Chlamydia trachomatis as a Current Health Problem: Challenges and Opportunities
Abstract
1. Introduction
2. Chlamydia trachomatis Epidemiology and Screening Programs
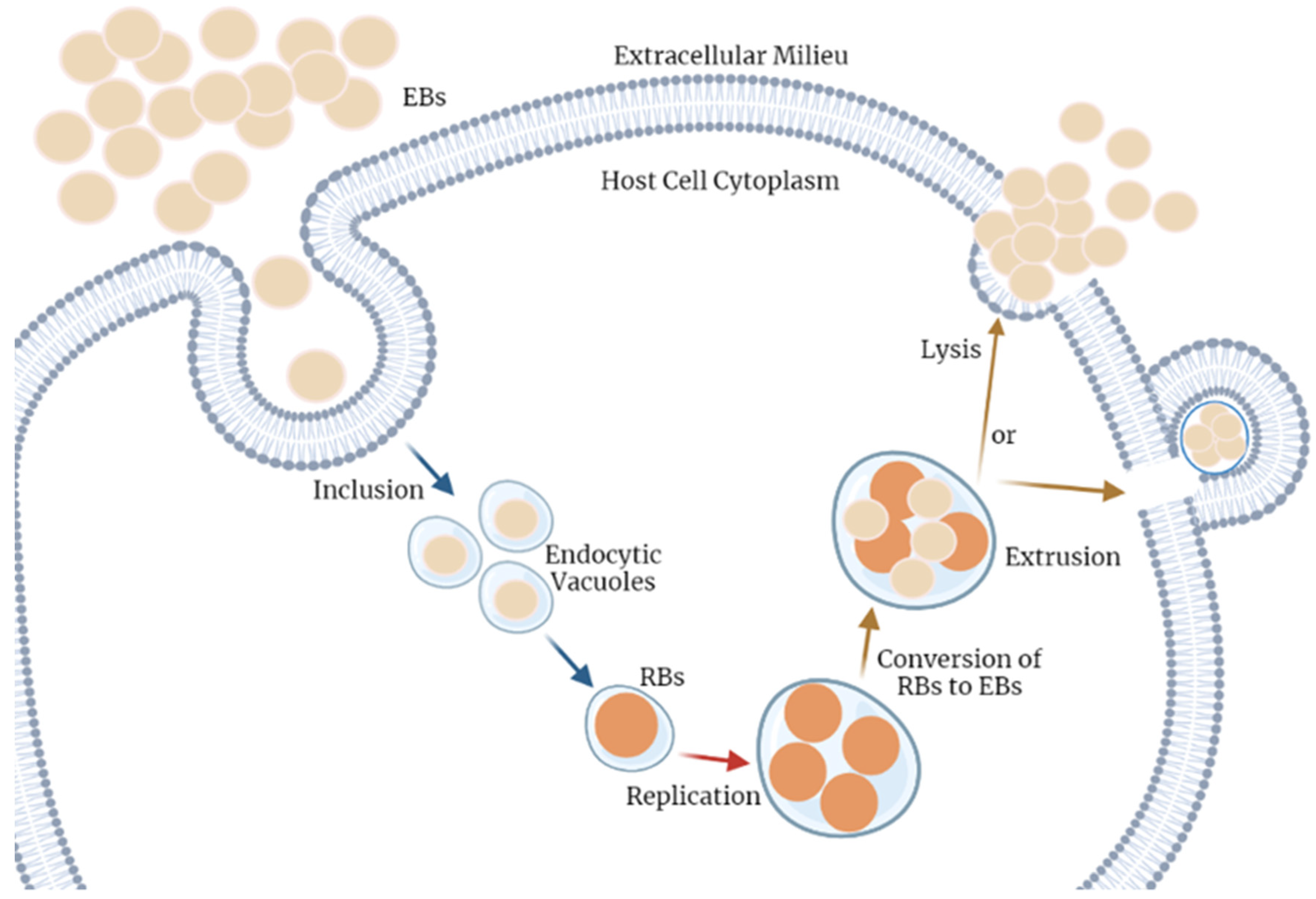
3. Chlamydia trachomatis Development and Immune Response
4. Chlamydia Diagnostic Methods
5. Current Therapeutic Options and Challenges
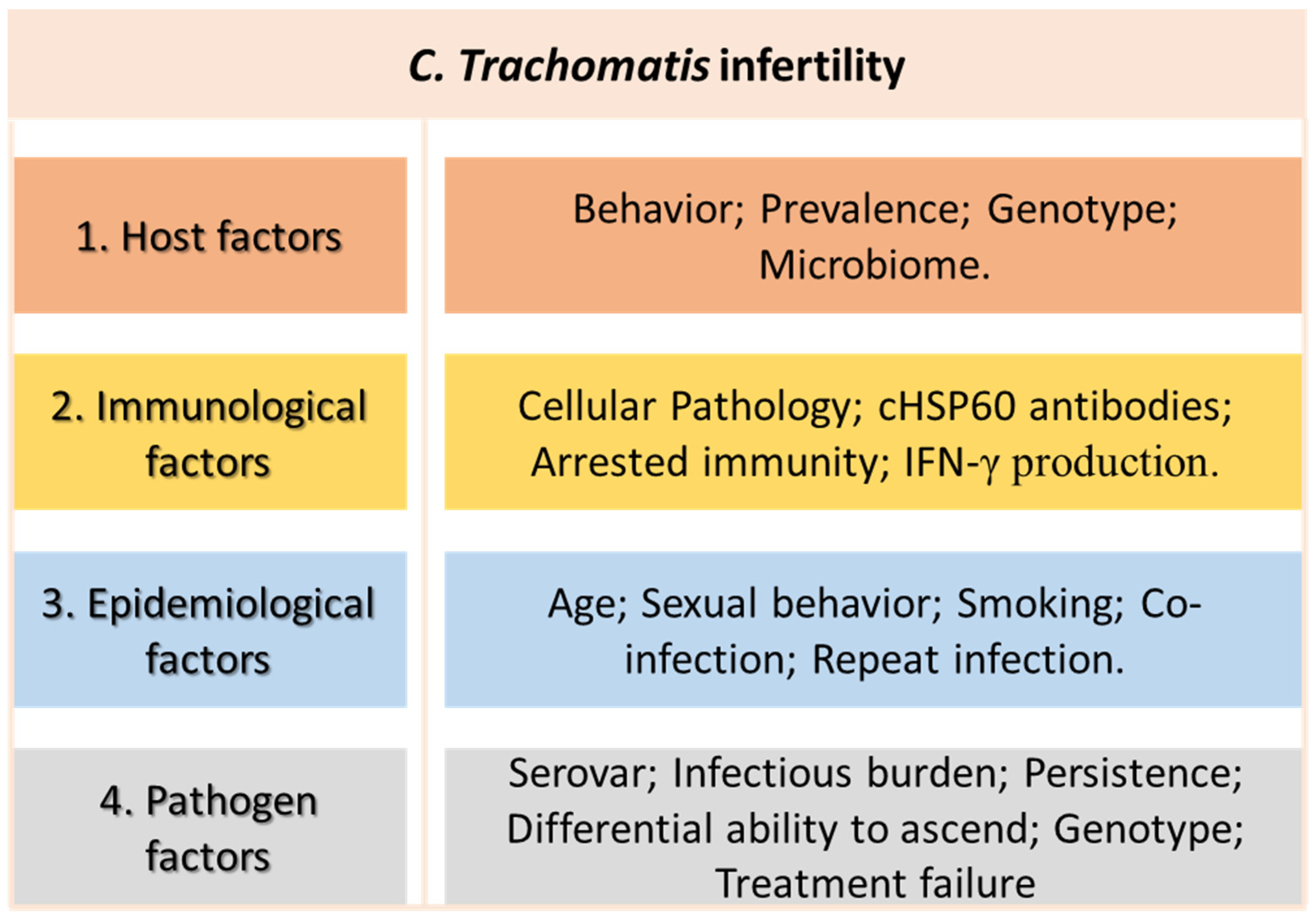
6. Chlamydia trachomatis as a Risk Factor for Infertility
7. Chlamydia and Tumorigenesis Association
8. Discussion of the Future Perspectives concerning Chlamydia Screening Programs
9. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Mohseni, M.; Sung, S.; Takov, V. Chlamydia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Lesiak-Markowicz, I.; Schötta, A.-M.; Stockinger, H.; Stanek, G.; Markowicz, M. Chlamydia trachomatis serovars in urogenital and ocular samples collected 2014–2017 from Austrian patients. Sci. Rep. 2019, 9, 18327. [Google Scholar] [CrossRef] [PubMed]
- Morré, S.A.; Rozendaal, L.; van Valkengoed, I.G.; Boeke, A.J.; van Voorst Vader, P.C.; Schirm, J.; de Blok, S.; van Den Hoek, J.A.; van Doornum, G.J.; Meijer, C.J.; et al. Urogenital Chlamydia trachomatis serovars in men and women with a symptomatic or asymptomatic infection: An association with clinical manifestations? J. Clin. Microbiol. 2000, 38, 2292–2296. [Google Scholar] [CrossRef] [PubMed]
- Land, J.A.; Van Bergen, J.E.A.M.; Morré, S.A.; Postma, M.J. Epidemiology of Chlamydia trachomatis infection in women and the cost-effectiveness of screening. Hum. Reprod. Update 2009, 16, 189–204. [Google Scholar] [CrossRef] [PubMed]
- Witkin, S.S.; Minis, E.; Athanasiou, A.; Leizer, J.; Linhares, I.M. Chlamydia trachomatis: The Persistent Pathogen. Clin. Vaccine Immunol. 2017, 24, e00203-17. [Google Scholar] [CrossRef] [PubMed]
- Schautteet, K.; De Clercq, E.; Vanrompay, D. Chlamydia trachomatis Vaccine Research through the Years. Infect. Dis. Obstet. Gynecol. 2011, 2011, 963513. [Google Scholar] [CrossRef]
- Detels, R.; Green, A.M.; Klausner, J.D.; Katzenstein, D.; Gaydos, C.; Handsfield, H.H.; Pequegnat, W.; Mayer, K.; Hartwell, T.D.; Quinn, T.C. The Incidence and Correlates of Symptomatic and Asymptomatic Chlamydia trachomatis and Neisseria gonorrhoeae Infections in Selected Populations in Five Countries. Sex. Transm. Dis. 2011, 38, 503–509. [Google Scholar] [CrossRef]
- Rantsi, T.; Öhman, H.; Puolakkainen, M.; Bloigu, A.; Paavonen, J.; Surcel, H.M.; Tiitinen, A.; Joki-Korpela, P. Predicting tubal factor infertility by using markers of humoral and cell-mediated immune response against Chlamydia trachomatis. Am. J. Reprod. Immunol. 2018, 80, e13051. [Google Scholar] [CrossRef]
- Tsevat, D.G.; Wiesenfeld, H.C.; Parks, C.; Peipert, J.F. Sexually transmitted diseases and infertility. Am. J. Obstet. Gynecol. 2017, 216, 1–9. [Google Scholar] [CrossRef]
- Peipert, J.F. Clinical practice. Genital chlamydial infections. N. Engl. J. Med. 2003, 349, 2424–2430. [Google Scholar] [CrossRef]
- Thompson, S.E.; Washington, A.E. Epidemiology of sexually transmitted Chlamydia trachomatis infections. Epidemiol. Rev. 1983, 5, 96–123. [Google Scholar] [CrossRef]
- Bellaminutti, S.; Seraceni, S.; De Seta, F.; Gheit, T.; Tommasino, M.; Comar, M. HPV and Chlamydia trachomatis co-detection in young asymptomatic women from high incidence area for cervical cancer. J. Med. Virol. 2014, 86, 1920–1925. [Google Scholar] [CrossRef] [PubMed]
- Escarcega-Tame, M.A.; López-Hurtado, M.; Escobedo-Guerra, M.R.; Reyes-Maldonado, E.; Castro-Escarpulli, G.; Guerra-Infante, F.M. Co-infection between genotypes of the human papillomavirus and Chlamydia trachomatis in Mexican women. Int. J. STD AIDS 2020, 31, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.A.; Olson, K.M.; Ratliff, A.E.; Xiao, L.; Van Der Pol, B.; Waites, K.B.; Geisler, W.M. Mycoplasma genitalium Coinfection in Women with Chlamydia trachomatis Infection. Sex. Transm. Dis. 2019, 46, e101–e104. [Google Scholar] [CrossRef] [PubMed]
- Borgogna, J.-L.C.; Shardell, M.D.; Yeoman, C.J.; Ghanem, K.G.; Kadriu, H.; Ulanov, A.V.; Gaydos, C.A.; Hardick, J.; Robinson, C.K.; Bavoil, P.M.; et al. The association of Chlamydia trachomatis and Mycoplasma genitalium infection with the vaginal metabolome. Sci. Rep. 2020, 10, 3420. [Google Scholar] [CrossRef] [PubMed]
- Lim, R.B.T.; Wong, M.L.; Cook, A.R.; Brun, C.; Chan, R.K.W.; Sen, P.; Chio, M. Determinants of Chlamydia, Gonorrhea, and Coinfection in Heterosexual Adolescents Attending the National Public Sexually Transmitted Infection Clinic in Singapore. Sex. Transm. Dis. 2015, 42, 450–456. [Google Scholar] [CrossRef]
- Seo, Y.; Choi, K.H.; Lee, G. Characterization and Trend of Co-Infection with Neisseria gonorrhoeae and Chlamydia trachomatis from the Korean National Infectious Diseases Surveillance Database. World J. Mens Health 2021, 39, 107–115. [Google Scholar] [CrossRef]
- Lima, L.M.; Hoelzle, C.R.; Simões, R.T.; Lima, M.I.M.; Fradico, J.R.B.; Mateo, E.C.C.; Zauli, D.A.G.; Melo, V.H. Sexually Transmitted Infections Detected by Multiplex Real Time PCR in Asymptomatic Women and Association with Cervical Intraepithelial Neoplasia. Rev. Bras. Ginecol. Obs. 2018, 40, 540–546. [Google Scholar] [CrossRef]
- Paavonen, J.; Eggert-Kruse, W. Chlamydia trachomatis: Impact on human reproduction. Hum. Reprod. Update 1999, 5, 433–447. [Google Scholar] [CrossRef]
- Beagley, K.W.; Timms, P. Chlamydia trachomatis infection: Incidence, health costs and prospects for vaccine development. J. Reprod. Immunol. 2000, 48, 47–68. [Google Scholar] [CrossRef]
- Páez-Canro, C.; Alzate, J.P.; González, L.M.; Rubio-Romero, J.A.; Lethaby, A.; Gaitán, H.G. Antibiotics for treating urogenital Chlamydia trachomatis infection in men and non-pregnant women. Cochrane Database Syst. Rev. 2019, 1, Cd010871. [Google Scholar] [CrossRef]
- Shao, L.; Guo, Y.; Jiang, Y.; Liu, Y.; Wang, M.; You, C.; Liu, Q. Sensitivity of the Standard Chlamydia trachomatis Culture Method Is Improved After One Additional In Vitro Passage. J. Clin. Lab. Anal. 2016, 30, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Stamm, W.E. Diagnosis of Neisseria gonorrhoeae and Chlamydia trachomatis infections using antigen detection methods. Diagn. Microbiol. Infect. Dis. 1986, 4 (Suppl. S3), 93s–99s. [Google Scholar] [CrossRef]
- Novak, D.P.; Lindholm, L.; Jonsson, M.; Karlsson, R.B. A Swedish cost-effectiveness analysis of community-based Chlamydia trachomatis PCR testing of postal urine specimens obtained at home. Scand. J. Public Health 2004, 32, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Sylvan, S.P.; Von Krogh, G.; Tiveljung, A.; Siwerth, B.M.; Henriksson, L.; Norén, L.; Asp, A.K.; Grillner, L. Screening and genotyping of genital Chlamydia trachomatis in urine specimens from male and female clients of youth-health centers in Stockholm County. Sex Transm. Dis. 2002, 29, 379–386. [Google Scholar] [CrossRef] [PubMed]
- de Vries, H.J.; Schim van der Loeff, M.F.; Bruisten, S.M. High-resolution typing of Chlamydia trachomatis: Epidemiological and clinical uses. Curr. Opin. Infect. Dis. 2015, 28, 61–71. [Google Scholar] [CrossRef]
- Batteiger, B.E.; Tu, W.; Ofner, S.; Van Der Pol, B.; Stothard, D.R.; Orr, D.P.; Katz, B.P.; Fortenberry, J.D. Repeated Chlamydia trachomatis genital infections in adolescent women. J. Infect. Dis. 2010, 201, 42–51. [Google Scholar] [CrossRef]
- Schillinger, J.A.; Katz, B.P.; Markowitz, L.E.; Braslins, P.G.; Shrier, L.A.; Madico, G.; Van Der Pol, B.; Orr, D.P.; Rice, P.A.; Batteiger, B.E. Genotype-Specific Concordance of Chlamydia trachomatis Genital Infection Within Heterosexual Partnerships. Sex. Transm. Dis. 2016, 43, 741–749. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ditkowsky, J.; Shah, K.H.; Hammerschlag, M.R.; Kohlhoff, S.; Smith-Norowitz, T.A. Cost-benefit analysis of Chlamydia trachomatis screening in pregnant women in a high burden setting in the United States. BMC Infect. Dis. 2017, 17, 155. [Google Scholar] [CrossRef]
- Chlamydia trachomatis genital infections—United States, 1995. MMWR Morb. Mortal Wkly Rep. 1997, 46, 193–198.
- Kumar, S.; Chesson, H.; Gift, T.L. Estimating the Direct Medical Costs and Productivity Loss of Outpatient Chlamydia and Gonorrhea Treatment. Sex. Transm. Dis. 2021, 48, e18–e21. [Google Scholar] [CrossRef]
- Washington, A.E.; Katz, P. Cost of and Payment Source for Pelvic Inflammatory Disease: Trends and Projections, 1983 Through 2000. JAMA 1991, 266, 2565–2569. [Google Scholar] [CrossRef]
- Greydanus, D.E.; Cabral, M.D.; Patel, D.R. Pelvic inflammatory disease in the adolescent and young adult: An update. Dis.-A-Mon. 2022, 68, 101287. [Google Scholar] [CrossRef]
- Sheringham, J.; Baraitser, P.; Simms, I.; Hart, G.; Raine, R. Chlamydia screening in England: A qualitative study of the narrative behind the policy. BMC Public Health 2012, 12, 317. [Google Scholar] [CrossRef]
- Low, N.; Hocking, J.S.; van Bergen, J. The changing landscape of chlamydia control strategies. Lancet 2021, 398, 1386–1388. [Google Scholar] [CrossRef]
- Kretzschmar, M.; Welte, R.; van den Hoek, A.; Postma, M.J. Comparative Model-based Analysis of Screening Programs for Chlamydia trachomatis Infections. Am. J. Epidemiol. 2001, 153, 90–101. [Google Scholar] [CrossRef]
- van Aar, F.; de Moraes, M.; Morré, S.A.; van Bergen, J.E.; van der Klis, F.R.; Land, J.A.; van der Sande, M.A.; van den Broek, I.V. Chlamydia trachomatis IgG seroprevalence in the general population of the Netherlands in 1996 and in 2007: Differential changes by gender and age. Sex Transm. Infect. 2014, 90, 434–440. [Google Scholar] [CrossRef]
- Chernesky, M.; Jang, D.; Martin, I.; Arias, M.; Shah, A.; Smieja, M.; Ratnam, S.; Getman, D.; Schachter, J. Mycoplasma genitalium, Chlamydia trachomatis, and Neisseria gonorrhoeae detected with aptima assays performed on self-obtained vaginal swabs and urine collected at home and in a clinic. Sex. Transm. Dis. 2019, 46, e87–e89. [Google Scholar] [CrossRef]
- Graseck, A.S.; Shih, S.L.; Peipert, J.F. Home versus clinic-based specimen collection for Chlamydia trachomatis and Neisseria gonorrhoeae. Expert. Rev. Anti. Infect. 2011, 9, 183–194. [Google Scholar] [CrossRef]
- Hoenderboom, B.M.; van Ess, E.F.; van den Broek, I.V.F.; van Loo, I.H.M.; Hoebe, C.; Ouburg, S.; Morré, S.A. Chlamydia trachomatis antibody detection in home-collected blood samples for use in epidemiological studies. J. Microbiol. Methods 2018, 144, 164–167. [Google Scholar] [CrossRef]
- LaMontagne, D.S.; Fenton, K.A.; Randall, S.; Anderson, S.; Carter, P. Establishing the National Chlamydia Screening Programme in England: Results from the first full year of screening. Sex. Transm. Infect. 2004, 80, 335–341. [Google Scholar] [CrossRef]
- Yuguero, O.; Fernández-Armenteros, J.M.; Vilela, Á.; Aramburu, J.; Laín, R.; Godoy, P. Preliminary Results of a Screening Programme for Chlamydia in an Asymptomatic Young Population in Spain. Front. Public Health 2021, 9, 615110. [Google Scholar] [CrossRef]
- Low, N.; Cassell, J.A.; Spencer, B.; Bender, N.; Martin Hilber, A.; van Bergen, J.; Andersen, B.; Herrmann, B.; Dubois-Arber, F.; Hamers, F.F.; et al. Chlamydia control activities in Europe: Cross-sectional survey. Eur. J. Public Health 2011, 22, 556–561. [Google Scholar] [CrossRef]
- Cha, S.; Newman, D.R.; Rahman, M.; Peterman, T.A. High Rates of Repeat Chlamydial Infections Among Young Women-Louisiana, 2000–2015. Sex. Transm. Dis. 2019, 46, 52–57. [Google Scholar] [CrossRef]
- Paul, K.J.; Garcia, P.J.; Giesel, A.E.; Holmes, K.K.; Hitti, J.E. Generation C: Prevalence of and risk factors for Chlamydia Trachomatis among adolescents and young women in Lima, Peru. J. Womens Health 2009, 18, 1419–1424. [Google Scholar] [CrossRef]
- Simons, J.L.; McKenzie, J.S.; Wright, N.C.; Sheikh, S.A.; Subramaniam, A.; Tita, A.T.N.; Dionne-Odom, J. Chlamydia Prevalence by Age and Correlates of Infection Among Pregnant Women. Sex. Transm. Dis. 2021, 48, 37–41. [Google Scholar] [CrossRef]
- Pavlin, N.L.; Gunn, J.M.; Parker, R.; Fairley, C.K.; Hocking, J. Implementing chlamydia screening: What do women think? A systematic review of the literature. BMC Public Health 2006, 6, 221. [Google Scholar] [CrossRef]
- Söderqvist, J.; Gullsby, K.; Stark, L.; Wikman, M.; Karlsson, R.; Herrmann, B. Internet-based self-sampling for Chlamydia trachomatis testing: A national evaluation in Sweden. Sex. Transm. Infect. 2020, 96, 160–165. [Google Scholar] [CrossRef]
- Goulet, V.; de Barbeyrac, B.; Raherison, S.; Prudhomme, M.; Semaille, C.; Warszawski, J. Prevalence of Chlamydia trachomatis: Results from the first national population-based survey in France. Sex. Transm. Infect. 2010, 86, 263–270. [Google Scholar] [CrossRef]
- Huai, P.; Li, F.; Chu, T.; Liu, D.; Liu, J.; Zhang, F. Prevalence of genital Chlamydia trachomatis infection in the general population: A meta-analysis. BMC Infect. Dis. 2020, 20, 589. [Google Scholar] [CrossRef]
- Honey, E.; Augood, C.; Templeton, A.; Russell, I.; Paavonen, J.; Mårdh, P.A.; Stary, A.; Stray-Pedersen, B. Cost effectiveness of screening for Chlamydia trachomatis: A review of published studies. Sex. Transm. Infect. 2002, 78, 406–412. [Google Scholar] [CrossRef]
- Rowley, J.; Vander Hoorn, S.; Korenromp, E.; Low, N.; Unemo, M.; Abu-Raddad, L.J.; Chico, R.M.; Smolak, A.; Newman, L.; Gottlieb, S.; et al. Chlamydia, gonorrhoea, trichomoniasis and syphilis: Global prevalence and incidence estimates, 2016. Bull. World Health Organ. 2019, 97, 548–562. [Google Scholar] [CrossRef] [PubMed]
- Murray, S.M.; McKay, P.F. Chlamydia trachomatis: Cell biology, immunology and vaccination. Vaccine 2021, 39, 2965–2975. [Google Scholar] [CrossRef] [PubMed]
- Poston, T.B.; Darville, T. Chlamydia trachomatis: Protective Adaptive Responses and Prospects for a Vaccine. Curr. Top. Microbiol. Immunol. 2018, 412, 217–237. [Google Scholar] [PubMed]
- Gottlieb, S.L.; Brunham, R.C.; Byrne, G.I.; Martin, D.H.; Xu, F.; Berman, S.M. Introduction: The natural history and immunobiology of Chlamydia trachomatis genital infection and implications for chlamydia control. J. Infect. Dis. 2010, 201 (Suppl. 2), S85–S87. [Google Scholar] [CrossRef]
- Fields, K.A.; Hackstadt, T. The chlamydial inclusion: Escape from the endocytic pathway. Annu. Rev. Cell Dev. Biol. 2002, 18, 221–245. [Google Scholar] [CrossRef]
- Hogan, R.J.; Mathews, S.A.; Mukhopadhyay, S.; Summersgill, J.T.; Timms, P. Chlamydial Persistence: Beyond the Biphasic Paradigm. Infect. Immun. 2004, 72, 1843–1855. [Google Scholar] [CrossRef]
- Bastidas, R.J.; Elwell, C.A.; Engel, J.N.; Valdivia, R.H. Chlamydial intracellular survival strategies. Cold Spring Harb. Perspect. Med. 2013, 3, a010256. [Google Scholar] [CrossRef]
- Shiragannavar, S.; Madagi, S.; Hosakeri, J.; Barot, V. In silico vaccine design against Chlamydia trachomatis infection. Netw. Model Anal. Health Inf. Bioinform. 2020, 9, 39. [Google Scholar] [CrossRef]
- Aslam, S.; Ahmad, S.; Noor, F.; Ashfaq, U.A.; Shahid, F.; Rehman, A.; Tahir Ul Qamar, M.; Alatawi, E.A.; Alshabrmi, F.M.; Allemailem, K.S. Designing a Multi-Epitope Vaccine against Chlamydia trachomatis by Employing Integrated Core Proteomics, Immuno-Informatics and In Silico Approaches. Biology 2021, 10, 997. [Google Scholar] [CrossRef]
- de la Maza, L.M.; Darville, T.L.; Pal, S. Chlamydia trachomatis vaccines for genital infections: Where are we and how far is there to go? Rev. Vaccines 2021, 20, 421–435. [Google Scholar] [CrossRef]
- Budai, I. Chlamydia trachomatis: Milestones in clinical and microbiological diagnostics in the last hundred years: A review. Acta Microbiol. Immunol. Hung 2007, 54, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Tosic-Pajic, J.; Sazdanovic, P.; Sorak, M.; Cukic, J.; Arsovic, A.; Milovanovic, D.; Baskic, D. Chlamydia trachomatis screening in resource-limited countries—Comparison of diagnostic accuracy of 3 different assays. J. Infect. Dev. Ctries. 2018, 12, 733–740. [Google Scholar] [CrossRef] [PubMed]
- Abdella, R.M.; Abdelmoaty, H.I.; Elsherif, R.H.; Sayed, A.M.; Sherif, N.A.; Gouda, H.M.; El Lithy, A.; Almohamady, M.; Abdelbar, M.; Hosni, A.N.; et al. Screening for Chlamydia trachomatis in Egyptian women with unexplained infertility, comparing real-time PCR techniques to standard serology tests: Case control study. BMC Womens Health 2015, 15, 45. [Google Scholar] [CrossRef] [PubMed]
- de Haro-Cruz, M.J.; Guadarrama-Macedo, S.I.; López-Hurtado, M.; Escobedo-Guerra, M.R.; Guerra-Infante, F.M. Obtaining an ELISA test based on a recombinant protein of Chlamydia trachomatis. Int. Microbiol. 2019, 22, 471–478. [Google Scholar] [CrossRef] [PubMed]
- Rabenau, H.F.; Köhler, E.; Peters, M.; Doerr, H.W.; Weber, B. Low correlation of serology with detection of Chlamydia trachomatis by ligase chain reaction and antigen EIA. Infection 2000, 28, 97–102. [Google Scholar] [CrossRef]
- Bianchi, S.; Frati, E.R.; Canuti, M.; Colzani, D.; Fasoli, E.; Amendola, A.; Tanzi, E. Molecular epidemiology and genotyping of Chlamydia trachomatis infection in a cohort of young asymptomatic sexually active women (18–25 years) in Milan, Italy. J. Prev. Med. Hyg. 2016, 57, E128–E134. [Google Scholar]
- Jaton, K.; Bille, J.; Greub, G. A novel real-time PCR to detect Chlamydia trachomatis in first-void urine or genital swabs. J. Med. Microbiol. 2006, 55, 1667–1674. [Google Scholar] [CrossRef]
- Rockett, R.; Goire, N.; Limnios, A.; Turra, M.; Higgens, G.; Lambert, S.B.; Bletchly, C.; Nissen, M.D.; Sloots, T.P.; Whiley, D.M. Evaluation of the cobas 4800 CT/NG test for detecting Chlamydia trachomatis and Neisseria gonorrhoeae. Sex. Transm. Infect. 2010, 86, 470. [Google Scholar] [CrossRef]
- Parra, M.; Palomares, J.C.; Bernal, S.; Sivianes, N.; Pérez, L.; Pueyo, I.; Almeida, C.; Martín-Mazuelos, E. Evaluation of the cobas 4800 CT/NG test for detecting Chlamydia trachomatis. Enferm. Infecc. Microbiol. Clin. 2011, 29, 421–424. [Google Scholar] [CrossRef]
- Parra-Sánchez, M.; Palomares, J.C.; Bernal, S.; González, M.T.; Sivianes, N.; Pérez, L.; Pueyo, I.; Martín-Mazuelos, E. Evaluation of the cobas 4800 CT/NG Test for detecting Chlamydia trachomatis and Neisseria gonorrhoeae DNA in urogenital swabs and urine specimens. Diagn. Microbiol. Infect. Dis. 2012, 74, 338–342. [Google Scholar] [CrossRef]
- Papp, J.R.; Schachter, J.; Gaydos, C.A.; Van Der Pol, B. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae—2014. MMWR Recomm. Rep. 2014, 63, 1–19. [Google Scholar]
- Jalal, H.; Stephen, H.; Al-Suwaine, A.; Sonnex, C.; Carne, C. The superiority of polymerase chain reaction over an amplified enzyme immunoassay for the detection of genital chlamydial infections. Sex. Transm. Infect. 2006, 82, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Köksal, M.O.; Beka, H.; Demirci, M.; Kadioglu, A.; Agacfidan, A.; Akgül, B. Prevalence and genotyping of Chlamydia trachomatis in symptomatic male patients from Istanbul, Turkey. Springerplus 2016, 5, 1706. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef]
- Badman, S.G.; Bell, S.F.; Dean, J.A.; Lemoire, J.; Coffey, L.; Debattista, J.; Redmond, A.M.; Williams, O.D.; Gilks, C.F.; Whiley, D.M. Reduced sensitivity from pooled urine, pharyngeal and rectal specimens when using a molecular assay for the detection of chlamydia and gonorrhoea near the point of care. Sex. Health 2020, 17, 15–21. [Google Scholar] [CrossRef]
- Brook, G. The performance of non-NAAT point-of-care (POC) tests and rapid NAAT tests for chlamydia and gonorrhoea infections. An assessment of currently available assays. Sex. Transm. Infect. 2015, 91, 539–544. [Google Scholar]
- Gaydos, C.A. Review of use of a new rapid real-time PCR, the Cepheid GeneXpert® (Xpert) CT/NG assay, for Chlamydia trachomatis and Neisseria gonorrhoeae: Results for patients while in a clinical setting. Expert. Rev. Mol. Diagn. 2014, 14, 135–137. [Google Scholar] [CrossRef]
- Adamson, P.C.; Loeffelholz, M.J.; Klausner, J.D. Point-of-Care Testing for Sexually Transmitted Infections: A Review of Recent Developments. Arch. Pathol. Lab. Med. 2020, 144, 1344–1351. [Google Scholar] [CrossRef]
- Harding-Esch, E.M.; Fuller, S.S.; Chow, S.C.; Nori, A.V.; Harrison, M.A.; Parker, M.; Piepenburg, O.; Forrest, M.S.; Brooks, D.G.; Patel, R.; et al. Diagnostic accuracy of a prototype rapid chlamydia and gonorrhoea recombinase polymerase amplification assay: A multicentre cross-sectional preclinical evaluation. Clin. Microbiol. Infect. 2019, 25, 380.e1–380.e7. [Google Scholar] [CrossRef]
- Potroz, M.G.; Cho, N.J. Natural products for the treatment of trachoma and Chlamydia trachomatis. Molecules 2015, 20, 4180–4203. [Google Scholar] [CrossRef]
- Mestrovic, T.; Ljubin-Sternak, S. Molecular mechanisms of Chlamydia trachomatis resistance to antimicrobial drugs. Front. Biosci. 2018, 23, 656–670. [Google Scholar] [CrossRef] [PubMed]
- Kong, F.Y.; Hocking, J.S. Treatment challenges for urogenital and anorectal Chlamydia trachomatis. BMC Infect. Dis. 2015, 15, 293. [Google Scholar] [CrossRef] [PubMed]
- Shima, K.; Kaufhold, I.; Eder, T.; Käding, N.; Schmidt, N.; Ogunsulire, I.M.; Deenen, R.; Köhrer, K.; Friedrich, D.; Isay, S.E.; et al. Regulation of the Mitochondrion-Fatty Acid Axis for the Metabolic Reprogramming of Chlamydia trachomatis during Treatment with β-Lactam Antimicrobials. mBio 2021, 12, e00023-21. [Google Scholar] [CrossRef] [PubMed]
- Shima, K.; Kaeding, N.; Ogunsulire, I.M.; Kaufhold, I.; Klinger, M.; Rupp, J. Interferon-γ interferes with host cell metabolism during intracellular Chlamydia trachomatis infection. Cytokine 2018, 112, 95–101. [Google Scholar] [CrossRef]
- Belland, R.J.; Nelson, D.E.; Virok, D.; Crane, D.D.; Hogan, D.; Sturdevant, D.; Beatty, W.L.; Caldwell, H.D. Transcriptome analysis of chlamydial growth during IFN-γ;-mediated persistence and reactivation. Proc. Natl. Acad. Sci. USA 2003, 100, 15971–15976. [Google Scholar] [CrossRef]
- Kintner, J.; Lajoie, D.; Hall, J.; Whittimore, J.; Schoborg, R.V. Commonly prescribed β-lactam antibiotics induce C. trachomatis persistence/stress in culture at physiologically relevant concentrations. Front. Cell. Infect. Microbiol. 2014, 4, 44. [Google Scholar] [CrossRef]
- Lewis, M.E.; Belland, R.J.; AbdelRahman, Y.M.; Beatty, W.L.; Aiyar, A.A.; Zea, A.H.; Greene, S.J.; Marrero, L.; Buckner, L.R.; Tate, D.J.; et al. Morphologic and molecular evaluation of Chlamydia trachomatis growth in human endocervix reveals distinct growth patterns. Front. Cell. Infect. Microbiol. 2014, 4, 71. [Google Scholar] [CrossRef]
- Wyrick, P.B.; Knight, S.T. Pre-exposure of infected human endometrial epithelial cells to penicillin in vitro renders Chlamydia trachomatis refractory to azithromycin. J. Antimicrob. Chemother. 2004, 54, 79–85. [Google Scholar] [CrossRef]
- Rajeeve, K.; Vollmuth, N.; Janaki-Raman, S.; Wulff, T.F.; Baluapuri, A.; Dejure, F.R.; Huber, C.; Fink, J.; Schmalhofer, M.; Schmitz, W.; et al. Reprogramming of host glutamine metabolism during Chlamydia trachomatis infection and its key role in peptidoglycan synthesis. Nat. Microbiol. 2020, 5, 1390–1402. [Google Scholar] [CrossRef]
- Brunham, R.C.; Pourbohloul, B.; Mak, S.; White, R.; Rekart, M.L. The unexpected impact of a Chlamydia trachomatis infection control program on susceptibility to reinfection. J. Infect. Dis. 2005, 192, 1836–1844. [Google Scholar] [CrossRef]
- Hocking, J.S.; Vodstrcil, L.A.; Huston, W.M.; Timms, P.; Chen, M.Y.; Worthington, K.; McIver, R.; Tabrizi, S.N. A cohort study of Chlamydia trachomatis treatment failure in women: A study protocol. BMC Infect. Dis. 2013, 13, 379. [Google Scholar] [CrossRef] [PubMed]
- Nasr El-Din, A.; Sorour, H.; Fattouh, M.; Abu El-Hamd, M. Evaluation of the role of Chlamydia trachomatis in primary male infertility. Int. J. Clin. Pr. 2021, 75, e14702. [Google Scholar] [CrossRef] [PubMed]
- Moazenchi, M.; Totonchi, M.; Salman Yazdi, R.; Hratian, K.; Mohseni Meybodi, M.A.; Ahmadi Panah, M.; Chehrazi, M.; Mohseni Meybodi, A. The impact of Chlamydia trachomatis infection on sperm parameters and male fertility: A comprehensive study. Int. J. STD AIDS 2018, 29, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Carey, A.J.; Beagley, K.W. Chlamydia trachomatis, a hidden epidemic: Effects on female reproduction and options for treatment. Am. J. Reprod. Immunol. 2010, 63, 576–586. [Google Scholar] [CrossRef]
- den Heijer, C.D.J.; Hoebe, C.; Driessen, J.H.M.; Wolffs, P.; van den Broek, I.V.F.; Hoenderboom, B.M.; Williams, R.; de Vries, F.; Dukers-Muijrers, N. Chlamydia trachomatis and the Risk of Pelvic Inflammatory Disease, Ectopic Pregnancy, and Female Infertility: A Retrospective Cohort Study Among Primary Care Patients. Clin. Infect. Dis. 2019, 69, 1517–1525. [Google Scholar] [CrossRef]
- Menon, S.; Timms, P.; Allan, J.A.; Alexander, K.; Rombauts, L.; Horner, P.; Keltz, M.; Hocking, J.; Huston, W.M. Human and Pathogen Factors Associated with Chlamydia trachomatis-Related Infertility in Women. Clin. Microbiol. Rev. 2015, 28, 969–985. [Google Scholar] [CrossRef]
- Zheng, X.; Zhong, W.; O’Connell, C.M.; Liu, Y.; Haggerty, C.L.; Geisler, W.M.; Anyalechi, G.E.; Kirkcaldy, R.D.; Wiesenfeld, H.C.; Hillier, S.L.; et al. Host Genetic Risk Factors for Chlamydia trachomatis-Related Infertility in Women. J. Infect. Dis. 2021, 224 (Suppl. S2), S64–S71. [Google Scholar] [CrossRef]
- Benamri, I.; Azzouzi, M.; Sanak, K.; Moussa, A.; Radouani, F. An overview of genes and mutations associated with Chlamydiae species’ resistance to antibiotics. Ann. Clin. Microbiol. Antimicrob. 2021, 20, 59. [Google Scholar] [CrossRef]
- den Hartog, J.E.; Land, J.A.; Stassen, F.R.M.; Kessels, A.G.H.; Bruggeman, C.A. Serological markers of persistent C. trachomatis infections in women with tubal factor subfertility. Hum. Reprod. 2005, 20, 986–990. [Google Scholar] [CrossRef]
- Ohman, H.; Tiitinen, A.; Halttunen, M.; Birkelund, S.; Christiansen, G.; Koskela, P.; Lehtinen, M.; Paavonen, J.; Surcel, H.M. IL-10 polymorphism and cell-mediated immune response to Chlamydia trachomatis. Genes Immun. 2006, 7, 243–249. [Google Scholar] [CrossRef]
- van Ess, E.F.; Eck-Hauer, A.; Land, J.A.; Morré, S.A.; Ouburg, S. Combining individual Chlamydia trachomatis IgG antibodies MOMP, TARP, CPAF, OMP2, and HSP60 for tubal factor infertility prediction. Am. J. Reprod. Immunol. 2019, 81, e13091. [Google Scholar] [PubMed]
- Paavonen, J.; Karunakaran, K.P.; Noguchi, Y.; Anttila, T.; Bloigu, A.; Dillner, J.; Hallmans, G.; Hakulinen, T.; Jellum, E.; Koskela, P.; et al. Serum antibody response to the heat shock protein 60 of Chlamydia trachomatis in women with developing cervical cancer. Am. J. Obs. Gynecol. 2003, 189, 1287–1292. [Google Scholar] [CrossRef]
- Berestoviy, V.O.; Sokol, I.V.; Mahmood, A.A.; Ginzburg, V.G.; Govsieiev, D.O. A prospective study to analyze the specificity of chlamydial heat shock protein (chsp60) antibodies to diagnose tubal infertility. Wiad Lek. 2021, 74, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Shen, Z.; Luo, H.; Zhang, W.; Zhu, X. Chlamydia Trachomatis Infection-Associated Risk of Cervical Cancer: A Meta-Analysis. Med. 2016, 95, e3077. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Siddique, A.; Khan, A.A.; Wang, Q.; Malik, A.; Jan, A.T.; Rudayni, H.A.; Chaudhary, A.A.; Khan, S. Chlamydia Trachomatis Infection: Their potential implication in the Etiology of Cervical Cancer. J. Cancer 2021, 12, 4891–4900. [Google Scholar] [CrossRef]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Grieshaber, S.S.; Grieshaber, N.A.; Miller, N.; Hackstadt, T. Chlamydia trachomatis causes centrosomal defects resulting in chromosomal segregation abnormalities. Traffic 2006, 7, 940–949. [Google Scholar] [CrossRef]
- Johnson, K.A.; Tan, M.; Sütterlin, C. Centrosome abnormalities during a Chlamydia trachomatis infection are caused by dysregulation of the normal duplication pathway. Cell Microbiol 2009, 11, 1064–1073. [Google Scholar] [CrossRef]
- Knowlton, A.E.; Brown, H.M.; Richards, T.S.; Andreolas, L.A.; Patel, R.K.; Grieshaber, S.S. Chlamydia trachomatis infection causes mitotic spindle pole defects independently from its effects on centrosome amplification. Traffic 2011, 12, 854–866. [Google Scholar] [CrossRef]
- Prozialeck, W.C.; Fay, M.J.; Lamar, P.C.; Pearson, C.A.; Sigar, I.; Ramsey, K.H. Chlamydia trachomatis disrupts N-cadherin-dependent cell-cell junctions and sequesters beta-catenin in human cervical epithelial cells. Infect. Immun. 2002, 70, 2605–2613. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Discacciati, M.G.; Gimenes, F.; Pennacchi, P.C.; Faião-Flores, F.; Zeferino, L.C.; Derchain, S.M.; Teixeira, J.C.; Costa, M.C.; Zonta, M.; Termini, L.; et al. MMP-9/RECK Imbalance: A Mechanism Associated with High-Grade Cervical Lesions and Genital Infection by Human Papillomavirus and Chlamydia trachomatis. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1539–1547. [Google Scholar] [CrossRef] [PubMed]
- Jensen, K.E.; Thomsen, L.T.; Schmiedel, S.; Frederiksen, K.; Norrild, B.; van den Brule, A.; Iftner, T.; Kjær, S.K. Chlamydia trachomatis and risk of cervical intraepithelial neoplasia grade 3 or worse in women with persistent human papillomavirus infection: A cohort study. Sex. Transm. Infect. 2014, 90, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Silins, I.; Ryd, W.; Strand, A.; Wadell, G.; Törnberg, S.; Hansson, B.G.; Wang, X.; Arnheim, L.; Dahl, V.; Bremell, D.; et al. Chlamydia trachomatis infection and persistence of human papillomavirus. Int. J. Cancer 2005, 116, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Insinga, R.P.; Perez, G.; Wheeler, C.M.; Koutsky, L.A.; Garland, S.M.; Leodolter, S.; Joura, E.A.; Ferris, D.G.; Steben, M.; Brown, D.R.; et al. Incidence, duration, and reappearance of type-specific cervical human papillomavirus infections in young women. Cancer Epidemiol. Biomark. Prev. 2010, 19, 1585–1594. [Google Scholar] [CrossRef] [PubMed]
- Paba, P.; Bonifacio, D.; Di Bonito, L.; Ombres, D.; Favalli, C.; Syrjänen, K.; Ciotti, M. Co-expression of HSV2 and Chlamydia trachomatis in HPV-positive cervical cancer and cervical intraepithelial neoplasia lesions is associated with aberrations in key intracellular pathways. Intervirology 2008, 51, 230–234. [Google Scholar] [CrossRef]
- Simonetti, A.C.; Melo, J.H.; de Souza, P.R.; Bruneska, D.; de Lima Filho, J.L. Immunological’s host profile for HPV and Chlamydia trachomatis, a cervical cancer cofactor. Microbes Infect. 2009, 11, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Anttila, T.; Saikku, P.; Koskela, P.; Bloigu, A.; Dillner, J.; Ikäheimo, I.; Jellum, E.; Lehtinen, M.; Lenner, P.; Hakulinen, T.; et al. Serotypes of Chlamydia trachomatis and risk for development of cervical squamous cell carcinoma. Jama 2001, 285, 47–51. [Google Scholar] [CrossRef]
- Koskela, P.; Anttila, T.; Bjørge, T.; Brunsvig, A.; Dillner, J.; Hakama, M.; Hakulinen, T.; Jellum, E.; Lehtinen, M.; Lenner, P. Chlamydia trachomatis infection as a risk factor for invasive cervical cancer. Int. J. Cancer 2000, 85, 35–39. [Google Scholar] [CrossRef]
- Dillner, J.; Lehtinen, M.; Björge, T.; Luostarinen, T.; Youngman, L.; Koskela, P.; Hallmans, G.; Paavonen, J.; Sapp, M.; Schiller, J.T. Prospective seroepidemiologic study of human papillomavirus infection as a risk factor for invasive cervical cancer. J. Natl. Cancer Inst. 1997, 89, 1293–1299. [Google Scholar] [CrossRef]
- Xie, X.; Yang, M.; Ding, Y.; Chen, J. Microbial infection, inflammation and epithelial ovarian cancer. Oncol. Lett. 2017, 14, 1911–1919. [Google Scholar] [CrossRef]
- Shanmughapriya, S.; Senthilkumar, G.; Vinodhini, K.; Das, B.C.; Vasanthi, N.; Natarajaseenivasan, K. Viral and bacterial aetiologies of epithelial ovarian cancer. Eur. J. Clin. Microbiol. Infect. Dis. 2012, 31, 2311–2317. [Google Scholar] [CrossRef] [PubMed]
- Bodzek, P.; Partyka, R.; Damasiewicz-Bodzek, A. Antibodies against Hsp60 and Hsp65 in the sera of women with ovarian cancer. J. Ovarian. Res. 2014, 7, 30. [Google Scholar] [CrossRef] [PubMed]
- Hosseininasab-nodoushan, S.-A.; Ghazvini, K.; Jamialahmadi, T.; Keikha, M.; Sahebkar, A. Association of Chlamydia and Mycoplasma infections with susceptibility to ovarian cancer: A systematic review and meta-analysis. Semin. Cancer Biol. 2021, in press. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, S.; Lundin, E.; Elgh, F.; Ottander, U.; Idahl, A. Chlamydia trachomatis and Anti-MUC1 Serology and Subsequent Risk of High-Grade Serous Ovarian Cancer: A Population-Based Case-Control Study in Northern Sweden. Transl. Oncol. 2020, 13, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Pol, B.V.D.; Fife, K.; Taylor, S.N.; Nye, M.B.; Chavoustie, S.E.; Eisenberg, D.L.; Crane, L.; Hirsch, G.; Arcenas, R.; Marlowe, E.M.; et al. Evaluation of the Performance of the Cobas CT/NG Test for Use on the Cobas 6800/8800 Systems for Detection of Chlamydia trachomatis and Neisseria gonorrhoeae in Male and Female Urogenital Samples. J. Clin. Microbiol. 2019, 57, e01996-18. [Google Scholar]
- Sabbatucci, M.; Salfa, M.C.; Regine, V.; Pezzotti, P.; Suligoi, B. Estimated burden of Chlamydia trachomatis female infection and consequent severe pelvic inflammatory disease, Italy, 2005–2016. Ann. Ist. Super Sanita 2019, 55, 217–223. [Google Scholar]
- Borrego, M.J.; Gomes, J.P.; Lefebvre, J.F.; Eb, F.; Orfila, J.; Catry, M.A. Genotyping of Portuguese Chlamydia trachomatis urogenital isolates. Genitourin. Med. 1997, 73, 561–563. [Google Scholar] [CrossRef]
| WHO Region, by Sex | ||
|---|---|---|
| Chlamydia | ||
| 2012 | 2016 | |
| Women | ||
| African Region | 3.7 (2.7–5.2) | 5.0 (3.8–6.6) |
| Region of the Americas | 7.6 (6.7–8.7) | 7.0 (5.8–8.3) |
| South-East Asia Region | 1.8 (1.4–2.2) | 1.5 (1.0–2.5) |
| European Region | 2.2 (1.6–2.9) | 3.2 (2.5–4.2) |
| Eastern Mediterranean Region | 3.5 (2.4–5.0) | 3.8 (2.6–5.4) |
| Western Pacific Region | 6.2 (5.1–7.5) | 4.3 (3.0–5.9) |
| Global total | 4.2 (3.7–4.7) | 3.8 (3.3–4.5) |
| Men | ||
| African Region | 2.5 (1.7–3.6) | 4.0 (2.4–6.1) |
| Region of the Americas | 1.8 (1.3–2.6) | 3.7 (2.1–5.5) |
| South-East Asia Region | 1.3 (0.9–1.8) | 1.2 (0.6–2.1) |
| European Region | 1.5 (0.9–2.6) | 2.2 (1.5–3.0) |
| Eastern Mediterranean Region | 2.7 (1.6–4.3) | 3.0 (1.7–4.8) |
| Western Pacific Region | 5.2 (3.4–7.2) | 3.4 (2.0–5.3) |
| Global total | 2.7 (2.0–3.6) | 2.7 (1.9–3.7) |
| Country | Female | Male |
|---|---|---|
| Portugal (n = 240) | Serovar E, H, F, G, and D | Serovar E, D/F, H, and G |
| Sweden (n = 449, 2 different cohorts) | Serovar E, D, F, and K | Serovar E, F, K, and D |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodrigues, R.; Sousa, C.; Vale, N. Chlamydia trachomatis as a Current Health Problem: Challenges and Opportunities. Diagnostics 2022, 12, 1795. https://doi.org/10.3390/diagnostics12081795
Rodrigues R, Sousa C, Vale N. Chlamydia trachomatis as a Current Health Problem: Challenges and Opportunities. Diagnostics. 2022; 12(8):1795. https://doi.org/10.3390/diagnostics12081795
Chicago/Turabian StyleRodrigues, Rafaela, Carlos Sousa, and Nuno Vale. 2022. "Chlamydia trachomatis as a Current Health Problem: Challenges and Opportunities" Diagnostics 12, no. 8: 1795. https://doi.org/10.3390/diagnostics12081795
APA StyleRodrigues, R., Sousa, C., & Vale, N. (2022). Chlamydia trachomatis as a Current Health Problem: Challenges and Opportunities. Diagnostics, 12(8), 1795. https://doi.org/10.3390/diagnostics12081795








