Abstract
Immunization is a preventive measure of crucial importance. As with any other medication, side effects are a possibility and include the rare occurrence of severe infections, such as osteomyelitis. We report an unusual case of pediatric osteomyelitis following vaccination and provide a review of similar reports submitted to the Vaccine Adverse Event Report System (VAERS), aiming to explore the association between the vaccination procedure and the occurrence of osteomyelitis in childhood. A previously healthy infant, with no history of trauma or infection, presented with hyperpyrexia, swelling, and functional impairment in the left leg and was eventually diagnosed with osteomyelitis of the left femur. An edema was noted at the site of the injection that he received days before for immunization purposes. The infection required surgical drainage and a four-week-long intravenous antibiotic treatment, and the patient was discharged upon showing improved clinical conditions. Forty-seven reports of similar cases submitted to VAERS between 1994 and 2023 were collected, and several cases from the literature, including a case of femoral osteomyelitis in a newborn vaccinated against Hepatitis B, attributed to improper injection technique. Another case was reported in a 15-year-old girl, which aligned with six similar cases of osteomyelitis in adolescents following HPV vaccines collected from VAERS. Despite the small sample number, the findings that in 77% of cases the infection was localized in the vaccinated limb and that symptoms appeared on average 4.3 days (IQR 1.0–5.7 days) post-vaccination suggest a possible link to the injection procedure and highlight the need to adhere to recommendations regarding skin preparation and the selection of the appropriate needle length and injection site.
1. Background
Vaccinations are the most effective and safe intervention for preventing major infectious diseases and represent an essential component of global public health programs. Over the last millennium, they have contributed to eradicating or reducing the burden of many diseases, saving more human lives than any other medical procedure in history [1].
The surveillance of adverse effects following immunization is mandatory. In Italy, data are collected by a national pharmacovigilance network known as “Rete Nazionale di Farmacovigilanza (RNF)”, which includes the Italian Medicine Agency (Agenzia Italiana del Farmaco, AIFA), the 20 regions and the autonomous provinces of Trento and Bolzano, 204 local health authorities, 112 hospitals, 38 research institutes, and 561 pharmaceutical companies [2,3].
The most common side effects of vaccination are warmth, swelling, pain at the injection site, fever, headache, and fatigue [4]. Allergic reactions can also occur; therefore, a 15–30 min long observation period is recommended post-procedure [4,5]. Bacterial infections following vaccination, however rare, have been occasionally reported and include cellulitis [6,7], abscesses [8,9,10,11], septic arthritis [12,13,14,15], bacteremia [16], and osteomyelitis [17,18,19]. In the United States, between 1991 and 2001, more than 1.9 billion doses of vaccines were distributed and 128,717 adverse events were reported to the Vaccine Adverse Event Report System (VAERS) [20]. Of these, 2148 (1.7%) were reports of cellulitis and 426 (0.3%) were reports of abscesses at the injection site [20].
Common responsible agents for these infections are Gram-positive cocci, especially Staphylococcus aureus (S. aureus), an opportunistic commensal residing in the nostrils, oropharynx, and skin of healthy individuals [21]. S. aureus can cause a broad spectrum of conditions, including skin and soft tissue infections, osteomyelitis, pneumonia, bacteremia, endocarditis, sepsis, and toxic shock syndrome (TSS) [22].
We describe a case of osteomyelitis in a previously healthy infant, likely caused by the direct inoculation of the microorganism occurring during the vaccination procedure and provide a review of pediatric cases of osteomyelitis following vaccination collected from VAERS, with the objective of exploring the association between the vaccination procedure and the occurrence of osteomyelitis in childhood.
2. Case Report
A 3-month-old previously healthy infant was admitted to our pediatric infectious disease department in November 2023 because of swelling at the site of injection in the left lower limb, functional impairment, and hyperpyrexia. He had been immunized against meningococcus B and rotavirus about 12 days before admission.
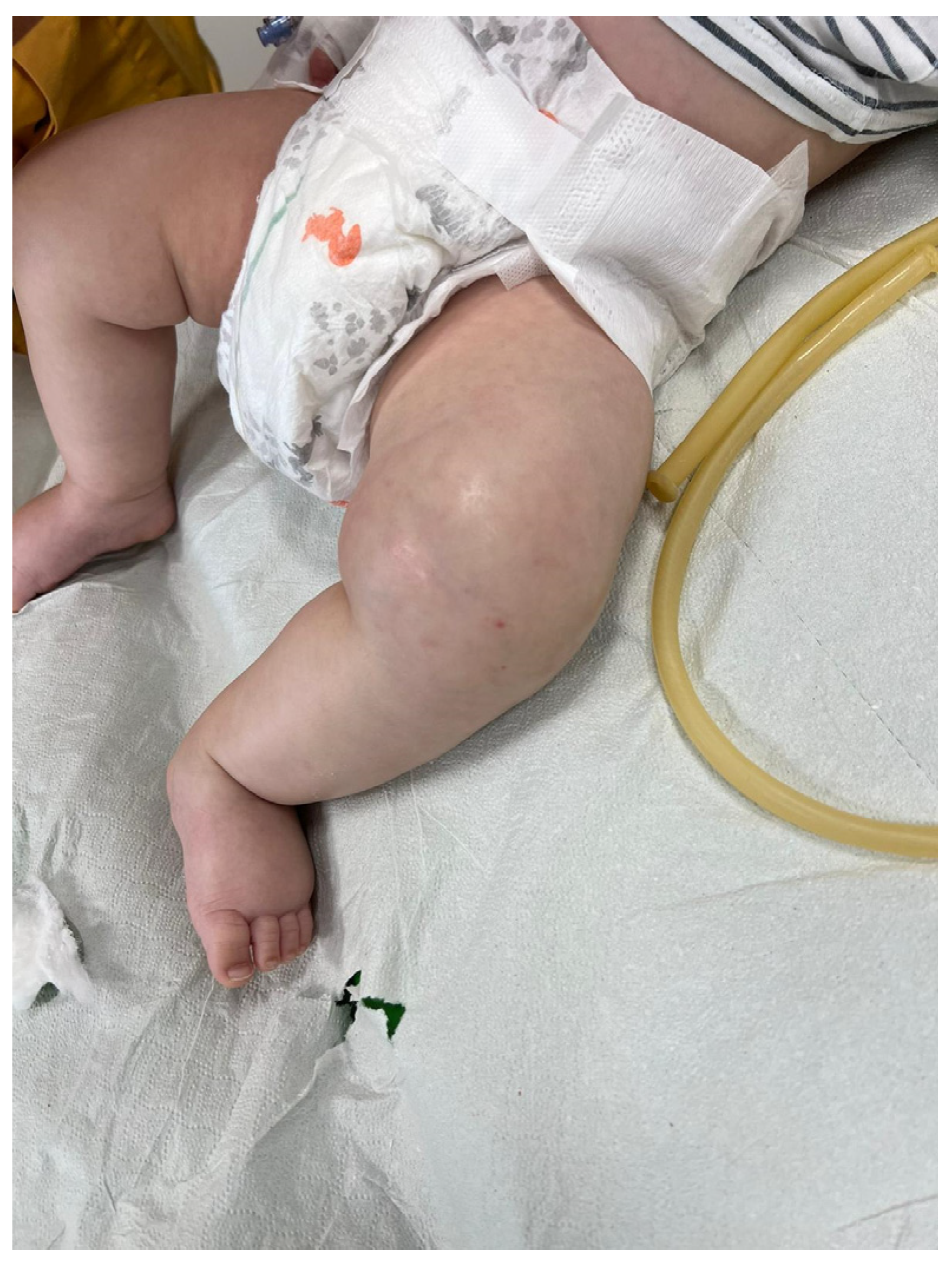
The patient’s parents did not report any prior medical conditions, and the infant exhibited good adaptation to extrauterine life and regular growth. The following findings were noted upon physical examination: SpO2, 98% in room air; heart rate, 170 bpm; respiratory rate, 35 breaths/min; and temperature, 38.5 °C. The left lower limb appeared externally rotated and flexed. The supra-patellar and thigh regions were edematous, with taut, warm, and tender skin (Figure 1).
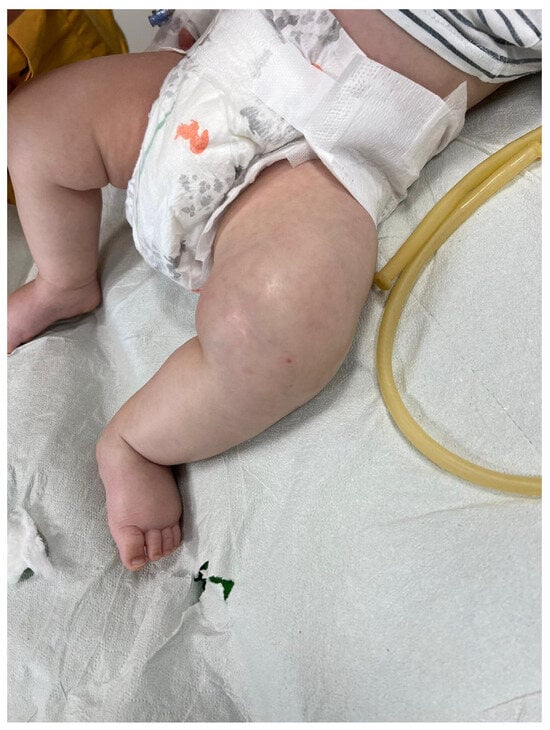
Figure 1.
Edematous left lower limb with tense skin in a 3-month-old infant affected by osteomyelitis and soft tissue infection.
Upon admission, antibiotic therapy was initiated with daptomycin 10 mg/kg/day and meropenem at the dosage of 60 mg/kg/day, divided into three administrations. The outcomes of haemato-chemical exams performed at various times during recovery are detailed in Table 1, and an ultrasound showed a voluminous collection in the proximity of the left-femoral region measuring 39 × 40 mm, with elevation.

Table 1.
Results of the haemato-chemical laboratory tests performed at different times during the hospital stay.
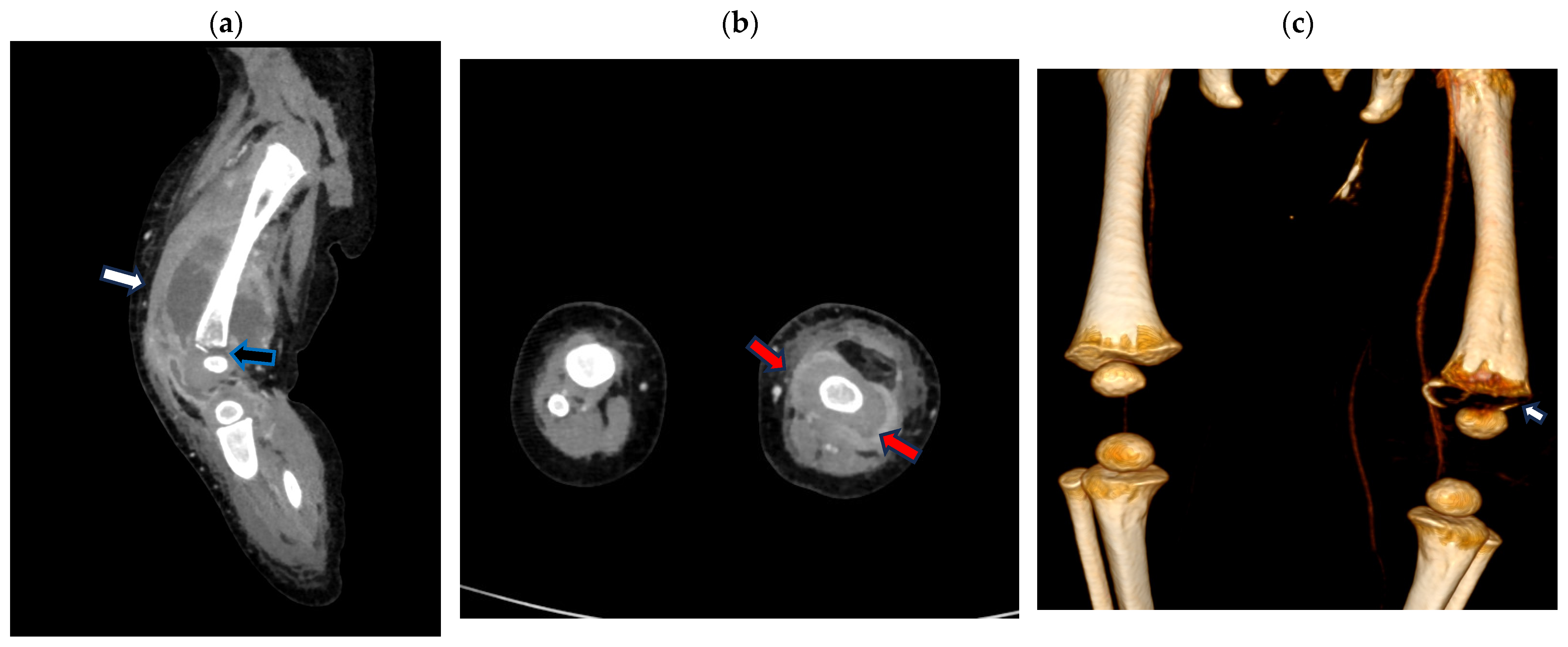
A CT scan performed on the second day of hospitalization evidenced a large, encapsulated abscess collection, 9 × 4 cm in size, surrounding the distal metaphyseal femoral bone and extending to the joint. The cortical erosion of the metaphyseal distal bone, which appeared frayed, and irregularities in the ossification core were observed (Figure 2).
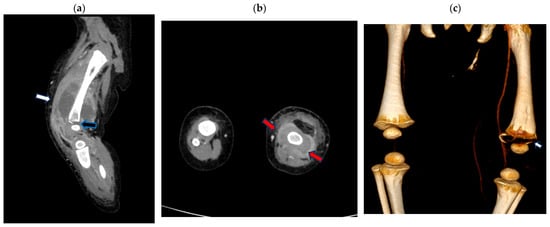
Figure 2.
Contrast-enhanced computed tomography (CT) scan of the left leg (a) sagittal multi-planar (MPR) reconstruction: left fluid collection surrounding the distal metaphyseal femoral bone (white arrow) extending to the joint with the cortical erosion/destruction of the metaphyseal distal bone (black arrow); (b) axial MPR reconstruction: left fluid collection surrounding the distal metaphyseal femur (red arrow); (c) volume rendering CT reconstruction bone window demonstrating distal metaphyseal bone erosion (arrow).
The surgical drainage of the abscess collection was performed, and a suction drain was placed in the left thigh. A cultural exam of the drained fluid revealed the presence of methicillin-sensitive S. aureus (MSSA). Therefore, antibiotic therapy was modified to cefazolin 150 mg/kg/day in three administrations for 34 days and rifampicin 10 mg/kg/day in two administrations for 32 days.
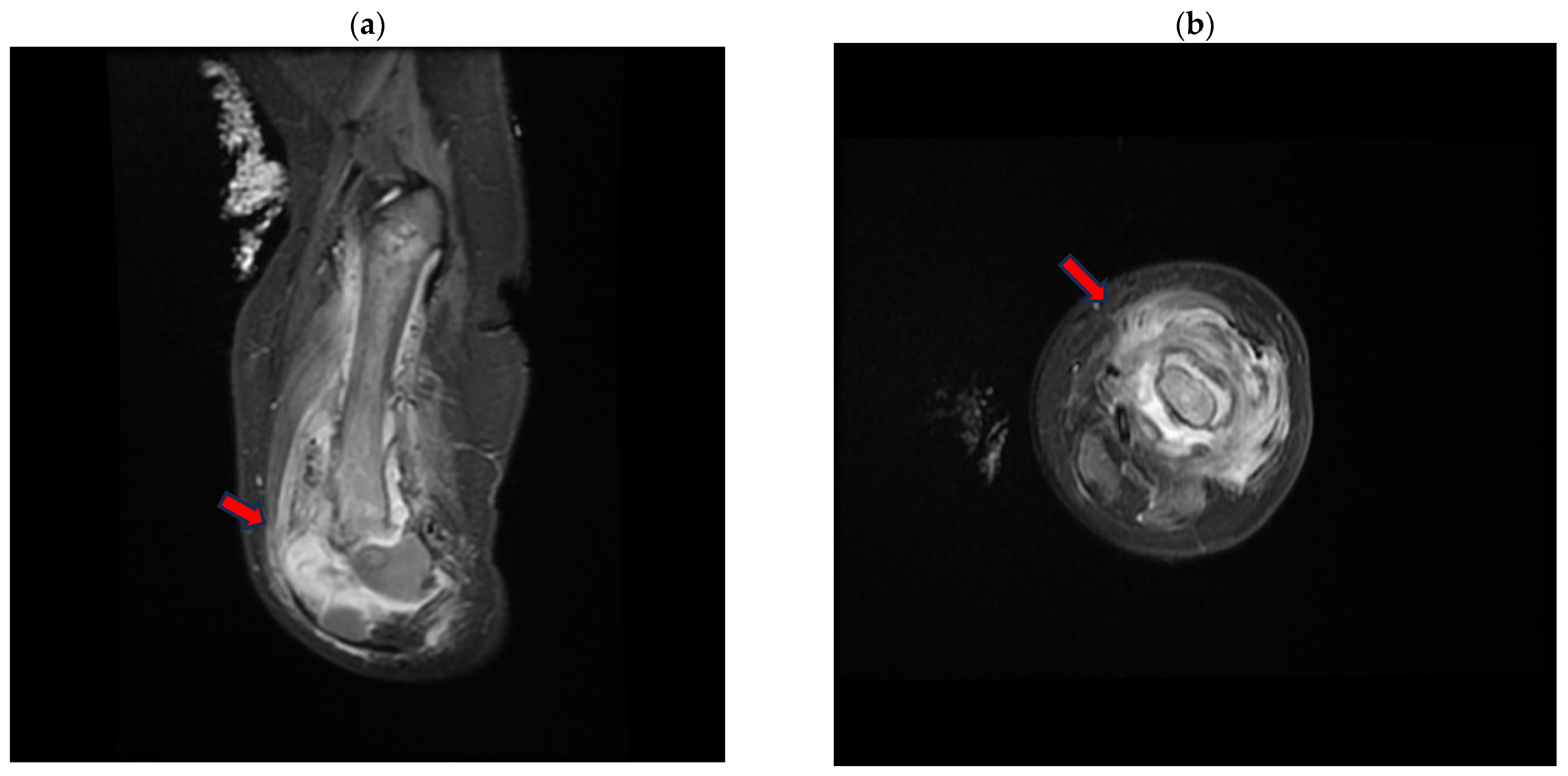
Fifteen days after the start of specific antibiotic therapy, an MRI was performed and showed a size reduction in the peripheral collection at the left-femoral diaphysis and the distal meta-epiphyseal region (Figure 3).
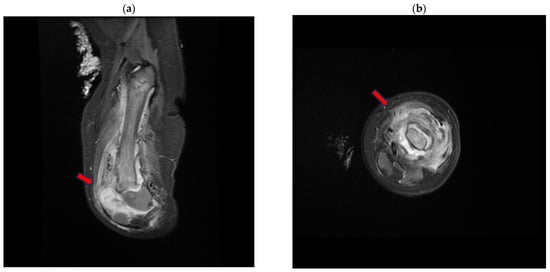
Figure 3.
Magnetic resonance (MR) short tau inversion recovery (STIR) of the left-lower-limb coronal (a) and axial (b) planes, demonstrating the large distal metaphyseal collection (arrow) expanding in the vastus lateralis muscle with an inhomogeneous signal from the distal femoral bone.
The maximum thickness was approximately 9 mm in the anterior distal location, compared to the previous 16 mm.
During hospitalization, a gradual yet progressive improvement in the patient’s clinical condition was observed, with the resolution of fever and the normalization of inflammatory markers (Table 1). Signs and symptoms, including pain, improved steadily, and the active and passive mobilization of the left lower limb resumed. Three weeks after admission, the child contracted influenza A.
Four weeks of intravenous antibiotic treatment were completed, and the patient was discharged in good clinical condition after receiving a single dose of dalbavancin at the dosage of 22.5 mg/kg. Three weeks after discharge, an outpatient re-evaluation and a total body PET scan were performed, and a modest radiotracer uptake, possibly a consequence of the inflammatory process, was noted in the distal part of the left femur. Despite the steady recovery, a discrepancy in limb length was reported at follow-up.
3. Methodology
We searched VAERS in December 2023 for reports of osteomyelitis in children (ages 0 to 17 years) vaccinated between 1990 and 2023. The following manifestations were selected, with the corresponding codes: osteomyelitis (10031252); osteomyelitis acute (10031253); osteomyelitis bacterial (10065237); osteomyelitis chronic (10031256); and staphylococcal osteomyelitis (10064250) [23].
We included all cases where a diagnosis of osteomyelitis in childhood was clearly reported and followed a vaccination administered subcutaneously or intramuscularly.
Information gathered by VAERS is subject to reporting biases, inaccuracy, and incompleteness. Reports can be submitted by the public at large, and the numerosity of reports of events following vaccinations is not sufficient to infer causation. To counter these limitations, we excluded layperson reports, reports where data were insufficient to determine a diagnosis of osteomyelitis, and cases where a correlation between the vaccination procedure and osteomyelitis was not plausible, for instance, because of the vaccination route, the timing of symptom onset, or the occurrence of a trauma that better explained the infection. Data collected from this resource should be interpreted within the context of the existing literature.
4. Results
The search returned 113 total events. Sixty-six were excluded for various reasons. In 32 cases, a diagnosis of osteomyelitis was not confirmed; in 11 cases, osteomyelitis was related to the Bacillus of Calmette–Guérin (BCG) vaccine; 17 cases were excluded because a correlation with the vaccination procedure was unlikely due to the vaccination route (i.e., oral vaccines), the identified pathogens (Pneumococcus in ten cases, Mycobacterium Avium Complex in one case, and Hemophilus in one case) or the timing of symptom onset; and 6 reports were excluded because they were duplicates or not submitted by health professionals. A total of 47 cases of pediatric osteomyelitis following vaccination were selected (detailed in Table 2).

Table 2.
Summary of the reports included, collected from VAERS.
Twenty-four were female (52%). The mean age was approximately 4.7 years, with an interquartile range (IQR) between 0.85 and 10.5, and the median age was 2 years. Three were newborns (6%), 66% were up to 4 years old (n = 31), 6% were between 5 and 9 years old (n = 3), and 28% were between 10 and 17 years old (n = 13).
In 77% of the cases where this information was available, osteomyelitis affected the same limb into which the vaccine was injected. The time elapsed between vaccination and symptom onset was reported in all cases but five. The mean time to onset was 4.3 days (IQR, 1.0–5.7; range, between 0 and 23 days). The median time to onset was 2 days. In 86% of cases where the time to onset was known (n = 36), symptoms manifested within a week. Only in 7% of cases (n = 3) did more than 15 days pass before the onset of clinical manifestations (19, 20, and 23 days).
Ultrasound findings included soft-tissue edema and fluid collections near the infection site or underneath the periosteum. X-ray findings included new bone formation, alterations in bone density, and pathological fractures. MRI was the most frequently reported imaging technique, and findings included subperiosteal or soft-tissue fluid collections (indicative of abscesses or phlegmons), soft-tissue edema and the involvement of muscles and joints (myositis or arthritis), and periosteal reaction or elevation. In four instances, scintigraphy findings of hypercaptation indicative of increased metabolic activity were reported.
In 56% of cases (13 out of 23) where an etiological diagnosis was reported, S. aureus species were isolated, either from a wound or blood sample. In two cases, Group A Streptococcus pyogenes was isolated from the blood; in one case, Citrobacter freundii; in another case, it was unspecified “Gram-positive cocci”; and in another case, S. capitis, S. epidermidis, and Pseudomonas aeruginosa were isolated together with S. aureus. In six cases, cultures were negative. Blood samples were used for cultures in all cases but one, in which a wound sample was used.
Information on antibiotic sensitivity was reported only in seven of these cases: in four, MRSA was isolated, and in three, MSSA was isolated.
Treatment included antibiotics and the incision and drainage of abscesses.
In several cases, reports included information on long-term sequelae, such as permanent disability, persistent unstable gait, limb length discrepancy, bone deformities, and impaired bone growth.
The outcome was positive in 25 cases out of 30 where information on recovery was available. In the remaining five cases, the children had not yet recovered at the time that the report was submitted.
5. Discussion
Vaccines are crucial in the prevention of infectious diseases and have saved millions of lives over the course of the last century [1]. Immunization side effects are infrequent and are mild in most cases [24]. Although rare, infections following intramuscular or subcutaneous injections, including vaccines, can occur due to the direct inoculation of skin flora bacteria. Skin bacteria that might enter the body through injection are nearly always non-pathogenic [25,26]; however, the possibility of severe infections, especially in children and immunocompromised individuals, warrants consideration, as it can inform preventive measures.
A literature search revealed that a case of femoral osteomyelitis occurred in a newborn with no known infective risk factors one day after Hepatitis B vaccination [18]. The authors attributed the infection to inadequate site preparation and excessive depth of needle insertion [18]. About a decade later, a case of osteomyelitis localized in the arm was reported in a 15-year-old girl following HPV vaccination [17]. In this case, the infection was linked to the overpenetration of the needle during injection [17]. Reports of six similar cases of osteomyelitis in adolescents (five girls and one boy) following HPV vaccination were gathered from VAERS (Table 2). Most recently, a case of osteomyelitis caused by C. freundii following immunization with the Moderna COVID-19 vaccine was reported [19]. The patient, a 15-year-old boy, also developed deep vein thrombosis (DVT), a pulmonary embolism, and acute respiratory distress syndrome. The authors noted that both the vaccine itself and the bone infection could explain the thrombotic event [19].
Of the cases collected in this review, the median time to the onset of symptoms (such as fever, pain, and functional impairment) was 2 days, and in most cases (77%), the infection affected the same limb into which the vaccine was injected. Similarly, in the case report, the affected limb was the same as that of vaccination, and signs and symptoms manifested within two weeks, indicating a possible link to the injection procedure.
According to the World Health Organization (WHO), a safe injection is one that does not cause harm to the recipient, does not produce dangerous waste, and does not put the provider at risk [27]. Unsafe injections can cause viral, bacterial, or fungal infections, and recommended safety practices include hand hygiene, using gloves when appropriate or other single-use protective materials, and skin preparation through disinfection [27].
The recommended skin disinfection procedure, while varying based on the type of injection, generally consists of the use of a swab soaked with a 60–70% alcohol solution to wipe the injection site area from the center outwards [27]. The solution should be applied for 30 s, and the site should be allowed to dry for an additional 30 s [27]. Of note, according to WHO recommendations, skin preparation for vaccines should be performed with soap and water rather than with alcohol solutions [27]. According to the CDC general best practice guidelines for immunization, health practitioners administering vaccines should follow general precautions to lower the risk of infections, such as hand cleansing with an alcohol-based antiseptic or hand washing with soap and water before the procedure [28].
A review conducted by the Canadian Agency for Drugs and Technologies in Health concluded that the question of whether skin disinfection prior to injections should be performed is difficult to answer [29]. However, the criteria for inclusion in this review were met by only one randomized controlled trial (RCT) [30], where no statistically significant difference was found in infection incidence between the group of children where swabbing at the injection site was performed prior to vaccination and the control group, where alcohol swabbing was performed adjacent to the vaccination site. However, the RCT did not have sufficient statistical power to accurately assess its main outcome, skin infection, as there were no cases of pus leaking from the injection site, cellulitis, or abscesses. Additional RCTs, with a broadened focus on various types of injections, would be needed to assess the effectiveness of skin disinfection [29,30].
Several authors have questioned the utility and necessity of skin disinfection prior to injections over time, highlighting limitations in related studies, such as small sample sizes and recall bias [31,32,33,34]. A recent review on the importance of skin disinfection prior to vaccination found that only 421 out of 1534 infectious events following injections (27.4%) were due to exogenous contamination, demonstrating the role of endogenous contamination as an infective source during vaccination [35].
The Advisory Committee on Immunization Practices (ACIP) specifies that vaccines should be injected into areas where tissue injury at the neural, vascular, or cutaneous level is unlikely. Furthermore, needle length should be established based on the age and body mass of the patient, in order to reach the muscle mass while leaving nerves, blood vessels, and bone untouched [36,37,38,39]. For newborns, a 5/8” needle (22–25 gauge) is indicated, while for infants, (1–12 months) a 1” needle (22–25 gauge) is recommended [40,41]. The recommended injection site is the anterolateral thigh muscle [40,41]. Regarding the reported case, we lack details on the thoroughness of disinfection, penetration depth, or needle length used. However, inflammatory manifestations at the injection site, in the absence of a history of trauma or other infection sources, and the temporal correlation support the possibility of causality in this case.
Children are more vulnerable to osteomyelitis due to their highly vascularized, developing skeletal system combined with an immature immune system, and they face a higher risk of sequelae such as bone asymmetry and gait disturbances. In the reported case, a previously healthy child with no risk factors developed limb asymmetry due to femoral osteomyelitis following a simple preventive procedure.
Osteomyelitis is an inflammatory process with accompanied bone destruction, with long bones (like the femur and tibia) being the most commonly affected sites in pediatric age [42]. The infection can spread to the adjacent joint space in areas where the metaphysis is intracapsular, such as the proximal ends of the femur, humerus, and tibia. Bone infection can develop due to the hematogenous dissemination, contiguous spread, or direct inoculation of bacteria [43], and, in children, it typically follows trauma, which acts as an infective source leading to bacteremia and bone infection [42]. In our case, we hypothesize that the infection was caused by either direct inoculation in the bone tissue due to excessive needle penetration or the hematogenous dissemination of cutaneous bacteria.
The pathogen most commonly responsible for osteomyelitis is S. aureus, a frequent colonizer of human skin and mucous membranes [44,45], which was responsible for most of the cases of osteomyelitis that we reviewed with a known etiological diagnosis. In our case report, MSSA was isolated from drained fluid. S. aureus possesses surface receptors known as MSCRAMMs, microbial surface components that bind to adhesive matrix molecules, mediating its adhesion to bone matrix components [42,46]. Once attached to the bone tissue, S. aureus survives in the intracellular space favored by its metabolically quiescent phenotype [47], which makes its eradication through antibiotics challenging.
Other bacteria responsible for pediatric osteomyelitis are S. pyogenes, S. pneumoniae, Salmonella, H. influenzae, and K. kingae [43,44]. Among the reviewed cases, other isolated pathogens besides S. aureus included S. pyogenes, S. capitis, S. epidermidis, and P. aeruginosa. These microorganisms can be part of the skin flora, reinforcing a possible causal link with the injection procedure.
Acute osteomyelitis is more common in children, particularly under 16, than adults [48,49,50]. In children, the rich vascularization and the stagnant blood in venous sinusoids, especially at the distal ends of long-bone metaphyses, can favor the establishment of microorganisms when bacteremia occurs [51]. Bone microtrauma can also favor infection, as it affects local blood supply and causes the exposure of host matrix proteins to which bacteria can adhere.
The treatment of acute osteomyelitis in children includes an antibiotic course over 4–6 weeks [52], pain medications, and sometimes surgical procedures. In our case, empirical intravenous treatment was started and later rationalized based on the cultural exam.
Regarding the minimum duration of oral treatment, the current data are insufficient to establish a clear recommendation. We decided on a long-course intravenous treatment given the young age of the patient and the severity of the clinical picture. We opted for a 4-week treatment followed by one dose of dalbavancin, instead of a 6-week-long treatment, to reduce the length of hospitalization and its associated risks. The use of dalbavancin, approved in the pediatric population to treat acute bacterial skin and skin structure infections (ABSSSIs), has the advantage of single-dose administration, with subsequent pain reduction and early discharge, minimizing the risk of nosocomial infections and hospitalization costs [53].
In conclusion, the necessity of skin preparation prior to injection is debated, and more blinded RCTs would be necessary to remove doubts on this matter. However, to minimize the risks, costs, and potentially severe sequelae of infections related to the immunization procedure, it is important to adhere to appropriate healthcare practices concerning vaccinations. This includes the use of single-use materials, the selection of the correct needle length and site, and proper skin preparation.
Author Contributions
Conceptualization, C.C. (Claudia Colomba), V.G. and L.V.; methodology, C.C. (Claudia Colomba), V.G. and L.V.; validation, C.C. (Claudia Colomba), A.C. (Antonio Cascio) and V.G.; formal analysis, C.C. (Claudia Colomba), V.G. and L.V.; investigation, V.G., L.V., C.C. (Costanza Caruana), A.C. (Anna Condemi), A.C. (Alessandra Cuccia), C.A., G.B. and V.F.P.; data curation, V.G., L.V., C.C. (Costanza Caruana), A.C. (Alessandra Cuccia), A.C. (Anna Condemi), and G.B.; writing—original draft preparation, V.G., L.V., C.C. (Costanza Caruana), A.C. (Alessandra Cuccia), and C.A.; writing—review and editing, V.G., L.V. and C.C. (Claudia Colomba); visualization, S.S. and V.F.P.; supervision, C.C. (Claudia Colomba), A.C. (Antonio Cascio), and S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this study because it involves a review of secondary data from the VAERS (Vaccine Adverse Event Reporting System) database, which is publicly available and contains only anonymous information.
Informed Consent Statement
Signed informed consent for publication was obtained from the child’s father.
Data Availability Statement
All relevant data are included in the article.
Conflicts of Interest
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Abbreviations
| AB | Antibiotics |
| ABSSSI | Acute bacterial skin and skin structure infections |
| ACIP | Advisory Committee on Immunization Practices |
| AIFA | Agenzia Italiana del Farmaco (Italian Medicine Agency) |
| ARDS | Acute respiratory distress syndrome |
| CDC | Centers for Disease Control and Prevention |
| CT | Computed tomography |
| DTaP | Diphtheria, Tetanus, and Pertussis Vaccine |
| DT-IPV | Diphtheria and Tetanus Toxoids and Inactivated Poliovirus Vaccine |
| DVT | Deep vein thrombosis |
| Hep B | Hepatitis B vaccine |
| Hib | Haemophilus influenzae type b vaccine |
| HPV | Human papillomavirus vaccine |
| IPV | Inactivated poliovirus vaccine |
| LA | Left arm |
| LL | Left leg |
| MMR | Measles, Mumps, and Rubella Vaccine |
| MO | Mouth (in the context of vaccine administration) |
| MPR | Multi-planar reconstruction |
| MRSA | Methicillin-resistant Staphylococcus aureus |
| MRI | Magnetic resonance imaging |
| MSSA | Methicillin-sensitive Staphylococcus aureus |
| NR | Not reported |
| Pneumo | Pneumococcal vaccine |
| RA | Right arm |
| RCT | Randomized controlled trial |
| RL | Right leg |
| RNF | Rete Nazionale di Farmacovigilanza (National Pharmacovigilance Network) |
| ROM | Range of motion |
| STIR | Short tau inversion recovery |
| Tdap (Adacel, Boostrix) | Tetanus, Diphtheria, and Pertussis Vaccine (specific brands) |
| US | Ultrasound |
| VAERS | Vaccine Adverse Event Reporting System |
| WHO | World Health Organization |
| XR | X-ray |
References
- A Brief History of Vaccination. Available online: https://www.who.int/news-room/spotlight/history-of-vaccination/a-brief-history-of-vaccination (accessed on 17 February 2024).
- EMA Pubblica i Risultati del Progetto Europeo sul Monitoraggio e la Valutazione dei Farmaci (PROTECT). Available online: https://www.aifa.gov.it/-/ema-pubblica-i-risultati-del-progetto-europeo-sul-monitoraggio-e-la-valutazione-dei-farmaci-protect- (accessed on 17 February 2024).
- La Rete Nazionale di Farmacovigilanza (RNF). Available online: https://www.aifa.gov.it/rete-nazionale-di-farmacovigilanza (accessed on 17 February 2024).
- Possible Side Effects from Vaccines|CDC. 2023. Available online: https://www.cdc.gov/vaccines/vac-gen/side-effects.htm (accessed on 24 February 2024).
- Vaccine Administration Route and Site|CDC. 2023. Available online: https://www.cdc.gov/vaccines/hcp/admin/administer-vaccines.html (accessed on 24 February 2024).
- AlBassri, T.K.; AlShaibi, S.; Khan, F.; Masud, N. A rare case of cellulitis after tetanus toxoid (TT) vaccination. J. Fam. Med. Prim. Care. 2020, 9, 1762–1764. [Google Scholar] [CrossRef]
- Lapphra, K.; Scheifele, D. Vaccination site reaction or bacterial cellulitis? Paediatr. Child. Health 2009, 14, 245. [Google Scholar] [CrossRef][Green Version]
- Mohanty, S.; Mohapatra, P.R.; Devi, S.; Saha, S. Post-COVID-19 vaccination shoulder abscess and pleuroparenchymal pulmonary lesion due to Burkholderia pseudomallei. IDCases 2022, 27, e01457. [Google Scholar] [CrossRef]
- Addison, M.; Lazzaro, T.; Crawford, N. Injection site abscess (ISA) following an infant vaccine. Vaccine 2019, 37, 1151–1152. [Google Scholar] [CrossRef]
- Bernier, R.H. Abscesses Complicating DTP Vaccination. Arch. Pediatr. Adolesc. Med. 1981, 135, 826–828. [Google Scholar] [CrossRef]
- Silva, L.; Lemos, S.; João Cabral, C.A. Intramuscular abscess following routine newborn immunization: A case report. J. Med. Case Rep. Case Ser. 2022. Available online: https://www.acquaintpublications.com/article/intramuscular_abscess_following_routine_newborn_immunization_a_case_report (accessed on 24 February 2024).
- Klabklay, P.; Chuaychoosakoon, C. Septic arthritis of shoulder joint following a COVID-19 vaccination: A case report. Int. J. Surg. Case Rep. 2022, 99, 107686. [Google Scholar] [CrossRef]
- Massel, D.H.; Haziza, S.; Rivera, S.; Mohile, N.; Subhawong, T.K.; Hernandez, V.H. Septic Arthritis of the Shoulder After SARS-CoV-2 Pfizer Vaccination: A Case Report. JBJS Case Connect. 2021, 11, e21.00090. [Google Scholar] [CrossRef]
- Damisa, J.; Zulkeefli, Z.; Rohman, L.; Burton, D. Shoulder septic arthritis following routine influenza vaccination. J. Surg. Case Rep. 2020, 2020, rjaa458. [Google Scholar] [CrossRef]
- DeRogatis, M.J.; Parameswaran, L.; Lee, P.; Mayer, T.G.; Issack, P.S. Septic Shoulder Joint after Pneumococcal Vaccination Requiring Surgical Debridement. HSS J. 2018, 14, 299–301. [Google Scholar] [CrossRef]
- Kus, M.S.P. 335. Staphylococcus aureus Bacteremia as a Potential and Severe Complication from Intramuscular COVID-19 Vaccine Injection. Open Forum Infect. Dis. 2021, 8 (Suppl. S1), S272. [Google Scholar] [CrossRef]
- Smith, S.S.; Lee, Y.; Wang, L. Adolescent with osteomyelitis after intramuscular administration of a vaccine: A case report. J. Am. Pharm. Assoc. JAPhA. 2020, 60, e357–e360. [Google Scholar] [CrossRef] [PubMed]
- Gupta, M.K.; Gupta, A.K. Neonatal osteomyelitis: An unusual but serious complication of intramuscular vaccination. J. Neonatol. 2011, 25, 105–106. [Google Scholar] [CrossRef]
- Adhikari, S.; Kc, S.; Dev, S.; Bhandari, S.; Parajuli, P.; Poudel, M.B.; Adhikari, Y.R.M.; Poudel, B.M. Citrobacter freundii-associated osteomyelitis and thromboembolic events following Moderna COVID-19 vaccination: A rare case report from Nepal. Ann. Med. Surg. 2023, 85, 5789–5794. [Google Scholar] [CrossRef] [PubMed]
- Surveillance for Safety After Immunization: Vaccine Adverse Event Reporting System (VAERS)—United States, 1991–2001. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/ss5201a1.htm (accessed on 12 February 2024).
- Liu, G.Y. Molecular Pathogenesis of Staphylococcus aureus Infection. Pediatr. Res. 2009, 65, 71–77. [Google Scholar] [CrossRef]
- Tong, S.Y.C.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G. Staphylococcus aureus Infections: Epidemiology, Pathophysiology, Clinical Manifestations, and Management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef]
- United States Department of Health and Human Services, Department of Health and Human Services (DHHS), Public Health Service (PHS), Food and Drug Administration (FDA)/Centers for Disease Control (CDC). Vaccine Adverse Events Reporting System (VAERS). 1990. Available online: https://wonder.cdc.gov/ (accessed on 17 February 2024).
- Vaccines and Immunization: Vaccine Safety. Available online: https://www.who.int/news-room/questions-and-answers/item/vaccines-and-immunization-vaccine-safety (accessed on 21 February 2024).
- Hutin, Y.; Hauri, A.; Chiarello, L.; Catlin, M.; Stilwell, B.; Ghebrehiwet, T.; Garner, J. Best infection control practices for intradermal, subcutaneous, and intramuscular needle injections. Bull. World Health Organ. 2003, 81, 491–500. [Google Scholar]
- Experimental Staphylococcal Infections in the Skin of Man—Elek—1956—Annals of the New York Academy of Sciences—Wiley Online Library. Available online: https://nyaspubs.onlinelibrary.wiley.com/doi/10.1111/j.1749-6632.1956.tb36626.x (accessed on 24 February 2024).
- WHO. Background. In WHO Best Practices for Injections and Related Procedures Toolkit; World Health Organization: Geneva, Switzerland, 2010. Available online: https://www.ncbi.nlm.nih.gov/books/NBK138493/ (accessed on 12 February 2024).
- General Recommendations on Immunization. Recommendations of the Advisory Committee on Immunization Practices (ACIP). Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/rr5515a1.htm (accessed on 13 February 2024).
- Dulong, C.; Brett, K.; Argáez, C. Skin Preparation for Injections: A Review of Clinical Effectiveness, Cost-Effectiveness and Guidelines; Canadian Agency for Drugs and Technologies in Health: Ottawa, ON, Canada, 2020; (CADTH Rapid Response Reports). Available online: http://www.ncbi.nlm.nih.gov/books/NBK562932/ (accessed on 9 February 2024).
- Wong, H.; Moss, C.; Moss, S.M.; Shah, V.; Halperin, S.A.; Ito, S.; Mithal, P.; Qu, A.; Taddio, A. Effect of alcohol skin cleansing on vaccination-associated infections and local skin reactions: A randomized controlled trial. Hum. Vaccines Immunother. 2019, 15, 995–1002. [Google Scholar] [CrossRef]
- Dann, T.C. Routine Skin Preparation Before Injection: An Unnecessary Procedure. Lancet 1969, 294, 96–98. [Google Scholar] [CrossRef]
- Koivisto, V.A.; Felig, P. Is skin preparation necessary before insulin injection? Lancet. Lond. Engl. 1978, 1, 1072–1075. [Google Scholar] [CrossRef]
- McCarthy, J.A.; Covarrubias, B.; Sink, P. Is the traditional alcohol wipe necessary before an insulin injection? Dogma disputed. Diabetes Care 1993, 16, 402. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, J.; Grinager, H.; Smith, S.D.; Sibley, S.; Harrison, A.R.; Lee, M.S. Isopropyl alcohol skin antisepsis does not reduce incidence of infection following insulin injection. Am. J. Infect. Control 2013, 41, 755–756. [Google Scholar] [CrossRef] [PubMed]
- Cook, I.F. Sepsis, parenteral vaccination and skin disinfection. Hum. Vaccines Immunother. 2016, 12, 2546–2559. [Google Scholar] [CrossRef] [PubMed]
- Zuckerman, J.N. The importance of injecting vaccines into muscle. Different patients need different needle sizes. BMJ 2000, 321, 1237–1238. [Google Scholar] [CrossRef] [PubMed]
- Bergeson, P.S.; Singer, S.A.; Kaplan, A.M. Intramuscular Injections in Children. Pediatrics 1982, 70, 944–948. [Google Scholar] [CrossRef]
- Poland, G.A.; Borrud, A.; Jacobson, R.M.; McDermott, K.; Wollan, P.C.; Brakke, D.; Charboneau, J.W. Determination of deltoid fat pad thickness. Implications for needle length in adult immunization. JAMA 1997, 277, 1709–1711. [Google Scholar] [CrossRef]
- Groswasser, J.; Kahn, A.; Bouche, B.; Hanquinet, S.; Perlmuter, N.; Hessel, L. Needle Length and Injection Technique for Efficient Intramuscular Vaccine Delivery in Infants and Children Evaluated Through an Ultrasonographic Determination of Subcutaneous and Muscle Layer Thickness. Pediatrics 1997, 100, 400–403. [Google Scholar] [CrossRef]
- General Recommendations on Immunization. Available online: https://www.cdc.gov/mmwr/preview/mmwrhtml/rr6002a1.htm (accessed on 15 February 2024).
- How to Administer Intramuscular and Subcutaneous Vaccine Injections. Available online: https://www.immunize.org/wp-content/uploads/catg.d/p2020.pdf (accessed on 15 February 2024).
- Kaushik, A.; Kest, H. Pediatric Methicillin-Resistant Staphylococcus aureus Osteoarticular Infections. Microorganisms 2018, 6, 40. [Google Scholar] [CrossRef]
- Gigante, A.; Coppa, V.; Marinelli, M.; Giampaolini, N.; Falcioni, D.; Specchia, N. Acute osteomyelitis and septic arthritis in children: A systematic review of systematic reviews. Eur. Rev. Med. Pharmacol. Sci. 2019, 23 (Suppl. S2), 145–158. [Google Scholar]
- Iliadis, A.D.; Ramachandran, M. Paediatric bone and joint infection. EFORT Open Rev. 2017, 2, 7–12. [Google Scholar] [CrossRef]
- Dodwell, E.R. Osteomyelitis and septic arthritis in children: Current concepts. Curr. Opin. Pediatr. 2013, 25, 58–63. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, R.; Cockayne, A.; Humphreys, H. Clinical and molecular aspects of the pathogenesis of Staphylococcus aureus bone and joint infections. J. Med. Microbiol. 1996, 44, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Tuchscherr, L.; Bischoff, M.; Lattar, S.M.; Noto Llana, M.; Pförtner, H.; Niemann, S.; Geraci, J.; Van de Vyver, H.; Fraunholz, M.J.; Cheung, A.L.; et al. Sigma Factor SigB Is Crucial to Mediate Staphylococcus aureus Adaptation during Chronic Infections. PLoS Pathog. 2015, 11, e1004870. [Google Scholar] [CrossRef] [PubMed]
- Walter, N.; Bärtl, S.; Alt, V.; Rupp, M. The Epidemiology of Osteomyelitis in Children. Children. 2021, 8, 1000. [Google Scholar] [CrossRef] [PubMed]
- Carek, P.J.; Dickerson, L.M.; Sack, J.L. Diagnosis and Management of Osteomyelitis. Am. Fam. Physician. 2001, 63, 2413–2421. [Google Scholar]
- Thakolkaran, N.; Shetty, A.K. Acute Hematogenous Osteomyelitis in Children. Ochsner J. 2019, 19, 116–122. [Google Scholar] [CrossRef]
- Trueta, J.; Morgan, J.D. The vascular contribution to osteogenesis. I Studies by the injection method. J. Bone Jt. Surg. Br. 1960, 42-B, 97–109. [Google Scholar] [CrossRef]
- Saavedra, J.; Falup-Pecurariu, O.; Faust, S.; Girschick, H.; Hartwig, N.; Kaplan, S.; Lorrot, M.; Mantadakis, E.; Peltola, H.; Rojo, P.; et al. Bone and Joint Infections. Bone Jt. Infect. 2017, 36, 788–799. [Google Scholar]
- Giorgobiani, M.; Burroughs, M.H.; Antadze, T.; Carrothers, T.J.; Riccobene, T.A.; Patel, R.; Lin, T.; Stefanova, P. The Safety and Efficacy of Dalbavancin and Active Comparator in Pediatric Patients with Acute Bacterial Skin and Skin Structure Infections. Pediatr. Infect. Dis. J. 2023, 42, 199–205. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).