Abstract
Thermodynamic analysis and computations of the equilibrium solution for the multivariable secondary phase in steels have been conducted, based on the chemical equilibrium. Solid solution precipitation in the Ti–N–C microalloy steel for the secondary phase was analyzed, and its engineering application in the development of carburizing gear steels was investigated. The complete dissolution temperature increases with more C, N, or Ti in gear steels, and the effects of Ti and N are more significant than C. At a given temperature, the amount of [Ti] increases naturally with more Ti element content, while it decreases with more C or N elements content. It declines rapidly in the complete dissolution temperature range in steels until 1100 °C. The effective activity coefficient k1 increases with the temperature decreases, and increases with the higher content of Ti or C, while it decreases with the higher N content under the same temperature. Thermodynamic analysis shows that nitrogen has been precipitated largely as TiN micron-particles above 1400 °C in gear steels. Then, titanium precipitated mainly as TiC nano-particles, thus this secondary phase can hinder grain coarsening during heat treatment.
1. Introduction
Microalloying elements in steel form solid solutions with the iron matrix and contribute to the formation of carbonitrides, having different effects [1,2]. It is known that the niobium, vanadium, and titanium microalloying elements, in combination with C or N, can form multiple secondary phases and significantly improve the comprehensive performance of the high-strength low-alloy (HSLA) steels through retarding grain growth at high temperature and precipitation strengthening in the iron matrix [3,4]. Thus, it is possible to improve the strength and toughness of steels based on reasonable control of the secondary phase in the solid solution and precipitation in the iron matrix [5]. It is important to conduct a rigorous theoretical analysis of the multiple secondary phases in special steels to effectively control steel microstructure and properties [6,7,8,9].
Quantitative computations of the equilibrium solution of complex secondary phases is one of the challenging problems in this area [10,11,12]. In this paper, according to the thermodynamic analysis model of the equilibrium solution for the multivariable secondary phases in special steels [13,14] based on the mass balance and solubility product equations-solid solution precipitation of the secondary phase in the Ti–N–C microalloy steel was analyzed, along with analysis of the effects of different elements on the solid solution and precipitation temperature. Taking its engineering application in carburizing gear steels as an example, and combined with heat treatment, the effects of grain coarsening by secondary phase precipitates in 20CrMnTi high quality gear steel at different temperatures were studied, the microstructure of the secondary phase in steel was observed, providing technical support for the enterprise production process development, and optimizing the microalloy composition. In turn, scientific design of the components in multivariate microelements could be conducted based on the specific production process.
2. Thermodynamic Analysis
The microalloying elements in combination with C or N, can form multiple secondary phases; these multiple secondary phases are comprised of carbides and nitrides with similar crystal structure, exhibiting continuous or extended mutual solubility. Therefore, the resulting multivariable secondary phases can be described by the chemical formula (M1, M2) (C, N). Valid for small element concentrations, the microalloying elements M1 and M2, as well as the interstitial elements C and N form dilute solutions in the matrix and their activities obey the Henry’s law. In this case, the effective activity coefficients of the components M1C, M2C, M1N and M2N are assumed to be k1, k2, m1 and m2, respectively, and the total molar fraction of the M1(k1+m1)M2(k2+m2)C(k1+k2)N(m1+m2) carbonitride formed in the steel is t moles [14,15]. This carbonitride can be seen as a mixture of the following amounts of pure binary carbides and nitrides: k1t mole M1C, k2t mole M2C, m1t mole M1N and m2t mole M2N. Therefore, based on the chemical equilibrium, the thermodynamic analysis model and computing method of the equilibrium solution for the multivariable secondary phase in steels have been developed, according to the mass balance and solubility product equations for the quarternary secondary phase, or more. Therefore, the solid solution precipitation of the secondary phases formed in steel is Ti(Ck1Nm1) in the Ti–N–C microalloy steel system, and the coefficient of solid solubility of the product is taken as in the reference [14]. Therefore,
where M1, C and N are the mass percentages of Ti, C and N, respectively; Am1, AC and AN are the atomic weights of Ti, C, and N, respectively; and ATi = 47.9, AN = 14, AC = 12, [M1], [C] and [N] are the concentrations (in wt. %) of the respective elements dissolved in the solution; T is the temperature; and t is the total molar fraction of the carbonitride Ti(Ck1Nm1) formed in the steel; thus, these ternary secondary phases can be seen as a mixture of the following amounts of pure carbides and nitrides: k1t mole TiC, m1t mole TiN. Equations (1)–(6) have six unknowns, which are solved for numerically to determine the equilibrium state. For a given steel at any appropriate temperature, the equilibrium matrix composition, precipitate composition, and precipitate volume fraction can be determined (i.e., [M1], [C], [N], k1, m1 and t).
It is important to control the combined precipitate in the Ti-bearing gear steels, however, little information is currently available regarding gear steels. In this paper, the numerical iteration method was used for a series of Ti–N–C microalloyed steels with (0.05%–0.35%) C, (0.0025%–0.015%) N and (0.01%–0.08%) Ti composition. The equilibrium solution thermodynamic state, including concentrations of the respective elements in solution [C], [N] and [Ti] from 950 °C to complete dissolution temperature, k1 and m1 constants, as well as the total molar fraction of carbonitrides have been investigated. The complete dissolution temperatures for different Ti–N–C system microalloyed steels are shown in Table 1. The carbonitride complete dissolution temperature increases with higher C, N, or Ti levels in Ti–N–C system microalloyed steels. It should be noted that the effects of N and Ti additions on the complete dissolution temperature are expected to be greater compared with C. As is known for microalloyed gear steels, if the complete dissolution temperature is above the liquidus temperature, constitutional liquation of the carbonitrides would occur, so it is important to scientifically optimize the content of Ti, N and C elements in the actual production.

Table 1.
Complete dissolution temperature changes with C, N and Ti composition variation in the Ti–C–N system steels.
The thermodynamic analysis results of the Ti–C–N system microalloyed gear steels are shown in Figure 1, Figure 2 and Figure 3. At a given temperature, the dissolved Ti content increases naturally with the content of microalloyed element Ti increasing, and decreases obviously with the increase in the content of element N, while there seems to be no obvious change with the variation of element C content. The dissolved Ti content ([Ti]) decreases naturally as the temperature decreases, especially in the complete dissolution temperature range until 1100 °C, thus Ti is precipitated easily in the high temperature phase. The coefficient k1 increases as the temperature decreases, and at a given temperature, the coefficient k1 increases with the content of C or Ti increasing, while it decreases obviously with the content of N increasing. Therefore, in engineering application, for higher [Ti] dissolved in Ti-bearing microalloyed steels, the addition of N should be decreased during the microalloy composition design, which is significant to control the secondary phase precipitate at the required temperature.
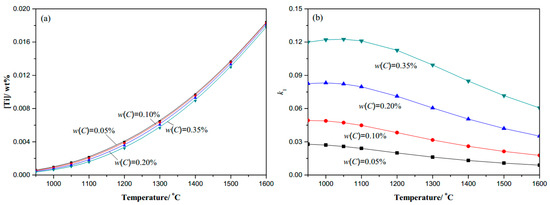
Figure 1.
C-0.0065N-0.02Timicroalloy steel: (a) Ti content changes with temperature; (b) k1 coefficient changes with temperature.
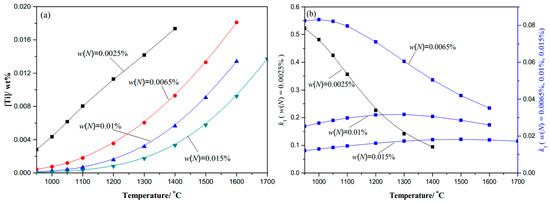
Figure 2.
0.20C-N-0.02Ti microalloy steel: (a) Ti content changes with temperature; (b) k1 coefficient changes with temperature.
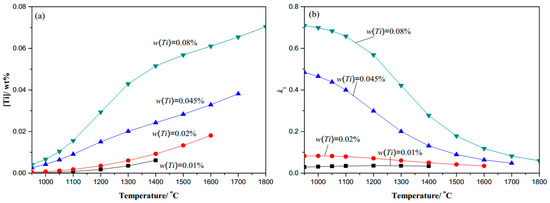
Figure 3.
0.20C-0.0065N-Ti microalloy steel: (a) Ti content changes with temperature; (b) k1 coefficient changes with temperature.
3. Engineering Applications
At present, the most widely used carburizing gear steel is 20CrMnTi. The main machining process is blanking → heating (950–1150 °C) → hot forging → heat preservation (880 °C for 8 h) → turning → gear milling → carburizing (950–1000 °C for 6 h) → quenching → low temperature tempering → finishing. The steel is required to have lower grain growth tendency at high temperature, which is ensured by keeping the fine matrix grain in the process of carburizing, thereby obtaining good toughness.
To optimize the composition with balanced comprehensive performance of Ti-bearing microalloyed steel, an engineering application of microalloyed gear steel 20CrMnTi was taken as an example here. The 20CrMnTi steel was produced by Fangda Special Steel Technology Co., Ltd (Nanchang, China); the production process of this steel was converter smelting → LF refining → VD vacuum processing → continuous casting → continuous rolling → testing processes. The dimensions of the rolling slab used were 160 mm × 160 mm × 9500 mm, and the rolling temperature was about 1050 °C. After rough rolling, middle rolling and finishing rolling, the steel was rolled into the required product specification (Φ30 mm); the finishing rolling temperature was controlled at about 950 °C. The investigated samples have the chemical composition shown in Table 2. The Ti–N–C system gear steels were heat treated at 850 °C, 900 °C, 950 °C and 1000 °C for 1 h in the intermediate frequency furnace, respectively, then air-cooled to ambient temperature, and the grain coarsening was studied under different temperatures. Metallographic specimens were prepared using conventional grinding, the polishing method and etched with 3% nitric acid alcohol; microstructures were characterized by 9XB-PC light microscopy (LM) (Shanghai optical instrument factory, Shanghai, China), Zeiss Auriga scanning electron microscope (SEM) (Zeiss, Dresden, Germany) and JEOL JEM-2100 transmission electron microscopy (TEM) (JEOL, Tokyo, Japan). The TEM samples were prepared by cutting 3 mm disks from the hot-rolled bars. The disks were mechanically thinned to a thickness of 100 μm and then the foils were thinned further for electron transparent area using a focused ion-beam, and the hardness was tested by a HB-3000 Brinell hardness tester, etc.

Table 2.
Chemical composition of the 20CrMnTi gear steel.
Based on the developed thermodynamic model, the change of the solid solution content of the microalloying elements and the relative content of binary precipitates with temperature can be obtained, as shown in Figure 4. Verified by the thermodynamic model, the amount of precipitates depends on Ti and N content as the temperature is reduced. The actual chemical composition of the carbonitrides as a function of temperature, expressed by the values of k1 and m1, strongly depends on the steel composition. The real content change of the TiC and TiN components with the temperature can be approximated, based on Figure 4b. The thermodynamic analysis shows that the dissolved Ti content is 0.03915%, the dissolved C content is 0.21843%, while the dissolved N content is only 0.00067% in the gear steel, and the coefficient k1 is 0.23238, and the coefficient m1 is 0.76762 at 1400 °C, thus nitrogen has been precipitated primarily in the TiN micro-particles above 1400 °C. The complete dissolution temperature is calculated to be 1915.02 °C by the model, which is 400 °C higher than the corresponding liquidus temperature of 1514.89 °C [16]. It is obvious that constitutional liquation of the strong carbonitrides would occur in this sample.
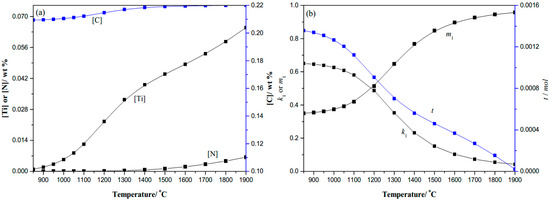
Figure 4.
The 0.22%C-0.0067%N-0.066%Ti gear steel: (a) Solid solution contents change with temperature; (b) k1, m1 and t change with temperature.
The carburizing gear steel 20CrMnTi microstructure is shown in Figure 5, where yellow secondary phase in the form of water chestnuts is visible in the etched and un-etched images. The corresponding SEM image is shown in Figure 6a, along with the energy dispersive spectroscopy (EDS) results presented in Figure 6b. The spectral components of the secondary phase are listed in Table 3. The secondary phase precipitation is mainly TiN; this result once again proved that constitutional liquation of the strong carbonitrides has occurred in the sample, which is consistent with the thermodynamic analysis results. In order to accurately control the size and shape of the secondary phase in steel, the content of each element must be scientifically selected, especially Ti, N and C to ensure that the multivariable secondary phase would precipitate at the required temperature.
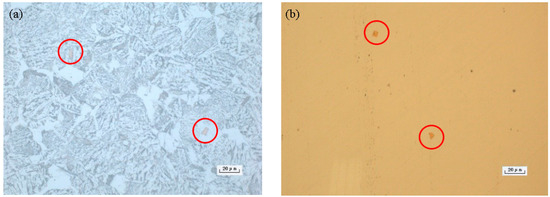
Figure 5.
The 20CrMnTi steel microstructure: (a) Optical image after etching; (b) Optical image without etching.
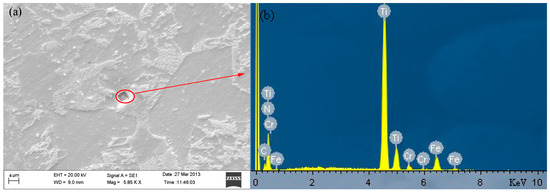
Figure 6.
(a) scanning electron microscope (SEM) image of the secondary phase in 20CrMnTi; (b) Energy dispersive spectroscopy (EDS) results.

Table 3.
Spectral components of the secondary phase in 20CrMnTi.
According to the processing schemes, the gear steels were heat treated at 850 °C, 900 °C, 950 °C and 1000 °C, respectively, and the grain coarsening was studied under these treatment temperatures. The microstructure and hardness of the 20CrMnTi steel after heat treatment are shown in Table 4 and Figure 7. The main phases are ferrite and pearlite with the grain size of No. 11.5–12.0 grade at 850 °C and 900 °C. However, when the temperature is 950 °C, the phases are ferrite, pearlite and some bainite, and the material hardness increases. The grain size is further increased to the No. 5.0 grade when the temperature is 1000 °C, and the main phases are ferrite, bainite and a small amount of pearlite with greatly increased hardness. It is known that when the grains begin to coarsen sharply in steel, which increases the stability of austenite significantly, it is advantageous to form the bainite, and the material hardness increases greatly.

Table 4.
Microstructure and hardness of the 20CrMnTi gear steel after heat treatment.

Figure 7.
Microstructure of 20CrMnTi under different heat treatment temperatures: (a) 850 °C; (b) 900 °C; (c) 950 °C.
Ti is added frequently in many steels to control the austenitic grain size in the 1200–1250 °C temperature range [16]. However, in this paper, when the temperature is 950 °C, grain coarsening began in the gear steel, so the secondary phase is not effective to control the matrix grain size at this temperature. This is consistent with the thermodynamic analysis results, because nitrogen has been precipitated largely as TiN micro-particles above 1400 °C. However, the larger particles have no effect on precipitation strengthening and hinder grain growth, and in contrast, will seriously affect the fatigue life of gear steel. Then, titanium precipitated mainly as TiC nano-particles, as shown in Figure 8a, along with the energy dispersive spectroscopy (EDS) results presented in Figure 8b, so the secondary phase hinders the grain coarsening mainly due to the TiC during heat treatment. In the practical production process, one should reduce the content of Ti element and control nitrogen. It makes sense to select the content of each element, and then ensure that the multivariable secondary phase precipitated in the required temperature range to accurately control the size and shape of the secondary phase and to improve the comprehensive performance of the product.
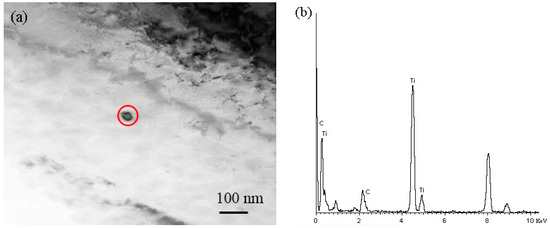
Figure 8.
Secondary phase in 20CrMnTi gear steel: (a) transmission electron microscopy (TEM) image; (b) energy dispersive spectroscopy (EDS) results.
4. Conclusions
According to the thermodynamic analysis model and computations, the composition and relative amounts of equilibrium carbonitrides, as functions of steel composition and temperature, are calculated. Taking the Ti–C–N alloy system as an example, the following general trends are predicted and verified:
- (1)
- Thermodynamic calculation results show that the complete dissolution temperature increases with higher levels of either C, N, or Ti, and the effects of Ti and N are more significant compared with C. At a given temperature, the amount of dissolved Ti increases naturally as the content of Ti microalloyed element increases, while it decreases obviously with more N. It will decline rapidly in the complete dissolution temperature range, until 1100 °C in steels. The coefficient k1 increases as the temperature decreases, and increases with the higher content of Ti or C, but decreases with the higher N content under the same temperature.
- (2)
- For the 20CrMnTi gear steels produced by Fangda Special Steel Technology Co., Ltd., the heat treatment shows that when the temperature is above 950 °C, the grains begin to coarsen in this steel. When the temperature is 1000 °C, the grains coarsen sharply, the grain size is No. 5.0 grade, and the phases are ferrite and bainite, containing a small amount of pearlite.
- (3)
- Thermodynamic analysis shows that the nitrogen has been precipitated largely as TiN micron-particles above 1400 °C in the gear steels, and then the titanium is precipitated mainly as TiC nano-particles, so this secondary phase hinders grain growth during heat treatment, consistent with experimental results.
Acknowledgments
This research was supported by the China Postdoctoral Science Foundation (2016M591072), Science and Technology Support Project of the Jiangxi Province (20112BBE50006) and the Outstanding Youth Fund Projects of Jiangxi Province (20133BCB23032).
Author Contributions
Yanlin Wang, Meng zhou and Xiaolu Pang are the main contributor of this research work. They mainly performed the experimental work, results analysis and made draft the research paper; Mingwen Chen, Kewei Gao and Alex A. Volinsky analyzed the data and proofread the paper.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Kimura, Y.; Inoue, T.; Yin, F.; Tsuzaki, K. Inverse temperature dependence of toughness in an ultrafine grain-structure steel. Science 2008, 320, 1057–1060. [Google Scholar] [CrossRef] [PubMed]
- Adrian, H. Thermodynamic model for precipitation of carbonitrides in high strength low alloy steels containing up to three microalloying elements with or without additions of aluminium. Mater. Sci. Technol. 1992, 8, 406–420. [Google Scholar] [CrossRef]
- Gao, N.; Baker, T. Influence of AlN precipitation on thermodynamic parameters in Al-VN microalloyed steels. ISIJ Int. 1997, 37, 596–604. [Google Scholar] [CrossRef]
- Yuan, S.; Zhang, X.; Hao, B. Precipitation behaviour aged in α phase after relaxation for different times in niobium bearing microalloyed steel. Mater. Res. Innov. 2013, 17, 26–28. [Google Scholar] [CrossRef]
- Uhm, S.; Moon, J.; Lee, C.; Yoon, J.; Lee, B. Prediction model for the austenite grain size in the coarse grained heat affected zone of Fe-C-Mn steels: Considering the effect of initial grain size on isothermal growth behavior. ISIJ Int. 2004, 44, 1230–1237. [Google Scholar] [CrossRef]
- Fu, J.; Li, G.; Yu, Y.; Mao, X.; Fang, K. Comprehensive strengthening mechanism of steel based on nano-scale cementite precipitates. Eng. Sci. 2011, 13, 31–42. [Google Scholar]
- Medina, S.F. Determination of precipitation–time–temperature (PTT) diagrams for Nb, Ti or V micro-alloyed steels. J. Mater. Sci. 1997, 32, 1487–1492. [Google Scholar] [CrossRef]
- Akamatsu, S.; Hasebe, M.; Senuma, T.; Matsumura, Y.; Kisue, O. Thermodynamic Calculation of Solute Carbon and Nitrogen in Nb and Ti Added Extra-low Carbon Steels. ISIJ Int. 1994, 34, 9–16. [Google Scholar] [CrossRef]
- Hong, S.; Jun, H.; Kang, K.; Park, C. Evolution of precipitates in the Nb–Ti–V microalloyed HSLA steels during reheating. Scr. Mater. 2003, 48, 1201–1206. [Google Scholar] [CrossRef]
- Suzuki, K.-I.; Miyagawa, S.; Saito, Y.; Shiotani, K. Effect of microalloyed nitride forming elements on precipitation of carbonitride and high temperature ductility of continuously cast low carbon Nb containing steel slab. ISIJ Int. 1995, 35, 34–41. [Google Scholar] [CrossRef]
- Wang, C.; Li, J.; Zhao, H.; Chen, Y. Influence factors on solid-solution of carbonitride of niobium in steel. J. Univ. Sci. Technol. B 2009, 31, 194–198. [Google Scholar]
- Xiang, S.; Liu, G.; Li, C.; Wang, A.; Han, Q. Thermodynamic model for carbonitride precipitation in low carbon steels. J. Univ. Sci. Technol. B 2006, 28, 818–822. [Google Scholar]
- Wang, Y.; Zhu, J. Numerical Analysis Model of Equilibrium Solution for the Multivariate Second-Phase in Steels. Patent CN201310,300,994.4, 18 July 2013. [Google Scholar]
- Wang, Y.; Zhuo, L.; Chen, M.; Wang, Z. Thermodynamic model for precipitation of carbonitrides in microalloyed steels and its application in Ti–V–C–N system. Rare Met. 2016, 35, 735–741. [Google Scholar] [CrossRef]
- Wang, Y.; Zhuo, L.; Chen, M.; Wang, Z. Precipitation behaviour of carbonitrides in Ti-Nb-C-N microalloyed steels and an engineering application with homogenously precipitated nano-particles. Mater. Sci. 2015, 21, 527–531. [Google Scholar] [CrossRef]
- Gan, Y. Practical Manual of Modern Continuous Casting Steel; Metallurgical Industry Press: Beijing, China, 2010. [Google Scholar]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).