1. Introduction
Environmentally induced hyperthermia is a global-scale threat to the dairy industry in many ways, with its effects including economic loss, animal health issues, and productivity. The adverse effects of heat stress (HS) on the productivity of dairy animals, in terms of milk yield, composition, and quality, have been well-documented [
1]. Despite advances in cooling systems and environmental management [
2], HS continues to negatively affect the diversity of dairy production characteristics. As such, lactating dairy cows under HS experience limited energy intake and, thus, are unable to meet the demands of their bodies for maintaining milk production and health, resulting in reduced milk yield and quality and leaving the animals susceptible to diseases [
1,
3,
4]. The harm of HS is particularly more important in high-yield dairy cows, due to their higher susceptibility to stressors which periodically happen during early stage of lactation [
5,
6,
7].
The temperature humidity index (THI) has exclusively been employed as an index of HS status in cattle [
3]. However, short-term and long-term exposure to HS may reflect variations in responses of the animals. Furthermore, THI is based only on the air temperature and humidity [
3] and, as such, is not a direct biomarker of metabolic alterations in response to HS. Thus, alterations in physiological parameters, such as rectal temperature, heart rate, and respiratory dynamics, reflect the degree of HS in cattle [
8], as well as feed intake [
1,
4], as the first indicator of HS and, consequently, its influence on milk yield and characteristics [
1]. Feed intake can establish certain blood metabolite alteration changes during HS. Besides those, metabolic profiling (known as metabolomics) has increasingly been used in clinical pharmacology and is an ideal tool for the acquisition of the several thousand metabolite alterations that are applied in the relationship between endogenous metabolite metabolism and body metabolism [
9]. Metabolomics has been applied in cow investigations, in order to predict the risk of diseases [
10], and has been used for biomarker and pathway discovery in some metabolic diseases in cows [
10]. Given this review, metabolomics analyses provide a powerful platform for the identification of animals and humans associated with pathophysiological alterations resulting from exposures to specific environmental factors [
10]. However, the metabolic changes related to short-term HS during the early lactation stage in dairy cattle still remain unclear. The activation process of the hypothalamus–pituitary–adrenal axis enhances the production and circulation of cortisol in the animal body. It is the primary indicator that ruminants can be identified when they are stressed [
11]. Additionally, haptoglobin is one of the acute phase protein indicators commonly identified to animal health and inflammatory responses [
11]. Hence, measuring these two correlated hormones in biological samples will allow better understating the mechanism behind the effect of short-term HS in dairy cows. HS response is a complex molecular process that involves the transcriptional and post-transcriptional regulation of stress-related genes. Acute environmental change initiates the heat stress response at the cellular level. Furthermore, microRNAs (miRNAs) was recognized as an important regulator of gene expression beyond the transcriptional stage and various biological reactions such as development, apoptosis, differentiation, and viral infection [
12]. Additionally, miRNAs regulate numerous functions of bovine mammary epithelial cells (bMECs), which play a putative role in milk secretion [
13]. In humans, it has been documented that miR-216 can regulate cell apoptosis through repressing target genes in several cancer cells [
13]. Likewise in this study, we aimed at exploring the potential involvement of miR-216 in the milk of heat stressed cows. Furthermore, to better deal with the consequences of HS, we aimed to characterize the short-term effects of HS in terms of not only the alterations of productive performance characteristics but also the narrative response of metabolomics and gene expression.
Therefore, the objective of this study was to characterize short-term HS (4 day) in Holstein dairy cows using altered indicators of metabolomics, milk miRNA-216 and characteristics, and blood metabolites.
On the other hand, our traditional knowledge regarding HS effects is limited to long-term HS, in which the animals were housed outdoors or underwent uncontrolled environmental housing conditions, where the temperature and humidity were subject to many fluctuations over time, such as from being severe around noon to cooling in the evening. These phenomena may compromise our understanding of HS effects, due to disparities in environmental and housing situations, thus misleading the characterization of a framework of HS influence. In this regard, the outcome of this study provides insights towards better understanding how dairy cows exposed to chamber-controlled short-term HS can be characterized, not only through production performance parameters but also using newly presented indicators, such as metabolomics and miRNA analyses.
4. Discussion
A decrease in FI is a very usual phenomenon in response to heat stress (HS) [
1,
4]. Under HS conditions, the amount of energy expended by the cow to maintain homeothermy increases (e.g., 20% more at 35 °C, compared to 20 °C). Panting also increases the maintenance requirement by 7–25% under HS. Therefore, the FI must increase to cover this additional energy cost [
16]. However, during HS, FI decreases; this means that the energy status of the cow gets a double hit—greater energy costs to try to maintain homeothermy and lower energy intake [
17]. In this regard, it is not surprising that milk production decreases. Indirectly, however, HS-decreased FI provokes consequences in connection with various physiological, metabolic, and blood parameters, in an attempt to lessen the effects of HS by activating homeostatic mechanisms across the body of the animal [
5,
8,
18]. In this study, the mechanism behind the significant decrease in FI can indicate the direct effect of HTHH conditions on animals and the partial inability of cows to dissipate the excess of heat from their body, and thus, less intake will help them to reduce heat production (i.e., metabolic heat production and physically generated heat, known as heat increment) in their body. These results are in alignment with numerous previous studies that have claimed similar phenomena in response to HS [
18].
Water intake has a high correlation with FI in animals and humans [
1,
5]. A normal phenomenon occurring in attempt to deal with HS is that cows tend to intake much more water than under normal conditions, in order to accommodate their body to dissipate heat by evaporation and by alterations in blood circulation. Additionally, under HS conditions, the cow loses water through its skin and respiration, in order to minimize the rise in body temperature [
19]. Although HS may cause higher water consumption, on the other hand, decreased FI could also alleviate this water intake. In this study, HTHH did not seem to indicate the influence of FI in increasing water intake; however, water intake was partially induced by HTHH, showing a numerical increase. There is also the possibility that a larger sample size and longer period of HTHH exposure may have allowed us to observe a significant difference in water intake, which should be further studied.
Several studies have shown that physiological indicators such as heart rate (HR) and rectal temperature (RT) are the foremost induced by short-term HS exposure in beef calves [
8] and dairy cattle [
20]. Kim et al. [
8] have pinpointed that HR and RT are closely associated indicators, in response to HS, and are the most sensitive markers to be elevated. Therefore, HR and RT are likely to change in animals under HS, which is in agreement with the present results.
The body processes stressful information and elicits a response, depending on the degree of stress [
21]. The body’s autonomic nervous system is broken down into the sympathetic nervous system (SNS) and the parasympathetic nervous system (PNS). In times of stress, the SNS is activated. The SNS is responsible for a cascade of hormonal and physiological responses [
21]. The hypothalamus subsequently activates the SNS, and the adrenal glands release a surge of catecholamines, such as epinephrine [
21]. This results in effects such as increased heart rate and respiratory rate. As the body continues to perceive stress, the hypothalamus activates the HPA axis. Cortisol is released from the adrenal cortex, which allows the body to continue to stay on high alert. Acutely, catabolic mechanisms of cortisol provide energy to the body [
13]. The higher cortisol levels observed in HTHH group were in line with the higher heart rate in the corresponding group in this study. Kim et al. [
8] provided a correlation study regarding the relationship between cortisol levels and blood and physiological parameters, including HR and RT. They mentioned that HR is correlated with the concentration of blood cortisol; as such, it has been utilized as an index for the regulation of animal body homeostasis [
22]. Furthermore, it has been reported that the rise in blood pressure is associated with increased HR [
23]. RT is also an important indicator for the homeostasis regulation of body temperature. The positive correlation between cortisol levels and RT may provide evidence that there exists a metabolic relationship between cortisol levels and RT. A previous study has suggested that the concentration of serum cortisol is a sensitive indicator of HS, and there is a significant correlation between cortisol and RT [
24]. Therefore, the significant correlations between HR, RT, and the concentration of serum cortisol can be used to determine HS severity through physiological parameters in high-yielding dairy cows. Given the above review, the reasons behind increased HR and RT in the HTHH group, compared with the LTLH group, can be elucidated.
Blood hematology encompasses several immune factors, including WBC, LYM, MON, and GRA, which are expected to be suppressed as a result of stress and an increase in cortisol, due to adverse correlations [
25]. HS can cause immunosuppression in ruminants by inhibiting rumination [
26], thereby leading to more chances of disease occurrence in the animals [
25]. However, in the current study, most of the hematological parameters (except for RBC, which showed a decreased tendency) were not affected by short-term HS exposure. The unaffected parameters could be attributed to either the short time of HS exposure (only 4 day) [
27,
28] or low sample size in this study, due to natural fluctuations in blood parameters. In line with the present RBC results, a previous study has shown increases in the fraction of RBCs, including erythrocyte number, hematocrit value, and hemoglobin content, in HS group cows [
29]. The tendency for higher RBC has been pinpointed, by Kumar and Pachauri [
30], to be due to RBC release from the spleen and/or changes in erythrocyte stimulating factor (ESF) release, which is governed by the relationship between the oxygen demand of tissues and the amount of oxygen carried by the blood [
30], in an attempt to dissipate heat from the body. In this study, the tendency of increase in RBC was also confirmed through higher serum haptoglobin, which we discuss, in detail, later (with respect to the hormonal effect on the HTHH group). On the contrary, Morar and Hutu [
31] have reported that RBC, Hct, and Hb were decreased significantly in dairy cows under HS. Casella et al. [
32] revealed the reduction in RBC, Hct, and Hb to be associated with a hemodilution effect due to increased water consumption in an attempt to facilitate evaporative cooling under HS [
30]. However, we did not observe a significant increase in water intake in the HTHH group.
Blood metabolites may be directly (e.g., by FI [
18]) or indirectly influenced by HS, in an attempt to reduce the deleterious effect of HS by activating body hemostasis mechanisms [
8,
18]. In this study, the serum metabolite profile, including GLU, NEFA, BUN, TP, ALB, r-GLU, Ca, P, MG, CHO, and GOT, showed no significant difference between two groups, LTLH and HTHH. As HS was accompanied with decreased FI, we expected to observe changes in some of the aforementioned profiles. However, contrary to our expectation, there were no changes in any of the metabolites. We could speculate some hypotheses behind this unchanged profile. One hypothesis could be the short application of HS, which may have compromised the ability of the body to challenge, with the first line of defense, against HS; while body lipolysis, gluconeogenesis, and other pathway activation or metabolite changes require time to elaborate [
33]. Another hypothesis could be attributed to the low sample size (e.g., due to fluctuations in blood metabolites). It is well-known that blood metabolite alterations in response to stress conditions are subject to fluctuations, particularly when the size of the experimental unit is low [
18]. In order to lessen these fluctuations, a larger sample size is recommended, in order to elaborate the variation effects within the data. In this way, we could possibly find significant differences in some blood metabolite profiles in future research.
A natural phenomenon in response to HS is a decrease in FI (direct effect), where such a decrease may alternatively cause a decrease in milk yield and some of its characteristics (indirect effects of HS). One study has established that 35% of decrease in milk yield was due to decreased FI, whereas 65% was governed by the direct physiological effect of HS [
4,
25]. However, decreases in FI can be improved by feeding the cows early in the morning and at night. In these cooler periods of the day, cows can consume up to 80% of their total daily DM intake [
16]. However, in cases where the night and morning times still exceed the upper critical THI (of 72)—such as the situation in this study, where cows were exposed to constant 24 h HS—the amount of feed consumed will not compensate for the greatly depressed intake during the day. Beyond THI = 69, each point increase in THI can cause milk reduction of 0.2 kg [
1,
25]. In other words, for each 1 °C raise in air temperature above the thermal comfort zone, an 0.85 kg decrease in FI occurs, which causes a milk yield decline of approximately 36% [
1,
4,
25]. Given the above discussion, the significant decrease in milk yield of HTHH group in this study could be speculatively explained.
HS not only may lessen milk yield, but can also negatively affect milk constituents—particularly in high yielding dairy cows [
25]. HS is widely responsible for a decline in milk fat, mainly due to higher concentrate ration and less fiber content or consumption of ration and, consequently, a disruption in fat synthesis in mammary glands due to increased body temperature [
34]. The decline in milk protein content [
13] may be due to specific down-thermoregulation activity of mammary protein synthesis [
25]. These assumptions could explain the decline in milk fat and the tendency for decreased protein content in the HTHH group of this study. A raise in THI compromises the ability of dairy cows to dissipate excessive heat from the bodies [
5], resulting in physiological changes such as reduced milk fat and protein contents [
13,
34]. Lower FI and thus less protein consumption aligned with decreased milk protein can also explain the decreased tendency of MUN. The mechanism may also rely on the lower urease activity in the wall of rumen and disruption in rumination [
25,
26], due to higher THI in HTHH group. Energy-corrected milk (ECM) determines the amount of energy in the milk, based upon milk fats and proteins, adjusted to 3.5% fat and 3.2% percent protein. Given the definition of ECM, it is obvious that the ECM showed a significant decrease in HTHH group, compared with the LTLH group, due to the lower milk fat and protein. The reasons for other milk constituents to remain unaffected in the HTHH group are unknown.
Cortisol is the first hormone to look at in blood, saliva, or hair, when assessing stress situations, such as HS. Circulating cortisol has been shown to be a very sensitive index of heat stress, heralding the onset of poor tolerance of severe climates [
5]. A high-temperature environment as a source of stress triggers a series of stress responses of the body [
18]. The cortisol level adapts to the adverse environment, which is the evaluated index for the degree of stress and plays an extremely important role in the body [
18]. Exposure to HS will shortly induce the production and release of cortisol from adrenal glands into the blood stream, in effect triggering a flood of glucose, which provides an immediate energy source for the body use. Given the above review, and as expected, the higher cortisol concentration in HTHH group was the result of short-term HS exposure in the corresponding animals. This result is consistent with numerous other studies, whether animals were exposed to short- or long-term HS [
35,
36]. It is worth noting that, as cortisol is one of primary responses of body to HS, it is not surprising to see its increase in less than an hour of stress exposure, and thus, cortisol increases can reflect acute stress conditions, which was observed under the conditions of this study.
Haptoglobin is an acute phase protein produced by the liver, which the body uses to clear free hemoglobin (found outside of red blood cells) from circulation [
8,
22,
35]. In other words, haptoglobin is a hemoglobin-binding protein which prevents oxidative damage by utilizing free hemoglobin and is integral in the formation of the haptoglobin–hemoglobin complex [
36]. Hemoglobin is the iron-containing protein complex that transports oxygen throughout the body. It is normally found within red blood cells (RBCs) [
35]. Haptoglobin binds to free hemoglobin in the blood. This forms a haptoglobin–hemoglobin complex, which is rapidly cleared out of circulation by the liver such that it can be broken down and the iron recycled. When an increased number of RBCs are damaged and/or break apart (hemolysis), they release their hemoglobin into the blood, increasing the amount of free hemoglobin in circulation, which is consistent with the obtained result of increased tendency of RBC, as hemoglobin carriers, in this study. On the other hand, when large numbers of RBCs are destroyed, haptoglobin levels in the blood will temporarily decrease, as the haptoglobin is used up faster than the liver can produce it [
22,
35]. A decrease in the amount of haptoglobin may be a sign of a condition that is causing red blood cells to be destroyed or to break apart [
35,
36], which was not the case in the present study. When the binding capacity of haptoglobin is exceeded, the free hemoglobin level in circulation goes up, which may cause tissue damage and/or organ dysfunction due to oxidative stress by free hemoglobin. In agreement with the presented results, an earlier study in beef calves showed higher haptoglobin supported by higher cortisol concentrations in blood [
8]; in addition, another study [
24] has reported an increase in serum haptoglobin in response to physical stress in cattle.
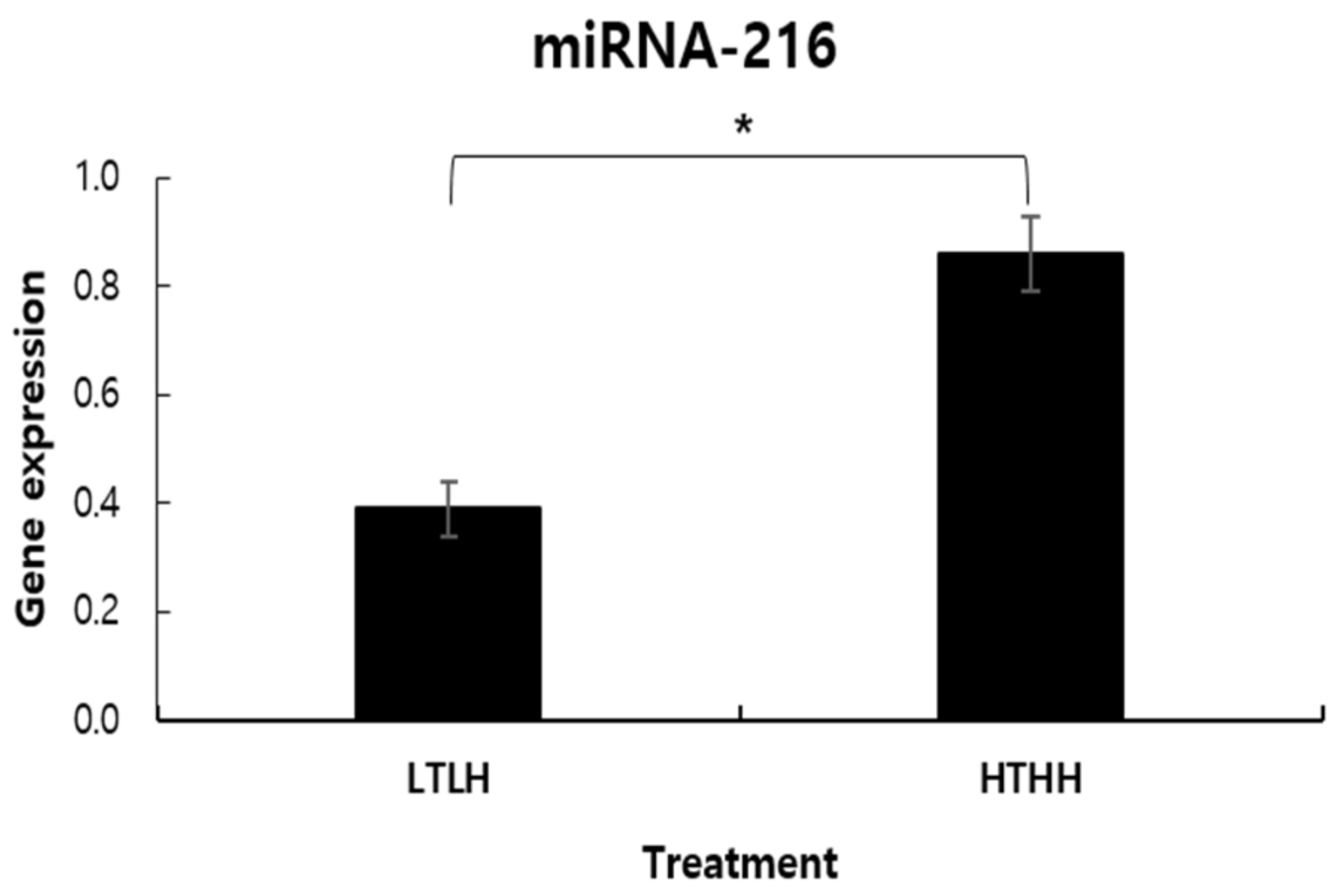
Thau et al. [
21] explained the mechanism behind higher gene expression due to stress conditions as follows: Steroid hormones, such as cortisol, are primary messengers. They can cross the cytoplasmic membrane due to their fat-soluble properties. Cell membranes are composed of phospholipid bilayers, which prevent fat-insoluble molecules from passing through. Once cortisol passes through the cell membrane and enters into the cell, it binds to specific receptors in the cytoplasm. In the absence of cortisol, the glucocorticoid receptor binds to a heat shock protein (HSP) 90 chaperone protein in the cytosol. The binding of cortisol to the glucocorticoid receptor dissociates HSP90. The cortisol–receptor complex then enters the nucleus of the cell and affects gene transcription. In addition, MicroRNAs (miRNAs) are small single-stranded non-coding RNA, which repress post-transcriptional gene expression that can be altered by cortisol via targeting HSPs to modulate HS responses in dairy cattle [
37]. Kumar et al. [
38] reported increased miRNA expression due to summer HS in Tharparkar and Sahiwal cattle. Subsequently, Kishore et al. [
39] reported higher expression of HSP40 transcript in Holstein Friesian, compared to Sahiwal cows, during summer HS [
37]. After a short-term HS (2 h) exposure, Shandilya et al. [
40] also reported induced mRNA expression of HSP40 and HPS70 in fibroblasts of zebu cattle. Taken together, the increase in miRNA expression in the HTHH group, compared to the LTLH group, in this study can be explained.
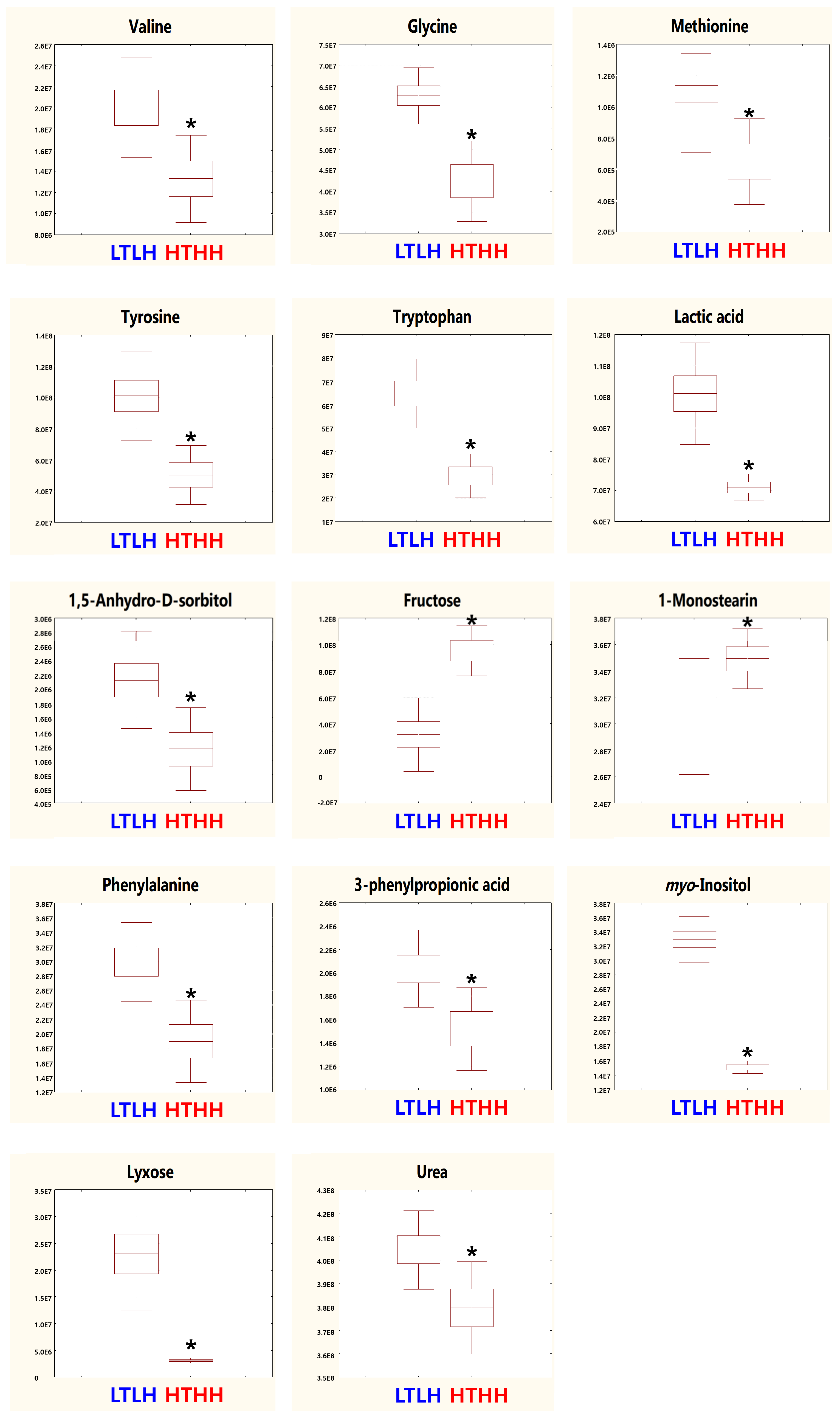
In our study, serum metabolic pathway analysis showed that the phenylalanine, tyrosine, and tryptophan biosynthesis and phenylalanine metabolism pathways were downregulated. Phenylalanine, tyrosine, and tryptophan are aromatic amino acids (AAA) which belong to the α-amino acid family for protein synthesis [
41]. Phenylalanine, tyrosine, and tryptophan have been reported to play regulatory roles under heat stress, through their co-expression network [
41]. In addition, AAAs play the role of precursors for numerous metabolomics related to protecting against stress, including melatonin, alkaloids, auxin, and phenolic compounds [
41]. AAAs play important roles in the metabolic processes of microflora in all animal bodies. The AAAs and metabolites derived from them also play integral roles in the health of animals [
41]. TAT1 is a T-system AA transporter, which plays an essential role in transporting AAAs. This transporter has been reported to be reduced in the chest and ileum of chickens affected by heat stress of 35 °C [
42]. Phenylalanine had reduced levels in the brain and liver of chickens, as well as embryos, after exposure to heat stress of 38 °C. Phenylalanine is converted to a phenylamine neurotransmitter in response to the PLP-dependent aromatic enzyme decarboxylase [
41]. In addition, it is often converted to tyrosine in the animal body, which is synthesized for epinephrine, dopamine, and norepinephrine neurotransmitters. Phenylalanine is also in charge of the biosynthesis of bacterial cell walls for inhibiting mureidomycins [
43] and antibodies classes [
44]. Tyrosine is changed to p-hydroxyphenylacetic acid by a mixture of bacteria and protozoa cultures, then converted to p-cresol [
41], which plays an important role in the production of antioxidants [
41]. Tyrosine is the precursor of the catecholamine neurotransmitters—dopamine and norepinephrine—which can administrate the behavioral, physiological, and neurochemical consequences, under a cold or heat stress environment, by adjusting the release of norepinephrine, thus demonstrating the role of tyrosine in protecting against the adverse effects of heat or cold stress [
45]. In addition, when tyrosine is activated to its thiol ester form, it can be attached to the enzyme modular thiosteraease enzyme for use in antibiotic synthesis [
41]. Tryptophan is the precursor for synthesis of serotonin, tryptamine, the neurohormone melatonin, and enzyme cofactors, which act as neurotransmitters [
41]. A study in steers has shown that tryptophan supplementation can slowly increase in RT in response to acute heat stress through an increase in brain 5-HT, followed by a presumable increase in evaporative heat loss from the skin surface in cattle [
46]. Among others, serotonin is involved in the melatonin synthesis metabolic process, which regulates growth activities in response to various biological stresses, such as pathogens, environmental toxins, and extreme temperature [
46]. In a previous study, myo-inositol has been reported to be involved in glucose uptake and insulin signaling regulation, as well as adipogenesis regulation [
9]. In the northern drosophila fly, high levels of myo-inositol were observed under a cold environment, which decreased in a warm or hot environment [
47]. In our current study, we also found the myo-inositol level to be significantly lower in the HTHH group; however, we did not find any significant change in the pathway analysis. The accumulation of glycine betaine can reduce the effect of HS and improve productivity in lactating dairy cows [
48]. A previous study has suggested that methionine supplementation can protect proteins from degradation by upregulating genes related to protein synthesis and decreasing genes related to protein breakdown [
49]. In our current study, we observed the downregulation of serum glycine and methionine, which indicated that the heat stress in the HTHH group may have adverse effects in early lactating cows. However, from the pathway analysis, we did not find any change related to phenylalanine metabolism, inositol phosphate metabolism, glyoxylate and dicarboxylate metabolism, cysteine and methionine metabolism, glycine, serine and threonine metabolism, and tryptophan metabolism, which implies that these parameters had limited adjustment in the pathway analysis. As per the aforementioned review, the downregulation of these two pathways and their final impact metabolomics resulted in negative modulation in immune parameters and biological polymers (e.g., proteins, muscle cells, and so on) due to HTHH conditions, even when only exposed in the short-term (4 d). Thus, we postulate the importance of the negative association observed between HTHH and the final products of the resulting pathways in this study.