Simple, Reproducible Synthesis of Pure Monohydrocalcite with Low Mg Content
Abstract
1. Introduction
2. Materials and Methods
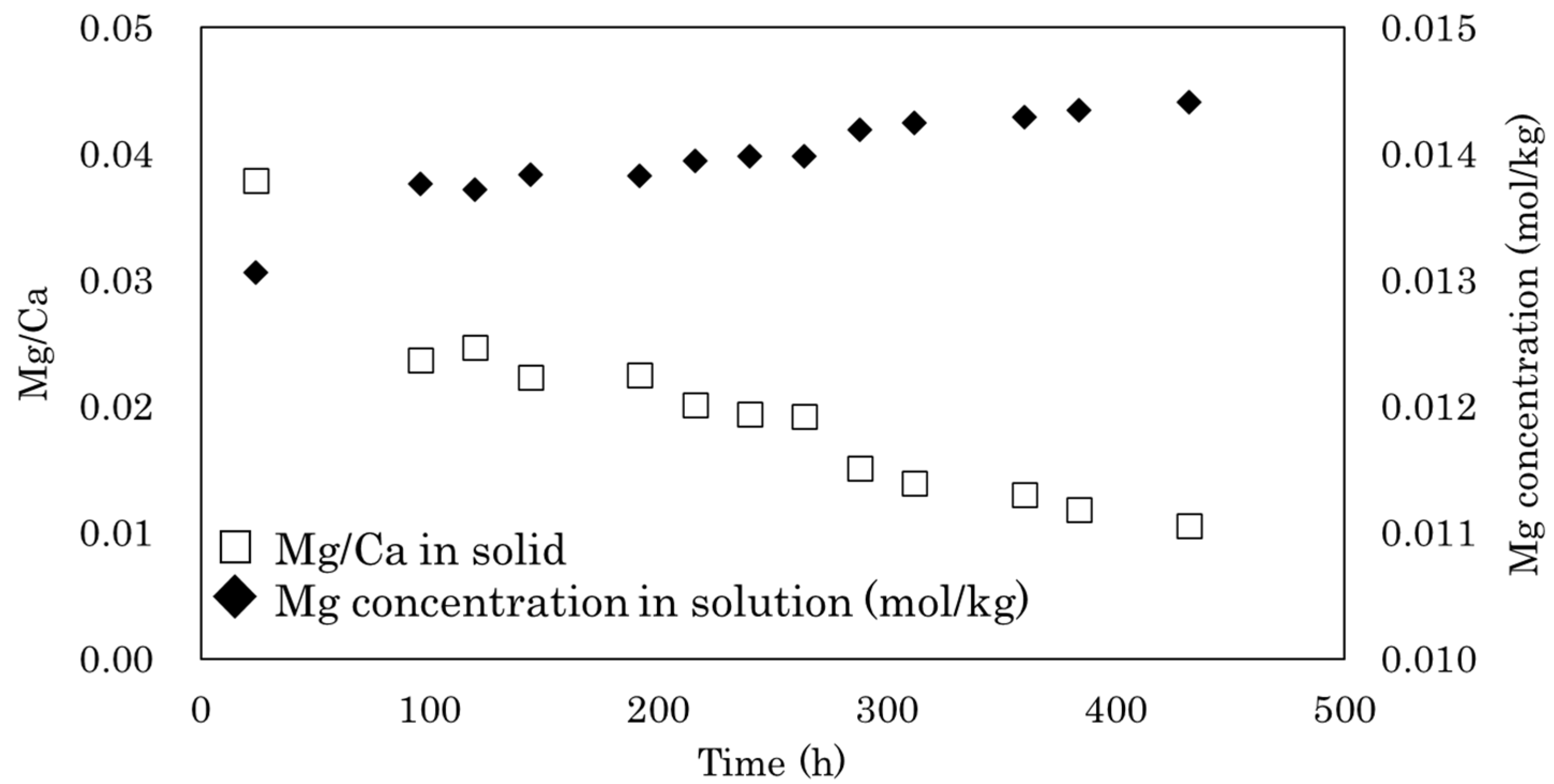
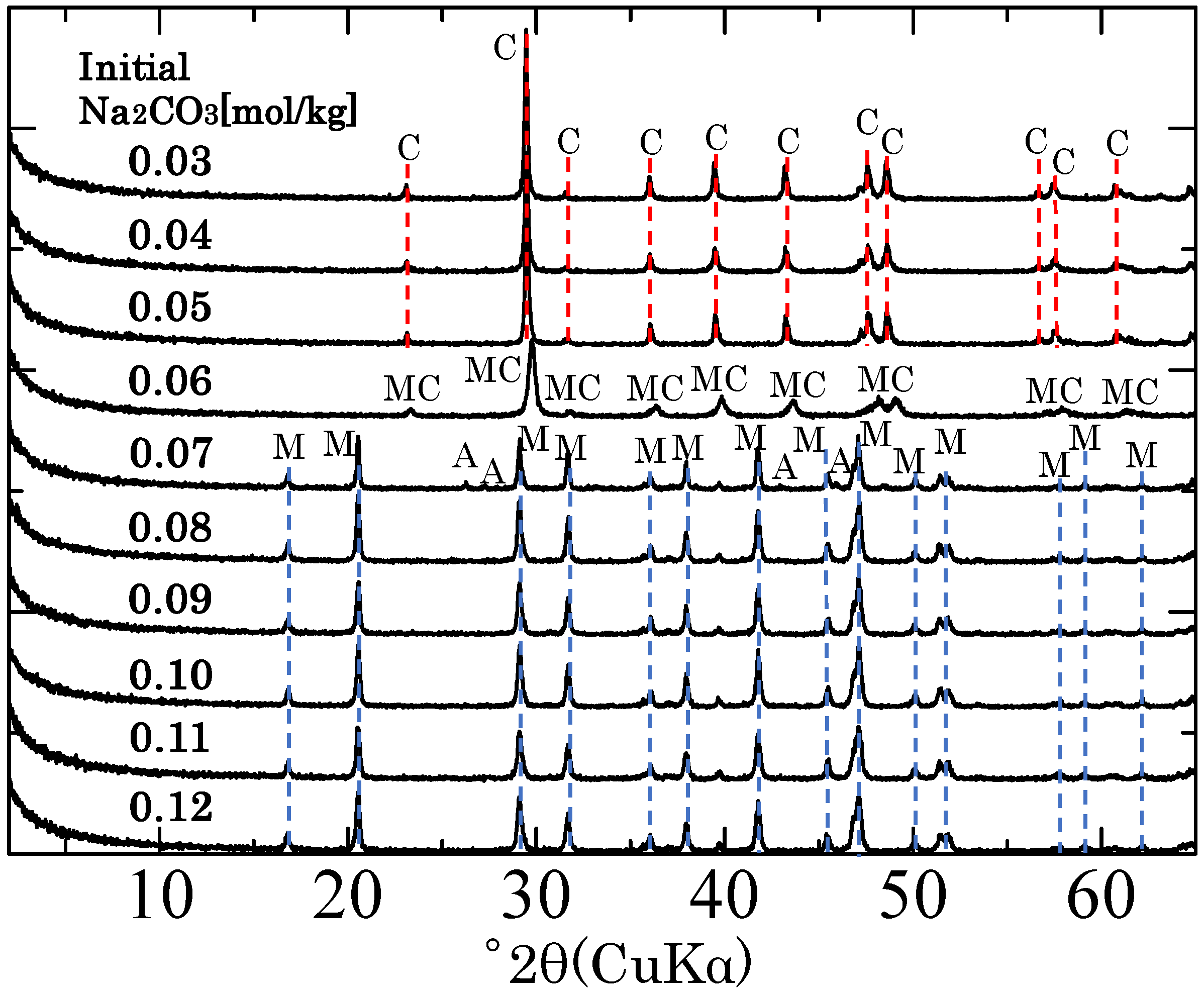
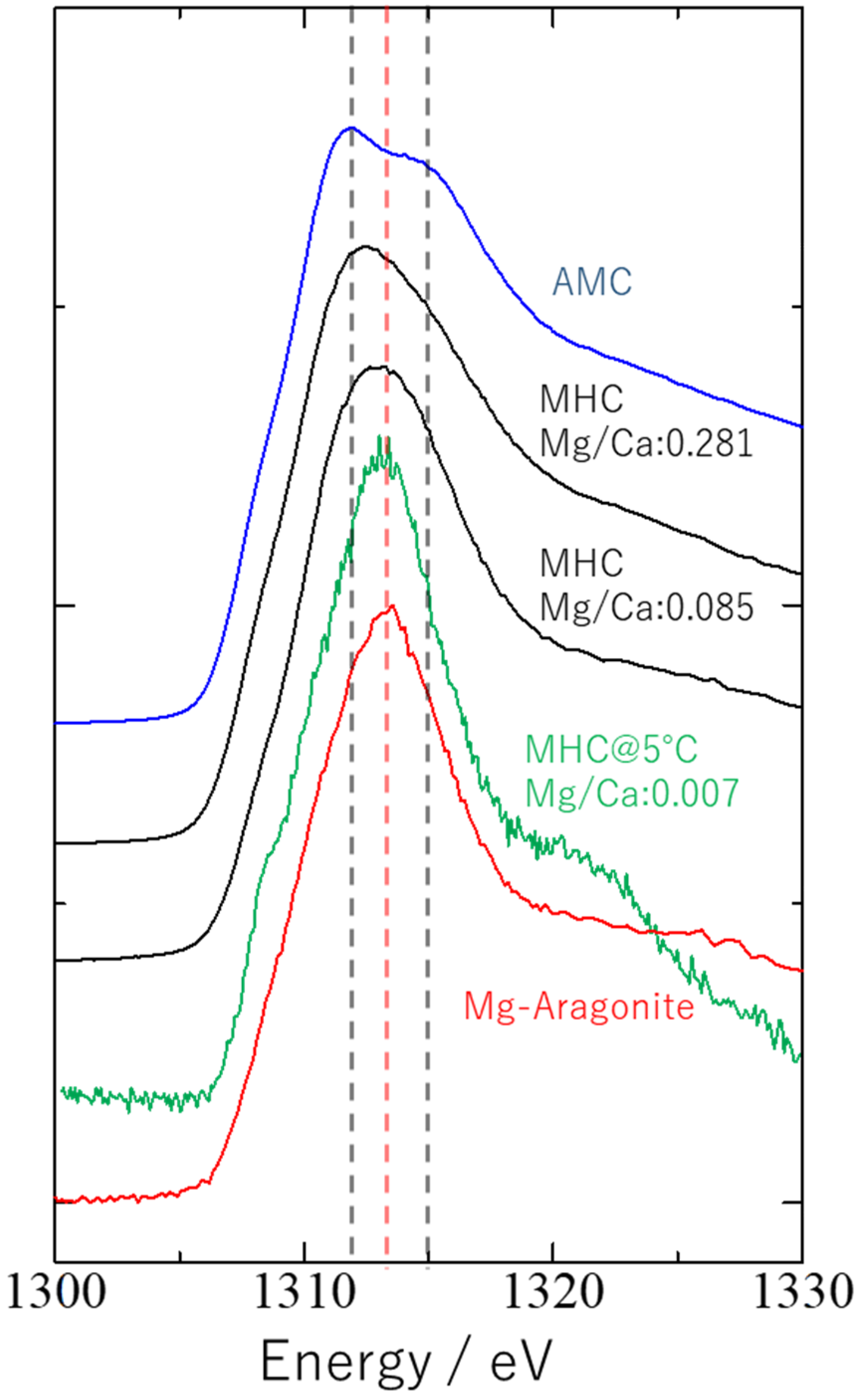
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Fukushi, K.; Munemoto, T.; Sakai, M.; Yagi, S. Monohydrocalcite: A Promising Remediation Material for Hazardous Anions. Sci. Technol. Adv. Mater. 2011, 12, 64702. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Blanco, J.D.; Shaw, S.; Bots, P.; Roncal-Herrero, T.; Benning, L.G. The Role of Mg in the Crystallization of Monohydrocalcite. Geochim. Cosmochim. Acta 2014, 127, 204–220. [Google Scholar] [CrossRef]
- Taylor, G.F. The Occurrence of Monohydrocalcite in Two Small Lakes in the South-East of South Australia. Am. Mineral. 1975, 60, 690–697. [Google Scholar]
- Last, F.M.; Last, W.M.; Halden, N.M. Carbonate Microbialites and Hardgrounds from Manito Lake, an Alkaline, Hypersaline Lake in the Northern Great Plains of Canada. Sediment. Geol. 2010, 225, 34–49. [Google Scholar] [CrossRef]
- Fukushi, K.; Matsumiya, H. Control of Water Chemistry in Alkaline Lakes: Solubility of Monohydrocalcite and Amorphous Magnesium Carbonate in CaCl2–MgCl2–Na2CO3 Solutions. ACS Earth Space Chem. 2018, 2, 735–744. [Google Scholar] [CrossRef]
- Fukushi, K.; Sakai, M.; Munemoto, T.; Yokoyama, Y.; Takahashi, Y. Arsenate Sorption on Monohydrocalcite by Coprecipitation during Transformation to Aragonite. J. Hazard. Mater. 2016, 304, 110–117. [Google Scholar] [CrossRef]
- Munemoto, T.; Fukushi, K.; Kanzaki, Y.; Murakami, T. Redistribution of Pb during Transformation of Monohydrocalcite to Aragonite. Chem. Geol. 2014, 387, 133–143. [Google Scholar] [CrossRef]
- Yagi, S.; Fukushi, K. Removal of Phosphate from Solution by Adsorption and Precipitation of Calcium Phosphate onto Monohydrocalcite. J. Colloid Interface Sci. 2012, 384, 128–136. [Google Scholar] [CrossRef]
- Sakai, M.; Munemoto, T.; Fukushi, K. Arsenate Uptake by Monohydrocalcite. Dep. Bull. Pap. K-INET Kanazawa Univ. 2010, 66–68, In Japanese. Available online: https://kanazawa-u.repo.nii.ac.jp/?action=pages_view_main&active_action=repository_view_main_item_detail&item_id=29857&item_no=1&page_id=13&block_id=21 (accessed on 13 April 2020).
- Yagi, S.; Fukushi, K. Phosphate Sorption on Monohydrocalcite. J. Mineral. Petrol. Sci. 2011, 106, 109–114. [Google Scholar] [CrossRef]
- Nishiyama, R.; Munemoto, T.; Fukushi, K. Formation Condition of Monohydrocalcite from CaCl2-MgCl2-Na2CO3 Solutions. Geochim. Cosmochim. Acta 2013, 100, 217–231. [Google Scholar] [CrossRef]
- Blue, C.R.; Giuffre, A.; Mergelsberg, S.; Han, N.; De Yoreo, J.J.; Dove, P.M. Chemical and Physical Controls on the Transformation of Amorphous Calcium Carbonate into Crystalline CaCO3 polymorphs. Geochim. Cosmochim. Acta 2017, 196, 179–196. [Google Scholar] [CrossRef]
- Kimura, T.; Koga, N. Monohydrocalcite in Comparison with Hydrated Amorphous Calcium Carbonate: Precipitation Condition and Thermal Behavior. Cryst. Growth Des. 2011, 11, 3877–3884. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Yao, Q.-Z.; Zhou, G.-T.; Fu, S.-Q. Transformation of Amorphous Calcium Carbonate into Monohydrocalcite in Aqueous Solution: A Biomimetic Mineralization Study. Eur. J. Mineral. 2015, 27, 717–729. [Google Scholar] [CrossRef]
- Fukushi, K.; Suzuki, Y.; Kawano, J.; Ohno, T.; Ogawa, M.; Yaji, T.; Takahashi, Y. Speciation of Magnesium in Monohydrocalcite: XANES, Ab Initio and Geochemical Modeling. Geochim. Cosmochim. Acta 2017, 213, 457–474. [Google Scholar] [CrossRef]
- Lin, C.Y.; Turchyn, A.V.; Steiner, Z.; Bots, P.; Lampronti, G.I.; Tosca, N.J. The Role of Microbial Sulfate Reduction in Calcium Carbonate Polymorph Selection. Geochim. Cosmochim. Acta 2018, 237, 184–204. [Google Scholar] [CrossRef]
- Morse, J.W.; Bender, M.L. Partition coefficients in calcite: Examination of factors influencing the validity of experimental results and their application to natural systems. Chem. Geol. 1990, 82, 265–277. [Google Scholar] [CrossRef]
- Zhang, Z.; Xie, Y.; Xu, X.; Pan, H.; Tang, R. Transformation of Amorphous Calcium Carbonate into Aragonite. J. Cryst. Growth 2012, 343, 62–67. [Google Scholar] [CrossRef]
- Harrison, A.L.; Mavromatis, V.; Oelker, E.H.; Bénézeth, P. Solubility of the hydrated Mg-carbonates nesquehonite and dypingite from 5 to 35 °C: Implications for CO2 storage and the relative stability of Mg-carbonates. Chem. Geol. 2019, 504, 123–135. [Google Scholar] [CrossRef]
- Munemoto, T.; Fukushi, K. Transformation Kinetics of Monohydrocalcite to Aragonite in Aqueous Solutions. J. Mineral. Petrol. Sci. 2008, 103, 345–349. [Google Scholar] [CrossRef]
- Scherrer, P. Estimation of the Size and Internal Structure of Colloidal Particles by Means of Röntgen. Nachr. Ges. Wiss. Göttingen. 1918, 2, 96–100. [Google Scholar]

| Batch# | Temperature (°C) | Initial Concentrations (mol/kg) | Reacted Solution Concentrations (mol/kg) | Mg/Ca in Solids | Partitioning Coefficient log D | Mineralogy | Crystallite Size | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca | Mg | CO3 | Ca | Mg | (nm) | ||||||
| 1 | 5 | 0.047 | 0.0095 | 0.029 | 1.6 × 10−2 | 1.0 × 10−2 | - | - | Calcite | - | This study |
| 2 | 5 | 0.050 | 0.010 | 0.040 | 1.1 × 10−2 | 1.0 × 10−2 | - | - | Calcite | - | This study |
| 3 | 5 | 0.051 | 0.010 | 0.050 | 2.8 × 10−3 | 8.8 × 10−3 | - | - | Calcite | - | This study |
| 4 | 5 | 0.050 | 0.010 | 0.060 | 4.1 × 10−4 | 3.7 × 10−3 | - | - | Mg-Calcite | - | This study |
| 5 | 5 | 0.051 | 0.010 | 0.071 | 1.2 × 10−4 | 9.4 × 10−3 | - | - | MHC, Ara | - | This study |
| 6 | 5 | 0.050 | 0.010 | 0.080 | 1.1 × 10−4 | 9.5 × 10−3 | 0.0077 | −4.1 | MHC | 37.0 ± 7.4 | This study |
| 7 | 5 | 0.050 | 0.010 | 0.089 | 8.4 × 10−5 | 1.0 × 10−2 | 0.0083 | −4.2 | MHC | 36.3 ± 4.5 | This study |
| 8 | 5 | 0.050 | 0.010 | 0.10 | 6.7 × 10−5 | 9.8 × 10−3 | - | - | MHC | 38.2 ± 2.0 | This study |
| 9 | 5 | 0.050 | 0.010 | 0.11 | 6.5 × 10−5 | 9.5 × 10−3 | 0.0101 | −4.2 | MHC | 37.2 ± 3.1 | This study |
| 10 | 5 | 0.050 | 0.010 | 0.12 | 6.3 × 10−5 | 9.7 × 10−3 | 0.0102 | −4.2 | MHC | 38.5 ± 3.9 | This study |
| N1 | 25 | 0.052 | 0.010 | 0.060 | 8.1 × 10−4 | 7.4 × 10−3 | - | - | Ara, Cal | - | Nishiyama et al. [11] |
| N2 | 25 | 0.052 | 0.010 | 0.077 | 1.6 × 10−4 | 9.1 × 10−3 | 0.02 † | −3.5 | MHC | - | Nishiyama et al. [11] |
| N3 | 25 | 0.052 | 0.010 | 0.120 | 1.9 × 10−4 | 7.3 × 10−3 | 0.06 † | −2.8 | MHC | - | Nishiyama et al. [11] |
| F1 | 25 | 0.050 | 0.035 | 0.090 | 2.4 × 10−4 | 1.9 × 10−2 | 0.085 ± 0.003 | −3.0 | MHC | 28.8 ± 1.2 | Fukushi et al. [15] |
| F2 | 25 | 0.050 | 0.015 | 0.100 | 1.3 × 10−4 | 6.7 × 10−3 | 0.136 ± 0.002 | −2.6 | MHC | 21.3 ± 2.1 | Fukushi et al. [15] |
| F3 | 25 | 0.160 | 0.040 | 0.200 | 3.5 × 10−4 | 2.7 × 10−2 | 0.281 ± 0.005 | −2.4 | MHC | 11.7 ± 0.9 | Fukushi et al. [15] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kitajima, T.; Fukushi, K.; Yoda, M.; Takeichi, Y.; Takahashi, Y. Simple, Reproducible Synthesis of Pure Monohydrocalcite with Low Mg Content. Minerals 2020, 10, 346. https://doi.org/10.3390/min10040346
Kitajima T, Fukushi K, Yoda M, Takeichi Y, Takahashi Y. Simple, Reproducible Synthesis of Pure Monohydrocalcite with Low Mg Content. Minerals. 2020; 10(4):346. https://doi.org/10.3390/min10040346
Chicago/Turabian StyleKitajima, Takuma, Keisuke Fukushi, Masahiro Yoda, Yasuo Takeichi, and Yoshio Takahashi. 2020. "Simple, Reproducible Synthesis of Pure Monohydrocalcite with Low Mg Content" Minerals 10, no. 4: 346. https://doi.org/10.3390/min10040346
APA StyleKitajima, T., Fukushi, K., Yoda, M., Takeichi, Y., & Takahashi, Y. (2020). Simple, Reproducible Synthesis of Pure Monohydrocalcite with Low Mg Content. Minerals, 10(4), 346. https://doi.org/10.3390/min10040346






