On the Affinities and Systematic Position of Lachnaeus Schoenherr and Rhinocyllus Germar in the Tribe Lixini (Coleoptera: Curculionidae: Lixinae) Based on the Morphological Characters of the Immature Stages
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Material Sampling
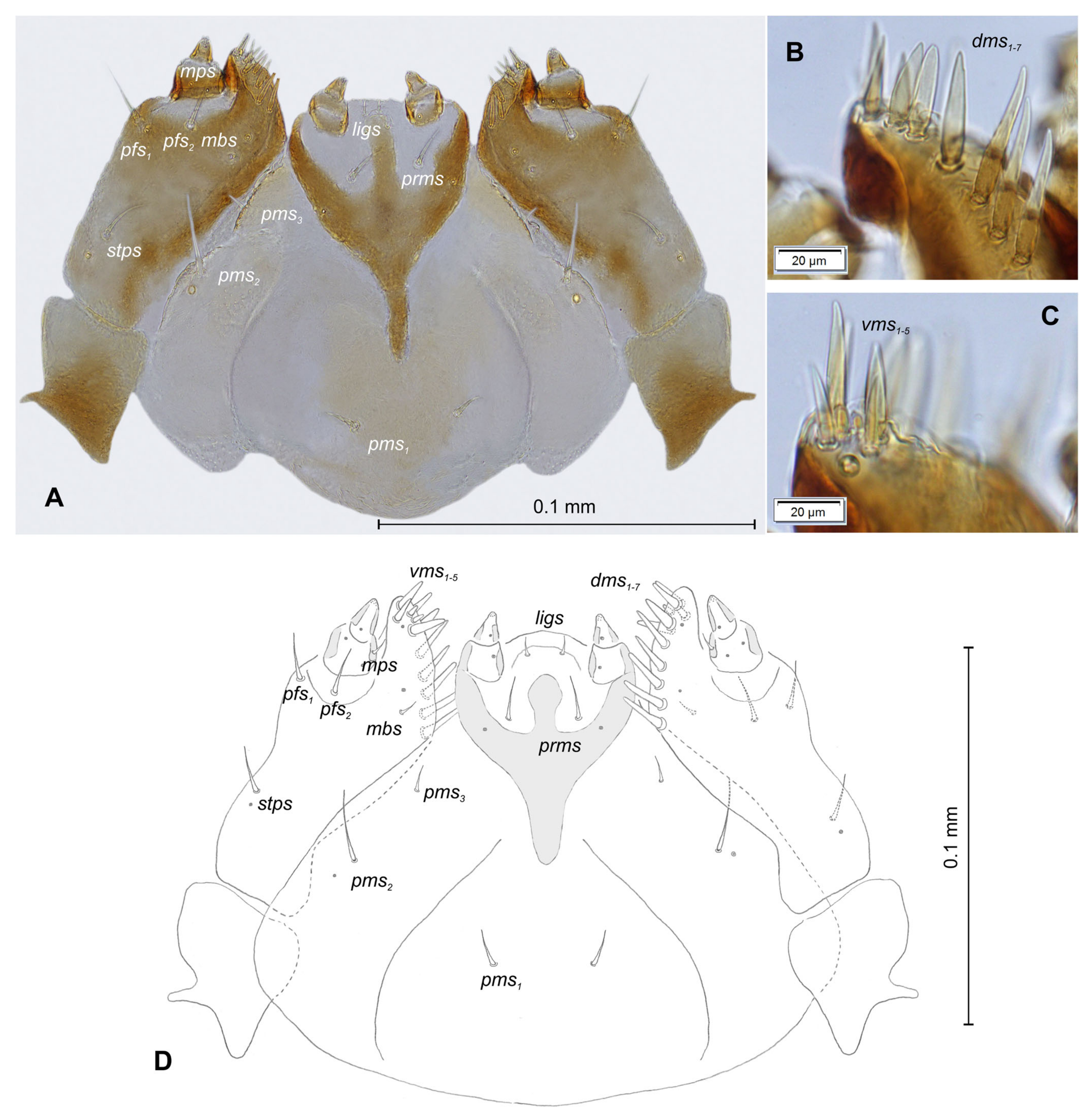
2.2. Morphological Description
3. Results
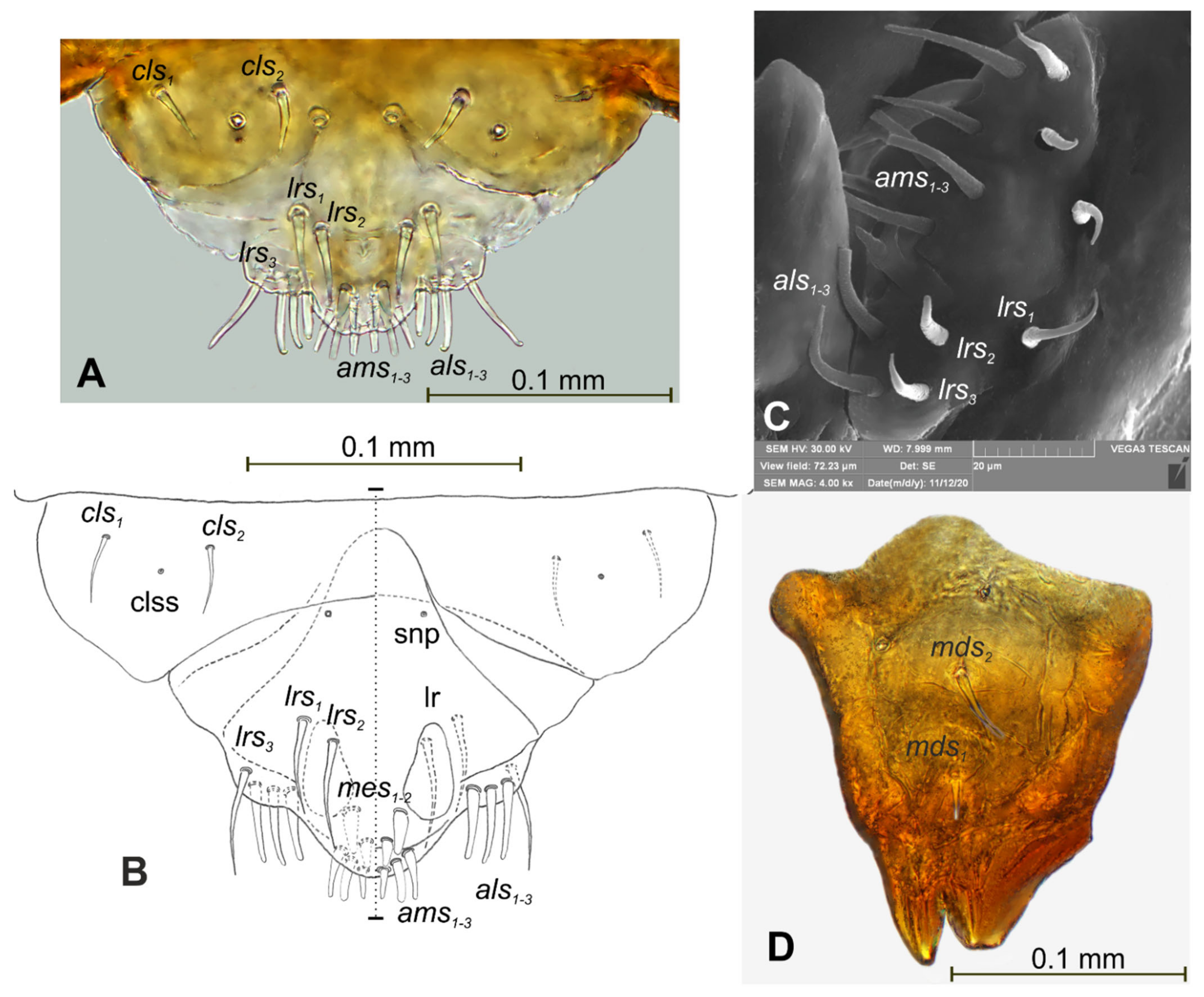
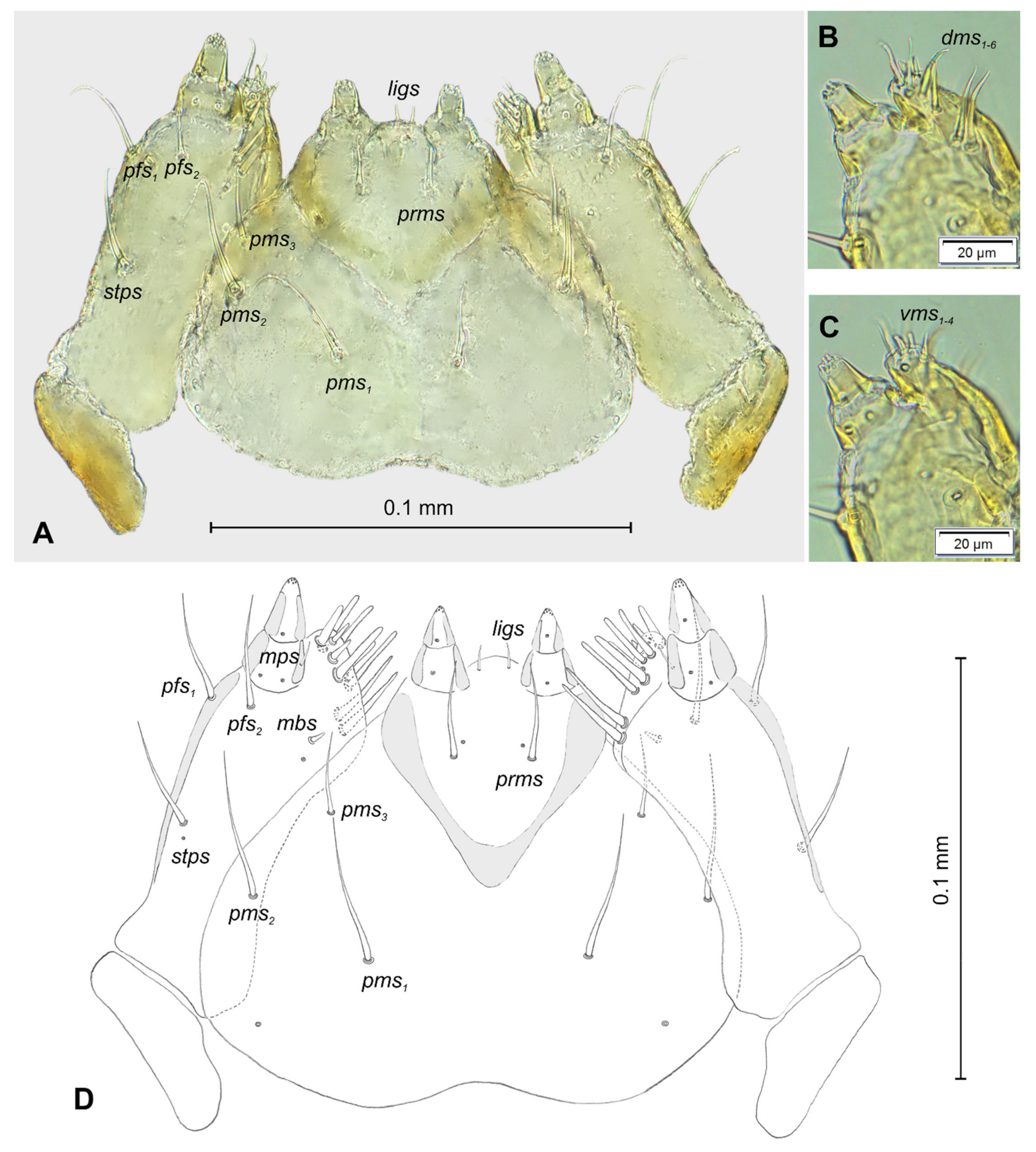
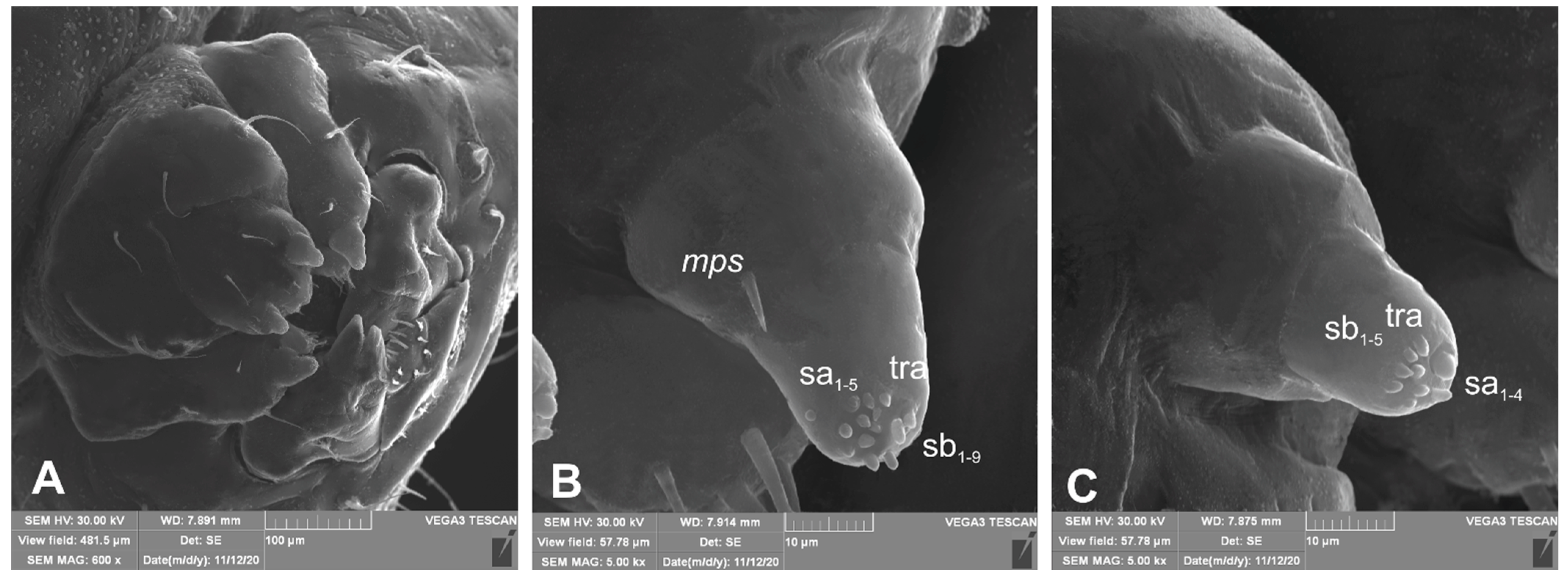
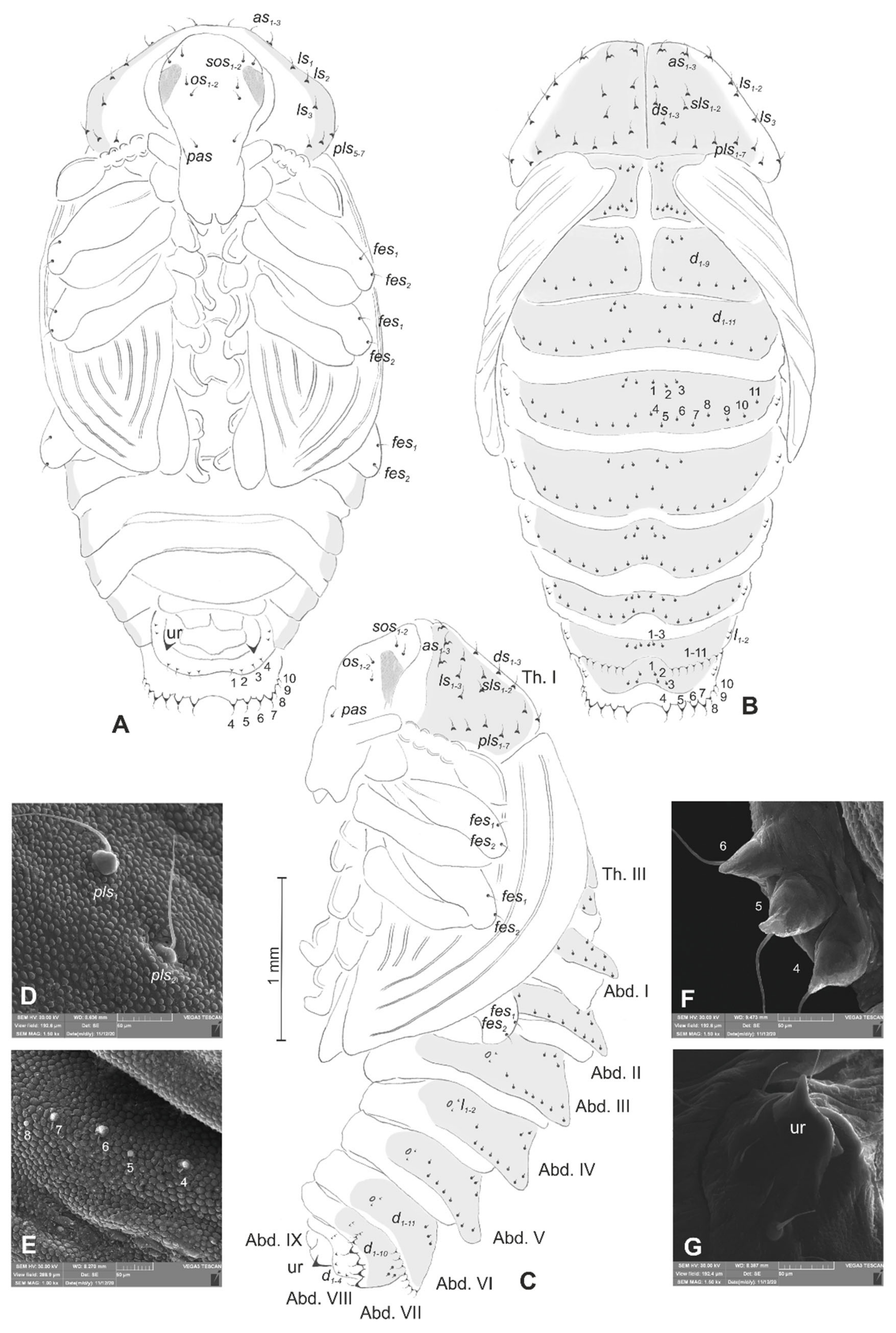
3.1. The Morphology of Immature Stages of Lachnaeus crinitus
3.1.1. Material Examined

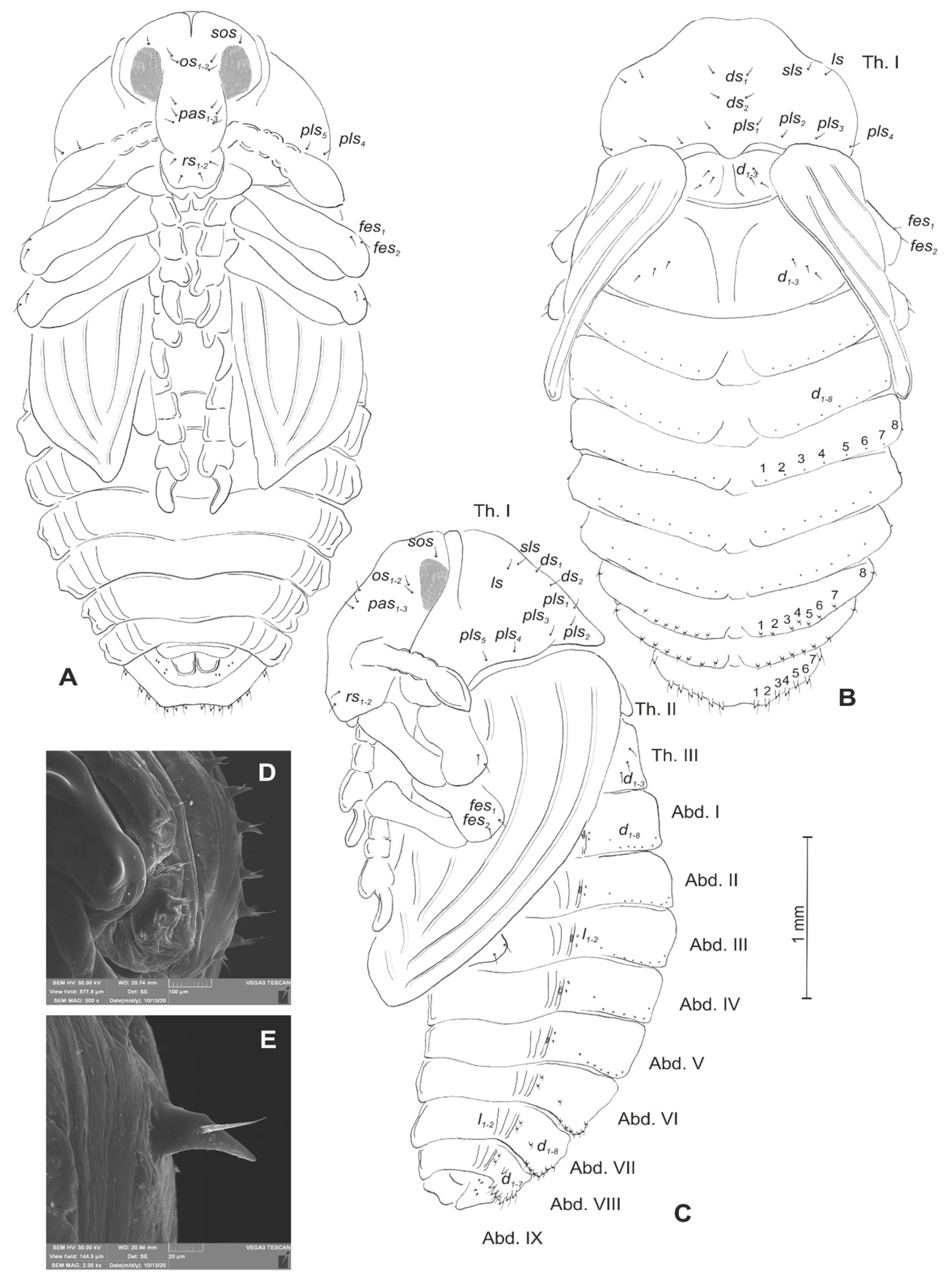
3.1.2. Description of Mature Larva
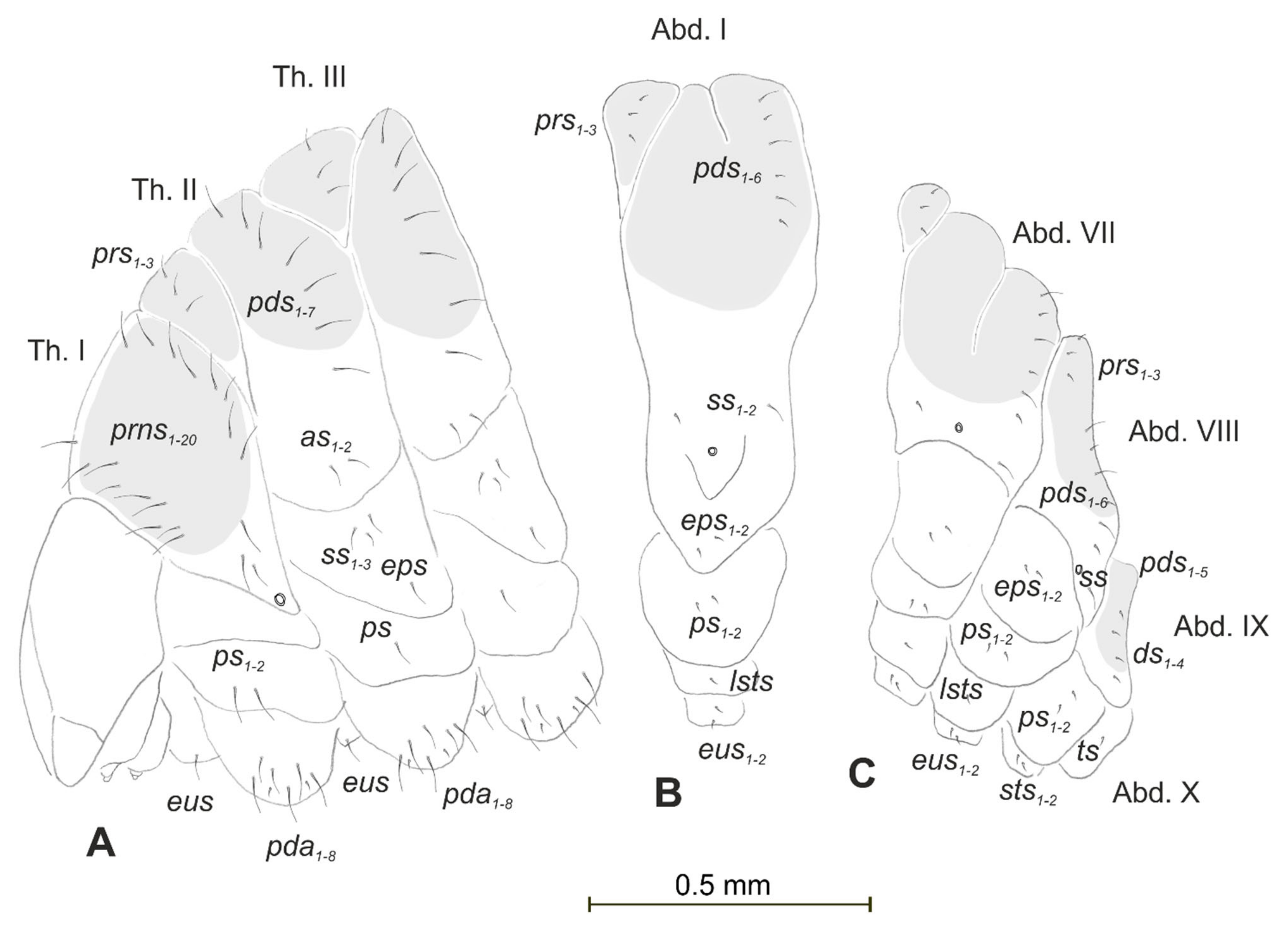
3.1.3. Description of Pupa
3.2. The Morphology of Immature Stages of Rhinocyllus alpinus
3.2.1. Material Examined
3.2.2. Description of Mature Larva
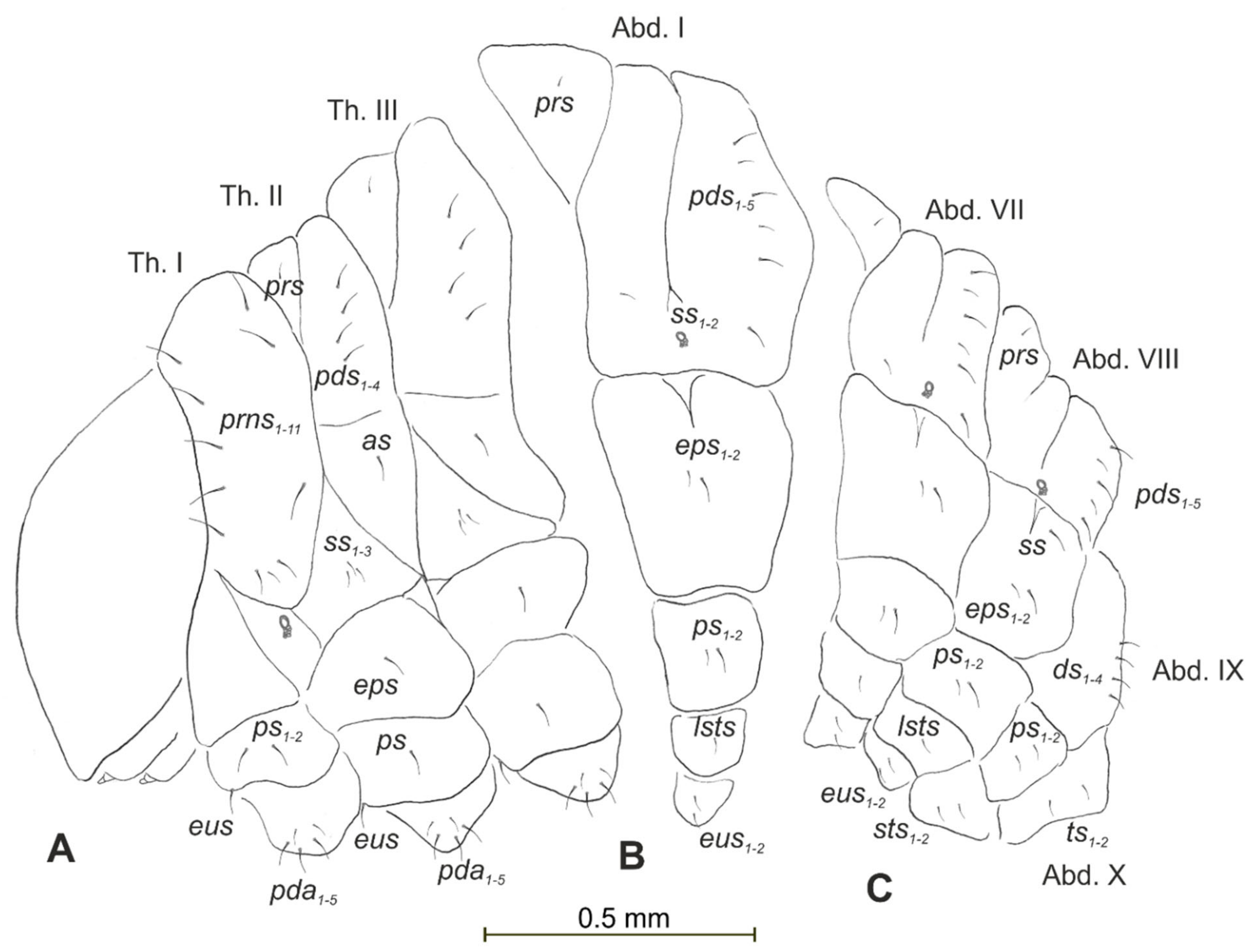
3.2.3. Description of Pupa
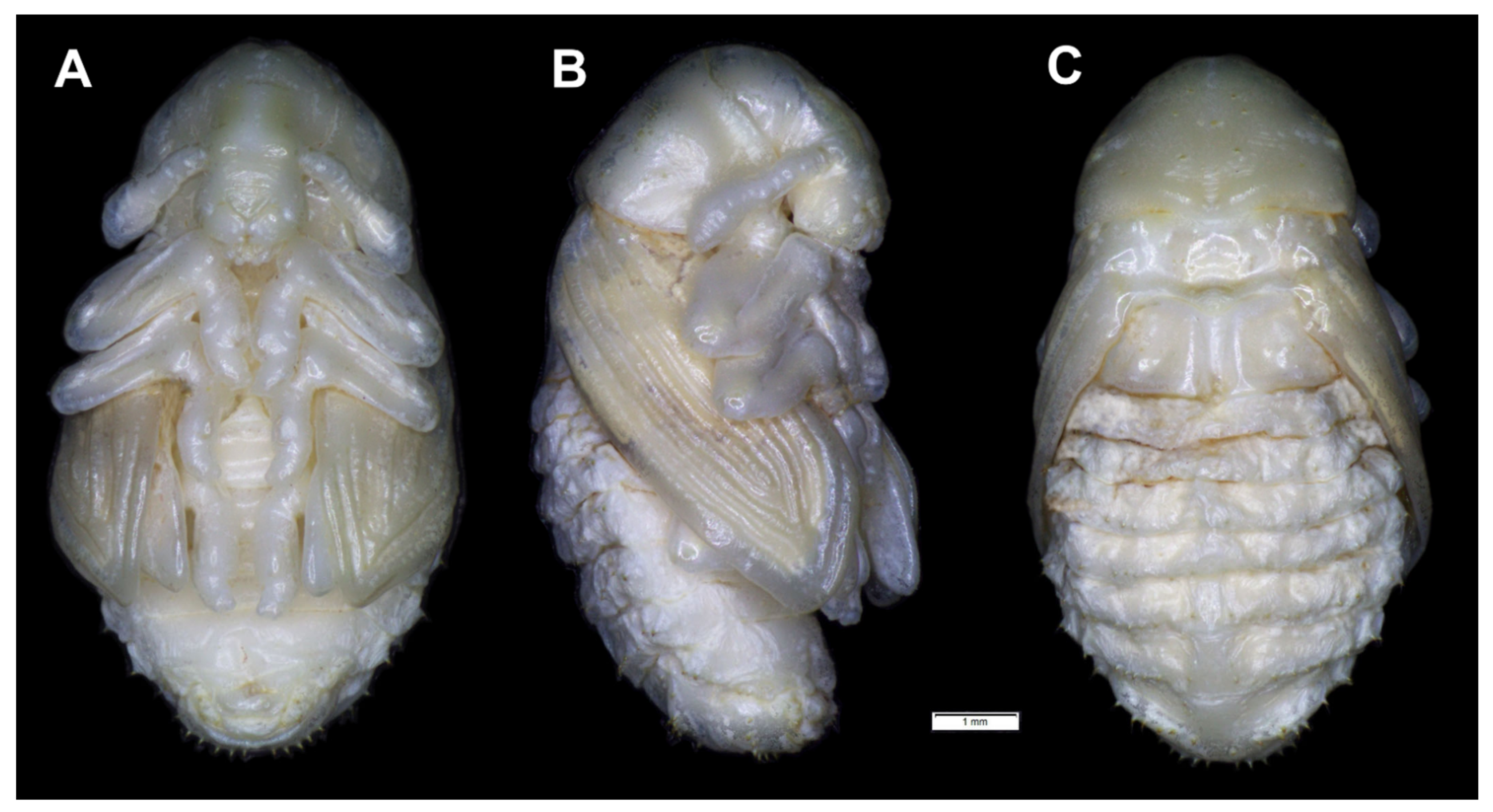
3.3. The Morphology of Immature Stages of Rhinocyllus Conicus
3.3.1. Material Examined
3.3.2. Description of Pupa
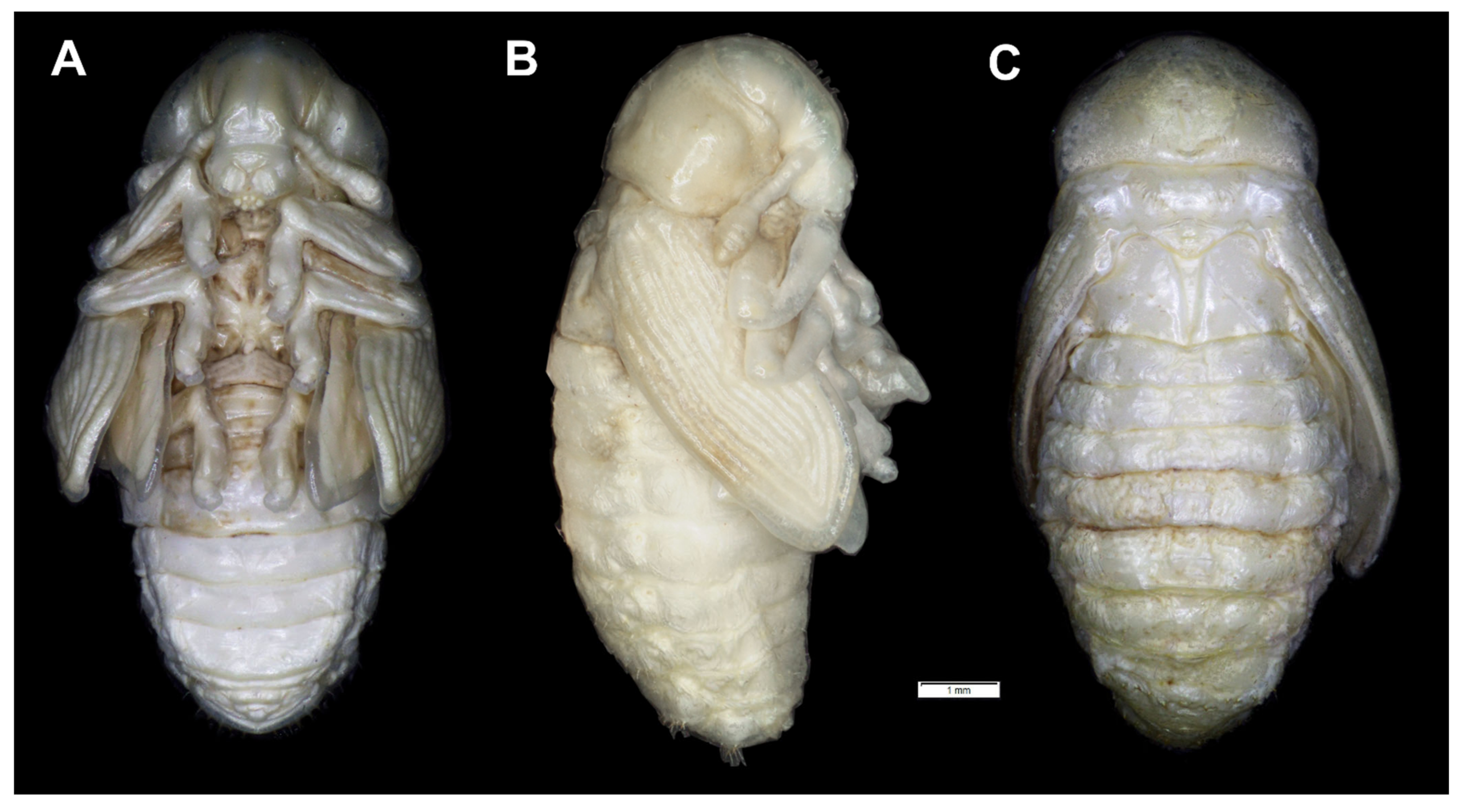
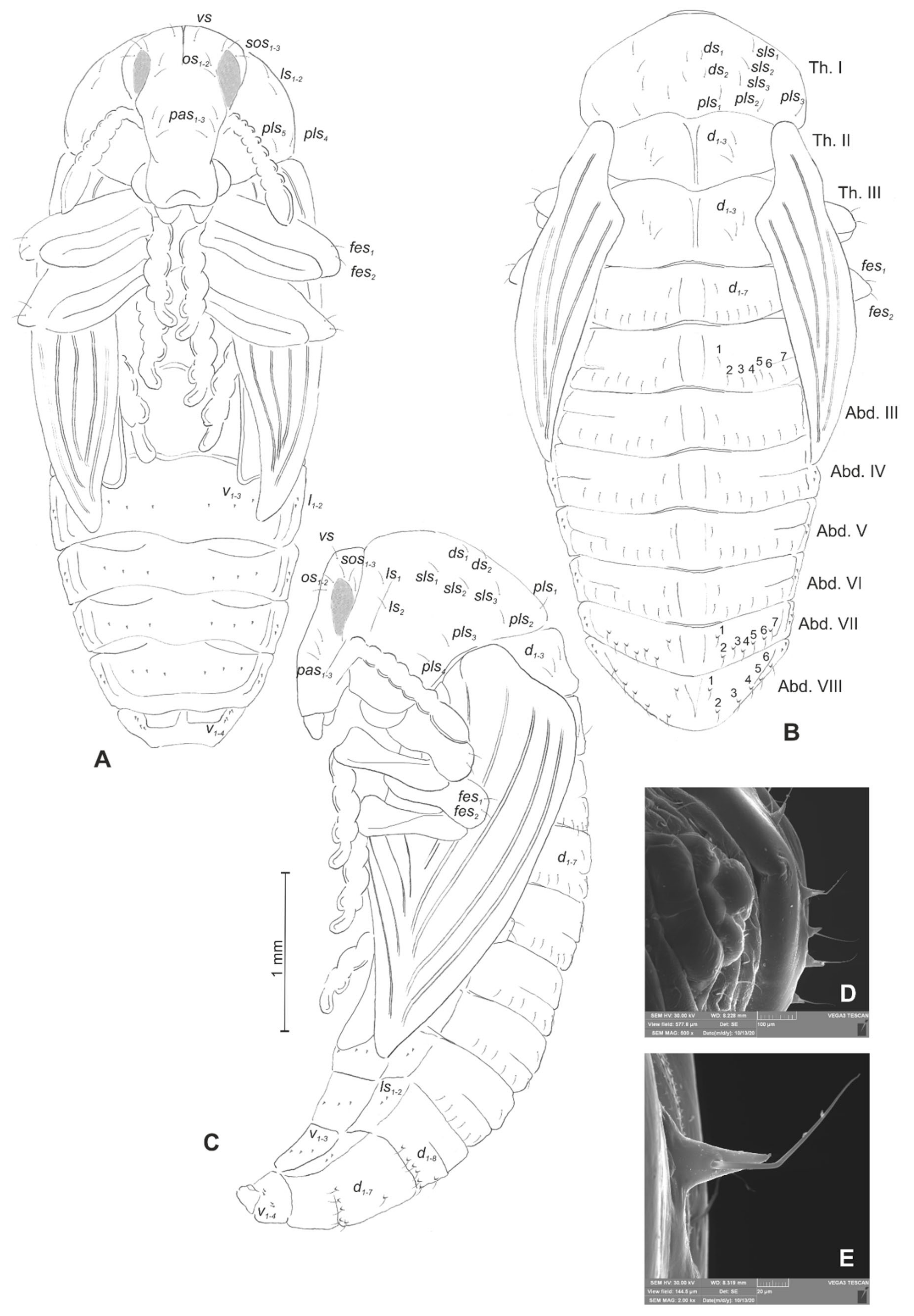
4. Discussion
4.1. A Comparison with Larvae of Other Lixini
4.2. Remarks on Lachnaeus
4.3. Remarks on Rhinocyllus
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
References
- Csiki, E. Curculionidae: Subfam. Cleoninae, Pars 134. In Coleopterorum Catalogus Auspiciis et Auxilio; Schenkling, S., Junk, W., Eds.; W. Junk: Berlin, Germany, 1934; p. 152. [Google Scholar]
- Alonso-Zarazaga, M.A.; Lyal, C.H.C. A World Catalogue of Families and Genera of Curculionoidea (Insecta: Coleoptera) (Excepting Scolytidae and Platypodidae); Entomopraxis: Barcelona, Spain, 1999; p. 315. [Google Scholar]
- Nikulina, O.N.; Gültekin, L.; Güçlü, Ş. Larval morphology of the capitulum weevil, Larinus latus (Herbst) (Coleoptera, Curculionidae). N. Z. J. Zool. 2004, 31, 23–26. [Google Scholar] [CrossRef]
- Seastedt, T.; Knochel, D.; Garmoe, M.; Shosky, S. Interactions and effects of multiple biological control insects on diffuse and spotted knapweed in the Front Range of Colorado. Biol. Control. 2007, 42, 345–354. [Google Scholar] [CrossRef]
- Skuhrovec, J.; Volovnik, S.; Gosik, R.; Stejskal, R.; Trnka, F. Cleonis pigra (Scopoli, 1763) (Coleoptera: Curculionidae: Lixinae): Morphological Re-Description of the Immature Stages, Keys, Tribal Comparisons and Biology. Insects 2019, 10, 325. [Google Scholar] [CrossRef]
- Lacordaire, J.T. Histoire Naturelle Des Insectes. Genera Des Coléoptères Ou Exposé Méthodique Et Critique De Tous Les Genres Proposés Jusqu’ici Dans Cet Ordre D’insectes; Librairie Encyclopédique de Roret: Paris, France, 1863; p. 637. [Google Scholar]
- Capiomont, G. Monographie des Rhinocyllides, Mise en ordre d’après les manuscrits de l’auteur par M. C.-E. Leprieur. Ann. Soc. Entomol. Fr. 1873, 3, 273–296. [Google Scholar]
- Petri, K. Bestimmungs-Tabelle der Gattungen Larinus Germ. (incl. Stolatus Muls.), Microlarinus Hochhuth, Rhinocyllus Germar und Bangasternus Gozis aus dem europäischen, mediterran, west- und nordasiatischen Faunengebiete. In Verhandlungen Des Naturforschenden Vereines in Brünn; Herausgegeben mit Unterstützung der deutschen Gesellschaft für Wissenschaft und Kunst in Brünn: Bonn, Germany, 1914; Volume 45, pp. 51–146. [Google Scholar]
- Pesarini, C. Tabelle per la determinazione dei generi di Curculionidi italiani (Coleoptera). In L’Informatore del Giovane Entomologo, Supplemento al Bollettino della Società Entomologica Italiana; Società Entomologica Italiana: Genova, Italy, 1977; Volume 84, pp. 17–24. [Google Scholar]
- Meregalli, M. 3.7.6. Lixinae Schoenherr, 1823. In Handbook of Zoology. Arthropoda: Insecta. Coleoptera, Beetles: Morphology and Systematics; Kristensen, N.P., Beutel, R.G., Eds.; De Gruyter: Göttingen, Germany, 2014; Volume 3, pp. 523–529. [Google Scholar]
- Arzanov, Y.G.; Grebennikov, V.V. Cleonini (Coleoptera: Curculionidae: Lixinae) are monophyletic and flightless: Tribe over-view, rampant adult homoplasy and illustrated global diversity. Zootaxa 2017, 4329, 1–63. [Google Scholar] [CrossRef] [PubMed]
- Reitter, E. Bestimmungs-Schlüssel der mir bekannten europäischen Gattungen der Curculionidae, mit Einschluss der mir bekannten Gattungen aus dem palaearktischen Gebiete. In Verhandlungen Des Naturforschenden Vereines In Brünn; Herausgegeben mit Unterstützung der deutschen Gesellschaft für Wissenschaft und Kunst in Brünn: Bonn, Germany, 1913; Volume 51, pp. 1–90. [Google Scholar]
- Reitter, E. Fauna Germanica. Die Käfer Des Deutsches Reiches. Nach Der Analytische Methode Bearbeitet; K. G. Lutz’ Verlag: Roetgen, Germany, 1916; p. 343. [Google Scholar]
- Hustache, A. Curculionidae Gallo-Rhénans. Ann. Soc. Entomol. Fr. 1932, 100, 153–470. [Google Scholar]
- Hoffmann, A. Faune De France 59 Coléoptères Curculionides (Deuxième Partie); Fédération Française des Sociétés de Sciences Naturelles: Paris, France, 1955; pp. 487–1208. [Google Scholar]
- Ter-Minasian, M.E. Zhuki-Dolgonosiki Podsemejstva Cleoninae Fauny SSSR. Svetozhily I Stebleedy (Triba Lixini). Nauka, Leningrad, 140 [+ 1 Unnumbered] P. (Translated 1978. Weevils Of The Subfamily Cleoninae In The Fauna Of The USSR. Tribe Lixini; ARS-USDA and National Science Foundation: Washington, DC, USA; Amerind Publishing Co.: New Delhi, India, 1967; p. 166. [Google Scholar]
- Colonnelli, E. A revised checklist of Italian Curculionoidea (Coleoptera). Zootaxa 2003, 337, 1–142. [Google Scholar] [CrossRef]
- Gültekin, L. A new weevil genus Nefis gen. nov. (Coleoptera: Curculionidae: Lixinae): Systematics and taxonomic revision. J. Insect Biodivers. 2013, 1, 1–51. [Google Scholar] [CrossRef]
- Gültekin, L.; Fremuth, J. Tribe Lixini. In Catalogue of Palaearctic Coleoptera, Volume 8, Curculionoidea II; Löbl, I., Smetana, A., Eds.; BRILL: Leiden, The Netherlands, 2013; pp. 456–472. [Google Scholar]
- Skuhrovec, J.; Volovnik, S. Biology and morphology of immature stages of Lixus canescens (Coleoptera: Curculionidae: Lixinae). Zootaxa 2015, 4033, 350–362. [Google Scholar] [CrossRef]
- Trnka, F.; Stejskal, R.; Skuhrovec, J. The morphology of the immature stages of two rare Lixus species (Coleoptera, Curculionidae, Lixinae) and notes on their biology. ZooKeys 2016, 604, 87–116. [Google Scholar] [CrossRef]
- Skuhrovec, J.; Volovnik, S.; Gosik, R. Description of the immature stages of Larinus vulpes and notes on its biology (Coleoptera, Curculionidae, Lixinae). ZooKeys 2017, 679, 107–137. [Google Scholar] [CrossRef]
- Skuhrovec, J.; Bogusch, P. The morphology of the immature stages of Metadonus vuillefroyanus (Capiomont, 1868) (Coleoptera, Curculionidae, Hyperini) and notes on its biology. ZooKeys 2016, 589, 123–142. [Google Scholar] [CrossRef]
- May, B.M. An introduction to the immature stages of Australian Curculionoidea, subfamily Bagoinae. In Australian Weevils (Coleoptera: Curculionidae); Zimmerman, E.C., Ed.; CSIRO: Melbourne, Australia, 1994; Volume 2, pp. 533–535. [Google Scholar]
- Hille Ris Lambers, D. On mounting aphids and other soft–skinned insects. Entomol. Ber. 1950, 13, 55–58. [Google Scholar]
- Anderson, W.H. A terminology for the anatomical characters useful in the taxonomy of weevil larvae. Proc. Entomol. Soc. 1947, 49, 123–132. [Google Scholar]
- Marvaldi, A.E. Larvae of South American Rhytirrhininae (Coleoptera: Curculionidae). Coleopt. Bull. 1998, 52, 71–89. [Google Scholar]
- Marvaldi, A.E. Morfología larval en Curculionidae. Acta Zoológica Lilloana 1999, 45, 7–24. [Google Scholar]
- Skuhrovec, J.; Gosik, R.; Caldara, R.; Košťál, M. Immatures of Palaearctic species of the weevil genus Sibinia (Coleoptera, Curculionidae): New descriptions and new bionomic data with suggestions on their potential value in a phylogenetic reconstruction of the genus. Zootaxa 2015, 3955, 151–187. [Google Scholar] [CrossRef]
- Zacharuk, R.Y. Antennae and sensilla. In Comparative Insects Physiology, Chemistry and Pharmacology; Kerkut, G.A., Gilbert, L.I., Eds.; Pergamon Press: Oxford, UK, 1985; Volume 6, pp. 1–69. [Google Scholar]
- Gosik, R.; Wanat, M. Descriptions of immature stages of the weevil Lixus punctiventris Boheman, 1835 (Coleoptera, Curculionidae, Lixini). Zootaxa 2014, 3754, 159–172. [Google Scholar] [CrossRef]
- Gardner, J.C.M. Immature stages of Indian Coleoptera (14) (Curculionidae). Indian For. Rec. 1934, 20, 1–48. [Google Scholar]
- Scherf, H. Die Entwicklungsstadien der Mitteleuropäischen Curculioniden (Morphologie, Bionomie, Ökologie). Abh. Senckenbergischen Nat. Ges. 1964, 506, 1–335. [Google Scholar]
- Lee, C.-Y.; Morimoto, K. Larvae of the Weevil Family Curculionidae of Japan Part 2. Hyperinae to Cioninae (Insecta: Coleoptera). J. Fac. Agric. Kyushu Univ. 1988, 33, 131–152. [Google Scholar] [CrossRef]
- Zotov, A.A. Morphology of the preimaginal stages of three species of weevil of the Lixini (Coleoptera: Curculionidae). Cauc. Èntomol. Bull. 2009, 5, 81–90. [Google Scholar] [CrossRef]
- Zotov, A.A. Morphology of preimaginal stages of the genus Larinus Dejean, 1821 (Coleoptera: Curculionidae). Part I. Cauc. Èntomol. Bull. 2010, 6, 171–178. [Google Scholar] [CrossRef]
- Gosik, R.; Skuhrovec, J. Descriptions of mature larvae and pupae of the genus Larinus (Coleoptera: Curculionidae, Lixinae). Zootaxa 2011, 3019, 1–25. [Google Scholar] [CrossRef]
- Nikulina, O.N.; Gültekin, L. New data on the larvae of the weevil genus Larinus Dejean, 1821 (Coleoptera, Curculionidae) from northeastern Turkey. Èntomol. Rev. 2014, 94, 1010–1018. [Google Scholar] [CrossRef]
- Nikulina, O.N. New data on larvae of weevils of the genus Lixus (Coleoptera, Curculionidae) from central Asia. Èntomol. Rev. 2007, 87, 750–756. [Google Scholar] [CrossRef]
- Zotov, A.A. Morphology of the preimaginal stages of weevil Lixus iridis Olivier, 1807 (Coleoptera: Curculionidae). Cauc. Èntomol. Bull. 2009, 5, 249–252. [Google Scholar] [CrossRef]
- Nikulina, O.N.; Gültekin, L. Larval morphology of Lixus cardui Olivier and Lixus filiformis (Fabricius) (Coleoptera: Curculionidae): Biological control agents for scotch and musk thistles. Aust. J. Èntomol. 2011, 50, 253–257. [Google Scholar] [CrossRef]
- Gosik, R.; Skuhrovec, J.; Toševski, I.; Caldara, R. Morphological evidence from immature stages further suggests Lignyodina being close to Tychiina (Coleoptera, Curculionidae, Curculioninae, Tychiini). Zootaxa 2017, 4320, 426–446. [Google Scholar] [CrossRef]
- Gosik, R.; Sprick, P.; Skuhrovec, J.; Deruś, M.; Hommes, M. Morphology and identification of the mature larvae of several species of the genus Otiorhynchus (Coleoptera, Curculionidae, Entiminae) from Central Europe with an update of the life history traits. Zootaxa 2016, 4108, 1–67. [Google Scholar] [CrossRef]
- Jiang, C.; Caldara, R.; Skuhrovec, J.; Zhang, R. Description and biological notes of the larva of Cionus olivieri Rosenschoeld, 1838 (Coleoptera, Curculionidae), with a comparison with other species of the tribe Cionini. ZooKeys 2020, 976, 131–145. [Google Scholar] [CrossRef]
- Skuhrovec, J.; Gosik, R.; Caldara, R.; Toševski, I.; Łętowski, J.; Szwaj, E. Morphological characters of immature stages of Palaearctic species of Cleopomiarus and Miarus and their systematic value in Mecinini (Coleoptera, Curculionidae, Curculioninae). ZooKeys 2018, 808, 23–92. [Google Scholar] [CrossRef] [PubMed]
- Gosik, R.; Skuhrovec, J.; Caldara, R.; Toševski, I. Immature stages of Palearctic Mecinus species (Coleoptera, Curculionidae, Curculioninae): Morphological characters diagnostic at genus and species levels. ZooKeys 2020, 939, 87–165. [Google Scholar] [CrossRef]
- Caldara, R.; Skuhrovec, J.; Gosik, R.; Marvaldi, A.E. On the affinities and systematic position of Pachytychius Jekel, a genus currently incertae sedis in Coleoptera, Curculionidae: Evidence from immature stages and the COI gene support its placement in Smicronychini. Zool. Anz. A J. Comp. Zool. 2018, 277, 218–230. [Google Scholar] [CrossRef]
- Hlaváč, P.; Skuhrovec, J.; Pelikán, J. A new, peculiar genus of Cossoninae (Coleoptera, Curculionidae) from Oman with description of a new species, larva and notes on biology. Zootaxa 2020, 4768, 129–142. [Google Scholar] [CrossRef] [PubMed]
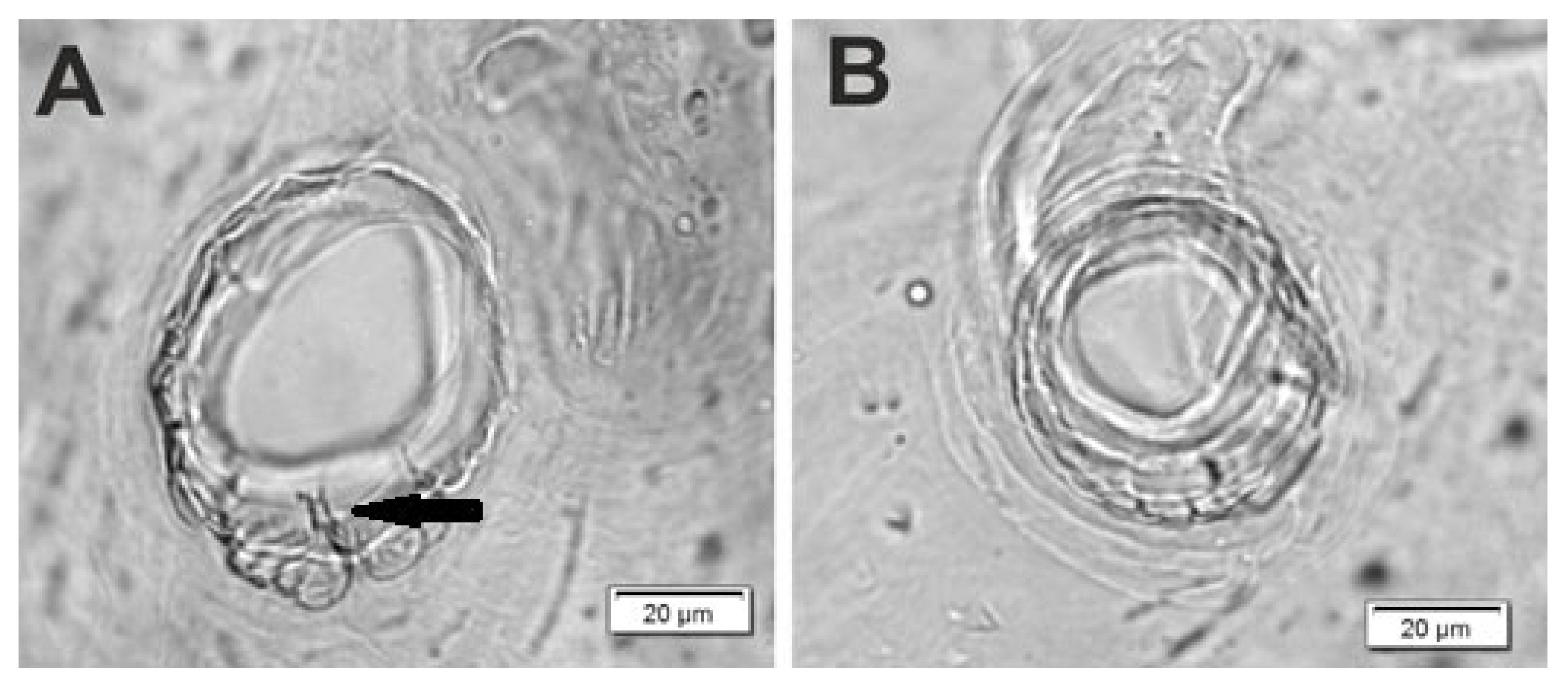
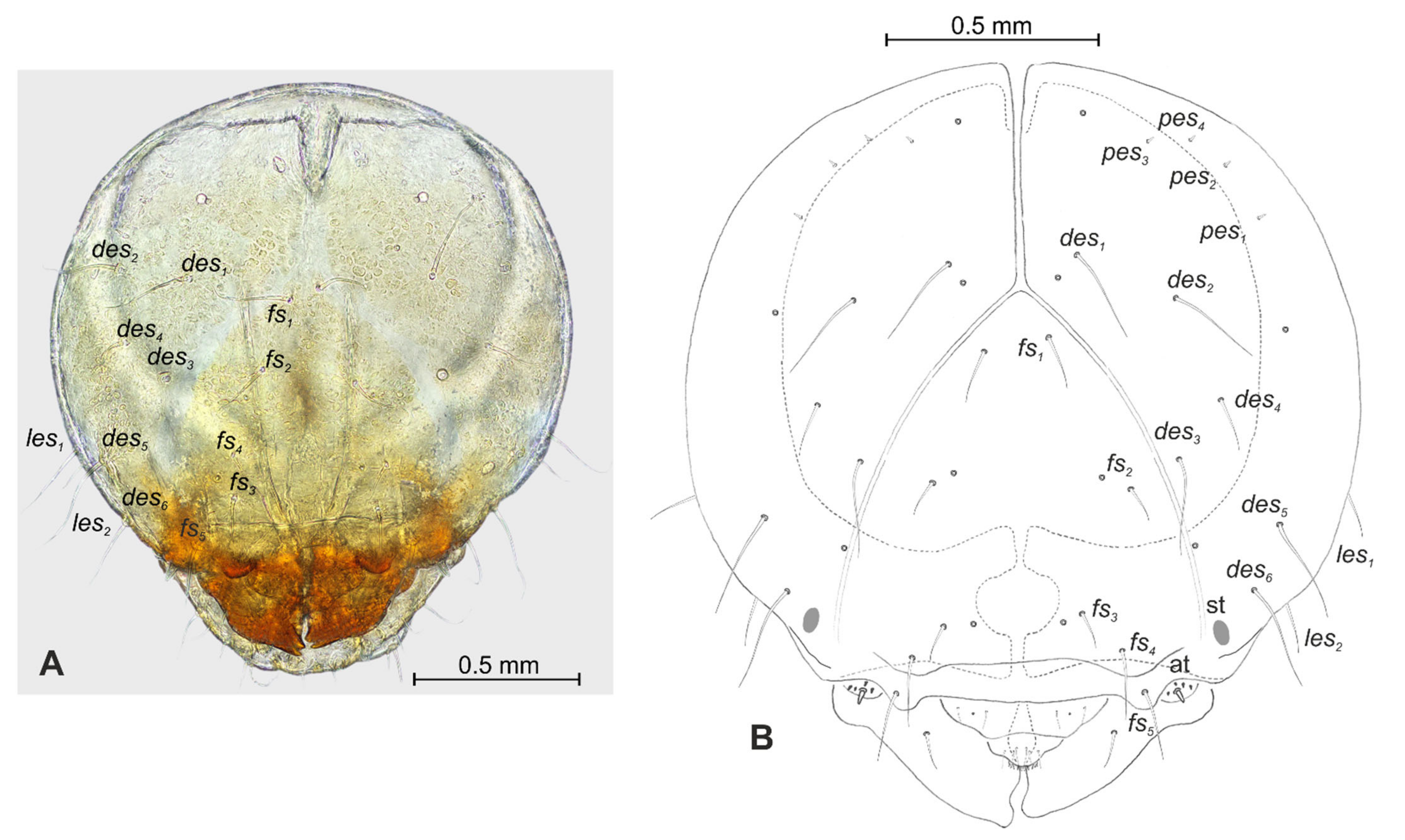


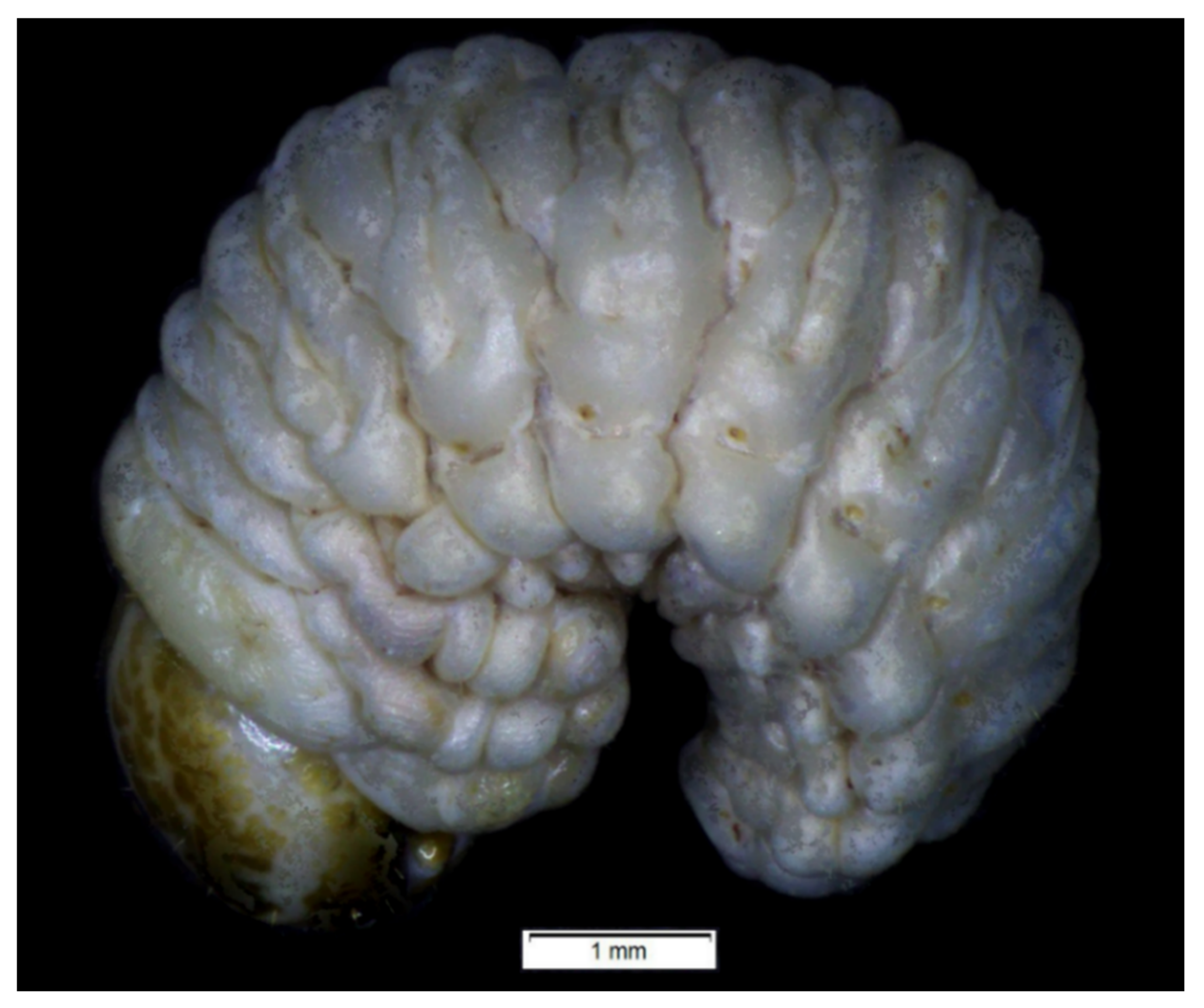
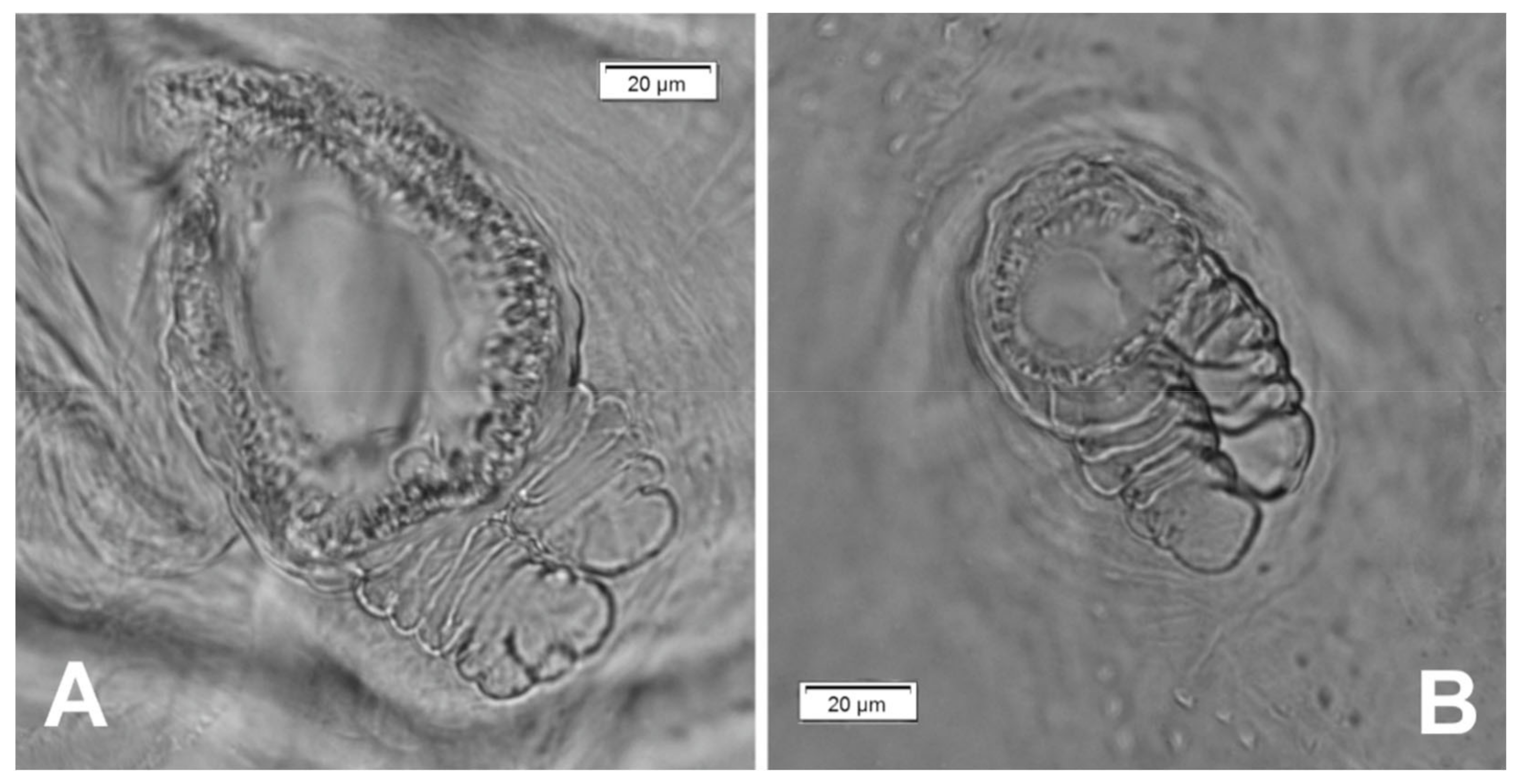

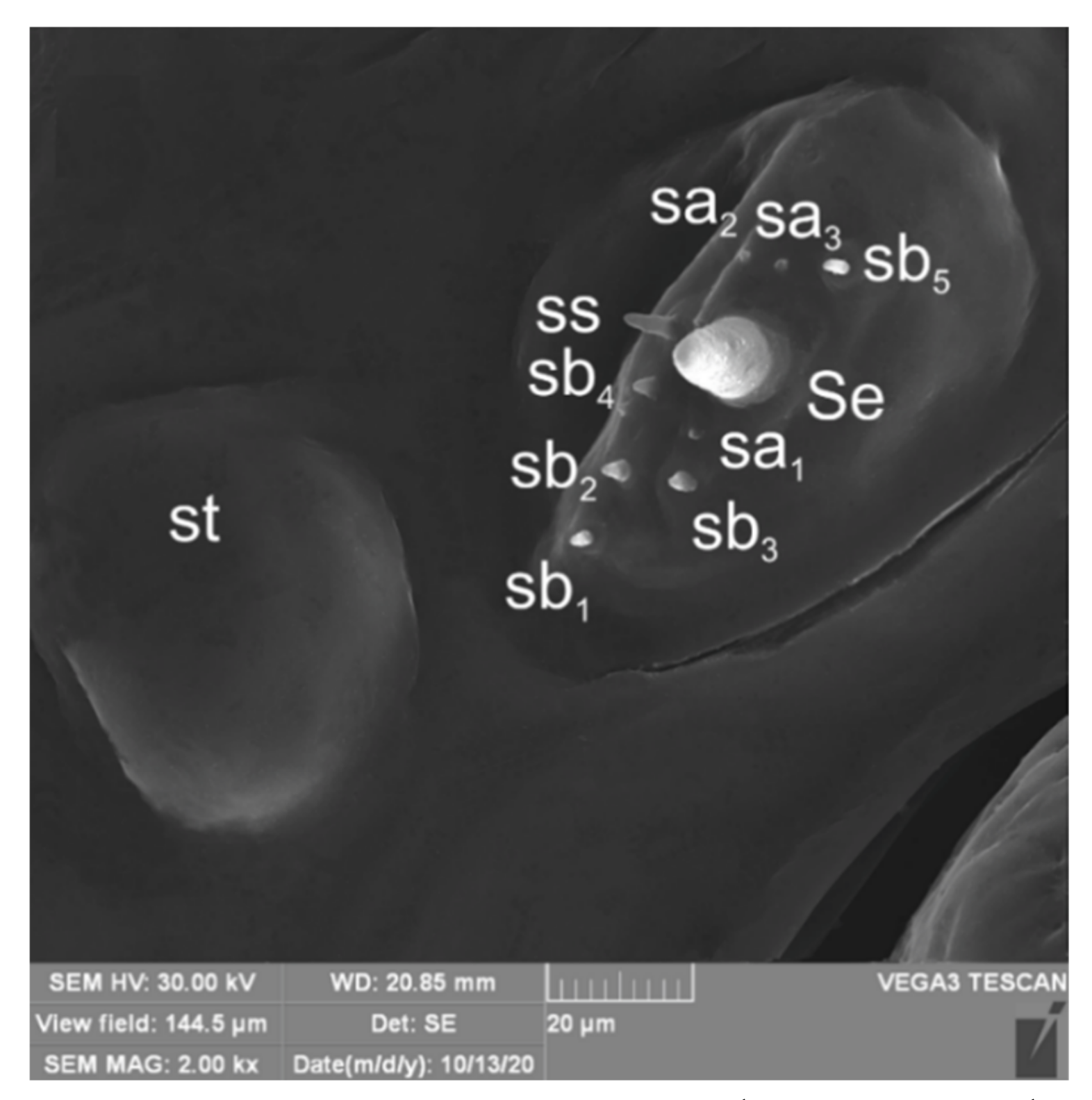
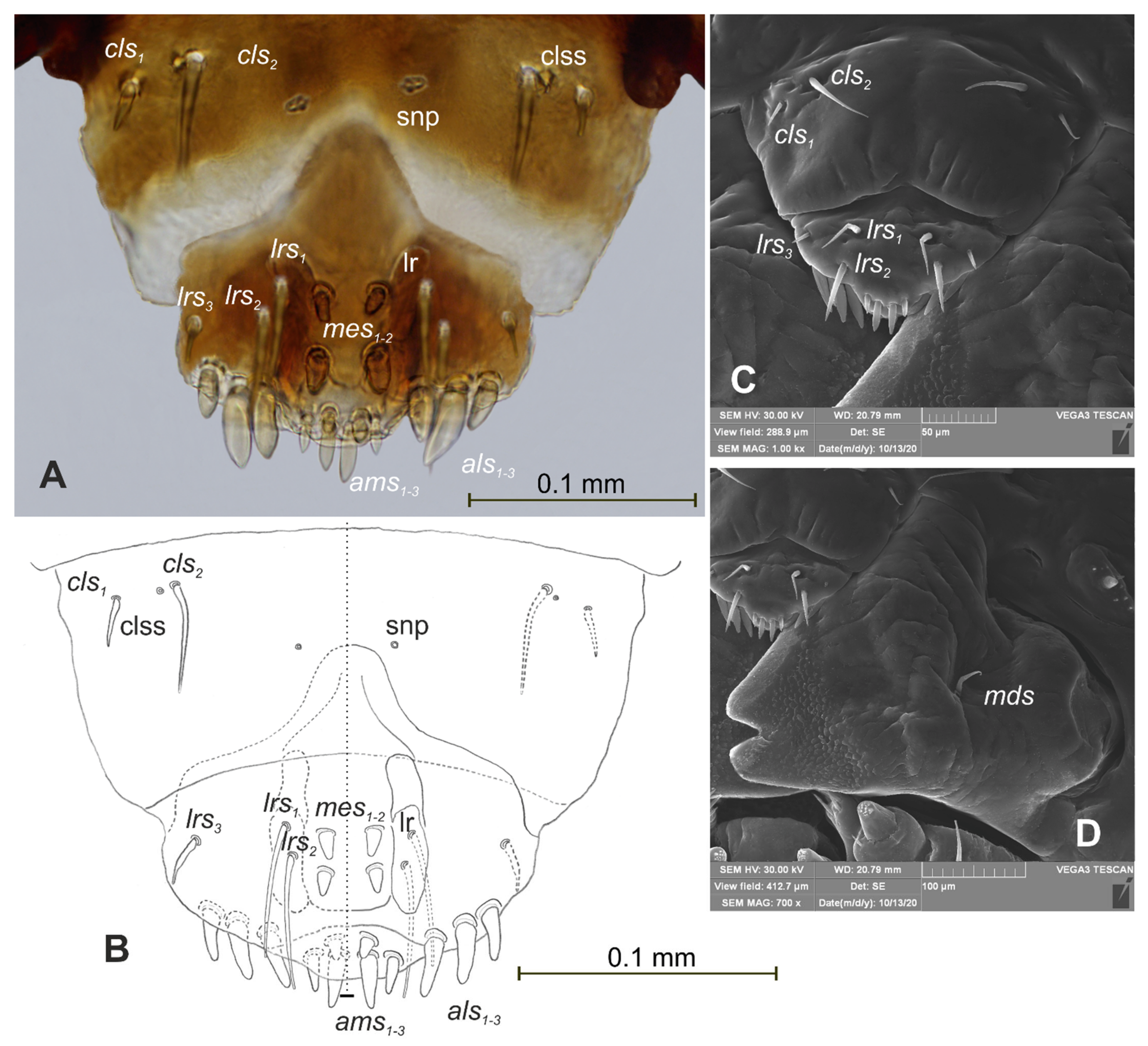

| Stages | Lachnaeus crinitus | Rhinocyllus alpinus | Rhinocyllus conicus | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Larva | HW | BL | BW | HW | BL | BW | |||
| 0.90 1, 1.25 1, 1.40 1, 1.50 1, 1.75 1 | 2.50 1, 3.25 1, 4.00 2, 4.25 1 | 1.00 1, 1.20 4 | 1.20 3, 1.25 19, 1.30 7 (mv: 1.25) | 3.25 2, 3.50 5, 4.00 2, 4.25 5, 4.50 10, 4.80 2, 5.00 2 (mv: 4.50) | 1.75 4, 2.00 6, 2.25 5, 2.50 8, 2.75 5, 3.00 1 (mv: 2.80) | ||||
| Pupa ♀ | THW | BL | BW | THW | BL | BW | THW | BL | BW |
| 1.55 1 | 6.50 1 | 3.501 | 2.10 2, 2.30 2 (mv: 2.20) | 5.50 1, 6.00 3 (mv: 6.00) | 3.30 1, 3.40 3 (mv: 3.40) | 1.60 2, 1.80 2, 2.00 1 (mv: 1.80) | 4.50 1, 5.20 3, 5.50 1 (mv: 5.20) | 2.20 2, 2.40 2, 2.90 1 (mv: 2.40) | |
| Pupa ♂ | 1.50 1, 1.70 4 (mv: 1.70) | 5.00 1, 6.40 4 (mv: 6.00) | 3.00 1, 3.50 4 (mv: 3.40) | 2.10 10, 2.20 2 (mv: 2.10) | 5.00 3, 5.50 4, 6.50 5 (mv: 6.50) | 3.00 7, 3.40 3, 3.50 2 (mv: 3.00) | 1.50 3, 2.00 6, 2.10 1 (mv: 2.00) | 4.50 1, 5.20 3, 6.10 4, 7.00 2 (mv: 6.10) | 2.10 4, 3.00 6, 3.40 2 (mv: 3.00) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skuhrovec, J.; Caldara, R.; Gosik, R.; Trnka, F.; Stejskal, R. On the Affinities and Systematic Position of Lachnaeus Schoenherr and Rhinocyllus Germar in the Tribe Lixini (Coleoptera: Curculionidae: Lixinae) Based on the Morphological Characters of the Immature Stages. Insects 2021, 12, 489. https://doi.org/10.3390/insects12060489
Skuhrovec J, Caldara R, Gosik R, Trnka F, Stejskal R. On the Affinities and Systematic Position of Lachnaeus Schoenherr and Rhinocyllus Germar in the Tribe Lixini (Coleoptera: Curculionidae: Lixinae) Based on the Morphological Characters of the Immature Stages. Insects. 2021; 12(6):489. https://doi.org/10.3390/insects12060489
Chicago/Turabian StyleSkuhrovec, Jiří, Roberto Caldara, Rafał Gosik, Filip Trnka, and Robert Stejskal. 2021. "On the Affinities and Systematic Position of Lachnaeus Schoenherr and Rhinocyllus Germar in the Tribe Lixini (Coleoptera: Curculionidae: Lixinae) Based on the Morphological Characters of the Immature Stages" Insects 12, no. 6: 489. https://doi.org/10.3390/insects12060489
APA StyleSkuhrovec, J., Caldara, R., Gosik, R., Trnka, F., & Stejskal, R. (2021). On the Affinities and Systematic Position of Lachnaeus Schoenherr and Rhinocyllus Germar in the Tribe Lixini (Coleoptera: Curculionidae: Lixinae) Based on the Morphological Characters of the Immature Stages. Insects, 12(6), 489. https://doi.org/10.3390/insects12060489






