Abstract
Two patients with a history of multiple myeloma experienced a recurrence of the disease.18F-FDG PET/CT revealed prominent extramedullary disease as well as multi-foci in the bone marrow, both with increased FDG uptake. However, on 68Ga-Pentixafor PET/CT, all the myeloma lesions showed significantly lower tracer uptake in comparison with 18F-FDG PET. This false-negative result of recurrent multiple myeloma with extramedullary disease may be a potential limitation of 68Ga-Pentixafor in assessing multiple myeloma.
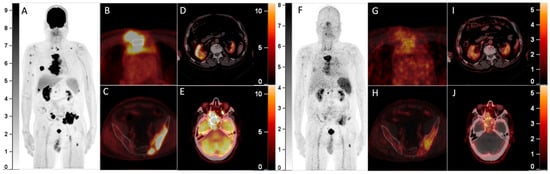
Figure 1.
A 64-year-old man with a history of multiple myeloma (MM) for 8 years, recently presented with a parasternal mass and blindness of the right eye. Serum protein electrophoresis and immunofixation electrophoresis showed positivity for the monoclonal protein (16.2 g/L, IgA-λ). A biopsy of the parasternal mass confirmed plasmacytoma. Considering the recurrence of MM, 18F-FDG PET/CT was referred. The maximum intensity projection (MIP) of the PET (A) detected multi-foci with intense radioactivity all over the body. The axial fusion images (B,C, bone window) showed most of the FDG-avid foci were located in bone marrow, most prominently in the sternum and pelvis (SUVmax 34.0), accompanied by lytic bone destruction and paramedullary masses. Additionally, the axial fusion images (D,E, soft tissue window) demonstrated extramedullary disease (EMD) with intense FDG uptake in the right kidney and paranasal sinus (SUVmax 22.3), which also involved the right orbit and temporal lobe. Since 68Ga-Pentixafor has been reported to be advantageous over 18F-FDG in assessing MM1,2, he was included in the clinical trial of 68Ga-Pentixafor (NCT03436342). In the MIP (F) and corresponding axial fusion images of 68Ga-Pentixafor PET (G–J), the above hypermetabolic foci showed significantly lower tracer uptake as compared with 18F-FDG PET (bone marrow lesions: SUVmax 16.5; EMD: SUVmax 7.4).
Figure 1.
A 64-year-old man with a history of multiple myeloma (MM) for 8 years, recently presented with a parasternal mass and blindness of the right eye. Serum protein electrophoresis and immunofixation electrophoresis showed positivity for the monoclonal protein (16.2 g/L, IgA-λ). A biopsy of the parasternal mass confirmed plasmacytoma. Considering the recurrence of MM, 18F-FDG PET/CT was referred. The maximum intensity projection (MIP) of the PET (A) detected multi-foci with intense radioactivity all over the body. The axial fusion images (B,C, bone window) showed most of the FDG-avid foci were located in bone marrow, most prominently in the sternum and pelvis (SUVmax 34.0), accompanied by lytic bone destruction and paramedullary masses. Additionally, the axial fusion images (D,E, soft tissue window) demonstrated extramedullary disease (EMD) with intense FDG uptake in the right kidney and paranasal sinus (SUVmax 22.3), which also involved the right orbit and temporal lobe. Since 68Ga-Pentixafor has been reported to be advantageous over 18F-FDG in assessing MM1,2, he was included in the clinical trial of 68Ga-Pentixafor (NCT03436342). In the MIP (F) and corresponding axial fusion images of 68Ga-Pentixafor PET (G–J), the above hypermetabolic foci showed significantly lower tracer uptake as compared with 18F-FDG PET (bone marrow lesions: SUVmax 16.5; EMD: SUVmax 7.4).
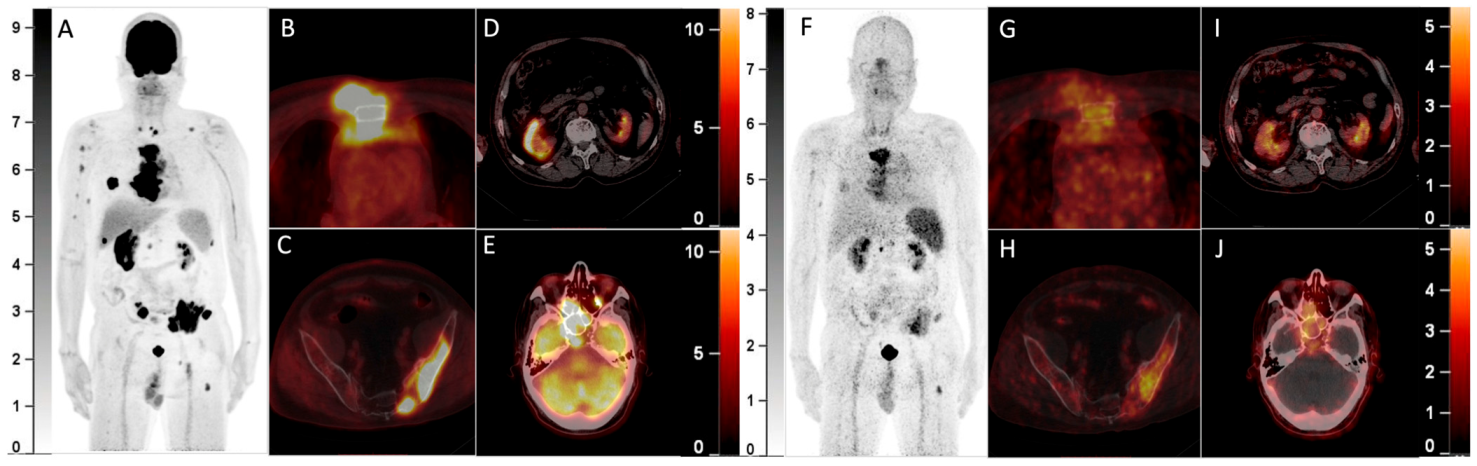
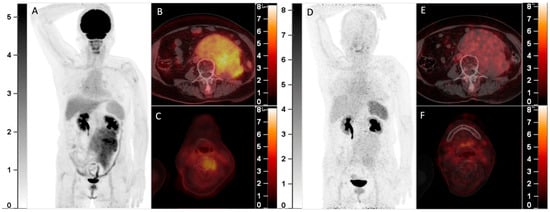
Figure 2.
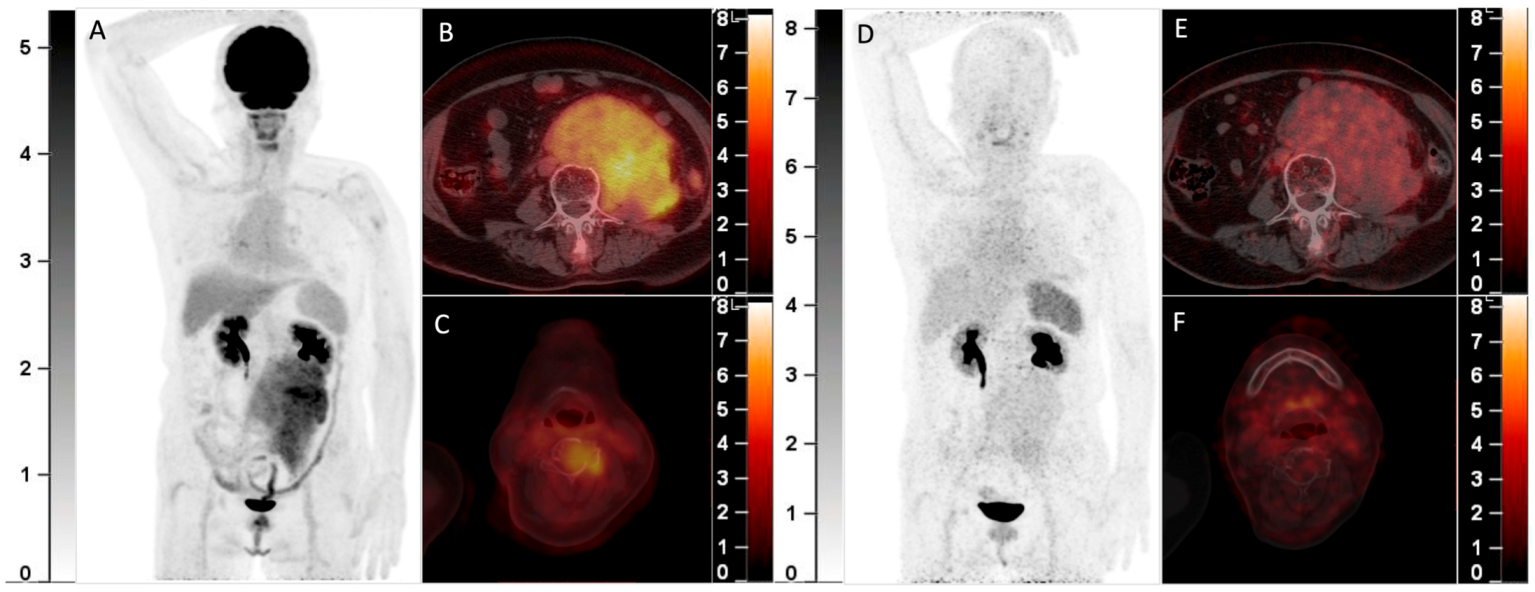
A 70-year-old woman with smoldering MM was found to have a solitary plasmacytoma in the frontal bone that was surgically resected one year ago. Recently, she complained of backache and was found with a retroperitoneal mass. Elevation of the monoclonal protein (19.0 g/L) and the presence of IgA-λ in serum immunofixation electrophoresis and infiltration of plasma cells (17.5%) in bone marrow aspiration confirmed the recurrence of MM. 18F-FDG PET/CT was then performed. The MIP image (A) showed an FDG-avid mass in the abdomen. The axial fusion images (B,C) showed the mass was located in the retroperitoneum with uneven FDG distribution (B, SUVmax 5.7). Furthermore, several bone marrow lesions with increased FDG uptake and lytic bone destruction were found in the occipital bone, C4 vertebra, and right 8th rib (C, SUVmax 4.1). She was also included in the clinical trial and underwent 68Ga-Pentixafor PET/CT (D, MIP image; E,F, axial fusion images). However, the retroperitoneal mass and the bone marrow lesions did not demonstrate increased uptake of 68Ga-Pentixafor. She then received chemotherapy against MM, and the retroperitoneal mass disappeared after 2 cycles of chemotherapy. 68Ga-Pentixafor, a CXCR4-targeted agent, has recently been introduced in MM [1,2,3,4]. Our recent study demonstrated 68Ga-Pentixafor had a significantly higher sensitivity than 18F-FDG in detecting newly diagnosed MM2. However, 68Ga-Pentixafor was inferior to 18F-FDG in the current two cases of recurrent MM with extensive EMD. CXCR4 is overexpressed in myeloma cells and is responsible for plasma cells’ homing to the bone marrow niche [5,6]. Development of EMD in MM is associated with CXCR4/CXCL12 downregulation through cell adhesion disruption [7,8,9]. In line with the current cases, Lapa C. et al.’s study found that some EMDs were exclusively identified by 18F-FDG and were not sensitive to 68Ga-Pentixafor [1]. Thus, the significantly lower uptake than 18F-FDG of recurrent MM with EMD may be a potential limitation of 68Ga-Pentixafor in assessing MM.
Figure 2.
A 70-year-old woman with smoldering MM was found to have a solitary plasmacytoma in the frontal bone that was surgically resected one year ago. Recently, she complained of backache and was found with a retroperitoneal mass. Elevation of the monoclonal protein (19.0 g/L) and the presence of IgA-λ in serum immunofixation electrophoresis and infiltration of plasma cells (17.5%) in bone marrow aspiration confirmed the recurrence of MM. 18F-FDG PET/CT was then performed. The MIP image (A) showed an FDG-avid mass in the abdomen. The axial fusion images (B,C) showed the mass was located in the retroperitoneum with uneven FDG distribution (B, SUVmax 5.7). Furthermore, several bone marrow lesions with increased FDG uptake and lytic bone destruction were found in the occipital bone, C4 vertebra, and right 8th rib (C, SUVmax 4.1). She was also included in the clinical trial and underwent 68Ga-Pentixafor PET/CT (D, MIP image; E,F, axial fusion images). However, the retroperitoneal mass and the bone marrow lesions did not demonstrate increased uptake of 68Ga-Pentixafor. She then received chemotherapy against MM, and the retroperitoneal mass disappeared after 2 cycles of chemotherapy. 68Ga-Pentixafor, a CXCR4-targeted agent, has recently been introduced in MM [1,2,3,4]. Our recent study demonstrated 68Ga-Pentixafor had a significantly higher sensitivity than 18F-FDG in detecting newly diagnosed MM2. However, 68Ga-Pentixafor was inferior to 18F-FDG in the current two cases of recurrent MM with extensive EMD. CXCR4 is overexpressed in myeloma cells and is responsible for plasma cells’ homing to the bone marrow niche [5,6]. Development of EMD in MM is associated with CXCR4/CXCL12 downregulation through cell adhesion disruption [7,8,9]. In line with the current cases, Lapa C. et al.’s study found that some EMDs were exclusively identified by 18F-FDG and were not sensitive to 68Ga-Pentixafor [1]. Thus, the significantly lower uptake than 18F-FDG of recurrent MM with EMD may be a potential limitation of 68Ga-Pentixafor in assessing MM.
Author Contributions
Conceptualization, Y.L.; investigation, Q.P.; writing—original draft preparation, Q.P.; writing—review and editing, Y.L.; visualization, X.C., J.L. and F.L.; supervision, X.C., J.L. and F.L.; funding acquisition, Q.P., Y.L. and X.C. All authors have read and agreed to the published version of the manuscript.
Funding
This work is supported by National High Level Hospital Clinical Research Funding (2022-PUMCH-A-127, 2022-PUMCH-B-070, 2022-PUMCH-B-071), the CAMS Initiative for Innovative Medicine (CAMS-I2M, 2017-I2M-3-001), and the Beijing Natural Science Foundation (7202160).
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the institutional review board of Peking Union Medical College Hospital (IRB protocol #ZS-1113).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study. Written informed consent has been obtained from the patients to publish this paper.
Data Availability Statement
No new data were created or analyzed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lapa, C.; Schreder, M.; Schirbel, A.; Samnick, S.; Kortüm, K.M.; Herrmann, K.; Kropf, S.; Einsele, H.; Buck, A.K.; Wester, H.J.; et al. [(68)Ga]Pentixafor-PET/CT for imaging of chemokine receptor CXCR4 expression in multiple myeloma-Comparison to [(18)F]FDG and laboratory values. Theranostics 2017, 7, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Cao, X.; Luo, Y.; Li, J.; Feng, J.; Li, F. Chemokine receptor-4 targeted PET/CT with (68)Ga-Pentixafor in assessment of newly diagnosed multiple myeloma: Comparison to (18)F-FDG PET/CT. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Philipp-Abbrederis, K.; Herrmann, K.; Knop, S.; Schottelius, M.; Eiber, M.; Lückerath, K.; Pietschmann, E.; Habringer, S.; Gerngroß, C.; Franke, K.; et al. In vivo molecular imaging of chemokine receptor CXCR4 expression in patients with advanced multiple myeloma. EMBO Mol. Med. 2015, 7, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Pan, Q.; Luo, Y.; Cao, X.; Ma, Y.; Li, F. Multiple myeloma presenting as a superscan on 68Ga-Pentixafor PET/CT. Clin. Nucl. Med. 2018, 43, 462–463. [Google Scholar] [CrossRef] [PubMed]
- Alsayed, Y.; Ngo, H.; Runnels, J.; Leleu, X.; Singha, U.K.; Pitsillides, C.M.; Spencer, J.A.; Kimlinger, T.; Ghobrial, J.M.; Jia, X.; et al. Mechanisms of regulation of CXCR4/SDF-1 (CXCL12)-dependent migration and homing in multiple myeloma. Blood 2007, 109, 2708–2717. [Google Scholar] [CrossRef] [PubMed]
- Roccaro, A.M.; Mishima, Y.; Sacco, A.; Moschetta, M.; Tai, Y.T.; Shi, J.; Zhang, Y.; Reagan, M.R.; Huynh, D.; Kawano, Y.; et al. CXCR4 regulates extra-medullary myeloma through epithelial-mesenchymal-transition-like transcriptional activation. Cell Rep. 2015, 12, 622–635. [Google Scholar] [CrossRef] [PubMed]
- Stessman, H.A.F.; Mansoor, A.; Zhan, F.; Janz, S.; Linden, M.A.; Baughn, L.B.; Van Ness, B. Reduced CXCR4 expression is associated with extramedullary disease in a mouse model of myeloma and predicts poor survival in multiple myeloma patients treated with bortezomib. Leukemia 2013, 27, 2075–2077. [Google Scholar] [CrossRef] [PubMed]
- Ghobrial, I.M. Myeloma as a model for the process of metastasis: Implications for therapy. Blood 2012, 120, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Bladé, J.; Fernández de Larrea, C.; Rosiñol, L. Extramedullary disease in multiple myeloma in the era of novel agents. Br. J. Haematol. 2015, 169, 763–765. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
