The Influence of the Ratio of Au and Pt Nanoparticles in Ternary Composites with TiO2
Abstract
1. Introduction
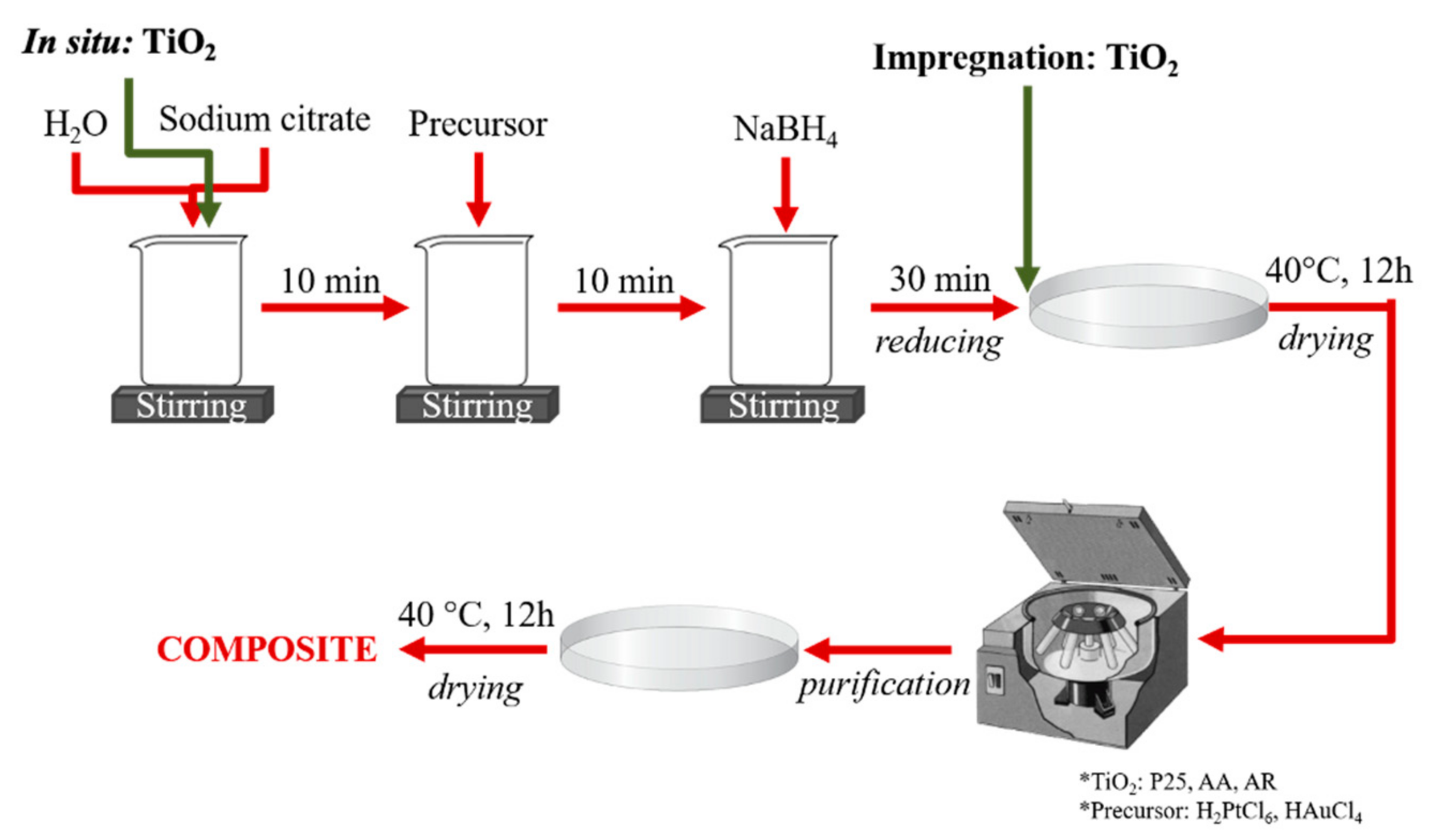
2. Materials and Methods
- 0.25% Au and 0.75% Pt;
- 0.75% Au and 0.25% Pt;
- 1% Au and 0% Pt—reference sample;
- 0% Au and 1% Pt—reference sample.
3. Results and Discussion
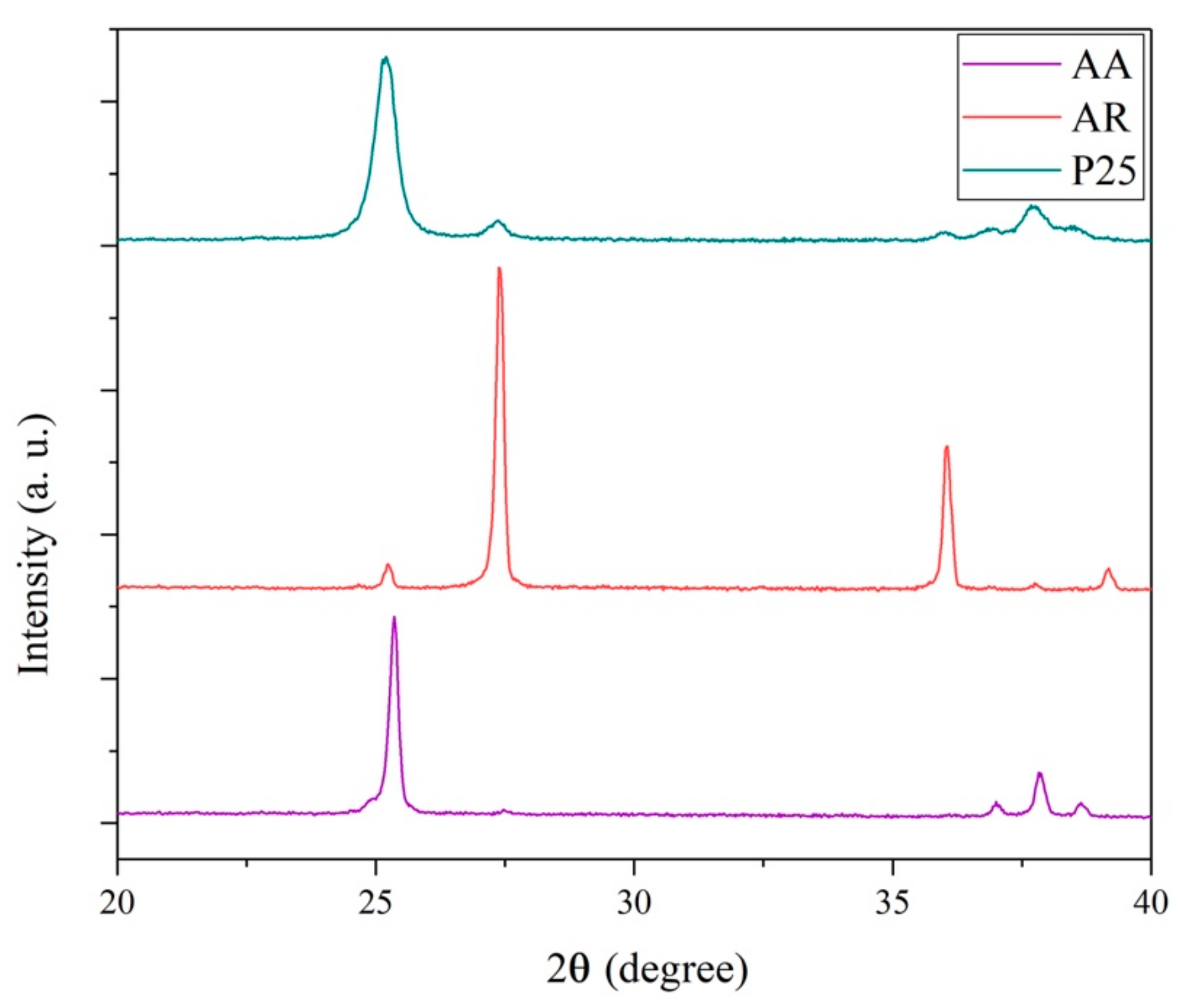
3.1. X-ray Diffraction
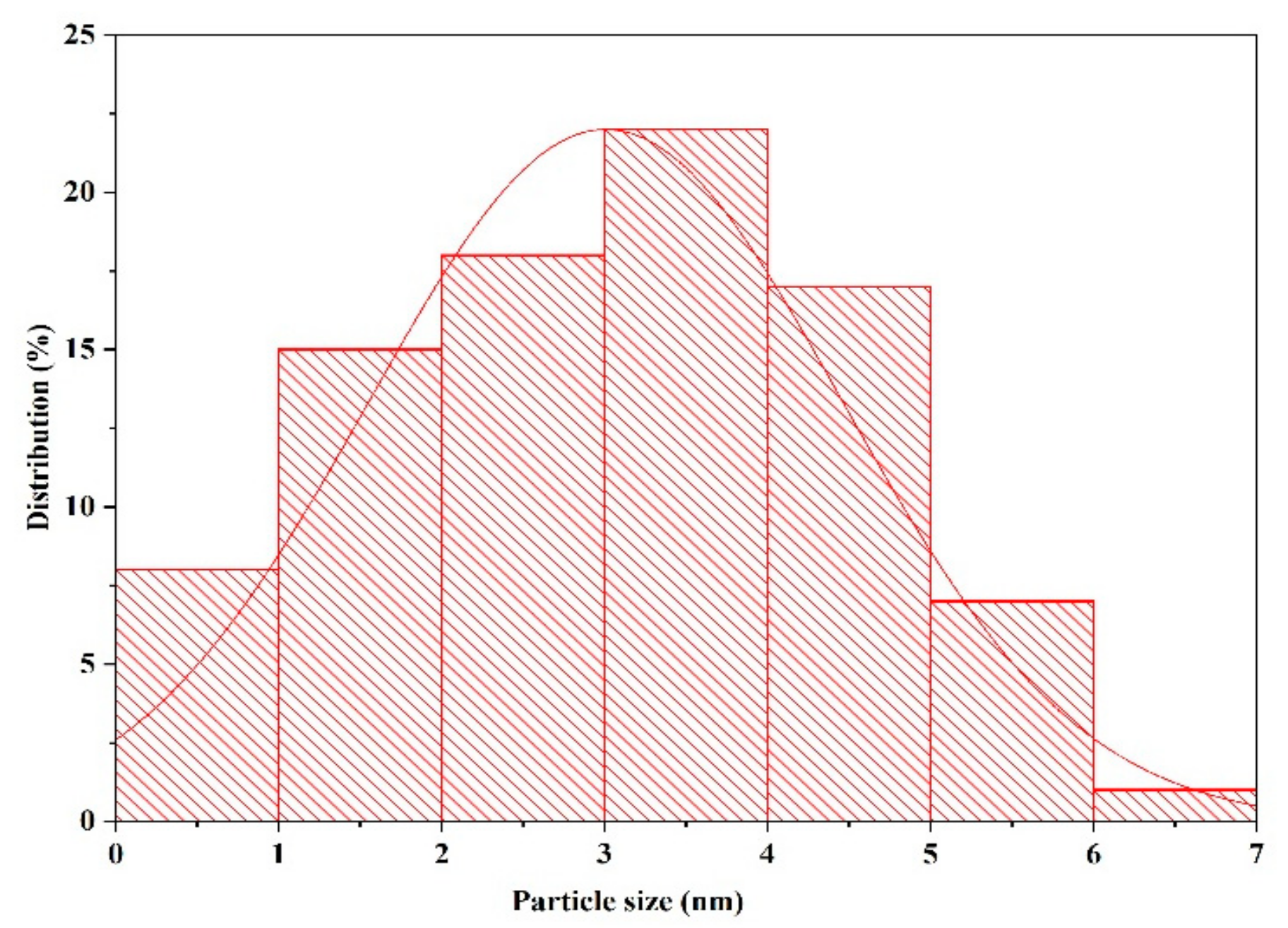
3.2. Transmission Electron Microscopy (TEM)
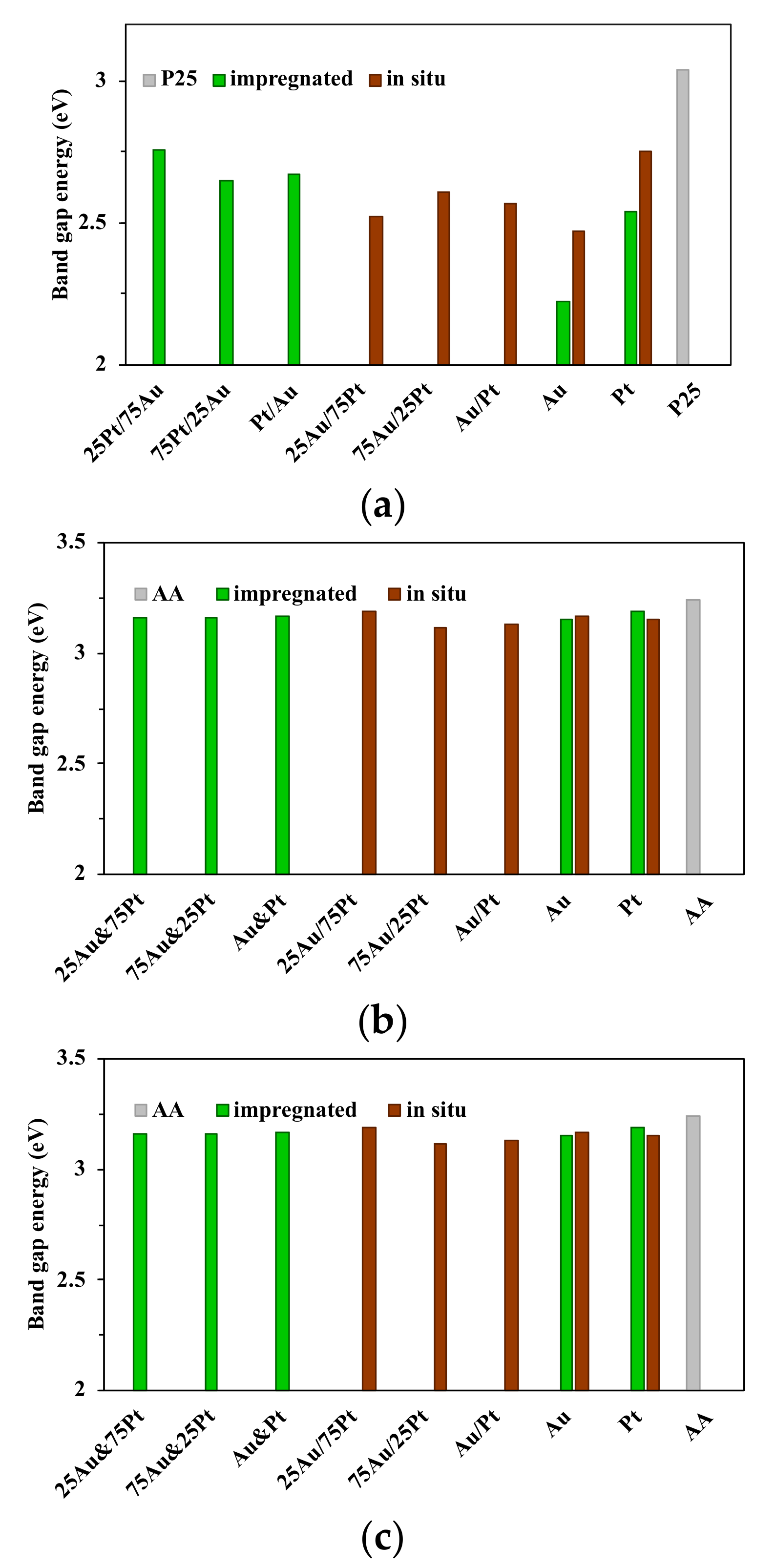
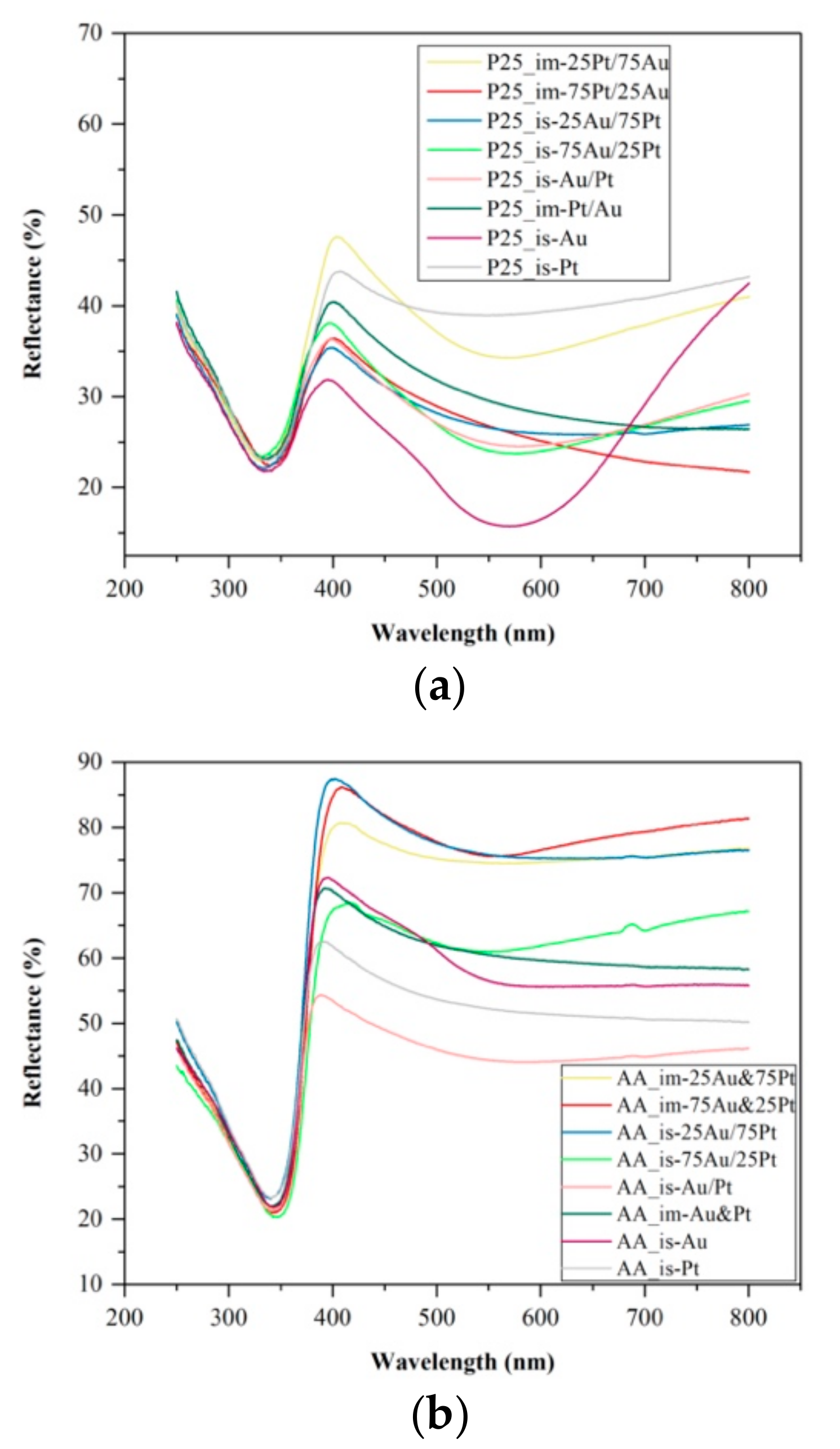
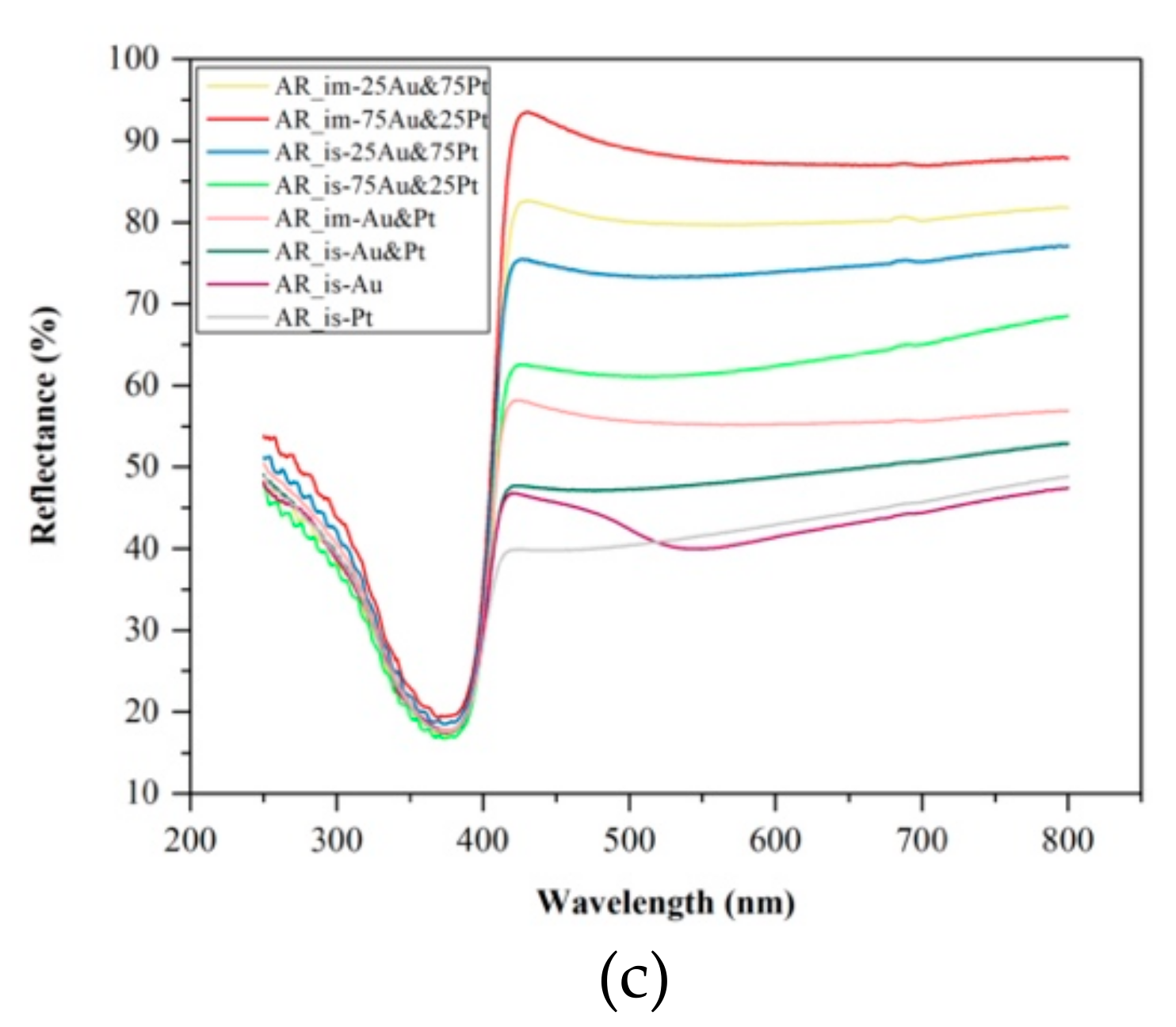
3.3. Diffuse Reflectance Spectroscopy (DRS)
- For the P25-based catalysts lowering the ratio of Pt nanoparticles resulted in the decrease of the bandgap energy (from 3.04 eV to 2.65 eV for P25_im-75Pt/25Au, to 2.52 eV for P25_is-25Au/75Pt);
- For AA-based photocatalysts obtained by impregnation, the decrease of the bandgap energy values cannot be linked with the ratio of the noble metals, while the differences between the values were also insignificant (the values vary from 3.15 to 3.17 eV). Using the in situ reductions, the same trend was observed as in the case of P25 (a decrease from 3.19 eV to 3.12 eV for AA_is-75Au/25Pt);
- When AR-based photocatalyst was investigated, the lowest bandgap energy was achieved when the ratio of the Au and Pt was 1:1 (2.89 eV for AR_is-Au&Pt and 2.91 eV for AR_im-Au&Pt).
3.4. Photocatalytic Performance of the Composites
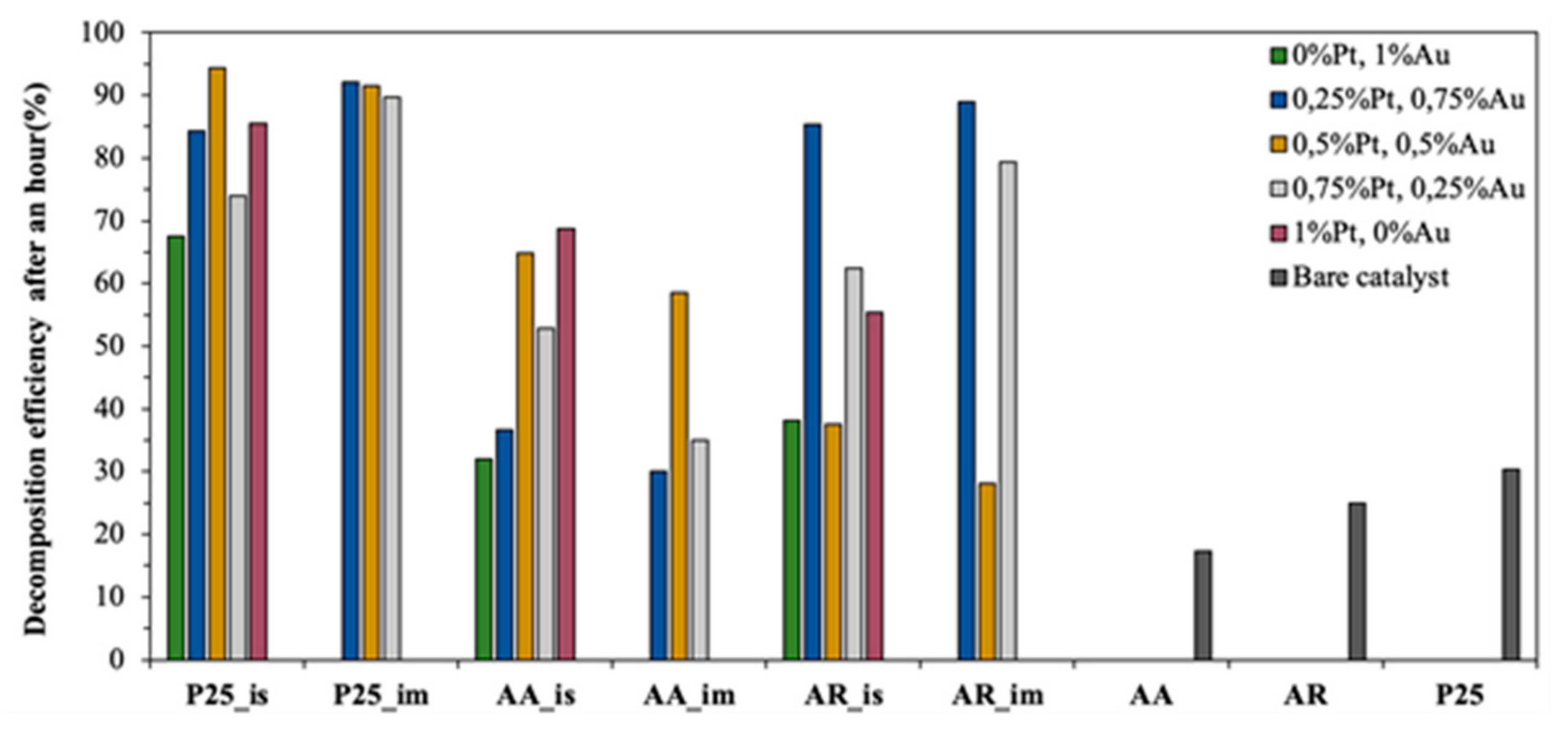
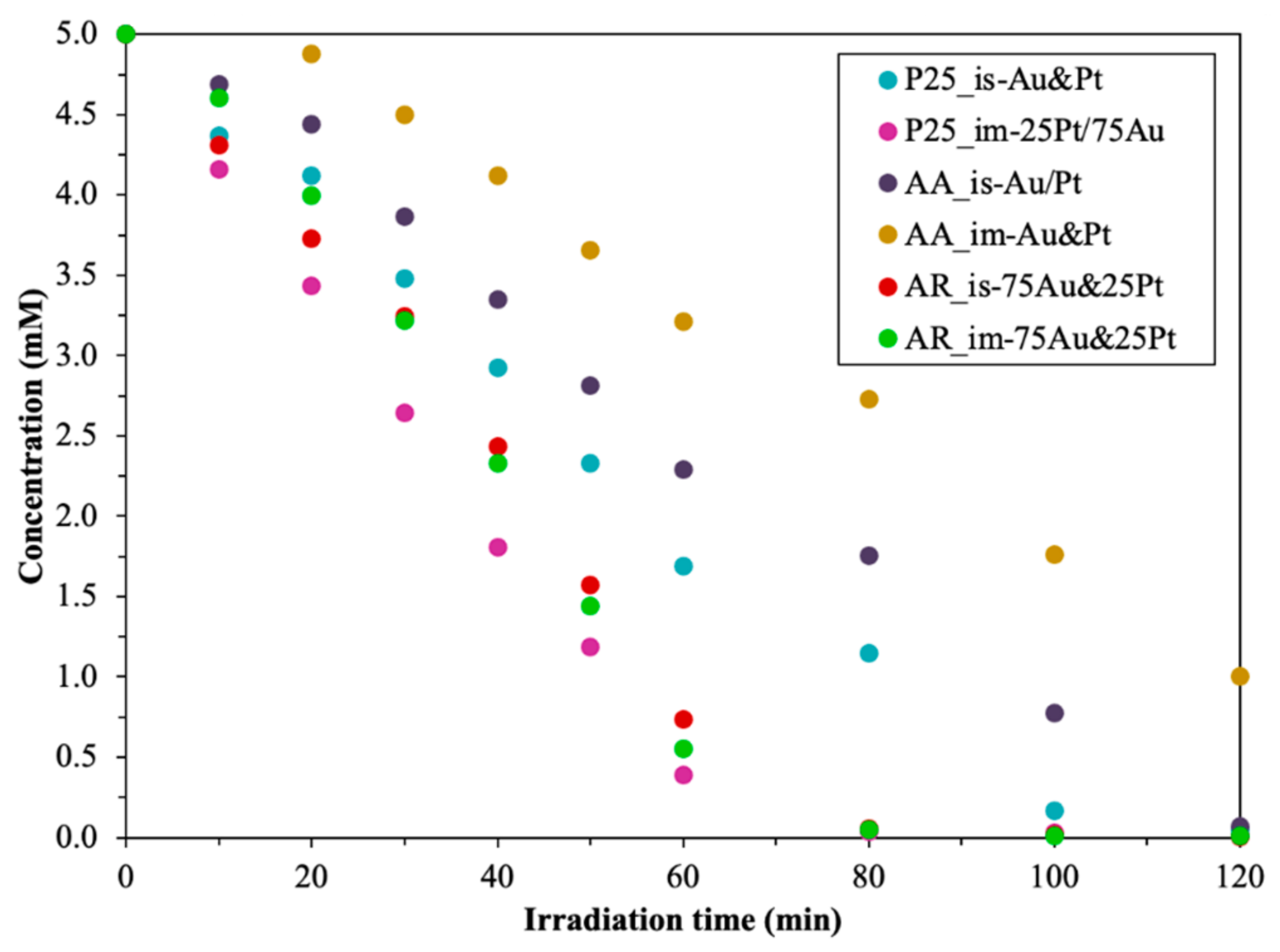
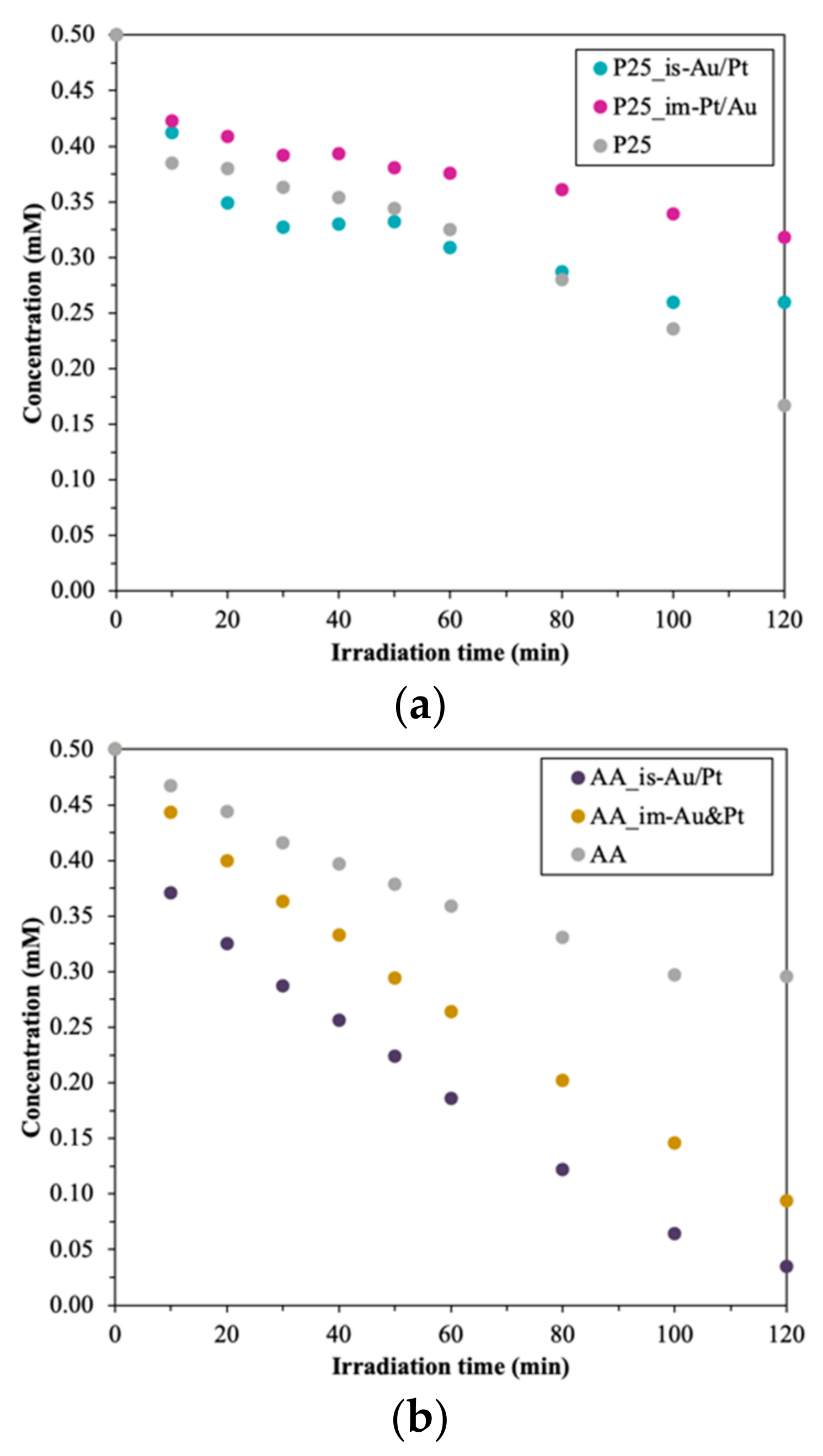
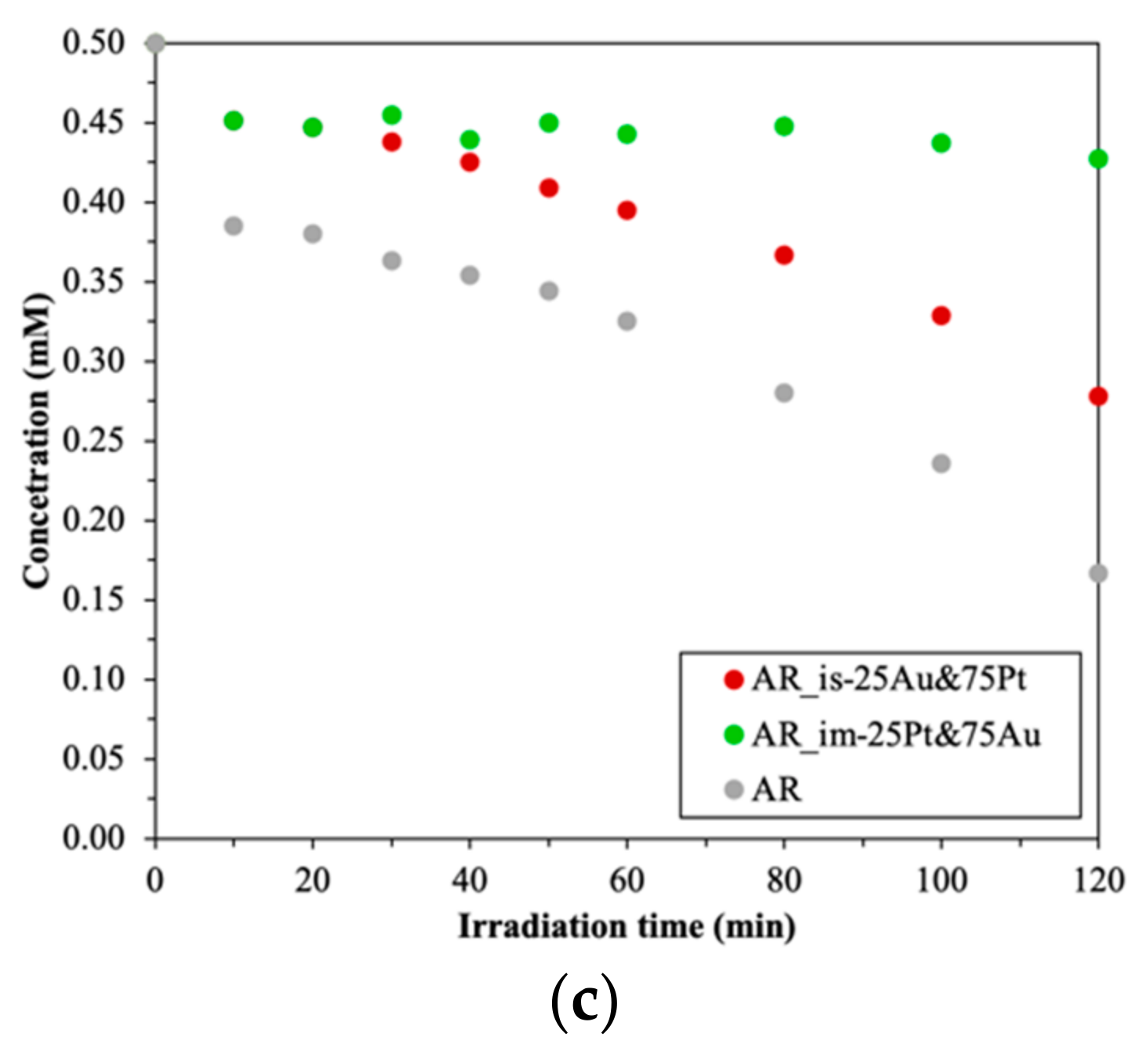
3.4.1. Photodegradation of Oxalic Acid
- In situ method: for the P25 and AA-based composites, the best photocatalytic activity was achieved when the sequential reduction was used—first Au then Pt (Au/Pt). For the AR-based composited the simultaneous reduction of the noble metals proved to be the best (Au&Pt);
- Impregnation: for the P25-based composites, the sequential reduction was the best as well as for the other method (first Pt then Au—Pt/Au). For the AR and AA-based catalysts, the simultaneous reduction of the nanoparticles proved to be the best according to oxalic acid degradation (Au&Pt);
- Regarding the order of the reduction of the two noble metals:
- ⚬
- First Au then Pt (Au/Pt): when the base catalyst was AA or P25, using in situ reductions in both cases (AA_is-Au/Pt, P25_is-Au/Pt);
- ⚬
- First Pt then Au (Pt/Au): when the base catalyst was the P25, using impregnation method (P25_im-Pt/Au);
- ⚬
- AU and Pt at the same time (Au&Pt): the AR-based composites (in situ and impregnation as well—(AR_is-Au&Pt, (AR_im-Au&Pt) and AA-based ones, using impregnation (AA_im-Au&Pt);
- The ratio between the noble metals: the change in the composition improved the photocatalytic activity only in the case of the AR-based composites.
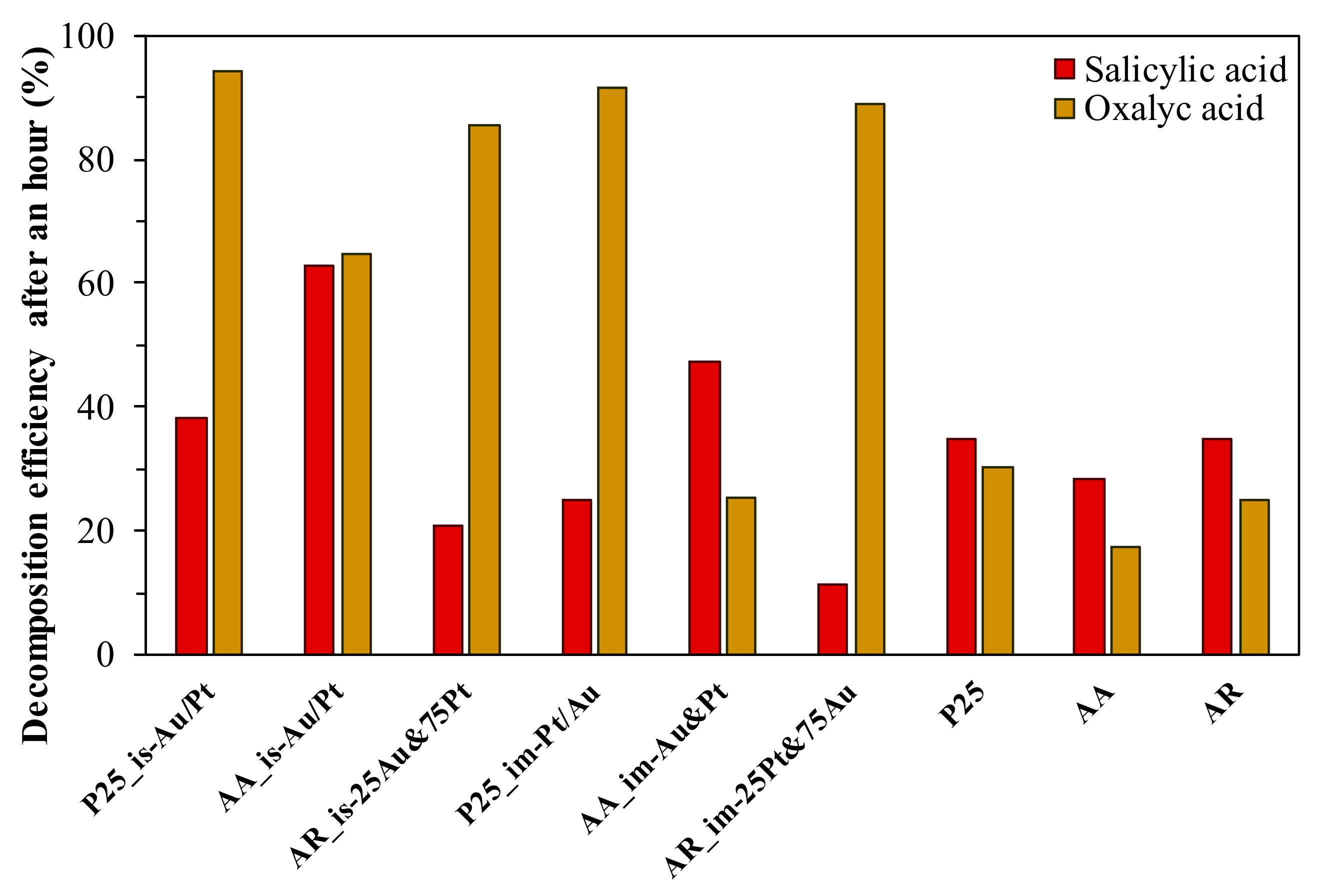
3.4.2. Photodegradation of Salicylic Acid
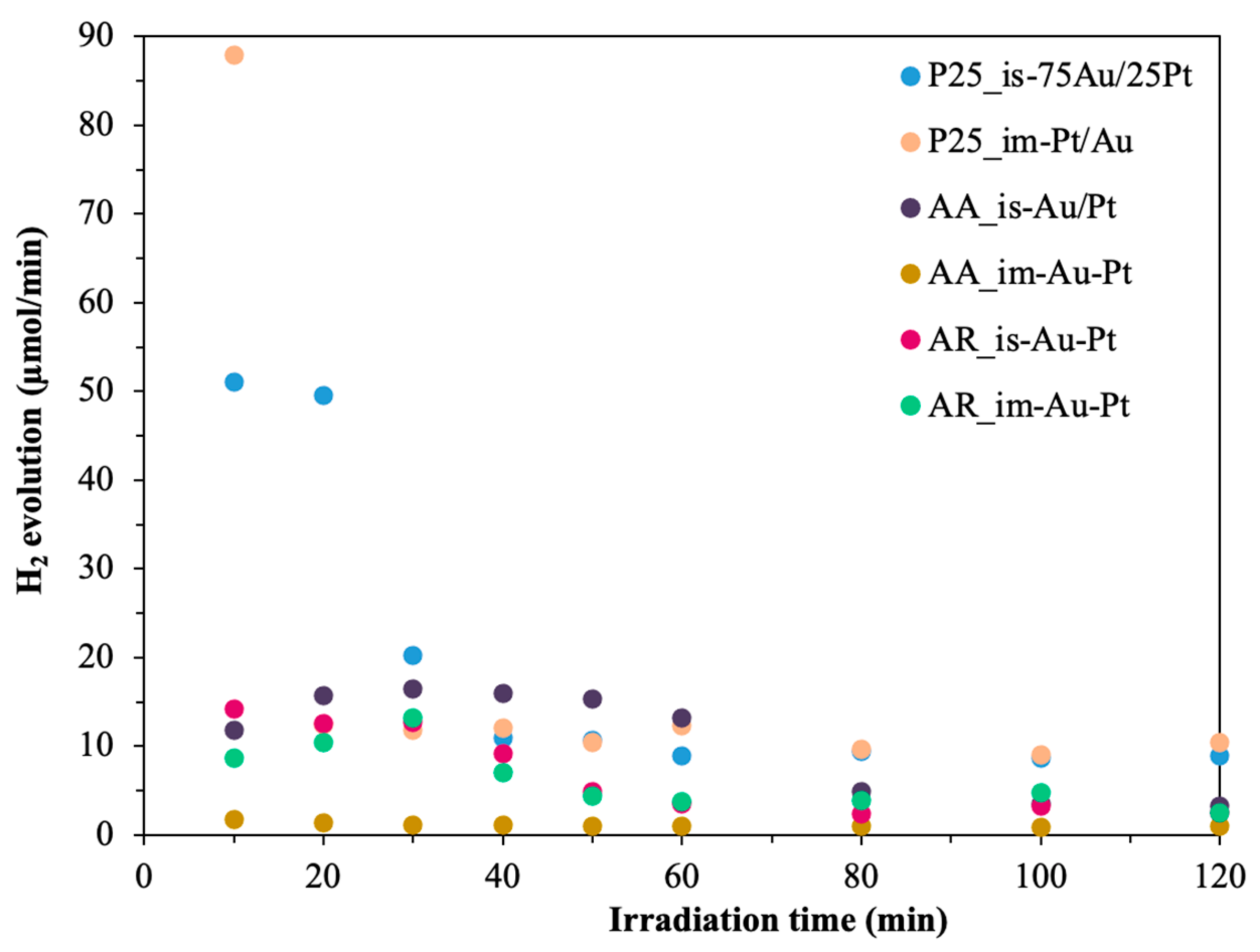
3.4.3. Photocatalytic H2 Production
4. Conclusions
- In terms of the base catalyst, only in the case of the AR-based composites improved the photocatalytic activity the change in the composition for the oxalic acid degradation (AR_im-75Au&25Pt);
- The hydrogen production capacity was improved only in the case of P25-based catalysts when the concentration of the noble metals was changed by the composite named P25_is-75Au/25Pt.
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, L.; Gu, F.; Chan, J.; Wang, A.; Langer, R.; Farokhzad, O. Nanoparticles in Medicine: Therapeutic Applications and Developments. Clin. Pharmacol. Ther. 2008, 83, 761–769. [Google Scholar] [CrossRef] [PubMed]
- Schaming, D.; Remita, H. Nanotechnology: From the ancient time to nowadays. Found. Chem. 2015, 17, 187–205. [Google Scholar] [CrossRef]
- Liu, H.; Song, C.; Zhang, L.; Zhang, J.; Wang, H.; Wilkinson, D.P. A review of anode catalysis in the direct methanol fuel cell. J. Power Sources 2006, 155, 95–110. [Google Scholar] [CrossRef]
- Forster, S.P.; Olveira, S.; Seeger, S. Nanotechnology in the market: Promises and realities. Int. J. Nanotechnol. 2011, 8, 592–613. [Google Scholar] [CrossRef]
- Siegrist, M.; Cousin, M.E.; Kastenholz, H.; Wiek, A. Public acceptance of nanotechnology foods and food packaging: The influence of affect and trust. Appetite 2007, 49, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Xie, B.; Jin, L.; Chen, W.; Li, J. Hydrothermal Synthesis and Responsive Characteristics of Hierarchical Zinc Oxide Nanoflowers to Sulfur Dioxide. J. Nanotechnol. 2016, 2016. [Google Scholar] [CrossRef]
- Zhang, W.X. Nanoscale iron particles for environmental remediation: An overview. J. Nanoparticle Res. 2003, 5, 323–332. [Google Scholar] [CrossRef]
- Rasalingam, S.; Peng, R.; Koodali, R.T. Removal of hazardous pollutants from wastewaters: Applications of TiO2-SiO2 mixed oxide materials. J. Nanomater. 2014, 2014. [Google Scholar] [CrossRef]
- Huang, L.; Peng, F.; Yu, H.; Wang, H. Preparation of cuprous oxides with different sizes and their behaviors of adsorption, visible-light driven photocatalysis and photocorrosion. Solid State Sci. 2009, 11, 129–138. [Google Scholar] [CrossRef]
- Kim, J.; Lee, C.W.; Choi, W. Platinized WO3 as an environmental photocatalyst that generates OH radicals under visible light. Environ. Sci. Technol. 2010, 44, 6849–6854. [Google Scholar] [CrossRef]
- Daneshvar, N.; Salari, D.; Khataee, A.R. Photocatalytic degradation of azo dye acid red 14 in water on ZnO as an alternative catalyst to TiO2. J. Photochem. Photobiol. A Chem. 2004, 162, 317–322. [Google Scholar] [CrossRef]
- Panudda, P.; Ekthammathat, N.; Phuruangrat, A.; Thongtem, S.; Thongtem, T. BiOX (X = Cl, Br, and I) Nanoplates Prepared by Surfactant-Free Microwave Synthesis and Their Photocatalytic Performance. Russ. J. Phys. Chem. A 2018, 92, 2289–2295. [Google Scholar] [CrossRef]
- Gaya, U.I.; Abdullah, A.H. Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: A review of fundamentals, progress and problems. J. Photochem. Photobiol. C Photochem. Rev. 2008, 9, 1–12. [Google Scholar] [CrossRef]
- El Rouby, W.M.A.; Comesaña-Hermo, M.; Testa-Anta, M.; Carbó-Argibay, E.; Salgueiriño, V.; Pérez-Lorenzo, M.; Correa-Duarte, M.A. Au-decorated sodium titanate nanotubes as high-performance selective photocatalysts for pollutant degradation. J. Phys. D Appl. Phys. 2017, 50. [Google Scholar] [CrossRef]
- Dawson, A.; Kamat, P.V. Semiconductor-metal nanocomposites. Photoinduced fusion and photocatalysis of gold-capped TiO2 (TiO2/Gold) nanoparticles. J. Phys. Chem. B 2001, 105, 960–966. [Google Scholar] [CrossRef]
- Yu, C.; Cao, F.; Li, G.; Wei, R.; Yu, J.C.; Jin, R.; Fan, Q.; Wang, C. Novel noble metal (Rh, Pd, Pt)/BiOX (Cl, Br, I) composite photocatalysts with enhanced photocatalytic performance in dye degradation. Sep. Purif. Technol. 2013, 120, 110–122. [Google Scholar] [CrossRef]
- Subramanian, V.; Wolf, E.E.; Kamat, P.V. Catalysis with TiO2/Gold Nanocomposites. Effect of Metal Particle Size on the Fermi Level Equilibration. J. Am. Chem. Soc. 2004, 126, 4943–4950. [Google Scholar] [CrossRef]
- Zhang, H.; Lv, X.; Li, Y.; Wang, Y.; Li, J. P25-graphene composite as a high performance photocatalyst. ACS Nano 2010, 4, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; He, X.; Zhou, B.; Shen, J.; Du, A. Hot electrons coupling-enhanced photocatalysis of super black carbon aerogels/titanium oxide composite. MRS Commun. 2018, 8, 521–526. [Google Scholar] [CrossRef]
- Stroyuk, A.L.; Shvalagin, V.V.; Kuchmii, S.Y. Photochemical synthesis and optical properties of binary and ternary metal-semiconductor composites based on zinc oxide nanoparticles. J. Photochem. Photobiol. A Chem. 2005, 173, 185–194. [Google Scholar] [CrossRef]
- Su, R.; Tiruvalam, R.; Logsdail, A.J.; He, Q.; Downing, C.A.; Jensen, M.T.; Dimitratos, N.; Kesavan, L.; Wells, P.P.; Bechstein, R.; et al. Designer titania-supported Au-Pd nanoparticles for efficient photocatalytic hydrogen production. ACS Nano 2014, 8, 3490–3497. [Google Scholar] [CrossRef] [PubMed]
- Arora, N.; Mehta, A.; Mishra, A.; Basu, S. 4-Nitrophenol reduction catalysed by Au-Ag bimetallic nanoparticles supported on LDH: Homogeneous vs. heterogeneous catalysis. Appl. Clay Sci. 2018, 151, 1–9. [Google Scholar] [CrossRef]
- Chanu, I.; Krishnamurthi, P.; Manoharan, P.T. Effect of Silver on Plasmonic, Photocatalytic, and Cytotoxicity of Gold in AuAgZnO Nanocomposites. J. Phys. Chem. C 2017, 121, 9077–9088. [Google Scholar] [CrossRef]
- Singh, S.; Srivastava, P.; Singh, G. Synthesis, characterization of Co-Ni-Cu trimetallic alloy nanocrystals and their catalytic properties, Part-91. J. Alloys Compd. 2013, 562, 150–155. [Google Scholar] [CrossRef]
- Yan, W.; Tang, Z.; Wang, L.; Wang, Q.; Yang, H.; Chen, S. PdAu alloyed clusters supported by carbon nanosheets as efficient electrocatalysts for oxygen reduction. Int. J. Hydrog. Energy 2017, 42, 218–227. [Google Scholar] [CrossRef]
- Henam Sylvia, D.; Henam Premananda, S.; Thiyam David, S. Green synthesized urchin like Pt/Cu bimetallic photonanocatalysts: Understanding composition effect. Mater. Res. Bull. 2017, 86, 277–286. [Google Scholar] [CrossRef]
- Hampel, B.; Kovács, G.; Czekes, Z.; Hernádi, K.; Danciu, V.; Ersen, O.; Girleanu, M.; Focşan, M.; Baia, L.; Pap, Z. Mapping the Photocatalytic Activity and Ecotoxicology of Au, Pt/TiO2 Composite Photocatalysts. ACS Sustain. Chem. Eng. 2018, 6, 12993–13006. [Google Scholar] [CrossRef]
- Kmetykó, Á.; Mogyorósi, K.; Pusztai, P.; Radu, T.; Kónya, Z.; Dombi, A.; Hernádi, K. Photocatalytic H2 evolution using different commercial TiO2 catalysts deposited with finely size-tailored au nanoparticles: Critical dependence on au particle size. Materials 2014, 7, 7615–7633. [Google Scholar] [CrossRef]
- Scherrer, P. Bestimmung der Grosse und der inneren Struktur von Kolloidterilchen mittels Rontgestrahlen. In Nachrichten von der Gesellschaft der Wissenschaften zu Göttingen, Mathematisch-Physikalische Klasse; Weidmannsche Buchhandlung: Berlin, Germany, 1918; pp. 98–100. [Google Scholar]
- Scarpelli, F.; Mastropietro, T.F.; Poerio, T.; Godbert, N. Mesoporous TiO2 Thin Films: State of the Art. In Titanium Dioxide—Material for a Sustainable Environment; Yang, D., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Kovács, G.; Fodor, S.; Vulpoi, A.; Schrantz, K.; Dombi, A.; Hernádi, K.; Danciu, V.; Pap, Z.; Baia, L. Polyhedral Pt vs. spherical Pt nanoparticles on commercial titanias: Is shape tailoring a guarantee of achieving high activity? J. Catal. 2015, 325, 156–167. [Google Scholar] [CrossRef]
- Baia, L.; Vulpoi, A.; Radu, T.; Karácsonyi, É.; Dombi, A.; Hernádi, K.; Danciu, V.; Simon, S.; Norén, K.; Canton, S.E.; et al. TiO2/WO3/Au nanoarchitectures’ photocatalytic activity “from degradation intermediates to catalysts’’ structural peculiarities" Part II: Aerogel based composites—fine details by spectroscopic means. ” Appl. Catal. B Environ. 2014, 148–149, 589–600. [Google Scholar] [CrossRef]
- Murphy, A.B. Band-gap determination from diffuse reflectance measurements of semiconductor films, and application to photoelectrochemical water-splitting. Sol. Energy Mater. Sol. Cells 2007, 91, 1326–1337. [Google Scholar] [CrossRef]
- Ohtani, B.; Prieto-Mahaney, O.O.; Li, D.; Abe, R. What is Degussa (Evonic) P25? Crystalline composition analysis, reconstruction from isolated pure particles and photocatalytic activity test. J. Photochem. Photobiol. A Chem. 2010, 216, 179–182. [Google Scholar] [CrossRef]
- Xue, J.; Ma, S.; Zhou, Y.; Zhang, Z.; He, M. Facile photochemical synthesis of Au/Pt/g-C3N4 with plasmon-enhanced photocatalytic activity for antibiotic degradation. ACS Appl. Mater. Interfaces 2015, 7, 9630–9637. [Google Scholar] [CrossRef] [PubMed]
- Ribao, P.; Rivero, M.J.; Ortiz, I. TiO2 structures doped with noble metals and/or graphene oxide to improve the photocatalytic degradation of dichloroacetic acid. Environ. Sci. Pollut. Res. Int. 2017, 12628–12637. [Google Scholar] [CrossRef]
- Bian, Z.; Tachikawa, T.; Zhang, P.; Fujitsuka, M.; Majima, T. Au/TiO2 superstructure-based plasmonic photocatalysts exhibiting efficient charge separation and unprecedented activity. J. Am. Chem. Soc. 2014, 136, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Mohamed, H.H.; Dillert, R.; Bahnemann, D. Kinetics and mechanisms of charge transfer processes in photocatalytic systems: A review. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 263–276. [Google Scholar] [CrossRef]
- Regazzoni, A.E.; Mandelbaum, P.; Matsuyoshi, M.; Schiller, S.; Bilmes, S.A.; Blesa, M.A. Adsorption and photooxidation of salicylic acid on titanium dioxide: A surface complexation description. Langmuir 1998, 14, 868–874. [Google Scholar] [CrossRef]
- Guo, Q.; Zhou, C.; Ma, Z.; Yang, X. Fundamentals of TiO2 Photocatalysis: Concepts, Mechanisms, and Challenges. Adv. Mater. 2019, 31, 1–26. [Google Scholar] [CrossRef] [PubMed]


| Reduction Approach | Au/Pt (eV) | Pt/Au (eV) | Au&Pt (eV) | |||
|---|---|---|---|---|---|---|
| Synthesis strategy | P25_is | AA_is | P25_im | AA_im | AR_is | AR_im |
| 0.25 Au 0.75 Pt | 2.52 | 3.19 | 2.65 | 3.16 | 2.93 | 2.95 |
| 0.50 Au 0.50 Pt | 2.57 | 3.13 | 2.67 | 3.17 | 2.89 | 2.91 [27] |
| 0.75 Au 0.25 Pt | 2.61 | 3.12 | 2.76 | 3.16 | 2.94 | 2.96 |
| Au | 2.47 | 3.15 | 2.22 | 3.17 | 2.88 | 2.88 |
| Pt | 2.75 | 3.19 | 2.54 | 3.15 | 2.82 | 2.93 |
| P25 | AA | AR | ||||
| 3.04 | 3.24 | 2.99 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hampel, B.; Baia, L.; Hernadi, K.; Pap, Z. The Influence of the Ratio of Au and Pt Nanoparticles in Ternary Composites with TiO2. Metals 2021, 11, 628. https://doi.org/10.3390/met11040628
Hampel B, Baia L, Hernadi K, Pap Z. The Influence of the Ratio of Au and Pt Nanoparticles in Ternary Composites with TiO2. Metals. 2021; 11(4):628. https://doi.org/10.3390/met11040628
Chicago/Turabian StyleHampel, Boglárka, Lucian Baia, Klara Hernadi, and Zsolt Pap. 2021. "The Influence of the Ratio of Au and Pt Nanoparticles in Ternary Composites with TiO2" Metals 11, no. 4: 628. https://doi.org/10.3390/met11040628
APA StyleHampel, B., Baia, L., Hernadi, K., & Pap, Z. (2021). The Influence of the Ratio of Au and Pt Nanoparticles in Ternary Composites with TiO2. Metals, 11(4), 628. https://doi.org/10.3390/met11040628









