, G.; Liotta, L.F.; Zanella, R.; Kaszkur, Z.; Tabakova, T. Effect of Y modified ceria support in mono and bimetallic Pd-Au catalysts for complete benzene oxidation. Catalysts 2018, 8, 283. [Google Scholar] [CrossRef]
- Centeno, M.A.; Paulis, M.; Montes, M.; Odriozola, J.A. Catalytic combustion of volatile organic compounds on Au/CeO2/Al2O3 and Au/Al2O3 catalysts. Appl. Catal. A Gen. 2002, 234, 65–78. [Google Scholar] [CrossRef]
- Gluhoi, A.C.; Bogdanchikova, N.; Nieuwenhuys, B.E. The effect of different types of additives on the catalytic activity of Au/Al2O3 in propene total oxidation: Transition metal oxides and ceria. J. Catal. 2005, 229, 154–162. [Google Scholar] [CrossRef]
- Aznárez, A.; Korili, S.A.; Gil, A. The Promoting effect of cerium on the characteristics and catalytic performance of palladium supported on alumina pillared clays for the combustion of propene. Appl. Catal. A Gen. 2014, 474, 95–99. [Google Scholar] [CrossRef]
- Sherwood, P.M.A. Practical Surface Analysis; Briggs, D., Seah, M.P., Eds.; Wiley: New York, NY, USA, 1990; pp. 555–585. [Google Scholar]
- Reina, T.R.; Ivanova, S.; Idakiev, V.; Delgado, J.J.; Ivanov, I.; Tabakova, T.; Centeno, M.A.; Odriozola, J.A. Impact of Ce–Fe synergism on the catalytic behavior of Au/CeO2–FeOx/Al2O3 for pure H2 production. Catal. Sci. Technol. 2013, 3, 779–787. [Google Scholar] [CrossRef]
- Ilieva, L.; Petrova, P.; Pantaleo, G.; Zanella, R.; Sobczak, J.W.; Lisowski, W.; Kaszkur, Z.; Munteanu, G.; Yordanova, I.; Liotta, L.F.; et al. Alumina supported Au/Y-doped ceria catalysts for pure hydrogen production via PROX. Int. J. Hydrogen Energy 2019, 44, 233–245. [Google Scholar] [CrossRef]
- Ilieva, L.; Petrova, P.; Pantaleo, G.; Zanella, R.; Sobczak, J.W.; Lisowski, W.; Ivanov, I.; Kaszkur, Z.; Liotta, L.F.; Venezia, A.M.; et al. Impact of ceria loading on the preferential CO oxidation over gold catalysts on CeO2/Al2O3 and Y-doped CeO2/Al2O3 supports prepared by mechanical mixing. Catal. Today 2019. [Google Scholar] [CrossRef]
- Liotta, L.F.; Pantaleo, G.; Puleo, F.; Venezia, A.M. Au/CeO2-SBA-15 catalysts for CO oxidation: Effect of ceria loading on physicochemical properties and catalytic performances. Catal. Today 2012, 187, 10–19. [Google Scholar] [CrossRef]
- Hosseini, M.; Siffert, S.; Tidahy, H.L.; Cousin, R.; Lamonier, J.-F.; Aboukais, A.; Vantomme, A.; Roussel, M.; Su, B.-L. Promotional effect of gold added to palladium supported on a new mesoporous TiO2 for total oxidation of volatile organic compounds. Catal. Today 2007, 122, 391–396. [Google Scholar] [CrossRef]
- Menegazzo, F.; Burti, P.; Signoretto, M.; Manzoli, M.; Vankova, S.; Boccuzzi, F.; Pinna, F.; Strukul, G. Effect of the addition of Au in zirconia and ceria supported Pd catalysts for the direct synthesis of hydrogen peroxide. J. Catal. 2008, 257, 369–381. [Google Scholar] [CrossRef]
- Sanchez, M.G.; Gazquez, J.L. Oxygen vacancy model in strong metal-support interaction. J. Catal. 1987, 104, 120–135. [Google Scholar] [CrossRef]
- Laachir, A.; Perrichon, V.; Bardi, A.; Lamotte, J.; Catherine, E.; Lavalley, J.C.; El Faallah, J.; Hilaire, L.; Le Normand, F.; Quemere, E. Reduction of CeO2 by hydrogen. Magnetic susceptibility and Fourier-transform infrared, ultraviolet and X-ray photoelectron spectroscopy measurements. J. Chem. Soc. Faraday Trans. 1991, 87, 1601–1609. [Google Scholar] [CrossRef]
- Gil, S.; Garcia-Vargas, J.M.; Liotta, L.F.; Pantaleo, G.; Ousmane, M.; Retailleau, L.; Giroir-Fendler, A. Catalytic Oxidation of Propene over Pd Catalysts Supported on CeO2, TiO2, Al2O3 and M/Al2O3 Oxides (M = Ce, Ti, Fe, Mn). Catalysts 2015, 5, 671–689. [Google Scholar] [CrossRef]
- Green, U.; Aizanshtat, Z.; Ruthstein, S.; Cohen, H. Stable radicals formation in coals undergoing weathering: Effect of coal rank. Phys. Chem. Chem. Phys. 2012, 14, 13046–13052. [Google Scholar] [CrossRef] [PubMed]
- Ge, L.; Chen, T.; Liu, Z.; Chen, F. The effect of gold loading on the catalytic oxidation performance of CeO2/H2O2 system. Catal. Today 2014, 224, 209–215. [Google Scholar] [CrossRef]
- Aboukais, A.; Aouad, S.; Skaf, M.; Hany, S.; Labaki, M.; Cousin, R.; Abi-Aad, E. EPR investigation of the nature of oxygen species present on the surface of gold impregnated cerium oxide. Mater. Chem. Phys. 2016, 170, 285–293. [Google Scholar] [CrossRef]
- Xie, S.H.; Liu, Y.X.; Deng, J.G.; Zhao, X.T.; Yang, J.; Zhang, K.F.; Han, Z.; Dai, H.X. Three-dimensionally ordered macroporous CeO2-supported Pd@Co nanoparticles: Highly active catalysts for methane oxidation. J. Catal. 2016, 342, 17–26. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, B.; Wang, Q.; Li, C.; Hu, W.; Liu, Y.; Jing, P.; Zhao, W.; Zhang, J. Three-dimensionally ordered macroporous Au/CeO2-Co3O4 catalysts with mesoporous walls for enhanced CO preferential oxidation in H2-rich gases. J. Catal. 2012, 296, 65–76. [Google Scholar] [CrossRef]
- Bu, Y.; Chen, Y.; Jiang, G.; Hou, X.; Li, S.; Zhang, Z. Understanding of Au-CeO2 interface and its role in catalytic oxidation of formaldehyde. Appl. Catal. B. Environ. 2020, 260, 118138. [Google Scholar] [CrossRef]
- Huang, H.; Xu, Y.; Feng, Q.; Leung, D.Y.C. Low temperature catalytic oxidation of volatile organic compounds: A review. Catal. Sci. Technol. 2015, 5, 2649–2669. [Google Scholar] [CrossRef]
- Roudesly, F.; Oble, J.; Poli, G. Metal-catalyzed CH activation/functionalization: The fundamentals. J. Mol. Catal. A 2017, 426, 275–296. [Google Scholar] [CrossRef]
- Hea, C.; Li, J.; Li, P.; Chenga, J.; Haoa, Z.; Xu, Z.-P. Comprehensive investigation of Pd/ZSM-5/MCM-48 composite catalysts with enhanced activity and stability for benzene oxidation. Appl. Catal. B Environ. 2010, 96, 466–475. [Google Scholar] [CrossRef]
- Chlala, D.; Labaki, M.; Giraudon, J.-M.; Gardoll, O.; Denicourt-Nowicki, A.; Roucoux, A.; Lamonier, J.-F. Toluene total oxidation over Pd and Au nanoparticles supported on hydroxyapatite. C.R. Chimie 2016, 19, 525–537. [Google Scholar] [CrossRef]


| Sample | SBET (m2·g−1) | Vpore (cm3·g−1) | Dpore (nm) | Dceria (nm) | αceria (nm) |
|---|---|---|---|---|---|
| γ-Al2O3 | 200 | 0.53 | 10.6 | - | - |
| Au/Al | 204 | 0.51 | 10.0 | - | - |
| Pd/Al | 195.8 | 0.52 | 9.7 | - | - |
| Pd-Au/Al | 193.7 | 0.52 | 9.6 | - | - |
| CeO2 | 63.0 | 0.29 | 20.3 | - | - |
| Au/Ce | 65.0 | 0.28 | 17.1 | 4.9 | 0.5418(7) |
| Pd/Ce | 67.6 | 0.28 | 16.5 | 6.2 | 0.5424(4) |
| Pd-Au/Ce | 62.5 | 0.29 | 22.5 | 5.1 | 0.5421(7) |
| CeAl | 165.0 | 0.39 | 9.5 | - | - |
| Au/CeAl | 147.9 | 0.39 | 9.8 | 6.2 | 0.5426(1) |
| Pd/CeAl | 161.2 | 0.45 | 9.5 | 5.8 | 0.5424(1) |
| Pd-Au/CeAl | 151.6 | 0.37 | 9.6 | 5.6 | 0.5425(1) |
| YCeAl | 168.0 | 0.43 | 10.0 | - | - |
| Au/YCeAl | 160.0 | 0.39 | 9.7 | 6.2 | 0.5428(1) |
| Pd/YCeAl | 166.4 | 0.40 | 9.6 | 5.8 | 0.5420(1) |
| Pd-Au/YCeAl | 157.9 | 0.38 | 9.6 | 5.8 | 0.5425(1) |
| Au/CeAl IM | 127.0 (142 *) | 0.27 | 8.6 | 16.8 | 0.5398(1) |
| Au/YCeAl IM | 125.0 (135 *) | 0.27 | 8.7 | 12.6 | 0.5402(7) |
| Catalyst | Ce 3d5/2 (eV) | Au 4f7/2 (eV) | Pd 3d5/2 (eV) | Y 3d5/2 (eV) | Au/Pd (1.1) * | Y/Pd (0.28) * | Y/Au (0.25) * | Ce3+/(Ce3++Ce4+) |
|---|---|---|---|---|---|---|---|---|
| Au/Ce | 882.1 | 84.5 | - | - | - | - | - | 0.30 |
| Au/CeAl | 881.9 | 84.3 | - | - | - | - | - | 0.22 |
| Au/YCeAl | 881.7 | 84.5 | - | 157.8 | - | - | 1.1 | 0.31 |
| Pd/Al | - | - | 336.7 | - | - | - | - | - |
| Pd/Ce | 881.7 | - | 337.1 | - | - | - | - | 0.35 |
| Pd/CeAl | 881.8 | - | 336.6 | - | - | - | - | 0.25 |
| Pd/YCeAl | 881.8 | - | 336.7 | 157.8 | - | 0.21 | - | 0.38 |
| Pd-Au/Al | - | 84.4 | 336.5 | - | 0.21 | - | - | - |
| Pd-Au/Ce | 881.7 | 84.2 | 337.0 | - | 0.46 | - | - | 0.41 |
| Pd-Au/CeAl | 881.9 | 84.2 | 336.7 | - | 0.20 | - | - | 0.29 |
| Pd-Au/YCeAl | 881.7 | 84.3 | 336.7 | 158.2 | 0.18 | 0.23 | 1.2 | 0.33 |
| Catalyst | HC (mmol·g−1) |
|---|---|
| Au/Ce | 0.54 |
| Au/CeAl | 0.26 |
| Au/YCeAl | 0.32 |
| Pd/Ce | 0.43 |
| Pd/CeAl | 0.23 |
| Pd/YCeAl | 0.29 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tabakova, T.; Ilieva, L.; Petrova, P.; Venezia, A.M.; Karakirova, Y.; Liotta, L.F.; Avdeev, G. Complete Benzene Oxidation over Mono and Bimetallic Pd—Au Catalysts on Alumina-Supported Y-Doped Ceria. Appl. Sci. 2020, 10, 1088. https://doi.org/10.3390/app10031088
Tabakova T, Ilieva L, Petrova P, Venezia AM, Karakirova Y, Liotta LF, Avdeev G. Complete Benzene Oxidation over Mono and Bimetallic Pd—Au Catalysts on Alumina-Supported Y-Doped Ceria. Applied Sciences. 2020; 10(3):1088. https://doi.org/10.3390/app10031088
Chicago/Turabian StyleTabakova, Tatyana, Lyuba Ilieva, Petya Petrova, Anna Maria Venezia, Yordanka Karakirova, Leonarda Francesca Liotta, and Georgi Avdeev. 2020. "Complete Benzene Oxidation over Mono and Bimetallic Pd—Au Catalysts on Alumina-Supported Y-Doped Ceria" Applied Sciences 10, no. 3: 1088. https://doi.org/10.3390/app10031088
APA StyleTabakova, T., Ilieva, L., Petrova, P., Venezia, A. M., Karakirova, Y., Liotta, L. F., & Avdeev, G. (2020). Complete Benzene Oxidation over Mono and Bimetallic Pd—Au Catalysts on Alumina-Supported Y-Doped Ceria. Applied Sciences, 10(3), 1088. https://doi.org/10.3390/app10031088