Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Clinicopathologic Information
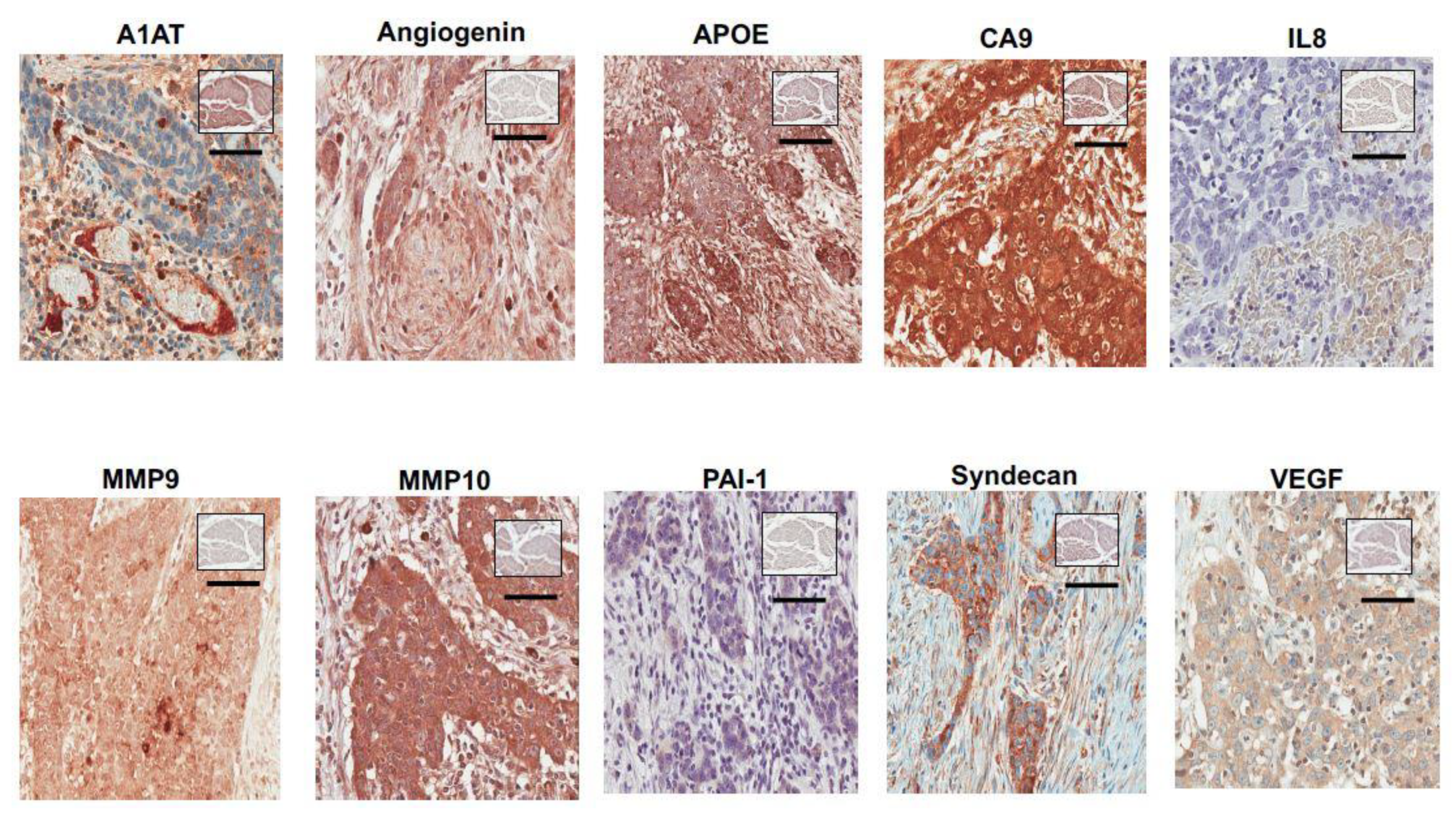
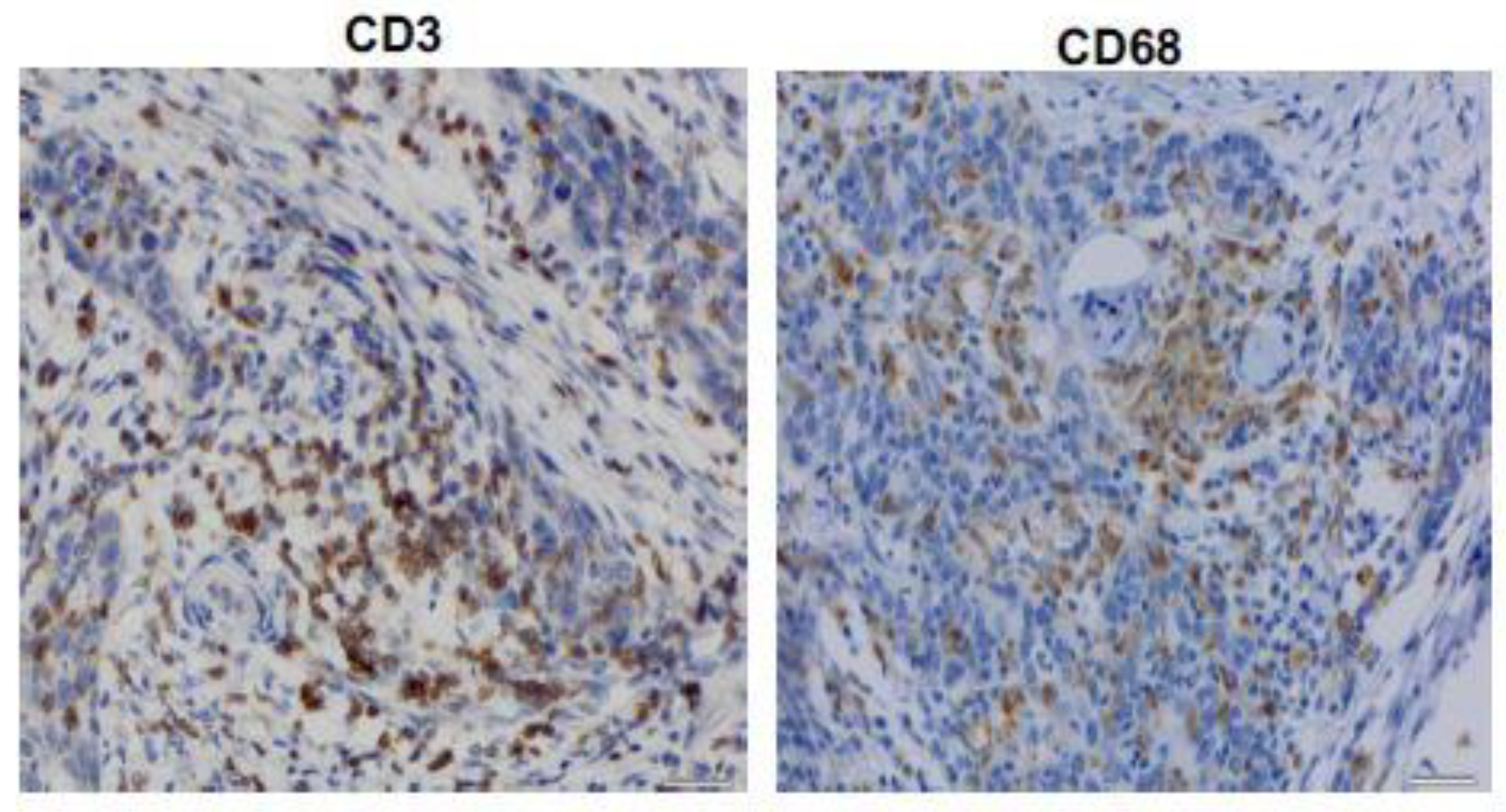
2.2. Immunohistochemistry
2.3. Image Analysis
2.4. Statistical Analysis
3. Results
3.1. Demographics of the Patients and Tumor Characteristics
3.2. Bladder Cancer Associated Diagnostic Signature Immunohistochemical Results
3.3. CD3+ and CD68+ Immunohistochemical Results
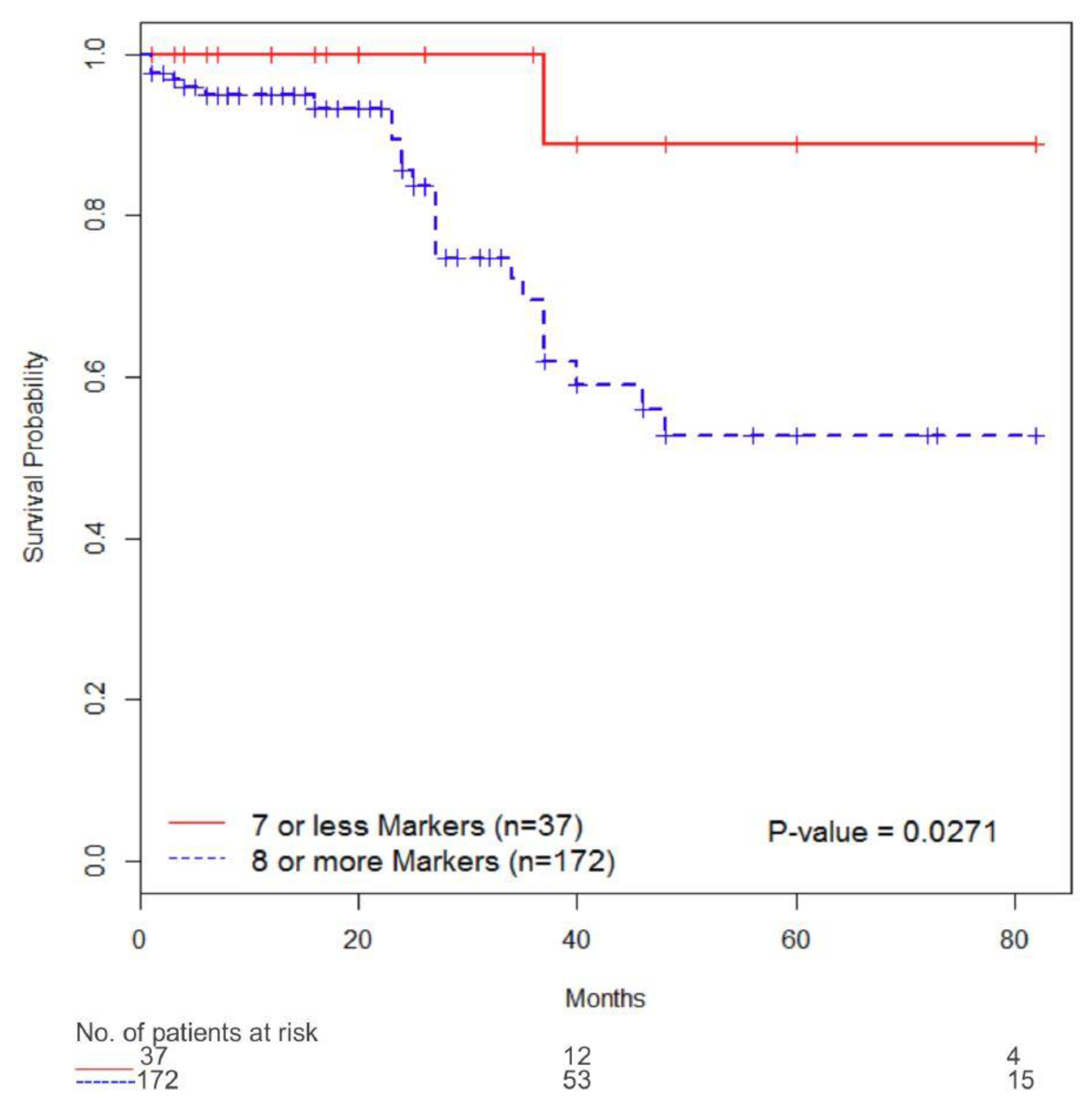
3.4. Immunophenotype and Survival
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Han, K.S.; You, D.; Jeong, I.G.; Kwon, T.; Hong, B.; Hong, J.H.; Ahn, H.; Ahn, T.Y.; Kim, C.S. Is intravesical Bacillus Calmette-Guerin therapy superior to chemotherapy for intermediate-risk non-muscle-invasive bladder cancer? An ongoing debate. J. Korean Med. Sci. 2015, 30, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Anastasiadis, A.; de Reijke, T.M. Best practice in the treatment of nonmuscle invasive bladder cancer. Ther. Adv. Urol. 2011, 4, 13–32. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.S.; Bochner, B.H.; Chou, R.; Dreicer, R.; Kamat, A.M.; Lerner, S.P.; Lotan, Y.; Meeks, J.J.; Michalski, J.M.; Morgan, T.M.; et al. Treatment of Non-Metastatic Muscle-Invasive Bladder Cancer: AUA/ASCO/ASTRO/SUO Guideline. J. Urol. 2017, 198, 552–559. [Google Scholar] [CrossRef]
- Chade, D.C.; Shariat, S.F.; Godoy, G.; Savage, C.J.; Cronin, A.M.; Bochner, B.H.; Donat, S.M.; Herr, H.W.; Dalbagni, G. Clinical Outcomes of Primary Bladder Carcinoma In Situ in a Contemporary Series. J. Urol. 2010, 184, 74–80. [Google Scholar] [CrossRef]
- CenterWatch. Valstar New FDA Drug Approval. 2013. Available online: https://www.centerwatch.com/drug-information/fda-approved-drugs/drug/498/valstar%20on%20August% (accessed on 13 August 2018).
- Patel, M.I.; Bang, A.; Gillatt, D.; Smith, D.P. Contemporary radical cystectomy outcomes in patients with invasive bladder cancer: A population-based study. BJU Int. 2015, 116, 18–25. [Google Scholar] [CrossRef]
- Stenzl, A.; Cowan, N.C.; De Santis, M.; Kuczyk, M.A.; Merseburger, A.S.; Ribal, M.J.; Sherif, A.; Witjes, J.A.; European Association of Urology (EAU). Treatment of Muscle-invasive and Metastatic Bladder Cancer: Update of the EAU Guidelines. Eur. Urol. 2011, 60, e53. [Google Scholar] [CrossRef]
- Sternberg, C.N.; Donat, S.M.; Bellmunt, J.; Millikan, R.E.; Stadler, W.; De Mulder, P.; Sherif, A.; von der Maase, H.; Tsukamoto, T.; Soloway, M.S. Chemotherapy for Bladder Cancer: Treatment Guidelines for Neoadjuvant Chemotherapy, Bladder Preservation, Adjuvant Chemotherapy, and Metastatic Cancer. Urology 2007, 69, 62–79. [Google Scholar] [CrossRef]
- Robertson, A.G. Comprehensive Molecular Characterization of Muscle-Invasive Bladder Cancer. Cell 2017, 171, 540–556. [Google Scholar] [CrossRef]
- Chen, L.M.; Chang, M.; Dai, Y.; Chai, K.X.; Dyrskjot, L.; Sanchez-Carbayo, M.; Szarvas, T.; Zwarthoff, E.C.; Lokeshwar, V.; Jeronimo, C.; et al. External Validation of a Multiplex Urinary Protein Panel for the Detection of Bladder Cancer in a Multicenter Cohort. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1804–1812. [Google Scholar] [CrossRef]
- Goodison, S.; Chang, M.; Dai, Y.; Urquidi, V.; Rosser, C.J. A Multi-Analyte Assay for the Non-Invasive Detection of Bladder Cancer. PLoS ONE 2012, 7, e47469. [Google Scholar] [CrossRef] [PubMed]
- Rosser, C.J.; Chang, M.; Dai, Y.; Ross, S.; Mengual, L.; Alcaraz, A.; Goodison, S. Urinary Protein Biomarker Panel for the Detection of Recurrent Bladder Cancer. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1340–1345. [Google Scholar] [CrossRef] [PubMed]
- Rosser, C.J.; Ross, S.; Chang, M.; Dai, Y.; Mengual, L.; Zhang, G.; Kim, J.; Urquidi, V.; Alcaraz, A.; Goodison, S. Multiplex Protein Signature for the Detection of Bladder Cancer in Voided Urine Samples. J. Urol. 2013, 190, 2257–2262. [Google Scholar] [CrossRef] [PubMed]
- Urquidi, V.; Chang, M.; Dai, Y.; Kim, J.; Wolfson, E.D.; Goodison, S.; Rosser, C.J. IL8 as a urinary biomarker for the detection of bladder cancer. BMC Urol. 2012, 12, 12. [Google Scholar] [CrossRef]
- Urquidi, V.; Goodison, S.; Kim, J.; Chang, M.; Dai, Y.; Rosser, C.J. Vascular Endothelial Growth Factor, Carbonic Anhydrase 9, and Angiogenin as Urinary Biomarkers for Bladder Cancer Detection. Urology 2012, 79, e1181–e1185. [Google Scholar] [CrossRef]
- Urquidi, V.; Goodison, S.; Rosser, C.J. Diagnostic Potential of Urinary α1-Antitrypsin and Apolipoprotein E in the Detection of Bladder Cancer. J. Urol. 2013. [Google Scholar] [CrossRef]
- Urquidi, V.; Kim, J.; Chang, M.; Dai, Y.; Rosser, C.J.; Goodison, S. CCL18 in a Multiplex Urine-Based Assay for the Detection of Bladder Cancer. PLoS ONE 2012, 7, e37797. [Google Scholar] [CrossRef]
- Tashiro, Y.; Nishida, C.; Sato-Kusubata, K.; Ohki-Koizumi, M.; Ishihara, M.; Sato, A.; Gritli, I.; Komiyama, H.; Sato, Y.; Dan, T.; et al. Inhibition of PAI-1 induces neutrophil-driven neoangiogenesis and promotes tissue regeneration via production of angiocrine factors in mice. Blood 2012, 119, 6382–6393. [Google Scholar] [CrossRef]
- Xu, J.; Zhu, D.; Sonoda, S.; He, S.; Spee, C.; Ryan, S.J.; Hinton, D.R. Over-expression of BMP4 inhibits experimental choroidal neovascularization by modulating VEGF and MMP-9. Angiogenesis 2012, 15, 213–227. [Google Scholar] [CrossRef]
- Zhang, G.; Miyake, M.; Lawton, A.; Goodison, S.; Rosser, C.J. Matrix metalloproteinase-10 promotes tumor progression through regulation of angiogenic and apoptotic pathways in cervical tumors. BMC Cancer 2014, 14, 310. [Google Scholar] [CrossRef]
- Folkman, J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1995, 1, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J.; Klagsbrun, M. Angiogenic factors. Science 1987, 235, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Folkman, J. Patterns and Emerging Mechanisms of the Angiogenic Switch during Tumorigenesis. Cell 1996, 86, 353–364. [Google Scholar] [CrossRef]
- Blood, C. Tumor interactions with the vasculature: Angiogenesis and tumor metastasis. Biochim. Biophys. Acta (Bba)—Rev. Cancer 1990, 1032, 89–118. [Google Scholar] [CrossRef]
- Giacoia, E.G.; Miyake, M.; Lawton, A.; Goodison, S.; Rosser, C.J. PAI-1 Leads to G 1 -Phase Cell-Cycle Progression through Cyclin D3/cdk4/6 Upregulation. Mol. Cancer Res. 2014, 12, 322–334. [Google Scholar] [CrossRef]
- Miyake, M.; Goodison, S.; Lawton, A.; Gomes-Giacoia, E.; Rosser, C.J. Angiogenin promotes tumoral growth and angiogenesis by regulating matrix metallopeptidase-2 expression via the ERK1/2 pathway. Oncogene 2014, 34, 890–901. [Google Scholar] [CrossRef]
- Zhang, G.; Gomes-Giacoia, E.; Dai, Y.; Lawton, A.; Miyake, M.; Furuya, H.; Goodison, S.; Rosser, C.J. Validation and clinicopathologic associations of a urine-based bladder cancer biomarker signature. Diagn. Pathol. 2014, 9, 200. [Google Scholar] [CrossRef]
- Yang, N.; Feng, S.; Shedden, K.; Xie, X.; Liu, Y.; Rosser, C.J.; Lubman, D.M.; Goodison, S. Urinary Glycoprotein Biomarker Discovery for Bladder Cancer Detection Using LC/MS-MS and Label-Free Quantification. Clin. Cancer Res. 2011, 17, 3349–3359. [Google Scholar] [CrossRef]
- Kreunin, P.; Zhao, J.; Rosser, C.; Urquidi, V.; Lubman, D.M.; Goodison, S. Bladder Cancer Associated Glycoprotein Signatures Revealed by Urinary Proteomic Profiling. J. Proteome Res. 2007, 6, 2631–2639. [Google Scholar] [CrossRef]
- Rosser, C.J.; Liu, L.; Sun, Y.; Villicana, P.; McCullers, M.; Porvasnik, S.; Young, P.R.; Parker, A.S.; Goodison, S. Bladder Cancer-Associated Gene Expression Signatures Identified by Profiling of Exfoliated Urothelia. Cancer Epidemiol. Biomark. Prev. 2009, 18, 444–453. [Google Scholar] [CrossRef]
- Urquidi, V.; Goodison, S.; Cai, Y.; Sun, Y.; Rosser, C.J. A Candidate Molecular Biomarker Panel for the Detection of Bladder Cancer. Cancer Epidemiol. Biomark. Prev. 2012, 21, 2149–2158. [Google Scholar] [CrossRef] [PubMed]
- Aleman, A.; Cebrian, V.; Alvarez, M.; Lopez, V.; Orenes, E.; Lopez-Serra, L.; Algaba, F.; Bellmunt, J.; Lopez-Beltran, A.; Gonzalez-Peramato, P.; et al. Identification of PMF1 Methylation in Association with Bladder Cancer Progression. Clin. Cancer Res. 2008, 14, 8236–8243. [Google Scholar] [CrossRef] [PubMed]
- Dyrskjøt, L.; Zieger, K.; Kissow Lildal, T.; Reinert, T.; Gruselle, O.; Coche, T.; Borre, M.; Ørntoft, T.F. Expression of MAGE-A3, NY-ESO-1, LAGE-1 and PRAME in urothelial carcinoma. Br. J. Cancer 2012, 107, 116–122. [Google Scholar] [CrossRef] [PubMed]
- van Oers, J.M.M.; Zwarthoff, E.C.; Rehman, I.; Azzouzi, A.-R.; Cussenot, O.; Meuth, M.; Hamdy, F.C.; Catto, J.W.F. FGFR3 Mutations Indicate Better Survival in Invasive Upper Urinary Tract and Bladder Tumours. Eur. Urol. 2009, 55, 650–658. [Google Scholar] [CrossRef] [PubMed]
- García-Baquero, R.; Puerta, P.; Beltran, M.; Alvarez, M.; Sacristan, R.; Alvarez-Ossorio, J.L.; Sánchez-Carbayo, M. Methylation of a Novel Panel of Tumor Suppressor Genes in Urine Moves Forward Noninvasive Diagnosis and Prognosis of Bladder Cancer: A 2-Center Prospective Study. J. Urol. 2013, 190, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Kandimalla, R.; Masius, R.; Beukers, W.; Bangma, C.H.; Orntoft, T.F.; Dyrskjot, L.; van Leeuwen, N.; Lingsma, H.; van Tilborg, A.A.G.; Zwarthoff, E.C. A 3-Plex Methylation Assay Combined with the FGFR3 Mutation Assay Sensitively Detects Recurrent Bladder Cancer in Voided Urine. Clin. Cancer Res. 2013, 19, 4760–4769. [Google Scholar] [CrossRef] [PubMed]
- Reinert, T.; Borre, M.; Christiansen, A.; Hermann, G.G.; Ørntoft, T.F.; Dyrskjøt, L. Diagnosis of Bladder Cancer Recurrence Based on Urinary Levels of EOMES, HOXA9, POU4F2, TWIST1, VIM, and ZNF154 Hypermethylation. PLoS ONE 2012, 7, e46297. [Google Scholar] [CrossRef]
- Goodison, S.; Ogawa, O.; Matsui, Y.; Kobayashi, T.; Miyake, M.; Ohnishi, S.; Fujimoto, K.; Dai, Y.; Shimizu, Y.; Tsukikawa, K.; et al. A multiplex urinary immunoassay for bladder cancer detection: Analysis of a Japanese cohort. J. Transl. Med. 2016, 14, 287. [Google Scholar] [CrossRef]
- Shimizu, Y.; Furuya, H.; Bryant Greenwood, P.; Chan, O.; Dai, Y.; Thornquist, M.D.; Goodison, S.; Rosser, C.J. A multiplex immunoassay for the non-invasive detection of bladder cancer. J. Transl. Med. 2016, 14, 31. [Google Scholar] [CrossRef]
- Masuda, N.; Ogawa, O.; Park, M.; Liu, A.Y.; Goodison, S.; Dai, Y.; Kozai, L.; Furuya, H.; Lotan, Y.; Rosser, C.J.; et al. Meta-analysis of a 10-plex urine-based biomarker assay for the detection of bladder cancer. Oncotarget 2018, 9, 7101. [Google Scholar] [CrossRef]
- Bremnes, R.M.; Dønnem, T.; Al-Saad, S.; Al-Shibli, K.; Andersen, S.; Sirera, R.; Camps, C.; Marinez, I.; Busund, L.-T. The Role of Tumor Stroma in Cancer Progression and Prognosis: Emphasis on Carcinoma-Associated Fibroblasts and Non-small Cell Lung Cancer. J. Thorac. Oncol. 2011, 6, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Williams, S.G.; Stein, J.P. Molecular pathways in bladder cancer. Urol. Res. 2004, 32, 373–385. [Google Scholar] [CrossRef] [PubMed]
- van Kessel, K.E.M.; van der Keur, K.A.; Dyrskjøt, L.; Algaba, F.; Welvaart, N.Y.C.; Beukers, W.; Segersten, U.; Keck, B.; Maurer, T.; Simic, T.; et al. Molecular Markers Increase Precision of the European Association of Urology Non–Muscle-Invasive Bladder Cancer Progression Risk Groups. Clin. Cancer Res. 2018, 24, 1586–1593. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Carbayo, M.; Cordon-Cardó, C. Molecular Alterations Associated With Bladder Cancer Progression. Semin. Oncol. 2007, 34, 75–84. [Google Scholar] [CrossRef]
- Sjödahl, G.; Lövgren, K.; Lauss, M.; Chebil, G.; Patschan, O.; Gudjonsson, S.; Månsson, W.; Fernö, M.; Leandersson, K.; Lindgren, D.; et al. 790 Infiltration of CD3+ and CD68+ cells in bladder cancer is subtype specific and affects outcome of patients with muscle-invasive tumors. Eur. Urol. Suppl. 2014, 13, e790. [Google Scholar] [CrossRef]
- Banat, G.A.; Tretyn, A.; Pullamsetti, S.S.; Wilhelm, J.; Weigert, A.; Olesch, C.; Ebel, K.; Stiewe, T.; Grimminger, F.; Seeger, W.; et al. Immune and Inflammatory Cell Composition of Human Lung Cancer Stroma. PLoS ONE 2015, 10, e0139073. [Google Scholar] [CrossRef]
- Ohnuki, H.; Jiang, K.; Wang, D.; Salvucci, O.; Kwak, H.; Sanchez-Martin, D.; Maric, D.; Tosato, G. Tumor-infiltrating myeloid cells activate Dll4/Notch/TGF-beta signaling to drive malignant progression. Cancer Res. 2014, 74, 2038–2049. [Google Scholar] [CrossRef]
| Features | Bladder Cancer (%) n = 213 | Controls (%) n = 74 | p-Value |
|---|---|---|---|
| Age (years) | |||
| <65 | 50 (23.9%) | 74 (100.0%) | <0.0001 |
| ≥65 | 150 (70.4%) | 0 (0.0%) | |
| Unavailable | 12 (5.6%) | ||
| Sex | |||
| Female | 48 (22.5%) | 16 (21.6%) | 0.154 |
| Male | 153 (71.8%) | 58 (78.4%) | |
| Unavailable | 12 (5.6%) | ||
| Race | |||
| Caucasian | 162 (76.5%) | 61 (82.4%) | 0.672 |
| Other | 30 (14.1%) | 7 (9.5%) | |
| Unavailable | 20 (9.4%) | 6 (8.1%) | |
| Tumor grade | |||
| High-grade | 176 (82.6%) | ||
| Low-grade | 26 (12.2%) | ||
| Unavailable | 11 (5.2%) | ||
| Tumor stage | |||
| Ta | 50 (23.5%) | ||
| Tis | 20 (9.4%) | ||
| T1 | 62 (29.1%) | ||
| T2 | 31 (14.6%) | ||
| ≥T3, N+ or M+ | 39 (18.2%) | ||
| Unavailable | 11 (5.2%) | ||
| Recurrence | |||
| Yes | 70 (32.9%) | ||
| No | 143 (67.1%) |
| Target Expression | Bladder Cancer, % | Benign, % | p-Value | Target Expression | Bladder Cancer, % | Benign, % | p-Value |
|---|---|---|---|---|---|---|---|
| VEGFA | APOE | ||||||
| 1 | 35 (21.3%) | 24 (32.4%) | 0.2361 | 1 | 36 (18.7%) | 30 (42.3%) | <0.0001 |
| 2 | 43 (26.2%) | 17 (23.0%) | 2 | 41 (21.2%) | 25 (35.2%) | ||
| 3 | 45 (27.4%) | 14 (18.9%) | 3 | 56 (29.0%) | 10 (14.1%) | ||
| 4 | 41 (25.0%) | 19 (25.7%) | 4 | 60 (31.1%) | 6 (8.5%) | ||
| MMP9 | A1AT | ||||||
| 1 | 22 (12.1%) | 42 (56.8%) | <0.0001 | 1 | 60 (32.3%) | 5 (6.8%) | <0.0001 |
| 2 | 41 (22.5%) | 23 (31.1%) | 2 | 55 (29.6%) | 10 (13.5%) | ||
| 3 | 57 (31.3%) | 7 (9.5%) | 3 | 44 (23.7%) | 21 (28.4%) | ||
| 4 | 62 (34.1%) | 2 (2.7%) | 4 | 27 (14.5%) | 38 (51.4%) | ||
| MMP10 | SDC1 | ||||||
| 1 | 36 (19.7%) | 28 (37.8%) | <0.0001 | 1 | 22 (11.9%) | 41 (58.6%) | <0.0001 |
| 2 | 30 (16.4%) | 34 (45.9%) | 2 | 44 (23.8%) | 20 (28.6%) | ||
| 3 | 55 (30.1%) | 9 (12.2%) | 3 | 57 (30.8%) | 7 (10.0%) | ||
| 4 | 62 (33.9%) | 3 (4.1%) | 4 | 62 (33.5%) | 2 (2.9%) | ||
| PAI1 | ANG | ||||||
| 1 | 13 (7.1%) | 50 (69.4%) | <0.0001 | 1 | 5 (2.6%) | 61 (84.7%) | <0.0001 |
| 2 | 51 (27.9%) | 13 (18.1%) | 2 | 55 (28.5%) | 11 (15.3%) | ||
| 3 | 60 (32.8%) | 4 (5.6%) | 3 | 66 (34.2%) | 0 (0%) | ||
| 4 | 59 (32.2%) | 5 (6.9%) | 4 | 67 (34.7%) | 0 (0%) | ||
| IL8 | |||||||
| 1 | 62 (31.6%) | 4 (5.7%) | <0.0001 | ||||
| 2 | 49 (25.0%) | 18 (25.7%) | |||||
| 3 | 39 (19.9%) | 27 (38.6%) | |||||
| 4 | 46 (23.5%) | 21 (30.0%) | |||||
| CA9 | |||||||
| 1 | 23 (11.7%) | 43 (61.4%) | <0.0001 | ||||
| 2 | 48 (24.5%) | 19 (27.1%) | |||||
| 3 | 60 (30.6%) | 6 (8.6%) | |||||
| 4 | 65 (33.2%) | 2 (2.9%) |
| Target Expression | Low-Grade, % | High-Grade, % | p-Value * | Target Expression | Low-Grade, % | High-Grade, % | p-Value |
|---|---|---|---|---|---|---|---|
| VEGFA | APOE | ||||||
| 1 | 2 (8.3%) | 33 (23.7%) | 0.146 | 1 | 3 (12.5%) | 33 (20.2%) | 0.630 |
| 2 | 5 (20.8%) | 37 (26.6%) | 2 | 6 (25.0%) | 35 (21.5%) | ||
| 3 | 7 (29.2%) | 38 (27.3%) | 3 | 9 (37.5%) | 44 (27.0%) | ||
| 4 | 10 (41.7%) | 31 (22.3%) | 4 | 6 (25.0%) | 51 (31.3%) | ||
| MMP9 | A1AT | ||||||
| 1 | 3 (12.5%) | 18 (11.8%) | 0.673 | 1 | 6 (27.3%) | 52 (33.1%) | 0.257 |
| 2 | 3 (12.5%) | 36 (23.5%) | 2 | 10 (45.5%) | 43 (27.4%) | ||
| 3 | 8 (33.3%) | 48 (31.4%) | 3 | 5 (22.7%) | 36 (22.9%) | ||
| 4 | 10 (41.7%) | 51 (33.3%) | 4 | 1 (4.5%) | 26 (16.6%) | ||
| MMP10 | SDC1 | ||||||
| 1 | 8 (33.3%) | 27 (17.6%) | 0.016 | 1 | 1 (4.0%) | 21 (13.2%) | 0.135 |
| 2 | 0 (0%) | 29 (19.0%) | 2 | 3 (12.0%) | 40 (25.2%) | ||
| 3 | 10 (41.7%) | 43 (28.1%) | 3 | 8 (32.0%) | 49 (30.8%) | ||
| 4 | 6 (25.0%) | 54 (35.3%) | 4 | 13 (52.0%) | 49 (30.8%) | ||
| PAI1 | ANG | ||||||
| 1 | 3 (12.5%) | 8 (5.3%) | 0.292 | 1 | 1 (4.2%) | 4 (2.5%) | 0.248 |
| 2 | 5 (20.8%) | 46 (30.3%) | 2 | 6 (25.0%) | 48 (29.4%) | ||
| 3 | 10 (41.7%) | 47 (30.9%) | 3 | 5 (20.8%) | 58 (35.6%) | ||
| 4 | 6 (25.0%) | 51 (33.6%) | 4 | 12 (50.0%) | 53 (32.5%) | ||
| IL8 | CD3+ | ||||||
| 1 | 8 (33.3%) | 52 (31.7%) | 0.592 | 1 | 6 (28.6%) | 32 (25.0%) | 0.904 |
| 2 | 5 (20.8%) | 40 (24.4%) | 2 | 5 (23.8%) | 32 (25.0%) | ||
| 3 | 3 (12.5%) | 35 (21.3%) | 3 | 4 (19.0%) | 33 (25.8%) | ||
| 4 | 8 (33.3%) | 37 (22.6%) | 4 | 6 (28.6%) | 31 (24.2%) | ||
| CA9 | CD68+ | ||||||
| 1 | 3 (13.0%) | 20 (12.0%) | 0.464 | 1 | 5 (22.7%) | 34 (25.4%) | 0.482 |
| 2 | 5 (21.7%) | 40 (24.1%) | 2 | 6 (27.3%) | 33 (24.6%) | ||
| 3 | 10 (43.5%) | 48 (28.9%) | 3 | 8 (36.4%) | 32 (23.9%) | ||
| 4 | 5 (21.7%) | 58 (34.9%) | 4 | 3 (13.6%) | 35 (26.1%) |
| Target Expression | Ta | Tis | T1 | T2 | ≥T3, N+ or M+ | p-Value |
|---|---|---|---|---|---|---|
| VEGFA | ||||||
| 1 | 7 (17.5%) | 4 (23.5%) | 11 (20.4%) | 8 (33.3%) | 5 (17.9%) | 0.854 |
| 2 | 10 (25.0%) | 6 (35.3%) | 12 (22.2%) | 7 (29.2%) | 7 (25.0%) | |
| 3 | 11 (27.5%) | 4 (23.5%) | 17 (31.5%) | 3 (12.5%) | 10 (35.7%) | |
| 4 | 12 (30.0%) | 3 (17.6%) | 14 (25.9%) | 6 (25.0%) | 6 (21.4%) | |
| MMP9 | ||||||
| 1 | 5 (11.6%) | 1 (7.1%) | 5 (8.9%) | 7 (24.1%) | 3 (8.6%) | 0.1389 |
| 2 | 6 (14.0%) | 7 (50.0%) | 16 (28.6%) | 3 (10.3%) | 7 (20.0%) | |
| 3 | 17 (39.5%) | 3 (21.4%) | 16 (28.6%) | 7 (24.1%) | 13 (37.1%) | |
| 4 | 15 (34.9%) | 3 (21.4%) | 19 (33.9%) | 12 (41.4%) | 12 (34.3%) | |
| MMP10 | ||||||
| 1 | 14 (32.6%) | 5 (35.7%) | 12 (21.8%) | 3 (10.3%) | 1 (2.8%) | 0.0103 |
| 2 | 3 (7.0%) | 4 (28.6%) | 9 (16.4%) | 6 (20.7%) | 7 (19.4%) | |
| 3 | 15 (34.9%) | 4 (28.6%) | 17 (30.9%) | 5 (17.2%) | 12 (33.3%) | |
| 4 | 11 (25.6%) | 1 (7.1%) | 17 (30.9%) | 15 (51.7%) | 16 (44.4%) | |
| PAI1 | ||||||
| 1 | 6 (14.3%) | 2 (13.3%) | 1 (1.8%) | 2 (7.1%) | 0 (0%) | 0.013 |
| 2 | 11 (26.2%) | 8 (53.3%) | 18 (32.1%) | 6 (21.4%) | 8 (22.9%) | |
| 3 | 13 (31.0%) | 5 (33.3%) | 14 (25.0%) | 8 (28.6%) | 17 (48.6%) | |
| 4 | 12 (28.6%) | 0 (0%) | 23 (41.1%) | 12 (42.9%) | 10 (28.6%) | |
| IL8 | ||||||
| 1 | 18 (39.1%) | 7 (43.8%) | 17 (28.3%) | 5 (17.2%) | 13 (35.1%) | 0.7881 |
| 2 | 8 (17.4%) | 3 (18.8%) | 17 (28.3%) | 8 (27.6%) | 9 (24.3%) | |
| 3 | 9 (19.6%) | 4 (25.0%) | 12 (20.0%) | 6 (20.7%) | 7 (18.9%) | |
| 4 | 11 (23.9%) | 2 (12.5%) | 14 (23.3%) | 10 (34.5%) | 8 (21.6%) | |
| CA9 | ||||||
| 1 | 3 (6.7%) | 0 (0%) | 6 (10.0%) | 8 (27.6%) | 6 (15.8%) | 0.0192 |
| 2 | 16 (35.6%) | 5 (29.4%) | 9 (15.0%) | 3 (10.3%) | 12 (31.6%) | |
| 3 | 13 (28.9%) | 7 (41.2%) | 18 (30.0%) | 7 (24.1%) | 13 (34.2%) | |
| 4 | 13 (28.9%) | 5 (29.4%) | 27 (45.0%) | 11 (37.9%) | 7 (18.4%) | |
| APOE | ||||||
| 1 | 9 (19.1%) | 2 (12.5%) | 14 (23.7%) | 6 (20.7%) | 5 (13.9%) | 0.967 |
| 2 | 12 (25.5%) | 3 (18.8%) | 10 (16.9%) | 8 (27.6%) | 8 (22.2%) | |
| 3 | 14 (29.8%) | 6 (37.5%) | 15 (25.4%) | 7 (24.1%) | 11 (30.6%) | |
| 4 | 12 (25.5%) | 5 (31.3%) | 20 (33.9%) | 8 (27.6%) | 12 (33.3%) | |
| A1AT | ||||||
| 1 | 16 (36.4%) | 4 (26.7%) | 22 (37.9%) | 6 (21.4%) | 10 (29.4%) | 0.403 |
| 2 | 18 (40.9%) | 4 (26.7%) | 15 (25.9%) | 8 (28.6%) | 8 (23.5%) | |
| 3 | 7 (15.9%) | 4 (26.7%) | 15 (25.9%) | 7 (25.0%) | 8 (23.5%) | |
| 4 | 3 (6.8%) | 3 (20.0%) | 6 (10.3%) | 7 (25.0%) | 8 (23.5%) | |
| SDC1 | ||||||
| 1 | 3 (7.1%) | 7 (36.8%) | 2 (3.4%) | 5 (17.2%) | 5 (14.3%) | <0.0001 |
| 2 | 5 (11.9%) | 5 (26.3%) | 10 (16.9%) | 7 (24.1%) | 16 (45.7%) | |
| 3 | 10 (23.8%) | 2 (10.5%) | 24 (40.7%) | 11 (37.9%) | 10 (28.6%) | |
| 4 | 24 (57.1%) | 5 (26.3%) | 23 (39.0%) | 6 (20.7%) | 4 (11.4%) | |
| ANG | ||||||
| 1 | 2 (4.3%) | 0 (0%) | 0 (0%) | 3 (10.3%) | 0 (0%) | 0.003 |
| 2 | 17 (37.0%) | 11 (64.7%) | 14 (23.7%) | 4 (13.8%) | 8 (22.2%) | |
| 3 | 13 (28.3%) | 1 (5.9%) | 22 (37.3%) | 10 (34.5%) | 17 (47.2%) | |
| 4 | 14 (30.4%) | 5 (29.4%) | 23 (39.0%) | 12 (41.4%) | 11 (30.6%) | |
| CD3+ | ||||||
| 1 | 12 (31.6%) | 1 (12.5%) | 14 (29.2%) | 1 (4.8%) | 10 (29.4%) | 0.444 |
| 2 | 8 (21.1%) | 1 (12.5%) | 10 (20.8%) | 8 (38.1%) | 10 (29.4%) | |
| 3 | 7 (18.4%) | 3 (37.5%) | 13 (27.1%) | 5 (23.8%) | 9 (26.5%) | |
| 4 | 11 (28.9%) | 3 (37.5%) | 11 (22.9%) | 7 (33.3%) | 5 (14.7%) | |
| CD68+ | ||||||
| 1 | 17 (42.5%) | 5 (55.6%) | 9 (18.0%) | 2 (8.7%) | 6 (17.7%) | 0.010 |
| 2 | 11 (27.5%) | 1 (11.1%) | 15 (30.0%) | 5 (21.7%) | 7 (20.6%) | |
| 3 | 9 (22.5%) | 3 (33.3%) | 13 (26.0%) | 7 (30.4%) | 8 (23.5%) | |
| 4 | 3 (7.5%) | 0 (0.0%) | 13 (26.0%) | 9 (39.1%) | 13 (38.2%) |
| Univariate Analyses 1 | Multivariate Analyses 1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | Median 2 | HR 3 | LCL | UCL | p4 | HR 3 | LCL | UCL | p4 | |
| Age (years) | ||||||||||
| ≤65 | 56 | 0.356 | 0.107 | 1.188 | 0.075 | 0.376 | 0.103 | 1.368 | 0.138 | |
| >65 | 153 | |||||||||
| Sex | ||||||||||
| Female | 46 | 1.222 | 0.513 | 2.908 | 0.646 | |||||
| Male | 163 | |||||||||
| Tumor Grade | ||||||||||
| Low | 28 | 0.489 | 0.115 | 2.072 | 0.314 | |||||
| High | 181 | |||||||||
| Tumor Stage | ||||||||||
| <T2 | 144 | 0.093 | 0.036 | 0.235 | <0.0001 | 0.107 | 0.034 | 0.337 | 0.0001 | |
| ≥T2 | 65 | 34 | ||||||||
| VEGFA | ||||||||||
| 1–2 | 79 | 1.000 | 0.375 | 2.670 | 0.999 | |||||
| 3–4 | 78 | |||||||||
| MMP9 | ||||||||||
| 1–2 | 84 | 0.950 | 0.425 | 2.125 | 0.900 | |||||
| 3–4 | 84 | |||||||||
| MMP10 | ||||||||||
| 1–2 | 85 | 46 | 1.224 | 0.547 | 2.741 | 0.616 | ||||
| 3–4 | 85 | |||||||||
| PAI1 | ||||||||||
| 1–2 | 86 | 0.708 | 0.304 | 1.646 | 0.416 | |||||
| 3–4 | 86 | |||||||||
| IL8 | ||||||||||
| 1–2 | 91 | 0.706 | 0.320 | 1.557 | 0.379 | |||||
| 3–4 | 90 | |||||||||
| CA9 | ||||||||||
| 1–2 | 91 | 1.897 | 0.789 | 4.563 | 0.141 | 1.348 | 0.536 | 3.390 | ||
| 3–4 | 91 | |||||||||
| APOE | ||||||||||
| 1–2 | 93 | 0.803 | 0.352 | 1.828 | 0.595 | |||||
| 3–4 | 87 | |||||||||
| A1AT | ||||||||||
| 1–2 | 84 | 0.475 | 0.213 | 1.062 | 0.060 | 0.750 | 0.292 | 1.926 | ||
| 3–4 | 83 | 37 | ||||||||
| SDC1 | ||||||||||
| 1–2 | 86 | 48 | 1.492 | 0.670 | 3.325 | 0.318 | ||||
| 3–4 | 90 | |||||||||
| ANG | ||||||||||
| 1–2 | 90 | 0.969 | 0.423 | 2.222 | 0.940 | |||||
| 3–4 | 90 | |||||||||
| CD3+ | ||||||||||
| 1–2 | 74 | 37 | 1.125 | 0.464 | 2.729 | 0.792 | ||||
| 3–4 | 74 | |||||||||
| CD68+ | ||||||||||
| 1–2 | 78 | 0.524 | 0.223 | 1.230 | 0.126 | 1.334 | 0.505 | 3.524 | 0.561 | |
| 3–4 | 77 | 37 | ||||||||
| CD68+/CD3+ | ||||||||||
| CD3+ 1–2 & CD68+ 1–2 | 40 | 0.434 | 0.112 | 1.685 | 0.258 | |||||
| CD3+ 1–2 & CD68+ 3–4 | 33 | 0.523 | 0.153 | 1.792 | ||||||
| CD3+ 3–4 & CD68+ 1–2 | 32 | 35 | 1.422 | 0.446 | 4.534 | |||||
| CD3+ 3–4 & CD68+ 3–4 | 41 | 48 | ||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furuya, H.; Chan, O.T.M.; Hokutan, K.; Tsukikawa, Y.; Chee, K.; Kozai, L.; Chan, K.S.; Dai, Y.; Wong, R.S.; Rosser, C.J. Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma. Diagnostics 2020, 10, 14. https://doi.org/10.3390/diagnostics10010014
Furuya H, Chan OTM, Hokutan K, Tsukikawa Y, Chee K, Kozai L, Chan KS, Dai Y, Wong RS, Rosser CJ. Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma. Diagnostics. 2020; 10(1):14. https://doi.org/10.3390/diagnostics10010014
Chicago/Turabian StyleFuruya, Hideki, Owen T.M. Chan, Kanani Hokutan, Yutaro Tsukikawa, Keanu Chee, Landon Kozai, Keith S. Chan, Yunfeng Dai, Regan S. Wong, and Charles J. Rosser. 2020. "Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma" Diagnostics 10, no. 1: 14. https://doi.org/10.3390/diagnostics10010014
APA StyleFuruya, H., Chan, O. T. M., Hokutan, K., Tsukikawa, Y., Chee, K., Kozai, L., Chan, K. S., Dai, Y., Wong, R. S., & Rosser, C. J. (2020). Prognostic Significance of Lymphocyte Infiltration and a Stromal Immunostaining of a Bladder Cancer Associated Diagnostic Panel in Urothelial Carcinoma. Diagnostics, 10(1), 14. https://doi.org/10.3390/diagnostics10010014






