The Canine Gut Health: The Impact of a New Feed Supplement on Microbiota Composition
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Study Design
2.2. Formulated Feed Composition
2.3. Safety and Nutritional Parameters
2.4. Fecal Sample Collection
2.5. Fecal DNA Extraction, PCR Amplification, and Amplicon Target Sequencing
2.6. Computation and Sequence Analysis
2.7. Taxonomic Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Giulio, P. Intestinal Dysbiosis: Definition, Clinical Implications, and Proposed Treatment Protocol (Perrotta Protocol for Clinical Management of Intestinal Dysbiosis, PID) for the Management and Resolution of Persistent or Chronic Dysbiosis. Arch. Clin. Gastroenterol. 2021, 7, 056–063. [Google Scholar] [CrossRef]
- Meineri, G.; Martello, E.; Atuahene, D.; Miretti, S.; Stefanon, B.; Sandri, M.; Biasato, I.; Corvaglia, M.R.; Ferrocino, I.; Cocolin, L.S. Effects of Saccharomyces boulardii Supplementation on Nutritional Status, Fecal Parameters, Microbiota, and Mycobiota in Breeding Adult Dogs. Vet. Sci. China 2022, 9, 389. [Google Scholar] [CrossRef] [PubMed]
- Suchodolski, J.S. Analysis of the Gut Microbiome in Dogs and Cats. Vet. Clin. Pathol. 2022, 50 (Suppl. 1), 6–17. [Google Scholar] [CrossRef] [PubMed]
- Jandhyala, S.M.; Talukdar, R.; Subramanyam, C.; Vuyyuru, H.; Sasikala, M.; Nageshwar Reddy, D. Role of the Normal Gut Microbiota. World J. Gastroenterol. 2015, 21, 8787–8803. [Google Scholar] [CrossRef] [PubMed]
- Pilla, R.; Suchodolski, J.S. The Role of the Canine Gut Microbiome and Metabolome in Health and Gastrointestinal Disease. Front. Vet. Sci. 2019, 6, 498. [Google Scholar] [CrossRef] [PubMed]
- Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M.-C.C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Corral, G.H.; et al. Correction to: Microbiome Definition Re-Visited: Old Concepts and New Challenges. Microbiome 2020, 8, 119. [Google Scholar] [CrossRef] [PubMed]
- Garrigues, Q.; Apper, E.; Chastant, S.; Mila, H. Gut Microbiota Development in the Growing Dog: A Dynamic Process Influenced by Maternal, Environmental and Host Factors. Front. Vet. Sci. 2022, 9, 964649. [Google Scholar] [CrossRef] [PubMed]
- Rhimi, S.; Kriaa, A.; Mariaule, V.; Saidi, A.; Drut, A.; Jablaoui, A.; Akermi, N.; Maguin, E.; Hernandez, J.; Rhimi, M. The Nexus of Diet, Gut Microbiota and Inflammatory Bowel Diseases in Dogs. Metabolites 2022, 12, 1176. [Google Scholar] [CrossRef]
- Ramirez, J.; Guarner, F.; Bustos Fernandez, L.; Maruy, A.; Sdepanian, V.L.; Cohen, H. Antibiotics as Major Disruptors of Gut Microbiota. Front. Cell. Infect. Microbiol. 2020, 10, 572912. [Google Scholar] [CrossRef] [PubMed]
- Prescott, J.F.; Hanna, W.J.B.; Reid-Smith, R.; Drost, K. Antimicrobial Drug Use and Resistance in Dogs. Can. Vet. J. 2002, 43, 107–116. [Google Scholar]
- Caneschi, A.; Bardhi, A.; Barbarossa, A.; Zaghini, A. The Use of Antibiotics and Antimicrobial Resistance in Veterinary Medicine, a Complex Phenomenon: A Narrative Review. Antibiotics 2023, 12, 487. [Google Scholar] [CrossRef] [PubMed]
- Ishak, K.A.; Mohamad Annuar, M.S.; Ahmad, N. Chapter 9—Nano-Delivery Systems for Nutraceutical Application. In Nanotechnology Applications in Food; Oprea, A.E., Grumezescu, A.M., Eds.; Academic Press: Cambridge, MA, USA, 2017; pp. 179–202. ISBN 9780128119426. [Google Scholar]
- Kelsey, N.A.; Wilkins, H.M.; Linseman, D.A. Nutraceutical Antioxidants as Novel Neuroprotective Agents. Molecules 2010, 15, 7792–7814. [Google Scholar] [CrossRef]
- Atuahene, D.; Costale, A.; Martello, E.; Mannelli, A.; Radice, E.; Ribaldone, D.G.; Chiofalo, B.; Stefanon, B.; Meineri, G. A Supplement with Bromelain, Lentinula Edodes, and Quercetin: Antioxidant Capacity and Effects on Morphofunctional and Fecal Parameters (Calprotectin, Cortisol, and Intestinal Fermentation Products) in Kennel Dogs. Vet. Sci. 2023, 10, 486. [Google Scholar] [CrossRef]
- Pavan, R.; Jain, S.; Shraddha; Kumar, A. Properties and Therapeutic Application of Bromelain: A Review. Biotechnol. Res. Int. 2012, 2012, 976203. [Google Scholar] [CrossRef]
- Kostiuchenko, O.; Kravchenko, N.; Markus, J.; Burleigh, S.; Fedkiv, O.; Cao, L.; Letasiova, S.; Skibo, G.; Fåk Hållenius, F.; Prykhodko, O. Effects of Proteases from Pineapple and Papaya on Protein Digestive Capacity and Gut Microbiota in Healthy C57BL/6 Mice and Dose-Manner Response on Mucosal Permeability in Human Reconstructed Intestinal 3D Tissue Model. Metabolites 2022, 12, 1027. [Google Scholar] [CrossRef] [PubMed]
- Anand David, A.V.; Arulmoli, R.; Parasuraman, S. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar]
- Porras, D.; Nistal, E.; Martínez-Flórez, S.; Pisonero-Vaquero, S.; Olcoz, J.L.; Jover, R.; González-Gallego, J.; García-Mediavilla, M.V.; Sánchez-Campos, S. Protective Effect of Quercetin on High-Fat Diet-Induced Non-Alcoholic Fatty Liver Disease in Mice Is Mediated by Modulating Intestinal Microbiota Imbalance and Related Gut-Liver Axis Activation. Free Radic. Biol. Med. 2017, 102, 188–202. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xie, W.; Sun, H.; Cao, K.; Yang, X. Effect of the Structural Characterization of the Fungal Polysaccharides on Their Immunomodulatory Activity. Int. J. Biol. Macromol. 2020, 164, 3603–3610. [Google Scholar] [CrossRef] [PubMed]
- Ziaja-Sołtys, M.; Radzki, W.; Nowak, J.; Topolska, J.; Jabłońska-Ryś, E.; Sławińska, A.; Skrzypczak, K.; Kuczumow, A.; Bogucka-Kocka, A. Processed Fruiting Bodies of Lentinus Edodes as a Source of Biologically Active Polysaccharides. NATO Adv. Sci. Inst. Ser. E Appl. Sci. 2020, 10, 470. [Google Scholar] [CrossRef]
- Hatvani, N. Antibacterial Effect of the Culture Fluid of Lentinus Edodes Mycelium Grown in Submerged Liquid Culture. Int. J. Antimicrob. Agents 2001, 17, 71–74. [Google Scholar] [CrossRef]
- Handayani, D.; Chen, J.; Meyer, B.J.; Huang, X.F. Dietary Shiitake Mushroom (Lentinus Edodes) Prevents Fat Deposition and Lowers Triglyceride in Rats Fed a High-Fat Diet. J. Obes. 2011, 2011, 258051. [Google Scholar] [CrossRef] [PubMed]
- Kitzberger, C.S.G.; Smânia, A.; Pedrosa, R.C.; Ferreira, S.R.S. Antioxidant and Antimicrobial Activities of Shiitake (Lentinula Edodes) Extracts Obtained by Organic Solvents and Supercritical Fluids. J. Food Eng. 2007, 80, 631–638. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Liang, Q.; Fenix, K.; Tamang, B.; Hauben, E.; Ma, L.; Zhang, W. Combining the Anticancer and Immunomodulatory Effects of Astragalus and Shiitake as an Integrated Therapeutic Approach. Nutrients 2021, 13, 2564. [Google Scholar] [CrossRef] [PubMed]
- Jayachandran, M.; Xiao, J.; Xu, B. A Critical Review on Health Promoting Benefits of Edible Mushrooms through Gut Microbiota. Int. J. Mol. Sci. 2017, 18, 1934. [Google Scholar] [CrossRef] [PubMed]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of General 16S Ribosomal RNA Gene PCR Primers for Classical and Next-Generation Sequencing-Based Diversity Studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Zuniga-Chaves, I.; Eggers, S.; Kates, A.E.; Safdar, N.; Suen, G.; Malecki, K.M.C. Neighborhood Socioeconomic Status Is Associated with Low Diversity Gut Microbiomes and Multi-Drug Resistant Microorganism Colonization. NPJ Biofilms Microbiomes 2023, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, Interactive, Scalable and Extensible Microbiome Data Science Using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- R Core Team, R. Others R: A Language and Environment for Statistical Computing; GBIF: Copenhagen, Denmark, 2013. [Google Scholar]
- McMurdie, P.J.; Holmes, S. Phyloseq: An R Package for Reproducible Interactive Analysis and Graphics of Microbiome Census Data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef]
- Oksanen, J.; Kindt, R.; Legendre, P.; O’Hara, B.; Stevens, M.H.H.; Oksanen, M.J.; Suggests, M. The Vegan Package. Community Ecol. Package 2007, 10, 719. [Google Scholar]
- Dixon, P. VEGAN, a Package of R Functions for Community Ecology. J. Veg. Sci. 2003, 14, 927–930. [Google Scholar] [CrossRef]
- Lin, H.; Peddada, S.D. Analysis of Compositions of Microbiomes with Bias Correction. Nat. Commun. 2020, 11, 3514. [Google Scholar] [CrossRef] [PubMed]
- Sunvold, G.D.; Hussein, H.S.; Fahey, G.C., Jr.; Merchen, N.R.; Reinhart, G.A. In Vitro Fermentation of Cellulose, Beet Pulp, Citrus Pulp, and Citrus Pectin Using Fecal Inoculum from Cats, Dogs, Horses, Humans, and Pigs and Ruminal Fluid from Cattle. J. Anim. Sci. 1995, 73, 3639–3648. [Google Scholar] [CrossRef] [PubMed]
- Vinolo, M.A.R.; Rodrigues, H.G.; Hatanaka, E.; Sato, F.T.; Sampaio, S.C.; Curi, R. Suppressive Effect of Short-Chain Fatty Acids on Production of Proinflammatory Mediators by Neutrophils. J. Nutr. Biochem. 2011, 22, 849–855. [Google Scholar] [CrossRef] [PubMed]
- Suchodolski, J.S. Companion Animals Symposium: Microbes and Gastrointestinal Health of Dogs and Cats. J. Anim. Sci. 2011, 89, 1520–1530. [Google Scholar] [CrossRef] [PubMed]
- Panasevich, M.R.; Daristotle, L.; Quesnell, R.; Reinhart, G.A.; Frantz, N.Z. Altered Fecal Microbiota, IgA, and Fermentative End-Products in Adult Dogs Fed Prebiotics and a Nonviable Lactobacillus Acidophilus. J. Anim. Sci. 2021, 99, skab347. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yang, K.; Jian, S.; Xin, Z.; Wen, C.; Zhang, L.; Huang, J.; Deng, B.; Deng, J. Effects of Softening Dry Food with Water on Stress Response, Intestinal Microbiome, and Metabolic Profile in Beagle Dogs. Metabolites 2022, 12, 1124. [Google Scholar] [CrossRef] [PubMed]
- Roager, H.M.; Licht, T.R.; Poulsen, S.K.; Larsen, T.M.; Bahl, M.I. Microbial Enterotypes, Inferred by the Prevotella-to-Bacteroides Ratio, Remained Stable during a 6-Month Randomized Controlled Diet Intervention with the New Nordic Diet. Appl. Environ. Microbiol. 2014, 80, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Tennoune, N.; Andriamihaja, M.; Blachier, F. Production of Indole and Indole-Related Compounds by the Intestinal Microbiota and Consequences for the Host: The Good, the Bad, and the Ugly. Microorganisms 2022, 10, 930. [Google Scholar] [CrossRef] [PubMed]
- Bansal, T.; Englert, D.; Lee, J.; Hegde, M.; Wood, T.K.; Jayaraman, A. Differential Effects of Epinephrine, Norepinephrine, and Indole on Escherichia Coli O157:H7 Chemotaxis, Colonization, and Gene Expression. Infect. Immun. 2007, 75, 4597–4607. [Google Scholar] [CrossRef]
- Ledala, N.; Malik, M.; Rezaul, K.; Paveglio, S.; Provatas, A.; Kiel, A.; Caimano, M.; Zhou, Y.; Lindgren, J.; Krasulova, K.; et al. Bacterial Indole as a Multifunctional Regulator of Klebsiella Oxytoca Complex Enterotoxicity. MBio 2022, 13, e0375221. [Google Scholar] [CrossRef]
- Li, G.; Young, K.D. Indole Production by the Tryptophanase TnaA in Escherichia Coli Is Determined by the Amount of Exogenous Tryptophan. Microbiology 2013, 159, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Mir, H.-D.; Milman, A.; Monnoye, M.; Douard, V.; Philippe, C.; Aubert, A.; Castanon, N.; Vancassel, S.; Guérineau, N.C.; Naudon, L.; et al. The Gut Microbiota Metabolite Indole Increases Emotional Responses and Adrenal Medulla Activity in Chronically Stressed Male Mice. Psychoneuroendocrinology 2020, 119, 104750. [Google Scholar] [CrossRef] [PubMed]
- Jaglin, M.; Rhimi, M.; Philippe, C.; Pons, N.; Bruneau, A.; Goustard, B.; Daugé, V.; Maguin, E.; Naudon, L.; Rabot, S. Indole, a Signaling Molecule Produced by the Gut Microbiota, Negatively Impacts Emotional Behaviors in Rats. Front. Neurosci. 2018, 12, 216. [Google Scholar] [CrossRef] [PubMed]
- Ayling, R.M.; Kok, K. Fecal Calprotectin. Adv. Clin. Chem. 2018, 87, 161–190. [Google Scholar]
- Herstad, K.M.V.; Vinje, H.; Skancke, E.; Næverdal, T.; Corral, F.; Llarena, A.-K.; Heilmann, R.M.; Suchodolski, J.S.; Steiner, J.M.; Nyquist, N.F. Effects of Canine-Obtained Lactic-Acid Bacteria on the Fecal Microbiota and Inflammatory Markers in Dogs Receiving Non-Steroidal Anti-Inflammatory Treatment. Animals 2022, 12, 2519. [Google Scholar] [CrossRef]
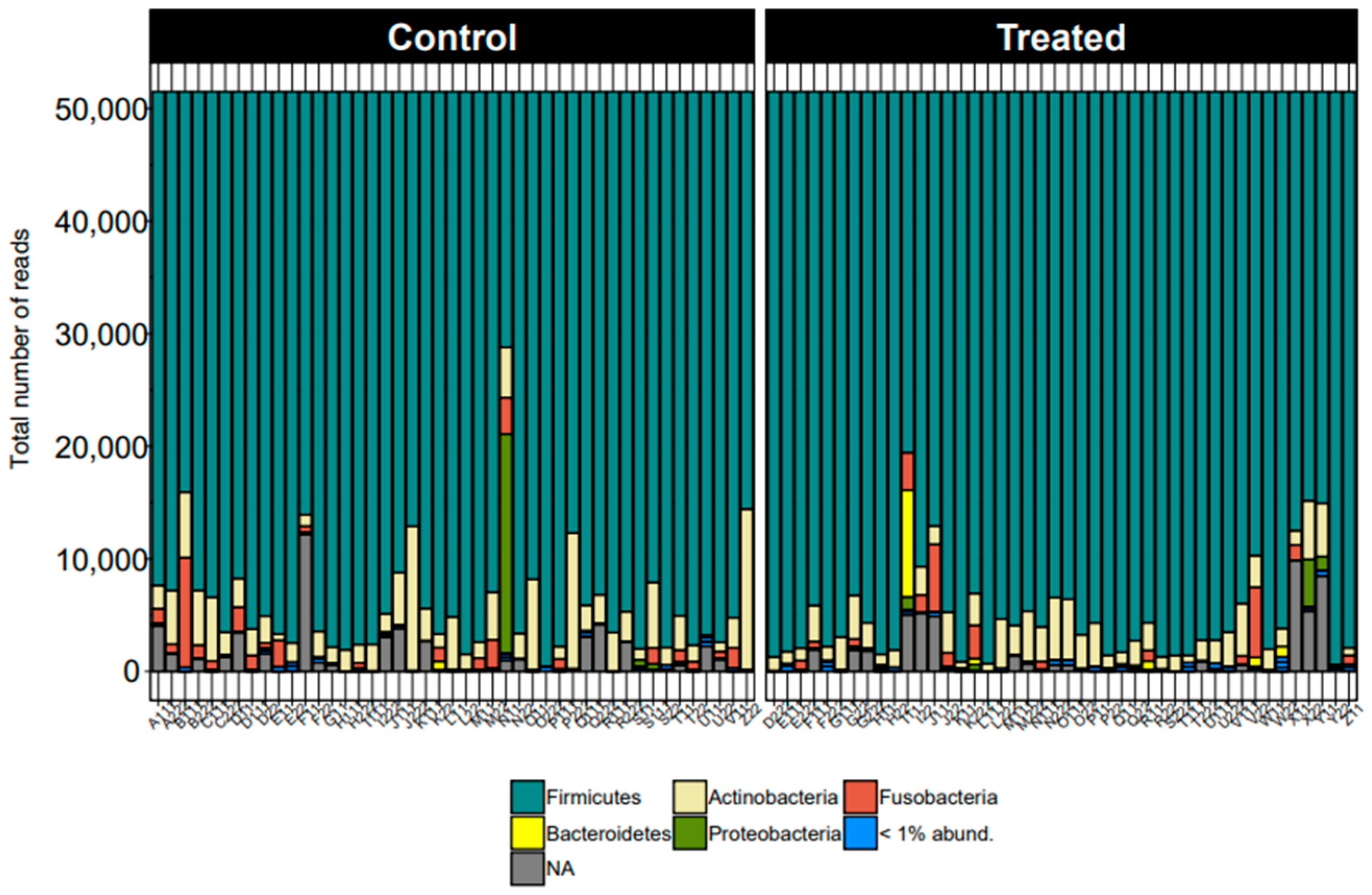


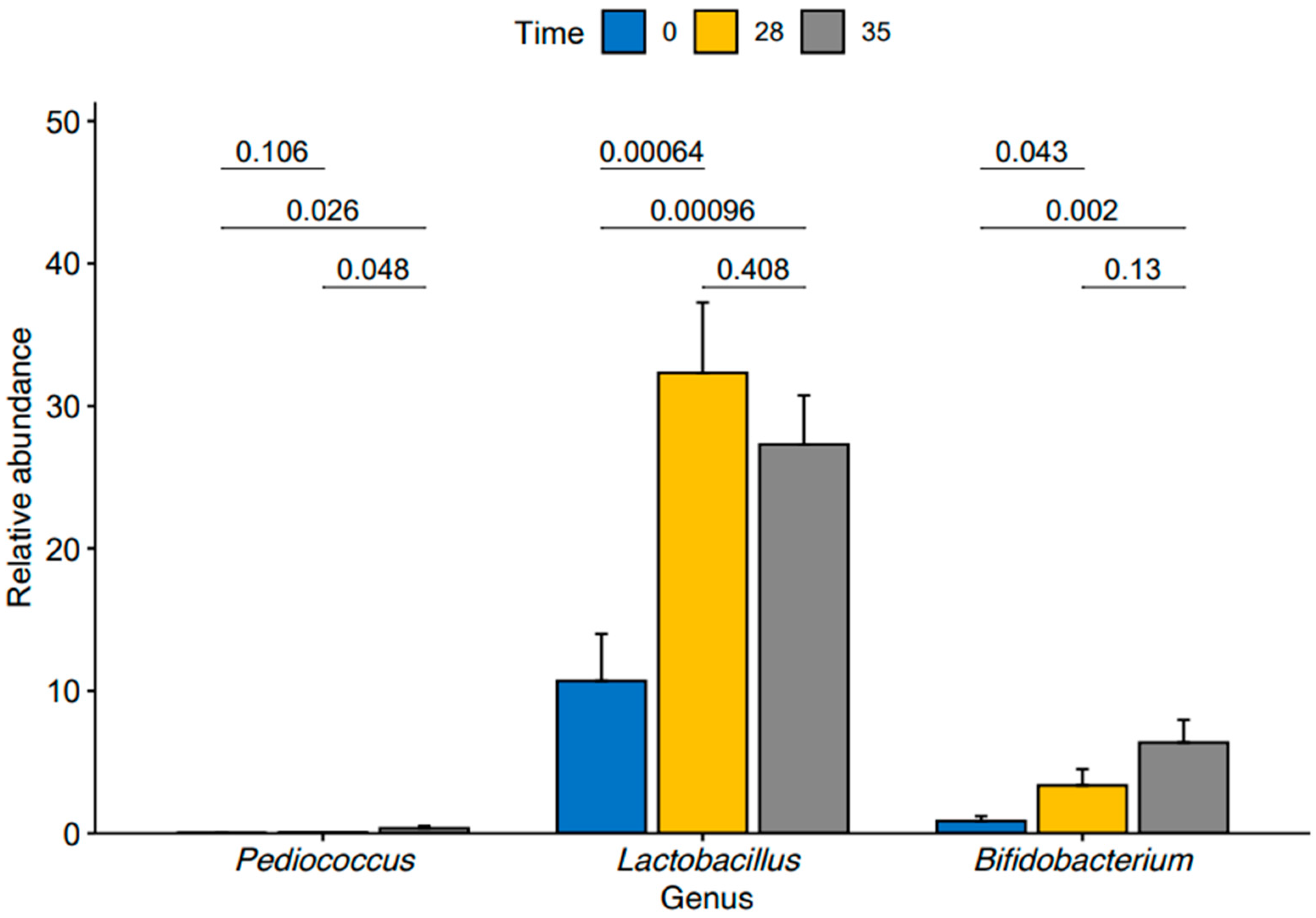
| Taxonomic Classification | Higher Abundance Time Point | Beta Coefficient | Standard Deviation | W Statistic | p-Value | Corrected p-Value | ASV Code |
|---|---|---|---|---|---|---|---|
| Fusobacteria_unclassified | T0 | −0.047 | 0.016 | −2.972 | 0.003 | 0.050 | ASV334 |
| Fusobacteria_unclassified | T0 | −0.070 | 0.023 | −3.025 | 0.002 | 0.049 | ASV029 |
| Bifidobacterium sp. | T28 | 0.065 | 0.014 | 4.698 | 0.000 | 0.000 | ASV073 |
| Bifidobacterium animalis | T28 | 0.089 | 0.023 | 3.925 | 0.000 | 0.005 | ASV117 |
| Bifidobacterium breve | T28 | 0.070 | 0.023 | 2.993 | 0.003 | 0.050 | ASV007 |
| Lactobacillaceae_unclassified | T28 | 0.104 | 0.022 | 4.697 | 0.000 | 0.000 | ASV256 |
| Lactobacillaceae_unclassified | T28 | 0.063 | 0.017 | 3.827 | 0.000 | 0.006 | ASV585 |
| Lactobacillaceae_unclassified | T28 | 0.111 | 0.025 | 4.489 | 0.000 | 0.001 | ASV098 |
| Lactobacillaceae hamsteri | T28 | 0.098 | 0.027 | 3.568 | 0.000 | 0.012 | ASV440 |
| Anaerorhabdus furcosa | T28 | 0.055 | 0.016 | 3.318 | 0.001 | 0.022 | ASV269 |
| Bacillaceae_unclassified | T28 | 0.047 | 0.016 | 2.847 | 0.004 | 0.070 | ASV706 |
| Lactobacillus hamsteri | T28 | 0.064 | 0.023 | 2.715 | 0.007 | 0.099 | ASV408 |
| Peptococcus sp. | T0 | −0.059 | 0.016 | −3.590 | 0.000 | 0.012 | ASV519 |
| Peptostreptococcaceae_unclassified | T28 | 0.050 | 0.015 | 3.387 | 0.001 | 0.019 | ASV003 |
| Peptostreptococcaceae_unclassified | T28 | 0.046 | 0.015 | 3.140 | 0.002 | 0.037 | ASV006 |
| Dorea longicatena | T0 | −0.053 | 0.015 | −3.418 | 0.001 | 0.019 | ASV262 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atuahene, D.; Zuniga-Chaves, I.; Martello, E.; Stefanon, B.; Suen, G.; Balouei, F.; Meineri, G. The Canine Gut Health: The Impact of a New Feed Supplement on Microbiota Composition. Animals 2024, 14, 1189. https://doi.org/10.3390/ani14081189
Atuahene D, Zuniga-Chaves I, Martello E, Stefanon B, Suen G, Balouei F, Meineri G. The Canine Gut Health: The Impact of a New Feed Supplement on Microbiota Composition. Animals. 2024; 14(8):1189. https://doi.org/10.3390/ani14081189
Chicago/Turabian StyleAtuahene, David, Ibrahim Zuniga-Chaves, Elisa Martello, Bruno Stefanon, Garret Suen, Fatemeh Balouei, and Giorgia Meineri. 2024. "The Canine Gut Health: The Impact of a New Feed Supplement on Microbiota Composition" Animals 14, no. 8: 1189. https://doi.org/10.3390/ani14081189
APA StyleAtuahene, D., Zuniga-Chaves, I., Martello, E., Stefanon, B., Suen, G., Balouei, F., & Meineri, G. (2024). The Canine Gut Health: The Impact of a New Feed Supplement on Microbiota Composition. Animals, 14(8), 1189. https://doi.org/10.3390/ani14081189








