Simple Summary
Eriophyoid mites are an ancient lineage of highly specialized plant parasites. Despite their minute size, certain species produce large protective silk nests. Using an integrative approach combining field observations, detailed morphology, and molecular data, we investigated web-spinning mites associated with the leaves of Mangifera indica (mango) in Asia, Africa, and North America and Lepisanthes rubiginosa (mertajam) in Vietnam. Our results demonstrate that silk-web production evolved convergently in phylogenetically distant lineages. We also show that on mango, the species Cisaberoptus kenyae and Aceria aegyptindicae frequently co-occur and likely cooperate in spinning shared webs. Finally, we provide evidence for a phylogenetic relationship linking the mango-associated web-spinners to the mite subfamily Cecidophyinae.
Abstract
Several lineages within the Eriophyoidea, an ancient group of phytoparasitic acariform mites, produce large protective silk webs on plant surfaces. This study examined the diversity and phylogeny of web-spinning eriophyoids associated with the leaves of Mangifera indica (mango) in Asia, Africa, and North America and Lepisanthes rubiginosa (mertajam) in Vietnam. We report, for the first time, silk-producing structures in Aceria aegyptindicae and Aculops knorri, as well as the presence of tibial seta l’ I and a lobular unpaired prosomal gland in Cisaberoptus kenyae. Molecular and morphological data indicate female dimorphism in A. knorri and reveal cryptic diversity within Cisaberoptus and mango-associated Aceria. The web-spinning species C. kenyae and Ac. aegyptindicae form a strongly supported clade and frequently coexist in syntopy on mango leaves, likely co-producing silk web. Phylogenetic analyses of COI and 28S genes place this clade as sister to Cecidophyinae, while grouping Aberoptus and A. knorri with Anthocoptini and Aceriini. These results demonstrate convergence, cryptic speciation, and unexpected mutualism in web-spinning eriophyoids, providing a framework for future research on the evolution of silk producing organs in this economically important mite group.
1. Introduction
The superfamily Eriophyoidea is an exceptionally ancient lineage of highly host-specific and morphologically simplified phytoparasitic acariform mites [1,2]. Phylogenetic reconstructions integrating fossil evidence and plant evolution suggest that, following their Devonian origin ~376 (314–441) Ma, eriophyoid mites underwent a transitory phase associated with the roots of early arborescent plants, co-evolved with now-extinct plant lineages, and later colonized gymnosperms and angiosperms [3,4,5,6]. The estimated divergence time of the crown group of Eriophyoidea, ~305 (260–364) Ma, closely aligns with the emergence of early conifers [6] and supports the hypothesis that gymnosperms were the primary host group for the radiation of extant eriophyoid lineages [3,7].
Current evidence indicates that the morphological evolution of Eriophyoidea is characterized by extensive convergence and homoplasy [2,8,9,10]. Key patterns include widespread morphological modifications like setal loss, dorsal opisthosomal transformations including the formation of ridges, furrows and pseudotagmata, structural variety of tarsal empodia, and leg segment fusion [1,11,12]. This is contrasted by divergence at higher taxonomic levels, defined by specific chaetotaxy of the prodorsal shield and topography of external and internal genitalia [2,13,14]. Speciation is frequently cryptic and driven by host shifts, which typically occur within a single plant genus or family, leading to complexes of subtly differentiated species sharing a similar design of prodorsal shield [9,15,16,17]. As a result, species delimitation usually requires the integration of detailed morphometric data with barcode gene comparisons [18,19,20,21,22,23].
The ability to produce large film-like silk webbing on plant surfaces (web-spinning) has long been considered an exotic adaptation that evolved in a few eriophyid taxa [24]. A series of recent publications has documented the presence of putative silk-producing structures in multiple unrelated lineages of Eriophyoidea, suggesting that this capability is more widespread [25,26,27,28,29]. Although the anatomy of gall mites is relatively well-studied [30], the detailed structure of their silk-producing organs has been examined in only a single species, Aberoptus schotiae Chetverikov et al. 2023 [27]. Current hypotheses for silk evolution and function in eriophyoid mites suggest that: (1) the silk-secreting system evolved from a symplesiomorphic complex of internal organs, including the anal glands, hindgut, and associated cuticular sacs; (2) this system is well-developed in taxa that produce large webs but reduced yet still functional in other species; (3) the web nests serve to regulate the micro-environment, protect mites from predators and pathogenic fungi, prevent dislodgement by rain, maximize occupation of the leaf surface, and isolate food resources from competitors; and (4) beyond web formation, the silk secretions of the anal glands may also enable attachment to plant surfaces, aid in ballooning during aerial dispersal, mark selected sites on the host, and facilitate intraspecific signaling to help conspecifics locate each other.
To date, ten species from four genera in the subfamilies Aberoptinae (Aberoptus Keifer, 1951 and Cisaberoptus Keifer, 1966), Phyllocoptinae (Anthocoptini: Aculops Keifer, 1966) and Eriophyinae (Aceriini: Aceria Keifer 1944) of the family Eriophyidae have been shown to produce large webs [24,27]. Nine of them are associated with arboreal host plant genera of the families Fabaceae, Ochnaceae, Anacardiaceae, and Sapindaceae (all within the eudicot superclade “rosids”), exhibiting pantropical and subtropical distributions and growing naturally across the Americas, Africa, Asia, and Australasia within frost-free, warm climates [27]. One species, Aceria gersoni Manson 1984, was described from the North Island of New Zealand living under webbing on the underside of the pinnules on the Rough tree fern, Dicksonia squarrosa (G. Forst.) Sw. (Polypodiopsida: Cyatheales: Dicksoniaceae) [31].
The genus Aberoptus comprises seven described species associated with plants in the Fabaceae (5 spp.), Ochnaceae (1 sp.), and Anacardiaceae (1 sp.), and reported from Brazil, South Africa, Taiwan, and Samoa. Female dimorphism has been confirmed in at least three species of Aberoptus (Ab. cerostructor Flechtmann 2001, Ab. inusitatus (Britto et al. 2008), and Ab. schotiae Chetverikov et al. 2023) through experimental studies [25,32] and genetic analysis [27]. In these species, the web-spinning form is specialized morphologically, featuring greatly modified swollen legs with heteromorphic empodia I and II, a spatulate structure on tarsus I, and two shortened terminal leg segments. The alternative female form resembles a typical Aceria, though it shares some general characteristics with the web-spinning form, such as swollen legs and a similar body shape.
The monotypic genus Cisaberoptus is well-known for its web-spinning pest species, C. kenyae Keifer, 1966, infesting mango (Anacardiaceae: Mangifera indica L.). It is present in most mango-cultivating regions worldwide, including southern Europe [33,34,35]. This species exhibits distinctive morphology, including a spatulate terminal palp segment forming a sucker-like structure; swollen legs I and II with large, pad-like empodia bearing a large number of rays (15–18); partially fused genua and femora; shortened tarsi and tibiae; and a putatively absent tibial seta l′ I. However, the functional correlation of this morphology with web-spinning behavior remains unknown. Although no specialized silk-producing structure has been described in Cisaberoptus, three hypotheses on the mechanism of silk production have been proposed: (1) regurgitation of intestinal content, (2) secretion from the salivary glands, or (3) production by anal glands and deposition via the hindgut [24,27,35,36]. Studies from Africa (Sudan, Egypt), Asia (Thailand), and South America (Brazil) have consistently reported Aceria-like specimens within the web nests of C. kenyae [34]. Researchers from Brazil interpreted these as a protogyne form of C. kenyae [37], whereas those working with Egyptian material described them as a new inquiline species, Aceria aegyptindicae Elhalawany et al. 2021 [34].
Aculops knorri Keifer, 1976 was described from Thailand, where it was found beneath a web coating on the leaves of Lepisanthes rubiginosa (Blume) Leenh. (Sapindaceae), mertajam, in tropical Asia and northern Australia [38]. Although serological tests confirmed the webbing as true proteinaceous silk [39], detailed anatomical data explaining how A. knorri produces silk are completely lacking. Apart from slightly swollen legs (primarily due to thickened femora), no specialized morphology correlated with its web-spinning ability has been reported. Taxonomically, A. knorri fits well within the tribe Anthocoptini (Eriophyidae: Phyllocoptinae). The species’ author noted that in the absence of specialized morphology it “… can only be assigned to the ‘waste-basket’ genus Aculops” [38]. This is a large anthocoptine genus comprising ~200 described species, which appears polyphyletic in molecular studies [9,40]. Remarkably, Aceria is another ‘waste-basket’ genus in Eriophyoidea, containing over 1000 species. Earlier authors proposed synonymies among Aceria, Cisaberoptus, and Aberoptus [11,32,36,37]; however, most recent studies consider them three valid genera [26,27,32,35].
No molecular data on web-spinning species of Aceria or Aculops are currently available. A single phylogenetic study focused on Aberoptus and Cisaberoptus recovered three 28S sequences of these genera in unrelated clades within Eriophyidae [27].
In this study, we (1) describe our field observations of populations of web-spinning mites associated with L. rubiginosa and M. indica, (2) investigate their genetic diversity using COI and 28S sequences, (3) report novel morphological characters observed in these mites, (4) test the conspecificity of C. kenyae and Ac. aegyptindicae, and (5) assess the monophyly of the ecological group of web-spinning eriophyid taxa based on an expanded dataset.
2. Materials and Methods
Collecting and slide-mounting. Web-spinning eriophyoid mites were collected from two eudicot tree species from Vietnam, South Africa, and USA and used for morphological investigation and genotyping (Table 1, Figure 1). The web-coated leaves of M. indica (Anacardiaceae) and L. rubiginosa (Sapindaceae) were examined under a stereo microscope and the mites were collected from under the web using a minuten pin. Some mites were slide-mounted in Hoyer’s medium [41] and cleared on a heating block at 95 °C for 30–40 s (for obtaining temporary slides with partially cleared mites) or for 4–5 h (for obtaining permanent slides of fully cleared mites). Some mites were mounted alive in glycerin for investigating their anatomy in vivo. The rest of the mites were stored in Eppendorf tubes filled with 96% ethanol in a freezer (−25 °C) or kept alive on leaves in a refrigerator (+4 °C) for further examination.

Table 1.
Material from Lepisanthes rubiginosa and Mangifera indica used for DNA extraction (N—number of mites used for extracting DNA).
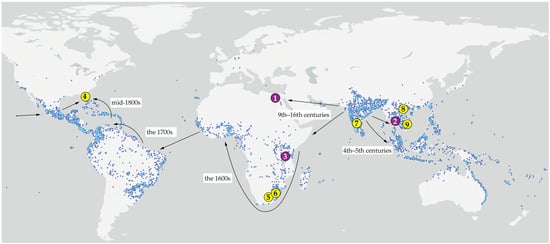
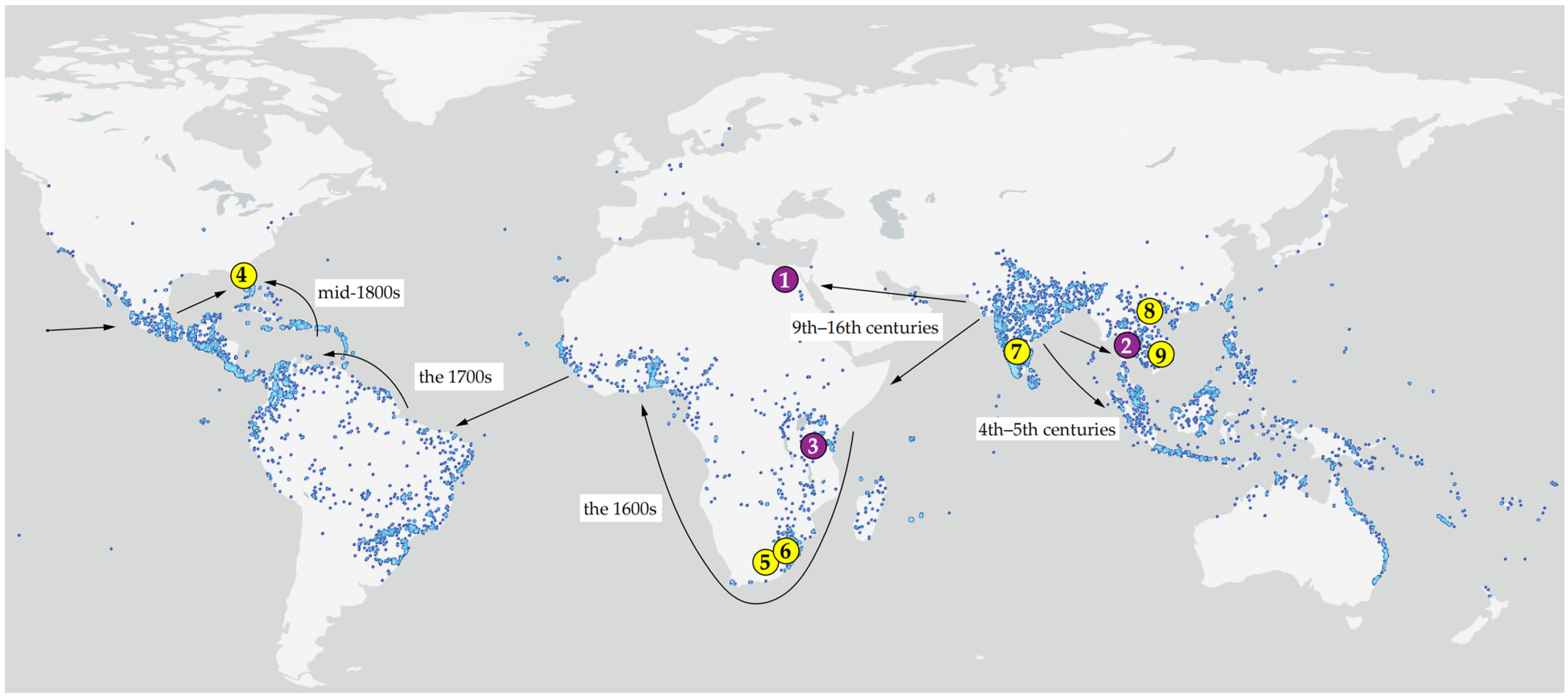
Figure 1.
Type localities (1–3, purple) and source localities of the studied material (4–9, yellow) of web-spinning eriophyoid mites from Mangifera indica (1, 3–8) and Lepisanthes rubiginosa (2, 9), mapped onto the global distribution of mango based on GBIF [42] Occurrence Download. Available online: https://www.gbif.org/species/3190638 (accessed on 10 January 2026). Arrows indicate inferred routes and chronology of the human-mediated migration of mango following its domestication in India c. 4000 BP (adapted from [43]). 1—Type locality of Aceria aegyptindicae; 2—Type locality of Aculops knorri; 3—Type locality of Cisaberoptus kenyae; 4, 5, 6, 7, 8—Collecting sites of mango mites in Florida (4), South Africa (5, 6), India (7), and Vietnam (8); 9—collecting site of Aculops knorri in Vietnam. Note: Specimens from locality 7 (India) were not available for examination; the sequence MW491351 was included in the molecular analysis. Additional information is given in the Table 1.
Mite populations used for microscopy observations. Two populations (1 and 2, see below) of the web-spinning mites from Vietnam were used for stereo microscopy, phase contrast (PC), and differential interference contrast (DIC) light microscopy (LM), while additional populations from Tam Dao (Vietnam), Mtunzini (South Africa), and Florida (USA) (Table 1) were examined only under PCLM and were used for morphological comparison and molecular phylogenetic analyses.
Population 1: A. knorri under a web coating on leaves of L. rubiginosa collected in early March 2024 in a small forest between road QL29 and the coast of Vung Ro Bay, approximately 80 km north of Nha Trang City in Hoa Xuan Commune, Dak Lak Province, 12°52′43.8″ N 109°25′20.7″ E.
Population 2: Sympatric C. kenyae and Aceria spp. under a web coating on leaves of approximately 40-year-old M. indica trees growing near the entrance of the Plant Protection Research Institute collected in mid-February 2023 in Hanoi, 21°04′18.8″ N, 105°46′30.2″ E.
Morphological measurements and comparison. The external morphology of the slide-mounted specimens was examined using conventional light microscopy (PC LM and DIC LM) with a Leica DM2500, and via confocal laser scanning microscopy (CLSM) using previously described methodologies and equipment [18,44]. For comparative analysis, morphometric data for A. knorri, C. kenyae, and Aceria spp. from mango were compiled from the literature [34,37,38,45] and supplemented with new measurements from populations 1 and 2 (described above). The terminology for eriophyoid morphology and the classification scheme for Eriophyoidea adhere to [1,11].
DNA extraction, PCR, and sequencing. For DNA extraction, from one to eight mite specimens were crushed separately with a fine pin in a 2 μL drop of sterile water on a cavity well microscope slide. Each drop was pipetted into a thin-walled PCR tube with 25 μL of 6% solution of Chelex® 100 Resin Bio Rad before being heated three times (5 min at 95 °C) in a thermostat with intermediate short vortexing. The solution above the Chelex® granules was used as the DNA template for PCR to amplify the fragments of the nuclear 28S rDNA and mitochondrial COI genes (Table 1). For the PCR and sequencing, we applied reported protocols and primers [29]. Sequences were obtained using BigDye Terminator v.3.1 chemistry (Applied Biosystems, Foster City, CA, USA) and a 3500xl Genetic Analyzer (Applied Biosystems). Trace files were checked and edited using GeneStudioTM Professional 2.2.0.0 (https://genestudio-pro.software.informer.com (accessed on 3 April 2023)). Additionally, we obtained a new COI sequence (PX794729) of Ab. schotiae using previously extracted DNA of the mites from the type population [27]. In order to test the genetic identity of the “narrower” and “wider” specimens from population 1 of A. knorri from L. rubiginosa (see Section 3.1.1 and Section 3.2.1), we obtained four DNA isolates representing only “wider specimens” (isolates d633 and d1057), only “narrower specimens” (d1059) and their mixture (d712, Table 1).
Analysis of COI and 28S sequence diversity. Prior to this study, the only available sequences for C. kenyae were a 28S sequence (KT070272; 1014 bp) from South Africa [14] and a single unpublished COI sequence (MW491351; 634 bp) from specimens collected by P. Dyamanagouda on 10 October 2019 in Coimbatore, India (Tamil Nadu Agricultural University, Paddy Breeding Station; 11°00′ N, 76°91′ E), from under a web coating on leaves of M. indica. Both reference sequences were aligned with our newly generated sequences (Table 1), trimmed to equal length, and analyzed using the Kimura 2-parameter (K2P) model in MEGA7 [46] to estimate evolutionary divergence.
Molecular phylogenetic analyses. Approximately 3400 COI sequences of Eriophyoidea were available in GenBank as of 15 January 2026. Most are short fragments of ~650 bp obtained with classical Folmer’s primers [47] or their variants, alongside a smaller series of ~1200 bp sequences amplified with primers developed by Klimov et al. [48] and complete COI sequences derived from recent mitogenomic studies [40,49]. Phylogenetic reconstructions using all available COI sequences from GenBank did not converge and yielded biologically inconsistent topologies, likely due to the high proportion of short fragments with insufficient phylogenetic signal.
To overcome this issue, we performed a BLAST+2.17.0 search with sequence PX794735 (C. kenyae, 1196 bp) against Eriophyoidea in GenBank, downloaded all sequences with query coverage 90–100%, removed erroneous sequences [49] and duplicates, excluded sequences from Phytoptidae and Nalepellidae (except for the outgroups specified below), and supplemented the selection with new sequences (Table 1). The resulting curated sequences were codon-aligned using MUSCLE in MEGA 7 [46] and translated to amino acids to screen for stop codons. The final COI dataset included 193 sequences and 456 amino acid positions.
To construct the 28S dataset, we conducted a BLAST search with sequence PX789905 of C. kenyae against GenBank entries for Eriophyidae, downloaded all sequences with 55–100% query coverage, and merged them with our novel sequences (Table 1) and outgroups. The 28S sequences were aligned using the E-INS-i algorithm in MAFFT, accessed via the web server [50], with default parameters. The final 28S dataset included 112 sequences and 1993 nucleotide positions.
Outgroup sequences (COI, 28S) for seven genera of Phytoptidae s. str. (Fragariocoptes, Oziella, Novophytoptus) and Nalepellidae (Boczekella, Nalepella, Setoptus, Trisetacus) were sourced from previous studies [10,40,48].
Maximum likelihood (ML) analyses were performed using IQ-TREE 2.2.2.6 [51]. The best-fit substitution models mtART + R6 (COI, amino acids) and GTR + F + R5 (28S) were selected via ModelFinder [52] within IQ-TREE based on the Bayesian Information Criterion. Branch support was assessed using the Ultrafast bootstrap approximation (UFBoot) with 10,000 replicates, 1000 maximum iterations, and a minimum correlation coefficient of 0.99. The resulting ML trees were annotated with values of UFBoot and approximate likelihood ratio test (aLRT) calculated in IQ-TREE.
3. Results
3.1. Stereomicroscopy Observations of Web-Spinning Mite Populations on the Host Plant Organs
3.1.1. Aculops knorri (Population 1)
Most leaves on the sampled twigs of L. rubiginosa were covered with white webbing produced by A. knorri. On younger, bright green leaves located near the terminal buds, the webbing was less developed and restricted to the veins (Figure 2A). In contrast, on older leaves, the webbing was more extensive, covering larger areas, including the interveinal spaces (Figure 2B). The leaf epidermis beneath the webbing appeared similar to unwebbed areas and showed no evident signs of damage.

Figure 2.
Web-nests of Aculops knorri on the upper surface of leaves of Lepisanthes rubiginosa (Central Vietnam). (A,B) Young (A) and old (B) leaf with web-coating; (C,D) mite clusters under young, semi-transparent webbing along the midrib (C) and secondary veins (D); (E–G) mite colonies under mature, dense white webbing between the veins (webbing partially removed to expose mites). Scale bar: (A,B)—1 cm; (C–G)—250 µm.
Small groups of approximately 5–10 mites were observed under a very thin, nearly transparent young web along the midrib and secondary veins on all infested leaves (Figure 2C,D). Within these groups, mites were predominantly oriented with their anal region toward the periphery of the webbing site, likely excreting silk through the spinnerets.
Among the females of A. knorri observed under a stereomicroscope, some individuals appeared slightly narrower and more elongated (“narrow” specimens) while others were slightly wider and shorter (“wider” specimens), indicating possible morphological femaledimorphism.
Mite density was higher on older leaves, where they formed large colonies containing brightly orange adults, light orange or whitish immatures, and eggs beneath notably denser and less transparent white webbing compared to younger leaves (Figure 2E–G). Approximately 2000 mites were washed with 96% ethanol from the single leaf shown in Figure 2B.
3.1.2. Cisaberoptus kenyae and Aceria aegyptindicae (Population 2)
The distribution of webbing observed on the mango leaves (Figure 3A) matched the pattern reported in the literature [34,35,36]. At an early stage, webbing was confined to areas near, but not directly covering, the veins. At a later stage, the webbing extended to cover the veins and interveinal spaces.

Figure 3.
Webbing of eriophyoid mites on the upper surface of mango leaves. (A) Young leaf showing early mite webbing on the petiole, along the midrib and base of the secondary veins. (B) Semitransparent young webbing along the secondary vein of a young leaf. (C) Necrotic spots under webbing along the midrib of an older leaf. (D) Mixed colony of Cisaberoptus kenyae (ck) and Aceria aegyptindicae (aa). (E) Three adults and one nymph of Ac. aegyptindicae under young webbing. Note: webbing was partially removed (C–E) to expose necrotic spots and mites. Scale bar: (A) 1 cm; (B,C) 1 mm; (D,E) 200 µm.
Irregular black spots, presumably of necrotic epidermis, were observed beneath the webbing. On younger leaves with lower mite density and early-stage infestation, these spots were confined to the epidermis adjacent to the veins (Figure 3B). On older leaves with heavier, more established infestations, the spots also occurred on the veins (Figure 3C).
Mixed colonies of two mite species, C. kenyae and Ac. aegyptindicae, were observed under the webbing on most investigated leaves. Adults of C. kenyae were light orange and slightly more triangular in shape than the adults of Ac. aegyptindicae, which were pale whitish and more elongated (Figure 3D,E).
Remarks. Beyond the mixed population 2 from Northern Vietnam, we repeatedly observed C. kenyae and Ac. aegyptindicae coexisting under the same webbing on mango leaves in two South African localities (Table 1). This co-occurrence aligns with previous reports of mixed colonies of C. kenyae and Ac. aegyptindicae from Sudan, Egypt, Thailand, and Brazil [34]. Overall, the current evidence indicates that C. kenyae and Ac. aegyptindicae frequently coexist under shared webbing, and such mixed colonies are widely distributed across mango-growing regions worldwide.
3.2. Microscopy Observations of Slide-Mounted Web-Spinning Mites
3.2.1. Morphological Remarks on Aculops knorri (Population 1)
While closely resembling the type Thai population [38], the studied Vietnamese specimens of A. knorri exhibited clear differences in prodorsal shield ornamentation and several morphometrics (Table S1). Contrary to the smooth prodorsal shield reported for the Thai specimens [38], the mites from Vietnam possess a prodorsal shield with two distinct, complete admedian lines, which delimit a central suboval area preceded by a large, broad-based, subtriangular frontal lobe (Figure 4G,H).
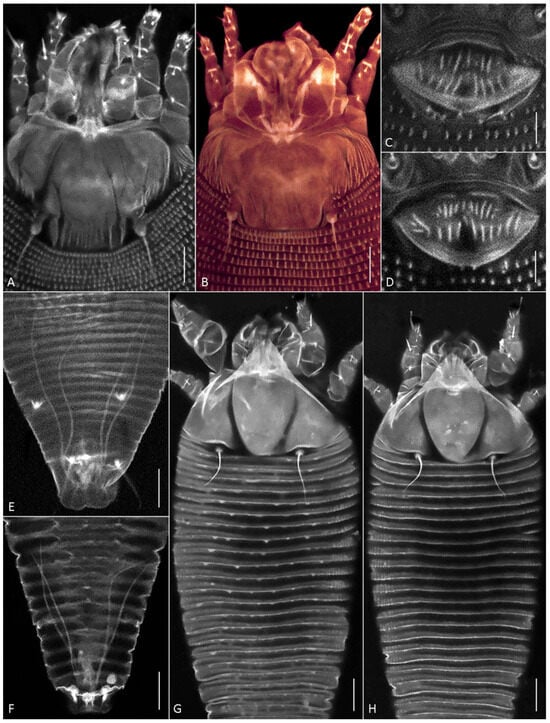
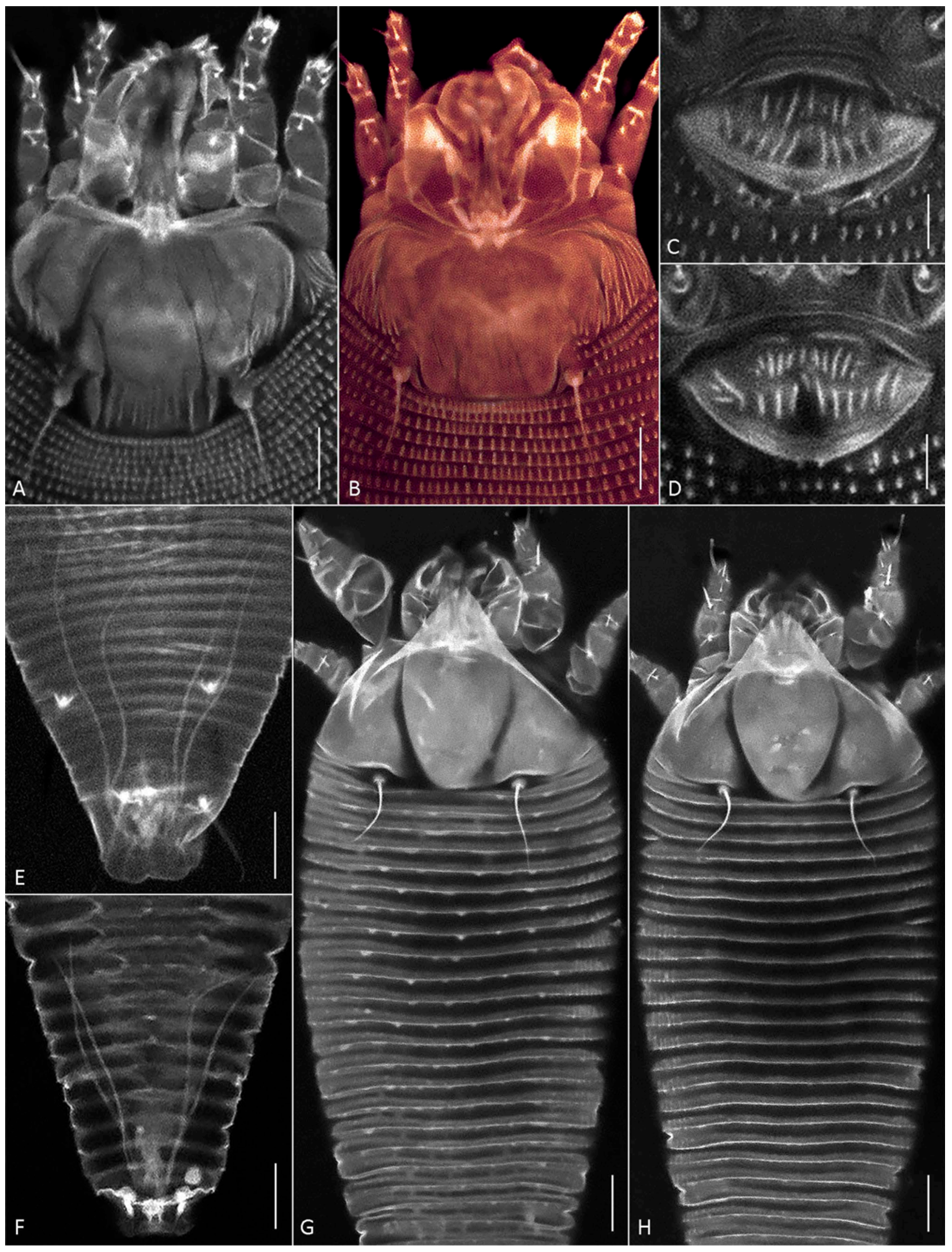
Figure 4.
CLSM images of females of Aceria aegyptindicae (A–E) and Aculops knorri (F–H). (A–D) Prodorsal shields (A,B) and external genitalia (C,D) in Ac. aegyptindicae; (E,F) cuticular ducts of silk-producing anal glands; (G,H) dorsal view of prosoma and anterior part of opisthosoma in “wider” (G), and “narrower” (H) females of A. knorri. Note small subtriangular microtubercles on the dorsal opisthosomal annuli in Figure 4G. Scale bar: (A,B,G,H) 10 µm; (C–F) 5 µm.
Within the Vietnamese population, females provisionally identified under a stereo microscope as “narrower” and “wider” forms (see Section 3.1.1.) differed in the following characteristics: the body width-to-length ratio, the lengths of setae 2a and l″ I, and the ornamentation of the dorsal opisthosomal annuli (Table S1). The wider specimens possessed subtriangular microtubercles that were more pronounced on the anterior half of the opisthosoma. In contrast, all dorsal opisthosomal annuli were smooth in the narrower specimens (Figure 4G,H). These observations suggest the presence of two distinct morphotypes within the Vietnamese population of A. knorri.
3.2.2. Morphological Remarks on Cisaberoptus kenyae (Population 2)
All investigated specimens of C. kenyae from our material possess a tibial seta l′ I 3–4 µm long, a feature not reported by previous authors (Figure S1).
Compared to specimens of C. kenyae from the type population in Kenya [45], our specimens from Vietnam were slightly wider, had longer setae sc, and fewer empodial rays on leg II (Table S2).
Specimens from Egypt [34] were morphometrically nearly identical to those from Kenya [45]. Notably, mites from both African populations (Kenya and Egypt) had a significantly narrower prodorsal shield than those in our material (Table S2). This difference, though consistent with geographical variation, is more likely caused by an artifact of measurement methodology or error since the recorded narrower prodorsal shield conflicts with the illustrated body proportions [34] (plate 1 DA); [45] (Figure 5D,F).
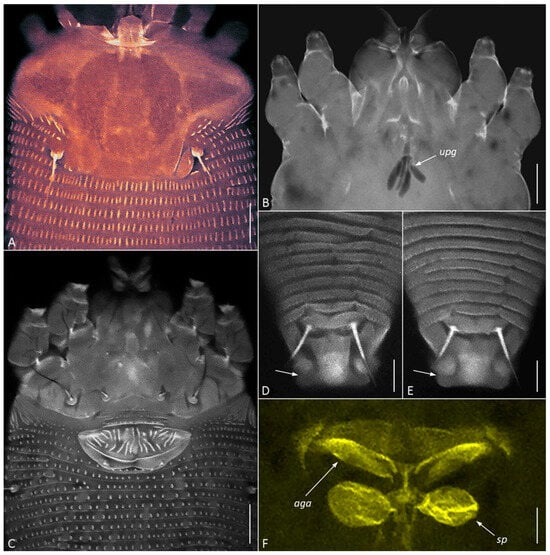
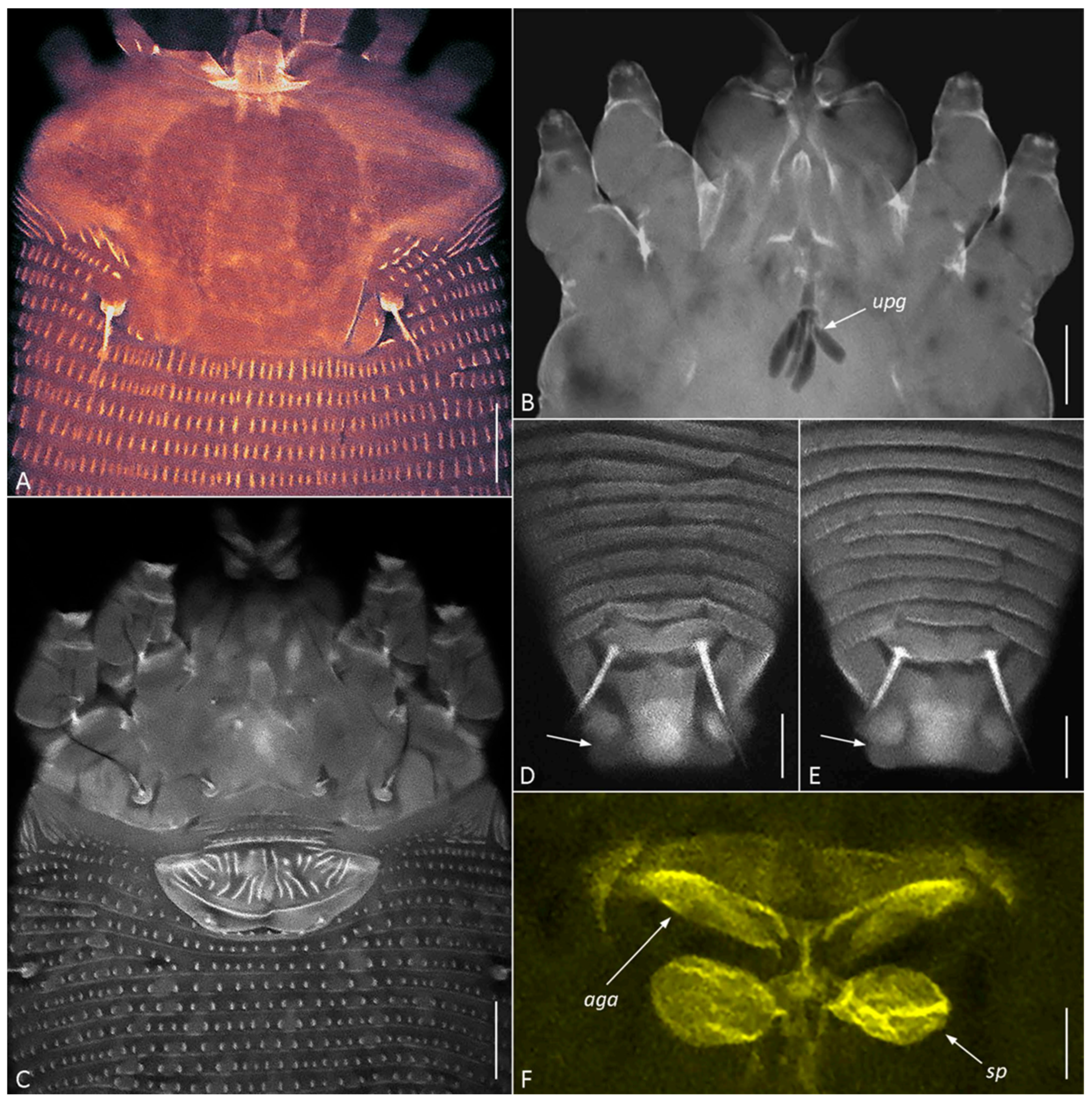
Figure 5.
CLSM images of females of Cisaberoptus kenyae from Northern Vietnam (population 2). (A) Prodorsal shield with conspicuous frontal lobe. (B) Outlines of lobular unpaired prosomal gland (upg) outlined by auto-fluorescing partially dissolved internal tissues, (C) coxigenital area, (D,E) dorsal view of telosoma and caudal lobe with lateral folding (arrows), (F) internal genitalia including ovoid spermathecae (sp) and plate-like anterior genital apodeme (aga). Scale bar: (A–C) 10 µm; (D–F) 5 µm.
A morphometric investigation of an additional population of C. kenyae from Florida showed these mites to be slightly smaller, with shorter setae sc and h2, while matching the other morphometrics of specimens from Hanoi (Table S2).
CLSM of C. kenyae from Vietnam revealed a small subrectangular frontal lobe of the prodorsal shield 4–5 µm long (Figure 5A) and outlines of a large, unpaired gland associated with the motivator (Figure 5B, upg), a structure rarely observed under conventional light microscopy. A tiny cuticular lateral folding of the caudal lobe (Figure 5D,E, arrow) was registered in all specimens.
CLSM of the female genitalia of C. kenyae (Figure 5C,F) showed their general resemblance to those in members of the subfamily Cecidophyinae, which are characterized by external genitalia appressed to coxae II, a genital coverflap ornamented with short ridges organized in two transverse rows, and an anterior genital apodeme situated in a plane orthogonal to the main body axis and anterior to small ovoid spermathecae [11,53,54,55].
3.2.3. Morphological Remarks on Aceria aegyptindicae (Population 2)
The Vietnamese specimens closely resembled those from the type population of Ac. aegyptindicae from Egypt and the specimens from Brazil, which were originally misidentified as protogynes of C. kenyae [34,37]. Morphometric differences among these three populations were minimal and largely overlapping (Table S3). Distinctive characters included: shorter setae (ft″ I, ft″ II, l″ I, l″ II, 1a, 2a, d, e) in Egyptian specimens; longer setae c2 and h2 in Vietnamese specimens; a greater number of ventral annuli and a larger distance between setae sc in Brazilian specimens; and a genital coverflap that was longest and narrowest in Egyptian specimens (Table S3).
The specimens from Vietnam showed variation in the ornamentation of the prodorsal shield and genital coverflap, delineating two putative forms. One form (identified as Ac. aegyptindicae) featured a prodorsal shield with distinct, numerous short lines laterally, a nearly smooth central area, and a genital coverflap with a greater number of basal ridges (Figure 4B,D). The other form (identified as Aceria cf. aegyptindicae) exhibited a shield with short subparallel lines medio-posteriorly and faint, longer lines resembling admedian or submedian lines, alongside a coverflap with notably fewer basal scores (Figure 4A,C).
3.2.4. Silk-Producing Apparatus (SPA) of Aceria aegyptindicae and Aculops knorri
In females of both species, a uniformly organized SPA was observed beneath the cuticle of the posterior opisthosoma (Figure 4E,F; Figure 6 and Figure 7). It comprised the same structures as those recently described in detail for the southern African web-spinning species Ab. schotiae [27], including large silk-producing anal glands extending along approximately one-fourth of the body length.
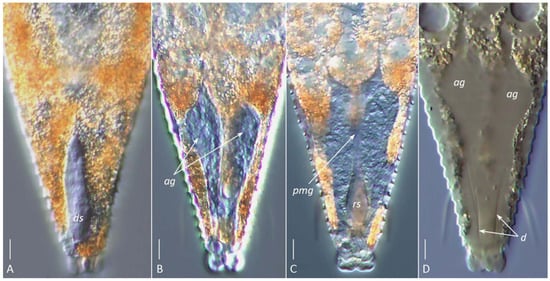
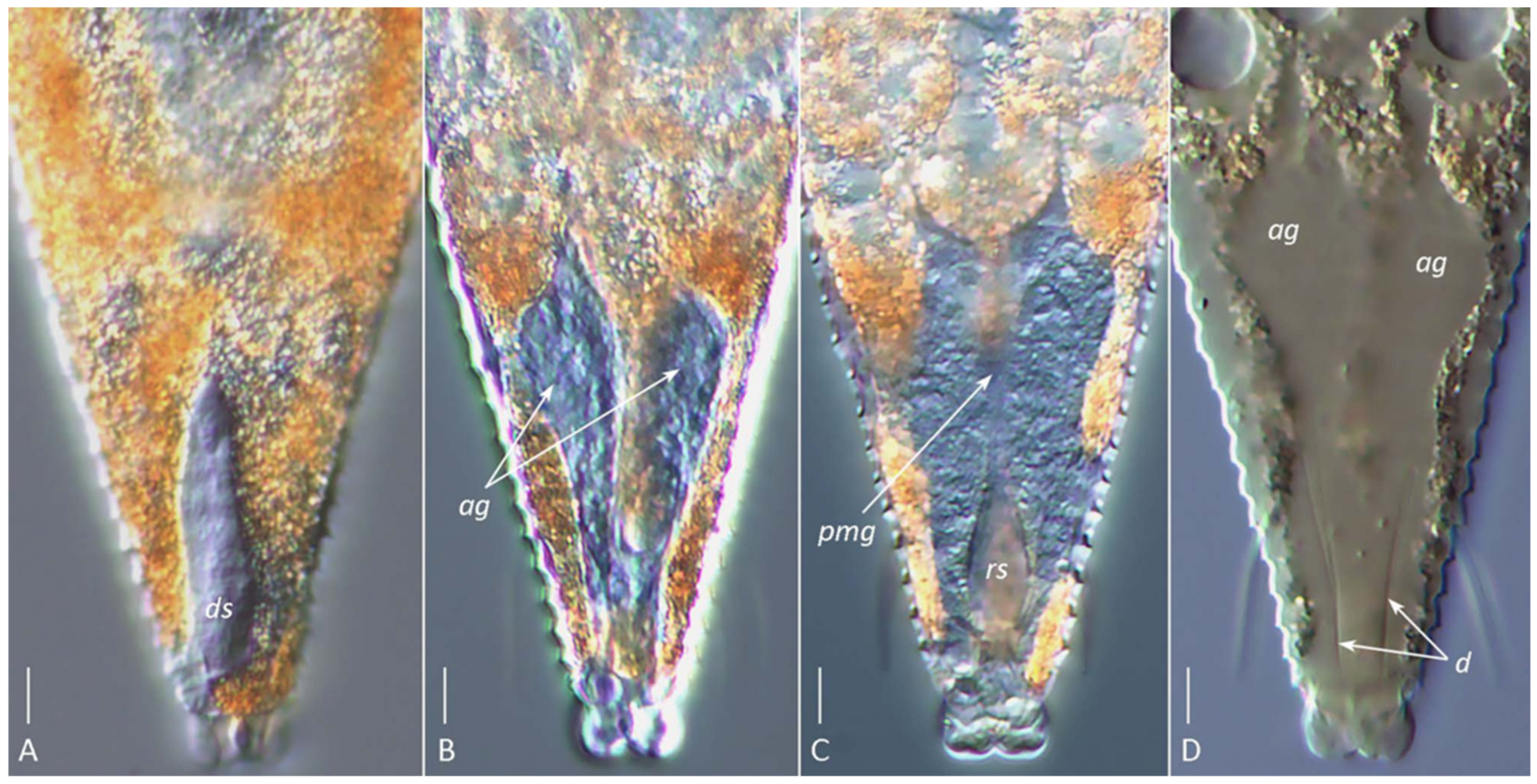
Figure 6.
DIC images of the silk-producing apparatus in live ((A–C), glycerin mounts) and partially cleared ((D), Hoyer’s medium) females of Aculops knorri. (A) Dorsal sac (ds); (B) paired hypertrophied anal glands (ag); (C) rectal sac (rs, corresponding to the anterior part of the rectum) and putative posterior midgut (pmg); (D) outlines of partially dissolved anal glands (ag) and their cuticular ducts (d). Note: Images (A–C) are from different specimens, highlighting the dorsal (A), medial (B), and ventral (C) structures of the silk-producing complex. Scale bar: 5 µm.
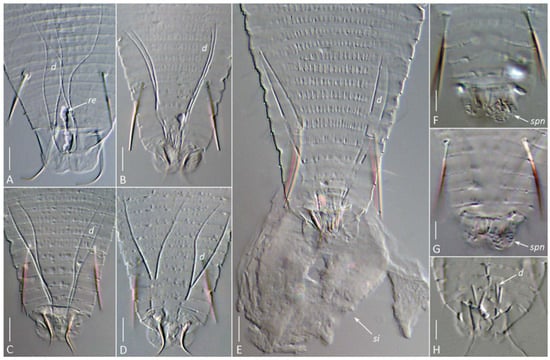
Figure 7.
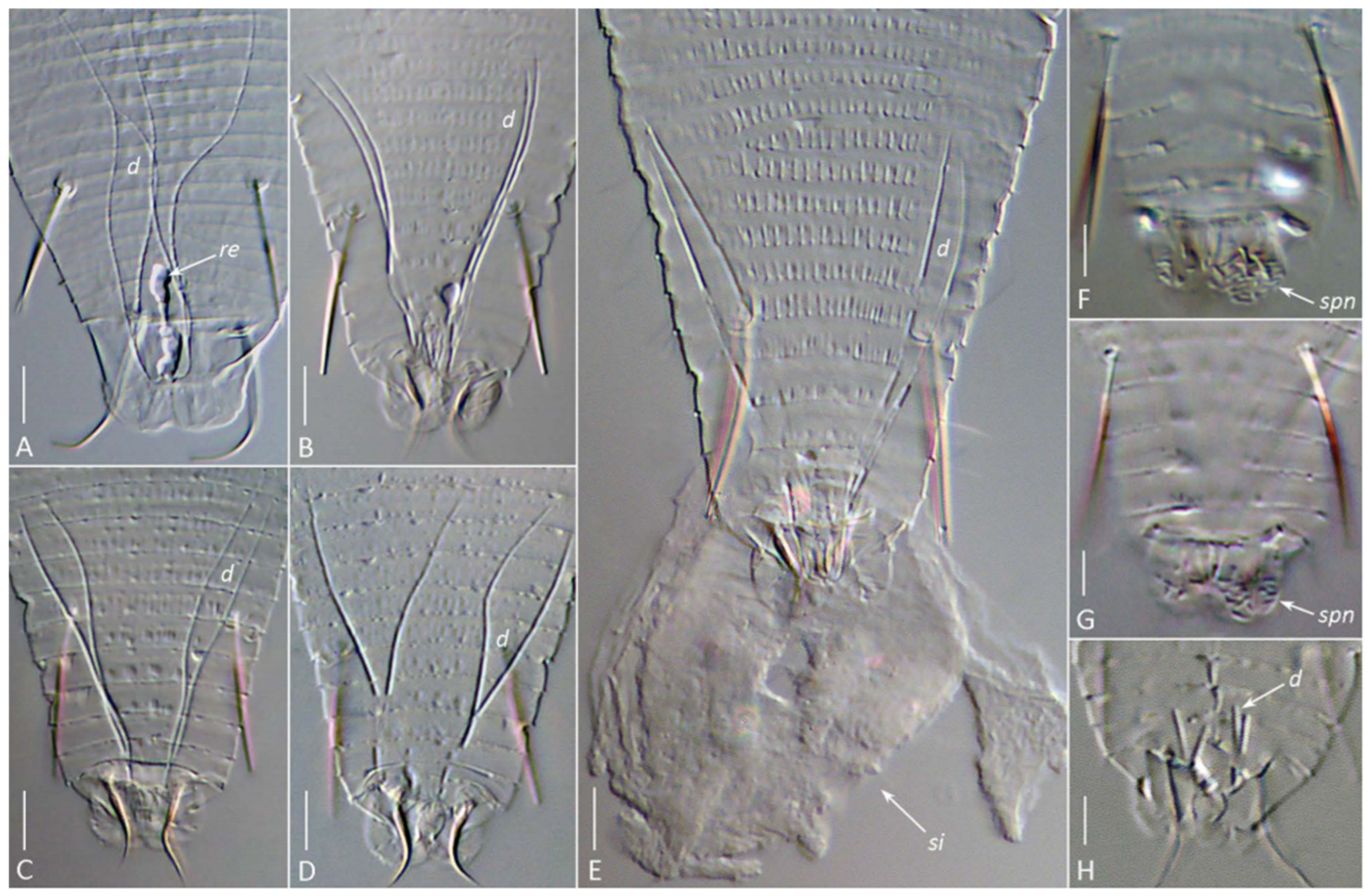
DIC images of the silk-producing apparatus in completely cleared (Hoyer’s medium) females (A–G) and males (H) of Aceria aegyptindicae (A) and Aculops knorri (B–H). Abbreviations: d—ducts of the anal glands; re—putative rectum filled with silk; si—silk extruded from the silk-producing organs of a slightly compressed mite; spn—areas of porose cuticle (the spinneret) on the caudal lobes. Scale bar: (A–D) 5 µm; (E–H) 3 µm.
The visibility of specific SPA elements depended on the mounting method. In glycerin mounts of live A. knorri females, the elongated silk-producing anal glands and, rarely, the reservoirs for silk storage (cuticular sacs and rectum) were visible (Figure 6A–C). In partially cleared specimens (Hoyer’s medium), the outlines of the rapidly dissolving anal glands and traces of their cuticular ducts were observed (Figure 6D).
Slight mechanical pressure applied to the cover slip with a dissection needle induced living A. knorri females in the early clearing process to extrude a droplet of silk material through the anus, forming a round silk deposit with a small central clearing at the anal opening (Figure 7E). In fully cleared specimens, a pair of two-branched anal gland ducts, remnants of the cuticular reservoirs between them (sometimes containing undissolved silk), and areas of porose cuticle on the anal lobes (the spinneret) were visible (Figure 7).
In males, the SPA was organized similarly to that of females but was generally smaller, with notably shorter anal gland ducts (Figure 7H).
3.3. Molecular Phylogenetics
3.3.1. COI and 28S Sequence Diversity
All DNA isolates of A. knorri (covering two morphotypes observed in the population 1, see Section 3.2.1) yielded identical (K2P = 0) COI and 28S sequences, confirming the conspecificity of the analyzed specimens.
COI sequences of Cisaberoptus and Aceria showed significant diversity (Table S4). Within the genus Aceria, three distinct clusters were identified: (i) isolates d392 and d398 of Ac. aegyptindicae from Vietnam (K2P = 0); (ii) isolate d395 of Ac. cf. aegyptindicae from Vietnam; and (iii) isolate PC22b of Aceria sp. from South Africa. The high genetic distance between two last clusters (K2P = 0.18) suggests that they may be cryptic species, each distinct from the Vietnamese lineage of Ac. aegyptindicae (distances of 0.20 and 0.23, respectively).
COI sequences of Cisaberoptus from Vietnam (isolates d400 and d401) and Florida (isolate d211) formed a genetically uniform cluster (K2P = 0), representing a single widespread species. However, the specimen from India (MW491351) was markedly divergent from this cluster (K2P = 0.24–0.25), indicating that it is a putative separate species within the genus.
K2P distances for the 28S gene revealed distinct patterns of sequence divergence (Table S5). Within Aceria, specimens from Hanoi identified as Ac. aegyptindicae (isolates d392, d393 and d398) and Ac. cf. aegyptindicae (isolates d395 and d1000) formed a highly homogeneous group (K2P = 0–0.007). In contrast, an Aceria specimen from South Africa (isolate d1063) was consistently divergent from all other Aceria sequences (K2P = 0.055–0.057), indicating that it represents a distinctly separate lineage. All analyzed specimens of C. kenyae from Florida (isolates d1060, d1061, d1062), South Africa (d1065, PC22a), and Vietnam (d400, d401, d636) were genetically uniform (K2P = 0–0.002), confirming their conspecificity across a broad geographic range.
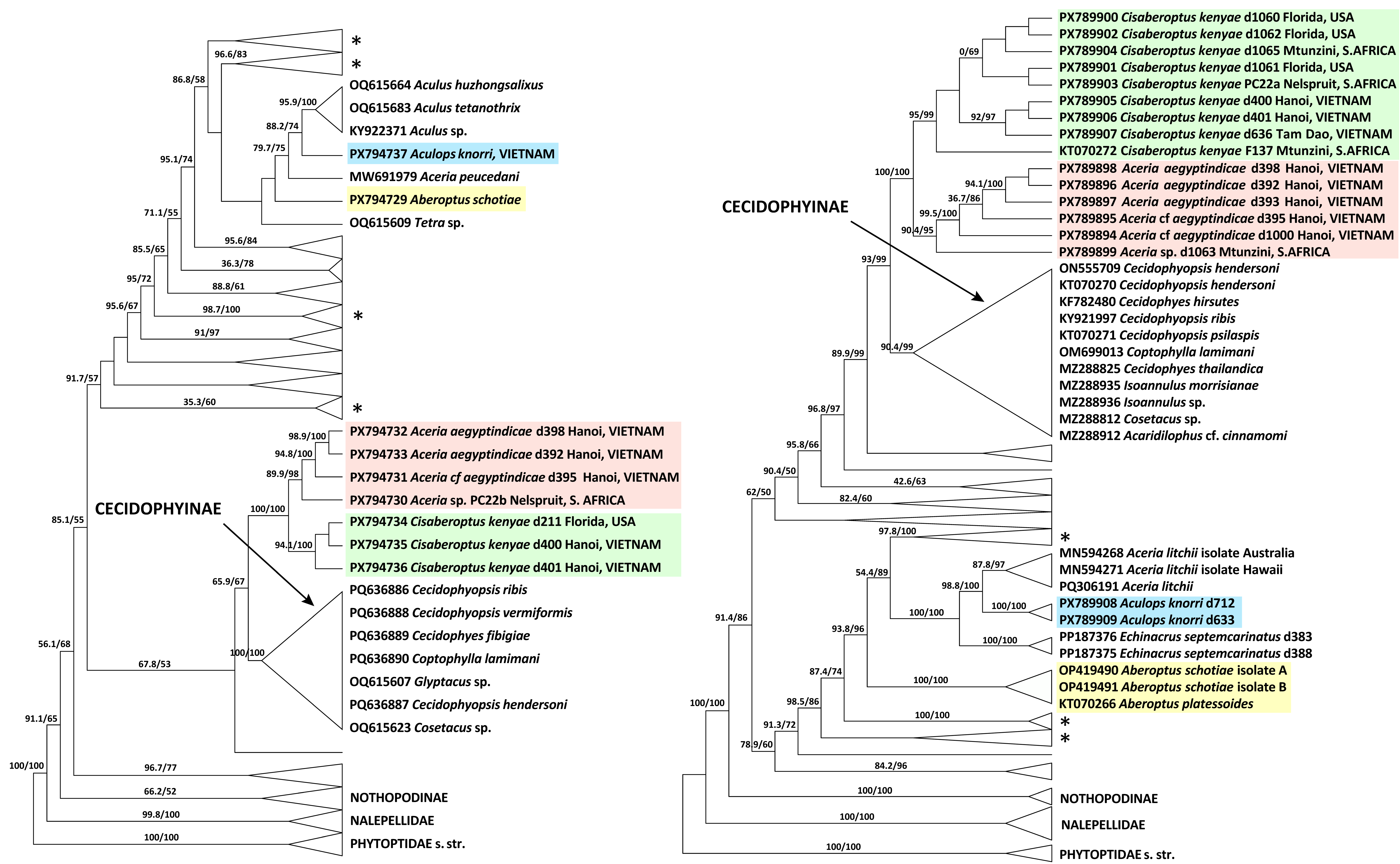
3.3.2. COI and 28S Phylogenetic Analyses (Figure 8)
Maximum likelihood analyses of COI and 28S sequences produced different overall tree topologies but shared several congruent, well-supported clades. Both analyses confirm the basal divergence of Eriophyoidea into three major lineages, Phytoptidae s. str., Nalepellidae, and Eriophyidae s. str., with poorly resolved internal relationships. Both datasets support the monophyly of the subfamily Cecidophyinae, placing it as sister to a lineage containing the monophyletic genus Cisaberoptus and a clade comprising mango-associated Aceria spp. The ecological group of web-producing species is not monophyletic; its members are dispersed, with one lineage (the mango-associated Cisaberoptus and Aceria) sister to Cecidophyinae, and another (comprising Aberoptus and A. knorri) positioned within clades dominated by members of Anthocoptinae and Aceriini.
The COI and 28S trees differ in the inferred sister taxa for A. knorri. The 28S topology suggests an ecologically based relationship, grouping A. knorri with Aceria litchii (both associated with Sapindaceae). In contrast, the COI topology—lacking data for Ac. litchii—supports a taxonomic relationship, placing A. knorri as sister to a clade of three Aculus species (all members of the tribe Anthocoptini).
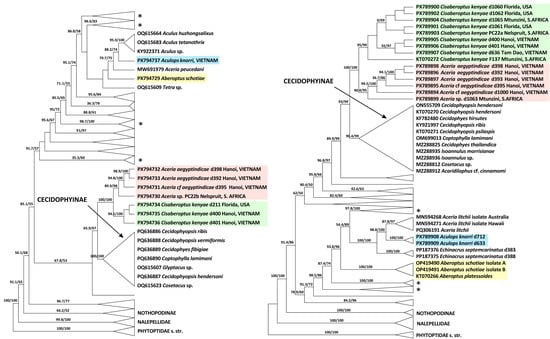
Figure 8.
COI (left) and 28S (right) ML phylogenies of Eriophyoidea showing the placement of Aberoptus (yellow), Cisaberoptus (green), Aculops knorri (blue), and mango-associated Aceria spp. (pink). Branches are labeled with aLRT values and UFBoot >50. Non-relevant clades are collapsed. Asterisks mark clades dominated by Anthocoptinae and Aceriini.
Figure 8.
COI (left) and 28S (right) ML phylogenies of Eriophyoidea showing the placement of Aberoptus (yellow), Cisaberoptus (green), Aculops knorri (blue), and mango-associated Aceria spp. (pink). Branches are labeled with aLRT values and UFBoot >50. Non-relevant clades are collapsed. Asterisks mark clades dominated by Anthocoptinae and Aceriini.
4. Discussion
This study, based on extensive material from Asia, Africa, and North America, revealed three central findings in web-spinning gall mites: (a) the convergent evolution of silk-webbing, (b) cryptic diversity within silk-producing lineages of Eriophyidae, and (c) cooperative web-production in sympatric eriophyid species associated with mango.
Cisaberoptus—Aceria sympatry on mango tree leaves. Our data demonstrate that similar to web-spinning mites of genus Aberoptus [25,27], Aceria aegyptindicae, Cisaberoptus kenyae (co-existing on mango), and Aculops knorri (from Lepisanthes rubiginosa) share a suite of related ecological traits. They produce large protective silk films, feed and reproduce beneath them, establish their younger colonies under transparent webbing along leaf veins, later expanding the silk webbing into the interveinular spaces, and show colony density and web coverage increasing on older leaves (Figure 2 and Figure 3). A notable divergence, however, is evident in their phytopathological impact. Contrary to A. knorri and Aberoptus, which cause no visible damage to their hosts, the mixed Cisaberoptus–Aceria colonies on mango from Vietnam were consistently associated with necrotic epidermal patches beneath the webbing (Figure 3C)—a type of damage previously reported in India for C. kenyae [56]. These data indicate a more injurious interaction on mango resulting in leaf tissue damage, potentially due to mite saliva or transmitted infectious agents.
Reports from geographically distant populations indicate that C. kenyae and Ac. aegyptindicae often coexist in syntopy under a common web on mango leaves and exhibit synchronized seasonal population dynamics [34]. Furthermore, we found that Ac. aegyptindicae possesses a well-developed silk-producing apparatus (Figure 4E), suggesting a capability for building large protective web—a trait shared with Cisaberoptus, Aberoptus, and A. knorri. The frequent co-occurrence, population synchrony, web-spinning capability, and shared use of a web support the hypothesis of a symbiotic association of C. kenyae and A. aegyptindicae on mango. We consider this type of relationship a case of sporadic mutualism between C. kenyae and A. aegyptindicae, genetically very close sister species. When their populations develop in syntopy, these relationships appear to involve the cooperative production and maintenance of a common web, resembling the symbiotic relationships observed between populations of some other arachnids [57,58,59].
Cryptic diversity in populations of web-spinning mites. Morphological and molecular investigations revealed undocumented variation in populations of web-spinning eriophyids from L. rubiginosa and mango. For A. knorri in Vietnam, two morphotypes (“narrow” and “wider”) were distinguished by body proportions and ornamentation of the dorsal opisthosomal annuli, both diverging from the type population from Thailand, which is characterized by a smooth prodorsal shield. Interestingly, a very similar species, Tetra lepisanthae Boczek & Chandrapatya 2000 from Thailand, most probably synonymous with A. knorri, has two faint admedian lines on the prodorsal shield [60], like our specimens from Vietnam, suggesting the presence of a common population of A. knorri in Indochina. We also found subtle but consistent morphometric differences in C. kenyae and Ac. aegyptindicae from our material compared to other geographical populations. Furthermore, two putative forms were observed within the Vietnamese Ac. aegyptindicae specimens, slightly differing in prodorsal shield and genital coverflap ornamentation.
Molecular data provided a crucial framework for interpreting this variation. For A. knorri, the genetic uniformity (K2P = 0) of COI and 28S sequences strongly indicates that the two morphotypes represent intraspecific variation, likely attributable to seasonal female dimorphism (deuterogeny) [61]. Conversely, for the mango-associated mites, molecular data clarified complex species boundaries. All Cisaberoptus specimens from Vietnam, Florida, and South Africa formed a single genetically uniform cluster, which suggest that C. kenyae may have been co-introduced globally with its host, M. indica (Figure 1), coinciding with the most intensive areas of contemporary mango cultivation across continents [43]. The divergent sequence (MW491351) of Cisaberoptus from India suggests a cryptic species within the genus, which may reflect genetic diversity and the presence of quasi-isolated old populations of mango in India—the center of origin for most contemporary mango cultivars [43,62,63].
For web-spinning Aceria associated with mango, the genetic data presented a more complicated picture: while Vietnamese Ac. aegyptindicae and Ac. cf. aegyptindicae were homogeneous in 28S, COI sequence analyses suggested that Ac. cf. aegyptindicae from Vietnam and an Aceria sp. from South Africa might be distinct cryptic species. This discordance between the mitochondrial and nuclear markers, as well as between morphology and COI data, highlights the potential for cryptic speciation and warrants further study with additional marker genes and populations.
Convergent evolution of silk-webbing in Eriophyoidea. The molecular phylogenetic analyses yielded two key insights into the evolution of web-spinning. First, the web- producing habit is a homoplasy; it has evolved at least twice independently within Eriophyidae—once in the lineage leading to Aberoptus and A. knorri, and once in the monophyletic, mango-associated Cisaberoptus + Aceria (mCA) lineage. This supports the hypothesis that web-spinning is a convergent adaptation, which evolved based on a symplesiomorphic internal anatomy [30] and resulted in the formation of a rectum-associated silk-producing apparatus (SPA) [27]. Our novel documentation of the SPA in A. knorri and Ac. aegyptindicae confirms its structural homology with that of Aberoptus, underscoring this convergence in phylogenetically remote clades.
Secondly, the consistent sister-group relationship between the web-spinning mCA lineage and the subfamily Cecidophyinae is intriguing, as it coincides with notable structural similarities in their external and internal genitalia. If confirmed, this relationship would provide a strong phylogenetic rationale for including the mCA clade within Cecidophyinae and for excluding the mango-associated Aceria species from the polyphyletic genus Aceria. The supported sister relationship between C. kenyae and Ac. aegyptindicae further suggests that they likely share a homologous SPA and web-spinning mechanism, though this will require direct morphological confirmation.
5. Conclusions
Our molecular and morphological evidence establishes the convergent evolution of silk-webbing in Eriophyoidea and reveals a novel, testable phylogenetic hypothesis linking web-spinning eriophyid mites from mango to Cecidophyinae. The following future perspectives will be crucial to testing the phylogenetic relationships proposed here and to build a comprehensive understanding of silk-use and evolution across Eriophyoidea: (a) a comparative histomorphology of Cisaberoptus and other web-spinning taxa, (b) phylogenetics based on new molecular datasets, including more conserved sequences such as single-copy nuclear protein genes, (c) investigation of silk genetics and proteomics, (d) targeted searches for new web-spinning taxa, and (e) experimental studies elucidating the functions of silk in eriophyoids that do not produce large web nests.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/insects17030259/s1, Figure S1: Tibial seta l’ I in two females of Cisaberoptus kenyae; Table S1: Morphological differences between females of A. knorri from Thailand [38] and Vietnam; Table S2: Morphological differences between females of C. kenyae from Kenya [45], Egypt [34], Vietnam, and Florida; Table S3: Morphological differences between females of Ac. aegyptindicae from Egypt [34], Brazil [37], and Vietnam; Table S4: K2P genetic distances for the COI gene sequences among studied isolates of Aceria and Cisaberoptus from mango; Table S5: K2P genetic distances for the 28S gene sequences among the studied isolates of Aceria and Cisaberoptus from mango.
Author Contributions
Conceptualization, supervision & project administration, P.E.C.; methodology, P.E.C., C.C., A.E.V. and A.E.R.; validation, A.E.V., A.G.D., J.A. and V.D.N.; formal analysis, P.E.C.; investigation, P.E.C., A.T.K. and N.S.K., C.C.; resources, P.E.C., C.C., H.X.T. and A.E.R.; data curation, V.D.N., A.T.K., N.S.K. and A.E.R.; writing: original draft—P.E.C.; writing: review & editing—P.E.C., J.A., A.G.D. and C.C.; visualization, P.E.C., A.T.K. and A.E.R.; funding acquisition, P.E.C., C.C. and H.X.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partially funded by Zoological Institute of Russian Academy of Sciences (ZIN RAS project #125013001089-0).
Data Availability Statement
The new sequences of eriophyoid mites have been deposited in the National Center for Biotechnology Information (NCBI) GenBank database (https://www.ncbi.nlm.nih.gov/genbank (accessed on 10 January 2026); accession numbers: PX789894-PX789911, PX794729-PX794740.
Acknowledgments
We thank S. Bolton (USA) for the comparative material of C. kenyae and P. Dyamanagouda (India) for the detailed collection data for COI sequence MW491351. We acknowledge the support from the St. Petersburg State University (project ID 125022803066-3) and Centre for Molecular and Cell Technologies and Center for Microscopy and Microanalysis (Research Park, St. Petersburg State University) for technical assistance in confocal microscopy and sequencing.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Lindquist, E.E. External anatomy and notation of structures. In Eriophyoid Mites: Their Biology, Natural Enemies and Control; Lindquist, E.E., Sabelis, M.W., Bruin, J., Eds.; World Crop Pests; Elsevier Science Publishing: Amsterdam, The Netherlands, 1996; Volume 6, pp. 3–31. [Google Scholar] [CrossRef]
- Lindquist, E.E. Evolution and phylogeny. 1.5.2. Phylogenetic relationships. In Eriophyoid Mites: Their Biology, Natural Enemies and Control; Lindquist, E.E., Bruin, J., Sabelis, M.W., Eds.; World Crop Pests; Elsevier: Amsterdam, The Netherlands, 1996; Volume 6, pp. 301–327. [Google Scholar] [CrossRef]
- Bagnjuk, I.G.; Sukhareva, S.I.; Shevchenko, V.G. Major trends in the evolution of four-legged mites as a specialized group using families Pentasetacidae Shev., Nalepellidae Roiv. and Phytoptidae Murray (Acari: Tetrapodili) as examples. Acarina 1998, 6, 59–76. [Google Scholar]
- Bolton, S.J.; Chetverikov, P.E.; Klompen, H. Morphological support for a clade comprising two vermiform mite lineages: Eriophyoidea (Acariformes) and Nematalycidae (Acariformes). Syst. Appl. Acarol. 2017, 22, 1096–1131. [Google Scholar] [CrossRef]
- Bolton, S.J.; Chetverikov, P.E.; Ochoa, R.; Klimov, P.B. Where Eriophyoidea (Acariformes) belong in the tree of life. Insects 2023, 14, 527. [Google Scholar] [CrossRef]
- Klimov, P.B.; Kolesnikov, V.B.; Vorontsov, D.D.; Ball, A.D.; Bolton, S.J.; Mellish, C.; Edgecombe, G.D.; Pepato, A.R.; Chetverikov, P.E.; He, Q.; et al. The evolutionary history and timeline of mites in ancient soils. Sci. Rep. 2025, 15, 13555. [Google Scholar] [CrossRef] [PubMed]
- Shevchenko, V.G.; Bagnyuk, I.G.; Sukhareva, S.I. Novoe semejstvo chetyrekhnogih kleshchej Pentasetacidae (Acariformes, Tetrapodili) i ego znachenie dlya traktovki proiskhozhdeniya gruppy. Zool. Zh. 1991, 70, 47–53. [Google Scholar]
- Craemer, C. A Systematic Appraisal of the Eriophyoidea (Acari: Prostigmata). Ph.D. Dissertation, University of Pretoria, Pretoria, South Africa, 2010. [Google Scholar]
- Li, H.S.; Xue, X.F.; Hong, X.Y. Homoplastic evolution and host association of Eriophyoidea (Acari, Prostigmata) conflict with the morphological-based taxonomic system. Mol. Phylogenet. Evol. 2014, 78, 185–198. [Google Scholar] [CrossRef]
- Chetverikov, P.E.; Craemer, C.; Cvrković, T.; Klimov, P.B.; Petanović, R.U.; Romanovich, A.E.; Sukhareva, S.I.; Zukoff, S.N.; Bolton, S.; Amrine, J. Molecular phylogeny of the phytoparasitic mite family Phytoptidae (Acariformes: Eriophyoidea) identified the female genitalic anatomy as a major macroevolutionary factor and revealed multiple origins of gall induction. Exp. Appl. Acarol. 2021, 83, 31–68. [Google Scholar] [CrossRef]
- Amrine, J.W., Jr.; Stasny, T.A.H.; Flechtmann, C.H.W. Revised Keys to the World Genera of the Eriophyoidea (Acari: Prostigmata); Indira Publishing House: Michigan, MI, USA, 2003; pp. 1–244. [Google Scholar]
- Chetverikov, P.E.; Craemer, C.; Bolton, S. Exoskeletal transformations in Eriophyoidea: New pseudotagmic taxon Pseudotagmus africanus n.g. & n. sp. from South Africa and remarks on pseudotagmosis in eriophyoid mites. Syst. Appl. Acarol. 2017, 22, 2093–2118. [Google Scholar] [CrossRef]
- Sukhareva, S.I. Family Phytoptidae Murray 1877 (Acari: Tetrapodili), its consisting, structure and suggested ways of evolution. Acarina 1994, 2, 47–72. [Google Scholar]
- Chetverikov, P.E.; Cvrković, T.; Makunin, A.; Sukhareva, S.; Vidović, B.; Petanović, R. Basal divergence of Eriophyoidea (Acariformes, Eupodina) inferred from combined partial COI and 28S gene sequences and CLSM genital anatomy. Exp. Appl. Acarol. 2015, 67, 219–245. [Google Scholar] [CrossRef]
- Skoracka, A. Host specificity of eriophyoid mites: Specialists or generalists. Biol. Lett. 2006, 43, 289–298. [Google Scholar]
- Skoracka, A.; Smith, L.; Oldfield, G.; Cristofaro, M.; Amrine, J.W. Host-plant specificity and specialization in eriophyoid mites and their importance for the use of eriophyoid mites as biocontrol agents of weeds. In Eriophyoid Mites: Progress and Prognoses; Ueckermann, E.A., Ed.; Springer: Dordrecht, The Netherlands, 2010; pp. 93–113. [Google Scholar] [CrossRef]
- Skoracka, A.; Magalhães, S.; Rector, B.G.; Kuczyński, L. Cryptic speciation in the acari: A function of species lifestyles or our ability to separate species? Exp. Appl. Acarol. 2015, 67, 165–182. [Google Scholar] [CrossRef]
- de Lillo, E.; Craemer, C.; Amrine, J.W., Jr.; Nuzzaci, G. Recommended procedures and techniques for morphological studies of Eriophyoidea (Acari: Prostigmata). Exp. Appl. Acarol. 2010, 51, 283–307. [Google Scholar] [CrossRef]
- de Lillo, E.; Marini, F.; Cristofaro, M.; Valenzano, D.; Petanović, R.; Vidović, B.; Cvrković, T.; Bon, M.-C. Integrative taxonomy and synonymization of Aculus mosoniensis (Acari: Eriophyidae), a potential biological control agent for tree of heaven (Ailanthus altissima). Insects 2022, 13, 489. [Google Scholar] [CrossRef] [PubMed]
- Navia, D.; Ferreira, C.B.; Reis, A.C.; Gondim, M.G. Traditional and geometric morphometrics supporting the differentiation of two new Retracrus (Phytoptidae) species associated with heliconias. Exp. Appl. Acarol. 2015, 67, 87–121. [Google Scholar] [CrossRef] [PubMed]
- Petanović, R. Towards an integrative approach to taxonomy of Eriophyoidea (Acari, Prostigmata)—An overview. Ecol. Montenegrina 2016, 7, 580–599. [Google Scholar] [CrossRef]
- Buttachon, S.; Arikit, S.; Nuchchanart, W.; Puangmalee, T.; Duanchay, T.; Jampameung, N.; Sanguansub, S. Geometric morphometric analysis and molecular identification of coconut mite, Aceria guerreronis Keifer (Acari: Eriophyidae) collected from Thailand. Insects 2022, 13, 1022. [Google Scholar] [CrossRef]
- Saccaggi, D.L.; Maboeei, P.; Powell, C.; Ngubane-Ndhlovu, N.P.; Allsopp, E.; Wesley-Smith, J.; van Asch, B. Towards solving the “Colomerus vitis conundrum”: Genetic evidence reveals a complex of highly diverged groups with little morphological differentiation. Diversity 2022, 14, 342. [Google Scholar] [CrossRef]
- Manson, D.C.M.; Gerson, U. Web spinning, wax secretion and liquid secretion by eriophyoid mites. In Eriophyoid Mites: Their Biology, Natural Enemies and Control; Lindquist, E.E., Sabelis, M.W., Bruin, J., Eds.; World Crop Pests; Elsevier Science Publishing: Amsterdam, The Netherlands, 1996; Volume 6, pp. 251–258. [Google Scholar] [CrossRef]
- Flechtmann, C.H.W. Aberoptus cerostructor n. sp., a deuterogynous species from Brazil (Acari: Eriophyidae). Int. J. Acarol. 2001, 27, 199–204. [Google Scholar] [CrossRef]
- Chetverikov, P.E.; Bolton, S.J.; Gubin, A.I.; Letukhova, V.Y.; Vishnyakov, A.E.; Zukoff, S. The anal secretory apparatus of Eriophyoidea and description of Phyllocoptes bilobospinosus n. sp. (Acariformes: Eriophyidae) from Tamarix (Tamaricaceae) from Ukraine, Crimea and USA. Syst. Appl. Acarol. 2019, 24, 139–157. [Google Scholar] [CrossRef]
- Chetverikov, P.E.; Craemer, C.; Gankevich, V.D.; Vishnyakov, A.E.; Zhuk, A.S. A new webbing Aberoptus species from South Africa provides insight in silk production in gall mites (Eriophyoidea). Diversity 2023, 15, 151. [Google Scholar] [CrossRef]
- Chetverikov, P.E.; Ngubane-Ndhlovu, N.P.; Situngu, S.; Elhalawany, A.; Amrine, J. Confocal Microscopy and Molecular Analyses Reveal Anal Secretory Apparatus in Immatures and Recover Transcontinental Clade of Gall Mites (Eriophyoidea) from Tamarisks. Forests 2024, 15, 715. [Google Scholar] [CrossRef]
- Chetverikov, P.E.; Bertone, M. First rhyncaphytoptine mite (Eriophyoidea, Diptilomiopidae) parasitizing american hazelnut (Corylus americana): Molecular identification, confocal microscopy, and phylogenetic position. Exp. Appl. Acarol. 2022, 88, 75–95. [Google Scholar] [CrossRef]
- Nuzzaci, G.; Alberti, G. Internal anatomy and physiology. In Eriophyoid Mites: Their Biology, Natural Enemies and Control; Lindquist, E.E., Sabelis, M.W., Bruin, J., Eds.; World Crop Pests; Elsevier Science Publishing: Amsterdam, The Netherlands, 1996; Volume 6, pp. 101–150. [Google Scholar] [CrossRef]
- Manson, D.C.M. Eriophyinae (Arachnida: Acari: Eriophyoidea); Fauna of New Zealand, No 5; Science Information Publishing Centre, DSIR: Wellington, New Zealand, 1984; 128p. [Google Scholar]
- Britto, E.P.; Gondim, M.G., Jr.; Navia, D.; Flechtmann, C.H.W. A new deuterogynous eriophyid mite (Acari: Eriophyidae) with dimorphic males from Caesalpinia echinata (Caesalpiniaceae) from Brazil: Description and biological observations. Int. J. Acarol. 2008, 34, 307–316. [Google Scholar] [CrossRef]
- Knihinicki, D.K.; Boczek, J. New eriophyoid mites (Acari: Eriophyoidea) from Australia. Int. J. Acarol. 2002, 28, 241–249. [Google Scholar] [CrossRef]
- Elhalawany, A.S.; Amrine, J.W.; Ueckermann, E.A. A new species and new record of eriophyoid mites (Trombidiformes: Eriophyoidea) from mango in Egypt with a note on the population dynamics of four eriophyoid species. Acarines 2021, 15, 1–22. [Google Scholar] [CrossRef]
- Giordano, T.; Andolina, F.; Cerasa, G.; Marotta, I.; Rizzo, R.; Verde, G.L.; de Lillo, E.; Tsolakis, H. First report of mango leaf-coating mite Cisaberoptus kenyae (Acari: Eriophyidae) in Europe. Acarologia 2025, 65, 927–933. [Google Scholar] [CrossRef]
- Hassan, E.F.O.; Keifer, H.H. Mango leaf-coating mite, Cisaberoptus kenyae K. (Eriophyidae, Aberoptinae). Pan-Pac. Entomol. 1978, 54, 185–193. [Google Scholar]
- Navia, D.; Flechtmann, C.H.W. Eriophyid mites (Acari: Prostigmata) from mango, Mangifera indica L., in Brazil. Int. J. Acarol. 2000, 26, 73–80. [Google Scholar] [CrossRef]
- Keifer, H.H. Eriophyid Studies C-11; Agricultural Research Service, U.S. Department of Agriculture: Washington, DC, USA, 1976; 24p.
- Knorr, L.C.; Phatak, H.C.; Keifer, H.H. Web-spinning eriophyid mites. J. Wash. Acad. Sci. 1976, 66, 228–234. [Google Scholar]
- Zhang, Q.; Lu, Y.-W.; Liu, X.-Y.; Li, Y.; Gao, W.-N.; Sun, J.T.; Hong, X.-Y.; Sao, R.; Xue, X.-F. Phylogenomics resolves the higher-level phylogeny of herbivorous eriophyoid mites (Acariformes: Eriophyoidea). BMC Biol. 2024, 22, 70. [Google Scholar] [CrossRef]
- Amrine, J.W., Jr.; Manson, D.C.M. Preparation, mounting and descriptive study of eriophyoid mites. In Eriophyoid Mites: Their Biology, Natural Enemies and Control; Lindquist, E.E., Sabelis, M.W., Bruin, J., Eds.; World Crop Pests; Elsevier Science Publishing: Amsterdam, The Netherlands, 1996; Volume 6, pp. 383–396. [Google Scholar] [CrossRef]
- GBIF Secretariat. GBIF Backbone Taxonomy. Checklist Dataset. 2023. Available online: https://doi.org/10.15468/39omei (accessed on 18 February 2026).
- Warschefsky, E.J.; von Wettberg, E.J. Population genomic analysis of mango (Mangifera indica) suggests a complex history of domestication. New Phytol. 2019, 222, 2023–2037. [Google Scholar] [CrossRef]
- Chetverikov, P.E. Confocal laser scanning microscopy technique for the study of internal genitalia and external morphology of eriophyoid mites (Acari: Eriophyoidea). Zootaxa 2012, 3453, 56–68. [Google Scholar] [CrossRef]
- Keifer, H.H. Eriophyid Studies B-18; State Bureau of Entomology, California Department of Agriculture: Sacramento, CA, USA, 1966; pp. 1–20.
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Klimov, P.B.; OConnor, B.M.; Chetverikov, P.E.; Bolton, S.J.; Pepato, A.R.; Mortazavi, A.L.; Tolstikov, A.V.; Bauchan, G.R.; Ochoa, R. Comprehensive phylogeny of acariform mites (Acariformes) provides insights on the origin of the four-legged mites (Eriophyoidea), a long branch. Mol. Phylogenet. Evol. 2018, 119, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Gankevich, V.D.; Chetverikov, P.E. Mitogenomic evidence for the monophyly of blackcurrant gall mite subfamily Cecidophyinae (Eriophyoidea, Eriophyidae). Exp. Appl. Acarol. 2025, 95, 9. [Google Scholar] [CrossRef]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT online service: Multiple sequence alignment, interactive sequence choice and visualization. Brief. Bioinform. 2017, 20, 1160–1166. [Google Scholar] [CrossRef]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [PubMed]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Marinković, S.M.; Chetverikov, P.E.; Hörweg, C.; Petanović, R.U. Supplementary description of three species from the subfamily Cecidophyinae (Eriophyoidea: Eriophyidae) from the Nalepa collection. Syst. Appl. Acarol. 2018, 23, 838–859. [Google Scholar] [CrossRef]
- Marinković, S.M.; Chetverikov, P.E.; Cvrković, T.; Vidović, B.; Petanović, R. Supplementary description of five species from the genus Cecidophyopsis (Eriophyoidea: Eriophyidae: Cecidophyinae). Syst. Appl. Acarol. 2019, 24, 1555–1578. [Google Scholar] [CrossRef]
- Marinković, S.M.; Cvrković, T.; Jakovljević, M.; Vidović, B.; Petanović, R.U. Cecidophyinae (Acari: Acariformes) from Serbia: Description of a new species, supplementary description of six species and four new records for the fauna. Syst. Appl. Acarol. 2023, 28, 1855–1877. [Google Scholar] [CrossRef]
- Neravathu, R. Feeding impact of Cisaberoptus kenyae Keifer (Acari: Eriophyidae) on photosynthetic efficiency and biochemical parameters of Mangifera indica L. Acarol. Stud. 2019, 1, 84–94. [Google Scholar]
- Goodale, E.; Beauchamp, G.; Ruxton, G.D. Mixed-Species Groups of Animals: Behavior, Community Structure, and Conservation; Elsevier Academic Press: London, UK, 2017; 205p. [Google Scholar] [CrossRef]
- Escalante, I.; Domínguez, M.; Gómez-Ruiz, D.A.; Machado, G. Benefits and Costs of Mixed-Species Aggregations in Harvestmen (Arachnida: Opiliones). Front. Ecol. Evol. 2022, 9, 766323. [Google Scholar] [CrossRef]
- Agnarsson, I. Spiders as superhosts and secondary kleptoparasites. Front. Arachnid Sci. 2025, 4, 1544428. [Google Scholar] [CrossRef]
- Chandrapatya, A.; Konvipasruang, P.; Amrine, J.W. Catalog of Thai Eriophyoidea (Acari: Prostigmata) with Illustrations and Keys to Genera of Thai Mites; Ku Digital Printing Center, Kasetsart University: Bangkok, Thailand, 2017; 526p. [Google Scholar]
- Manson, D.C.M.; Oldfield, G.N. Biology and ecology 1.4.1. Life forms, deuterogyny, diapause and seasonal development. In Eriophyoid Mites: Their Biology, Natural Enemies and Control; Lindquist, E.E., Sabelis, M.W., Bruin, J., Eds.; World Crop Pests; Elsevier Science Publishing: Amsterdam, The Netherlands, 1996; Volume 6, pp. 173–183. [Google Scholar] [CrossRef]
- Sankaran, M.; Dinesh, M.R.; Abirami, K.; Murugan, C. Botany of Mango. In The Mango Genome; Kole, C., Ed.; Compendium of Plant Genomes; Springer International Publishing: Cham, Switzerland, 2021. [Google Scholar] [CrossRef]
- Liang, Q.; Pan, H.; He, X.; Wang, S.; Hou, Y.; Xiao, H.; Xu, G.; Yi, R.; Lin, D.; Yang, Z. Population structure and genetic diversity of mango (Mangifera indica L.) germplasm resources as revealed by single-nucleotide polymorphism markers. Front. Plant Sci. 2024, 15, 1328126. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.