Natural Products and Their Potential Anti-HAV Activity
Abstract
:1. Introduction
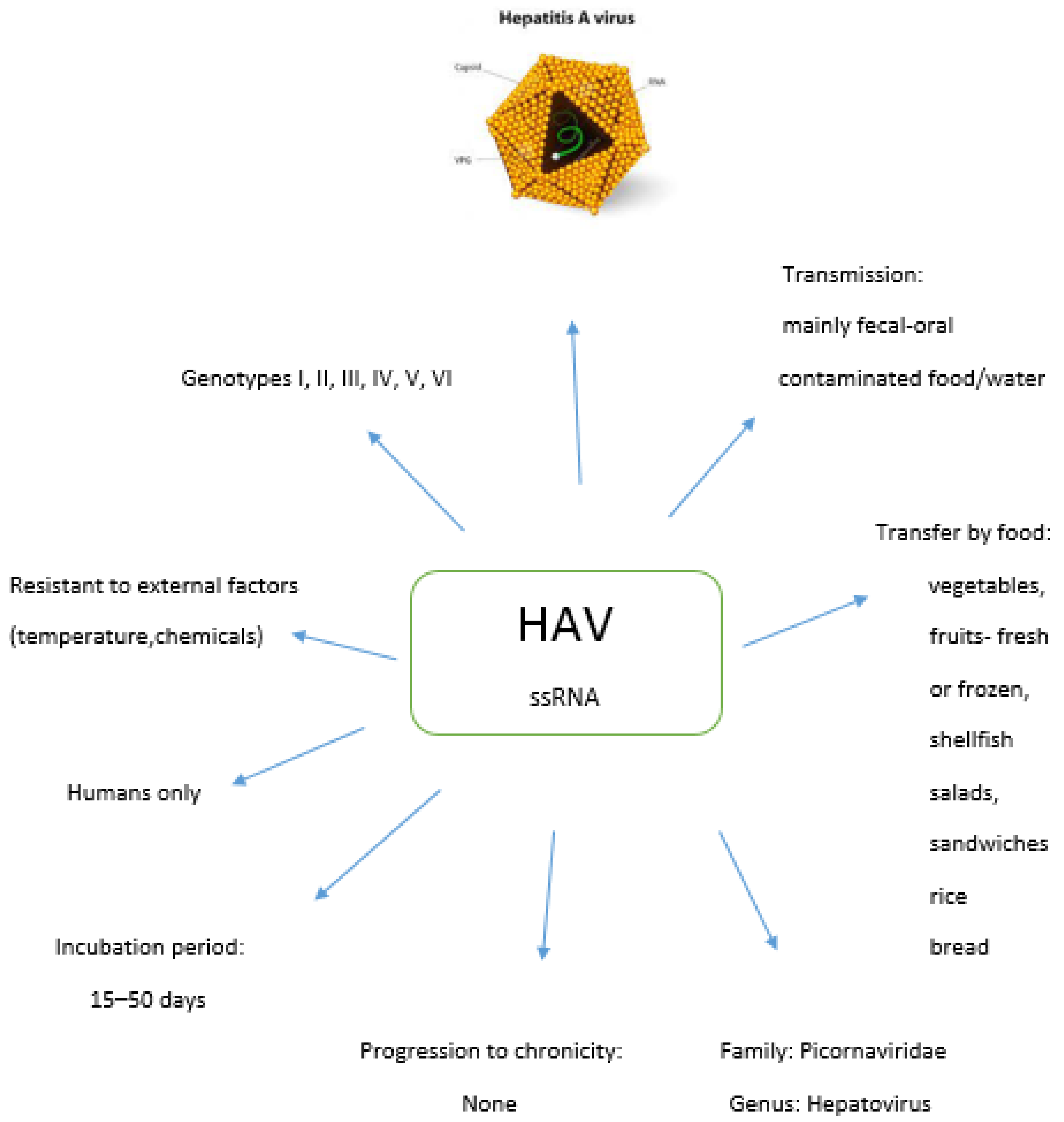
2. Hepatitis A Virus
2.1. Genotypes of HAV
2.2. Food Sources of HAV
2.3. Epidemiology of HAV
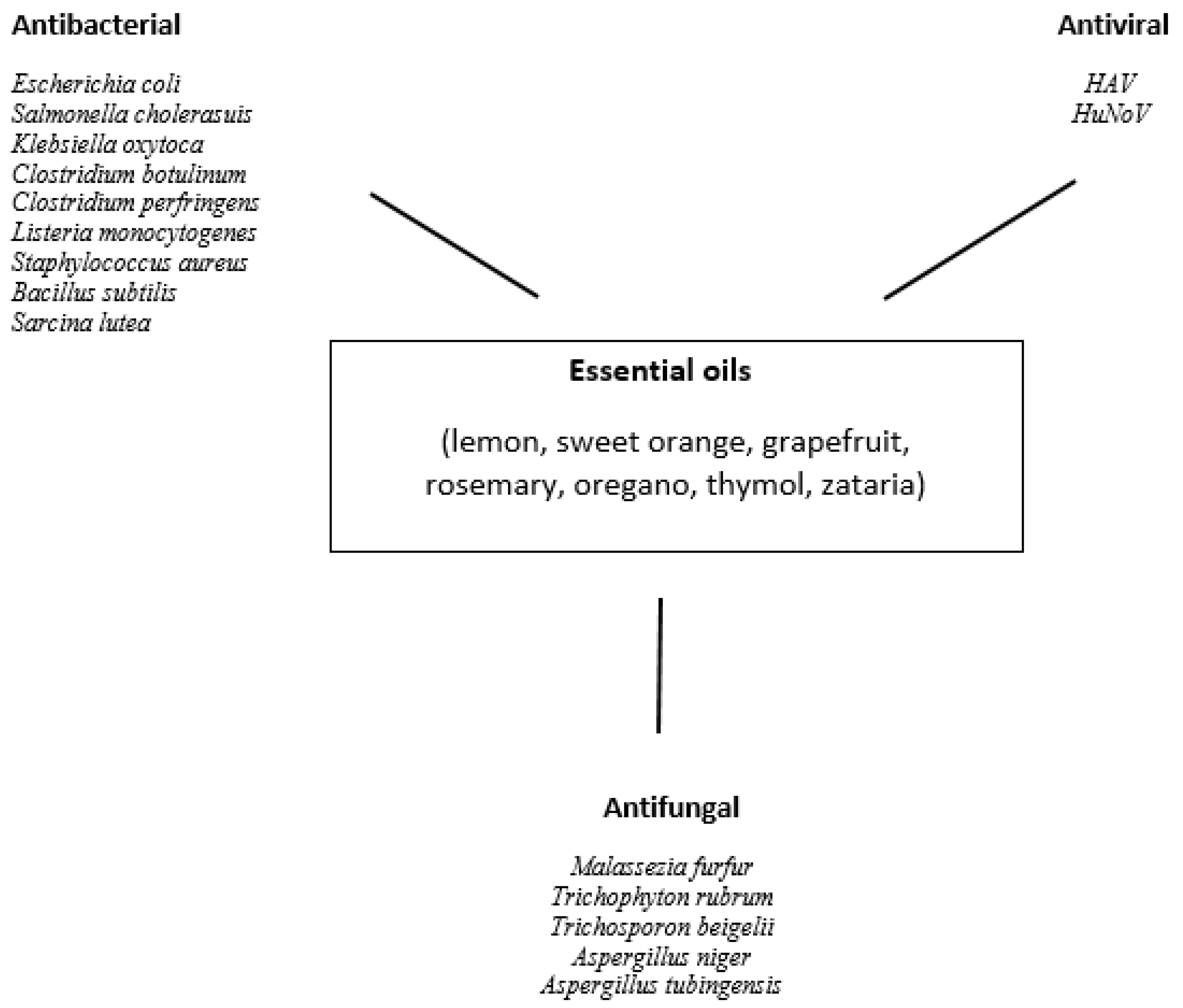
3. Composition and Biological Activity of Essential Oils
4. Review of Antiviral Use of Essential Oils, Juices, and Other Plant Extracts against HAV
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sánchez, G.; Bosch, A. Survival of Enteric Viruses in the Environment and Food. Vir. Food. 2016, 26, 367–392. [Google Scholar]
- Yeargin, T.; Gibson, K.E. Key characteristics of foods with an elevated risk for viral enteropathogen contamination. J. Appl. Microbiol. 2018, 26, 996–1010. [Google Scholar] [CrossRef] [Green Version]
- Koopmans, M.; Duizer, E. Foodborne viruses: An emerging problem. Int. J. Food Microbiol. 2004, 90, 23–41. [Google Scholar] [CrossRef]
- Marsh, Z.; Shah, M.P.; Wikswo, M.E.; Barclay, L.; Kisselburgh, H.; Kambhampati, A.; Cannon, J.L.; Parashar, U.D.; Vinjé, J.; Hall, A.J. Epidemiology of foodborne norovirus outbreaks–United States, 2009–2015. Food Saf. 2018, 6, 58–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deboosere, N.; Legeay, O.; Caudrelier, Y.; Lange, M. Modelling effect of physical and chemical parameters on heat inactivation kinetics of hepatitis A virus in a fruit model system. Int. J. Food Microbiol. 2004, 93, 73–85. [Google Scholar] [CrossRef]
- Hirneisen, K.A.; Black, E.P.; Cascarino, J.L.; Fino, V.R.; Hoover, D.G.; Kniel, K.E. Viral inactivation in foods: A review of traditional and novel food-processing technologies. Compr. Rev. Food Sci. Food Saf. 2010, 9, 3–20. [Google Scholar] [CrossRef] [PubMed]
- Aadil, R.H.; Roobab, U.; Mann, A.A. Effect of heat on food properties. In Encyclopedia of Food Chemistry; Melton, L., Shahidi, F., Varelis, P., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 70–75. [Google Scholar]
- Stumbo, C.R. Thermobacteriology in Food Processing; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Bartsch, C.; Plaza-Rodriguez, C.; Trojnar, E.; Filter, M.; Johne, R. Predictive models for thermal inactivation of human norovirus and surrogates in strawberry puree. Food Control. 2019, 96, 87–97. [Google Scholar] [CrossRef]
- Bozkurt, H.; D’Souza, D.H.; Davidson, P.M. Thermal inactivation of foodborne enteric viruses and their viral surrogates in foods. J. Food Prot. 2015, 78, 1597–1617. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Wu, X.; Sánchez, G.; Randazzo, W. Viability RT-qPCR to detect potentially infectious enteric viruses on heat-processed berries. Food Control. 2020, 107, 106818. [Google Scholar] [CrossRef]
- Shao, L.; Chen, H.; Hicks, D.; Wu, C. Thermal inactivation of human norovirus surrogates in oyster homogenate. Int. J. Food Microbiol. 2018, 281, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Deboosere, N.; Pinon, A.; Delobel, A.; Temmam, S.; Morin, T.; Merle, G.; Blaise-Boisseau, S.; Perelle, S.; Vialette, M. A predictive microbiology approach for thermal inactivation of Hepatitis A virus in acidified berries. Food Microbiol. 2010, 7, 962–967. [Google Scholar] [CrossRef] [PubMed]
- Bosch, A.; Gkogka, E.; Le Guyader, F.S.; Loisy-Hamon, F.; Lee, A.; van Lieshout, L.; Marthi, B.; Myrmel, M.; Sansom, A.; Schultz, A.C.; et al. Foodborne viruses: Detection, risk assesment, and control options in food processing. Int. J. Food Microbiol. 2018, 285, 110–128. [Google Scholar] [CrossRef] [PubMed]
- Koskiniemi, C.B.; Truong, V.D.; McFeeters, R.F.; Simunovic, J. Quality evaluation of packaged acidified vegetables subjected to continuous microwave pasteurization. LWT-Food Sci. Technol. 2013, 54, 157–164. [Google Scholar] [CrossRef]
- Zang, J.; Wang, D.; Zhao, G. Mechanism of discoloration in processed garlic and onion. Trends Food Sci. Technol. 2013, 30, 162–173. [Google Scholar] [CrossRef]
- Todd, E.C.D.; Greig, J.D. Viruses of foodborne origin: A review. Virus Adapt. Treat. 2015, 7, 25–45. [Google Scholar] [CrossRef] [Green Version]
- Carter, M.J. Enterically infecting viruses: Pathogenicity, transmission and significance for food and waterborne infection. J. Appl. Microbiol. 2005, 98, 1354–1380. [Google Scholar] [CrossRef] [PubMed]
- Kokkinos, P.; Kozyra, I.; Lazic, S.; Söderberg, K.; Vasickova, P.; Bouwknegt, M.; Rutjes, S.; Willems, K. Virological quality of irrigation water in leafy green vegetables and berry fruits production chains food. Environ. Virol. 2017, 9, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Matthews, K.R. Sources of enteric pathogen contamination of fruits and vegetables: Future directions of research. Stewart. Postharvest. Rev. 2013, 9, 1–5. [Google Scholar] [CrossRef]
- Doyle, M.P.; Erickson, M.C. Opportunities for mitigating pathogen contamination during on-farm food production. Int. J. Food Microbiol. 2012, 152, 54–74. [Google Scholar] [CrossRef] [PubMed]
- Beuchat, L.R. Vectors and conditions for preharvest contamination of fruits and vegetables with pathogens capable of causing enteric diseases. Br. Food J. 2006, 108, 38–53. [Google Scholar] [CrossRef]
- Matthews, K.R. Leafy vegetables. In The Produce Contamination Problem. Causes and Solutions; Matthews, K.R., Sapers, G.M., Gerba, C.P., Eds.; Elsevier: Waltham, MA, USA, 2014; pp. 187–205. [Google Scholar]
- Park, S.; Szonyi, B.; Gautam, R.; Nightingale, K.; Anciso, J.; Ivanek, R. Risk factors for microbial contamination in fruits and vegetables at the preharvest level: A systematic review. J. Food Prot. 2012, 75, 2055–2081. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Hepatitis Report 2017; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- European Centre for Disease Prevention and Control. Multi-Country Outbreak of Hepatitis A Virus Genotype IA Infections Affecting EU Countries in 2018—21 May 2018; European Centre for Disease Prevention and Control: Stockholm, Sweden, 2018. [Google Scholar]
- Debing, Y.; Neyts, J.; Thibaut, H.J. Molecular biology and inhibitors of hepatitis A virus. Med. Res. Rev. 2014, 34, 895–917. [Google Scholar] [CrossRef] [Green Version]
- Vaughan, G.; Xia, G.; Forbi, J.C.; Purdy, M.A.; Rossi, L.M.; Spradling, P.R.; Khudyakov, Y.E. Genetic relatedness among hepatitis A virus strains associated with food-borne outbreaks. PLoS ONE 2013, 8, e74546. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McKnight, K.L.; Lemon, S.M. Hepatitis A Virus Genome Organization and Replication Strategy. Cold Spring Harb. Perspect Med. 2018, 8, a033480. [Google Scholar] [CrossRef] [PubMed]
- Sattar, S.A.; Jason, T.; Bidawid, S.; Farber, J. Foodborne spread of hepatitis A: Recent studies on virus survival, transfer and inactivation. Can. J. Infect. Dis. 2000, 11, 159–163. [Google Scholar]
- Bozkurt, H.; Phan-Thien, K.Y.; van Ogtrop, F.; Bell, T.; McConchie, R. Outbreaks, occurrence, and control of norovirus and hepatitis a virus contamination in berries: A review. Crit. Rev. Food Sci. Nutr. 2021, 61, 116–138. [Google Scholar] [CrossRef] [PubMed]
- Nasheri, N.; Vester, A.; Petronella, N. Foodborne viral outbreaks associated with frozen produce. Epidemiol. Infect. 2019, 18, e291. [Google Scholar] [CrossRef] [PubMed]
- Trudel-Ferland, M.; Jubinville, E.; Jean, J. Persistence of Hepatitis A Virus RNA in water, on non-porous surfaces, and on blueberries. Front. Microbiol. 2021, 12, 618352. [Google Scholar] [CrossRef]
- Marosevic, D.; Belting, A.; Schönberger, K.; Carl, A.; Wenzel, J.J.; Brey, R. Hepatitis A outbreak in the general population due to a MSM-associated HAV genotype linked to a food handler, November 2017–February 2018, Germany. Food Environ. Virol. 2019, 11, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, G.; Goncalves Rossi, L.M.; Forbi, J.C.; de Paula, V.S.; Purdy, M.A.; Xia, G.; Khudyakov, Y.E. Hepatitis A virus: Host interactions, molecular epidemiology and evolution. Infect. Genet. Evol. 2014, 21, 227–243. [Google Scholar] [CrossRef]
- Department of Epidemiology and Surveillance of Infectious Diseases. Laboratory of Monitoring and Epidemiological Analysis. NIZP-PZH-GIS. Warszawa, Poland. 2020. Available online: http://wwwold.pzh.gov.pl/oldpage/epimeld/2019/index_mp.html (accessed on 6 May 2021).
- Polański, P.; Sadkowska-Todys, M. Hepatitis A in Poland in 2016. Epidemiol. Rev. 2018, 72, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Polański, P.; Radziszewski, F.; Księżak, E.; Wiktor, A.; Sadkowska-Todys, M. Foodborne infections and intoxications in Poland in 2017. Przegl. Epidemiol. 2019, 7, 451–462. [Google Scholar]
- Foster, M.A.; Hofmeister, M.G.; Kupronis, B.A.; Lin, Y.; Xia, G.L.; Yin, S.; Teshale, E. Increase in Hepatitis A Virus Infections—United States, 2013–2018. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 413–415. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Essoil Database. Available online: http://www.nipgr.ac.in/Essoildb/ (accessed on 6 May 2021).
- Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Vazquez-Olivo, G.; Heredia, J.B. Essential oils of oregano: Biological activity beyond their antimicrobial properties. Molecules. 2017, 22, 989. [Google Scholar] [CrossRef] [Green Version]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Antimicrobial properties of plant essential oils against human pathogens and their mode of action: An updated review. Evid. Based Complementary Altern. Med. 2016, 2016, 3012462. [Google Scholar] [CrossRef] [PubMed]
- Adam, K.; Sivropoulou, A.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antifungal activities of Origanum vulgare subsp. hirtum, Mentha spicata, Lavandula angustifolia, and Salvia fruticosa essential oils against human pathogenic fungi. J. Agric. Food Chem. 1998, 46, 1739–1745. [Google Scholar] [CrossRef]
- Cleff, M.B.; Madrid, I.; Meinerz, A.R.; Meireles, M.C.A.; Braga de Mello, J.R.; Rodrigues, M.R.; Escareño, J.J.H. Essential oils against Candida spp.: In vitro antifungal activity of Origanum vulgare. Afr. J. Microbiol. Res. 2013, 7, 2245–2250. [Google Scholar]
- Císarová, M.; Tančinová, D.; Medo, J. Antifungal activity of lemon, eucalyptus, thyme, oregano, sage and lavender essential oils against Aspergillus niger and Aspergillus tubingensis isolated from grapes. Potravinarstvo 2016, 10, 83–88. [Google Scholar]
- Lakhrissi, B.; Boukhraz, A.; Barrahi, M.; Hartiti, H.E.; Ouhssine, M. Antifungal activity of essential oil of oregano (Origanum vulgare), marjoram (Origanum majorana) and synergy of two essential oils against Candida albicans. Int. J. Res. Stud. Sci. Eng. 2016, 3, 14–17. [Google Scholar]
- Tuttolomondo, T.; La Bella, S.; Licata, M.; Virga, G.; Leto, C.; Saija, A.; Trombetta, D.; Tomaino, A.; Speciale, A.; Napoli, E.M.; et al. Biomolecular characterization of wild sicilian oregano: Phytochemical screening of essential oils and extracts, and evaluation of their antioxidant activities. Chem. Biodivers. 2013, 10, 411–433. [Google Scholar] [CrossRef] [PubMed]
- Martucci, J.F.; Gende, L.B.; Neira, L.M.; Ruseckaite, R.A. Oregano and lavender essential oils as antioxidant and antimicrobial additives of biogenic gelatin films. Ind. Crop. Prod. 2015, 71, 205–213. [Google Scholar] [CrossRef]
- Borugă, O.; Jianu, C.; Mişcă, C.; Goleţ, I.; Gruia, A.T.; Horhat, F.G. Thymus vulgaris essential oil: Chemical composition and antimicrobial activity. J. Med. Life 2014, 7, 56–60. [Google Scholar] [PubMed]
- Kryvtsova, M.V.; Salamon, I.; Koscova, J.; Bucko, D.; Spivak, M. Antimicrobial, antibiofilm and biochemichal properties of Thymus vulgaris essential oil against clinical isolates of opportunistic infections. Biosyst. Divers. 2019, 27, 270–275. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Niu, Y.; Luo, Y.; Ge, M.; Yang, T.; Yu, L.L.; Wang, Q. Fabrication, characterization, and antimicrobial activity of thymol-loaded zein nanoparticles stabilized by sodium caseinate–chitosan hydrochloride double layers. Food Chem. 2014, 142, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Mahboubi, M.; Heidarytabar, R.; Mahdizadeh, E. Antibacterial activity of Zataria multiflora essential oil and its main components against Pseudomonas aeruginosa. Herba Pol. 2017, 63, 18–24. [Google Scholar] [CrossRef] [Green Version]
- Mahboubi, M.; Bidgoli, F.G. Antistaphylococcal activity of Zataria multiflora essential oil and its synergy with vancomycin. Phytomedicine 2010, 17, 548–550. [Google Scholar] [CrossRef] [PubMed]
- Saei-Dehkordi, S.S.; Tajik, H.; Moradi, M.; Khalighi-Sigaroodi, F. Chemical composition of essential oils in Zataria multiflora Boiss. from different parts of Iran and their radical scavenging and antimicrobial activity. Food Chem. Toxicol. 2010, 48, 1562–1567. [Google Scholar] [CrossRef] [PubMed]
- Eftekhar, F.; Zamani, S.; Yusefzadi, M.; Hadian, J.; Ebrahimi, S.N. Antibacterial activity of Zataria multiflora Boiss essential oil against extended spectrum β lactamase produced by urinary isolates of Klebsiella pneumoniae. Jundishapur J. Microbiol. 2011, 4, S43–S49. [Google Scholar]
- Samadi, N.; Sharifan, A.; Emam-Djomeh, Z.; Salehi Sormaghi, M.H. Biopreservation of hamburgers by essential oil of Zataria multiflora. Nat. Prod. Res. 2012, 26, 665–668. [Google Scholar] [CrossRef]
- Kordsardouei, H.; Barzegar, M.; Sahari, M.A. Application of Zataria multiflora Boiss. and Cinnamon zeylanicum essential oils as two natural preservatives in cake. Avicenna J. Phytomed. 2013, 3, 238–247. [Google Scholar]
- Hsouna, A.B.; Halima, N.B.; Smaoui, S.; Hamdi, N. Citrus lemon essential oil: Chemical composition, antioxidant and antimicrobial activities with its preservative effect against Listeria monocytogenes inoculated in minced beef meat. Lipids Health Dis. 2017, 16, 146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Battistini, R.; Rossini, I.; Ercolini, C.; Goria, M.; Callipo, M.R.; Maurella, C.; Pavoni, E.; Serracca, L. Antiviral activity of essential oils against hepatitis A virus in soft fruits. Food Environ. Virol. 2019, 11, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Saeb, S.; Amin, M.; Gooybari, R.S.; Aghe, N. Evaluation of antibacterial activities of Citrus limon, Citrus reticulata, and Citrus grandis against pathogenic bacteria. Int. J. Enteric Pathog. 2016, 4, e37103. [Google Scholar] [CrossRef] [Green Version]
- Man, A.; Santacroce, L.; Jacob, R.; Mare, A.; Man, L. Antimicrobial activity of six essential oils against a group of human pathogens: A comparative study. Pathogens 2019, 8, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jing, L.; Lei, Z.; Li, L.; Xie, R.; Xi, W.; Guan, Y.; Sumner, L.W.; Zhou, Z. Antifungal activity of citrus essential oils: A review. J. Agric. Food Chem. 2014, 62, 3011–3033. [Google Scholar] [CrossRef]
- Ajayi-Moses, O.B.; Ogidi, C.O.; Akinyele, B.J. Bioactivity of Citrus essential oils (CEOs) against microorganisms associated with spoilage of some fruits. Chem. Biol. Technol. Agric. 2019, 6, 22. [Google Scholar] [CrossRef] [Green Version]
- Dosoky, N.S.; Setzer, W.N. Biological activities and safety of Citrus spp. essential oils. Int. J. Mol. Sci. 2018, 19, 1966. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.M.; Sheu, S.R.; Hsu, S.C.; Tsai, Y.H. Determination of bactericidal efficacy of essential oil extracted from orange peel on the food contact surfaces. Food Control 2010, 21, 1710–1715. [Google Scholar] [CrossRef]
- Bourgou, S.; Zohra, F.; Ourghemmi, I.; Saidani, M. Changes of peel essential oil composition of four Tunisian citrus during fruit maturation. Sci. World J. 2012, 528593. [Google Scholar] [CrossRef] [Green Version]
- Settani, L.; Palazzolo, E.; Guarrasi, V.; Aleo, A.; Mammina, C.; Moschetti, G.; Germaná, M. Inhibition of foodborne pathogen bacteria by essential oils extracted from citrus fruits cultivated in Sicily. Food Control 2012, 26, 326–330. [Google Scholar] [CrossRef]
- Sharma, N.; Tripathi, A. Effects of Citrus sinensis (L.) Osbeck epicarp essential oil on growth and morphogenesis of Aspergillus niger (L.) Van Tieghem. Microbiol. Res. 2008, 163, 337–344. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Ruiz-Navajas, Y.; Fernández-López, J.; Perez-Álvarez, J. Antifungal activity of lemon (Citrus limon L.), mandarin (Citrus reticulata L.), grapefruit (Citrus paradisi L.) and orange (Citrus sinensis L.) essential oils. Food Control 2008, 19, 1130–1138. [Google Scholar] [CrossRef]
- Singh, P.; Shukla, R.; Prakash, B.; Kumar, A.; Singh, S.; Mishra, P.K.; Dubey, N.K. Chemical profile, antifungal, antiaflatoxigenic and antioxidant activity of Citrus maxima Burm. and Citrus sinensis (L.) Osbeck essential oils and their cyclic monoterpene, dl-limonene. Food Chem. Toxicol. 2010, 48, 1734–1740. [Google Scholar] [CrossRef] [PubMed]
- Palazzolo, E.; Laudicina, V.A.; Germanà, M.A. Current and potential use of citrus essential oils. Curr. Org. Chem. 2013, 17, 3042–3049. [Google Scholar] [CrossRef]
- Wolffenbüttel, A.N.; Zamboni, A.; Becker, G.; dos Santos, M.K.; Borille, B.T.; de Cássia Mariotti, K.; Fagundes, A.C.; de Oliveira Salomón, J.L.; Coelho, V.R.; Ruiz, L.V. Citrus essential oils inhalation by mice: Behavioral testing, GCMS plasma analysis, corticosterone, and melatonin levels evaluation. Phytother. Res. 2018, 32, 160–169. [Google Scholar] [CrossRef]
- Vasek, O.M.; Cáceres, L.M.; Chamorro, E.R.; Velasco, G.A. Antibacterial activity of Citrus paradisi essential oil. J. Nat. Prod. 2015, 8, 16–26. [Google Scholar]
- Uysal, B.; Sozmen, F.; Aktas, O.; Oksal, B.S.; Kose, E.O. Essential oil composition and antibacterial activity of the grapefruit (Citrus paradisi L.) peel essential oils obtained by solvent-free microwave extraction: Comparison with hydrodistillation. Int. J. Food Sci. Technol. 2011, 46, 1455–1461. [Google Scholar] [CrossRef]
- Deng, W.; Liu, K.; Cao, S.; Sun, J.; Zhong, B.; Chun, J. Chemical composition, antimicrobial, antioxidant, and antiproliferative properties of grapefruit essential oil prepared by molecular distillation. Molecules 2020, 25, 217. [Google Scholar] [CrossRef] [Green Version]
- Denkova-Kostova, R.; Teneva, D.; Tomova, T.; Goranov, B.; Denkova, Z.; Shopska, V.; Slavchev, A.; Hristova-Ivanova, Y. Chemical composition, antioxidant and antimicrobial activity of essential oils from tangerine (Citrus reticulata L.), grapefruit (Citrus paradisi L.), lemon (Citrus lemon L.) and cinnamon (Cinnamomum zeylanicum Blume). Z. Naturforsch. 2021, 76, 175–185. [Google Scholar] [CrossRef]
- Luciardi, M.C.; Blázquez, M.A.; Alberto, M.R.; Cartagena, E.; Arena, M.E. Grapefruit essential oils inhibit quorum sensing of Pseudomonas aeruginosa. Food Sci. Technol. Int. 2019, 26, 231–241. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Lysakowska, M.; Pastuszka, M.; Bienias, W.; Kowalczyk, E. The potential of use Basil and Rosemary essential oils as effective antibacterial agents. Molecules 2013, 18, 9334–9351. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Hussain, A.I.; Anwar, F.; Chatha, S.A.S.; Jabbar, A.; Mahboob, S.; Nigam, P.S. Rosmarinus officinalis essential oil: Antiproliferative, antioxidant and antibacterial activities. Braz. J. Microbiol. 2010, 41, 1070–1078. [Google Scholar] [CrossRef] [Green Version]
- Baghloul, F.; Mansori, R.; Djahoudi, A. In vitro antifungal effect of Rosmarinus officinalis essential oil on Aspergillus niger. Natl. J. Physiol. Pharm. Pharmacol. 2017, 7, 285–289. [Google Scholar] [CrossRef]
- Da Silva Bomfim, N.; Kohiyama, C.Y.; Nakasugi, L.P.; Nerilo, S.B.; Mossini, S.A.G.; Romoli, J.C.Z.; Graton Mikcha, J.M.; Abreu Filho, B.A.; Machinski, M. Antifungal and antiaflatoxigenic activity of rosemary essential oil (Rosmarinus officinalis L.) against Aspergillus flavus. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2020, 37, 153–161. [Google Scholar] [CrossRef]
- Felšöciová, S.; Vukovic, N.; Jeżowski, P.; Kačániová, M. Antifungal activity of selected volatile essential oils against Penicillium sp. Open Life Sci. 2020, 15, 511–521. [Google Scholar] [CrossRef]
- Nieto, G.; Ros, G.; Castillo, J. Antioxidant and antimicrobial properties of rosemary (Rosmarinus officinalis L.): A review. Medicines 2018, 5, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jung, E.K. Chemical composition and antimicrobial activity of the essential oil of Chrysanthemum indicum against oral bacteria. J. Bacteriol. Virol. 2009, 39, 61–69. [Google Scholar] [CrossRef] [Green Version]
- Shunying, Z.; Yang, Y.; Huaidong, Y.; Yue, Y.; Guolin, Z. Chemical composition and antimicrobial activity of the essential oils of Chrysanthemum indicum. J. Ethnopharmacol. 2005, 96, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Shahrajabian, M.H.; Sun, W.; Zandi, P.; Cheng, Q. A review of chrysanthemum, the eastern queen in traditional chinese medicine with healing power in modern pharmaceutical sciences. Appl. Ecol. Environ. Res. 2019, 17, 13355–13369. [Google Scholar] [CrossRef]
- Hodaei, M.; Rahimmalek, M.; Arzani, A. Variation in morphological characters, chemical composition, and anthocyanin content of different Chrysanthemum morifolium cultivars from Iran. Biochem. System. Ecol. 2017, 74, 1–10. [Google Scholar] [CrossRef]
- Boukhebti, H.; Demirtas, I.; Omar, L.; Chaker, A.N. Chemical composition, antibacterial activity of essential oil and anatomical study of Chrysanthemum morifolium. J. Drug Deliv. Ther. 2020, 10, 7–13. [Google Scholar] [CrossRef]
- Zhang, K.; Jiang, Y.; Zhao, H.; Köllner, T.G.; Chen, S.; Chen, F.; Chen, F. Diverse terpenoids and their associated antifungal properties from roots of different cultivars of Chrysanthemum morifolium Ramat. Molecules 2020, 25, 2083. [Google Scholar] [CrossRef]
- Lin, L.T.; Hsu, W.C.; Lin, C.C. Antiviral natural products and herbal medicines. J. Tradit. Complement. Med. 2014, 4, 24–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Youssef, F.S.; Eid, S.Y.; Alshammari, E.; Ashour, M.L.; Wink, M.; El-Readi, M.Z. Chrysanthemum indicum and Chrysanthemum morifolium: Chemical composition of their essential oils and their potential use as natural preservatives with antimicrobial and antioxidant activities. Foods 2020, 9, 1460. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, G.; Aznar, R. Evaluation of natural compounds of plant origin for inactivation of enteric viruses. Food Environ. Virol. 2015, 7, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Raut, J.S.; Karuppayil, S.M. A Status Review on the Medicinal Properties of Essential Oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Behl, T.; Rocchetti, G.; Chadha, S.; Zengin, G.; Bungau, S.; Kumar, A.; Mehta, V.; Uddin, M.S.; Khullar, G.; Setia, D.; et al. Phytochemicals from Plant Foods as Potential Source of Antiviral Agents: An Overview. Pharmaceuticals 2021, 14, 381. [Google Scholar] [CrossRef]
- Randazzo, W.; Falcó-Ferrando, I.; Aznar, R.; Sánchez, G. Effect of green tea extract on enteric viruses and its application as natural sanitizer. Food Microbiol. 2017, 66, 150–156. [Google Scholar] [CrossRef] [PubMed]
- An, B.J.; Kwak, J.H.; Son, J.H.; Park, J.M.; Lee, J.Y.; Jo, C.; Byun, M.W. Biological and anti-microbial activity of irradiated green tea polyphenols. Food Chem. 2004, 88, 549–555. [Google Scholar] [CrossRef]
- Gadang, V.; Hettiarachchy, N.; Johnson, M.; Owens, C. Evaluation of antibacterial activity of whey protein isolate coating incorporated with nisin, grape seed extract, malic acid, and EDTA on a turkey frankfurter system. J. Food Sci. 2008, 73, 389–394. [Google Scholar] [CrossRef]
- Yilmaz, Y. Novel uses of catechins in foods. Trends Food Sci. 2006, 17, 64–71. [Google Scholar] [CrossRef]
- Falcó, I.; Díaz-Reolid, A.; Randazzo, W.; Sánchez, G. Green tea extract assisted low-temperature pasteurization to inactivate enteric viruses in juices. Int. J. Food Microbiol. 2020, 334, 108809. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.J.; McIntosh, J.; Pearce, P.; Camden, B.; Jordan, B.R. Anthocyanin and antioxidant capacity in Roselle (Hibiscus sabdariffa L.) extract. Food Res. Int. 2002, 35, 351–356. [Google Scholar] [CrossRef]
- Yang, Y.S.; Wang, C.J.; Huang, C.N.; Chen, M.L.; Chen, M.J.; Peng, C.H. Polyphenols of Hibiscus sabdariffa improved diabetic nephropathy via attenuating renal epithelial mesenchymal transition. J. Agric. Food Chem. 2013, 61, 7545–7551. [Google Scholar] [CrossRef] [PubMed]
- McKay, D.L.; Chen, C.Y.; Saltzman, E.; Blumberg, J.B. Hibiscus sabdariffa L. tea (tisane) lowers blood pressure in prehypertensive and mildly hypertensive adults. J. Nutr. 2010, 140, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.S.; Tsao, S.M.; Yin, M.C. In vitro antibacterial activity of roselle calyx and protocatechuic acid. Phytother. Res. 2005, 19, 942–945. [Google Scholar] [CrossRef]
- Joshi, S.S.; Dice, L.; D’Souza, D.H. Aqueous Extracts of Hibiscus sabdariffa Calyces Decrease Hepatitis A Virus and Human Norovirus Surrogate Titers. Food Environ. Virol. 2015, 7, 366–373. [Google Scholar] [CrossRef] [PubMed]
- El-Shiekh, R.A.; Abdelmohsen, U.R.; Ashour, H.M.; Ashour, R.M. Novel Antiviral and Antibacterial Activities of Hibiscus schizopetalus. Antibiotics 2020, 9, 756. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.S.; Howell, A.B.; D’Souza, D.H. Reduction of Enteric Viruses by Blueberry Juice and Blueberry Proanthocyanidins. Food Environ. Virol. 2016, 8, 235–243. [Google Scholar] [CrossRef]
- D’Souza, D.H. Phytocompounds for the control of human enteric viruses. Curr. Opin. Virol. 2014, 4, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasha, G.K.; Selvi, T.; Sakariah, K.K. Antibacterial and antioxidant activities of grape (Vitis vinifera) seed extracts. Food Res. Int. 2003, 36, 117–122. [Google Scholar] [CrossRef]
- Joshi, S.S.; Su, X.; D’Souza, D.H. Antiviral effects of grape seed extract against feline calicivirus, murine norovirus, and hepatitis A virus in model food systems and under gastric conditions. Food Microbiol. 2015, 52, 1–10. [Google Scholar] [CrossRef]
- Su, X.; D’Souza, D.H. Grape seed extract for control of human enteric viruses. Appl. Environ. Microbiol. 2011, 77, 3982–3987. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fabra, M.J.; Castro-Mayorga, J.L.; Randazzo, W.; Lagarón, J.M.; López-Rubio, A.; Aznar, R.; Sánchez, G. Efficacy of cinnamaldehyde against enteric viruses and its activity after incorporation into biodegradable multilayer systems of interest in food packaging. Food Environ. Virol. 2016, 8, 125–132. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.H.; Lee, B.H.; Jung, J.Y.; Cheon, D.S.; Kim, K.T.; Choi, C. Antiviral effect of korean red ginseng extract and ginsenosides on murine norovirus and feline calicivirus as surrogates for human norovirus. J. Ginseng Res. 2011, 35, 429–435. [Google Scholar] [CrossRef] [Green Version]
- Lee, M.H.; Lee, B.H.; Lee, S.; Choi, C. Reduction of hepatitis A virus on FRhK-4 cells treated with Korean red ginseng extract and ginsenosides. J. Food Sci. 2013, 78, 1412–1415. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Baert, L.; Uyttendaele, M. Inactivation of food-borne viruses using natural biochemical substances. Food Microbiol. 2013, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hamdy, A.H.; Mettwally, W.S.; El Fotouh, M.A.; Rodriguez, B.; El-Dewany, A.I.; El-Toumy, S.A.; Hussein, A.A. Bioactive phenolic compounds from the Egyptian Red Sea seagrass Thalassodendron ciliatum. Z. Nat. C 2012, 67, 291–296. [Google Scholar]
- Fiore, C.; Eisenhut, M.; Krausse, R.; Ragazzi, E.; Pellati, D.; Armanini, D.; Bielenberg, J. Antiviral effects of Glycyrrhiza species. Phytother. Res. 2008, 22, 141–148. [Google Scholar] [CrossRef]
| Name of Plant Source | Substance/ Essential Oil (EO)/Plant Extract | Reduction in HAV Titer | References |
|---|---|---|---|
| Citrus limon (lemon), Citrus sinensis (sweet orange), Citrus paradisi (grapefruit), Rosmarinus officinalis (rosemary cineole) | Lemon EO: limonene (71.18%), β-pinene (8.76%), and γ-terpinene (8.24%) Sweet orange EO: limonene 95.74%) Grapefruit EO (limonene 93.45%) Rosemary cineole EO: 1.8 cineole (51.79%), ɑ-pinene (16.54%), camphor (8.38%), and camphene (4.27%)) | 2.84 log TCID50/mL >2 log TCID50/mL 2.89 log TCID50/mL 2.94 log TCID50/mL | [59] |
| Chrysanthemum indicum, Chrysanthemum morifolium | EO from flower heads, major constituents: camphor, borneol, camphene, α-pinene, p-cymene and 1.8 cineole, | 2.21 log PFU/mL ID50 2.59 log PFU/mL ID50 | [92] |
| Origanum vulgare Thymus vulgaris Zataria multiflora | Oregano EO—carvacrol Thyme EO—thymol Zataria EO—carvacrol, thymol methyl ether | <0.5 log TCID50/mL 1.66 (1%); 2.45 (2%) log TCID50/mL <0.5 log TCID50/ml | [93] |
| Green tea | Green tea natural extract—soluble in water—content of epigallocatechin-3-gallate: 40–50% | 1 (pH 6.5), 1.15 (pH 7.2) log TCID50/mL | [96,100] |
| Hibiscus sabdariffa | Hibiscus sabdariffa extract: PCA (protocatechuic acid), FA (ferulic acid), MA (malic acid) | 1.29 ± 0.05–1.14 ± 0.01 (40mg/mL HE) log PFU/mL 1.37 ± 0.02–1.33 ± 0.01 (100mg/mL HE) log PFU/mL | [105] |
| Blueberry | Blueberry and blueberry extracts, blueberry juice (BJ) and blueberry proanthocyanidins (BB-PAC, B-type PAC structurally different from A-type PAC found in cranberries) | 1–2 log PFU/mL | [107] |
| Grape seed | Grape seed extract (GSE), Gravinol S, proanthocyanidins | 0.63 ± 0.2 log PFU/mL 1.86 log PFU/mL (0.25 mg/mL GSE), 2.26 log PFU/mL (0.5 mg/mL GSE); 2.89 log PFU/mL (1 mg/mL GSE) | [110,111,113] |
| Cinnamon | Cinnaaldehyde (CNMA)—3-Phenylprop-2-enal; ≥95% purity | 1 log10 TCID50/mL | [112] |
| Hibiscus schizopetalus | Dichloromethane Fraction (DCM-F), n-Butanol Fraction (Bu-F) | 1–2 log PFU/mL | [106] |
| Panax ginseng Meyer | Ginsenosides, saponin | 0.23 ± 0.48 to 0.57 ± 0.25 log 10 PFU/mL 0.45 ± 0.46 to 0.66 ± 0.52 log 10 PFU/mL | [114] |
| Thalassodendron ciliatum | Flavonoids: rutin, asebotin, 3-hydroxyasebotin, quercetin-3-O-β-D-xylopyranoside, and a racemic mixture of catechin | 100% (crude extract) | [116] |
| Glycyrrhiza | Glycyrrhizin | No data | [117] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jama-Kmiecik, A.; Sarowska, J.; Wojnicz, D.; Choroszy-Król, I.; Frej-Mądrzak, M. Natural Products and Their Potential Anti-HAV Activity. Pathogens 2021, 10, 1095. https://doi.org/10.3390/pathogens10091095
Jama-Kmiecik A, Sarowska J, Wojnicz D, Choroszy-Król I, Frej-Mądrzak M. Natural Products and Their Potential Anti-HAV Activity. Pathogens. 2021; 10(9):1095. https://doi.org/10.3390/pathogens10091095
Chicago/Turabian StyleJama-Kmiecik, Agnieszka, Jolanta Sarowska, Dorota Wojnicz, Irena Choroszy-Król, and Magdalena Frej-Mądrzak. 2021. "Natural Products and Their Potential Anti-HAV Activity" Pathogens 10, no. 9: 1095. https://doi.org/10.3390/pathogens10091095
APA StyleJama-Kmiecik, A., Sarowska, J., Wojnicz, D., Choroszy-Król, I., & Frej-Mądrzak, M. (2021). Natural Products and Their Potential Anti-HAV Activity. Pathogens, 10(9), 1095. https://doi.org/10.3390/pathogens10091095






