Equine Stomach Development in the Foetal Period of Prenatal Life—An Immunohistochemical Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
- The first age group (4th–5th month of gestation).
- The second age group (7th–8th month of gestation).
- The third age group (10th–11th month of gestation).
2.1. Immunohistochemical Staining
2.2. Immunohistochemical Reaction Scoring
2.3. Statistical Analysis
3. Results
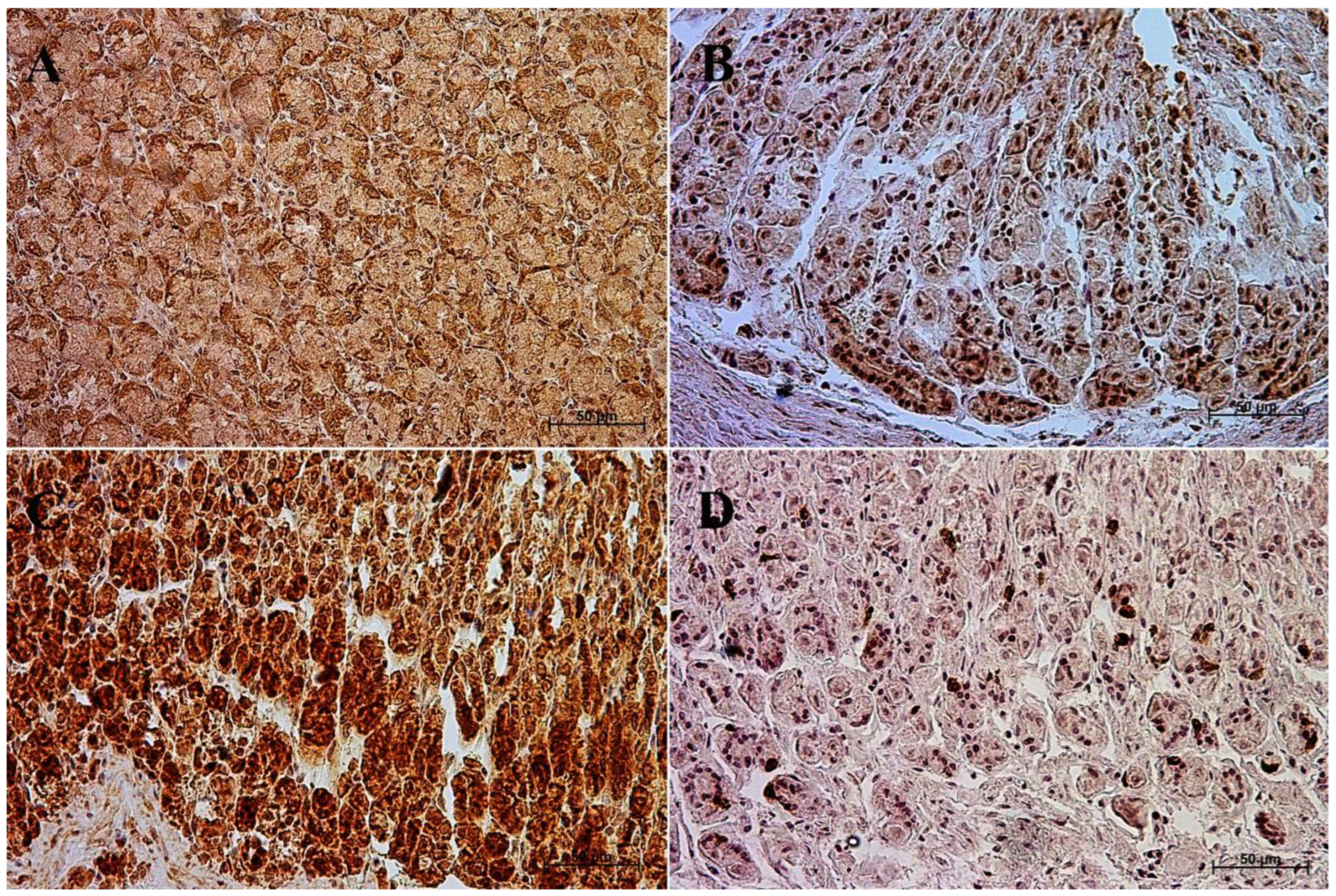
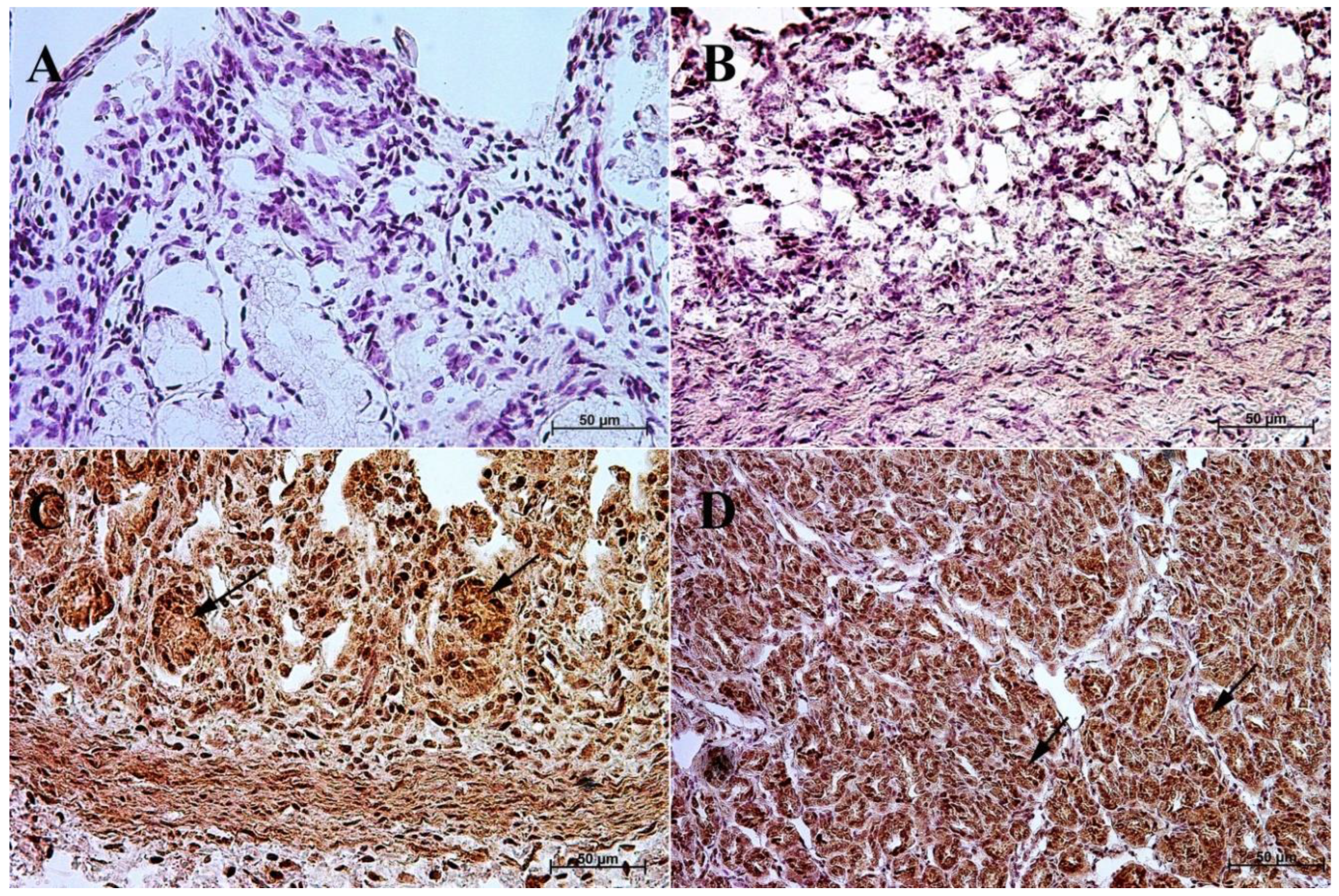
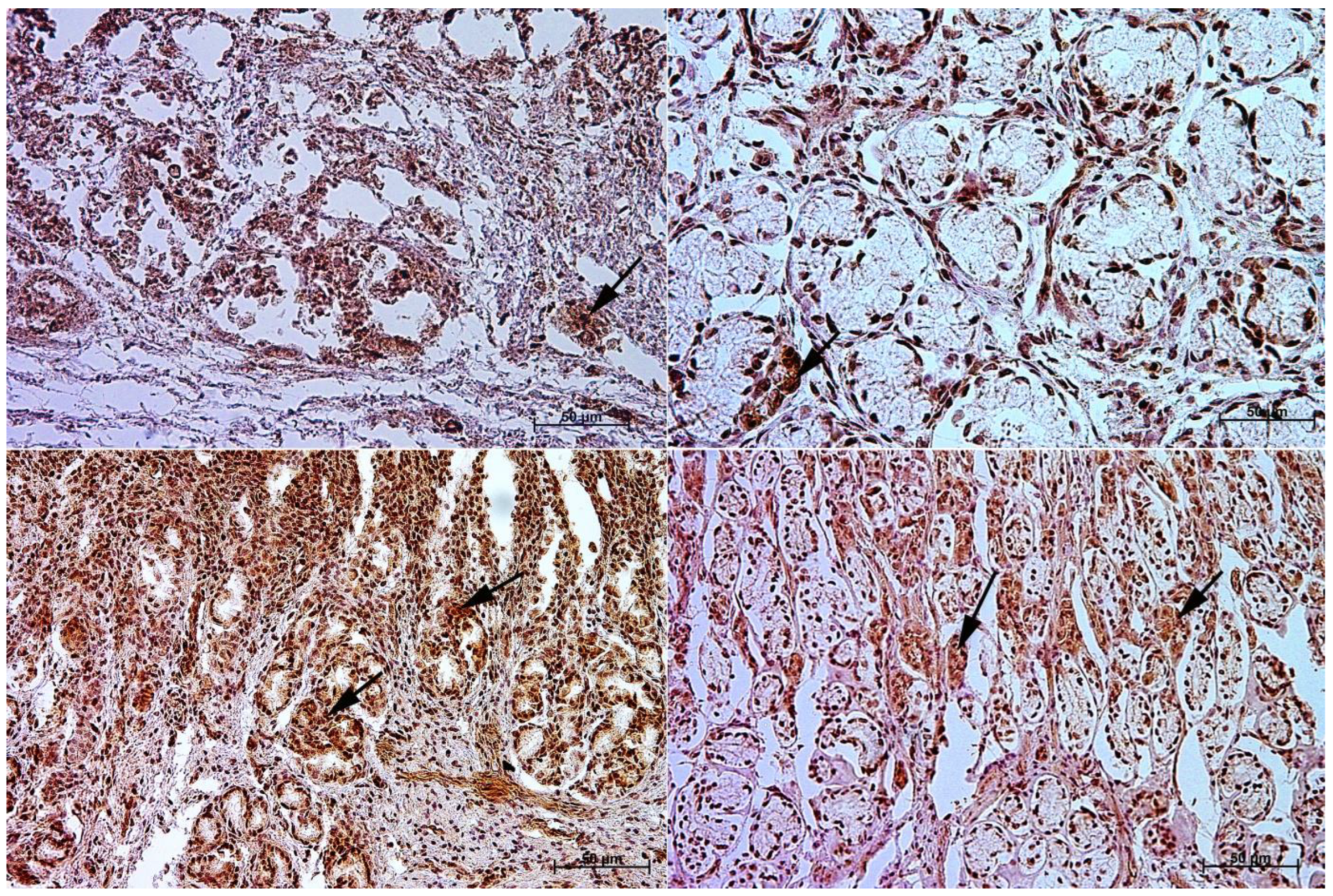
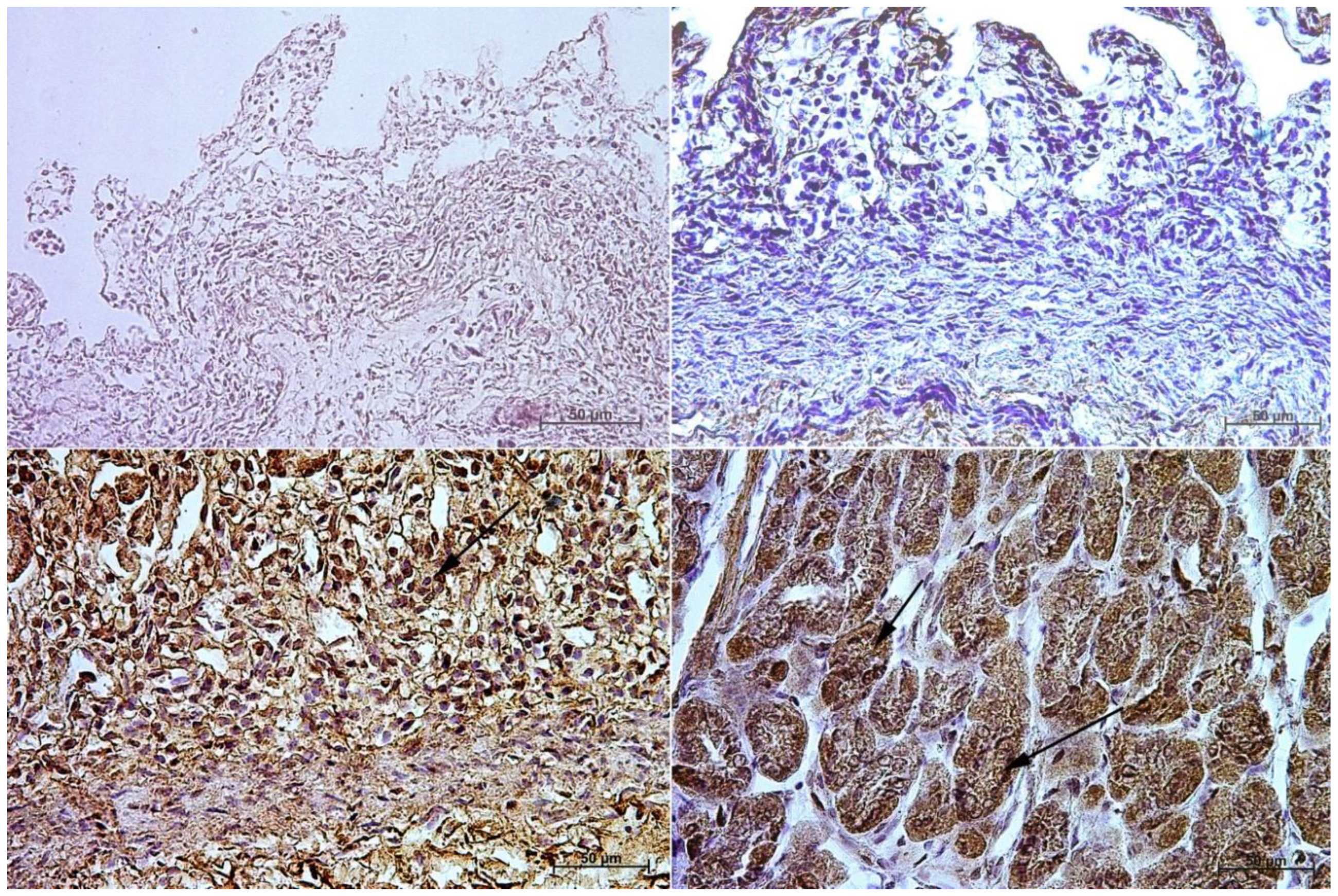
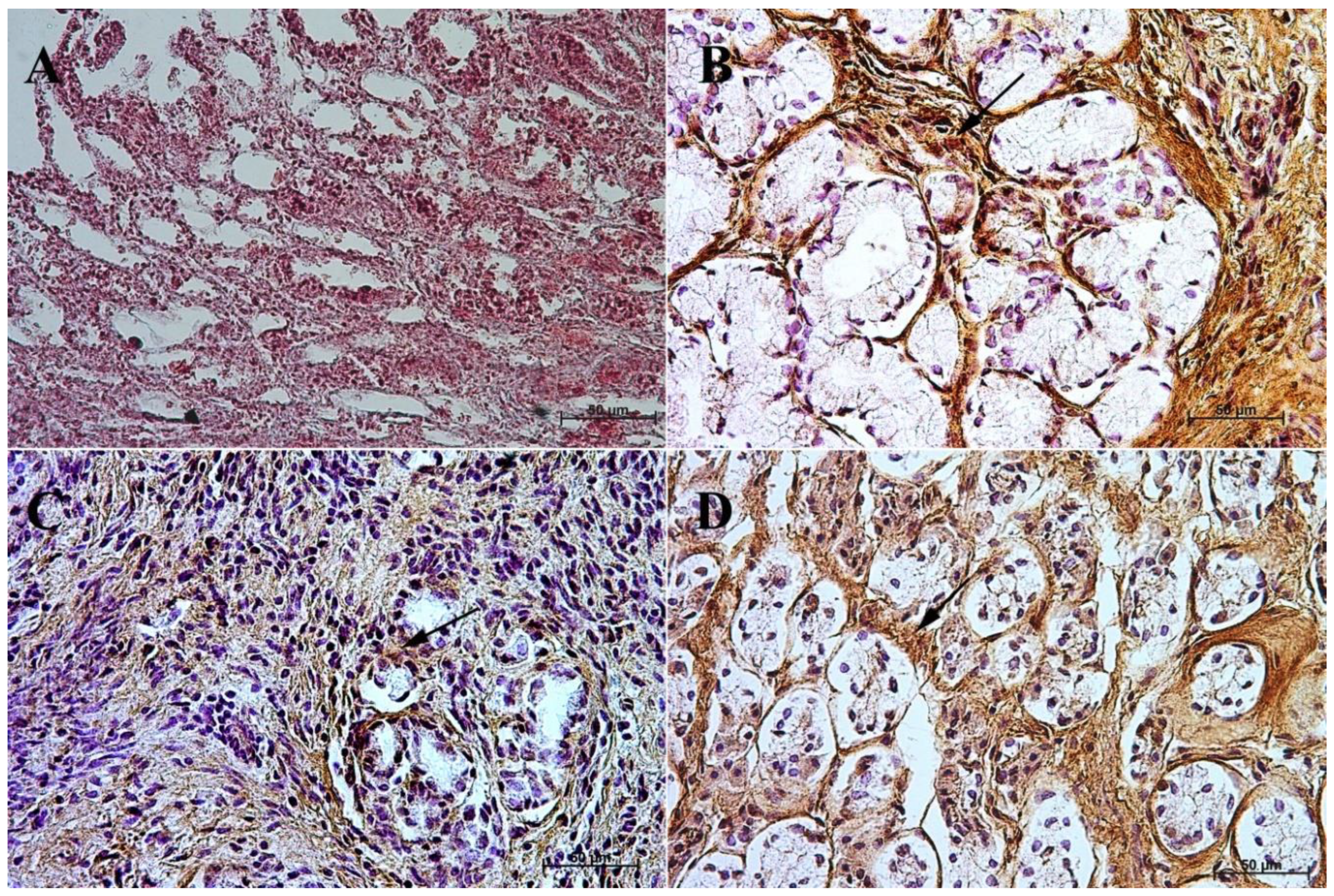
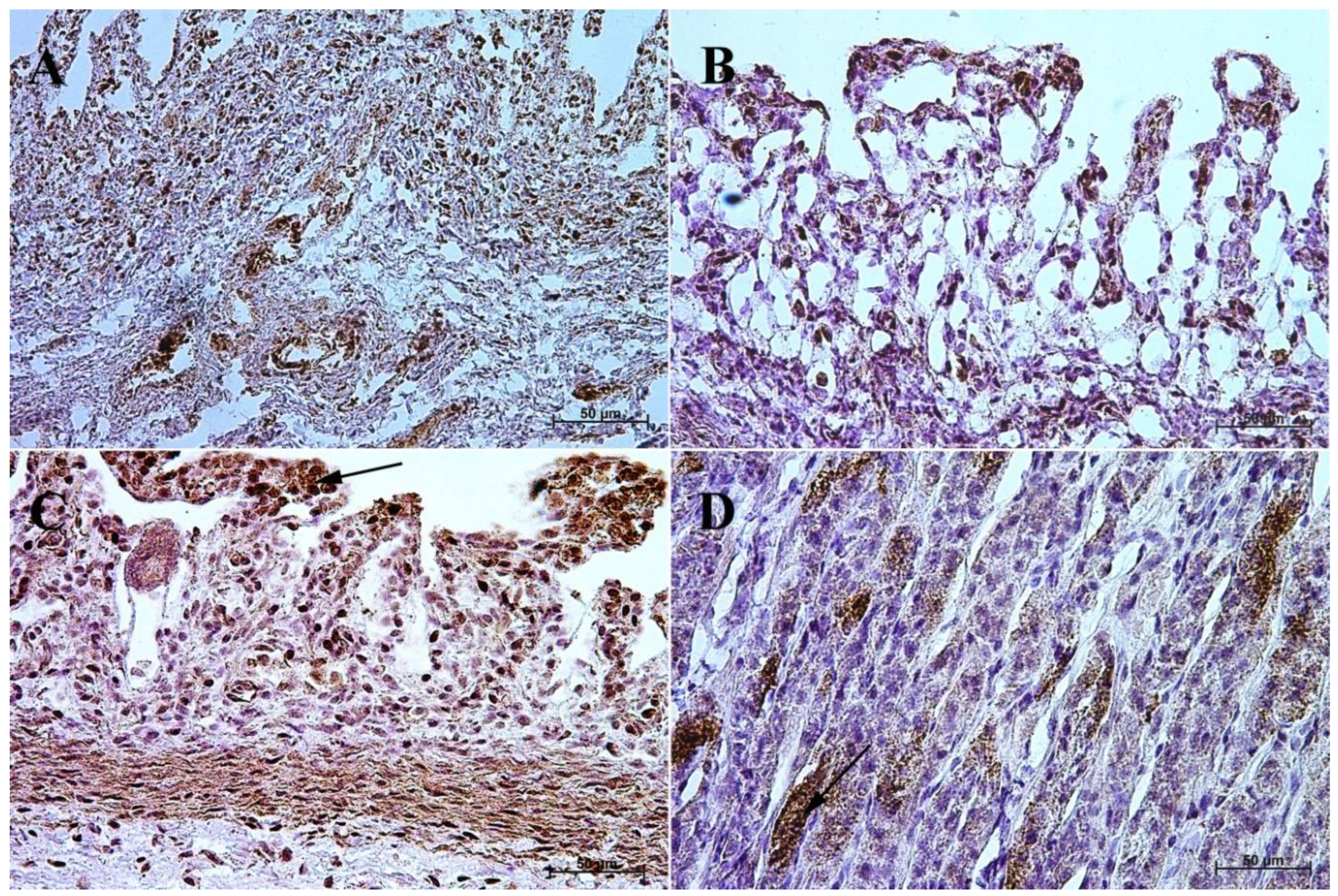
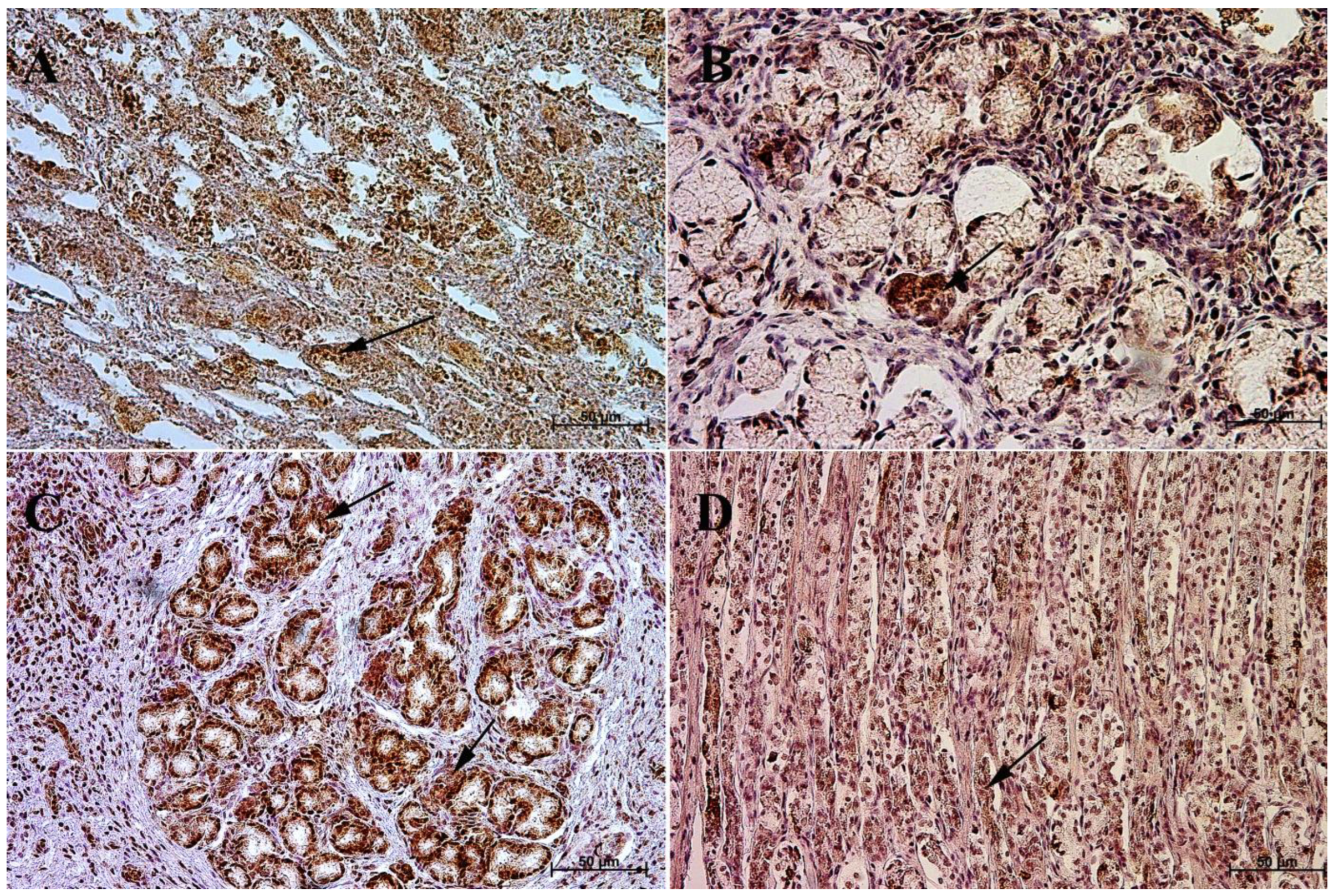
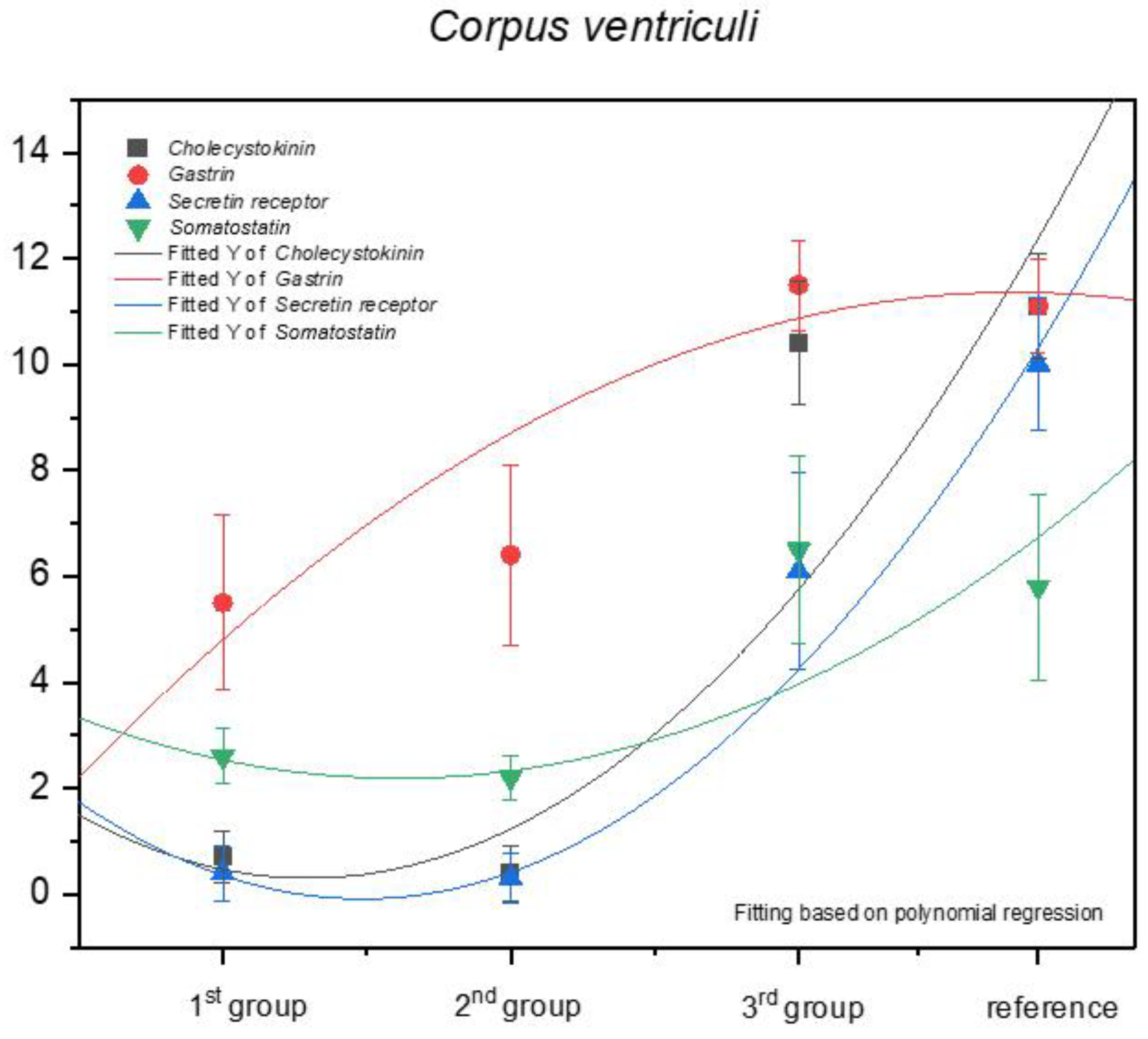
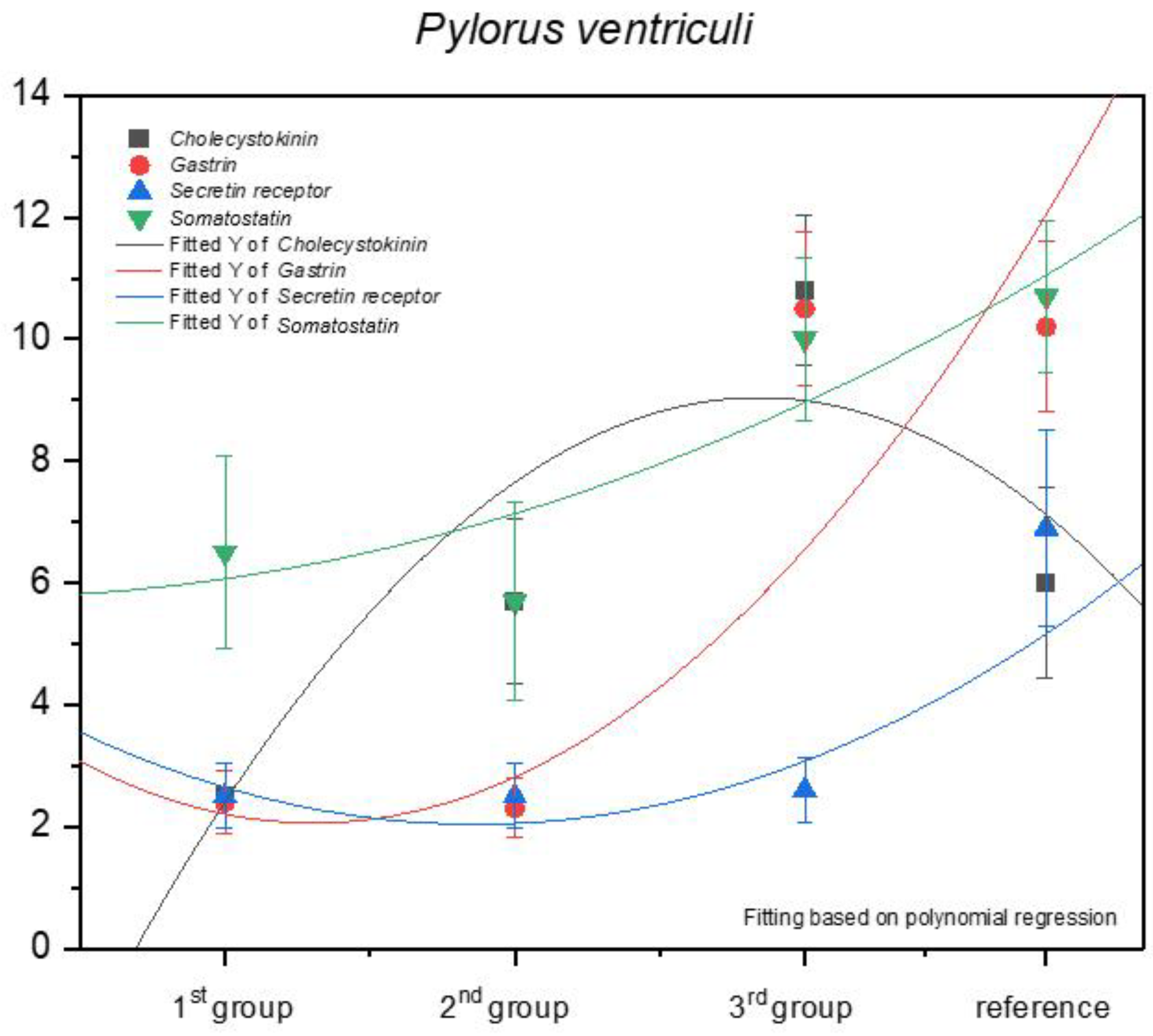
3.1. Immunohistochemistry of Anti-Gastrin Antibody
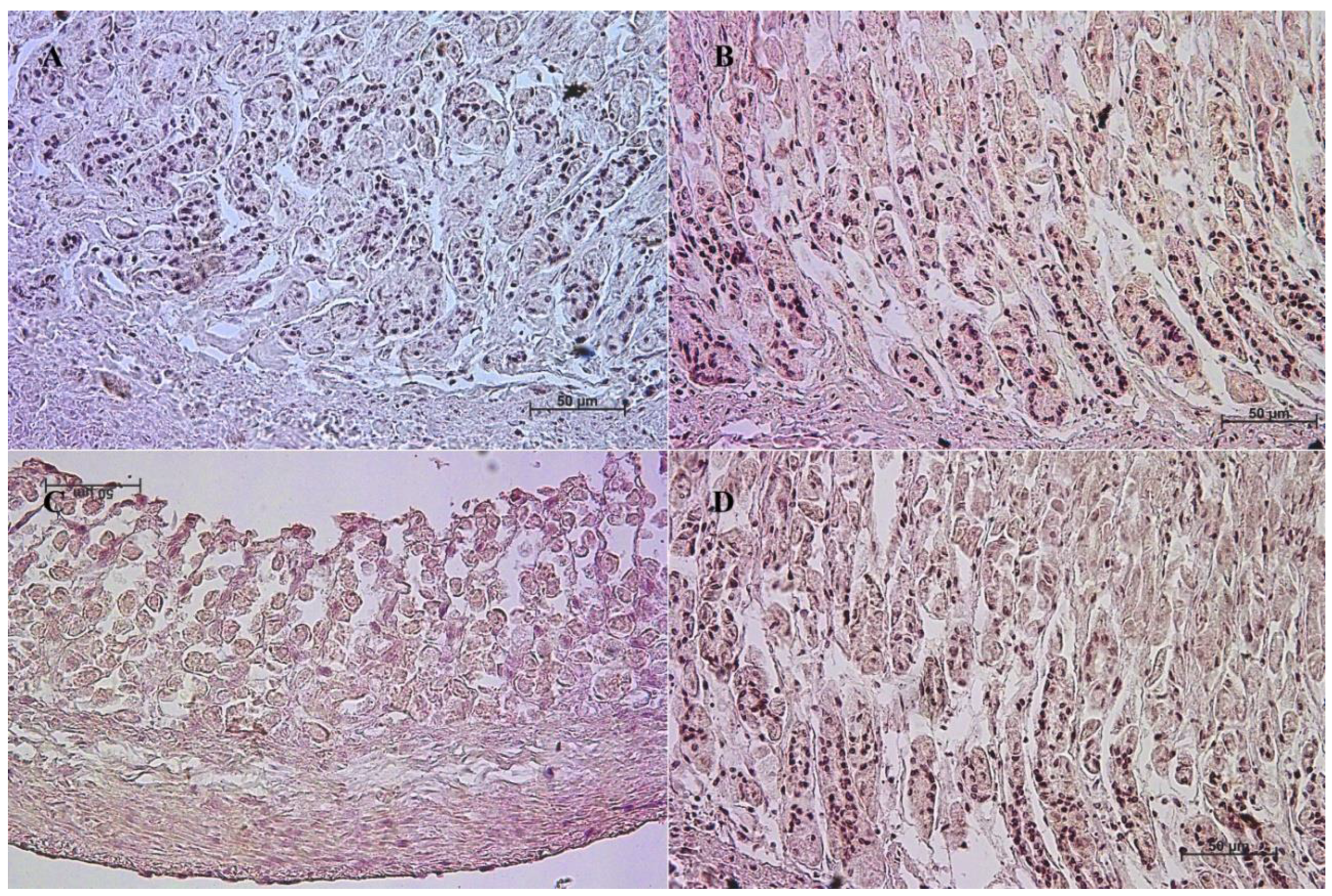
3.2. Immunohistochemistry of Anti-Cholecystokinin Antibody
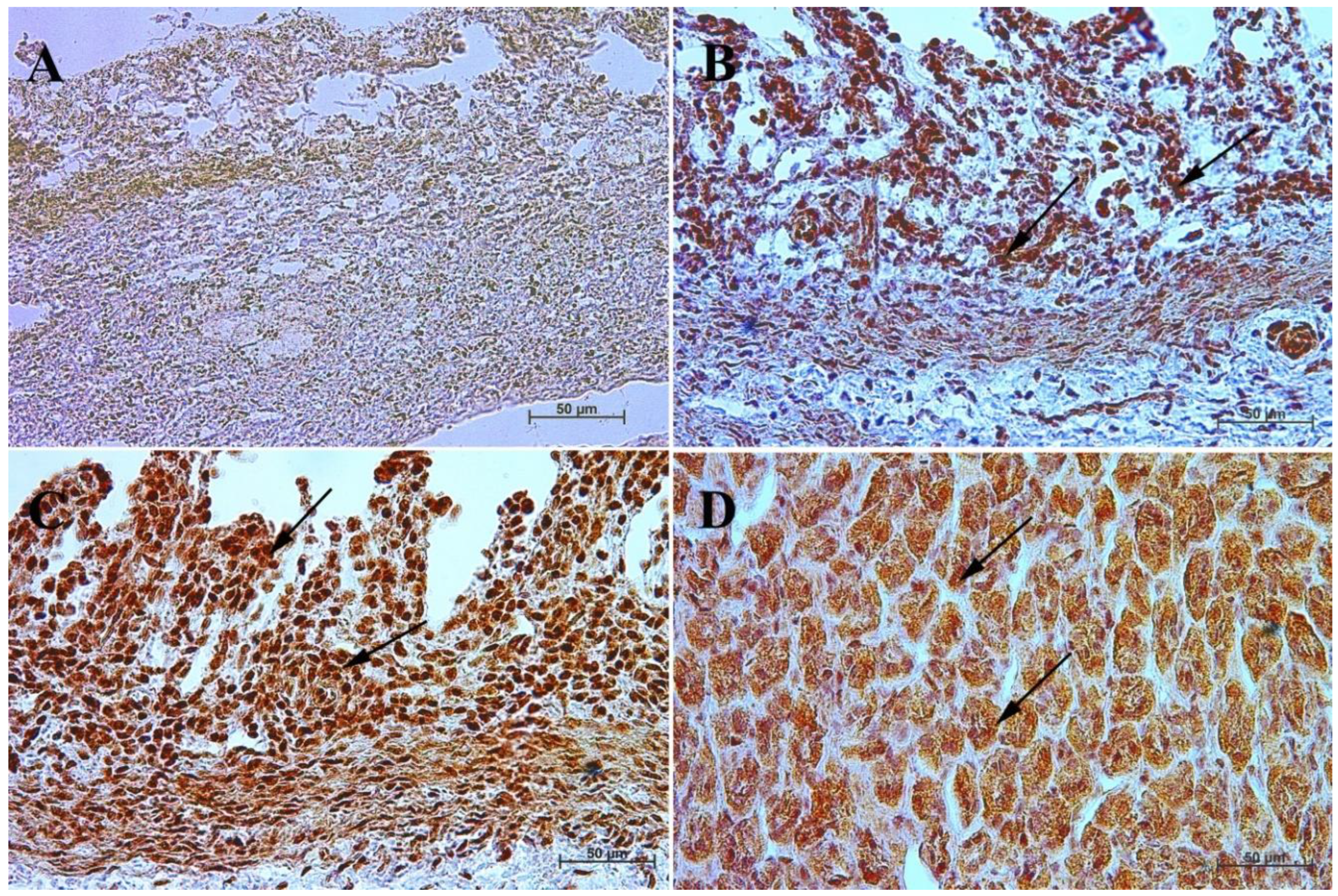
3.3. Immunohistochemistry of Anti-Secretin Receptor Antibody
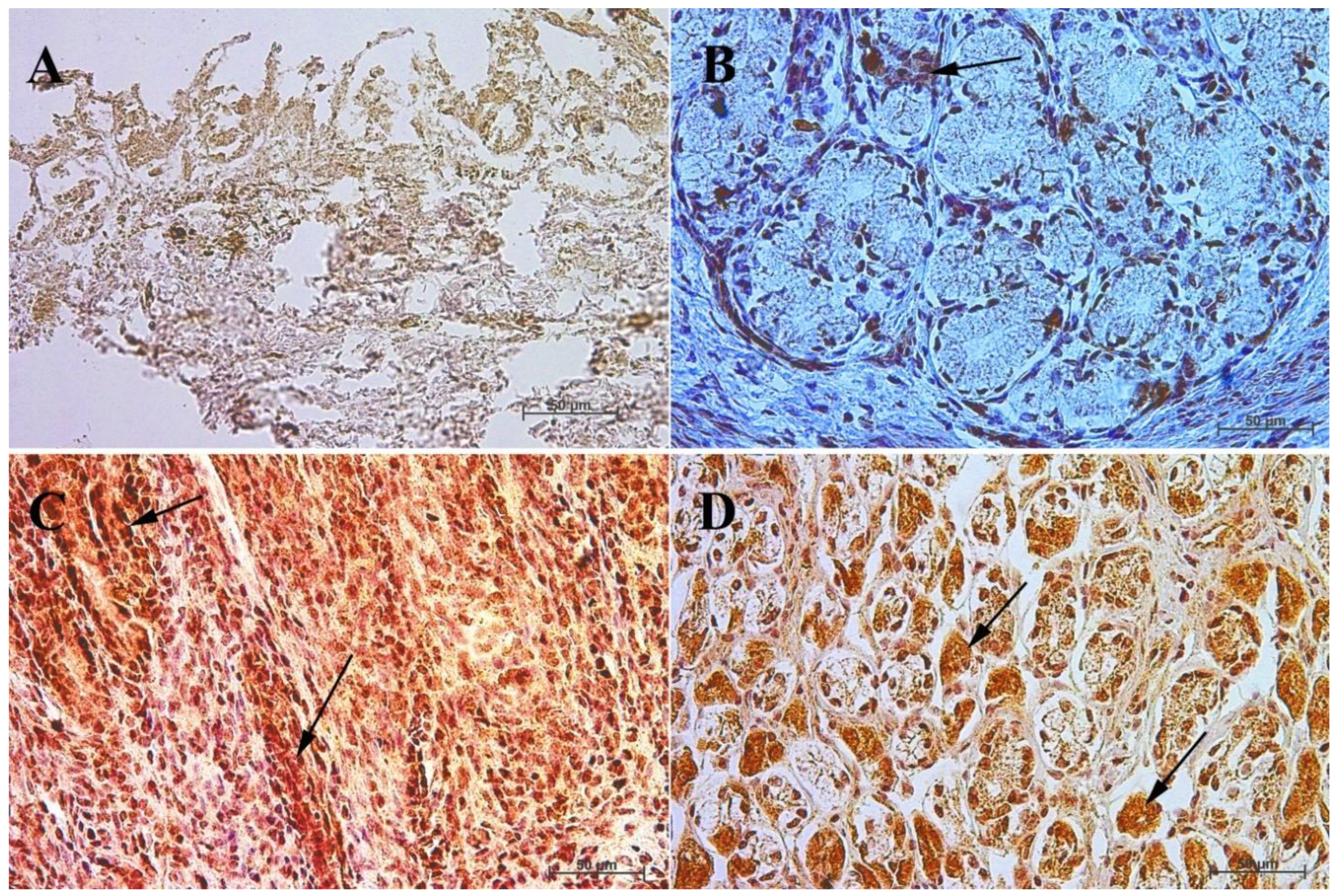
3.4. Immunohistochemistry of Anti-Somatostatin Antibody
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bolk, L.; Göppert, E.; Kallius, E.; Lubosch, W. Handbuch der Vergleichenden Anatomie der Wirbeltiere, 1st ed.; Urban & Schwarzenberg Verlag: Berlin, Germany; Vienna, Austria, 1937. [Google Scholar]
- Nickel, R.; Schummer, A.; Seiferle, E. Lehrbuch der Anatomie der Haustiere, 1st ed.; Parey Verlag: Stuttgart, Germany, 2004. [Google Scholar]
- Schnorr, B.; Kressin, M. Embriologie der Haustiere, 6th ed.; Enke Verlag: Stuttgart, Germany, 2011. [Google Scholar]
- Rüsse, I.; Sinowatz, F. Lehrbuch der Embriologie der Haustiere; Verlag Paul Parey: Berlin, Germany; Hamburg, Germany, 1991. [Google Scholar]
- Bielańska-Osuchowska, Z. Zarys Organogenezy, 1st ed.; PWN: Warszawa, Poland, 2004. [Google Scholar]
- Knospe, C. Die Entwickling der Magendrüssen der Katze (felis silvestris catus). Anat. Histol. Embryol. 1996, 25, 75–94. [Google Scholar] [CrossRef] [PubMed]
- Werner, E.D. The organogenesis and early histogenesis of the bovine stomach. Am. J. Anat. 1958, 102, 33–53. [Google Scholar] [CrossRef]
- McGeady, T.A.; Sack, W.O. The development of vagal nerve innervation of the bovine stomach. Am. J. Anat. 1967, 121, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, R.; Gerov, K. The morphological and functional differentiation of the alimentary canal of pig during ontogeny. I—Development and differentiation of the fundic portion of stomach. Anat. Anz. 1975, 137, 12–15. [Google Scholar]
- Georgieva, R.; Gerov, K. The morphological and functional differentiation of the alimentary canal of the pig during ontogeny. II development and differentiation of the jejunum. Anat. Anz. 1975, 137, 16–20. [Google Scholar] [PubMed]
- Pospieszny, N. Distribution of the vegal nerve of the stomach and certain lymph nodes of the sheep in the prenatal period. Anat. Anz. 1979, 146, 47–59. [Google Scholar]
- Cai, W.-Q.; Gabella, G. Structure and innervation of the musculature at the gastroduodenal junction of the guinea pig. J. Anat. 1984, 139, 93–104. [Google Scholar]
- Ginneken van, C.; Weyns, A.; Meir van, F.; Ooms, L.; Verhofstad, A. Intrinsic innervation of the stomach of the foetal pig: An immunohistochemical study of VIP-immunoreactive nerve fibres and cell bodies. Anat. Histol. Embryol. 1996, 25, 269–275. [Google Scholar] [CrossRef]
- Steele, M.A.; Penner, G.B.; Chaucheyras-Durand, F.; Guan, L.L. Development and physiology of the rumen and the lower gut: Targets for improving gut health. J. Dairy Sci. 2016, 99, 4955–4966. [Google Scholar] [CrossRef]
- Franciolli, A.L.R.; Cordeiro, B.M.; Fonseca, E.T.; Rodrigues, M.N.; Sarmento, C.A.P.; Ambrosio, C.E.; Carvalho, A.F.; Miglino, M.A.; Silva, L.A. Characteristics of equine embryo and fetus from days 15 to 107 of pregnancy. Theriogenology 2011, 76, 819–832. [Google Scholar] [CrossRef]
- Rodrigues, M.N.; Carvalho, R.C.; Franciolli, A.L.R.; Rodrigues, R.F.; Rigoglio, N.N.; Jacob, J.C.F.; Gastal, E.L.; Miglino, M.A. Prenatal development of the digestive system in horse. Anat. Rec. 2014, 297, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Poradowski, D.; Chrószcz, A. Equine Stomach Development in the Fetal Period: An Anatomical, Topographical, and Morphometric Study. Animals 2022, 12, 2966. [Google Scholar] [CrossRef] [PubMed]
- Poradowski, D.; Chrószcz, A. Equine Stomach Development in the Foetal Period of Prenatal Life—A Histological and Histometric Study. Animals 2022, 12, 3047. [Google Scholar] [CrossRef] [PubMed]
- Karam, S.M.; Leblond, C.P. Dynamics of epithelial cells in the corpus of the mouse. I–Identification of proliferative cell types and pin pointing of the stem cells. Anat. Rec. 1993, 236, 259–279. [Google Scholar] [CrossRef]
- Karam, S.M.; Leblond, C.P. Dynamics of epithelial cells in the corpus of the mouse. II–Outward migration of pit cells. Anat. Rec. 1993, 236, 280–296. [Google Scholar] [CrossRef]
- Karam, S.M.; Leblond, C.P. Dynamics of epithelial cells in the corpus of the mouse. III–Inward migration of neck cells followed by progressive transformation into zymogenic cells. Anat. Rec. 1993, 236, 297–313. [Google Scholar] [CrossRef]
- Karam, S.M.; Leblond, C.P. Dynamics of epithelial cells in the corpus of the mouse. IV–Bidirectional migration of parietal cells ending in their degeneration and loss. Anat. Rec. 1993, 236, 314–322. [Google Scholar] [CrossRef]
- Karam, S.M.; Leblond, C.P. Dynamics of epithelial cells in the corpus of the mouse. V–Behaviour of entero-endocrine and caveolated cells: General conclusions on cell kinetics in the oxyntic epithelium. Anat. Rec. 1993, 236, 333–340. [Google Scholar] [CrossRef]
- Karam, S.M.; Leblond, C.P. Origin and migratory pathways of the eleven epithelial cell types present in the body of mouse stomach. Microsc. Res. Tech. 1995, 31, 193–214. [Google Scholar] [CrossRef]
- Kablar, B. Structural study on the appearance of innervation in the stomach of mouse and rat embryos. Tissue Cell 1995, 27, 309–315. [Google Scholar] [CrossRef]
- Asar, M.; Bayram, Z.; Korgun, E.T.; Tertemiz, F.; Akkoyunlu, G.; Demir, R. Immunocytochemical Detection of Synaptophysin in Enteric Neurones during Prenatal Development in the Rat Stomach. Anat. Histol. Embryol. 2004, 33, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Çetin, A.; Eşrefoğlu, M. Prenatal and Postnatal Development of the Stomach in Wistar Albino Rats. J. Turgut Ozal Med. Cent. 2014, 21, 4–11. [Google Scholar]
- Chrószcz, A. The innervation and arterial blood supply of pig’s stomach in the fetal period. EJPAU 2008, 11, 3. [Google Scholar]
- Chrószcz, A. The morphometric and topographic study of pig’s stomach development between the 35th and 114th day of gestation. EJPAU 2008, 11, 4. [Google Scholar]
- Chrószcz, A. Morphology, development and histometry of swine gastric wall in the fetal period. EJPAU 2008, 11, 4. [Google Scholar]
- Pearse, A.G.E.; Takor, T. Neuroendocrine embryology and the APUD concept. Clin. Endocrinol. 1976, 5, 229–244. [Google Scholar] [CrossRef]
- Stave, R.; Myren, J.; Brandtzaeg, P.; Gjone, E. Quantitative studies of gastrin cells (G cells) and parietal cells in relation to the gastric acid secretion in patients with peptic ulcer disease. Scand. J. Gastroenterol. 1978, 13, 293–298. [Google Scholar] [CrossRef]
- Capella, C.; Solcia, E. The endocrine cells of the pig gastrointestinal mucosa and pancreas. Arch. Histol. Jap. 1972, 35, 1–29. [Google Scholar] [CrossRef]
- Grube, D.; Forssmann, W.G. Morphology and function of the entero-endocrine cells. Horm. Metab. Res. 1979, 11, 589–606. [Google Scholar] [CrossRef]
- Oomori, Y. Eight types of endocrine cells in the abomasus of scheep. Z. Mikrosk.-Anat. Forsch. 1983, 97, 369–385. [Google Scholar]
- Sangild, P.T.; Cranwell, P.D.; Hilsted, L. Ontogeny of gastric function in the pig: Acid secretion and the synthesis and secretion of gastrin. Nord. Vet. Med. 1992, 62, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Zabel, M.; Surdyk-Zasada, J.; Lesisz, I.; Jagoda, E.; Wysocka, T.; Seidel, J.; Zabel-Olejnik, J.; Grzeszkowiak, J. Immunocytochemical studies on endocrine cells of alimentary tract of the pig in the embryonic and foetal period of life. Folia Morphol. 1995, 54, 69–80. [Google Scholar]
- Pan, Q.S.; Fang, Z.P.; Huang, F.J. Identification, localization and morphology od APUD cells in gastroenteropancreatic system of stomach—Containing teleosts. World J. Gastroenterol. 2000, 6, 842–847. [Google Scholar] [CrossRef] [PubMed]
- Ameri, P.; Ferone, D. Diffuse endocrine system, neuroendocrine tumors and immunity: What’s new? Neuroendocrinology 2012, 95, 267–276. [Google Scholar] [CrossRef]
- Vaish, R.; Pandey, Y.; Gupta, N. APUD system: An anatomical perspective. Indo-Am. J. Agric. Vet. Sci. 2014, 2, 13–26. [Google Scholar]
- Mandal, A.K.; Tudu, N.K. Anatomical and clinical perspective of APUD cell series: An overview. Int. J. Pure App. Biosci. 2018, 6, 1310–1317. [Google Scholar] [CrossRef]
- Rodriguez, A.; Pena, L.; Flores, J.M.; Gonzalez, M.; Castano, M. Immunocytochemical study of the diffuse neuroendocrine system cells in equine lungs. Anat. Histol. Embryol. 1992, 21, 136–145. [Google Scholar] [CrossRef]
- Mahapatra, A.; Beaulah, J.V.; Gnanadevi, R.; Basha, S.H.; Ramesh, G. Histological study on APUD cells during prenatal and postnatal period in sheep. Indian J. Vet. Anat. 2016, 28, 24–27. [Google Scholar]
- Samuelson, D.A. Textbook of Veterinary Histology, 1st ed.; Saunders Elsevier: Sant Louis, Mo, USA, 2006. [Google Scholar]
- Fawcett, D.W. A Text Book of Histology, 12th ed.; Chapman and Hall: New York, NY, USA, 1994. [Google Scholar]
- Dellmann, H.D.; Eurell, J.A. Textbook of Veterinary Histology, 5th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 1998. [Google Scholar]
- Bożiłow, W.; Sawicki, K. Metody Badań Zmienności cech Anatomicznych Człowieka Podczas Rozwoju Prenatalnego i Okołoporodowego, 1st ed.; Akademia Medyczna: Wrocław, Poland, 1980. [Google Scholar]
- Remmele, W.; Stegner, H.E. Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 1987, 8, 138–140. [Google Scholar]
- Fedchenko, N.; Reifenrath, J. Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue—A review. Diagn. Pathol. 2014, 9, 221. [Google Scholar] [CrossRef]
- Moran-Ramos, S.; Tovar, A.R.; Torres, N. Diet: Friend or foe of enteroendocrine cells--how it interacts with enteroendocrine cells. Adv. Nutr. 2012, 3, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Grunddal, K.V.; Ratner, C.F.; Svendsen, B.; Sommer, F.; Engelstoft, M.S.; Madsen, A.N.; Pedersen, J.; Nøhr, M.K.; Egerod, K.L.; Nawrocki, A.R.; et al. Neurotensin is coexpressed, coreleased, and acts together with GLP-1 and PYY in enteroendocrine control of metabolism. Endocrinology 2016, 157, 176–194. [Google Scholar] [CrossRef] [PubMed]
- Sykaras, A.G.; Demenis, C.; Cheng, L.; Pisitkun, T.; Mclaughlin, J.T.; Fenton, R.A.; Smith, C.P. Duodenal CCK cells from male mice express multiple hormones including ghrelin. Endocrinology 2014, 155, 3339–3351. [Google Scholar] [CrossRef]
- Ye, L.; Liddle, R.A. Gastrointestinal hormones and the gut connectome. Curr. Opin. Endocrinol. Diabetes Obes. 2017, 24, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Dockray, G.J. Enteroendocrine cell signalling via the vagus nerve. Curr. Opin. Pharmacol. 2013, 13, 954–958. [Google Scholar] [CrossRef] [PubMed]
- Avetisyan, M.; Schill, E.M.; Heuckeroth, R.O. Building a second brain in the bowel. J. Clin. Investig. 2015, 125, 899–907. [Google Scholar] [CrossRef]
- Foong, J.P.; Hirst, C.S.; Hao, M.M.; McKeown, S.J.; Boesmans, W.; Young, H.M.; Bornstein, J.C.; Vanden, P. Changes in nicotinic neurotransmission during enteric nervous system development. J. Neurosci. 2015, 35, 7106–7115. [Google Scholar] [CrossRef]
- Hao, M.M.; Bornstein, J.C.; Vanden Berghe, P.; Lomax, A.E.; Young, H.M.; Foong, J.P.P. The emergence of neural activity and its role in the development of the enteric nervous system. Dev. Biol. 2013, 382, 365–374. [Google Scholar] [CrossRef]
- Uesaka, T.; Nagashimada, M.; Enomoto, H. Neuronal differentiation in schwann cell lineage underlies postnatal neurogenesis in the enteric nervous system. J. Neurosci. 2015, 35, 9879–9888. [Google Scholar] [CrossRef]
- Foltmann, B.; Cranwell, P.D.; Newport, M.J.; Howarth, G.L. Ontogeny of pig gastric proteases: Observations on fetal, stillborn, unsuckled newborn, suckled and growing pigs. Proc. Nutr. Soc. 1987, 46, 26A. [Google Scholar]
- Walsh, J.H. Peptides as regulators of gastric acid secretion. Annu. Rev. Physiol. 1988, 50, 41–63. [Google Scholar] [CrossRef] [PubMed]
- Andrews, F.M.; Buchanan, B.R.; Elliot, S.B.; Clariday, N.A.; Edwards, L.H. Gastric ulcers in horses. J. Anim. Sci. 2005, 83, E18–E21. [Google Scholar]
- Niezgoda, M.; Kasacka, I. Gastrointestinal neuroendocrine cells in various types of hypertension—A review. Prog. Health Sci. 2017, 7, 117–125. [Google Scholar] [CrossRef]
- Smith, M.E.; Morton, D.G. The Digestive Tract, 2nd ed.; Churchill Livingstone Elsevier: Edinburgh, UK; London, UK, 2010. [Google Scholar]
- Masot, A.J.; Franco, A.J.; Redondo, E. Morphometric and immunohistochemical study of the abomasum of red deer during prenatal development. J. Anat. 2007, 211, 376–386. [Google Scholar] [CrossRef] [PubMed]
- Yee, L.F.; Wong, H.C.; Calaustro, E.Q.; Mulvhill, S.J. Roles of gastrin and somatostatin in the regulation of gastric acid secretion in the fetal rabbit. J. Surg. Res. 1996, 63, 364–368. [Google Scholar] [CrossRef]
- Niewiesk, S. Maternal antibodies: Clinical significance, mechanism of interference with immune responses and possible vaccination strategies. Front. Immunol. 2014, 5, 446. [Google Scholar] [CrossRef]
| Name | Type | Dilution | Catalogue Number | Homology |
|---|---|---|---|---|
| Gastrin | Polyclonal | 1:500 | clone ab16035, Abcam, Cambridge, UK | >75% |
| Cholecystokinin | Polyclonal | 1:300 | clone PA5-103116, Invitrogen, Waltham, MA, USA | unknown |
| Secretin receptors | Polyclonal | 1:200 | clone bs-0089R, Biossusa, Woburn, MA, USA | unknown |
| Somatostatin | Polyclonal | 1:500 | clone ab108456, Abcam, Cambridge, UK | >75% |
| I—% of Positive Cells | II—Intensity of Staining | IRS Score |
|---|---|---|
| 0 = lack | 0 = lack of colour reaction | 0–1 = negative |
| 1 = <10% | 1 = mild | 2–3 = mild |
| 2 = 10–50% | 2 = moderate | 4–8 = moderate |
| 3 = 51–80% | 3 = intense | 9–12 = strongly positive |
| 4 = >80% |
| Age [Months] | The first age group (4–5 Month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 7 | 7 | 6 | 4 | 4 | 4 | 8 | 7 | 4 | 4 | 5.5 ± 1.65 | moderate |
| Pylorus ventriculi | 3 | 3 | 2 | 3 | 2 | 2 | 2 | 2 | 3 | 2 | 2.4 ± 0.52 | mild |
| Age [months] | The second age group (7–8 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 8 | 4 | 4 | 8 | 4 | 7 | 7 | 7 | 7 | 8 | 6.4 ± 1.71 | moderate |
| Pylorus ventriculi | 2 | 2 | 2 | 2 | 3 | 2 | 2 | 2 | 3 | 3 | 2.3 ± 0.48 | mild |
| Age [months] | The third age group (10–11 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 12 | 12 | 12 | 12 | 12 | 10 | 11 | 12 | 10 | 12 | 11.5 ± 0.85 | strongly positive |
| Pylorus ventriculi | 11 | 11 | 10 | 9 | 9 | 9 | 10 | 12 | 12 | 12 | 10.5 ± 1.27 | strongly positive |
| Age [months] | Adult reference group | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 10 | 12 | 12 | 11 | 11 | 11 | 12 | 10 | 10 | 12 | 11.1 ± 0.88 | strongly positive |
| Pylorus ventriculi | 9 | 10 | 9 | 9 | 11 | 12 | 12 | 12 | 9 | 9 | 10.2 ± 1.4 | strongly positive |
| Age [Months] | The first age group (4–5 Month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 1 | 1 | 1 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0.7 ± 0.48 | negative |
| Pylorus ventriculi | 2 | 3 | 3 | 3 | 2 | 2 | 3 | 2 | 3 | 2 | 2.5 ± 0.53 | mild |
| Age [months] | The second age group (7–8 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0.4 ± 0.52 | negative |
| Pylorus ventriculi | 5 | 5 | 8 | 4 | 6 | 6 | 7 | 5 | 4 | 7 | 5.7 ± 1.34 | moderate |
| Age [months] | The third age group (10–11 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 9 | 11 | 9 | 12 | 12 | 11 | 11 | 10 | 9 | 10 | 10.4 ± 1.17 | strongly positive |
| Pylorus ventriculi | 11 | 11 | 10 | 9 | 9 | 12 | 12 | 12 | 12 | 10 | 10.8 ± 1.23 | strongly positive |
| Age [months] | Adult reference group | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 12 | 12 | 10 | 12 | 11 | 11 | 9 | 12 | 11 | 11 | 11.1 ± 0.99 | strongly positive |
| Pylorus ventriculi | 6 | 6 | 8 | 7 | 7 | 8 | 6 | 4 | 4 | 4 | 6 ± 1.56 | moderate |
| Age [Months] | The first age group (4–5 Month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 0 | 1 | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 1 | 0.4 ± 0.52 | negative |
| Pylorus ventriculi | 2 | 3 | 2 | 3 | 2 | 2 | 3 | 2 | 3 | 3 | 2.5 ± 0.53 | mild |
| Age [months] | The second age group (7–8 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 | 1 | 0.3 ± 0.48 | negative |
| Pylorus ventriculi | 3 | 2 | 3 | 3 | 3 | 2 | 2 | 2 | 2 | 3 | 2.5 ± 0.53 | mild |
| Age [months] | The third age group (10–11 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 8 | 4 | 5 | 7 | 8 | 8 | 4 | 4 | 5 | 8 | 6.1 ± 1.85 | moderate |
| Pylorus ventriculi | 2 | 3 | 2 | 3 | 3 | 3 | 2 | 3 | 3 | 2 | 2.6 ± 0.52 | mild |
| Age [months] | Adult reference group | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 11 | 9 | 9 | 9 | 9 | 10 | 10 | 9 | 12 | 12 | 10 ± 1.25 | strongly positive |
| Pylorus ventriculi | 8 | 8 | 8 | 8 | 8 | 4 | 5 | 5 | 8 | 7 | 6.9 ± 1.6 | moderate |
| Age [Months] | The first age group (4–5 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 2 | 3 | 3 | 2 | 2 | 2 | 3 | 3 | 3 | 3 | 2.6 ± 0.52 | mild |
| Pylorus ventriculi | 8 | 7 | 8 | 5 | 5 | 5 | 8 | 7 | 8 | 4 | 6.5 ± 1.58 | moderate |
| Age [months] | The second age group (7–8 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 2 | 2 | 3 | 2 | 2 | 2 | 2 | 2 | 2 | 3 | 2.2 ± 0.42 | mild |
| Pylorus ventriculi | 4 | 7 | 4 | 4 | 6 | 6 | 4 | 6 | 8 | 8 | 5.7 ± 1.64 | moderate |
| Age [months] | The third age group (10–11 month of gestation) | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 8 | 4 | 5 | 4 | 5 | 8 | 8 | 8 | 7 | 8 | 6.5 ± 1.78 | moderate |
| Pylorus ventriculi | 12 | 9 | 9 | 8 | 12 | 10 | 10 | 10 | 11 | 9 | 10 ± 1.33 | strongly positive |
| Age [months] | Adult reference group | |||||||||||
| IRS mean/SD | Description | |||||||||||
| Corpus ventriculi | 5 | 5 | 5 | 4 | 8 | 8 | 7 | 8 | 4 | 4 | 5.8 ± 1.75 | moderate |
| Pylorus ventriculi | 12 | 12 | 11 | 12 | 10 | 10 | 12 | 10 | 9 | 9 | 10.7 ± 1.25 | strongly positive |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poradowski, D.; Chrószcz, A. Equine Stomach Development in the Foetal Period of Prenatal Life—An Immunohistochemical Study. Animals 2023, 13, 161. https://doi.org/10.3390/ani13010161
Poradowski D, Chrószcz A. Equine Stomach Development in the Foetal Period of Prenatal Life—An Immunohistochemical Study. Animals. 2023; 13(1):161. https://doi.org/10.3390/ani13010161
Chicago/Turabian StylePoradowski, Dominik, and Aleksander Chrószcz. 2023. "Equine Stomach Development in the Foetal Period of Prenatal Life—An Immunohistochemical Study" Animals 13, no. 1: 161. https://doi.org/10.3390/ani13010161
APA StylePoradowski, D., & Chrószcz, A. (2023). Equine Stomach Development in the Foetal Period of Prenatal Life—An Immunohistochemical Study. Animals, 13(1), 161. https://doi.org/10.3390/ani13010161





