Photobiomodulation Therapy Applied after 6 Months for the Management of a Severe Inferior Alveolar Nerve Injury
Abstract
1. Introduction
2. Case Report
- Complete numbness of the gingiva and the mucosa from the extracted wisdom tooth until the right mandibular canine.
- Robust contraction of the inferior lip.
- Deep cutaneous pain on the inferior lip.
- Complete numbness of more than half of the right chin.
- Generalized and vague pain in the right mandibular arch.
2.1. Diagnosis
2.2. Treatment
2.3. Symptoms Assessment during the Treatment
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ciardelli, G.; Chiono, V. Materials for Peripheral Nerve Regeneration. Macromol. Biosci. 2005, 6, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Grinsell, D.; Keating, C.P. Peripheral Nerve Reconstruction after Injury: A Review of Clinical and Experimental Therapies. BioMed Res. Int. 2014, 2014, 1–13. [Google Scholar] [CrossRef]
- Cheung, L.K.; Leung, Y.Y.; Chow, L.K.; Wong, M.C.; Chan, E.K.; Fok, Y.H. Incidence of neurosensory deficits and recovery after lower third molar surgery: A prospective clinical study of 4338 cases. Int. J. Oral Maxillofac. Surg. 2010, 39, 320–326. [Google Scholar] [CrossRef]
- Grötz, K.A.; Al-Nawas, B.; De Aguiar, E.G.; Schulz, A.; Wagner, W. Treatment of injuries to the inferior alveolar nerve after endodontic procedures. Clin. Oral Investig. 1998, 2, 73–76. [Google Scholar] [CrossRef]
- Shavit, I.; Juodzbalys, G. Inferior Alveolar Nerve Injuries Following Implant Placement—Importance of Early Diagnosis and Treatment: A Systematic Review. J. Oral Maxillofac. Res. 2014, 5, e2. [Google Scholar] [CrossRef]
- Grant, G.A.; Goodkin, R.; Kliot, M. Evaluation and Surgical Management of Peripheral Nerve Problems. Neurosurgery 1999, 44, 825–839. [Google Scholar] [CrossRef]
- Argyriou, A.A.; Polychronopoulos, P.; Iconomou, G.; Chroni, E.; Kalofonos, H. A review on oxaliplatin-induced peripheral nerve damage. Cancer Treat. Rev. 2008, 34, 368–377. [Google Scholar] [CrossRef]
- Borisov, A.B.; Carlson, B.M. Cell death in denervated skeletal muscle is distinct from classical apoptosis. Anat. Rec. 2000, 258, 305–318. [Google Scholar] [CrossRef]
- Higashino, K.; Matsuura, T.; Suganuma, K.; Yukata, K.; Nishisho, T.; Yasui, N. Early changes in muscle atrophy and muscle fiber type conversion after spinal cord transection and peripheral nerve transection in rats. J. Neuroeng. Rehabil. 2013, 10, 46–47. [Google Scholar] [CrossRef]
- Schmalbruch, H.; Lewis, D. Dynamics of nuclei of muscle fibers and connective tissue cells in normal and denervated rat muscles. Muscle Nerve 2000, 23, 617–626. [Google Scholar] [CrossRef]
- Selcen, D. Muscle diseases. In Goldman Cecil Medicine, 25th ed.; Goldman, L., Schafer, A.I., Eds.; Elsevier Saunders: Philadelphia, PA, USA, 2016; pp. 2537–2547. [Google Scholar]
- Hussain, G.; Wang, J.; Rasul, A.; Anwar, H.; Qasim, M.; Zafar, S.; Aziz, N.; Razzaq, A.; Hussain, R.; De Aguilar, J.-L.G.; et al. Current Status of Therapeutic Approaches against Peripheral Nerve Injuries: A Detailed Story from Injury to Recovery. Int. J. Biol. Sci. 2020, 16, 116–134. [Google Scholar] [CrossRef] [PubMed]
- Mandelbaum-Livnat, M.M.; Almog, M.; Nissan, M.; Loeb, E.; Shapira, Y.; Rochkind, S. Photobiomodulation Triple Treatment in Peripheral Nerve Injury: Nerve and Muscle Response. Photomed. Laser Surg. 2016, 34, 638–645. [Google Scholar] [CrossRef] [PubMed]
- El Mobadder, M.; Farhat, F.; El Mobadder, W.; Nammour, S. Photobiomodulation Therapy in the Treatment of Oral Mucositis, Dysphagia, Oral Dryness, Taste Alteration, and Burning Mouth Sensation Due to Cancer Therapy: A Case Series. Int. J. Environ. Res. Public Health 2019, 16, 4505. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R.; Liebert, A. Photobiomodulation Therapy Mechanisms beyond Cytochrome c Oxidase. Photobiomodul. Photomed. Laser Surg. 2021, 39, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Zecha, J.A.E.M.; Raber-Durlacher, J.E.; Nair, R.G.; Epstein, J.B.; Elad, S.; Hamblin, M.R.; Barasch, A.; Migliorati, C.A.; Milstein, D.M.J.; Genot, M.-T.; et al. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: Part 2: Proposed applications and treatment protocols. Support. Care Cancer 2016, 24, 2793–2805. [Google Scholar] [CrossRef]
- Zecha, J.A.E.M.; Raber-Durlacher, J.E.; Nair, R.G.; Epstein, J.B.; Sonis, S.T.; Elad, S.; Hamblin, M.R.; Barasch, A.; Migliorati, C.A.; Milstein, D.M.J.; et al. Low-level laser therapy/photobiomodulation in the management of side effects of chemoradiation therapy in head and neck cancer: Part 1: Mechanisms of action, dosimetric, and safety considerations. Support. Care Cancer 2016, 24, 2781–2792. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, Z.; Zhu, Z.; Liang, Z.; Zuo, X.; Ju, C.; Song, Z.; Li, X.; Hu, X.; Wang, Z. Photobiomodulation Promotes Repair Following Spinal Cord Injury by Regulating the Transformation of A1/A2 Reactive Astrocytes. Front. Neurosci. 2021, 15, 768262. [Google Scholar] [CrossRef]
- Lopes-Martins, R.A.; Leonardo, P.S.; Bjordal, J.M.; Marcos, R.L. Photobiomodulation: Inhibition or Resolution of the Inflam-matory Process? Photobiomodul. Photomed. Laser Surg. 2020, 38, 453–454. [Google Scholar] [CrossRef]
- Scalon, D.; Picada, J.N.; de Sousa, J.T.; da Silva, A.T.; Colares, J.R.; Marroni, N.A.P. Photobiomodulation intervention improves oxidative, inflammatory, and morphological parameters of skeletal muscle in cirrhotic Wistar rats. Lasers Med. Sci. 2021, 1–10. [Google Scholar] [CrossRef]
- Zuniga, J.R.; Radwan, A.M. Classification of nerve injuries. Trigeminal Nerve Injuries 2013, 17–25. [Google Scholar]
- On, S.-W.; Cho, S.-W.; Byun, S.-H.; Yang, B.-E. Clinical Significance of Intraoperative Exposure of Inferior Alveolar Nerve during Surgical Extraction of the Mandibular Third Molar in Nerve Injury. J. Clin. Med. 2021, 10, 4379. [Google Scholar] [CrossRef]
- Pogrel, M.A.; Bryan, J.; Reigzi, J. Nerve damage associated: With inferior alveolar nerve blocks. J. Am. Dent. Assoc. 1995, 126, 1150–1155. [Google Scholar] [CrossRef]
- Lopes, V.; Mumenya, R.; Feinmann, C.; Harris, M. Third molar surgery: An audit of the indications for surgery, post-operative complaints and patient satisfaction. Br. J. Oral Maxillofac. Surg. 1995, 33, 33–35. [Google Scholar] [CrossRef]
- Smith, W. The relative risk of neurosensory deficit following removal of mandibular third molar teeth: The influence of radiography and surgical technique. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2013, 115, 18–24. [Google Scholar] [CrossRef]
- Montazeri, K.; Farhadi, M.; Fekrazad, R.; Akbarnejad, Z.; Chaibakhsh, S.; Mahmoudian, S. Transcranial photobiomodulation in the management of brain disorders. J. Photochem. Photobiol. B Biol. 2021, 221, 112207. [Google Scholar] [CrossRef] [PubMed]
- Hanna, R.; Dalvi, S.; Bensadoun, R.J.; Raber-Durlacher, J.E.; Benedicenti, S. Role of Photobiomodulation Therapy in Neurological Primary Burning Mouth Syndrome. A Systematic Review and Meta-Analysis of Human Randomised Controlled Clinical Trials. Pharmaceutics 2021, 13, 1838. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Wu, C.; Tucker, L.; Dong, Y.; Li, Y.; Xu, P.; Zhang, Q. Photobiomodulation Therapy Attenuates Anxious-Depressive-Like Behavior in the TgF344 Rat Model. J. Alzheimers Dis. 2021, 83, 1415–1429. [Google Scholar] [CrossRef]
- Rochkind, S. Photobiomodulation in Neuroscience: A Summary of Personal Experience. Photomed. Laser Surg. 2017, 35, 604–615. [Google Scholar] [CrossRef] [PubMed]
- Silveira, P.C.L.; Da Silva, L.A.; Pinho, C.A.; De Souza, P.S.; Ronsani, M.M.; Scheffer, D.D.L.; Pinho, R. Effects of low-level laser therapy (GaAs) in an animal model of muscular damage induced by trauma. Lasers Med. Sci. 2012, 28, 431–436. [Google Scholar] [CrossRef]
- Barez, M.M.; Tajziehchi, M.; Heidari, M.H.; Bushehri, A.; Moayer, F.; Mansouri, N.; Naini, N.S.; Movafagh, A. Stimulation Effect of Low-Level Laser Therapy on Sciatic Nerve Regeneration in Rat. J. Lasers Med. Sci. 2017, 8, S32–S37. [Google Scholar] [CrossRef][Green Version]
| Sunderland Classification | Causes | Recovery | Pathophysiology | Surgical Intercessions |
|---|---|---|---|---|
| Grade IV | Nerve crush | Incomplete and variable—depending on the injury and treatment—months to years | Axon with myelin sheath, endoneurium disconnected | Typically required; procedure depends upon findings |
| Area | Classification | Description |
|---|---|---|
| Gingiva and mucosa from the second molar to the canine | S0 | No sensation at all |
| Cheek | S0 | No sensation at all |
| Chin | S0 | No sensation at all |
| Lower lip | S1 | Deep cutaneous pain in an autonomous zone |
| Area | Description |
|---|---|
| Teeth from the second right molar to the right canine | Cold test result: A mean value delay of 3.2 s between the stimulus and the sensation of pain |
| Right depressor labii inferioris muscle | Sensation of contraction with normal function |
| Right depressor anguli oris | |
| Right modiolus | |
| Right mentalis |
| Parameters | Values |
|---|---|
| Wavelength | 635 nanometers |
| Output Power | 0.1 Watt |
| Fiber diameter | 600 µm (0.6 mm) |
| Mode of irradiation | Continuous and contact mode |
| Time of irradiation | 40 s |
| Energy delivered | 4 Joules |
| Delivered energy related to fiber diameter | 1415 J/cm2 |
| Number of sessions in total | 42 sessions |
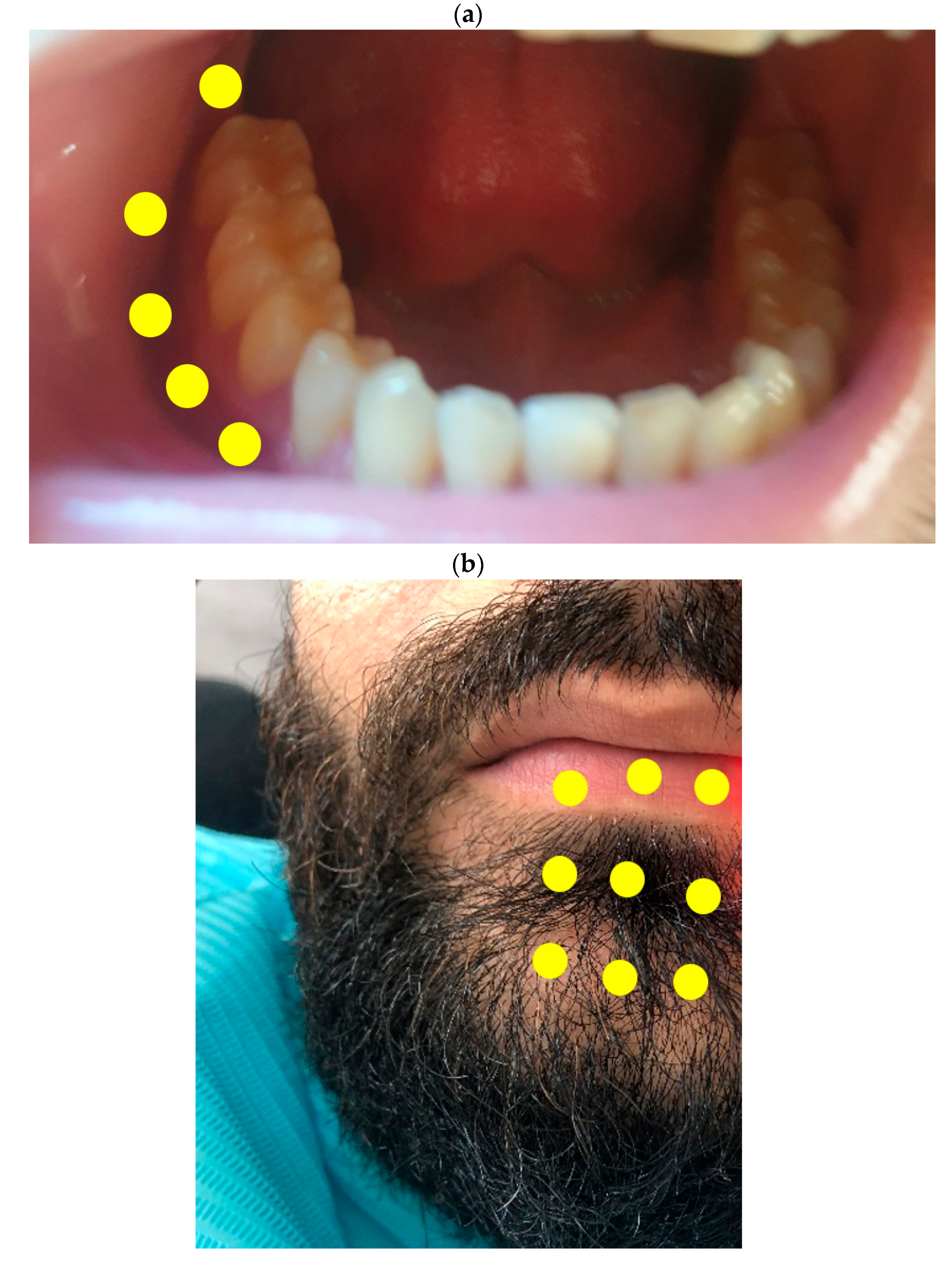
| Irradiation area | Intraorally and extraorally (Figure 1) |
| Symptoms in the Concerned Area | Follow-Up Period after Each Session | |||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | After S1 | After S2 | After S3 | After S4 | After S5 | After S6 | After S7 | |
| Gingiva and mucosa from the second molar to the canine | No sensation at all | Abnormal sensation with pain on touching | Light abnormal sensation on touching | |||||
| Cheek | No sensation at all | Abnormal sensation with deep cutaneous pain | Normal sensation | |||||
| Lower lip | Deep cutaneous pain in an autonomous zone | Abnormal sensation with deep cutaneous pain | Abnormal sensation only on touching | Normal sensation | ||||
| Cold Test on the tooth | 3 s delay with strong pain | Normal cold test | ||||||
| Muscle contraction of the mental region (chin) | Severe contraction | Moderate contraction | No contraction | |||||
| Muscle contraction of the lip | Severe sensation of contraction | Moderate sensation of contraction | Light sensation of contraction | No contraction | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
El Mobadder, M.; Nammour, S.; Ortega, M.; Grzech-Leśniak, K. Photobiomodulation Therapy Applied after 6 Months for the Management of a Severe Inferior Alveolar Nerve Injury. Life 2021, 11, 1420. https://doi.org/10.3390/life11121420
El Mobadder M, Nammour S, Ortega M, Grzech-Leśniak K. Photobiomodulation Therapy Applied after 6 Months for the Management of a Severe Inferior Alveolar Nerve Injury. Life. 2021; 11(12):1420. https://doi.org/10.3390/life11121420
Chicago/Turabian StyleEl Mobadder, Marwan, Samir Nammour, Marlin Ortega, and Kinga Grzech-Leśniak. 2021. "Photobiomodulation Therapy Applied after 6 Months for the Management of a Severe Inferior Alveolar Nerve Injury" Life 11, no. 12: 1420. https://doi.org/10.3390/life11121420
APA StyleEl Mobadder, M., Nammour, S., Ortega, M., & Grzech-Leśniak, K. (2021). Photobiomodulation Therapy Applied after 6 Months for the Management of a Severe Inferior Alveolar Nerve Injury. Life, 11(12), 1420. https://doi.org/10.3390/life11121420









