COVID-19 Severity and Mortality after Vaccination against SARS-CoV-2 in Central Greece
Abstract
:1. Introduction
2. Materials and Methods
2.1. Settings
2.2. Participants and Study Design
2.3. Ethical Considerations
2.4. Statistical Analysis
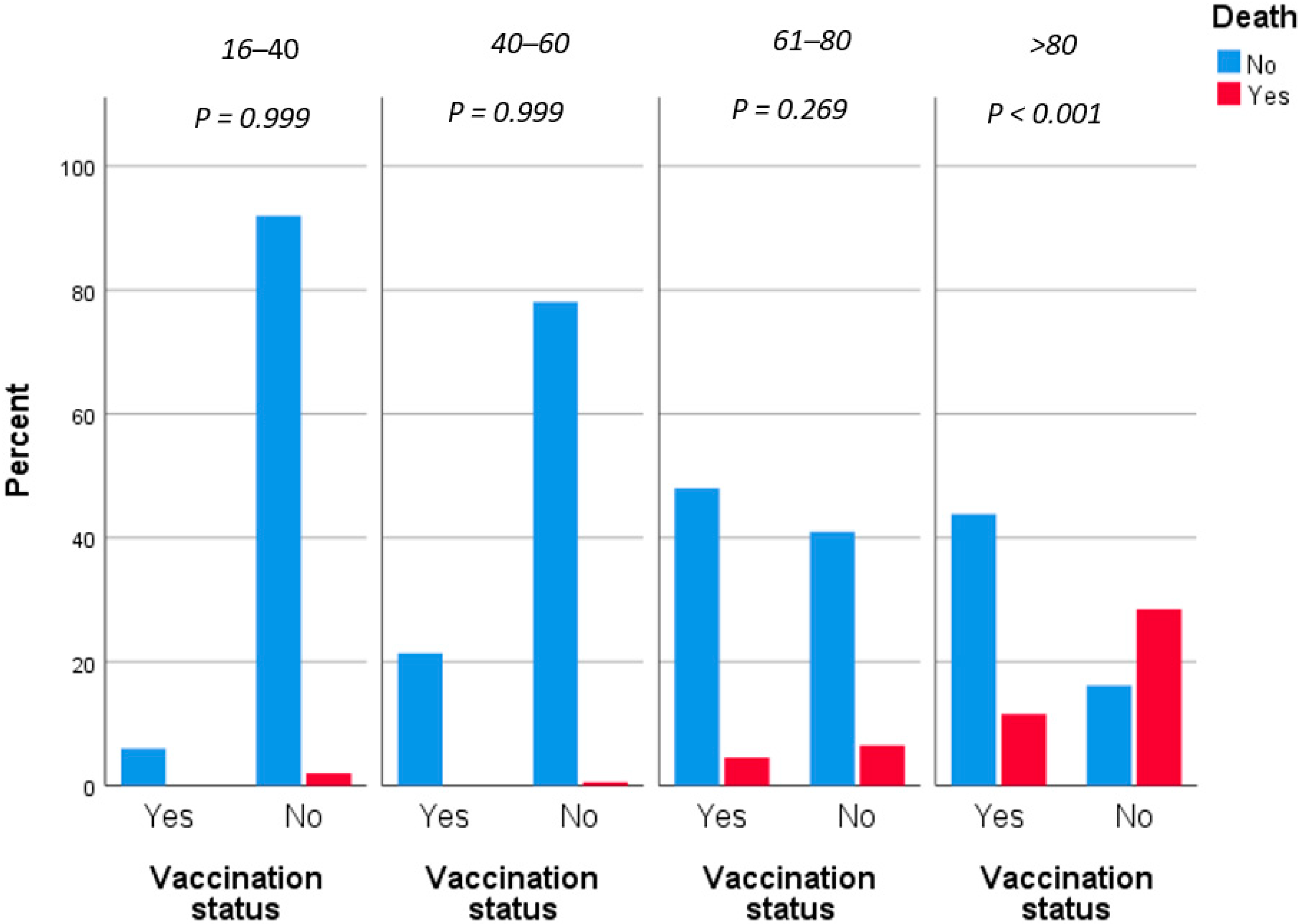
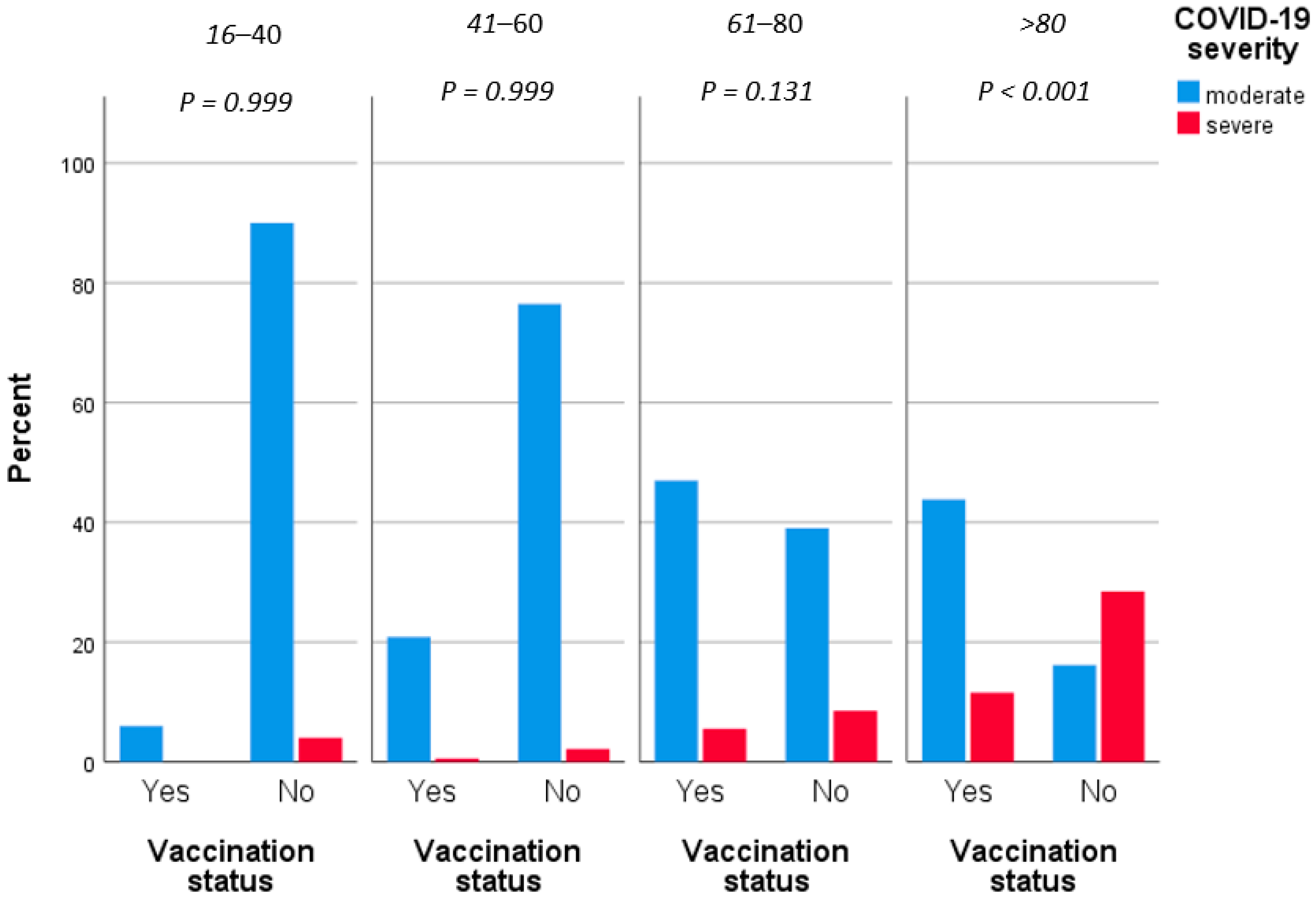
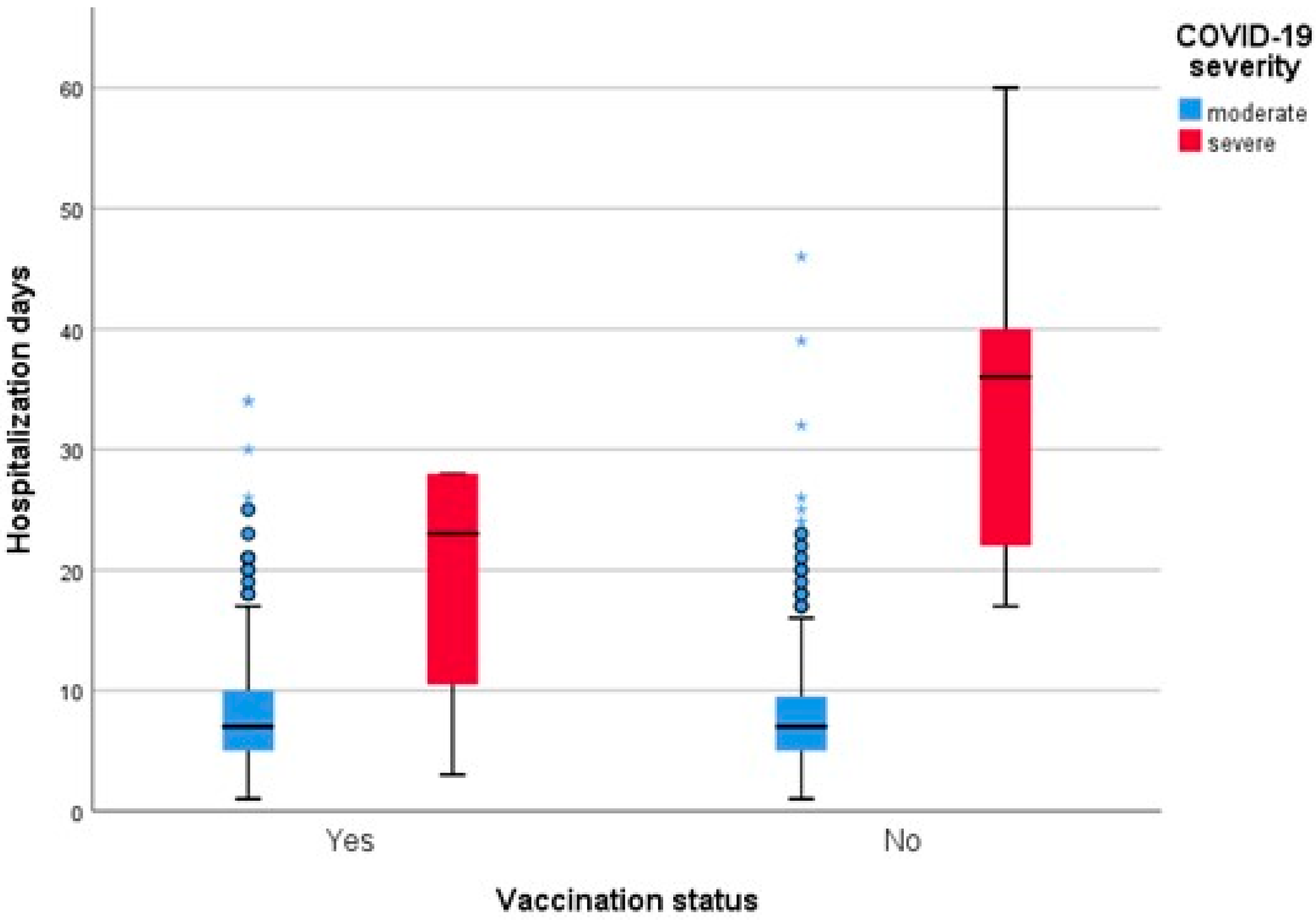
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 8 January 2022).
- Wang, Y.; Wang, Y.; Chen, Y.; Qin, Q. Unique epidemiological and clinical features of the emerging 2019 novel coronavirus pneumonia (COVID-19) implicate special control measures. J. Med. Virol. 2020, 92, 568–576. [Google Scholar] [CrossRef] [PubMed]
- Jain, U. Effect of COVID-19 on the Organs. Cureus 2020, 12, e9540. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Chen, K.; Zou, J.; Han, P.; Hao, J.; Han, Z. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front. Med. 2020, 14, 185–192. [Google Scholar] [CrossRef]
- Maximiano Sousa, F.; Roelens, M.; Fricker, B.; Thiabaud, A.; Iten, A.; Cusini, A.; Flury, D.; Buettcher, M.; Zukol, F.; Balmelli, C.; et al. Risk factors for severe outcomes for COVID-19 patients hospitalised in Switzerland during the first pandemic wave, February to August 2020: Prospective observational cohort study. Swiss Med. Wkly. 2021, 151, w20547. [Google Scholar] [CrossRef] [PubMed]
- Sotiriou, S.; Samara, A.A.; Lachanas, K.E.; Vamvakopoulou, D.; Vamvakopoulos, K.-O.; Vamvakopoulos, N.; Janho, M.B.; Perivoliotis, K.; Donoudis, C.; Daponte, A.; et al. Vulnerability of β-Thalassemia Heterozygotes to COVID-19: Results from a Cohort Study. J. Pers. Med. 2022, 12, 352. [Google Scholar] [CrossRef]
- Mohammed, I.; Nauman, A.; Paul, P.; Ganesan, S.; Chen, K.-H.; Jalil, S.M.S.; Jaouni, S.H.; Kawas, H.; Khan, W.A.; Vattoth, A.L.; et al. The efficacy and effectiveness of the COVID-19 vaccines in reducing infection, severity, hospitalization, and mortality: A systematic review. Hum. Vaccines Immunother. 2022, 18, 2027160. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Z.; Kuan, C.C. Vaccination to reduce severe COVID-19 and mortality in COVID-19 patients: A systematic review and meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 1770–1776. [Google Scholar] [CrossRef]
- Lopez Bernal, J.; Andrews, N.; Gower, C.; Robertson, C.; Stowe, J.; Tessier, E.; Simmons, R.; Cottrell, S.; Roberts, R.; O’Doherty, M.; et al. Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on COVID-19 related symptoms, hospital admissions, and mortality in older adults in England: Test negative case-control study. BMJ 2021, 373, n1088. [Google Scholar] [CrossRef]
- Gravelle, T.B.; Phillips, J.B.; Reifler, J.; Scotto, T.J. Estimating the size of “anti-vax” and vaccine hesitant populations in the US, UK, and Canada: Comparative latent class modeling of vaccine attitudes. Hum. Vaccines Immunother. 2022, 18, 2008214. [Google Scholar] [CrossRef]
- Joshi, G.; Poduri, R. Omicron, a new SARS-CoV-2 variant: Assessing the impact on severity and vaccines efficacy. Hum. Vaccines Immunother. 2022, 18, 2034458. [Google Scholar] [CrossRef]
- Karim, S.S.A.; Karim, Q.A. Omicron SARS-CoV-2 Variant: A New Chapter in the COVID-19 Pandemic. Lancet 2021, 398, 2126–2128. [Google Scholar] [CrossRef]
- He, X.M.; Hong, W.Q.; Pan, X.Y.; Lu, G.W.; Wei, X.W. SARS-CoV-2 Omicron variant: Characteristics and prevention. MedComm 2021, 2, 838–845. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Living Guidance for Clinical Management of COVID-19. Available online: https://apps.who.int/iris/bitstream/handle/10665/349321/WHO-2019-nCoV-clinical-2021.2-eng.pdf (accessed on 8 July 2022).
- Nonaka, C.K.; Franco, M.M.; Gräf, T.; de Lorenzo Barcia, C.A.; de Ávila Mendonça, R.N.; de Sousa, K.A.; Neiva, L.M.; Fosenca, V.; Mendes, A.V.; de Aguiar, R.S.; et al. Genomic Evidence of SARS-CoV-2 Reinfection Involving E484K Spike Mutation, Brazil. Emerg. Infect. Dis. 2021, 27, 1522–1524. [Google Scholar] [CrossRef] [PubMed]
- Halvatsiotis, P.; Vassiliu, S.; Koulouvaris, P.; Chatzantonaki, K.; Asonitis, K.; Charvalos, E.; Siatelis, A.; Houhoula, D. Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Mutational Pattern in the Fourth Pandemic Phase in Greece. Curr. Issues Mol. Biol. 2022, 44, 329–335. [Google Scholar] [CrossRef]
- Del Rio, C.; Omer, S.B.; Malani, P.N. Winter of Omicron—The Evolving COVID-19 Pandemic. JAMA 2022, 327, 319–320. [Google Scholar] [CrossRef]
- Chassalevris, T.; Chaintoutis, S.C.; Koureas, M.; Petala, M.; Moutou, E.; Beta, C.; Kyritsi, M.; Hadjichristodoulou, C.; Kostoglou, M.; Karapantsios, T.; et al. SARS-CoV-2 wastewater monitoring using a novel PCR-based method rapidly captured the Delta-to-Omicron BA.1 transition patterns in the absence of conventional surveillance evidence. Sci. Total Environ. 2022, 844, 156932. [Google Scholar] [CrossRef] [PubMed]
- Wrenn, J.O.; Pakala, S.B.; Vestal, G.; Shilts, M.H.; Brown, H.M.; Bowen, S.M.; Strickland, B.A.; Williams, T.; Mallal, S.A.; Jones, I.D.; et al. COVID-19 severity from Omicron and Delta SARS-CoV-2 variants. Influ. Other Respir. Viruses 2022, 16, 832–836. [Google Scholar] [CrossRef]
- Lundberg, A.L.; Lorenzo-Redondo, R.; Ozer, E.A.; Hawkins, C.A.; Hultquist, J.F.; Welch, S.B.; Prasad, P.V.; Oehmke, J.F.; Achenbach, C.J.; Murphy, R.L.; et al. Has Omicron Changed the Evolution of the Pandemic? JMIR Public Health Surveill. 2022, 8, e35763. [Google Scholar] [CrossRef]
- Di Fusco, M.; Lin, J.; Vaghela, S.; Lingohr-Smith, M.; Nguyen, J.L.; Sforzolini, T.S.; Judy, J.; Cane, A.; Moran, M.M. COVID-19 vaccine effectiveness among immunocompromised populations: A targeted literature review of real-world studies. Expert Rev. Vaccines 2022, 21, 435–451. [Google Scholar] [CrossRef]
- Lipsitch, M.; Krammer, F.; Regev-Yochay, G.; Lustig, Y.; Balicer, R.D. SARS-CoV-2 breakthrough infections in vaccinated individuals: Measurement, causes and impact. Nat. Rev. Immunol. 2022, 22, 57–65. [Google Scholar] [CrossRef]
- Livanou, E.; Rouka, E.; Sinis, S.; Dimeas, I.; Pantazopoulos, I.; Papagiannis, D.; Malli, F.; Kotsiou, O.; Gourgoulianis, K.I. Predictors of SARS-CoV-2 IgG Spike Antibody Responses on Admission and Clinical Outcomes of COVID-19 Disease in Fully Vaccinated Inpatients: The CoVax Study. J. Pers. Med. 2022, 12, 640. [Google Scholar] [CrossRef] [PubMed]
- Rouka, E.; Livanou, E.; Sinis, S.; Dimeas, I.; Pantazopoulos, I.; Papagiannis, D.; Malli, F.; Kotsiou, O.; Gourgoulianis, K.I. Immune response to the severe acute respiratory syndrome coronavirus 2 vaccines: Is it sustained in the diabetes population? J. Diabetes Investig. 2020, 13, 1461–1462. [Google Scholar] [CrossRef] [PubMed]
- Malli, F.; Lampropoulos, I.C.; Papagiannis, D.; Papathanasiou, I.V.; Daniil, Z.; Gourgoulianis, K.I. Association of SARS-CoV-2 Vaccinations with SARS-CoV-2 Infections, ICU Admissions and Deaths in Greece. Vaccines 2022, 10, 337. [Google Scholar] [CrossRef] [PubMed]
- Lytras, T.; Kontopidou, F.; Lambrou, A.; Tsiodras, S. Comparative effectiveness and durability of COVID-19 vaccination against death and severe disease in an ongoing nationwide mass vaccination campaign. J. Med. Virol. 2022, 94, 5044–5050. [Google Scholar] [CrossRef]
- Jabłońska, K.; Aballéa, S.; Toumi, M. The real-life impact of vaccination on COVID-19 mortality in Europe and Israel. Public Health 2021, 198, 230–237. [Google Scholar] [CrossRef]
- Zuckerman, N.; Nemet, I.; Kliker, L.; Atari, N.; Lustig, Y.; Bucris, E.; Bar Ilan, D.; Geva, M.; Sorek-Abramovich, R.; Weiner, C.; et al. The SARS-CoV-2 Lambda variant and its neutralisation efficiency following vaccination with Comirnaty, Israel, April to June 2021. Eurosurveillance 2021, 26, 2100974. [Google Scholar] [CrossRef]
- Lustig, Y.; Zuckerman, N.; Nemet, I.; Atari, N.; Kliker, L.; Regev-Yochay, G.; Sapir, E.; Mor, O.; Alroy-Preis, S.; Mendelson, E.; et al. Neutralising capacity against Delta (B.1.617.2) and other variants of concern following Comirnaty (BNT162b2, BioNTech/Pfizer) vaccination in health care workers, Israel. Eurosurveillance 2021, 26, 2100557. [Google Scholar] [CrossRef]
- Trabace, L.; Pace, L.; Morgese, M.G.; Santo, I.B.; Galante, D.; Schiavone, S.; Cipolletta, D.; Rosa, A.M.; Reveglia, P.; Parisi, A.; et al. SARS-CoV-2 Gamma and Delta Variants of Concern Might Undermine Neutralizing Activity Generated in Response to BNT162b2 mRNA Vaccination. Viruses 2022, 14, 814. [Google Scholar] [CrossRef]
- Davis, C.; Logan, N.; Tyson, G.; Orton, R.; Harvey, W.T.; Perkins, J.S.; Mollett, G.; Blacow, R.M.; The COVID-19 Genomics UK (COG-UK) Consortium; Peacock, T.P.; et al. Reduced neutralisation of the Delta (B.1.617.2) SARS-CoV-2 variant of concern following vaccination. PLOS Pathog. 2021, 17, e1010022. [Google Scholar] [CrossRef]
- Kodera, S.; Rashed, E.A.; Hirata, A. Estimation of Real-World Vaccination Effectiveness of mRNA COVID-19 Vaccines against Delta and Omicron Variants in Japan. Vaccines 2022, 10, 430. [Google Scholar] [CrossRef]
- Gao, Y.-D.; Ding, M.; Dong, X.; Zhang, J.-J.; Azkur, A.K.; Azkur, D.; Gan, H.; Sun, Y.-L.; Fu, W.; Li, W.; et al. Risk factors for severe and critically ill COVID-19 patients: A review. Allergy 2021, 76, 428–455. [Google Scholar] [CrossRef] [PubMed]
- Sotiriou, S.; Samara, A.A.; Vamvakopoulou, D.; Vamvakopoulos, K.-O.; Sidiropoulos, A.; Vamvakopoulos, N.; Janho, M.B.; Gourgoulianis, K.I.; Boutlas, S. Susceptibility of β-Thalassemia Heterozygotes to COVID-19. J. Clin. Med. 2021, 10, 3645. [Google Scholar] [CrossRef]
- Dessie, Z.G.; Zewotir, T. Mortality-related risk factors of COVID-19: A systematic review and meta-analysis of 42 studies and 423,117 patients. BMC Infect. Dis. 2021, 21, 855. [Google Scholar] [CrossRef] [PubMed]
- Izcovich, A.; Ragusa, M.A.; Tortosa, F.; Marzio, M.A.L.; Agnoletti, C.; Bengolea, A.; Ceirano, A.; Espinosa, F.; Saavedra, E.; Sanguine, V.; et al. Prognostic factors for severity and mortality in patients infected with COVID-19: A systematic review. PLoS ONE 2020, 15, e0241955. [Google Scholar] [CrossRef]
- Vimercati, L.; De Maria, L.; Quarato, M.; Caputi, A.; Gesualdo, L.; Migliore, G.; Cavone, D.; Sponselli, S.; Pipoli, A.; Inchingolo, F.; et al. Association between Long COVID and Overweight/Obesity. J. Clin. Med. 2021, 10, 4143. [Google Scholar] [CrossRef]
- Davis, C.J.; Golding, M.; McKay, R. Efficacy information influences intention to take COVID-19 vaccine. Br. J. Health Psychol. 2022, 27, 300–319. [Google Scholar] [CrossRef]
- Bianchi, F.P.; Tafuri, S.; Migliore, G.; Vimercati, L.; Martinelli, A.; Lobifaro, A.; Diella, G.; Stefanizzi, P.; on behalf of the Control Room Working Group. BNT162b2 mRNA COVID-19 Vaccine Effectiveness in the Prevention of SARS-CoV-2 Infection and Symptomatic Disease in Five-Month Follow-Up: A Retrospective Cohort Study. Vaccines 2021, 9, 1143. [Google Scholar] [CrossRef]
| Mortality | Univariate | Multivariate Binary Logistic Regression | ||||
|---|---|---|---|---|---|---|
| Yes (%) | Significance | OR with 95% CI | RR with 95% CI | Significance | aOR with 95% CI | |
| Male gender | 45 (13.9) | 0.805 (C) | 1.09 (0.67–1.78) | 1.08 (0.71–1.65) | 0.013 | 2.29 (1.19–4.39) |
| Age (median, IQR) | Dead: 83 (13) Alive: 61 (26) | <0.001 (M-W) | – | <0.001 | 1.12 (1.09–1.16) | |
| Smoking * | 2 (3.6) | 0.521 (F) | 0.48 (0.10–2.24) | 0.50 (0.11–2.19) | Insufficient data | |
| Vaccination | 24 (10.9) | 0.169 (C) | 0.70 (0.42–1.17) | 0.73 (0.46–1.15) | <0.001 | 0.20 (0.10–0.39) |
| Atrial fibrillation | 23 (29.9) | <0.001 (C) | 3.47 (1.98–6.10) | 2.73 (1.79–4.18) | 0.500 | 1.27 (0.63–2.56) |
| Chronic respiratory fisease | 14 (26.4) | 0.004 (C) | 2.59 (1.33–5.04) | 2.17 (1.31–3.60) | 0.437 | 1.40 (0.60–3.24) |
| Coronary disease | 27 (30.7) | <0.001 (C) | 3.83 (2.24–6.57) | 2.96 (1.97–4.46) | 0.549 | 1.24 (0.62–2.47) |
| Diabetes type II | 23 (24.2) | 0.001 (C) | 2.50 (1.44–4.32) | 2.13 (1.38–3.30) | 0.218 | 1.56 (0.77–2.18) |
| Malignancy | 7 (14.9) | 0.768 (C) | 1.14 (0.49–2.63) | 1.12 (0.54–2.28) | 0.803 | 1.14 (0.42–3.09) |
| Dyslipidemia | 39 (23.4) | <0.001 (C) | 2.94 (1.80–4.79) | 2.48 (1.65–3.74) | 0.090 | 1.75 (0.92–3.35) |
| Hypertension | 57 (20.2) | <0.001 (C) | 3.41 (1.99–5.84) | 2.92 (1.80–4.73) | 0.692 | 1.16 (0.56–2.38) |
| C: Chi-square test; F: Fisher’s exact test M-W: Mann-Whitney U test | ||||||
| Severe COVID-19 (N = 89) | Univariate | Multivariate Binary Logistic Regression | ||||
|---|---|---|---|---|---|---|
| Significance | OR with 95% CI | RR with 95% CI | Significance | aOR with 95% CI | ||
| Male gender | 51 (15.8) | 0.879 (C) | 1.04 (0.66–1.64) | 1.03 (0.70–1.52) | 0.025 | 1.92 (1.09–3.41) |
| Age (median, IQR) | Moderate: 61 (13) Severe: 83 (26) | <0.001 (M-W) | – | <0.001 | 1.09 (1.06–1.11) | |
| Smoking * | 5 (8.9) | 0.789 (F) | 1.16 (0.39–3.46) | 1.15 (0.42–3.11) | Insufficient data | |
| Vaccination | 27 (12.3) | 0.109 (C) | 0.67 (0.41–1.10) | 071 (0.47–1.09) | <0.001 | 0.25 (0.14–0.45) |
| Atrial fibrillation | 23 (29.9) | <0.001 (C) | 2.76 (1.59–4.80) | 2.24 (1.49–3.37) | 0.712 | 1.13 (0.59–2.19) |
| Chronic respiratory disease | 15 (28.3) 74 (14.3) | 0.007 (C) | 2.37 (1.24–4.52) | 1.98 (1.23–3.19) | 0.406 | 1.39 (0.64–3.04) |
| Coronary disease | 27 (30.7) 62 (12.8) | <0.001 (C) | 3.01 (1.78–5.08) | 2.39 (1.62–3.53) | 0.892 | 1.05 (0.55–2.00) |
| Diabetes type II | 25 (26.3) 64 (13.4) | 0.002 (C) | 2.30 (1.36–3.90) | 1.96 (1.30–2.94) | 0.272 | 1.44 (0.75–2.78) |
| Malignancy | 11 (23.4) 78 (14.9) | 0.123 (C) | 1.75 (0.85–3.58) | 1.57 (0.90–2.74) | 0.142 | 1.85 (0.81–4.23) |
| Dyslipidemia | 44 (26.3) 45 (11.1) | <0.001 (C) | 2.85 (1.80–4.54) | 2.37 (1.63–3.44) | 0.056 | 1.76 (0.99–3.12) |
| Hypertension | 63 (22.3) 26 (9.0) | <0.001 (C) | 2.91 (1.78–4.75) | 2.48 (1.62–3.80) | 0.748 | 1.11 (0.59–2.07) |
| C: Chi-square test; F: Fisher’s exact test M-W: Mann-Whitney U test | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samara, A.A.; Boutlas, S.; Janho, M.B.; Gourgoulianis, K.I.; Sotiriou, S. COVID-19 Severity and Mortality after Vaccination against SARS-CoV-2 in Central Greece. J. Pers. Med. 2022, 12, 1423. https://doi.org/10.3390/jpm12091423
Samara AA, Boutlas S, Janho MB, Gourgoulianis KI, Sotiriou S. COVID-19 Severity and Mortality after Vaccination against SARS-CoV-2 in Central Greece. Journal of Personalized Medicine. 2022; 12(9):1423. https://doi.org/10.3390/jpm12091423
Chicago/Turabian StyleSamara, Athina A., Stylianos Boutlas, Michel B. Janho, Konstantinos I. Gourgoulianis, and Sotirios Sotiriou. 2022. "COVID-19 Severity and Mortality after Vaccination against SARS-CoV-2 in Central Greece" Journal of Personalized Medicine 12, no. 9: 1423. https://doi.org/10.3390/jpm12091423
APA StyleSamara, A. A., Boutlas, S., Janho, M. B., Gourgoulianis, K. I., & Sotiriou, S. (2022). COVID-19 Severity and Mortality after Vaccination against SARS-CoV-2 in Central Greece. Journal of Personalized Medicine, 12(9), 1423. https://doi.org/10.3390/jpm12091423








