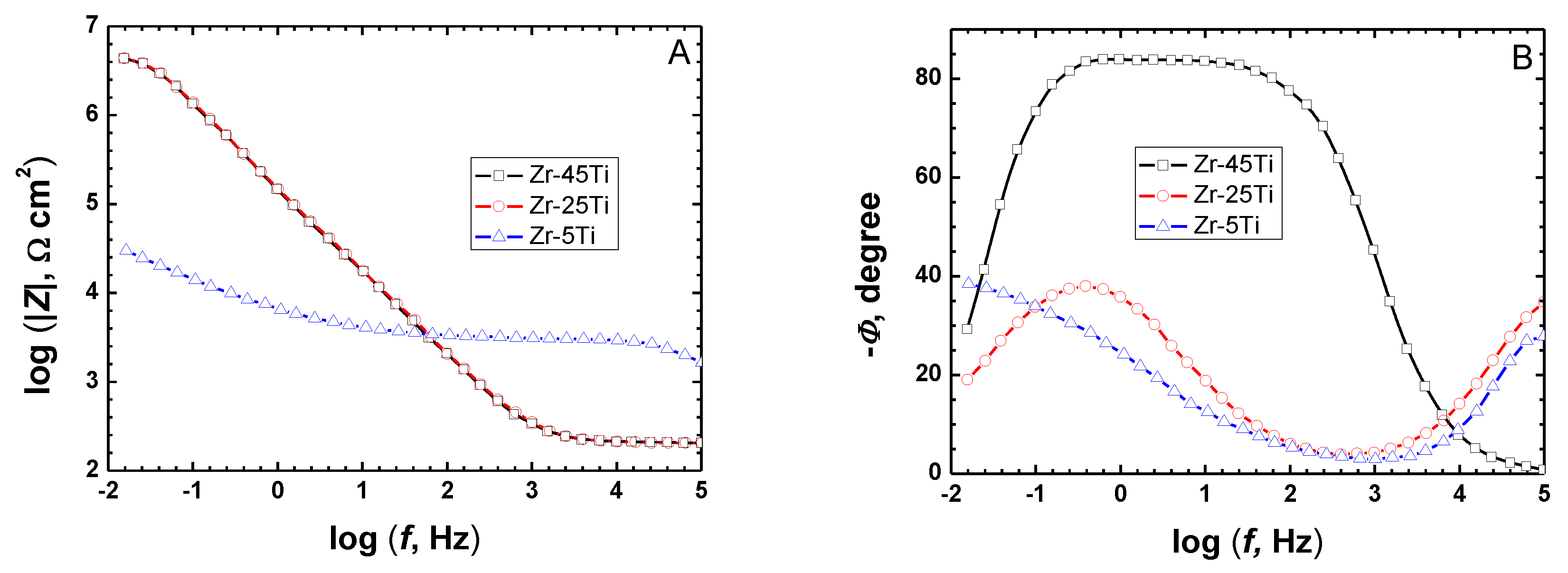
3.2. In Vitro Monitoring of the Formation of Passivating Surface Layers on Zr-Ti Alloys
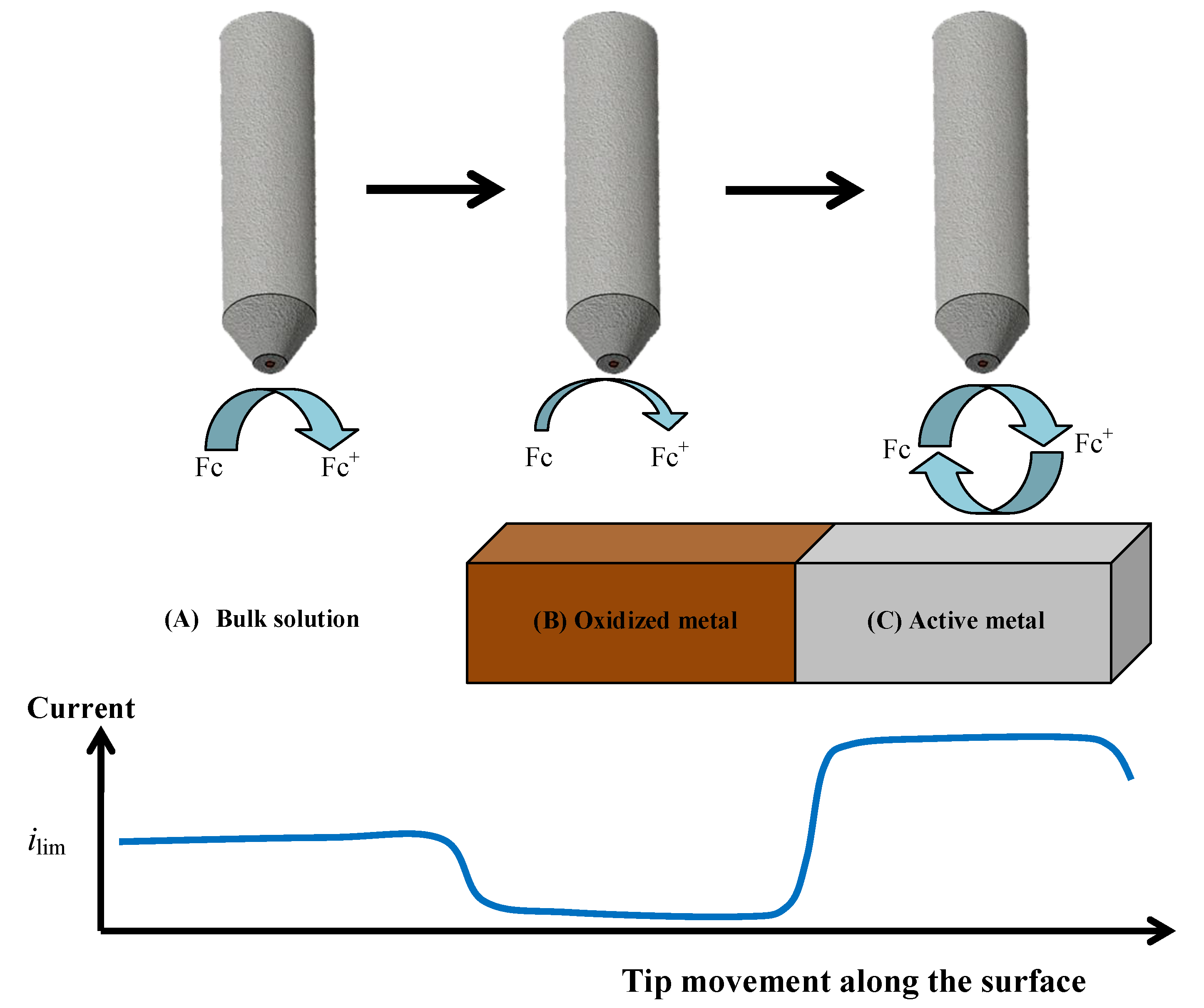
The formation of the oxide surface layers on Zr-Ti alloys upon immersion in simulated physiological solution was monitored by means of scanning electrochemical microscopy (SECM) operated in the amperometric feedback mode. In this way, the evolution of the electrochemical properties of these layers could be monitored in real time. Ferrocene-methanol was added to the Ringer physiological solution as redox mediator in order to monitor its diffusion-limited oxidation at the Pt microelectrode tip, which results in the limiting current,
ilim, sensed in bulk solution (see
Figure 3A). When this reaction is performed in the proximity of the flat surface of the oxidized Zr-Ti alloy sample, either the material will block the diffusion of the redox mediator towards the tip (that is, negative feedback at an insulating substrate, see
Figure 3B), or regeneration of the redox mediator will occur if the material undergoes an electron transfer reaction (i.e., positive feedback at an electroactive substrate, see
Figure 3C) [
40]. Thus, respectively smaller or bigger faradaic currents will be recorded at the tip placed in the proximity of the investigated material compared to the value monitored in the bulk of the electrolyte. When irreversible electron transfer, under kinetic control, is attained at the surface, the response in SECM approach curves depicts an intermediate behavior between positive and negative feedback, depending on the geometry of the electrode and the electron transfer rate constant. Finally, in order to avoid the potential risk of fouling effects in the Pt probe due to components of the electrolyte, the simpler Ringer’s solution was chosen instead of PBS, as to mimic the intracellular environment, acidified to pH 3 thereby simulating inflammatory conditions. The measurements were performed in the naturally aerated solution at 37 °C.
Zr-Ti alloys subjected to thermal oxidation in the optimum temperature range were thus selected for this microelectrochemical characterization during immersion in simulated physiological solution.
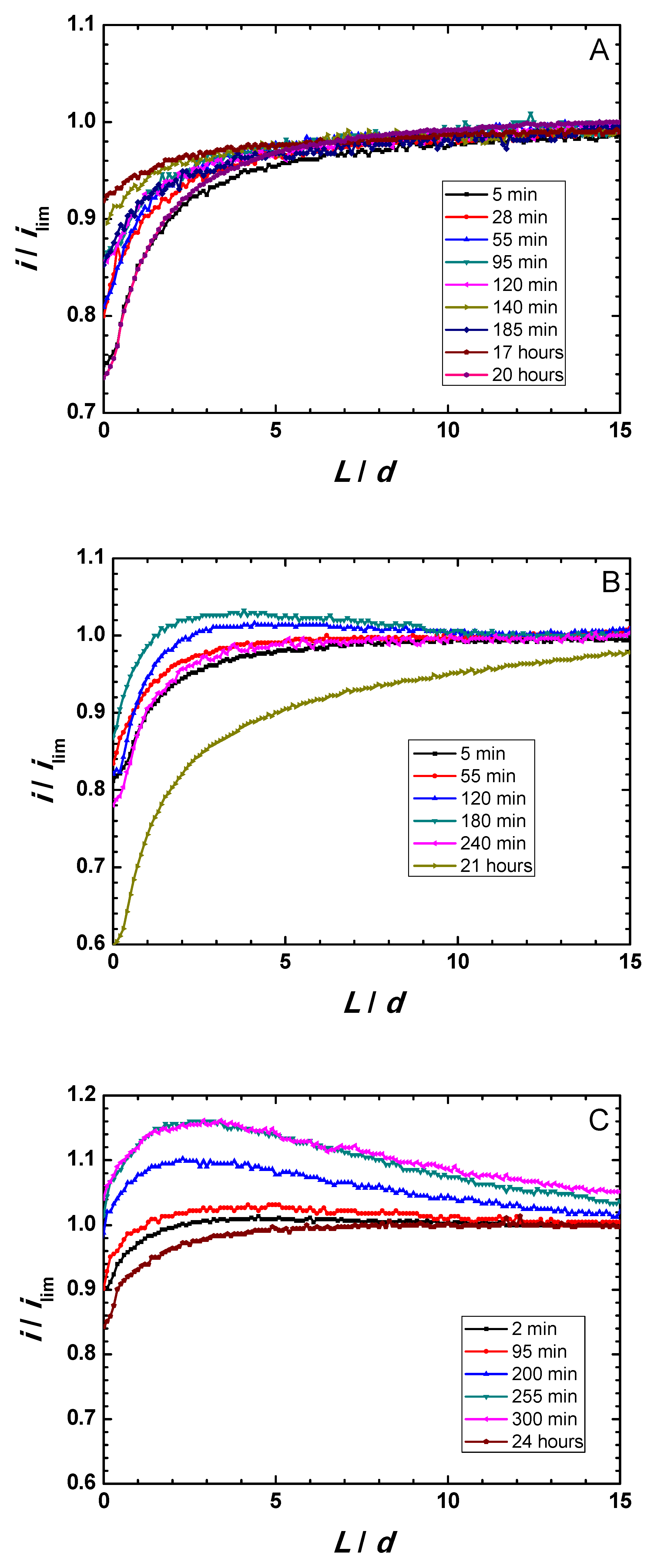
Figure 4A–C displays Z-approach curve results for the three thermally-oxidized Zr-Ti alloys measured during 1-day immersion in acidified Ringer’s solution.
Y-axis reflects the normalized current measured at each location, that is, the measured current divided by the current value registered in bulk solution. The
X-axis gives the normalized vertical position,
L, namely the
Z position divided by the Pt microdisk diameter (
d = 10 µm). Results show that the behavior of the surfaces was primarily insulating, although dynamic evolution occur in all samples. The measured currents were observed to slightly increase within the first 3–5 h immersion, attaining maximum ranges after 140, 180, and 300 min immersion for the Zr-5Ti, Zr-25Ti, and Zr-45Ti specimens, respectively. After sufficiently long exposure (i.e., 20–24 h immersion), the surfaces became more insulating, directly related to thickening of the oxide-hydroxide layer formed on the surface of the alloys exposed to the aqueous environment. This feature is clearly observed from the general trend towards smaller normalized feedback currents measured at the tip located in the vicinity of the substrate (i.e., for small normalized tip–substrate separations) occurring for the longest immersion times in all cases.
These observations also differentiate between the three Zr-Ti alloys in the early stages of immersion. Thus, whereas normalized currents smaller than one were always measured for the Zr-5Ti alloy (cf.
Figure 4A), surfaces promoting higher tip currents, presumably due to partial conductivity/electron transfer efficiency, were observed for Zr-25Ti and Zr-45Ti (see the normalized currents higher than one in
Figure 4B,C at the beginning of the experiment). Despite the observation of normalized currents greater than one for the Ti-rich alloys, it must be noticed that the
Z-approach curves do not display the actual positive feedback behavior characteristic of conductive surfaces, as there was not a steady growth of the normalized current as the tip approached the substrate, ideally increasing towards infinite for normalized tip–substrate separations tending to zero. Instead, a mixed behavior between those corresponding to positive and negative feedback effects is observed. That is, as the tip traveled towards the substrate from the bulk of the electrolyte, the proximity of the surface was first noticed from the increase in the normalized current related to partial conductivity, and then eventually changed towards smaller normalized currents in the vicinity of the substrate. In addition, probe-approach curves measured after 3–5 h immersion for Zr-45Ti depict a continuous increment of the tip current at relatively large distances (i.e., 10 to 15 times the diameter of the Pt microdisk), with the same slope maintained until ca.
L = 4, which has been previously reported with the same alloys [
19], but can hardly be ascribed or modeled considering exclusively feedback effects. Features eventually influencing the tip current must involve the generation of electro-active species diffusing towards the probe, either metal cations or complexes, or redox species catalytically formed at the surface of the metal following reactions of components of the electrolyte. The former is very unlikely, not only because of the stability of the passive films, but also considering that no titanium or zirconium-containing ion or complexes are expected to produce a measurable electrochemical signal at the given tip potential and weak acidic aqueous solution. However, (photo)catalytic reactions involving the generation of H
2 or H
2O
2, oxidizable at the tip potential, cannot be discarded. Further evidence must be attained in order to investigate this effect, which otherwise should not compromise the surface stability.
The heterogeneity of the surface response was evaluated by conducting 2-D scans over a fixed random area of 500 × 500 µm
2 dimensions for each specimen during the first 24 h.
Figure 5 displays representative area scans obtained during exposure of the materials to the acidified Ringer′s solution. Notice that green color in all scans represent normalized current equal to 1, that is, reproducing the same
ilim current value attained for the diffusion-limited oxidation of the redox mediator in the bulk solution. Hence, normalized current values greater than one (e.g., yellow and red colors) indicate higher surface reactivity (i.e., positive feedback), whereas the blue color corresponds to more insulating properties (i.e., negative feedback).
The behavior of the three specimens dynamically changed during the first hours of immersion, moving towards more insulating character when samples were exposed over 20 h. Zr-5Ti showed smoother current variations throughout the scanned area, whereas higher Ti content depicted regions with distinct reactivity. The response resembled closely that previously observed in the same solution at 25 °C [
20]. However, the behavior differed with the addition of higher Ti content at the simulated body temperature in this work. Scans performed on Zr-25Ti exhibited one region with rather insulating properties in all scans (close to the drawn origin of coordinates), and only the first measurement showed a nearby area with higher reactivity, which progressively deactivated in following scans. Conversely, it was observed that the Zr-45Ti surface presented single locations with much higher activity towards electron exchange with the redox mediator. These spots contrasted with the main insulating properties exhibited by the surface, in particular after 24 h immersion.
It must be noticed that the conductivity characteristics in the feedback mode reflect the efficiency of the electron transfer reaction at the very site examined by the microelectrode for
L/
d → 0, whereas wider screening of the surface occurs for longer distances. Then, normalized currents bigger than one in the
Z-approach curves of
Figure 4B,C indicate a surface that is not an efficient insulator, yet this phenomenon can also be explained considering the single active locations seen in the scans taken over the Zr-45Ti (cf.
Figure 5). As result, from the observation of the
Z-approach curves displayed in
Figure 4A–C it can be concluded that the surface films formed on Zr-5Ti exhibit low efficiency for electron transfer even at short exposures to the electrolyte, and subsequent thickening of the bilayer oxide film leads to even more insulating characteristics. In contrast, the richer Ti alloys require exposure to the electrolyte to develop the surface oxide layers with insulating characteristics, namely 1 h and approximately 1 day for Zr-25Ti and Zr-45Ti, respectively. The surface film developed on the Zr-25Ti surface would be the most insulating, with heterogeneities mostly depicting inert areas. Conversely, the oxide layer formed on the Zr-45Ti was partially conductive at single locations, suggesting that the passive character could be locally broken to facilitate electron donation.
These SECM observations, depicting electrochemical heterogeneity and local passivity breakdown on the surface of Zr-45Ti, apparently contradict the higher corrosion resistance exhibited by the Ti-enriched alloy from conventional averaging electrochemical (impedance) measurements. The developed oxide layer has shown to be equally or more inert in thermally oxidized Zr-45Ti, compared to Zr-25Ti and Zr-5Ti, when exposed to phosphate buffer saline solution at 37 °C (this work), neutral Ringer’s solution at 37 °C [
31], and Ringer’s acidified solution (pH 3) at 37 °C [
20]. However, SECM results obtained in the feedback mode reflect the ability of the sample to transfer electrons via heterogeneous electron exchange reactions at its surface, rather than the release of metal cations from the anodic degradation of the material. That is, the origin of the electrons collected by the ferrocenium ions does necessarily stem from the anodic dissolution of Ti or Zr. Instead, ferrocenium ions may promote the formation of oxides, thus accelerating the thickening of the oxide layer, providing that the single point location at which tip current increases behaves as electrically conductive and kinetically active for the heterogeneous electron donation. This thickening process results in a final surface state which behaves primarily insulating after 24 h immersion, supporting the formation of a protective layer. In addition, it is feasible that, if the excitation of electrons from the valence band to the conduction band occurs in such semiconductor material surfaces under illumination, electrons may be either collected following water splitting mechanism, or by the ferrocenium ions instead. All these early-stage phenomena might occur heterogeneously during the development of the passive layer, attaining homogeneous insulating and protective character after sufficiently long immersion time. Given the presence of redox proteins in the real physiological media [
41,
42], the ability of a metal implant to exchange electrons on the surface, and eventually promote the alteration of such biomolecules during the early stages of the implantation procedure, is regarded as critical and needs to be taken into consideration in regard to the biocompatibility of a newly proposed biomaterial.