The Performance of Protein Meal from Hermetia illucens Larvae in Hetero-Clarias Hybrid Farming
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
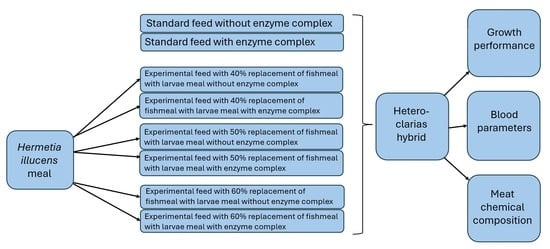
2.1. Diet Preparation and Experimental Design
2.2. Productive Performance Evaluations
2.3. Sampling Procedure
2.4. Hematological Analysis
2.5. Biochemical Analysis of Serum
2.6. Proximate Body Composition
2.7. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DM | Dry matter |
| LM− | Control group without Hostazyme X supplementation |
| LM+ | Control group with Hostazyme X supplementation |
| LE40− | Experimental group with 40% replacement of fishmeal with H. illucens larvae meal without Hostazyme X supplementation |
| LE40+ | Experimental group with 40% replacement of fishmeal with H. illucens larvae meal with Hostazyme X supplementation |
| LE50− | Experimental group with 50% replacement of fishmeal with H. illucens larvae meal without Hostazyme X supplementation |
| LE50+ | Experimental group with 50% replacement of fishmeal with H. illucens larvae meal with Hostazyme X supplementation |
| LE60− | Experimental group with 60% replacement of fishmeal with H. illucens larvae meal without Hostazyme X supplementation |
| LE60+ | Experimental group with 60% replacement of fishmeal with H. illucens larvae meal with Hostazyme X supplementation |
| ME | Metabolizable energy |
| SGR | Specific growth rate |
| FCR | Feed conversion ratio |
| FI | Daily feed intake |
| DWG | Daily weight gain |
| WG | Percentage weight gain |
| SR | Survival rate |
| PI | Production index |
| MGR | Metabolic growth rate |
| SOD | Superoxide dismutase |
| GPx | Glutathione peroxidase |
| ALAT | Alanine aminotransferase |
| ASAT | Aspartate aminotransferase |
References
- FAO. The State of World Fisheries and Aquaculture 2022. Towards Blue Transformation; FAO: Rome, Italy, 2022; Available online: https://doi.org/10.4060/cc0461en (accessed on 18 November 2025). [CrossRef]
- FAO.org. Available online: https://www.fao.org/publications/fao-flagship-publications/the-state-of-world-fisheries-and-aquaculture/en (accessed on 20 June 2025).
- OECD.org. Available online: https://www.oecd.org/en/publications/2021/07/oecd-fao-agricultural-outlook-2021-2030_31d65f37.html (accessed on 13 July 2025).
- Gopakumar, K.; Nai, M.R. Fatty acid composition of eight species of Indian marine fish. J. Sci. Food Agric. 1972, 23, 493–496. [Google Scholar] [CrossRef]
- Skaara, T.; Regenstein, J.M. The structure and properties of myofibrillar proteins in beef, poultry, and fish. J. Muscle Foods 1990, 1, 269–291. [Google Scholar] [CrossRef]
- Venugopal, V.; Shahidi, F. Structure and composition of fish muscle. Food Rev. Int. 1996, 12, 175–197. [Google Scholar] [CrossRef]
- Shaviklo, A.R. Development of fish protein powder as an ingredient for food applications: A review. J. Food Sci. Technol. 2013, 52, 648–661. [Google Scholar] [CrossRef] [PubMed]
- Henry, M.; Gasco, L.; Piccolo, G.; Fountoulaki, E. Review on the use of insects in the diet of farmed fish: Past and future. Anim. Feed. Sci. Technol. 2015, 203, 1–22. [Google Scholar] [CrossRef]
- Papuc, T.; Boaru, A.; Ladosi, D.; Struti, D.; Georgescu, B. Potential of black soldier fly (Hermetia illucens) as alternative protein source in salmonid feeds—A review. Indian J. Fish 2020, 67, 160–170. [Google Scholar] [CrossRef]
- Barroso, F.G.; Haro, C.; Sánchez-Muros, M.-J.; Venegas, E.; Martínez-Sánchez, A.; Pérez-Bañón, C. The potential of various insect species for use as food for fish. Aquaculture 2014, 422–423, 193–201. [Google Scholar] [CrossRef]
- Chia, S.Y.; Tanga, C.M.; Osuga, I.M.; Cheseto, X.; Ekesi, S.; Dicke, M.; van Loon, J.J. Nutritional composition of black soldier fly larvae feeding on agro-industrial by-products. Entomol. Exp. Appl. 2020, 168, 472–481. [Google Scholar] [CrossRef]
- Magee, K.; Halstead, J.; Small, R.; Young, I. Valorisation of organic waste by-products using black soldier fly (Hermetia illucens) as a bio-convertor. Sustainability 2021, 13, 8345. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Anim. Feed Sci. Technol. 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Georgescu, B.; Boaru, A.M.; Muntean, L.; Sima, N.; Struți, D.I.; Păpuc, T.A.; Georgescu, C. Modulating the fatty acid profiles of Hermetia illucens larvae fats by dietary enrichment with different oilseeds: A sustainable way for future use in feed and food. Insects 2022, 13, 801. [Google Scholar] [CrossRef]
- Lu, S.; Taethaisong, N.; Meethip, W.; Surakhunthod, J.; Sinpru, B.; Sroichak, T.; Archa, P.; Thongpea, S.; Paengkoum, S.; Purba, R.A.P.; et al. Nutritional composition of black soldier fly larvae (Hermetia illucens L.) and its potential uses as alternative protein sources in animal diets: A review. Insects 2022, 13, 831. [Google Scholar] [CrossRef]
- da-Silva, W.C.; Silva, É.B.R.d.; Silva, J.A.R.d.; Martorano, L.G.; Belo, T.S.; Sousa, C.E.L.; Camargo-Júnior, R.N.C.; Andrade, R.L.; Santos, A.G.d.S.; de Carvalho, K.C.; et al. Nutritional value of the larvae of the black soldier fly (Hermetia illucens) and the house fly (Musca domestica) as a food alternative for farm animals—A systematic review. Insects 2024, 15, 619. [Google Scholar] [CrossRef]
- Srifawattana, N.; Phimolsiripol, Y.; Boonchuay, P.; Na-Lampang, K.; Piboon, P.; Umsumarng, S.; Nganvongpanit, K. Black soldier fly (Hermetia illucens) larvae as a protein substitute in adverse food reactions for canine dermatitis: Preliminary results among patients. Vet. Sci. 2025, 12, 68. [Google Scholar] [CrossRef]
- Huang, J.; Yu, T.; Yuan, B.; Xiao, J.; Huang, D. The addition of Hermetia illucens to feed: Influence on nutritional composition, protein digestion characteristics, and antioxidant activity of Acheta domesticus. Foods 2025, 14, 1140. [Google Scholar] [CrossRef]
- Renna, M.; Schiavone, A.; Gai, F.; Dabbou, S.; Lussiana, C.; Malfatto, V.; Prearo, M.; Capucchio, M.T.; Biasato, I.; Biasibetti, E.; et al. Evaluation of the suitability of a partially defatted black soldier fly (Hermetia illucens L.) larvae meal as ingredient for rainbow trout (Oncorhynchus mykiss Walbaum) diets. J. Anim. Sci. Biotechnol. 2017, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Stamer, A.; Wesselss, S.; Neidigk, R.; Hoerstgen-Schwark, G. Black soldier fly (Hermetia illucens) larvae-meal as an example for a new feed ingredients’ class in aquaculture diets. In Proceedings of the 4th ISOFAR Scientific Conference, ‘Building Organic Bridges’, at the Organic World Congress, Istanbul, Turkey, 13–15 October 2014. [Google Scholar]
- Sobczak, M.; Panicz, R.; Sadowski, J.; Półgęsek, M.; Żochowska-Kujawska, J. Does production of Clarias gariepinus × Heterobranchus longifilis hybrids influence quality attributes of fillets? Foods 2022, 11, 2074. [Google Scholar] [CrossRef]
- Fawole, F.J.; Adeoye, A.A.; Tiamiyu, L.O.; Ajala, K.I.; Obadara, S.O.; Ganiyu, I.O. Substituting fishmeal with Hermetia illucens in the diets of African catfish (Clarias gariepinus): Effects on growth, nutrient utilization, haemato-physiological response, and oxidative stress biomarker. Aquaculture 2020, 518, 734849. [Google Scholar] [CrossRef]
- Adeoye, A.A.; Akegbejo-Samsons, Y.; Fawole, F.J.; Davies, S.J. Preliminary assessment of black soldier fly (Hermetia illucens) larval meal in the diet of African catfish (Clarias gariepinus): Impact on growth, body index, and hematological parameters. J. World Aquac. Soc. 2020, 51, 1024–1033. [Google Scholar] [CrossRef]
- Bartucz, T.; Csókás, E.; Nagy, B.; Gyurcsák, M.P.; Bokor, Z.; Bernáth, G.; Molnár, J.; Urbányi, B.; Csorbai, B. Black soldier fly (Hermetia illucens) meal as direct replacement of complex fish feed for rainbow trout (Oncorhynchus mykiss) and African catfish (Clarias gariepinus). Life 2023, 13, 1978. [Google Scholar] [CrossRef]
- Bake, G.G.; Ajibade, D.O.; Gana, A.B.; Yakubu, F.B.; Samaila, J.; Abdulkarim, I.A.; Igili, O.E.; Sadiku, S.; Gatlin, D.M. Growth performance, body composition, and apparent nutrient digestibility of hybrid catfish fingerlings fed with blended insect meal. Niger. J. Fish. 2024, 18, 2118–2128. [Google Scholar]
- Sándor, Z.J.; Banjac, V.; Vidosavljević, S.; Káldy, J.; Egessa, R.; Lengyel-Kónya, E.; Tömösközi-Farkas, R.; Zalán, Z.; Adányi, N.; Libisch, B.; et al. Apparent digestibility coefficients of black soldier fly (Hermetia illucens), yellow mealworm (Tenebrio molitor), and blue bottle fly (Calliphora vicina) insects for juvenile African catfish hybrids (Clarias gariepinus × Heterobranchus longifilis). Aquac. Nutr. 2022, 2022, 4717014. [Google Scholar] [CrossRef]
- Smets, R.; Verbinnen, B.; Van de Voorde, I.; Aerts, G.; Claes, J.; van der Borght, M. Sequential extraction and characterisation of lipids, proteins, and chitin from black soldier fly (Hermetia illucens) larvae, prepupae, and pupae. Waste Biomass Valor. 2020, 11, 6455–6466. [Google Scholar] [CrossRef]
- Rapatsa, M.M.; Moya, N.A.G. Enzyme activity and histological analysis of Clarias gariepinus fed on Imbrasia belina meal used for partial replacement of fishmeal. Fish Physiol. Biochem. 2019, 45, 1309–1320. [Google Scholar] [CrossRef]
- Ajayi, A.A.; Onibokun, E.A.; Adedeji, O.M.; George, F.O.A. Characterization of chitinase from the African catfish, Clarias gariepinus (Burchell, 1822). Int. J. Adv. Sci. Eng. Technol. 2015, 3, 89–96. [Google Scholar]
- Libisch, B.; Sándor, Z.J.; Keresztény, T.; Ozoaduche, C.L.; Papp, P.P.; Posta, K.; Biró, J.; Stojkov, V.; Banjac, V.; Adányi, N.; et al. Effects of short-term feeding with diets containing insect meal on the gut microbiota of African catfish hybrids. Animals 2025, 15, 1338. [Google Scholar] [CrossRef]
- Kroeckel, S.; Harjes, A.-G.E.; Roth, I.; Katz, H.; Wuertz, S.; Susenbeth, A.; Schulz, C. When a turbot catches a fly: Evaluation of a pre-pupae meal of the black soldier fly (Hermetia illucens) as fish meal substitute—Growth performance and chitin degradation in juvenile turbot (Psetta maxima). Aquaculture 2012, 364–365, 345–352. [Google Scholar] [CrossRef]
- Liang, Q.; Yuan, M.; Xu, L.; Lio, E.; Zhang, F.; Mou, H.; Secundo, F. Application of enzymes as a feed additive in aquaculture. Mar. Life Sci. Technol. 2022, 4, 208–221. [Google Scholar] [CrossRef]
- Hogsette, J.A. New diets for production of house flies and stable flies (Diptera: Muscidae) in the laboratory. J. Econ. Entomol. 1992, 85, 2291–2294. [Google Scholar] [CrossRef]
- National Research Council. Nutrient Requirements of Fish and Shrimp; The National Academies Press: Washington, DC, USA, 2011; 392p. [Google Scholar]
- Adeoye, A.A.; Akegbejo-Samsons, Y.; Fawole, F.J.; Olatunji, P.O.; Muller, N.; Wan, A.H.; Davies, S.J. From waste to feed: Dietary utilisation of bacterial protein from fermentation of agricultural wastes in African catfish (Clarias gariepinus) production and health. Aquaculture 2021, 531, 735850. [Google Scholar] [CrossRef]
- Bain, L. Statistical Analysis of Reliability and Life-Testing Models Theory and Methods, 2nd ed.; Routledge: New York, NY, USA, 1991. [Google Scholar]
- Fernandes, I.M.; Bastos, Y.F.; Barreto, D.S.; Lourenço, L.S.; Penha, J.M. The efficacy of clove oil as an anaesthetic and in euthanasia procedure for small-sized tropical fishes. Braz. J. Biol. 2016, 77, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Buzek, J.; Chastel, O. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes Text with EEA relevance. Off. J. Eur. Union 2010, 276, 33–74. [Google Scholar]
- AOAC. Official Methods of Analysis, 18th ed.; AOAC International: Rockville, MD, USA, 2005. [Google Scholar]
- Engle, R. GARCH 101: The use of ARCH/GARCH models in applied econometrics. J. Econ. Perspect. 2001, 15, 157–168. [Google Scholar] [CrossRef]
- Teye-Gaga, C.; Molnár, P.I.; Kertész, A.; Fehér, M.; Bársony, P. Preliminary assessment of BSF larval-based diets on growth performance of hybrid African catfish Heteroclarias. In Proceedings of the Application of One Health Concept in Farm Animal Nutrition: 21st International Symposium on Animal Nutrition and One Health Day, Kaposvár, Hungary, 6 November 2024; 2024. [Google Scholar]
- Tuychiev, K.; Khujamov, S. Effect of black soldier fly larvae meal on African catfish growth. Egypt. J. Aquat. Biol. Fish. 2025, 29, 935–944. [Google Scholar] [CrossRef]
- Mundida, G.B.; Manyala, J.O.; Madzimure, J.; Rono, K. Growth and economic performance of African catfish (Clarias gariepinus Burchell, 1822) fed diets containing black soldier fly larvae (Hermetia illucens Linnaeus, 1758). East Afr. J. Agric. Biotechnol. 2023, 6, 332–344. [Google Scholar] [CrossRef]
- Bonomini, M.G.; Prandi, B.; Caligiani, A. Black soldier fly (Hermetia illucens L.) whole and fractionated larvae: In vitro protein digestibility and effect of lipid and chitin removal. Food Res. Int. 2024, 196, 115102. [Google Scholar] [CrossRef]
- Tacon, A.G.J. Nutrițional Fish Pathology: Morphological Sings of Nutrient Deficiency and Toxicity in Farmed Fish; FAO Fisheries Technical Paper 330; Food and Agriculture Organization of the United Nations: Rome, Italy, 1992. [Google Scholar]
- Esmaeili, M. Blood performance: A new formula for fish growth and health. Biology 2021, 10, 1236. [Google Scholar] [CrossRef]
- Argungu, L.A.; Hashimu, J.; Magami, I.M.; Abdullahi, Y.M.; Ahmed, J.M. Seasonal influence on haematological parameters of male and female Heterobranchus bidorsalis (Geoffroy Saint-Hilaire, 1809) in river Rima Sokoto, Nigeria. Int. J. Fish. Aquac. Res. 2021, 7, 68–75. [Google Scholar]
- Marín-García, P.B.; Llobat, L.; López-Lujan, M.C.; Cambra-López, M.; Blas, E.; Pascual, J.J. Urea nitrogen metabolite can contribute to implementing the ideal protein concept in monogastric animals. Animals 2022, 12, 2344. [Google Scholar] [CrossRef] [PubMed]
- Chew, S.F.; Wilson, J.M.; Ip, Y.K.; Randall, D.J. Nitrogen excretion and defense against ammonia toxicity. Fish Physiol. 2005, 21, 307–395. [Google Scholar]
- Okorie-Kanu, C.O.; Unakalamba, N.J. Normal haematological and blood biochemistry values of cultured Heteroclarias hybrid in South East, Nigeria. Comp. Clin. Pathol. 2015, 24, 1445–1450. [Google Scholar] [CrossRef]
- Grădinariu, L.; Crețu, M.; Vizireanu, C.; Dediu, L. Oxidative stress biomarkers in fish exposed to environmental concentrations of pharmaceutical pollutants: A review. Biology 2025, 14, 472. [Google Scholar] [CrossRef] [PubMed]
- Adeshina, I.; Jenyo-Oni, A.; Emikpe, B.O. Use of Eugenia cayrophyllata oil as anaesthetic in farm raised African catfish Clarias gariepinus juveniles. Egypt. J. Exp. Biol. 2016, 12, 71–76. [Google Scholar]
| Specification | Experimental Groups | |||||||
|---|---|---|---|---|---|---|---|---|
| LC− | LC+ | LE40− | LE40+ | LE50− | LE50+ | LE60− | LE60+ | |
| Experimental diets (% in feed) | ||||||||
| Wheat gluten | 25.0 | 25.0 | 26.0 | 26.0 | 26.6 | 26.6 | 27.1 | 27.1 |
| Starch | 24.0 | 24.0 | 23.0 | 23.0 | 22.4 | 22.4 | 21.9 | 21.9 |
| Soybean meal | 22.0 | 22.0 | 22.0 | 22.0 | 22.0 | 22.0 | 22.0 | 22.0 |
| Fishmeal | 12.0 | 12.0 | 7.2 | 7.2 | 6.0 | 6.0 | 4.8 | 4.8 |
| Fish oil | 11.0 | 11.0 | 11.0 | 11.0 | 11.0 | 11.0 | 11.0 | 11.0 |
| Blood meal | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| H. illucens meal | - | - | 4.8 | 4.8 | 6.0 | 6.0 | 7.2 | 7.2 |
| Premix vit-min | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Enzymes | - | 0.02 | - | 0.02 | - | 0.02 | - | 0.02 |
| Nutritive value | ||||||||
| ME (kcal/kg) * | 4350.20 | 4350.20 | 4414.20 | 4414.20 | 4431.60 | 4431.60 | 4448.60 | 4448.60 |
| Moisture (%) | 8.6 | 8.6 | 8.05 | 8.05 | 7.73 | 7.73 | 7.34 | 7.34 |
| Crude protein (%) | 43.90 | 43.90 | 43.73 | 43.73 | 43.96 | 43.96 | 44.12 | 44.12 |
| Crude fat (%) | 2.00 | 2.00 | 2.84 | 2.84 | 3.05 | 3.05 | 3.26 | 3.26 |
| Fiber (%) | 1.01 | 1.01 | 1.43 | 1.43 | 1.54 | 1.54 | 1.65 | 1.65 |
| Ash (%) | 2.81 | 2.81 | 2.84 | 2.84 | 2.85 | 2.85 | 2.86 | 2.86 |
| Specification | Initial Weight | Final Weight | DFI (g/ind/day) | DWG (g/day) | FCR | SR% |
|---|---|---|---|---|---|---|
| Larval meal effect | ||||||
| LC− | 11.35 ± 0.32 a | 27.39 ± 1.07 a | 0.39 ± 0.03 a | 0.19 ± 0.01 a | 2.02 ± 0.16 a | 96.67 ± 3.33 a |
| LE40− | 11.19 ± 0.34 a | 30.85 ± 1.25 b | 0.40 ± 0.03 a | 0.24 ± 0.01 b | 1.65 ± 0.13 b | 100 a |
| LE50− | 11.39 ± 0.31 a | 29.26 ± 1.14 b | 0.38 ± 0.03 a | 0.22 ± 0.02 ab | 1.81 ± 0.20 c | 100 a |
| LE60− | 11.17 ± 0.33 a | 29.19 ± 1.18 b | 0.38 ± 0.03 a | 0.22 ± 0.01 ab | 1.77 ± 0.18 c | 100 a |
| Enzyme effect | ||||||
| LC+ | 11.43 ± 0.25 a | 26.9 ± 1.15 a | 0.39 ± 0.04 a | 0.20 ± 0.02 a | 2.07 ± 0.21 a | 96.67 ± 3.33 a |
| LE40+ | 11.75 ± 0.38 a | 34.09 ± 1.49 c | 0.43 ± 0.03 a | 0.30 ± 0.02 c | 1.44 ± 0.10 d | 96.67 ± 3.33 a |
| LE50+ | 11.73 ± 0.29 a | 32.45 ± 1.08 d | 0.42 ± 0.03 a | 0.26 ± 0.02 d | 1.62 ± 0.11 b | 100 a |
| LE60+ | 11.44 ± 0.31 a | 31.55 ± 1.14 d | 0.42 ± 0.03 a | 0.25 ± 0.01 b | 1.68 ± 0.15 b | 100 a |
| p-value | ||||||
| Meal effect | 0.820 | 0.001 | 0.199 | 0.001 | 0.001 | 0.158 |
| Enzyme effect | 0.523 | 0.012 | 0.103 | 0.001 | 0.049 | 0.912 |
| Meal × enzyme | 0.524 | 0.334 | 0.561 | 0.001 | 0.043 | 0.794 |
| Specification | PI | SGR% | MGR | WG% |
|---|---|---|---|---|
| Insect meal effect | ||||
| LC− | 18.28 ± 6.60 a | 1.09 ± 0.07 a | 4.51 ± 0.91 a | 132.08 ± 33.48 a |
| LE40− | 22.97 ± 8.25 bc | 1.31 ± 0.04 b | 5.29 ± 1.06 b | 169.96 ± 41.67 b |
| LE50− | 21.17 ± 7.40 ab | 1.17 ± 0.06 ac | 4.97 ± 1.15 ab | 154.30 ± 44.10 ab |
| LE60− | 21.03 ± 7.22 ab | 1.2 ± 0.07 acd | 4.98 ± 0.79 ab | 155.27 ± 31.26 ab |
| Enzyme effect | ||||
| LC+ | 18.02 ± 7.83 a | 1.08 ± 0.07 a | 4.45 ± 1.03 a | 129.40 ± 44.62 a |
| LE40+ | 27.00 ± 10.55 d | 1.31 ± 0.06 b | 5.72 ± 1.58 b | 188.42 ± 62.36 b |
| LE50+ | 24.58 ± 6.45 e | 1.26 ± 0.04 bcd | 5.48 ± 0.83 b | 174.34 ± 35.13 b |
| LE60+ | 23.16 ± 7.40 ce | 1.22 ± 0.05 bcd | 5.18 ± 0.75 ab | 157.63 ± 30.83 ab |
| p-value | ||||
| Meal effect | 0.002 | 0.0001 | 0.001 | 0.001 |
| Enzyme effect | 0.061 | 0.0009 | 0.102 | 0.149 |
| Meal × enzyme | 0.621 | 0.0041 | 0.612 | 0.522 |
| Hb (g/dL) | Hct % | Ery (mil/mm3) | GPx (U/g Hb) | SOD (U/g Hb) | ALAT (U/L) | ASAT (U/L) | |
|---|---|---|---|---|---|---|---|
| Insect meal effect | |||||||
| LC− | 6.72 ± 0.20 a | 34.50 ± 1.19 a | 2.55 ± 0.25 a | 146.90 ± 2.61 bc | 1493.0 ± 6.32 a | 8.50 ± 0.55 ab | 26.70 ± 0.08 a |
| LE40− | 7.52 ± 0.01 a | 33.00 ± 2.37 a | 2.46 ± 0.17 a | 140.00 ± 0.79 ab | 1393.0 ± 17.16 bc | 9.95 ± 0.75 ab | 31.50 ± 1.26 ab |
| LE50− | 6.33 ± 0.27 a | 33.50 ± 0.40 a | 3.48 ± 0.30 a | 147.50 ± 5.30 bc | 1436.0 ± 8.20 ab | 11.00 ± 0.87 b | 34.48 ± 9.19 b |
| LE60− | 6.85 ± 0.06 a | 33.00 ± 2.37 a | 2.26 ± 0.03 a | 154.00 ± 7.12 c | 1568.5 ± 16.50 de | 15.10 ± 3.32 c | 45.85 ± 0.20 c |
| Enzyme effect | |||||||
| LC+ | 7.55 ± 0.30 a | 35.00 ± 1.98 a | 2.86 ± 0.02 a | 139.95 ± 7.23 a | 1407.0 ± 8.70 bf | 7.38 ± 0.18 a | 25.55 ± 0.75 a |
| LE40+ | 7.33 ± 0.08 a | 34.00 ± 0.79 a | 3.05 ± 0.08 a | 133.60 ± 5.06 a | 1298.0 ± 7.94 g | 8.95 ± 0.20 ab | 30.65 ± 2.02 ab |
| LE50+ | 7.60 ± 0.46 a | 35.00 ± 0.01 a | 3.44 ± 0.27 a | 144.75 ± 4.94 abc | 1356.5 ± 9.39 cfg | 10.15 ± 1.38 ab | 34.00 ± 3.08 b |
| LE60+ | 7.16 ± 0.28 a | 34.50 ± 0.40 a | 3.36 ± 0.0.07 a | 152.65 ± 7.19 bc | 1499.5 ± 10.40 ad | 14.55 ± 2.65 c | 44.35 ± 1.86 c |
| p-value | |||||||
| Meal effect | 0.189 | 0.138 | 0.238 | 0.001 | 0.001 | 0.001 | 0.001 |
| Enzyme effect | 0.093 | 0.597 | 0.101 | 0.009 | 0.001 | 0.100 | 0.389 |
| Meal × enzyme | 0.078 | 0.109 | 0.211 | 0.126 | 0.029 | 0.982 | 0.990 |
| Specification | Total Protein (g/dL) | Albumins (g/dL) | Urea (mg/dL) | Creatinine (mg/dL) | γ-Glob (g/dL) | Cholesterol (mg/dL) | Triglycerides (mg/dL) | Total Lipids (mg/dL) |
|---|---|---|---|---|---|---|---|---|
| Insect meal effect | ||||||||
| LC− | 4.63 ± 0.02 a | 1.17 ± 0.08 ab | 1.69 ± 0.59 a | 0.37 ± 0.05 a | 0.02 ± 0.01 a | 114.70 ± 2.85 ab | 218.70 ± 7.75 a | 437.3 ± 32.29 ab |
| LE40− | 3.36 ± 0.17 b | 1.12 ± 0.07 ab | 1.28 ± 1.86 c | 0.32 ± 0.01 acd | 0.018 ± 0.01 a | 103.05 ± 3.01 ae | 210.50 ± 9.37 a | 461.0 ± 21.35 ab |
| LE50− | 3.94 ± 0.30 de | 1.35 ± 0.01 c | 1.62 ± 0.79 ab | 0.33 ± 0.02 a | 0.014 ± 0.01 a | 129.50 ± 6.02 b | 298.75 ± 8.86 b | 604.7 ± 39.7 cd |
| LE60− | 4.14 ± 0.11 e | 1.63 ± 0.13 d | 1.69 ± 1.62 a | 0.35 ± 0.03 ad | 0.015 ± 0.01 a | 135.60 ± 5.03 b | 359.20 ± 6.41 b | 743.2 ± 57.51 c |
| Enzyme effect | ||||||||
| LC+ | 2.99 ± 0.19 c | 1.11 ± 0.09 a | 1.44 ± 1.34 bc | 0.25 ± 0.08 b | 0.02 ± 0.01 a | 109.60 ± 5.08 ae | 193.30 ± 7.12 a | 377.0 ± 44.27 ab |
| LE40+ | 2.90 ± 0.04 c | 1.10 ± 0.04 ab | 0.83 ± 0.32 d | 0.24 ± 0.01 b | 0.014 ± 0.01 a | 71.35 ± 3.31 cd | 205.45 ± 7.27 a | 408.1 ± 58.42 ab |
| LE50+ | 3.17 ± 0.25 bc | 1.27 ± 0.09 bc | 1.38 ± 0.83 c | 0.26 ± 0.01 bc | 0.021 ± 0.01 a | 95.05 ± 6.81 de | 219.10 ± 7.81 a | 506.0 ± 21.35 b |
| LE60+ | 3.70 ± 0.13 d | 1.49 ± 0.09 cd | 1.34 ± 1.03 c | 0.24 ± 0.02 b | 0.019 ± 0.01 a | 95.35 ± 4.72 a | 328.45 ± 5.31 b | 682.1 ± 30.52 cd |
| p-value | ||||||||
| Meal effect | 0.001 | 0.001 | 0.001 | 0.344 | 0.156 | 0.001 | 0.001 | 0.001 |
| Enzyme effect | 0.001 | 0.009 | 0.001 | 0.001 | 0.087 | 0.001 | 0.003 | 0.001 |
| Meal × enzyme | 0.001 | 0.481 | 0.147 | 0.495 | 0.127 | 0.017 | 0.117 | 0.796 |
| Specification | Dry Matter (%) | Crude Protein (%) | Crude Fat (%) | Ash (%) |
|---|---|---|---|---|
| Insect meal effect | ||||
| LC− | 20.19 ± 0.04 a | 12.61 ± 0.11 a | 1.40 ± 0.07 ab | 1.09 ± 0.05 ab |
| LE40− | 19.70 ± 0.34 a | 12.62 ± 0.14 a | 1.54 ± 0.04 abc | 1.07 ± 0.07 ab |
| LE50− | 20.63 ± 0.49 a | 12.78 ± 0.25 a | 1.63 ± 0.25 c | 1.01 ± 0.06 b |
| LE60− | 20.44 ± 0.08 a | 12.75 ± 0.63 a | 1.62 ± 0.02 c | 1.09 ± 0.02 a |
| Enzyme effect | ||||
| LC+ | 20.29 ± 0.28 a | 12.74 ± 0.10 a | 1.39 ± 0.06 a | 1.06 ± 0.02 ab |
| LE40+ | 19.84 ± 0.53 a | 12.77 ± 0.11 a | 1.53 ± 0.07 abc | 0.98 ± 0.02 bc |
| LE50+ | 20.30 ± 0.18 a | 12.86 ± 0.07 a | 1.44 ± 0.04 abc | 1.06 ± 0.03 ab |
| LE60+ | 20.24 ± 0.42 a | 12.82 ± 0.63 a | 1.64 ± 0.17 c | 1.08 ± 0.03 ab |
| p-value | ||||
| Meal effect | 0.136 | 0.532 | 0.001 | 0.003 |
| Enzyme effect | 0.491 | 0.203 | 0.219 | 0.101 |
| Meal × enzyme | 0.347 | 0.984 | 0.207 | 0.007 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgescu, B.; Georgescu, C.; Păpuc, T.; Vasiu, M.; Struți, D.; Boaru, A. The Performance of Protein Meal from Hermetia illucens Larvae in Hetero-Clarias Hybrid Farming. Insects 2025, 16, 1279. https://doi.org/10.3390/insects16121279
Georgescu B, Georgescu C, Păpuc T, Vasiu M, Struți D, Boaru A. The Performance of Protein Meal from Hermetia illucens Larvae in Hetero-Clarias Hybrid Farming. Insects. 2025; 16(12):1279. https://doi.org/10.3390/insects16121279
Chicago/Turabian StyleGeorgescu, Bogdan, Carmen Georgescu, Tudor Păpuc, Marius Vasiu, Dănuț Struți, and Anca Boaru. 2025. "The Performance of Protein Meal from Hermetia illucens Larvae in Hetero-Clarias Hybrid Farming" Insects 16, no. 12: 1279. https://doi.org/10.3390/insects16121279
APA StyleGeorgescu, B., Georgescu, C., Păpuc, T., Vasiu, M., Struți, D., & Boaru, A. (2025). The Performance of Protein Meal from Hermetia illucens Larvae in Hetero-Clarias Hybrid Farming. Insects, 16(12), 1279. https://doi.org/10.3390/insects16121279