Abstract
The integration of sustainable and natural waste-derived materials into lightweight metals presents a promising strategy with both environmental and performance-related benefits. In this study, a biobased magnesium composite reinforced with dried leaf powder (DLP) derived from fallen waste leaves was synthesized using a controlled powder metallurgy method incorporating energy efficient hybrid microwave sintering, followed by hot extrusion at varying temperatures (350 °C, 250 °C, 150 °C). Microstructural analysis revealed that the addition of DLP had minimal effect on the overall grain morphology, while lower extrusion temperatures promoted finer grains due to restricted grain growth. Mg–5DLP composites consistently exhibited higher porosity than pure Mg, primarily due to the evaporation of organic constituents during sintering. The damping performance of the biomass-containing materials was improved (54.5% increase), particularly at lower extrusion temperatures (250 °C), though mechanical performance showed a trade-off with reduced hardness and compressive strength. A slight increase in yield strength at lower extrusion temperatures was attributed to retained dislocation density and grain refinement. Thermal stability remained largely unaffected, while corrosion behavior was strongly dependent on both DLP addition and extrusion temperature, with Mg–5DLP samples corroding faster than pure Mg when extruded at higher temperatures; interestingly, however, at the lowest extrusion temperature (150 °C), improved corrosion resistance to pure Mg (1.3 mm/year for Mg-5DLP vs. 2.0 mm/year for pure Mg) was observed. Overall, this work demonstrates that extrusion temperature is a critical factor in controlling the microstructure, thermal response, damping response, mechanical behavior and corrosion of biobased composites. The study not only highlights the potential of using direct biomass reinforcement of magnesium to synthesize lightweight, ecofriendly materials, but also lays a strong foundation for future investigations into biobased composite design, processing optimization, and property tailoring.
1. Introduction
Magnesium (Mg) makes up ~2.7% of the Earth’s crust by weight, making it the sixth most abundant element. Mg with a density of 1.74 g/cm−3 is ~35% and ~77% lighter than aluminum and steel, respectively [1]. It has gained widespread interest for both industrial and biomedical applications due to its high specific strength, good machinability, excellent damping capacity, and natural abundance. Its elastic modulus (~45 GPa) is closer to that of natural bone compared to conventional metallic biomaterials such as titanium or stainless steel, making Mg a promising candidate for orthopedic and biodegradable implant applications [2]. In the past decade, the potential for Mg as a biomaterial has inspired a lot of research, with a noticeable increase in the number of scientific publications investigating novel magnesium alloys and composites containing biocompatible reinforcements [3]. Mg exhibits superior biocompatibility, shows no indications of local or systemic toxicity [2,4,5], and is the second most abundant intracellular cation, being essential to the body’s metabolism [6,7,8]. Beyond its biomedical uses, Mg is seeing increasing use in the defense, electronics, sports and transportation sectors (aerospace, automotive, maritime, space and trains) where reducing weight without compromising performance is crucial [9].
Along with the growing emphasis on lightweight materials for improved energy efficiency, the recycling and re-use of waste resources such as biomass has also attracted significant attention. Converted biomass materials, such as leaf powder, have demonstrated promising potential in diverse applications, including ceramics, catalysts, supercapacitors, and even microwave-absorbing materials [10,11,12,13,14,15]. Such developments highlight the expanding scope of recycling, re-use, and upcycling approaches, which play a vital role in enhancing the sustainability and environmental compatibility of materials produced from waste [16]. El-Sayed and Mostafa conducted pyrolysis and ultimate analysis of three different types of mango leaves and reported that the carbon content comprises approximately 40.01–41.59% of the leaf’s dried weight [17]. Zang and Willison performed impedance measurements on two leaf species, showing that plant leaves possess very high electrical resistance and impedance, confirming their insulating nature [18]. Despite this, their fibrous organic structure and inherent carbon content make them promising starting materials for developing bio-derived composites. Wang conducted nanoindentation studies involving eight plant species and reported hardness values ranging from 0.01 GPa to 0.38 GPa (1.02 HV to 38.75 HV), with higher hardness observed in leaves rich in cellulose and lignin, such as Phyllostachys pubescens [19].
Our literature search did not indicate that development of metal-based composites containing biomass with naturally locked carbon has taken place. Metal-based composites traditionally utilize ceramics (e.g., oxides, carbides), which are durable and thermally stable, as reinforcement [20,21,22,23,24,25]. On the other hand, using biomass as an organic material possesses significant challenges in the context of metallurgy: it is not thermally stable at the temperatures that are typical during metals processing. Ergo, this work attempts to integrate waste fallen leaves (zero cost and material circularity) into pure magnesium using an energy-efficient processing approach, with a particular focus on the effect of extrusion temperature) on the microstructure, thermal, damping, mechanical and electrochemical responses of pure magnesium.
Accordingly, the present work is intended as an exploratory study to establish fundamental processing–microstructure–property relationships in a biobased magnesium composite; it will do this by leveraging powder metallurgy (PM), which enables the incorporation of low-density, non-metallic reinforcements while minimizing oxidation, excessive interfacial reactions, and the thermal degradation of temperature-sensitive reinforcements [1].
2. Materials and Methods
2.1. Materials and Synthesis
Fallen waste mango leaves (Mangifera indica) were collected, followed by microwave drying for 8 min in a 900 W microwave (Sharp R898C(S), Sharp Corporation, Osaka, Japan). These were then ball-milled using an Inversina 2 L tumbler mixer (Bioengineering AG, Wald, Switzerland) with 15 mm diameter stainless-steel balls (20:1 ball mill ratio) at a mixing speed of 65 rpm for a duration of 1 h. The resulting powder was separated from the coarse pieces of the midrib that were not well milled. Subsequently, it was dried in a Heraeus T6 drying oven at 90 °C for 1 h, producing dried leaf powder (DLP).
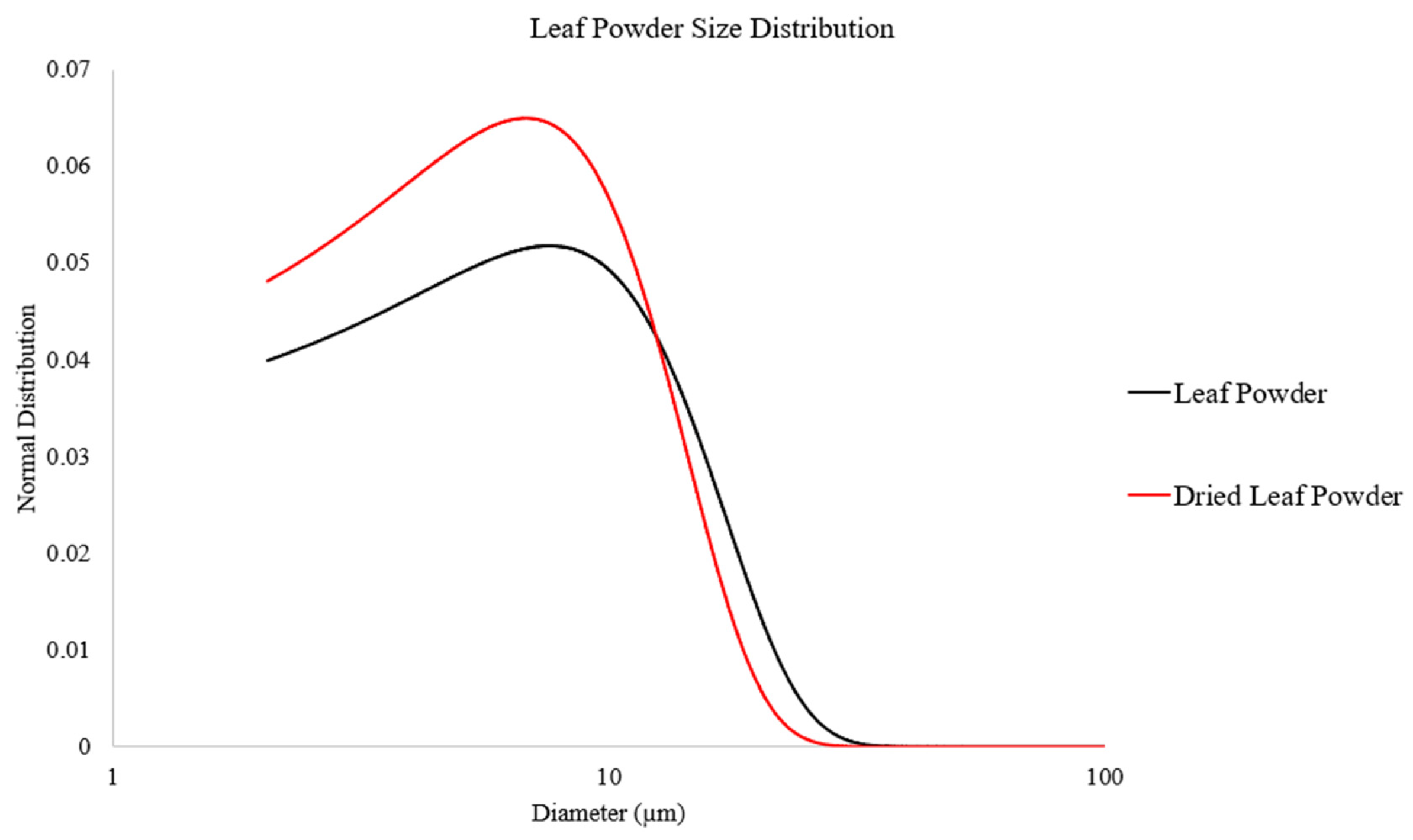
The DLP was then mixed with Mg powder (60–300 µm, Merck Group, Darmstadt, Germany) with proportions of 95 wt.% Mg and 5 wt.% DLP in a sealed vessel using an Inversina 2 L tumbler mixer at a speed of 50 rpm for 1 h to produce a mixture of Mg-5DLP using powder metallurgy. Figure 1 shows micrographs of the powder materials used in this work, with Figure 2 and Table 1 outlining the powder size of the leaf powder used, showing that drying is an effective method of size reduction.

Figure 1.
Micrographs of powder materials used in this work: (a) Mg, (b) DLP after ball milling, and (c) Mg-5DLP after blending.

Figure 2.
Powder size distribution of leaf powder used as biomass reinforcement in this study.
The blended powder was compacted into billets using a hydraulic press under a pressure of 600 psi with a holding time of 2 min, producing cylindrical billets of 35 mm diameter (Figure 3). The billets were then microwave-sintered to a temperature of 150 °C. While this is significantly lower than the conventional sintering temperatures typically employed for Mg-based materials (which are typically above 400 °C) [20], this temperature was intentionally selected to minimize pyrolysis and the degradation of the biomass reinforcement while retaining sufficient structural integrity for these biobased composites. Following sintering, hot extrusion was performed with an 8 mm diameter extrusion die after prior homogenization for 1 h. The resulting cylindrical rods were then machined and processed into samples for further characterization. Table 2 shows the extrusion and homogenization parameters employed in this work.

Figure 3.
Compacted pure Mg and biobased composite billets.

Table 1.
Further information on biomass used in this study.
Table 1.
Further information on biomass used in this study.
| Material | Average Circular Equivalent Diameter (µm) |
|---|---|
| Dried Mango Leaves | - |
| Leaf Powder | 7.61 ± 7.70 |
| Dried Leaf Powder | 5.02 ± 10.03 |

Table 2.
Extrusion and homogenization parameters used for biobased composites in this study.
Table 2.
Extrusion and homogenization parameters used for biobased composites in this study.
| Processing Condition | Extrusion Temperature (°C) | Homogenization Temperature (°C) |
|---|---|---|
| E350 | 350 | 400 |
| E250 | 250 | 300 |
| E150 | 150 | 200 |
For comparison, pure Mg was also synthesized in the same conditions (omitting the mixing/blending step). Furthermore, E350 materials were used as reference materials for subsequent comparisons in this study where applicable.
2.2. Characterization
2.2.1. Microstructure
Leaf powder particle size was characterized using a Morphologi 4 analyzer (Malvern Panalytical Ltd., Worcestershire, UK) with a sample size of 12,000 particles.
Flat and parallel samples were fine-finished using a 0.05 micron size alumina suspension and deionized water. Microstructural analysis was carried out using a Hitachi S-4300 Field Emission Scanning Electron Microscope (FESEM, Hitachi Ltd., Tokyo, Japan) and JEOL IT200 SEM (JEOL Ltd., Tokyo, Japan), equipped with Energy-Dispersive X-ray Spectroscopy (EDS).
Grain size characterization was conducted by etching polished sample surfaces with a solution of 5% citric acid in H2O for the durations listed in Table 3.

Table 3.
Etch durations of the materials explored in this work.
Grain morphology was captured using a Leica DM2500 optical microscope (Leica Microsystems (SEA) Pte Ltd., Singapore). Image processing and grain size quantification were performed with MATLAB software (version R2013b, Natick, MA, USA), following the procedures outlined in ASTM E112-13 (2021) [26].
2.2.2. X-Ray Diffraction
X-ray diffraction (XRD) analysis was performed using a Shimadzu XRD-6000 diffractometer (Shimadzu Corporation, Kyoto, Japan) equipped with Cu-Kα radiation (λ = 1.54056 Å). The scans were recorded in the 2θ range of 10–80° at a scanning speed of 2° per minute, where θ is the Bragg angle.
2.2.3. Density and Porosity
The experimental density was determined using the Archimedes principle with an AD-1653 density determination kit (AND Company, Ltd., Tokyo, Japan) attached to the GH-252 balance. A minimum of 6 representative samples were tested to ensure reproducibility.
Porosity analysis was carried out to examine the pore distribution on the sample’s surfaces. A Hitachi S-4300 FESEM was used to obtain sample images. Quantitative porosity values were obtained by examining representative polished surface images with ImageJ (version 1.54p, National Institutes of Health, Bethesda, MD, USA), and MATLAB (version R2013b) software, where the area fraction of pores was calculated to estimate the true porosity of the materials.
2.2.4. Damping Analysis
Rods measuring 50 mm in length were tested using the impulse excitation technique, coupled with the Response Frequency Damping Analyzer (RFDA, version 8.1.2) software (IMCE, Genk, Belgium). The recorded vibration responses were subsequently analyzed to determine the material’s damping characteristics, including its attenuation coefficient, damping capacity, and elastic modulus.
2.2.5. Mechanical Properties
Microhardness characterization was carried out using an Anton Paar Micro Indentation Tester (MHT, Anton Paar GmbH, Graz, Austria) equipped with a diamond indenter. The tests were performed at a maximum load of 250 mN with loading and unloading times of 10 s each and a 2 s holding period in accordance with standard ISO 14577 [27]. A minimum of 10 representative measurements were recorded across the sample surface of each material.
Compression tests were carried out using an MTS-810 servo-hydraulic testing system on specimens with an L/D ratio of 1, loaded until failure. The tests utilized a quasi-static loading condition as per ASTM E9-09 standards [28], with a strain rate of 0.0083%/s.
2.2.6. Thermal Properties
The ignition behavior of the samples (approximately 2 mm × 2 mm × 2 mm) was analyzed using a Shimadzu DTG-60H Thermogravimetric Analyzer (Shimadzu Corporation, Kyoto, Japan). Tests were conducted in purified air (flow rate: 50 mL/min) while heating from 30 °C to 1400 °C at a rate of 10 °C/min. The thermal response was further assessed using a Shimadzu DSC-60 (Shimadzu Corporation, Kyoto, Japan) under an argon atmosphere (flow rate: 25 mL/min), within a temperature range of 30 °C to 600 °C and a heating rate of 5 °C/min.
The coefficient of thermal expansion (CTE) was determined using a TMA PT1000 analyzer on specimens of approximately 6 mm height (Linseis Thermal Analysis, Selb, Germany). Measurements were performed between 50 °C and 400 °C at a heating rate of 5 °C/min under an argon gas flow of 100 mL/min.
2.2.7. Corrosion Response
Disks with a thickness of 1.5 mm were sectioned, polished, and immersed in Phosphate-Buffered Saline (PBS, Thermo Fisher Scientific Inc., Waltham, MA, USA) maintained at 37 °C. The samples were periodically cleaned every 24 h to remove corrosion products using a cleaning solution containing 1.9 g of AgNO3 and 20 g of CrO3 dissolved in 100 mL of deionized water, followed by rinsing with deionized water [29]. The specimens were then dried and weighed to determine the amount of weight lost. This was done until 28 days had elapsed or the sample disintegrated, whichever came earlier. Subsequently, the corrosion rate was calculated using the following equation [29]:
where ΔW—weight loss in mg; D—experimental density of the material in g/cm3; A—disk surface area in cm2; and T—immersion duration in hours.
Corrosion Rate (mm/year) = 87.6 × ΔW/(DAT)
3. Results and Discussion
3.1. Microstructure and Morphology
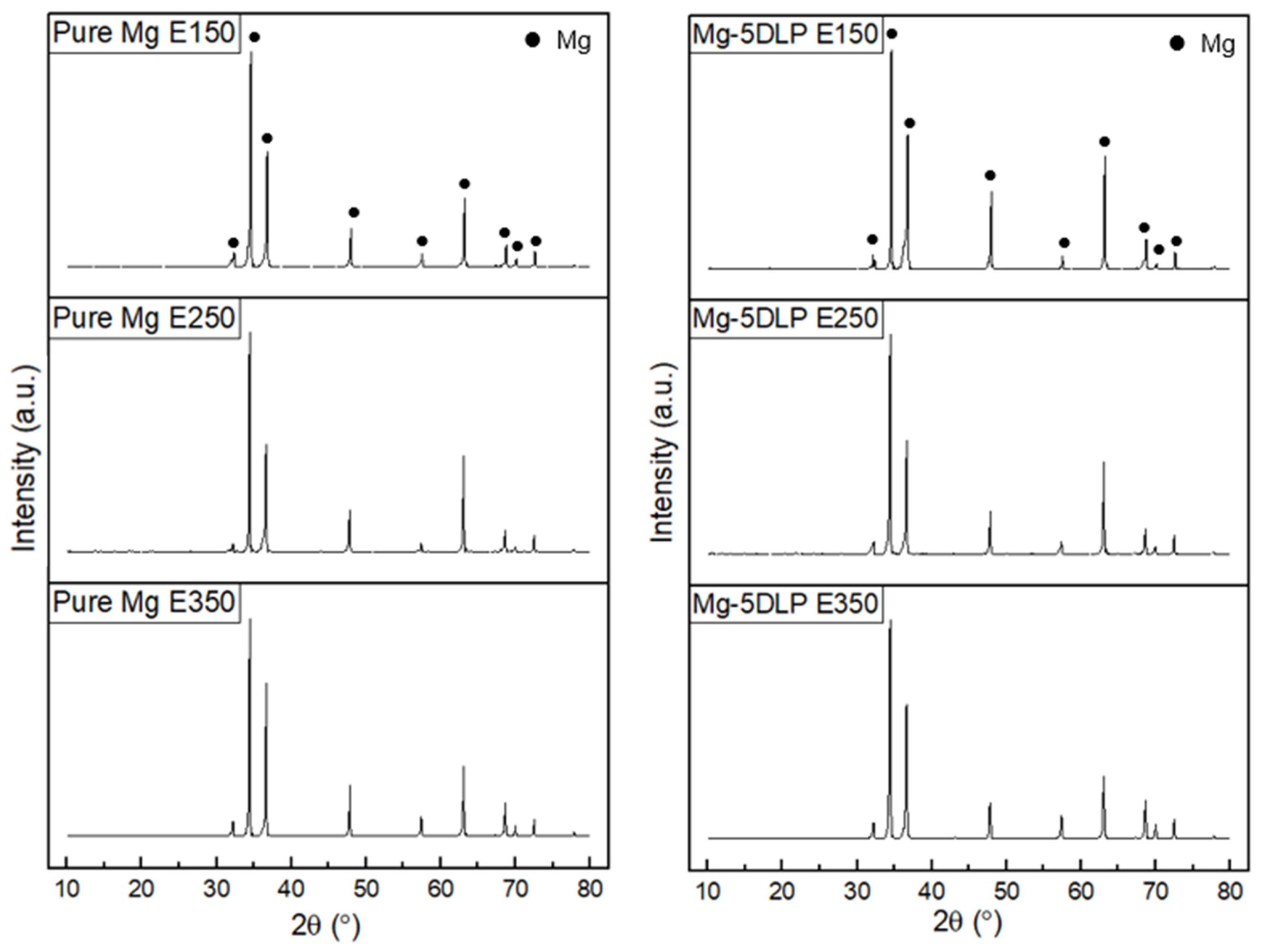
Figure 4 presents the XRD diffractograms of the materials in this work. Peak identification was carried out using JCPDS cards from the PDF-5+ (2024) database [30], where magnesium was indexed to card number 00-004-0770. The Mg-based materials in this study predominantly display characteristic Mg peaks with a dominant basal texture, as outlined in Table 4. It was observed that E250 processing resulted in a more dominant basal texture. The results indicate that the addition of DLP did not appreciably change the textural response, as all composite samples showed a dominant basal texture akin to pure Mg.
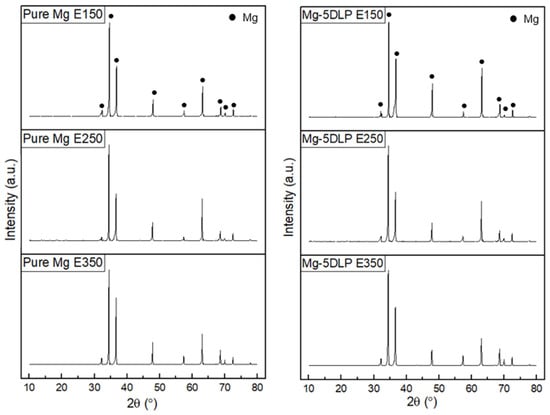
Figure 4.
X-Ray diffractograms of Mg materials explored in this work.

Table 4.
Crystallographic plane data of Mg materials explored in this work.
Figure 5 shows the micrographs and selected spectrum locations for EDS analysis, while Table 5 and Table 6 show the EDS results of the detected elements, corresponding to the various selected spectrum locations.

Figure 5.
SEM micrographs of Mg materials explored in this work, showing selected EDS spectrum locations.

Table 5.
Tabulated EDS results of pure Mg.

Table 6.
Tabulated EDS results of selected spectrum regions on biobased composites.
Closer examination of the SEM micrographs and corresponding EDS point analyses indicates that darker (porous) regions contain DLP-derived constituents [18,31]. This behavior is attributed to the evaporation of volatile components from the biomass during sintering, which leads to localized pore formation.
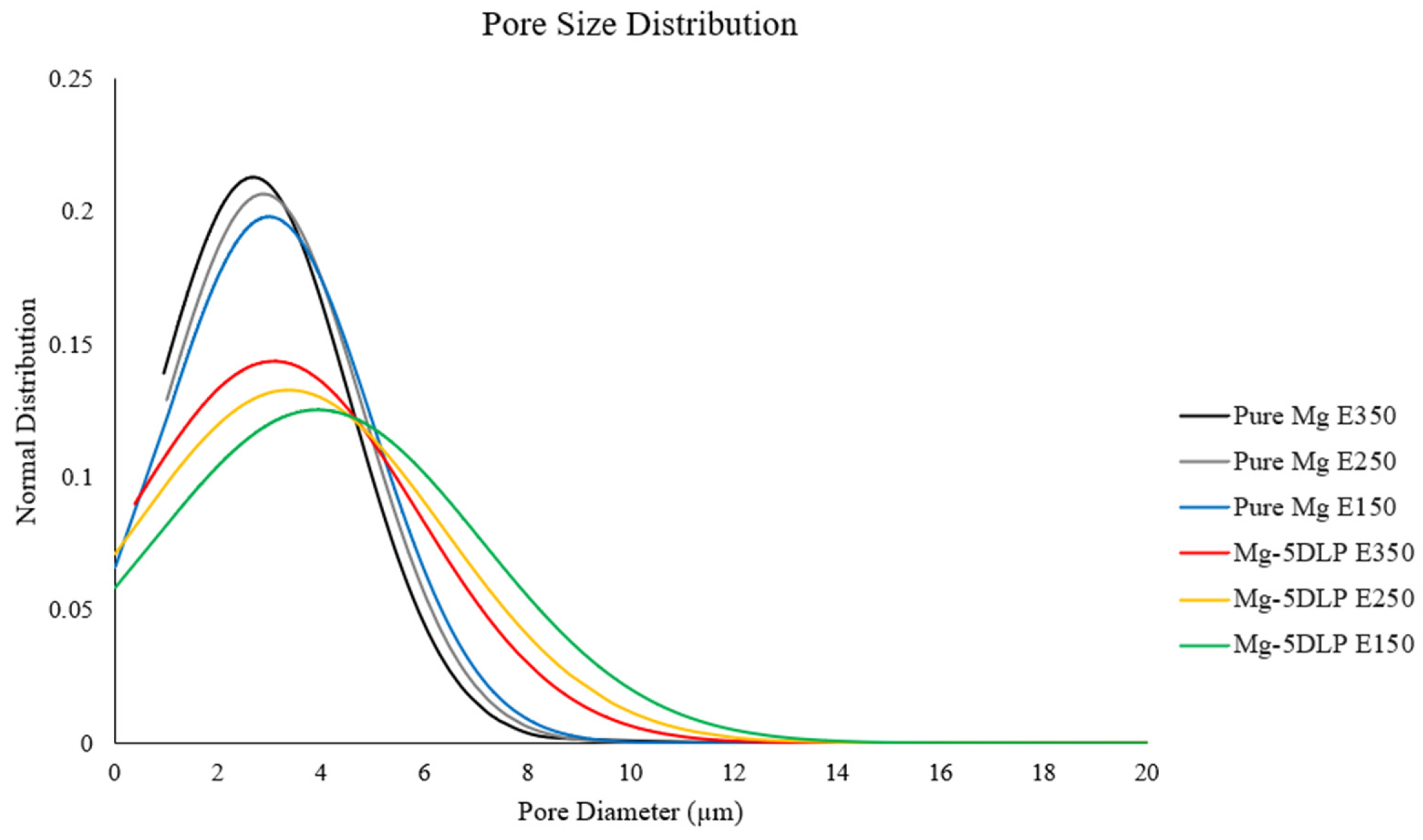
Table 7 and Figure 6 below showcase the higher porosity of the explored biobased composites. As previously described, this is attributed to the presence of DLP, resulting in the formation of localized pores in the material after sintering. Furthermore, compared to pure Mg, the inclusion of biomass particles introduces regions of weaker interfacial bonding and heterogeneous diffusion during sintering, which can further promote pore generation at the metal–biomass interfaces [32,33,34,35]. Finally, the pore size distribution was comparatively wider in Mg-5DLP composites. In the context of the extrusion temperature, the pore size variation was narrower at a 350 °C extrusion temperature for monolithic and composite samples.

Table 7.
The grain morphology, density, and porosity of the Mg materials explored in this work.
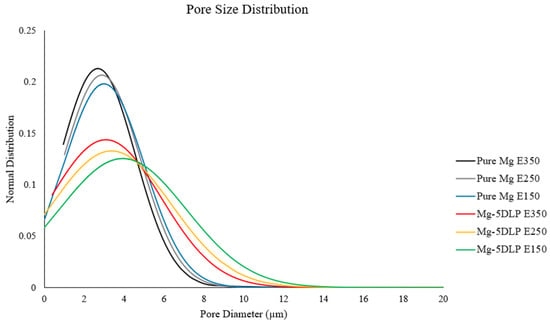
Figure 6.
Pore size distribution of the Mg materials in this work.
Porosity in powder metallurgy-processed Mg composites is affected by incomplete particle bonding, volatilization of non-metallic phases, and limited diffusion at lower processing temperatures [36]. Similar residual porosity has been reported in PM Mg matrix composites even after extrusion, particularly when low-density or non-metallic reinforcements are incorporated. Such behavior is intrinsic to PM-based processing routes and differs from cast-and-extruded Mg systems [37].
Considering the porosity values obtained, which were within normal range for PM-synthesized Mg composites in the literature (despite the lower sintering temperatures) [38], the material was consolidated effectively even with higher porosity compared to liquid-to-solid synthesis, e.g., casting [39].
Figure 7 and Table 7 show the grain size and morphology of the explored materials. The addition of DLP did not significantly alter the grain morphology (size and aspect ratio), suggesting that the DLP particles did not act as effective nucleation sites for grain refinement during processing.
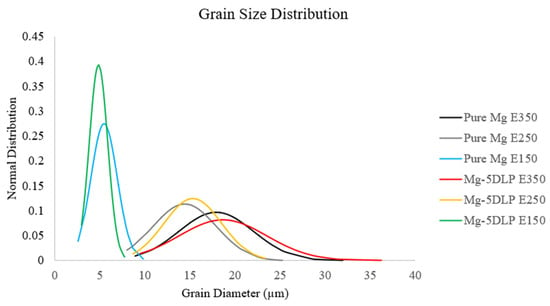
Figure 7.
Grain size distribution of Mg materials studied in this work.
Conversely, the decrease in extrusion temperature from E250 to E150 was accompanied by reductions in grain size (to 70.4% for pure Mg and to 72.6% for Mg-5DLP). This phenomenon can primarily be attributed to the reduced amount of energy available to allow grains to grow. The reduced extrusion temperatures suppress grain growth by decreasing the mobility of grain boundaries, thereby maintaining a finer grain structure; lower temperatures mean that there is less energy available to facilitate grain growth after the recrystallization [40]. These observations align with findings in other studies, where lower extrusion temperatures resulted in finer grain sizes in magnesium alloys [41,42].
3.2. Damping Analysis
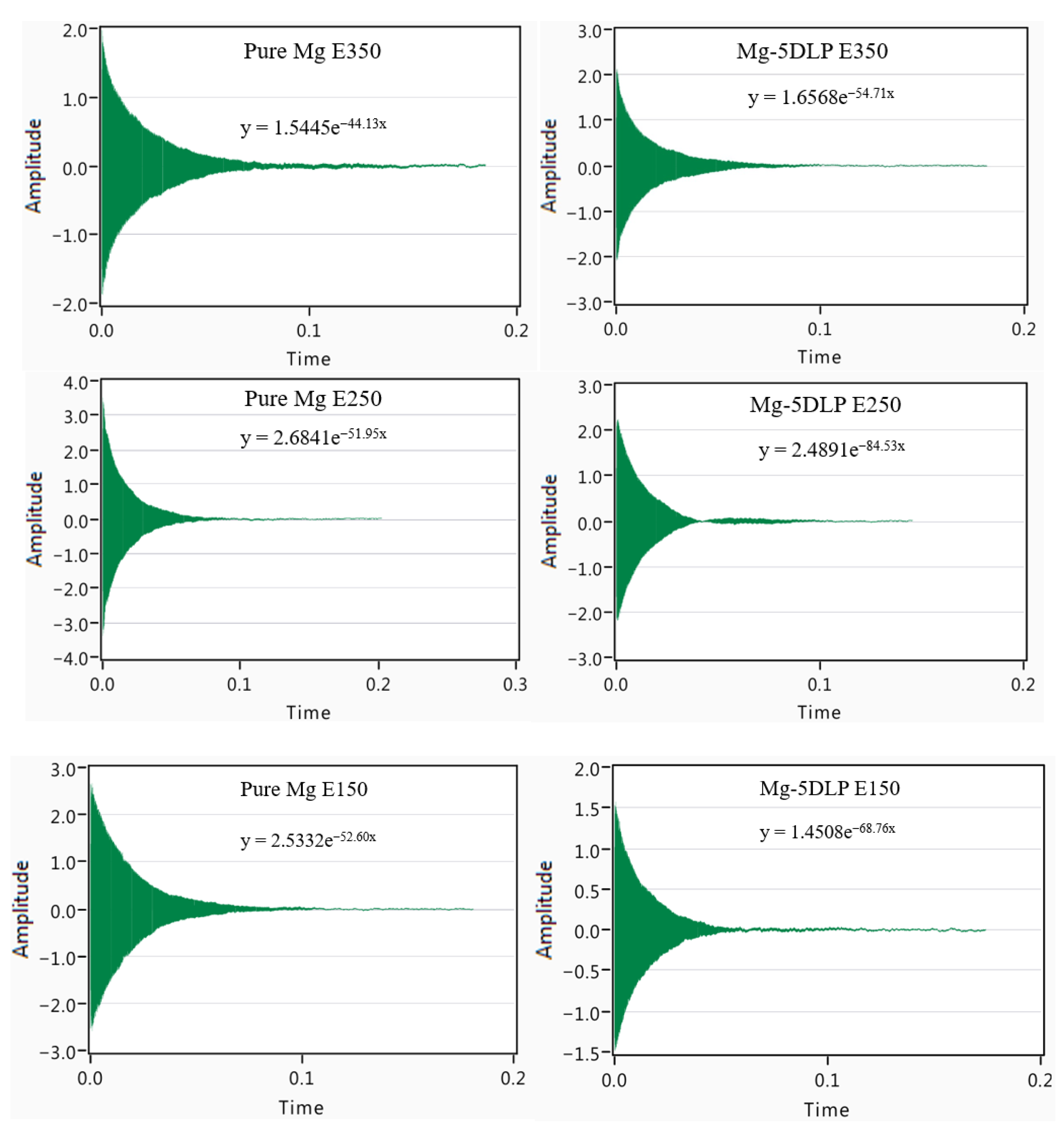
Figure 8 shows the vibration response curves of Mg materials in this work. A best-fit curve with a general form of Ae−bt was fitted, where b is the attenuation coefficient.

Figure 8.
Vibration response curves of explored Mg materials in this work.
Table 8 shows the damping properties of the materials explored in this work, with Pure Mg E150 and Mg-5DLP E250 exhibiting optimal performance. Compared with pure Mg, the biobased composite exhibits a superior damping performance because natural biomass (e.g., plant fibers/particles) promotes internal friction and energy dissipation at interfaces and within the particles themselves [43]. Studies on natural fiber composites report clear improvements in vibration damping and energy absorption when bio-reinforcements are used [44,45]. Furthermore, porous materials tend to show better damping performance because pores and internal voids allow local deformation, frictional motion, and viscous dissipation of mechanical energy. This explains why materials extruded at lower temperature (with higher porosity/air content) can exhibit better damping performance [46].

Table 8.
Damping properties and Young’s modulus of Mg materials in this work.
For the elastic modulus, pure Mg samples showed a reduction in the modulus with a decrease in extrusion temperature, and this can be attributed to a progressive increase in porosity [47]. However, Mg-5DLP composites exhibited the opposite trend (Table 7), and the addition of DLP negated the effect of porosity, which was higher compared to that of the monolithic samples. Further work is continuing in this area.
3.3. Mechanical Properties
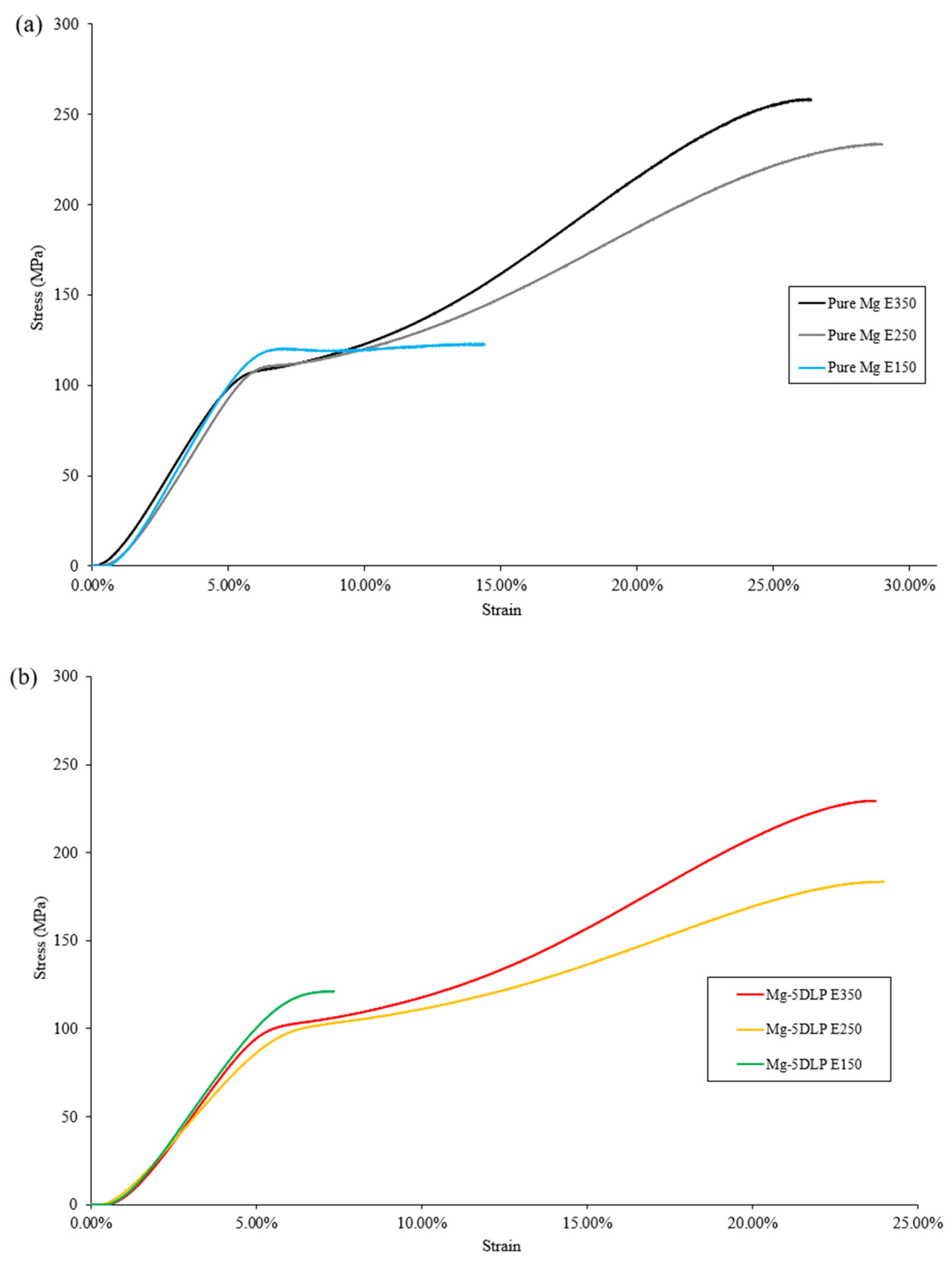
Table 9 outlines the mechanical properties of the Mg materials used in this study and Figure 9 shows the stress–strain curve for monolithic and composite samples studied at different extrusion temperatures.

Table 9.
Mechanical properties of Mg materials explored in this work.
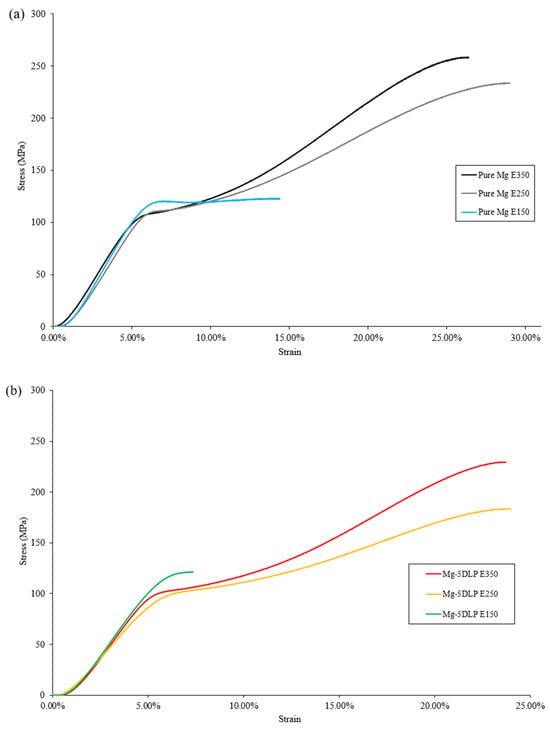
Figure 9.
Representative stress–strain curves for (a) pure Mg and (b) biobased composites in this work.
At lower extrusion temperatures, both hardness and ultimate compressive strength were reduced. This reduction in properties can be collectively attributed to higher porosity (hardness) and its ability to act as stress concentration site, lowering the material’s ability to resist plastic deformation (UCS). Similar behavior has also been reported in other metallic and Mg-based systems [48,49].
Additionally, according to the Hall–Petch relationship, finer grain structures obtained at lower extrusion temperatures contribute to increased yield strength, as smaller grains act as barriers to dislocation motion. Thus, the combined effects of grain refinement and retained dislocation density result in a moderate strengthening effect in the Mg–5DLP composites processed at lower extrusion temperatures [50,51,52]. The fracture strain for the E250 materials was similar to that of E350, showing good ductility at a moderate extrusion temperature. However, the E150 materials showed compromised ductility with low fracture strain. Similar trends are commonly seen within metallic composites exhibiting elevated porosity [53,54,55]. Overall, the results indicate that extrusion at 250 °C yielded the best combination of properties for pure Mg, while extrusion at 350 °C proved most suitable for Mg-5DLP composites.
Figure 10 shows the samples after compressive failure, with a 45-degree angle of fracture. Crushing was observed, which prevented more detailed examination of fracture surfaces.

Figure 10.
Macro-scale photographs of fractured Mg materials in this work.
3.4. Thermal Properties
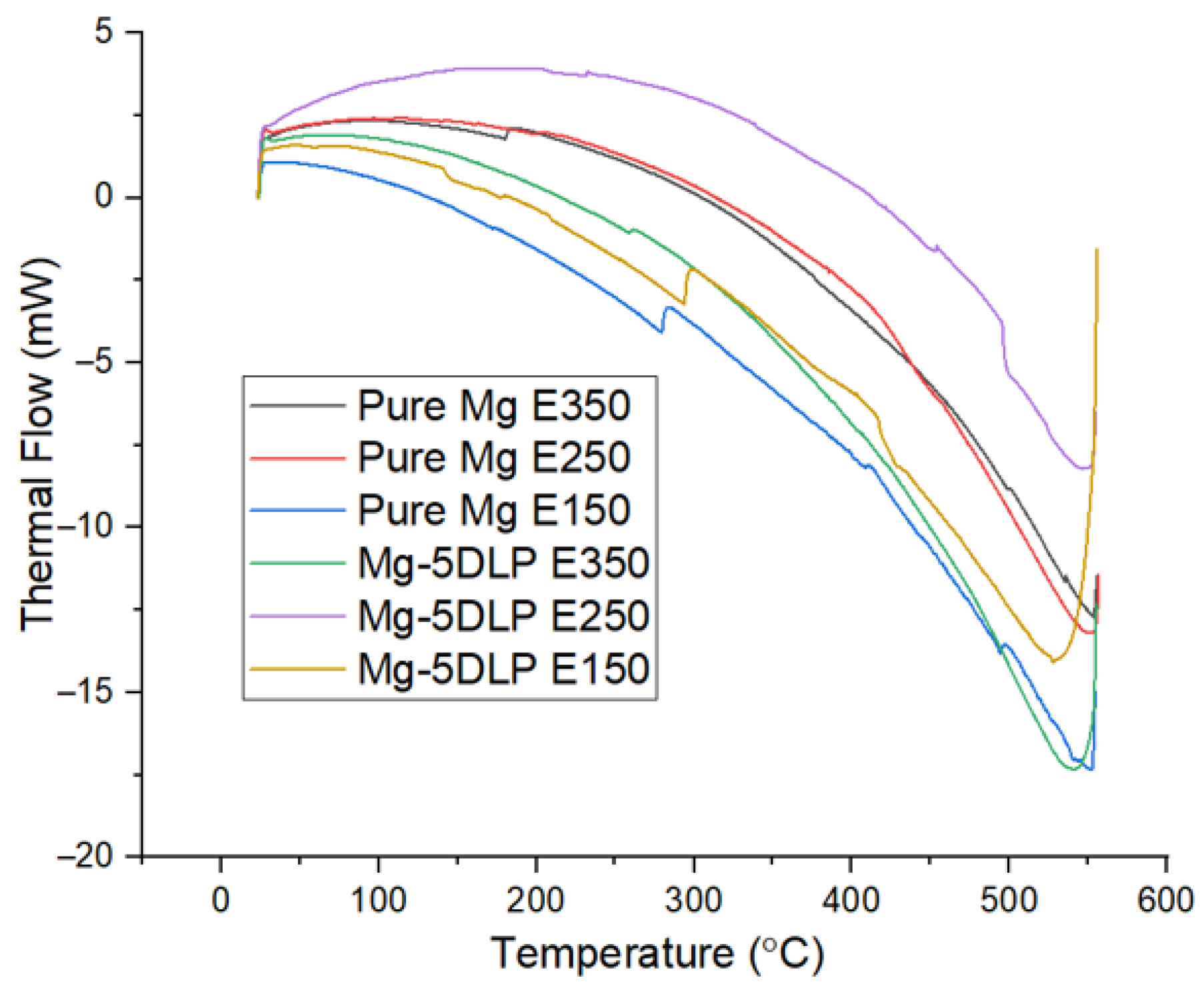
Figure 11 and Table 10 show the ignition responses of the pure Mg and biobased composites, while Figure 12 displays the thermal response of the Mg materials used in this work. The results indicate that neither ignition temperature nor thermal response were significantly affected by either the addition of DLP or the change in extrusion temperature, demonstrating that thermal stability remains largely uncompromised following the addition of DLP particles. Table 11 shows the CTE of the Mg materials. No significant changes in CTE were observed for the E350 and E250 materials, while a noticeable increase was seen for the E150 materials. This increase is likely due to higher porosity at lower extrusion temperatures, something which was also observed with stainless-steel materials used in a different study [56].
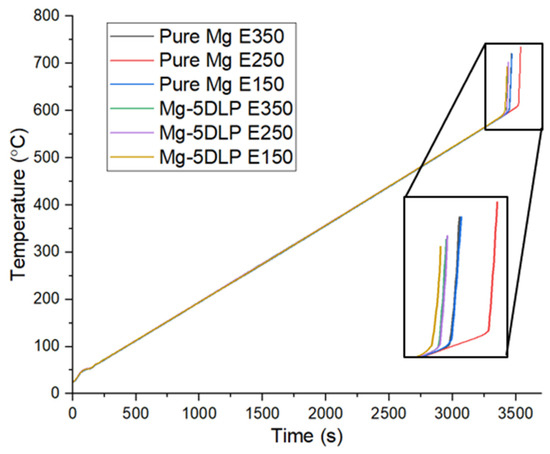
Figure 11.
Ignition response of Mg materials explored in this work.

Table 10.
Ignition temperature of Mg materials explored in this work.

Figure 12.
Thermal response of Mg materials explored in this work.

Table 11.
Coefficient of thermal expansion of Mg materials explored in this work.
3.5. Corrosion Response
Table 12 shows that corrosion response was affected by both extrusion temperature and the presence of DLP. At a higher extrusion temperature (E350), Mg–5DLP material corroded much faster than pure Mg (84.6 vs. 1.7 mm/year), and at E250 the composite still corroded more (15.6 vs. 3.5 mm/year). However, at the lowest extrusion temperature (E150) the Mg–5DLP composite showed a significantly lower corrosion rate (1.3 mm/year), even lower than that of pure Mg at the same temperature (2.0 mm/year). At higher processing temperatures (E350 materials), the DLP experienced greater thermal degradation (pyrolysis to biochar) which increased electrical conductivity [57,58,59,60], which in turn promoted galvanic corrosion and sped up corrosion rates. In contrast, the lower extrusion temperature in E150 processing minimized this effect, resulting in lower corrosion rates.

Table 12.
Corrosion rates of pure Mg and biobased composites explored in this work.
The much more significant improvements in corrosion rate for Mg-5DLP in correlation with the lower extrusion temperature suggest that chemical effects have a more significant effect on corrosion rates compared to morphological factors (i.e., porosity).
4. Conclusions
Pure Mg and Mg–5DLP composites were successfully synthesized using powder metallurgy incorporating energy-efficient microwave processing in this work. The key conclusions are as follows:
- (1)
- The inclusion of 5 wt.% Dried Leaf Powder (DLP) had minimal impact on the overall microstructure, but finer grains were observed at lower extrusion temperatures (a 70.4% reduction for pure Mg E150 and a 72.6% reduction for Mg-5DLP E150), which can be attributed to enhanced dynamic recrystallization and limited grain growth caused by lower processing temperatures.
- (2)
- The explored biobased composites consistently exhibited higher porosity, primarily due to evaporation of volatile organics, weak interfacial bonding, and limited diffusion between Mg and DLP particles at lower sintering temperatures necessitated by the thermal sensitivity of biomass-derived reinforcements.
- (3)
- The higher porosity enhanced the damping capacity and attenuation coefficient for both pure Mg and Mg–5DLP, particularly at lower extrusion temperatures (a 41.2% and a 54.5% increase, respectively, for E250 processing).
- (4)
- Mechanical performance showed a trade-off with decreasing extrusion temperature: lower temperatures led to increased porosity, which reduced hardness, compressive strength, and ductility, while a slight increase in yield strength was observed (a 5.9% increase for pure Mg E150 and a 14.0% increase for Mg–5DLP) due to retained dislocation density. Overall, the results indicate that extrusion at 250 °C yields the best combination of properties for pure Mg, while extrusion at 350 °C is most suitable for Mg-5DLP composites.
- (5)
- The thermal stability of Mg was largely retained after DLP addition.
- (6)
- Corrosion behavior was influenced by both DLP addition and extrusion temperature. The Mg-5DLP biobased composite corroded faster than pure Mg at higher extrusion temperatures; however, at the lowest extrusion temperature (E150), the Mg–5DLP composite exhibited corrosion resistance superior to that of pure Mg (1.3 mm/year as compared to 2.0 mm/year), highlighting the potential of processing conditions in property tailoring and the importance of chemical factors in comparison to morphological ones in relation to corrosion management.
- (7)
- Overall, adding DLP improves damping and further reduces weight but requires careful control of processing parameters to minimize negative effects on mechanical and corrosion properties.
Future work could involve further refinement of processing parameters, possible pre-processing of DLP, and compositional control (through the addition of alloying elements and a change in matrix material) to tailor properties and minimize compromises for high-performance applications.
These findings not only highlight the potential of incorporating natural biomass into metallic systems for developing lightweight and sustainable materials, but also establish a strong foundation for future investigations into metal–biomass composite design, processing optimization, and performance enhancement while minimizing the potential limitations.
The successful incorporation of natural biomass, despite the inherent thermal stability compromise of the initial raw biomass material, presents an encouraging and new research path for bio-derived reinforcements in future metallic composites. Considering the biocompatible nature of magnesium as is, one potential field in which it could prove useful is the biomedical sector, where functional properties (e.g., drug delivery, bioactive compounds) are paramount to not just recovery, but enhancement of general health.
Author Contributions
Conceptualization, M.G.; methodology, M.J. and M.G.; validation, M.J. and M.G.; formal analysis, A.G. and M.J.; investigation, A.G. and M.J.; resources, M.J. and M.G.; data curation, A.G. and M.J.; writing—original draft preparation, A.G.; writing—review and editing, M.J. and M.G.; visualization, A.G. and M.J.; supervision, M.J. and M.G.; project administration, M.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Acknowledgments
The authors would like to acknowledge Juraimi Bin Madon for the extrusion work and Ng Hong Wei for the assistance with DSC, CTE and TGA testing.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Gupta, M.; Sharon, N.M.L. Magnesium, Magnesium Alloys, and Magnesium Composites, 1st ed.; Wiley: Hoboken, NJ, USA, 2011. [Google Scholar]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Farraro, K.F.; Kim, K.E.; Woo, S.L.Y.; Flowers, J.R.; McCullough, M.B. Revolutionizing orthopaedic biomaterials: The potential of biodegradable and bioresorbable magnesium-based materials for functional tissue engineering. J. Biomech. 2014, 47, 1979–1986. [Google Scholar] [CrossRef]
- Sietsema, W.K. Animal models of cortical porosity. Bone 1995, 17, S297–S305. [Google Scholar] [CrossRef]
- Ramirez, J.M.; Hurt, W.C. Bone Remodeling in Periodontal Lesions. J. Periodontol. 1977, 48, 74–77. [Google Scholar] [CrossRef] [PubMed]
- Xin, Y.; Hu, T.; Chu, P.K. In vitro studies of biomedical magnesium alloys in a simulated physiological environment: A review. Acta Biomater. 2011, 7, 1452–1459. [Google Scholar] [CrossRef] [PubMed]
- Rude, R.K. Physiology of magnesium metabolism and the important role of magnesium in potassium deficiency. Am. J. Cardiol. 1989, 63, G31–G34. [Google Scholar] [CrossRef]
- Mooren, F.C.; Golf, S.W.; Lechtermann, A.; Völker, K. Alterations of ionized Mg2+ in human blood after exercise. Life Sci. 2005, 77, 1211–1225. [Google Scholar] [CrossRef]
- Han, J.Y.; Jung, H.Y.; Cho, J.R.; Choi, J.H.; Bae, W.B. Buckling analysis and test of composite shells under hydrostatic pressure. J. Mater. Process. Technol. 2008, 201, 742–745. [Google Scholar] [CrossRef]
- Galvagno, S.; Portofino, S.; Casciaro, G.; Casu, S.; d’Aquino, L.; Martino, M.; Russo, A.; Bezzi, G. Synthesis of beta silicon carbide powders from biomass gasification residue. J. Mater. Sci. 2007, 42, 6878–6886. [Google Scholar] [CrossRef]
- Teong, S.P.; Zhang, Y. Calcium carbide and its recent advances in biomass conversion. J. Bioresour. Bioprod. 2020, 5, 96–100. [Google Scholar] [CrossRef]
- Yu, J.; Luo, B.; Wang, Y.; Wang, S.; Wu, K.; Liu, C.; Chu, S.; Zhang, H. An efficient way to synthesize biomass-based molybdenum carbide catalyst via pyrolysis carbonization and its application for lignin catalytic pyrolysis. Bioresour. Technol. 2022, 346, 126640. [Google Scholar] [CrossRef]
- Negi, P.; Chhantyal, A.K.; Dixit, A.K.; Kumar, S.; Kumar, A. Activated carbon derived from mango leaves as an enhanced microwave absorbing material. Sustain. Mater. Technol. 2021, 27, e00244. [Google Scholar] [CrossRef]
- Zhang, Y.-N.; Su, C.-Y.; Chen, J.-L.; Huang, W.-H.; Lou, R. Recent progress of transition metal-based biomass-derived carbon composites for supercapacitor. Rare Met. 2023, 42, 769–796. [Google Scholar] [CrossRef]
- Xiong, C.; Zheng, C.; Jiang, X.; Xiao, X.; Wei, H.; Zhou, Q.; Ni, Y. Recent progress of green biomass based composite materials applied in supercapacitors, sensors, and electrocatalysis. J. Energy Storage 2023, 72, 108633. [Google Scholar] [CrossRef]
- Osman, A.I.; Abdelkader, A.; Farrell, C.; Rooney, D.; Morgan, K. Reusing, recycling and up-cycling of biomass: A review of practical and kinetic modelling approaches. Fuel Process. Technol. 2019, 192, 179–202. [Google Scholar] [CrossRef]
- El-Sayed, S.A.; Mostafa, M.E. Thermal pyrolysis and kinetic parameter determination of mango leaves using common and new proposed parallel kinetic models. RSC Adv. 2020, 10, 18160–18179. [Google Scholar] [CrossRef]
- Zhang, M.I.N.; Willison, J.H.M. Electrical Impedance Analysis in Plant Tissues: Impedance Measurement in Leaves. J. Exp. Bot. 1993, 44, 1369–1375. [Google Scholar] [CrossRef]
- Wang, S.; Ren, L.; Liu, Y.; Han, Z.; Yang, Y. Mechanical Characteristics of Typical Plant Leaves. J. Bionic Eng. 2010, 7, 294–300. [Google Scholar] [CrossRef]
- Parande, G.; Manakari, V.; Meenashisundaram, G.K.; Gupta, M. Enhancing the hardness/compression/damping response of magnesium by reinforcing with biocompatible silica nanoparticulates. Int. J. Mater. Res. 2016, 107, 1091–1099. [Google Scholar] [CrossRef]
- Johanes, M.; Gupta, M. An Investigation into the Potential of Turning Induced Deformation Technique for Developing Porous Magnesium and Mg-SiO2 Nanocomposite. Materials 2023, 16, 2463. [Google Scholar] [CrossRef] [PubMed]
- Johanes, M.; Mehtabuddin, S.; Venkatarangan, V.; Gupta, M. An Insight into the Varying Effects of Different Cryogenic Temperatures on the Microstructure and the Thermal and Compressive Response of a Mg/SiO2 Nanocomposite. Metals 2024, 14, 808. [Google Scholar] [CrossRef]
- Gupta, S.; Johanes, M.; Parande, G.; Gupta, M. An Investigation into the Effect of Length Scale of Reinforcement on the Cryogenic Response of a Mg/2wt.%CeO2 Composite. Micro 2024, 4, 170–184. [Google Scholar] [CrossRef]
- Tekumalla, S.; Joo Yuan, N.; Haghshenas, M.; Gupta, M. Enhancing Properties of Aerospace Alloy Elektron 21 Using Boron Carbide Nanoparticles as Reinforcement. Appl. Sci. 2019, 9, 5470. [Google Scholar] [CrossRef]
- Sharma, A.; Garg, H.; Ragit, S.S. An overview of the future prospects and challenges of TiC-reinforced magnesium matrix composites. Int. J. Sci. Res. Arch. 2025, 14, 643–655. [Google Scholar] [CrossRef]
- ASTM E112-13 (2021); Standard Test Methods for Determining Average Grain Size. ASTM International: West Conshohocken, PA, USA, 2024.
- ISO 14577-1:2015; Metallic Materials—Instrumented Indentation Test for Hardness and Materials Parameters. International Organization for Standardization: Geneva, Switzerland, 2015.
- ASTM E9-09; Standard Test Methods of Compression Testing of Metallic Materials at Room Temperature. ASTM International: West Conshohocken, PA, USA, 2018.
- Singh, I.B.; Singh, M.; Das, S. A comparative corrosion behavior of Mg, AZ31 and AZ91 alloys in 3.5% NaCl solution. J. Magnes. Alloys 2015, 3, 142–148. [Google Scholar] [CrossRef]
- Kabekkodu, S.N.; Dosen, A.; Blanton, T.N. PDF-5+: A comprehensive Powder Diffraction File™ for materials characterization. Powder Diffr. 2024, 39, 47–59. [Google Scholar] [CrossRef]
- Kumar, M.; Saurabh, V.; Tomar, M.; Hasan, M.; Changan, S.; Sasi, M.; Maheshwari, C.; Prajapati, U.; Singh, S.; Prajapat, R.K.; et al. Mango (Mangifera indica L.) Leaves: Nutritional Composition, Phytochemical Profile, and Health-Promoting Bioactivities. Antioxidants 2021, 10, 299. [Google Scholar] [CrossRef]
- Cheng, M.-Q.; Wahafu, T.; Jiang, G.-F.; Liu, W.; Qiao, Y.-Q.; Peng, X.-C.; Cheng, T.; Zhang, X.-L.; He, G.; Liu, X.-Y. A novel open-porous magnesium scaffold with controllable microstructures and properties for bone regeneration. Sci. Rep. 2016, 6, 24134. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Ogata, S. Magnesium based alloys for reinforcing biopolymer composites and coatings: A critical overview on biomedical materials. Adv. Ind. Eng. Polym. Res. 2023, 6, 341–355. [Google Scholar] [CrossRef]
- Diao, R.; Li, B.; Qi, F.; Zha, Z.; Ma, P. Insights into mineral removal, fusion, and evolution in biomass valorization through integrated pretreatment strategies from an ash chemistry perspective. Ind. Crops Prod. 2025, 233, 121445. [Google Scholar] [CrossRef]
- Arora, G.S.; Saxena, K.K.; Mohammed, K.A.; Prakash, C.; Dixit, S. Manufacturing Techniques for Mg-Based Metal Matrix Composite with Different Reinforcements. Crystals 2022, 12, 945. [Google Scholar] [CrossRef]
- Jiang, C.; Gu, L.; Xiong, H.; Wang, J.; Zhou, L.; Shen, M.; Wang, J.; Ying, T.; Zeng, X. Review of progress on fabrication technology of Mg matrix composites. J. Magnes. Alloys 2024, 12, 4768–4807. [Google Scholar] [CrossRef]
- Kumar, S.; Gupta, R.N. A Review on Fabrication and Properties of Magnesium Metal Matrix Composite Materials. In Recent Advancements in Mechanical Engineering; Springer: Singapore, 2024; pp. 183–195. [Google Scholar]
- Fu, X.; Deng, R.; Kong, X.; Parande, G.; Hu, J.; Peng, P.; Zhu, Z.; Shi, B.; Wang, G.; Gupta, M.; et al. Interfacial characterization and its influence on the corrosion behavior of Mg-SiO2 nanocomposites. Acta Mater. 2022, 230, 117840. [Google Scholar] [CrossRef]
- Čapek, J.; Vojtěch, D. Characterization of Porous Magnesium Prepared by Powder Metallurgy—Influence of Powder Shape. Manuf. Technol. J. 2014, 14, 271–275. [Google Scholar] [CrossRef]
- Li, Q.; Jiao, X. Recrystallization mechanism and activation energies of severely-deformed magnesium during annealing process. Materialia 2019, 5, 100188. [Google Scholar] [CrossRef]
- Wang, M.; Li, Z.; Hu, Y.; Dai, Y.; Fu, L.; Jiang, B.; Pan, F. Effect of extrusion temperature on microstructure, mechanical properties, and deformation mechanism of Mg-2.5Er-0.6Zr alloy. J. Mater. Res. Technol. 2023, 24, 4685–4697. [Google Scholar] [CrossRef]
- Li, Y.; Hou, P.; Wu, Z.; Feng, Z.; Ren, Y.; Choo, H. Dynamic recrystallization of a wrought magnesium alloy: Grain size and texture maps and their application for mechanical behavior predictions. Mater. Des. 2021, 202, 109562. [Google Scholar] [CrossRef]
- Wang, J.; Zou, Y.; Dang, C.; Wan, Z.; Wang, J.; Pan, F. Research Progress and the Prospect of Damping Magnesium Alloys. Materials 2024, 17, 1285. [Google Scholar] [CrossRef] [PubMed]
- Şimşir, E.; Akçin Ergün, Y.; Yavuz, İ. Investigation of Damping Properties of Natural Fiber-Reinforced Composites at Various Impact Energy Levels. Polymers 2024, 16, 3553. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Sharma, R.; Katarne, R. Experimental investigation on influence of damping response of composite material by natural fibers—A review. Mater. Today Proc. 2021, 47, 3035–3042. [Google Scholar] [CrossRef]
- Zhou, G.; Jiang, H.; Liu, C.; Huang, H.; Wei, L.; Meng, Z. Effect of porous particle layer on damping capacity and storage modulus of AlSi30p/5052Al composites. Mater. Lett. 2021, 300, 130162. [Google Scholar] [CrossRef]
- Liu, B.; Pan, Y. Effect of Pore Shape on Mechanical Properties of Porous Shape Memory Alloy. Micromachines 2022, 13, 566. [Google Scholar] [CrossRef]
- Rahmani Fard, R.; Akhlaghi, F. Effect of extrusion temperature on the microstructure and porosity of A356-SiCp composites. J. Mater. Process. Technol. 2007, 187–188, 433–436. [Google Scholar] [CrossRef]
- Dong, S.; Zhang, B.; Zhan, Y.; Liu, X.; Xin, L.; Yang, W.; Wu, G. Effect of Extrusion Temperature on the Microstructure and Mechanical Properties of SiCnw/2024Al Composite. Materials 2019, 12, 2769. [Google Scholar] [CrossRef]
- Zengin, H.; Ari, S.; Turan, M.E.; Hassel, A.W. Evolution of Microstructure, Mechanical Properties, and Corrosion Resistance of Mg–2.2Gd–2.2Zn–0.2Ca (wt%) Alloy by Extrusion at Various Temperatures. Materials 2023, 16, 3075. [Google Scholar] [CrossRef]
- Carvalho, A.P.; Figueiredo, R.B. An Overview of the Effect of Grain Size on Mechanical Properties of Magnesium and Its Alloys. Mater. Trans. 2023, 64, 1272–1283. [Google Scholar] [CrossRef]
- Liu, X.; Mao, P.; Zhou, L.; Wang, X.; Wang, Z.; Wang, F.; Wei, Z.; Liu, Z. Effect of Grain Size on Dynamic Compression Behavior and Deformation Mechanism of ZK60 Magnesium Alloy. Metals 2023, 13, 314. [Google Scholar] [CrossRef]
- Aqida, S.N.; Ghazali, M.I.; Hashim, J. Effect of Porosity on Mechanical Properties of Metal Matrix Composite: An Overview. J. Teknol. 2012, 40, 17–32. [Google Scholar] [CrossRef]
- Li, X.; Gao, W.; Jiang, Q. Understanding the intrinsic framework of the Hall-Petch relationship of metals from the view of the electronic-structure level. Acta Mater. 2025, 292, 121071. [Google Scholar] [CrossRef]
- Yu, H.; Xin, Y.; Wang, M.; Liu, Q. Hall-Petch relationship in Mg alloys: A review. J. Mater. Sci. Technol. 2018, 34, 248–256. [Google Scholar] [CrossRef]
- Kozlovskii, Y.M.; Stankus, S.V.; Igumenov, I.K. Linear thermal expansion coefficient of porous stainless steel. J. Phys. Conf. Ser. 2020, 1677, 012168. [Google Scholar] [CrossRef]
- Das, C.; Tamrakar, S.; Kiziltas, A.; Xie, X. Incorporation of Biochar to Improve Mechanical, Thermal and Electrical Properties of Polymer Composites. Polymers 2021, 13, 2663. [Google Scholar] [CrossRef] [PubMed]
- Gabhi, R.; Basile, L.; Kirk, D.W.; Giorcelli, M.; Tagliaferro, A.; Jia, C.Q. Electrical conductivity of wood biochar monoliths and its dependence on pyrolysis temperature. Biochar 2020, 2, 369–378. [Google Scholar] [CrossRef]
- Kane, S.; Ulrich, R.; Harrington, A.; Stadie, N.P.; Ryan, C. Physical and chemical mechanisms that influence the electrical conductivity of lignin-derived biochar. Carbon Trends 2021, 5, 100088. [Google Scholar] [CrossRef]
- Park, W.; Kim, H.; Park, H.; Choi, S.; Hong, S.J.; Bahk, Y.-M. Biochar as a low-cost, eco-friendly, and electrically conductive material for terahertz applications. Sci. Rep. 2021, 11, 18498. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.