Design of Gold Nanoparticle Vertical Flow Assays for Point-of-Care Testing
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. VFAs in POCT Diagnostics
3.2. Antibody Detection
3.3. Protein Biomarker Detection
3.4. Nucleic Acid and Small Molecule Detection
3.5. Options for Fabrication and Optimization of VFAs
3.5.1. Increasing the Flow Rate and Decreasing Membrane Pore Size Is a Good Combination for Improving VFA Assay Sensitivity
3.5.2. Buffer Optimization Is an Important Step in Assay Optimization and Troubleshooting
3.5.3. Altering the Reporter Is Another Strategy to Enhance the Limit of Detection (LoD)
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yetisen, A.K.; Akram, M.S.; Lowe, C.R. Paper-based microfluidic point-of-care diagnostic devices. Lab Chip 2013, 13, 2210–2251. [Google Scholar] [CrossRef] [PubMed]
- Parolo, C.; Sena-Torralba, A.; Bergua, J.F.; Calucho, E.; Fuentes-Chust, C.; Hu, L.; Rivas, L.; Álvarez-Diduk, R.; Nguyen, E.P.; Cinti, S.; et al. Tutorial: Design and fabrication of nanoparticle-based lateral-flow immunoassays. Nat. Protoc. 2020, 15, 3788–3816. [Google Scholar] [CrossRef] [PubMed]
- Peeling, R.W.; Holmes, K.K.; Mabey, D.; Ronald, A. Rapid tests for sexually transmitted infections (STIs): The way forward. Sex. Transm. Infect. 2006, 82 (Suppl. 5), v1–v6. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Hagen, J.A.; Papautsky, I. Point-of-care colorimetric detection with a smartphone. Lab Chip 2012, 12, 4240–4243. [Google Scholar] [CrossRef] [PubMed]
- Lei, R.; Huo, R.; Mohan, C. Current and emerging trends in point-of-care urinalysis tests. Expert Rev. Mol. Diagn. 2020, 20, 69–84. [Google Scholar] [CrossRef]
- Mansfield, M.A. The use of nitrocellulose membranes in lateral-flow assays. In Drugs of Abuse: Body Fluid Testing; Wong, R.C., Tse, H.Y., Eds.; Humana Press: Totowa, NJ, USA, 2005; pp. 71–85. [Google Scholar] [CrossRef]
- Zhang, G.; Guo, J.; Wang, X. Immunochromatographic lateral flow strip tests. Methods Mol. Biol. 2009, 504, 169–183. [Google Scholar] [CrossRef]
- Fenton, E.M.; Mascarenas, M.R.; López, G.P.; Sibbett, S.S. Multiplex lateral-flow test strips fabricated by two-dimensional shaping. ACS Appl. Mater. Interfaces 2009, 1, 124–129. [Google Scholar] [CrossRef]
- Danthanarayana, A.N.; Finley, E.; Vu, B.; Kourentzi, K.; Willson, R.C.; Brgoch, J. A multicolor multiplex lateral flow assay for high-sensitivity analyte detection using persistent luminescent nanophosphors. Anal. Methods 2020, 12, 272–280. [Google Scholar] [CrossRef]
- Luminostics. Available online: https://luminostics.com/ (accessed on 28 December 2021).
- INDICAID COVID-19 Rapid Antigen Test—Instructions for Use. Available online: https://www.fda.gov/media/151215/download (accessed on 28 December 2021).
- Rapid COVID-19 IgM/IgG Combo Test Kit—Instructions for Use. Available online: https://www.fda.gov/media/140297/download (accessed on 28 December 2021).
- SARS-CoV-2 Viral Mutations: Impact on COVID-19 Tests|FDA. Available online: https://www.fda.gov/medical-devices/coronavirus-covid-19-and-medical-devices/sars-cov-2-viral-mutations-impact-covid-19-tests (accessed on 28 December 2021).
- Ragnesola, B.; Jin, D.; Lamb, C.C.; Shaz, B.H.; Hillyer, C.D.; Luchsinger, L.L. COVID19 antibody detection using lateral flow assay tests in a cohort of convalescent plasma donors. BMC Res. Notes 2020, 13, 372. [Google Scholar] [CrossRef]
- Lei, R.; Mohan, C. Immunological biomarkers of COVID-19. Crit. Rev. Immunol. 2020, 40, 497–512. [Google Scholar] [CrossRef]
- Jiang, N.; Ahmed, R.; Damayantharan, M.; Ünal, B.; Butt, H.; Yetisen, A.K. Lateral and vertical flow assays for point-of-care diagnostics. Adv. Healthc. Mater. 2019, 8, e1900244. [Google Scholar] [CrossRef]
- Vashist, S.K.; Luppa, P.B.; Yeo, L.Y.; Ozcan, A.; Luong, J.H.T. Emerging technologies for next-generation point-of-care testing. Trends Biotechnol. 2015, 33, 692–705. [Google Scholar] [CrossRef]
- St. John, A.; Price, C.P. Existing and emerging technologies for point-of-care testing. Clin. Biochem. Rev. 2014, 35, 155–167. [Google Scholar]
- Lepowsky, E.; Ghaderinezhad, F.; Knowlton, S.; Tasoglu, S. Paper-based assays for urine analysis. Biomicrofluidics 2017, 11, 051501. [Google Scholar] [CrossRef]
- Warsinke, A. Point-of-care testing of proteins. Anal. Bioanal. Chem. 2009, 393, 1393–1405. [Google Scholar] [CrossRef]
- Simerville, J.A.; Maxted, W.C.; Pahira, J.J. Urinalysis: A comprehensive review. Am. Fam. Physician 2005, 71, 1153–1162, Erratum in Am. Fam. Phys. 2006, 74, 1096. [Google Scholar]
- Park, J.; Park, J.-K. Pressed region integrated 3D paper-based microfluidic device that enables vertical flow multistep assays for the detection of C-reactive protein based on programmed reagent loading. Sens. Actuators B Chem. 2017, 246. [Google Scholar] [CrossRef]
- Oh, Y.K.; Joung, H.A.; Kim, S.; Kim, M.G. Vertical flow immunoassay (VFA) biosensor for a rapid one-step immunoassay. Lab Chip 2013, 13, 768–772. [Google Scholar] [CrossRef]
- Frimpong, R.; Jang, W.; Kim, J.H.; Driskell, J.D. Rapid vertical flow immunoassay on AuNP plasmonic paper for SERS-based point of need diagnostics. Talanta 2021, 223 Pt 2, 121739. [Google Scholar] [CrossRef]
- Prajapati, A.; Verma, N.; Pandya, A. Highly sensitive vertical flow based point-of-care immunokit for rapid and early detection of human CRP as a cardiovascular risk factor. Biomed. Microdevices 2020, 22, 28. [Google Scholar] [CrossRef]
- Nybond, S.; Réu, P.; Rhedin, S.; Svedberg, G.; Alfvén, T.; Gantelius, J.; Svahn, H.A. Adenoviral detection by recombinase polymerase amplification and vertical flow paper microarray. Anal. Bioanal. Chem. 2019, 411, 813–822. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.; Hao, Y.; Miller, E.A.; Tay, D.M.Y.; Yee, E.; Kongsuphol, P.; Jia, H.; McBee, M.; Preiser, P.R.; Sikes, H.D. Vertical flow cellulose-based assays for SARS-CoV-2 antibody detection in human serum. ACS Sens. 2021, 6, 1891–1898. [Google Scholar] [CrossRef]
- HIV—Medmira. Available online: https://medmira.com/hiv/ (accessed on 28 December 2021).
- Clarke, O.J.R.; Goodall, B.L.; Hui, H.P.; Vats, N.; Brosseau, C.L. Development of a SERS-based rapid vertical flow assay for point-of-care diagnostics. Anal. Chem. 2017, 89, 1405–1410. [Google Scholar] [CrossRef] [PubMed]
- Ramachandran, S.; Singhal, M.; McKenzie, K.; Osborn, J.; Arjyal, A.; Dongol, S.; Baker, S.; Basnyat, B.; Farrar, J.; Dolecek, C.; et al. A rapid, multiplexed, high-throughput flow-through membrane immunoassay: A convenient alternative to ELISA. Diagnostics 2013, 3, 244–260. [Google Scholar] [CrossRef] [PubMed]
- Joung, H.-A.; Ballard, Z.S.; Ma, A.; Tseng, D.; Teshome, H.; Burakowski, S.; Garner, O.B.; Di Carlo, D.; Ozcan, A. Paper-based multiplexed vertical flow assay for point-of-care testing. Lab Chip 2019, 19, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Shi, F.; Sun, Y.; Wu, Y.; Zhu, M.; Feng, D.; Zhang, R.; Peng, L.; Chen, C. A novel, rapid and simple method for detecting brucellosis based on rapid vertical flow technology. J. Appl. Microbiol. 2019, 128, 794–802. [Google Scholar] [CrossRef]
- Reveal COVID-19. Available online: https://revealcovid19.com/ (accessed on 28 December 2021).
- Reuterswärd, P.; Gantelius, J.; Andersson Svahn, H. An 8-minute colorimetric paper-based reverse phase vertical flow serum microarray for screening of hyper IgE syndrome. Analyst 2015, 140, 7327–7334. [Google Scholar] [CrossRef] [PubMed]
- Chinnasamy, T.; Segerink, L.I.; Nystrand, M.; Gantelius, J.; Andersson Svahn, H. Point-of-care vertical flow allergen microarray assay: Proof of concept. Clin. Chem. 2014, 60, 1209–1216. [Google Scholar] [CrossRef] [Green Version]
- Chen, R.; Liu, B.; Ni, H.; Chang, N.; Luan, C.; Ge, Q.; Dong, J.; Zhao, X. Vertical flow assays based on core-shell SERS nanotags for multiplex prostate cancer biomarker detection. Analyst 2019, 144, 4051–4059. [Google Scholar] [CrossRef]
- Chen, P.; Gates-Hollingsworth, M.; Pandit, S.; Park, A.; Montgomery, D.; AuCoin, D.; Gu, J.; Zenhausern, F. Paper-based Vertical Flow Immunoassay (VFI) for detection of bio-threat pathogens. Talanta 2019, 191, 81–88. [Google Scholar] [CrossRef]
- Chen, R.; Du, X.; Cui, Y.; Zhang, X.; Ge, Q.; Dong, J.; Zhao, X. Vertical flow assay for inflammatory biomarkers based on nanofluidic channel array and SERS nanotags. Small 2020, 16, e2002801. [Google Scholar] [CrossRef]
- Mehta, S.M.; Mehta, S.; Muthurajan, H.; D’Souza, J.S. Vertical flow paper-based plasmonic device for cysteine detection. Biomed. Microdevices 2019, 21, 55. [Google Scholar] [CrossRef]
- Berger, A.G.; Restaino, S.M.; White, I.M. Vertical-flow paper SERS system for therapeutic drug monitoring of flucytosine in serum. Anal. Chim. Acta 2017, 949, 59–66. [Google Scholar] [CrossRef] [Green Version]
- Schonhorn, J.E.; Fernandes, S.C.; Rajaratnam, A.; Deraney, R.N.; Rolland, J.P.; Mace, C.R. A device architecture for three-dimensional, patterned paper immunoassays. Lab Chip 2014, 14, 4653–4658. [Google Scholar] [CrossRef]
- Bhardwaj, J.; Sharma, A.; Jang, J. Vertical flow-based paper immunosensor for rapid electrochemical and colorimetric detection of influenza virus using a different pore size sample pad. Biosens. Bioelectron. 2019, 126, 36–43. [Google Scholar] [CrossRef]
- Cretich, M.; Torrisi, M.; Daminelli, S.; Gagni, P.; Plavisch, L.; Chiari, M. Flow-through, viral co-infection assay for resource-limited settings. Talanta 2015, 132, 315–320. [Google Scholar] [CrossRef]
- Yee, E.H.; Lathwal, S.; Shah, P.P.; Sikes, H.D. Detection of biomarkers of periodontal disease in human saliva using stabilized, vertical flow immunoassays. ACS Sens. 2017, 2, 1589–1593. [Google Scholar] [CrossRef]
- Rivas, L.; Reuterswärd, P.; Rasti, R.; Herrmann, B.; Mårtensson, A.; Alfvén, T.; Gantelius, J.; Andersson-Svahn, H. A vertical flow paper-microarray assay with isothermal DNA amplification for detection of Neisseria meningitidis. Talanta 2018, 183, 192–200. [Google Scholar] [CrossRef]
- Serhan, M.; Jackemeyer, D.; Abi Karam, K.; Chakravadhanula, K.; Sprowls, M.; Cay-Durgun, P.; Forzani, E. A novel vertical flow assay for point of care measurement of iron from whole blood. Analyst 2021, 146, 1633–1641. [Google Scholar] [CrossRef]
- Moumita, M.; Shankar, K.M.; Abhiman, P.B.; Shamasundar, B.A. Development of a sandwich vertical flow immunogold assay for rapid detection of oxytetracycline residue in fish tissues. Food Chem. 2019, 270, 585–592. [Google Scholar] [CrossRef]
- Rong, Z.; Xiao, R.; Xing, S.; Xiong, G.; Yu, Z.; Wang, L.; Jia, X.; Wang, K.; Cong, Y.; Wang, S. SERS-based lateral flow assay for quantitative detection of C-reactive protein as an early bio-indicator of a radiation-induced inflammatory response in nonhuman primates. Analyst 2018, 143, 2115–2121. [Google Scholar] [CrossRef] [PubMed]
- Downs, C.; Milovancev, M.; Fu, E. Rational design and characterization of a lateral flow assay for canine C-reactive protein in wound exudate. Talanta 2020, 220, 121319. [Google Scholar] [CrossRef] [PubMed]
- Xiong, E.; Jiang, L.; Tian, T.; Hu, M.; Yue, H.; Huang, M.; Lin, W.; Jiang, Y.; Zhu, D.; Zhou, X. Simultaneous dual-gene diagnosis of SARS-CoV-2 based on CRISPR/Cas9-mediated lateral flow assay. Angew. Chem. Int. Ed. Engl. 2021, 60, 5307–5315. [Google Scholar] [CrossRef] [PubMed]
- Shelite, T.R.; Uscanga-Palomeque, A.C.; Castellanos-Gonzalez, A.; Melby, P.C.; Travi, B.L. Isothermal recombinase polymerase amplification-lateral flow detection of SARS-CoV-2, the etiological agent of COVID-19. J. Virol. Methods 2021, 296, 114227. [Google Scholar] [CrossRef]
- Lau, Y.L.; Ismail, I.B.; Mustapa, N.I.B.; Lai, M.Y.; Tuan Soh, T.S.; Haji Hassan, A.; Peariasamy, K.M.; Lee, Y.L.; Abdul Kahar, M.K.B.; Chong, J.; et al. Development of a reverse transcription recombinase polymerase amplification assay for rapid and direct visual detection of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). PLoS ONE 2021, 16, e0245164, Erratum in PLoS ONE 2021, 16, e0249100. [Google Scholar] [CrossRef]
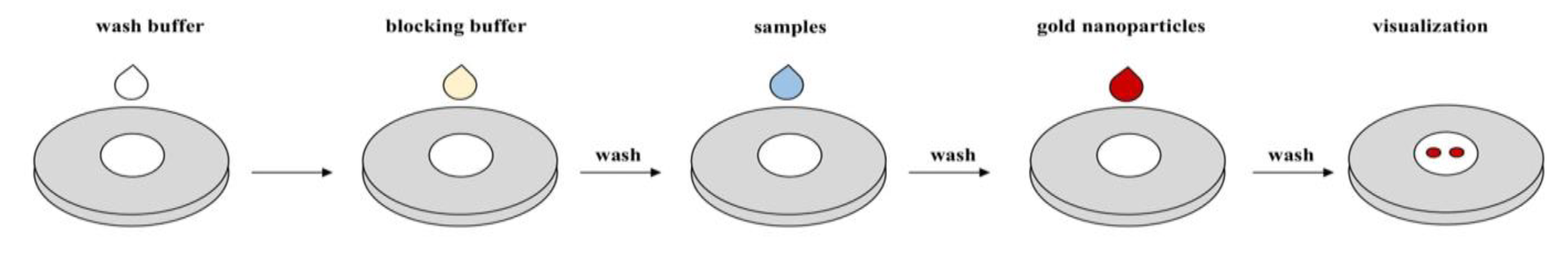
| Features | LFAs | VFAs |
|---|---|---|
| Sample flow | Capillary force [6] | External force; Gravity force; Capillary force [16] |
| Flow method | Passive [6] | Passive; Active [16] |
| Sensing response | Moderate [6] | Fast [16] |
| Washing steps | Not required [6] | Mostly yes [16] |
| Timed results | Required [6] | Not required [16] |
| Hook effect | Yes [2,6] | Mostly No [22,23] |
| Sample and conjugate separation | Mostly No [2,6] | Yes [24,25,26] No [22,23,27] |
| Sample volume | <100 µL [2,6] | 10–500 µL [22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47] |
| Sample type | Urine; Serum; Blood; Plasma; Sweat; Mucus; Saliva; Stool; Food; Cerebrospinal fluid [1,2,5] | Serum; Blood; Plasma [22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47] |
| Reagents volume | <100 µL [2] | <10 mL [22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47] |
| Multiplexing capacity | <10 [8,9] | >30 [30,35,43,45] |
| Detection method | Fluorescent NPs (QD; UCNP); Luminescent; NPs (Phosphors); Enzymatic reaction (HRP); Colorimetric NP (AuNP; CNP/CNT; Latex beads; MNP) [2] | Mostly colorimetric NPs (AgNPs; AuNP; SERS-AuNP) [16]; Enzymatic reaction (HRP; AP) [27] |
| Measurements | Qualitative or Quantitative coupled with portable reader [2,4] | Mostly qualitative or quantitative coupled with benchtop scanner [16] |
| Analyte (Ref) | Site of Use (Intended) | Indications | Detection Method | Time | Sample Type | LoD | Advantages | Disadvantages |
|---|---|---|---|---|---|---|---|---|
| Antibodies to HIV-1 and HIV-2 [28] | Clinic | HIV | GNP-colorimetric | 5 min | Serum, plasma, and venipuncture or fingerstick whole blood specimens | N/A | Sensitivity of 99.8% and a specificity of 99.7%. | Three-steps and expensive |
| Anti-HCV IgG [29] | Lab Clinic | Hepatitis C | GNP + SERS | 1 min | Commercial solution of monoclonal antibodies | Visual/SERS limit 63.1 and 3.32 µg/mL | Reproducible and intense results, little spot to spot variation | Raman spectrometer needed |
| Anti-LPS O9 IgM [30] | Clinic | Typhoid Fever | GNP-colorimetric | 30 min | Plasma | N/A | Multi-well multiplex assay; positive and negative controls can be performed in the same well as samples | Preparation of samples takes longer (~2 h), centrifuge and flatbed scanner required |
| Anti-MbpA IgG [31] | Clinic Home | Lyme disease | GNP-colorimetric | 20 min | Serum | 162.2 ng/mL | Mobile-phone based quantitative assay, inexpensive, no advanced medical equipment needed, no trained technicians needed, no need for sample dilution | Requires setup of a central server to interpret results, further studies needed for stability of test + buffers long term |
| Anti-OspC IgG [31] | 209.6 ng/mL | |||||||
| Anti-P41 IgG [31] | 1.05 μg/mL | |||||||
| Brucella antibodies [32] | Lab | Brucellosis | GNP-colorimetric | 5 min | Serum | 17:40 | 98% accuracy, potentially suitable for testing whole blood | Requires heating serum sample to 56 Celsius, samples must be incubated overnight |
| COVID-19 total Antibody [33] | Clinic | COVID-19 | GNP-colorimetric | 3 min | Serum, plasma or whole blood samples | N/A | Three-steps and expensive | |
| SARS-CoV2 Antibody [27] | Clinic | COVID-19 | NP-Biotin-Streptavidin-HRP colorimetric | 10 min | Serum | 0.5 nM | Cheap, rapid, easy to operate | Qualitative, in need of scanner for intensity analysis |
| IgE [34] | Clinic | Hyper IgE syndrome | GNP-colorimetric | 8 min | Serum | 1.9 μg/mL | Inexpensive; Simultaneously screen 113 samples where 1208 spots are available. | Overnight drying after serum sample added; flatbed scanner needed for detection |
| IgE-reactive allergens [35] | Clinic | Allergies | Neutravidin-GNP colorimetric | 15 min | Serum | 1 ng/mL IgG | Large multiplex capabilities (1480 spots available), low CV | Tabletop scanner and software required, skilled labor |
| mouse IgG [24] | Clinic | NA | SERS GNC-colorimetric | 2 min | Serum | 3 ng/mL | A plasmonic filter paper with pre-adsorbed goat anti-mouse IgG antibody to improve sensitivity; less than 2 min assays time | using FEI-Quanta 450 SEM to image; Enwave Optronics, Inc. ProRaman-L-785B instrument to analyze |
| Analyte (Ref) | Site of Use (Intended) | Indications | Detection Method | Time | Sample Type | LoD | Advantages | Disadvantages |
|---|---|---|---|---|---|---|---|---|
| AFP [36] | Clinic | Prostate cancer | SERS | 10 min | Serum | 0.26 pg/mL | Sensitive, rapid, one test zone to show three detections | Surface-enhanced Raman scattering (SERS)-based vertical flow assay in need of Raman microscope |
| CEA [36] | 0.43 pg/mL | |||||||
| PSA [36] | 0.37 pg/mL | |||||||
| Capsular polysaccharide [37] | Lab | B. pseudomallei (meliodosis) | GNP-colorimetric | <30 min | 0.02 ng/mL | Analyte concentration is highly consistent with signal intensity | Sample type unspecified. Assumed to have used purified B. pseudomallei, results may not be similar for whole blood/serum testing | |
| CRP [25] | Clinic Home | Asymptomatic cardiovascular disease | GNP-colorimetric | 1–2 min | Serum/Whole blood | 10 ng/mL | Inexpensive, highly accurate, no expensive equipment needed to interpret results | Semi-quantitative; signal intensity of low-risk CRP concentrations (<1 mg/L) looks similar to high-risk CRP concentrations |
| CRP [22] | Clinic | Cardiovascular disease | 15 min | Serum | 0.005 µg/mL | Upper limit of 5 ug/mL without hook effect, one-step assay (no need to sequentially add reagents) | Low sample volume capability; need of benchtop scanner for quantitative measurement | |
| CRP [23] | Clinic | Cardiovascular disease | GNP-colorimetric | 2 min | Serum | 10 ng/mL | One-step assay (no need to sequentially add reagents), upper limit of 10 ug/mL without hook effect | No mentioning on the stability; need of benchtop scanner for quantitative measurement |
| CRP [38] | Clinic | Inflammatory biomarker detection | Raman dyes encoded core–shell SERS nanotags | NA | Serum | 53.4 fg/mL | Ultra-sensitive; a linear range spanning five orders of magnitude; The proposed method shows acceptable accuracy and repeatability | In need of Raman spectrum measurement system to analyze; In need of longer assay time compared to other VFA. |
| IL-6 [38] | 4.72 fg/mL | |||||||
| SAA [38] | 48.3 fg/mL | |||||||
| Procalcitonin [38] | 7.53 fg/mL | |||||||
| Cysteine [39] | Clinic Home | Cystinuria | AgNP-colorimetric | 2 min | Standard cysteine solution | 10 nM visible limit; 0.1 nM quantification limit | Inexpensive, no skilled labor or sophisticated equipment, specific assay | Semi-quantitative, some normal ranges of cysteine concentration look visually positive |
| Flucytosine [40] | Clinic | Therapeutic drug monitoring | SERS | 15 min | Serum | 10 μg/mL | No serum dilution needed | Raman spectrometer needed |
| hCG [41] | Clinic | pregnancy | GNP-colorimetric | 10 min | urine | 0.5 mIU/mL | Cheap, rapid, small volume requirement (20 uL) | Need to peel the device to see the readout; need washing compared to commercial lateral flow test |
| HINI [42] | Clinic | Influenza | Electrochemical and colorimetric | 6 min | saliva | 4.7 PFU/mL in saliva by EIS, 2.27 PFU/mL in saliva by colorimetric | High sensitivity, simplicity of operation; duo-methods detection with higher accuracy | Electrochemical signal needs electrochemical impedance spectroscopy (EIS) for measurements; reduced signal after 30 days storage |
| HIV p24 and hepatitis B virus antigens [43] | Clinic | HIV and Hepatitis B | Streptavidin-AP + NBT/BCIP | 5 min | Pure bovine serum | 0.95 ng/mL for HIV p24 and 1.12 ng/mL for HBV-SAg | Multi-well multiplex, sensitive | Higher background in serum sample Need benchtop to analyze the signal |
| MMP-8 [44] | Lab Clinic | Periodontal disease | Eosin-based signal polymerization, colorimetric | N/A | ~1 nM 1.1 nM | 75% recovery of saliva sample after processing is finished, only about 15 μL original sample needed | Signals near the lower end of clinically relevant concentrations not easily discernible, qualitative, saliva must be centrifuged and frozen | |
| MMP-9 [44] |
| Analyte (Ref) | Site of use (Intended) | Indications | Detection Method | Time | Sample Type | LoD | Advantages | Disadvantages |
|---|---|---|---|---|---|---|---|---|
| Adenoviral DNA [26] | Lab Clinic | Adenoviral infection | GNP-colorimetric | 6 min | Constructed amplicons + nasopharyngeal aspirates from patients | 50 nM | Detect multiple strains of adenovirus w/low inter- and intra-assay variation | 1 h RPA and ssDNA generation; possible cross-reactivity |
| DNA [45] | Clinic | Neisseria meningitidis (meningitis) | Streptavidin-GNP colorimetric | 20 min | Constructed amplicons | 38–2.1 × 106 copies/assay | Multiplex detection based on different capture probes | DNA extraction, RPA, ssDNA generation required |
| Analyte (Ref) | Site of Use (Intended) | Indications | Detection Method | Time | Sample Type | LoD | Advantages | Disadvantages |
|---|---|---|---|---|---|---|---|---|
| Iron [46] | On-site clinic | NA | NA | NA | Whole blood | NA | the system consists of a smartphone and an in-house developed app | NA |
| Oxytetracycline [47] | On-site clinic | Drug abuse | Silver-enhanced GNP-colorimetric | 4 min | Fish tissue | 2 ng/mL | Simple, sensitive and rapid assay. Room for several test samples. | Multiple steps and lack of reference for semiquantitative measurements. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lei, R.; Wang, D.; Arain, H.; Mohan, C. Design of Gold Nanoparticle Vertical Flow Assays for Point-of-Care Testing. Diagnostics 2022, 12, 1107. https://doi.org/10.3390/diagnostics12051107
Lei R, Wang D, Arain H, Mohan C. Design of Gold Nanoparticle Vertical Flow Assays for Point-of-Care Testing. Diagnostics. 2022; 12(5):1107. https://doi.org/10.3390/diagnostics12051107
Chicago/Turabian StyleLei, Rongwei, David Wang, Hufsa Arain, and Chandra Mohan. 2022. "Design of Gold Nanoparticle Vertical Flow Assays for Point-of-Care Testing" Diagnostics 12, no. 5: 1107. https://doi.org/10.3390/diagnostics12051107
APA StyleLei, R., Wang, D., Arain, H., & Mohan, C. (2022). Design of Gold Nanoparticle Vertical Flow Assays for Point-of-Care Testing. Diagnostics, 12(5), 1107. https://doi.org/10.3390/diagnostics12051107





