Simple Summary
All the species in the Colocasiomyia gigantea group breed on monsteroid host plants (aroids in the subfamily Monsteroideae). So far, we have not resolved the phylogenetic relationship among these fly species, making it difficult to trace the origin and history of the species diversification, biogeography and host plant selection. In this study, we reconstructed the evolutionary relationships between these species using multilocus DNA sequence data, and we inferred their ancestral areas and host plants. According to the results, this group diverged from its sister taxon through a split between the northeastern Oriental region and Sundaland + Wallacea, with the subsequent diversification occurring largely in the first region. We inferred the most likely ancestral host genus of this group to be Rhaphidophora Hassk, with possible subsequent shifts to Scindapsus Schott and/or Epipremnum Schott. We discuss the potential of the group as a model system for studies in evolutionary ecology and developmental genetics.
Abstract
The gigantea species group of the genus Colocasiomyia de Meijere (Diptera: Drosophilidae) is among the four aroid-breeding species groups in this genus; however, it differs from the remaining three groups in the host use: all the flies in this group use plants from the subfamily Monsteroideae instead of from the subfamily Aroideae. So far, we have not resolved the phylogenetic relationship within this group, making it difficult to trace its geographical origin, pattern of species diversification and history of host plant use. In this study, we reconstructed the phylogenetic relationships within the C. gigantea group using DNA sequences of eight (two mitochondrial and six nuclear) gene markers, and we inferred the ancestral areas and host plants of the group based on the resulting phylogeny. According to the results, the C. gigantea group may have diverged from its sister group (i.e., the C. cristata group) through vicariance between the northeastern Oriental region and Sundaland + Wallacea, and the subsequent diversification of the C. gigantea group occurred mostly in the northeastern Oriental region, although an Oriental-to-Sundaland dispersal was followed by vicariance between these two areas, which finally gave rise to the C. gigantea-C. scindapsae lineage in the latter area. We inferred the most likely ancestral host plant of the C. gigantea group to be of the genus Rhaphidophora Hassk, with possible subsequent shifts to Scindapsus Schott and/or Epipremnum Schott plants. We discuss the potential for the egg filaments in the C. gigantea group to be used as a model system for comparative studies in pollination mutualism and developmental genetics concerning tubulogenesis.
1. Introduction
The genus Colocasiomyia de Meijere 1914 (Diptera, Drosophilidae) is an Old World pantropically distributed assemblage of 43 described and nearly 60 undescribed putatively new species, all feeding and breeding on the flowers of the lower angiosperms [1,2,3,4,5,6]. All these species were taxonomically assigned into six species groups, with each adapted to a specific host plant lineage: the crassipes group adapted to the family Magnoliaceae (the genus Michelia Linn.), the zeylanica group adapted to the family Arecaceae (the genus Pinanga Blume) and the remaining four groups adapted to the family Araceae: the toshiokai, baechlii and cristata groups adapted to the subfamily Aroideae, and the gigantea group adapted to the subfamily Monsteroideae (the genera Rhaphidophora Hassk, Epipremnum Schott and Scindapsus Schott, all belonging to the Rhaphidophora clade [7]) [3,4,5,8,9] (Table A1).
Since the 1980s, various researchers have investigated the phylogenetic relationships between the Colocasiomyia species via morphological comparison [1,2,3,4,10,11]. On the whole, a revised Colocasiomyia was supported as being monophyletic, with each of the four aroid-associated species groups (i.e., the cristata, toshiokai, baechlii and gigantea groups) and the zeylanica group of palm-breeding flies was supported. In a recent study [4], a cladistic analysis of 70 morphological characters of 24 species covering all six groups in Colocasiomyia was conducted, where the gigantea group (represented by C. gigantea, C. scindapsae and C. rhaphidophorae) was found to have the synapomorphy “foreleg tarsomere II elongated below”, and was placed as a sister to the cristata group. This synapomorphy in all the subsequently described species of the group, together with the appearance of egg filaments observed in the earliest member of the gigantea group, were confirmed in all the subsequently described species of the group, with the evolutionary significance of egg filaments readily recognized [4,5,8,9].
So far molecular phylogenetic analysis is still rare for Colocasiomyia, making it difficult to trace the evolutionary history of this genus. In a recent multilocus molecular phylogenetic analysis covering hundreds of species across the family Drosophilidae, a huge phylogenetic framework was constructed, in which the position of the genus Colocasiomyia was only poorly resolved. Among the three sampled species groups in this genus, i.e., gigantea, toshiokai and cristata, the former two were suggested with low confidence as closer to each other than either is to the third [12]. In the present study, we reconstructed the phylogenetic relationship in the C. gigantea group with DNA sequences of multiple gene loci and a full taxon sampling in the group. On the basis of this phylogenetic framework, we reconstructed and assessed the evolutionary history of this particular assemblage of species, paying special attention to the host use, biogeography and body sizes of the species.
2. Materials and Methods
2.1. DNA Markers, Taxon Sampling and DNA Sequencing
To reconstruct the phylogenetic relationship in the gigantea group, we used the same set of gene markers as in previous studies [6,13], including two mitochondrial genes (COI and COII: cytochrome c oxidase subunits I and II) and six nuclear genes (28S rRNA: 28S ribosomal RNA; ATPsyn-alpha: ATP synthases alpha gene; ATPsyn-gamma: ATP synthases gamma gene; alphaTub84b: alpha-Tublin at 84B; Hsc70cb: heat shock 70 kDa protein; EF-2: elongation factor 2) (Table 1). The corresponding sequences of five gigantea group species (i.e., C. gigantea, C. hailini, C. longifilamentata, C. longivalva and C. scindapsae), all used as outgroups in the abovementioned studies [6,13], were used again in this study, together with those of five species from the cristata group (i.e., colocasiae, cristata, sarawakana, xenalocasiae and ecornuta, representing the following lineages: clades I, IIa, IIb, IIc, and the independent branch leading to ecornuta, respectively), all of which were used as outgroup taxa considering their close morphological affinity to the gigantea group [4]. We did not consider the toshiokai groups, which was suggested as close to the gigantea group [12] since such a relationship was only weakly supported, as we discuss later in the Section 4.

Table 1.
Taxon sampling (and source of DNA sequences) in the present phylogenetic reconstruction.
We then determined the DNA sequences of the same set of markers for the remaining members of the gigantea group (i.e., C. yini, C. rhaphidophorae, C. liae, C. todai and C. daiae) (Table 1) using the same sets of PCR/sequencing primers as in a previous study [6], with adult specimens collected from host plants (or derived from eggs on host inflorescence) at varied sites in China (Table 1). We assembled the obtained trace files of the same target region in the SeqMan module of the DNAStar package version 7.1.0 (DNAStar Inc., Madison, WI, USA), with contigs made species by species, and manually examined and edited the ambiguities by eye. We then aligned the consensus sequences with the end nontarget regions trimmed, locus by locus, by using the CustalW tool in MEGA7 [14].
2.2. Data Partitioning and Model Selection
We concatenated the nucleotide sequences of all eight markers for the 15 sampled species, partitioned them into datasets by property (protein-coding gene: PCG vs. the 28S rRNA gene), source (nuclear vs. mitochondrial), locus and codon position (Table S1). For each dataset (or dataset combination), nucleotide substitution model was selected in MEGA7. We then selected the optimal data-partitioning strategy among ten competing ones according to the Bayes factor (BF) criterion [15]. For this, we performed two parallel MCMC runs for 106 generations (107 generations for the strategy P5) in the analysis of each strategy, allowing the average standard deviation (STDEV) of the split frequencies to fall well below 0.01. We analyzed the resulting trace files in Tracer 1.7.1 [16].
2.3. Phylogenetic Reconstruction
Using RAxML HPC [17], starting from random trees, we calculated 20 distinct ML trees for the gigantea group under the selected optimal partitioning strategy and the GTR-GAMMA model. We used the five species in cristata as the outgroup taxa, evaluated the branch confidences using 1000 rapid bootstrap replications and drew bipartitions from the 1000 bootstrapped trees onto the best-scoring ML tree. A 50% majority rule consensus Bayesian tree was constructed in MrBayes v 3.2.6 [18] with the same partitioning strategy as in the ML analysis through two runs of 107 MCMC generations. We generated a consensus tree after discarding the 20,000 initially sampled trees in either run.
2.4. Biogeographic Analyses
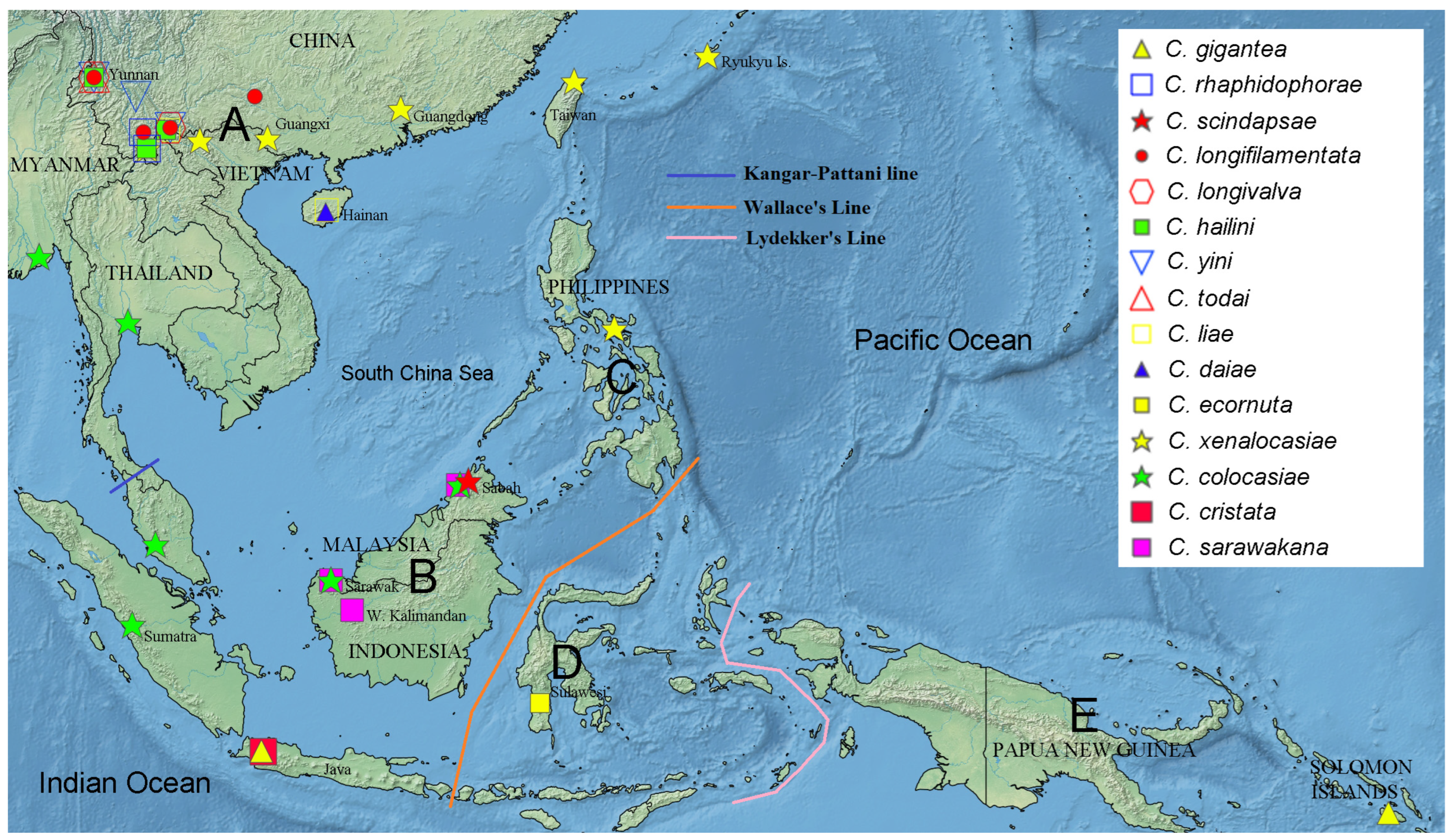
We used RASP version 4 [19] to reconstruct the biogeographic history of the gigantea group through S-DIVA (statistical dispersal-vicariance analysis) on the ML tree constructed above. For this, we compiled data on the geographical ranges of the ten species in the gigantea group and the outgroup species from the literature [2,3,4,5,6,8,9], with the total range divided into five areas that referred to the Kangar-Pattani Line [20], Wallace’s Line [21] and Lydekker’s Line [21] (Figure 1): A—the northeastern Oriental region (referred to as the “NE Oriental region” below for convenience) with southern China, including Hainan and Taiwan + Ryukyu Islands + Sino-Himalaya + Indochina Peninsula; B—Sundaland; C—the Philippines; D—Wallacea; E—the Australian region (referred to as “Australian” below). Then, a S-DIVA analysis was conducted with four allowed areas in ancestral distribution.
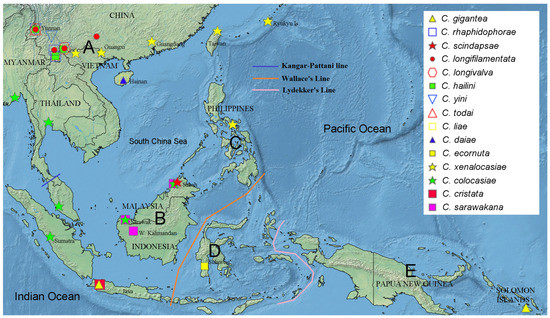
Figure 1.
Division of the five areas (A: NE Oriental region, B: Sundaland, C: the Philippines, D: Wallacea, E: Australian region) employed in the biogeographical analysis. Records of geographic distributions of all known species in the C. gigantea group and five representative species of C. cristata group employed in the present study are shown with legends on the map (created with Simplemappr (http://www.simplemappr.net/; accessed on 28 May 2022).
2.5. Reconstruction of the Evolution of Host Use and Body Size
We reconstructed the evolution of the host use and body size in the C. gigantea group with Mesquite V3.70 [22] on the ML tree constructed in the present study. For this, we created a standard categorial matrix for the 15 representative Colocasiomyia species and two characters: the host use and body size. For the host use, we discriminated four states (Table 2) by referring to the literature [3,5,8,9] or our unpublished data: 0 (the subfamily Aroideae), 1 (the genus Rhaphidophora), 2 (the genus Scindapsus) and 3 (the genus Epipremnum).

Table 2.
Statuses of the distribution areas, host plant uses and body sizes of ten species in the Colocasiomyia gigantea group and five representative species from the C. cristata group.
For the body size, we used the thorax length (ThL) (defined as the distance from the anterior notal margin to the apex of the scutellum [23]), with data compiled from the original descriptions of the type specimens in the taxonomic literature or newly collected (Table A2), For this character, we discriminated two states (small vs. large): 0 (small: ThL ≤ 1.0 mm) and 1 (large: ThL ≥ 1.2 mm). For Colocasiomyia liae, only two specimen types were employed based on their original descriptions [8]. In our analysis, we treated the state of the body size of this species as ambiguous (indicated with “?” by default). We then reconstructed the likelihood of the ancestral states at the nodes of the ML tree. We traced the history of the two characters: the host use and body size. We then edited and combined the resulting charts in Adobe® Photoshop® CS6.
3. Results
3.1. DNA Sequencing, Model Selection and Data Partitioning
3.1.1. DNA Sequencing
DNA sequences were newly determined for five species: yini, rhaphidophorae, liae, todai and daiae in the C. gigantea group (GenBank accession numbers: OM988102–106 for COII, OM943988–992 for 28S, ON107163–167 for ATPsyn-alpha, ON107168–172 for ATPsyn-gamma, ON107158–162 for alphaTub84B, ON157517–521 for Hsc70cb and ON157522–526 for EF-2; Table S2). The alignment of the concatenated sequences of all these loci and 15 species spanned 4750 nucleotide sites, among which 1127 were variable and 649 were parsimony informative.
3.1.2. Model Selection and Data Partitioning
The schemed DNA sequence data sets and respective selected models are shown in Table S1. Among the competitive partitioning strategies (Table 3), the strategy P5 (−lnL = 16,126.526) that making a distinction between the sources (mitochondrial vs. nuclear) and type (protein-coding or not) was selected as optimal by Bayes factor (Table 4).

Table 3.
Data-partitioning strategies and the mean-lnL and 95% credible interval of each strategy.

Table 4.
The 2ln BFs of comparisons between partitioning strategies (1 vs. 2). A positive value indicates evidence against the strategy 2.
3.2. Phylogenetic Relationship in the C. gigantea Group
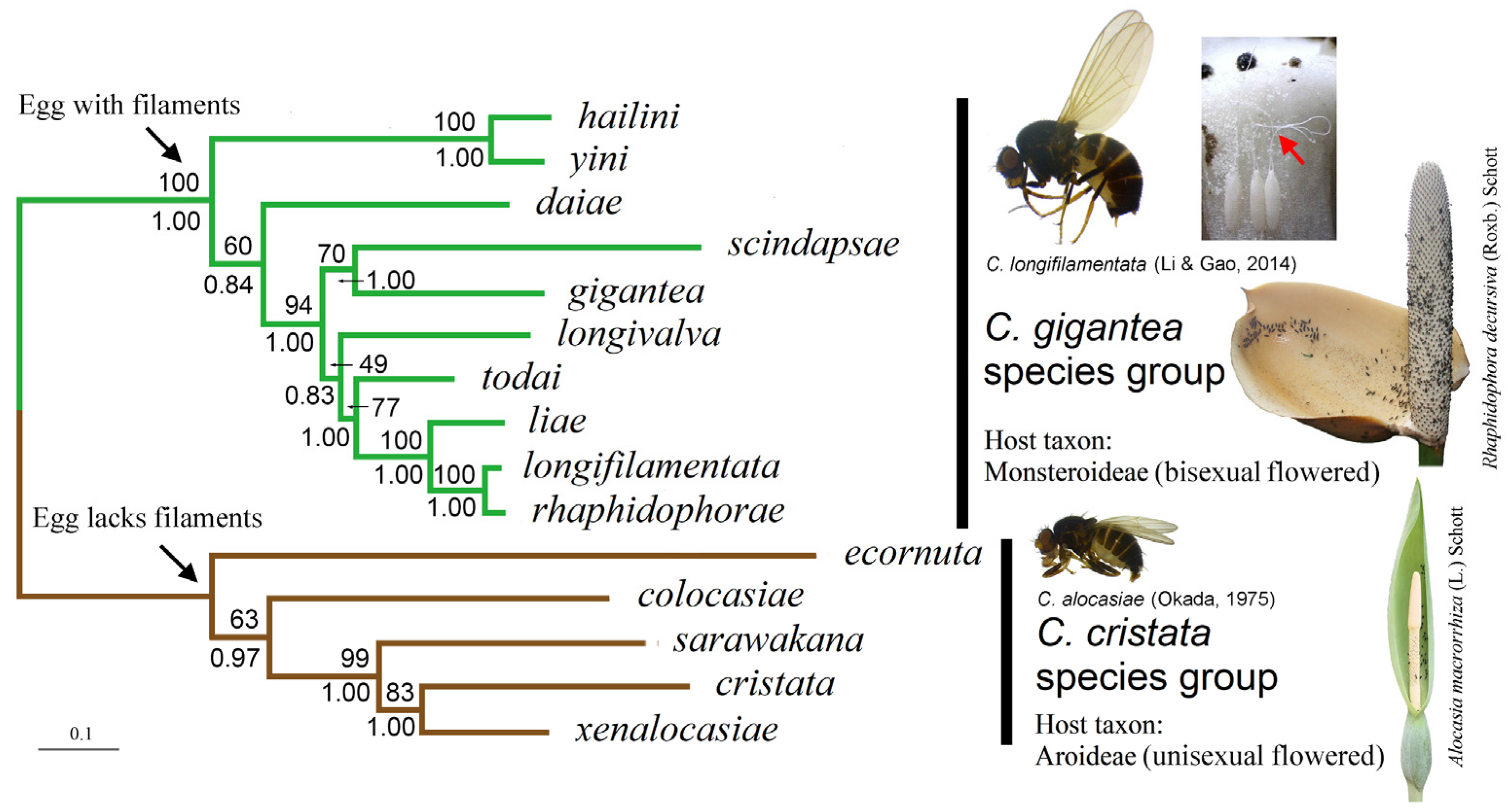
We present the ML tree of the C. gigantea group built with the concatenated DNA sequences of eight gene loci and the data-partitioning strategy P5 in Figure 2. The tree strongly supported (we considered any bootstrap percentage (BP) above 75% and/or a posterior probability (PP) above 0.95 as strong support) the mutually monophyletic relationship between the C. gigantea and C. cristata groups (BP = 100; PP = 1.00).

Figure 2.
ML tree of ten species in the C. gigantea group (rooted with C. cristata group species). The tree was constructed with concatenated DNA sequences of eight gene loci and the data-partitioning strategy P5. Numbers beside nodes are bootstrap percentages and Bayesian posterior probabilities, respectively. Adults of C. longifilamentata and C. alocasiae are shown along with inflorescences of respective common host plants. Eggs of C. longifilamentata (all attached to the side wall of a pistil of R. decursiva) are also shown, with their filaments indicated with the red arrowhead.
Within the gigantea group, the “C. hailini + C. yini” clade was strongly supported (BP = 100, PP = 1.00), with the remaining eight species forming a weakly supported species cluster (BP = 60, PP = 0.84). Within this cluster, C. daiae was assigned as a sister to the collection of the seven remaining species (BP = 94, PP = 1.00). These latter seven species were divided into two clusters: the first was of the Southeast Asian C. gigantea and C. scindapsae (BP = 70, PP = 1.00), and the second, which was only weakly supported (BP = 49, PP = 0.83), was of five Chinese species (C. longivalva, C. todai, C. liae, C. longifilamentata and C. rhaphidophorae). Within this latter cluster, the grouping of the four species C. todai, C. liae, C. longifilamentata and C. rhaphidophorae was strongly supported (BP = 77, PP = 1.00), with well-resolved relationships among the four.
Among the outgroup species (i.e., the C. cristata species group), the previously defined “clade II” (represented here by C. sarawakana, C. cristata and C. xenalocasiae) [6] was strongly supported with respect to “clade I” (represented by C. colocasiae) (BP = 99, PP = 1.00), although with a different subclade branching order: the “subclades IIa” (represented by C. cristata) and “subclade IIc” (represented by C. xenalocasiae) were closer to each other than either was to “subclade IIb” (represented by C. sarawakana) (BP = 83, PP = 1.00).
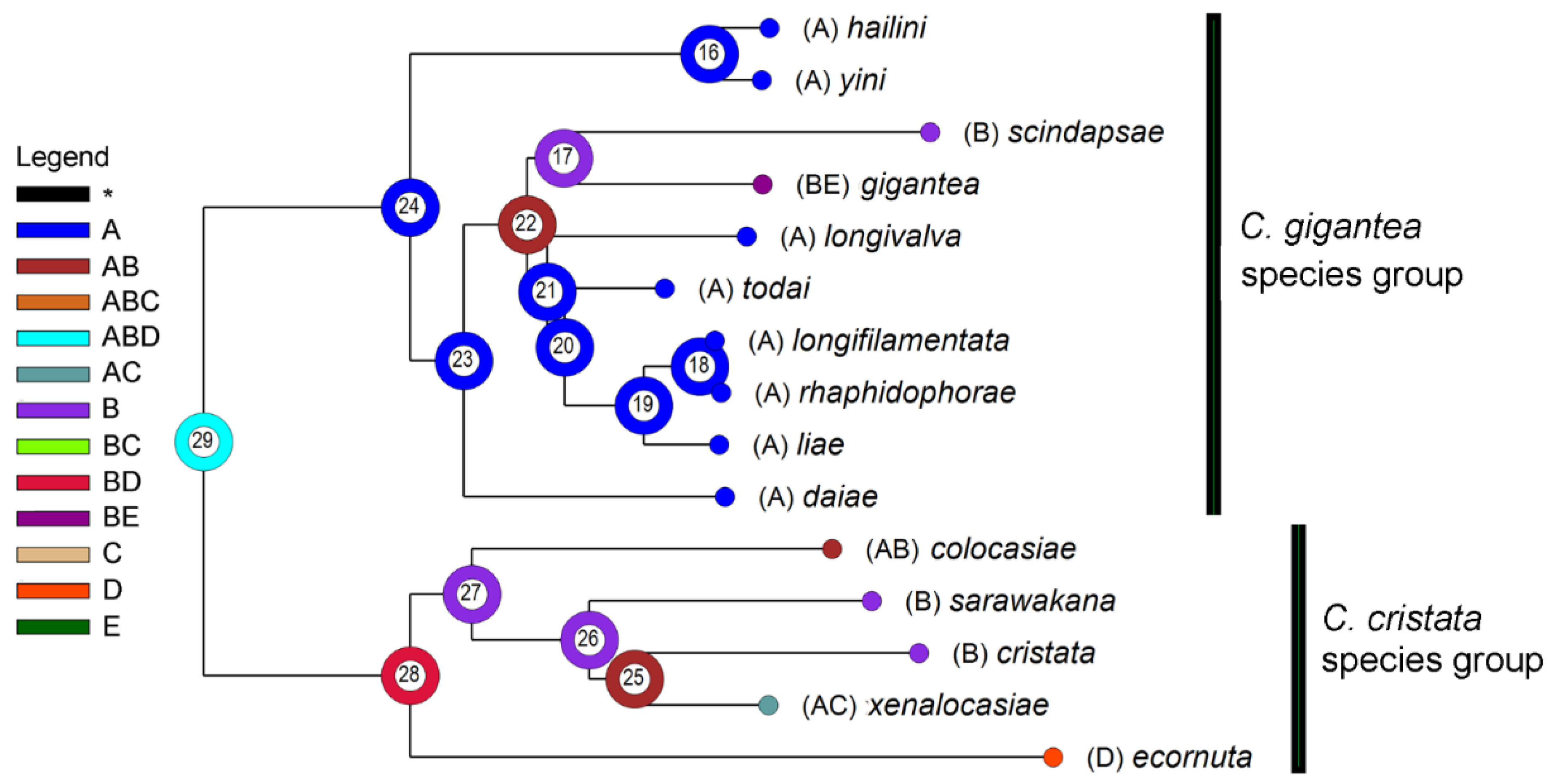
3.3. Biogeography
As shown in Figure 3 and Table 5, we inferred the range of the MRCA (most recent common ancestor) of the gigantea and cristata groups (node 29) as ABD (NE Oriental + Sundaland + Wallacea). Subsequent vicariance between A (NE Oriental) and BD (Sundaland + Wallacea) occurred then, and thereby gave rise to the MRCAs of the two groups. The MRCA of the gigantea group further split within A (node 24), and thereby gave rise to the C. hailini-C. yini lineage (node 16) and that of the remaining eight species (node 23). The former lineage split within A, and thereby gave rise to C. hailini and C. yini; the latter first dispersed to B and then split into two sublineages: one (restricted in A) finally gave rise to C. daiae, while the other (dispersed to B) gave rise to the MRCA of the remaining seven species (node 22). The further splitting of this latter sublineage between A and B gave rise to the MRCA of the C. scindapsae-C. gigantea pair in B and that of the NE Oriental pentad (C. longivalva, C. todai, C. liae, C. longifilamentata and C. rhaphidophorae) through a vicariance between A and B, with all the relevant speciation occurring within the respective area, except for C. gigantea, in which a further B-to-E dispersal (probably through D) occurred.
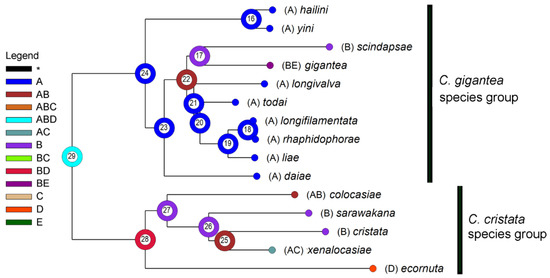
Figure 3.
A reconstruction of ancestral areas (A: NE Oriental region; B: Sundaland; C: the Philippines; D: Wallacea; E: Australian region) in the C. gigantea group and the C. cristata group (out-group). The most likely state on each of the ancestral nodes (#16–29) is represented with a color/letter or color/letter combination. Distributions of extant fly species are mapped onto terminal nodes.

Table 5.
Summary of the results of the S-DIVA analysis of ancestral distribution in the C. gigantea species group.
The ancestral area of the C. cristata group was reconstructed as BD (Sundaland + Wallacea), and it split into two sublineages through a vicariance between the areas B and D. The subsequent evolution may have involved a dispersal from Sundaland to the NE Oriental region (e.g., C. colocasiae) or the Philippines (e.g., C. xenalocasiae), and a vicariance between Sundaland and the NE Oriental region-the Philippines (C. sarawakana vs. C. xenalocasiae).
3.4. Ancestral Host Use and Evolution of Body Size
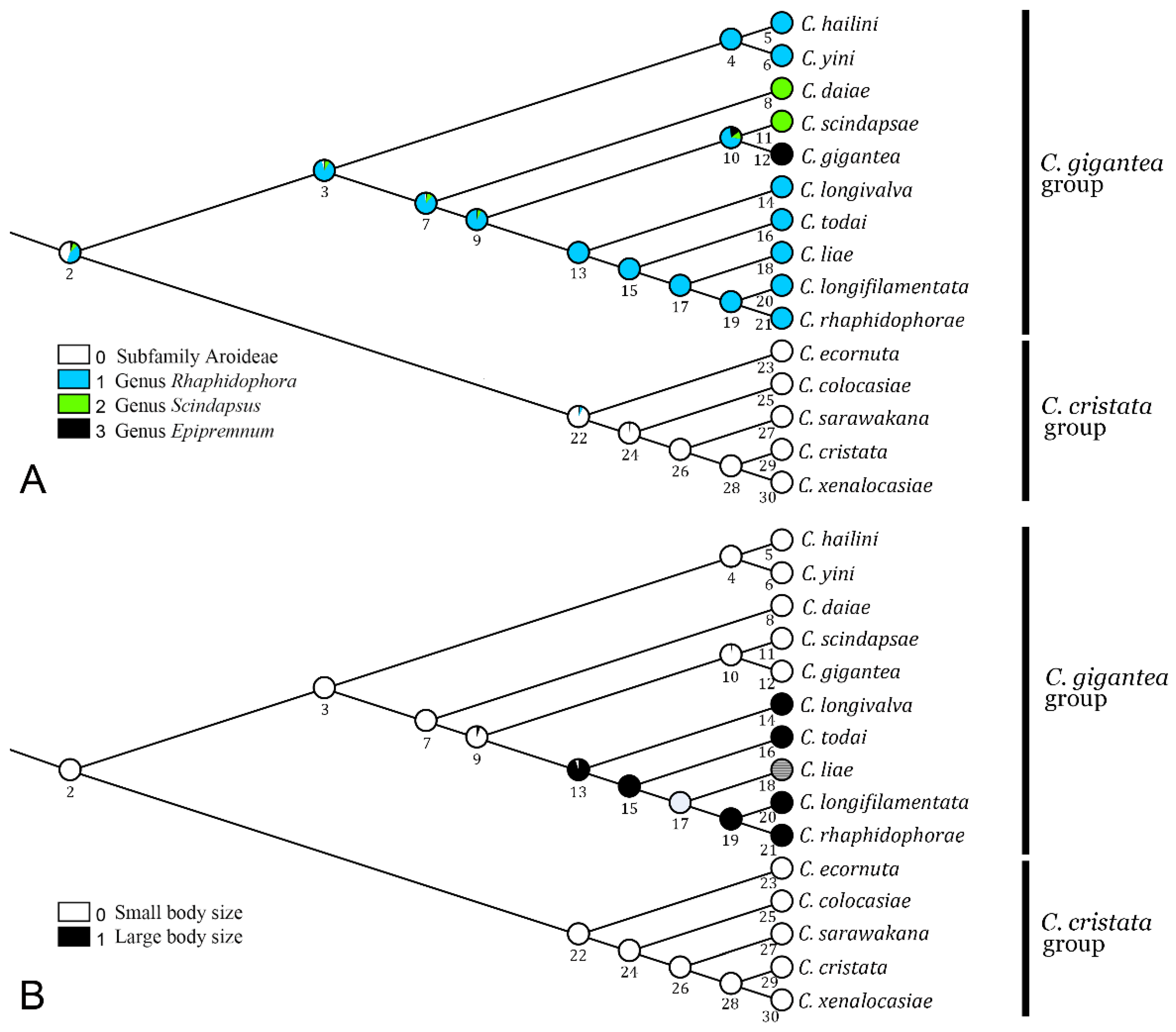
As shown in Figure 4A, the host use was ambiguously reconstructed for the MRCA of the C. gigantea and C. cristata groups, either from Monsteroideae (Rhaphidophora, or much less likely, Scindapsus or Epipremnum) or from the subfamily Aroideae. We inferred with confidence that the MRCA of the C. gigantea group (node 3) used Rhaphidophora as a host. Although most of its descendant species retained the host selection of Rhaphidophora, two independent shifts to Scindapsus occurred: one to C. daiae and the other to C. scindapsae. For C. gigantea, a Rhaphidophora-to-Epipremnum shift occurred.
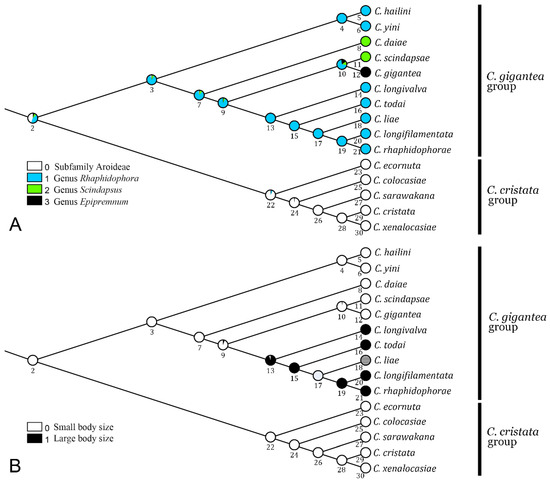
Figure 4.
Evolutionary scenarios in (A) host plant use and (B) body size in the C. gigantea and C. cristata species groups. See Table A2 for data on body size.
We inferred that the body size was ancestrally small in either the gigantea or the cristata group (Figure 4B). On the one hand, in the former group, the “large” body size may have evolved once in the MRCA of C. longivalva, C. todai, C. liae, C. longifilamentata and C. rhaphidophorae, even though in C. liae, the status was undetermined due to the deficiency of the type specimens [8]. On the other hand, for the cristata group, the body size was ancestrally “small”, which prevailed during the subsequent evolution of the group.
4. Discussion
4.1. Phylogeny of the C. gigantea Species Group
4.1.1. Monophyly of the Gigantea Group and Its Relationship to the Cristata Group
In the recent cladistic analyses of 70 morphological characters and 24 Colocasiomyia species covering all six species groups of this genus [4], the gigantea and cristata groups were supported as reciprocally monophyletic, with both forming the most derived clade in the genus. The autapomorphy that supported the monophyly of the gigantea group (i.e., the bilateral lobes of the oviscapt (corresponding to the “hypogynium” in the standardized nomenclature of Drosophila melanogaster [24])) was “fused to each other only apically” (vs. “not fused” or “fused submedially to subapically” in the other groups) and was affirmed in all the subsequently replenished members in the C. gigantea group, except for C. daiae; in this latter species, the lateral lobes of the oviscapt were found fused to each other “only subapically” [9]. This feature, together with some other morphological features of the species (e.g., lobes of oviscapt lack warts on basal half, thorax with an additional pair of dorsocentral setae, wing costa with extraordinarily long setae and male abdominal sternite VI absence) [9], may be attributable to some forms of evolutionary adaptation.
4.1.2. Phylogenetic Position of the C. hailini-C. yini Lineage
The pairing of these two species is consistent with the overall morphological similarity between them [5]. This pair was also recognized in the family-wide molecular tree [12], but was placed at a more or less derived position, forming a cluster with the Southeast Asian C. gigantea, and then with the cluster of C. rhaphidophorae, C. longifilamentata and C. longivalva. By closely examining the sequence dataset of this large-scale phylogenetic analysis [12], we found that it suffered from an overwhelming proportion of missing data. For example, ca. 81% of the schemed DNA sequences (272) were missing for the 16 Colocasiomyia species employed. Therefore, we relied on our results rather than those of the family-wide analysis. This was also why we chose to use the cristata group instead of the toshiokai group as the outgroup taxon in the present phylogenetic reconstruction.
4.1.3. The C. scindapsae-C. gigantea Pair and C. daiae
In our phylogenetic tree, C. daiae branched right after the C. hailini-C. yini pair. It was likely that C. daiae retained “primordial” type of surstylus seen in the other two species: the surstylus was broad and ornamented, with three large peg-like prensisetae on the distal margin, with the lowest one distinctly elongated below [5,9].
4.1.4. The C. longivalva-C. todai-C. liae-C. longifilamentata-C. rhaphidophorae Pentad
Despite the relatively lower bootstrap/posterior probability support for the clustering of the five large-bodied species (longivalva, todai, liae, longifilamentata and rhaphidophorae) in our phylogenetic analyses, two lines of morphological evidence support this pentad: (1) a relatively narrower and/or smaller surstylus, bearing only thin setae and/or tiny teeth (broad, with stout pegs or teeth in the small-bodied species), and (2) the presence of an epandrial posterior lobe (absent in the small-bodied species). Moreover, among these five species, the longifilamentata-rhaphidophorae pair exhibit some extraordinary morphological similarities with each other (e.g., epandrial posterior lobe well-developed and narrowly prolongated, scabbard-like, apically inlaid with a large peg [4,5,8,9]).
4.2. Biogeography, Major Host Shift, Diversification and Body-Size Evolution
According to our biogeographical analysis, a divergence between the C. gigantea and C. cristata groups took place through vicariance between the NE Oriental region and Sundaland + Wallacea, with the subsequent diversification of the former group occurring mostly within the NE Oriental region, despite two exceptions: (1) a NE Oriental→Sundaland dispersal followed by vicariance between these two areas, giving rise to the lineage of the large-bodied pentad in the NE Oriental region and to that of the C. scindapsae-C. gigantea pair in Sundaland; (2) the split between C. scindapsae and C. gigantea within Sundaland. However, taking into consideration the possible bias in our past surveys of the species diversity and host use of the C. gigantea group toward the NE Oriental region, the above scenario of the evolutionary history of the C. gigantea group was likely far from exhaustive. The genus Rhaphidophora, as the ancestral host taxa of the C. gigantea group, consists of ca. 100 species geographically occupying tropical Asia, and even extend as far as West Africa and the western Pacific, with the eastern Himalayas (from northern India and Nepal to southwestern China) recognized as one of its “hotspots” for endemic species [25,26]. It is thus intriguing to explore the dynamics underlying the evolution of the large body size in the lineage of the pentad C. longivalva, C. todai, C. liae, C. longifilamentata and C. rhaphidophorae, along with its adaptation to Rhaphidophora host plants, referring to the convergent feeding habit that occurred in the C. hailini-C. yini lineage. The other two host genera of the flies in the C. gigantea group, Scindapsus and Epipremnum, although of inferior species diversity (35 and 15 species, respectively [27]), are largely geographically overlapped with each other and with Rhaphidophora: Scindapsus occurs from northeastern India to western Polynesia [28], while Epipremnum ranges from southern Japan to Australia, and from India to the Cook Islands [29]. A future investigation of a wider geographical range, especially the NE Oriental region, is necessary to reconstruct the overall scenario of the diversification of the genus Colocasiomyia, including the C. gigantea group.
4.3. Adaptation of the C. gigantea Group to Monsteroid Host Plants
So far, varied evolutionary adaptations of the flies in the C. gigantea group have been assumed to evolved along with the Aroideae-to-Monsteroideae host shift of this fly group [4]. One character that accords with the host adaptation in the C. gigantea group is the blade-like oviscapt lobes, which are advantageous for laying eggs deep in the slits between the pistils on the host inflorescence [4,5,8,9]. Such an egg-laying mode protects the larvae (quiescent in the eggshell and harbored in the inflorescence) from biotic (e.g., predatory/parasitic natural enemies) and abiotic (low temperature (e.g., for species inhabiting the north tropical zone), drought, UV, etc.) stresses during the longstanding fruit-ripening process of the host plant [4]. We now know that for the eggs in all the species of the gigantea group, there are two thin, long and tubulate filaments that are horizontally arranged at the posterior end of the egg [4,5,8,9]. The filaments are indispensable to the respiration of the eggs and the embryo within the egg capsule hiding in the narrow slits between the pistils of the host inflorescence because the air exchange through the chorion is largely arrested within the slits [4,30,31]. Because egg filaments are not seen in the other Colocasiomyia, we can reasonably assume a single gain of such an apparatus in the MRCA of the gigantea group based on the principle of parsimony [32].
To date, no species in Colocasiomyia has ever been involved in evolutionary dating, making it difficult to explore its evolution against a concrete historical background. Nevertheless, we can make a rough judgment in the evolutionary context of the whole family Drosophilidae, taking into consideration the following date estimates: (1) 40–80 Mya (million years ago) for the splitting between the two subfamilies of Drosophilidae (i.e., Drosophilinae (with Colocasiomyia inside) and Steganinae) based on fossil and biogeographical data [33,34,35], and (2) 95 Mya (Nauheimer et al.; unpublished data as cited in a previous study [36]) or 89.2 Mya (95% highest probability density: 86.4–97.0 Mya) [37] for the splitting between the plant subfamilies Monsteroideae and Aroideae. Thus, we can reasonably interpret the initial adaptation of the C. gigantea group to some monsteroid host plant (and specifically a plant in the Rhaphidophora clade [7]) as a pollinator shift [38,39,40].
The evolution of egg filaments is a critical transformation that mediated the overall adaptation of the C. gigantea group to monsteroid host plants, which differ from those in the Aroideae according to a series of defining features [4], each of which may have exerted an essential influence on the flies’ adaptation to the monsteroid host plants (Table A1): (1) the spadices that are composed of bisexual florets are spatially homogeneous (i.e., without zonation and lacking any appendix), and thus, may provide only a few edibles for the larvae of the Colocasiomyia flies until the maturation of the spadix itself; (2) it is unlikely that the mass of the withered stamens of these hosts is sufficient to feed the larvae of the Colocasiomyia, and neither is the amount of pollen; (3) the whole spathe of the inflorescence, which is borne basal to the inflorescence, may thus hinder the Colocasiomyia larvae derived from eggs laid in the upper portion of the spadix from exploiting the bract itself; (4) the spathe falls off as a whole from its joint with the spadix soon after anthesis, which provides merely a momentary shelter for the Colocasiomyia larvae from environmental stress (i.e., the floral chamber, as in Alocasia odora [4]); and (5) in the NE Oriental region, where the C. gigantea group may have originated and where the major diversification of the group may have occurred, none of the presently known monsteroid plants bloom throughout the year (www.iplant.cn/info/Araceae?t=foc; accessed on 1 June 2022), which makes it difficult for the flies to depend solely on the spathe and/or pollen there.
Besides the flies in the C. gigantea group, a Neotropical Drosophila species was also found using the monsteroid plant (i.e., D. monsterae: the adults of this species are attracted into the floral chamber of Monstera lentii and copulate there during the female phase of the inflorescences, and they “stay at the spadix base where fallen pollen grains accumulated and later departed with pollen on their bodies.”) [41,42]. This finding indicates a parallel or convergent adaptation to the monsteroid host plant between Colocasiomyia and Drosophila flies. Moreover, according to the original description [42], the adults of D. monstera are characterized by a “small slightly circular compound eye” and a “conspicuously broad gena”, both commonly seen in Colocasiomyia flies.
4.4. The Potential of the C. gigantea Group in Evolutionary/Developmental Studies
So far, pollination mutualism have been examined in many aroid-breeding Colocasiomyia species and confirmed in some cristata species: the flies pollinate for their host plants. In turn, the plant inflorescence/infructescence rewards the flies with edibles for adults (exudates, solid substance from stamens) and larvae (the decaying pulp of fruits and/or basal soft tissue of dehisced pistils); a site for mating and oviposition; and a site/shelter for developing embryos, quiescent larval, pupae and even adults. In almost all such systems of pollinating mutualism, phenotypical synchrony between the lifecycles of the flies and their host plants has been observed [3,6,43,44,45]. The genus Colocasiomyia, with each of its groups specialized on a particular host plant lineage, provides us with an ideal system to explore the subtle mechanisms that underlie the evolution of intimate and obligate pollination mutualism, putting it on par with the fig-fig wasp and yucca-yucca moth systems [44]. A sister-group analysis allows control of “much potential noise and confounding variation which might otherwise afflict the analysis”, which makes it possible to “compare like with like” [46]. Among the groups in Colocasiomyia, the cristata and gigantea groups are of particular importance considering their sister-group relationship and their impressive intergroup divergence, according to the morphological, ecological and behavioral attributes, along with the major host shift that occurred in the latter group [4].
The distribution and phylogenetic importance of egg filaments in other drosophilid lineages at length, have been examined at length, with the gains and losses of such a structure in confirmed in varied taxa (e.g., the Hawaiian Drosophila and Scaptomyza) [47,48]. Indeed, some researchers considered the reduction in or absence of egg filaments to be a general property of tropical flower-breeding drosophilid species [49]. Within Colocasiomyia, egg filaments present in all the species in the gigantea group, but absent in the remaining groups [4,5,8,9]. In the fields of genetics and developmental biology, it is confirmed that such filaments of the eggshell, which are also known as dorsal appendages (DAs), arise at the end of oogenesis in Drosophila melanogaster; the tubulogenesis of this structure provides an excellent system that serves as a link between the patterning and morphogenesis phases, with eggshells of different drosophilid species used as the ideal testing model [50,51,52]. The species in the genus Colocasiomyia, especially those in the gigantea and cristata groups, provide an ideal comparative system for this field.
5. Conclusions
The present phylogenetic analysis lends strong support to the monophyly of the C. gigantea group with respect to the C. cristata group, placing the C. hailini-C. yini lineage as most basal within the C. gigantea group. Within the focal group, the Southeast Asian C. scindapsae-C. gigantea pair is well recognized; the northeastern Oriental, large-bodied C. longivalva-C. todai-C. liae-C. longifilamentata-C. rhaphidophorae pentad is supported with either molecular or morphological evidences.
The C. gigantea group diverged from the C. cristata group through a vicariance between the northeastern Oriental region and Sundaland + Wallacea. The subsequent diversification of this group occurred mostly within the former region. An Oriental-to-Sundaland dispersal occurred subsequently, followed by a vicariance between these two areas, finally gave rise to the C. gigantea-C. scindapsae lineage in Southeast Asian.
The most likely ancestral host genus of the C. gigantea species group is Rhaphidophora, with subsequent shift to Scindapsus and/or Epipremnum plants may had occurred.
Within the whole genus Colocasiomyia, egg filaments (dorsal appendages) evolved exclusively in the C. gigantea group, investing the flies in this group with potential as ideal model system for comparative studies of pollination mutualism and developmental genetics concerning tubulogenesis.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/insects13070647/s1. Table S1: Data sets of DNA sequences and corresponding selected model (using the BIC i.e., Bayesian information criterion). Table S2: GenBank accession numbers of DNA sequences of eight molecular markers employed in the present study.
Author Contributions
Conceptualization, J.-J.G.; validation, L.X. and N.-N.L.; investigation, L.X., N.-N.L., L.-K.Y. and J.-L.L.; resources, J.-J.G. and J.-L.L.; data curation, L.X., N.-N.L. and L.-K.Y.; writing, J.-J.G. and L.X.; funding acquisition, J.-J.G. and L.X. All authors have read and agreed to the published version of the manuscript.
Funding
This work is supported by the National Science Foundation of China (no. 32060112) to J.-J.G., and by the Postgraduate Research and Innovation Foundation of Yunnan University (no. 2021Y414) to L.X.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data available in a publicly accessible repository.
Acknowledgments
We thank the Administrations of the Gaoligongshan, Xishuangbanna, Huanglianshan and Wuzhishan National Nature Reserves for their support during the field surveys in the reserves; our Malaysian, Indonesian and Vietnamese colleagues for the fly samples; and M.J. Toda for comments on the draft.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Table A1.
Comparison between host taxa of four aroid-breeding species groups in Colocasiomyia.
Table A1.
Comparison between host taxa of four aroid-breeding species groups in Colocasiomyia.
| Host Subfamily | Character Status | |||
|---|---|---|---|---|
| Aroideae | Monsteroideae | |||
| Host tribe/clade/genus [27,53] | Genera: Homalomena Schott (Philodendron clade) and Aglaonema Schott (tribe Aglaonemateae) | Tribe: Schismatoglottideae (genera: Schismatoglottis and Piptospatha) | Colocasia clade; genera Colocasia, Alocasia, Leucocasia and Steudnera | Rhaphidophora clade (Rhaphidophora, Scindapsus, Epipremnum) |
| Corresponding species group in Colocasiomyia | C. toshiokai group (6 known species) [45] | C. baechlii group (2 known + 37 undescribed species) [54] | C. cristata group (21 known + 14 undescribed species) [6] | C. gigantea group (10 known species) [4] |
| Flower sexuality [27,53,55] | Unisexual | Bisexual | ||
| Spadix zonation [27,53,55] | Female/male | Female/sterile or bare/male | Female/sterile/male | No zonation |
| Spadix appendix [27,53,55] | Absent | Absent | Well developed | Absent |
| Spathe shape [27,53,55] | Without floral chamber (Aglomena) or varied (Homalomena) | Without floral chamber | With floral chamber | Boat-shaped (without floral chamber) |

Table A2.
Data of body size (thorax length) [23] of 15 Colocasiomyia species compiled from the literature or as newly collected data.
Table A2.
Data of body size (thorax length) [23] of 15 Colocasiomyia species compiled from the literature or as newly collected data.
| Species Group | Species | Thorax Length (ThL) | Code | |
|---|---|---|---|---|
| n (♂/♀) | Range (♂/♀; in mm) | |||
| C. gigantea group | hailini Li and Gao, 2014 [5] | 11/10 | (0.76–0.94)/(0.89–1.22) | A |
| yini Li and Gao, 2014 [5] | 6/5 | (0.90–1.12)/(0.93–1.20) | A | |
| daiae Xue and Gao, 2022 [9] | 6/5 | (0.90–1.07)/(0.86–1.24) | A | |
| scindapsae Fartyal and Toda, 2013 [4] | 10/10 | (0.88–1.03)/(0.85–1.24) | A | |
| gigantea Okada, 1987 [11] | n/a | n/a | A | |
| longivalva Li and Gao, 2014 [5] | 4/8 | (1.18–1.42)/(1.26–1.47) | B | |
| todai Jiao and Gao, 2020 [8] | 6/5 | (1.56–1.72)/(1.49–1.73) | B | |
| liae Jiao and Gao, 2014 [8] | 1/1 | 1.10/0.93 | ? | |
| longifilamentata Li and Gao, 2014 [5] | 7/5 | (1.17–1.37)/(1.33–1.47) | B | |
| rhaphidophorae Gao and Toda, 2013 [4] | 7/13 | (1.30–1.48)/(1.18–1.57) | B | |
| C. cristata group | ecornuta Toda and Takano, 2021 [6] | 1 (?) | ca. 0.80 | A |
| colocasiae (Duda, 1924) [6] | 1 (?) | ca. 0.94 | A | |
| sarawakana Toda and Yafuso, 2021 [6] | 1 (?) | ca. 0.86 | A | |
| cristata der Meijere, 1914 [6] | 1 (?) | ca. 1.05 | A | |
| xenalocasiae (Okada, 1980) * | 10/10 | (0.65–0.80)/(0.66–0.92) | A | |
* Newly collected data from specimens from Vietnam (Lot 201: Mot Tuc Vill., Vietnam, 20.x.2011, ex. inflorescence of Colocasia esculenta, #2).
References
- Okada, T. Synhospitalic evolution of the genus Drosophilella Duda (Diptera, Drosophilidae), with description of a new Species from Okinawa and Taiwan. Kontyû 1980, 48, 218–225. [Google Scholar]
- Grimaldi, D. Systematics of the genus Colocasiomyia de Meijere (Diptera, Drosophilidae)—Cladistics, a new generic synonym, new records, and a new species from Nepal. Entomol. Scand. 1991, 22, 417–426. [Google Scholar] [CrossRef]
- Sultana, F.; Hu, Y.G.; Toda, M.J.; Takenaka, K.; Yafuso, M. Phylogeny and classification of Colocasiomyia (Diptera, Drosophilidae), and its evolution of pollination mutualism with aroid plants. Syst. Entomol. 2006, 31, 684–702. [Google Scholar] [CrossRef]
- Fartyal, R.S.; Gao, J.J.; Toda, M.J.; Hu, Y.G.; Takano, K.T.; Suwito, A.; Katoh, T.; Takigahira, T.; Yin, J.T. Colocasiomyia (Diptera: Drosophilidae) revised phylogenetically, with a new species group having peculiar lifecycles on monsteroid (Araceae) host plants. Syst. Entomol. 2013, 38, 763–782. [Google Scholar] [CrossRef]
- Li, N.N.; Toda, M.J.; Fu, Z.; Chen, J.M.; Li, S.H.; Gao, J.J. Taxonomy of the Colocasiomyia gigantea species group (Diptera, Drosophilidae), with descriptions of four new species from Yunnan, China. Zookeys 2014, 406, 41–64. [Google Scholar] [CrossRef]
- Takano, K.T.; Gao, J.J.; Hu, Y.G.; Li, N.N.; Yafuso, M.; Suwito, A.; Repin, R.; Pungga, R.A.S.; Meleng, P.A.; Kaliang, C.H.; et al. Phylogeny, taxonomy and flower-breeding ecology of the Colocasiomyia cristata species group (Diptera: Drosophilidae), with descriptions of ten new species. Zootaxa 2021, 5079, 1–70. [Google Scholar] [CrossRef]
- Cusimano, N.; Bogner, J.; Mayo, S.J.; Boyce, P.C.; Wong, S.Y.; Hesse, M.; Hetterscheid, W.L.; Keating, R.C.; French, J.C. Relationships within the Araceae: Comparison of morphological patterns with molecular phylogenies. Am. J. Bot. 2011, 98, 654–668. [Google Scholar] [CrossRef]
- Jiao, R.J.; Bai, L.H.; Gao, J.J. Descriptions of two new species of the genus Colocasiomyia (Diptera, Drosophilidae) breeding on Rhaphidophora host plants in Yunnan, China. Zookeys 2020, 968, 127–141. [Google Scholar] [CrossRef]
- Xue, X.C.; Jiao, R.J.; Yu, L.I.; Gao, J.J. A new species of the Colocasiomyia gigantea species group (Diptera, Drosophilidae) breeding on inflorescences of Scindapsus maclurei (Araceae). Zootaxa 2022, 5120, 586–594. [Google Scholar] [CrossRef]
- Okada, T. Taximetrical analysis of costal chaetotaxy of the genus Drosophilella Duda, with Description of a new species from Sri Lanka (Diptera, Drosophilidae). Proc. Jpn. Soc. Syst. Zool. 1986, 34, 53–59. [Google Scholar]
- Okada, T. Further notes on the genus Drosophilella Duda with descriptions of two new species from Indonesia (Diptera, Drosophilidae). Proc. Jpn. Soc. Syst. Zool. 1987, 36, 38–45. [Google Scholar]
- Finet, C.; Kassner, V.A.; Carvalho, A.B.; Chung, H.; Al, E. DrosoPhyla: Genomic resources for drosophilid phylogeny and systematics. Genome Biol. Evol. 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Toda, M.J.; Li, N.N.; Zhang, Y.P.; Gao, J.J. A new genus of anthophilous drosophilids, Impatiophila (Diptera, Drosophilidae): Morphology, DNA barcoding and molecular phylogeny, with descriptions of thirty-nine new species. Zootaxa 2016, 4120, 1–100. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular evolutionary genetics analysis Version 7.0 for bigger datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef]
- Brandley, M.C.; Schmitz, A.; Reeder, T.W. Partitioned Bayesian analyses, partition choice, and the phylogenetic relationships of scincid lizards. Syst. Biol. 2005, 54, 373–390. [Google Scholar] [CrossRef]
- Rambaut, A.; Xie, D.; Suchard, M.A.; Drummond, A.J.; Baele, G. Posterior summarization in Bayesian phylogenetics Using Tracer 1.7. Syst. Biol. 2013, 67, 901–904. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML-VI-HPC: Maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 2006, 22, 2688–2690. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Hohna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Yu, Y.; Blair, C.; He, X. RASP 4: Ancestral state reconstruction tool for multiple genes and characters. Mol. Biol. Evol. 2019, 37, 604–606. [Google Scholar] [CrossRef]
- van Steenis, C.G.G.J. The delimitation of Malesia and its main plant geographical divisions. Flora Males. 1950, 1, 120–125. [Google Scholar]
- Lohman, D.J.; de Bruyn, M.; Page, T.; von Rintelen, K.; Hall, R.; Ng, P.K.L.; Shih, H.-T.; Carvalho, G.R.; von Rintelen, T. Biogeography of the Indo-Australian Archipelago. Ann. Rev. Ecol. Evol. Syst. 2011, 42, 205–226. [Google Scholar] [CrossRef]
- Maddison, W.P.M. Mesquite: A Modular System for Evolutionary Analysis. Version 3.70. Available online: http://www.mesquiteproject.org (accessed on 21 May 2022).
- Zhang, W.X.; Toda, M.J. A new species-subgroup of the Drosophila immigrans species-group (Diptera, Drosophilidae), with description of two new species from China and revision of taxonomic terminology. Jpn. J. Entomol. Scand. 1992, 60, 839–850. [Google Scholar]
- McQueen, E.W.; Afkhami, M.; Atallah, J.; Belote, J.M.; Gompel, N.; Heifetz, Y.; Kamimura, Y.; Kornhauser, S.C.; Masly, J.P.; O’Grady, P.; et al. A standardized nomenclature and atlas of the female terminalia of Drosophila melanogaster. Fly 2022, 16, 128–151. [Google Scholar] [CrossRef] [PubMed]
- Boyce, P. Plate 377. Rhaphidophora glauca. Curtis’s Bot. Mag. 1999, 16, 273–280. [Google Scholar] [CrossRef]
- Nguyen, D.V.; Bui, H.Q.; Ha, M.T.; Le, X.D.; Nguyen, C.S.; Boyce, P.C. Rhaphidophora sonlaensis (Araceae), a new species from northern Vietnam. Ann. Bot. Fenn. 2017, 54, 111–115. [Google Scholar] [CrossRef]
- Cole, T.C.H.; Wong, S.Y.; Li, H. Araceae Phylogeny Poster (Chinese). Available online: https://www.researchgate.net/publication/338718929 (accessed on 21 May 2022).
- Bogner, J.; Boyce, P.C. Scindapsus lucens (Araceae: Monsteroideae), a new species related to Scindapsus pictus. Kew Bull. 1994, 49, 789–792. [Google Scholar] [CrossRef]
- Boyce, P. A review of Epipremnum (Araceae) in Cultivation. Aroideana 2004, 27, 205–211. [Google Scholar]
- Hinton, H.E. Plastron respiration in the eggs of Drosophila and other flies. Nature 1959, 184, 280–281. [Google Scholar] [CrossRef]
- Hinton, H.E. Respiratory systems of insect egg shells. Annu. Rev. Entomol. 1969, 14, 343–368. [Google Scholar] [CrossRef]
- Sober, E. The Principle of Parsimony. Br. J. Philos. Sci. 1981, 32, 145–156. [Google Scholar] [CrossRef]
- Grimaldi, D.A. Relicts in the Drosophilidae. In Zoogeography of Caribbean Insects; Liebherr, J.K., Ed.; Cornell University Press: Ithaca, NY, USA, 1988. [Google Scholar]
- Hennig, W. Die Acalyptratae des Baltischen Bernsteins und ihre Bedeutung für die Erforschung der phylogenetischen Entwicklung dieser Dipterengruppe. Stuttg. Beiträge Nat. 1965, 145, 1–215. [Google Scholar]
- Powell, J.R.; DeSalle, R. Drosophila molecular phylogenies and their uses. In Evolutionary Biology; Hecht, M.K., MacIntyre, R.J., Clegg, M.T., Eds.; Plenum Press: New York, NY, USA, 1995. [Google Scholar]
- Schiestl, F.P.; Dotterl, S. The evolution of floral scent and olfactory preferences in pollinators: Coevolution or pre-existing bias? Evolution 2012, 66, 2042–2055. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Yang, Y.Y.; Qu, X.J.; Ma, H.; Hu, Y.; Li, H.T.; Yi, T.S.; Li, D.Z. Phylotranscriptomic analyses reveal multiple whole-genome duplication events, the history of diversification and adaptations in the Araceae. Ann. Bot. 2022. [Google Scholar] [CrossRef] [PubMed]
- Whittall, J.B.; Hodges, S.A. Pollinator shifts drive increasingly long nectar spurs in columbine flowers. Nature 2007, 447, 706–709. [Google Scholar] [CrossRef]
- Hodges, S.A.; Whittall, J.B. One-sided evolution or two? A reply to Ennos. Heredity 2008, 100, 541–542. [Google Scholar] [CrossRef]
- Johnson, S.D.; Anderson, B. Coevolution between Food-Rewarding Flowers and Their Pollinators. Evol. Educ. Outreach 2010, 3, 32–39. [Google Scholar] [CrossRef]
- Prieto, D.; Cascante-Marín, A. Pollination by nitidulid beetles in the hemi-epiphytic aroid Monstera lentii (Araceae: Monsteroideae). Flora 2017, 231, 57–64. [Google Scholar] [CrossRef][Green Version]
- Vilela, C.R.; Prieto, D. A new Costa Rican species of Drosophila visiting inflorescences of the hemi-epiphytic climber Monstera lentii (Araceae). Rev. Bras. Entomol. 2018, 62, 225–231. [Google Scholar] [CrossRef]
- Takenaka, K.; Yin, J.T.; Wen, S.Y.; Toda, M.J. Pollination mutualism between a new species of the genus Colocasiomyia de Meijere (Diptera: Drosophilidae) and Steudnera colocasiifolia (Araceae) in Yunnan, China. Entomol. Sci. 2006, 9, 79–91. [Google Scholar] [CrossRef]
- Takano, K.T.; Repin, R.; Mohamed, M.B.; Toda, M.J. Pollination mutualism between Alocasia macrorrhizos (Araceae) and two taxonomically undescribed Colocasiomyia species (Diptera: Drosophilidae) in Sabah, Borneo. Plant Biol. 2012, 14, 555–564. [Google Scholar] [CrossRef]
- Shi, T.; Toda, M.J.; Takano, K.T.; Yafuso, M.; Suwito, A.; Wong, S.Y.; Shang, S.Q.; Gao, J.J. A review of taxonomy and flower-breeding ecology of the Colocasiomyia toshiokai species group (Diptera: Drosophilidae), with description of a new species from Indonesia. Eur. J. Entomol. 2019, 116, 341–361. [Google Scholar] [CrossRef]
- Barraclough, T.G.; Nee, S.; Harvey, P.H. Sister-group analysis in identifying correlates of diversification—Comment. Evol. Ecol. 1998, 12, 751–754. [Google Scholar]
- Throckmorton, L.H. The Problem of Phylogeny in the Genus Drosophila; The University of Texas Press: Austin, TX, USA, 1962; Volume 6205, pp. 207–343. [Google Scholar]
- Throckmorton, L.H. The Relationships of the Endemic Hawaiian Drosophilidae; The University of Texas Press: Austin, TX, USA, 1966; Volume 6615, pp. 335–396. [Google Scholar]
- David, J.R.; Yassin, A.; Rasamizafi, L.A.; Ravaomanarivo, L.H.R.; Debat, V. Scratching for food: An original feeding behavior in an African flower breeding Drosophila. Fly 2011, 5, 285–290. [Google Scholar] [CrossRef]
- Berg, C.A. The Drosophila shell game: Patterning genes and morphological change. Trends Genet. 2005, 21, 346–355. [Google Scholar] [CrossRef]
- Osterfield, M.; Berg, C.A.; Shvartsman, S.Y. Epithelial patterning, morphogenesis, and evolution: Drosophila eggshell as a model. Dev. Cell 2017, 41, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, S.G.; Merrihew, G.E.; MacCoss, M.J.; Berg, C.A. Proteomics analysis identifies orthologs of human chitinase-like proteins as inducers of tube morphogenesis defects in Drosophila melanogaster. Genetics 2017, 206, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zhu, G.; Boyce, P.; Murata, J.; Hetterscheid, W.L.A.; Bogner, J.; Jacobsen, N. The Flora of China. Available online: http://www.efloras.org/flora_page.aspx?flora_id=2 (accessed on 21 May 2022).
- Toda, M.J.; Lakim, M.B. Genus Colocasiomyia (Drosophilidae: Diptera) in Sabah, Bornean Malaysia: High species diversity and use of host aroid inflorescences. Entomol. Sci. 2011, 14, 262–270. [Google Scholar] [CrossRef]
- Chartier, M.; Gibernau, M.; Renner, S.S. The evolution of pollinator-plant interaction types in the Araceae. Evolution 2014, 68, 1533–1543. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).