Extrapolation of Leaf Measurements to Obtain the Whole-Canopy Transpiration of C3 and C4 Xerophytic Shrubs
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Plant Description
2.2. Methods
2.2.1. Experimental Design
2.2.2. Gas Exchange Measurements
2.2.3. Transpiration Calculation Procedures
2.2.4. Precipitation and Meteorological Variables
2.2.5. Statistical Analysis
3. Results
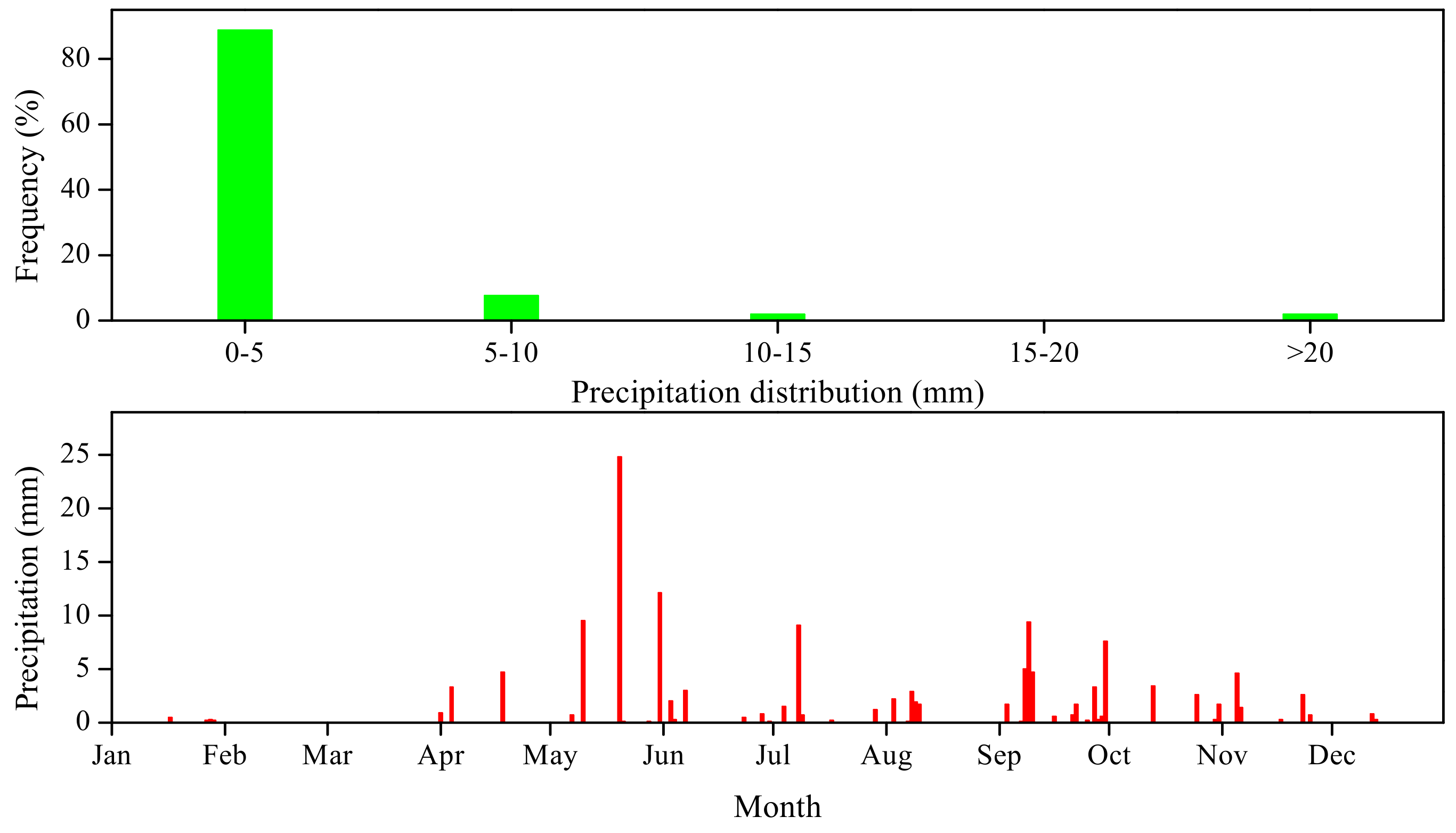
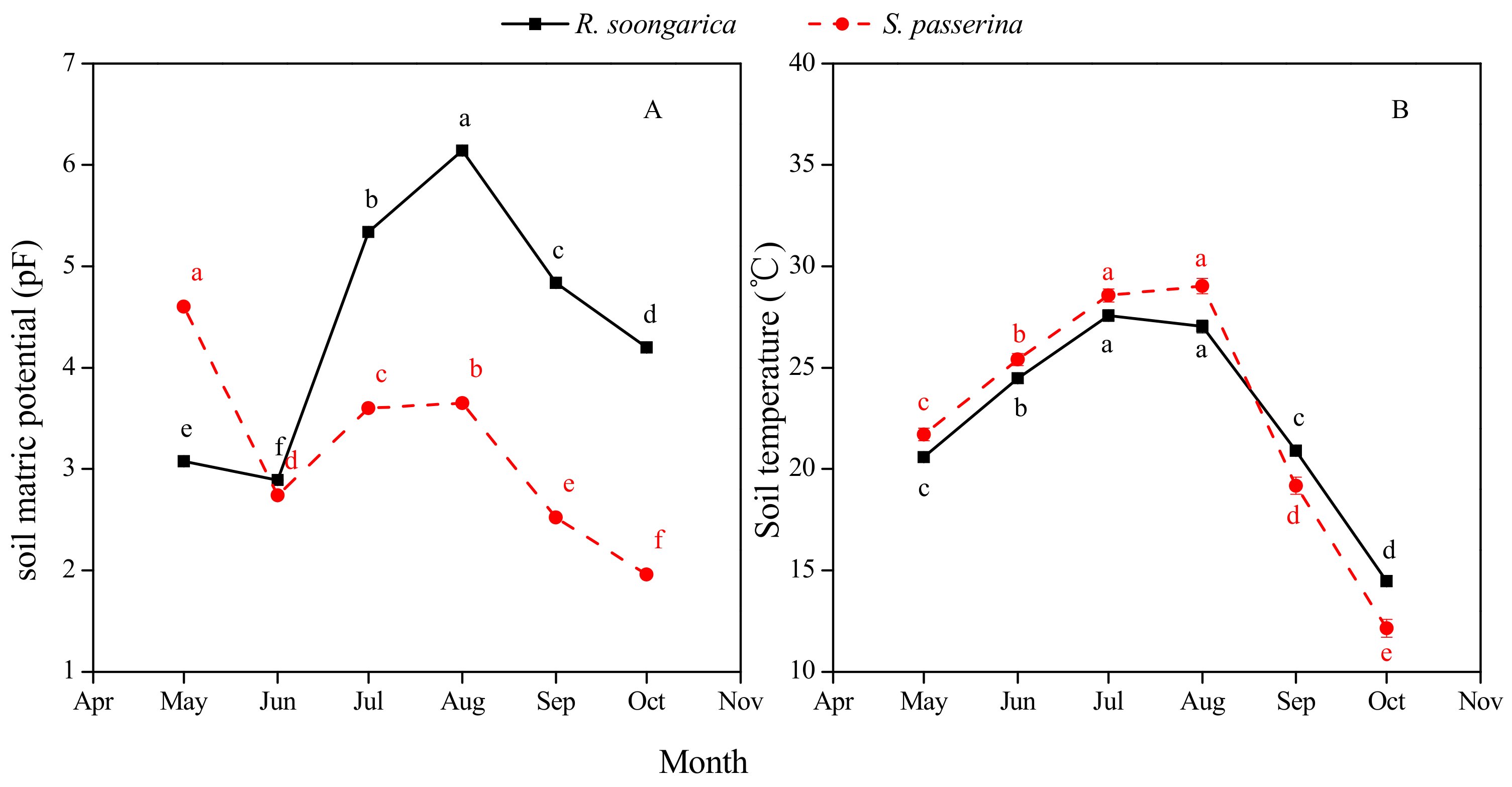
3.1. Precipitation and Soil Environment
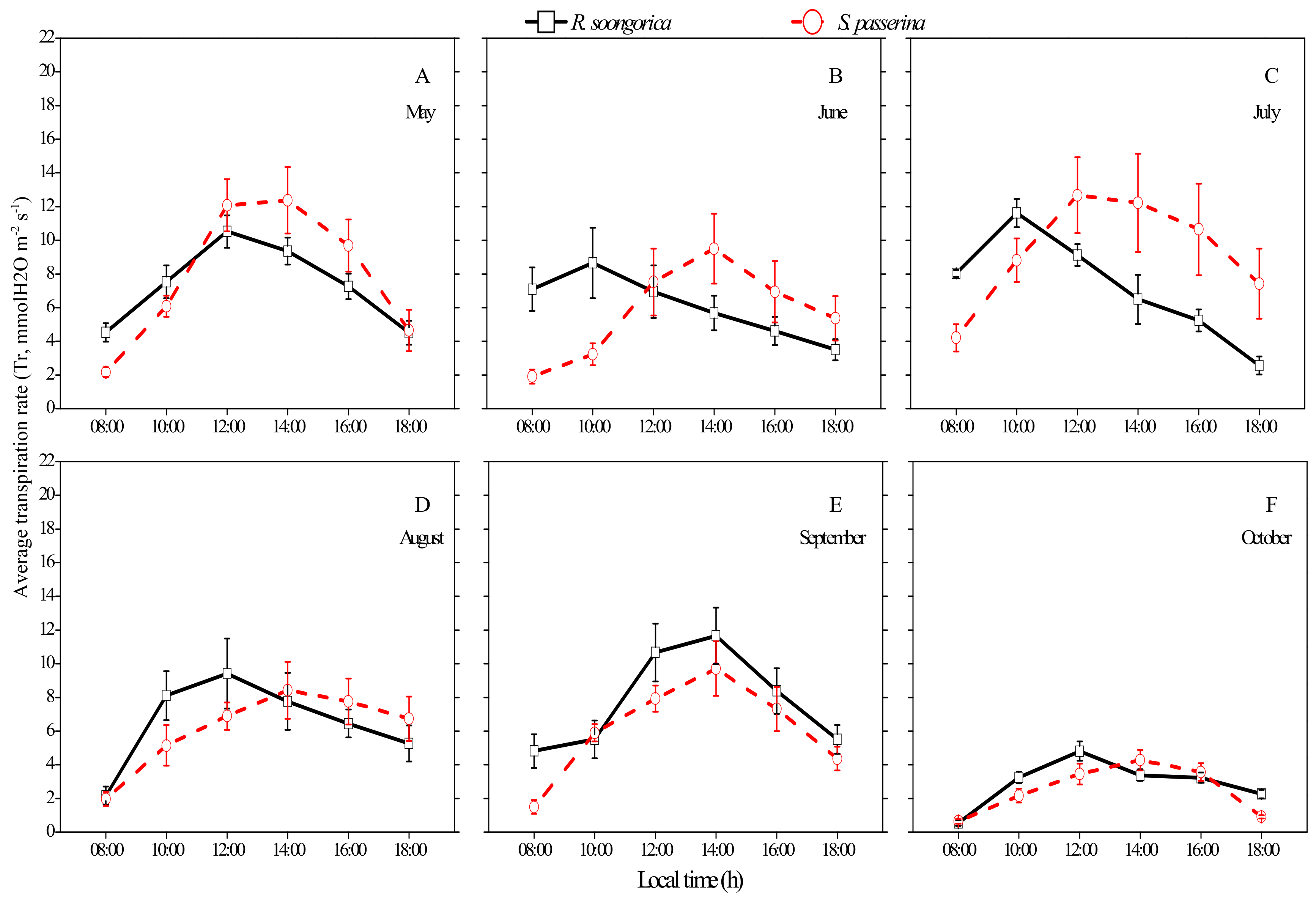
3.2. Daily and Seasonal Patterns of Tr
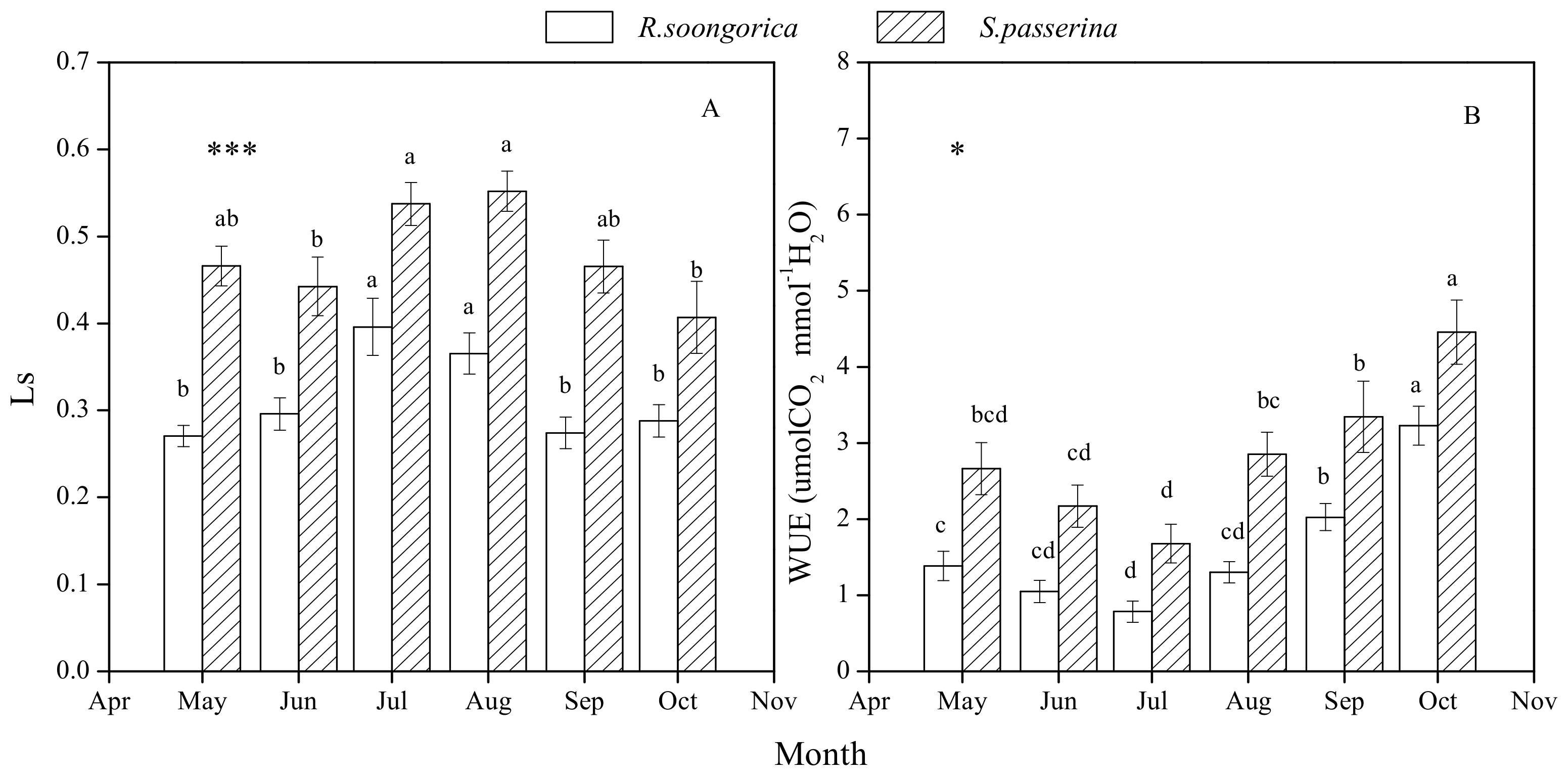
3.3. Ls and WUE Seasonal Changes
3.4. Analysis of the Factors Influencing Transpiration at the Leaf and Canopy Scale
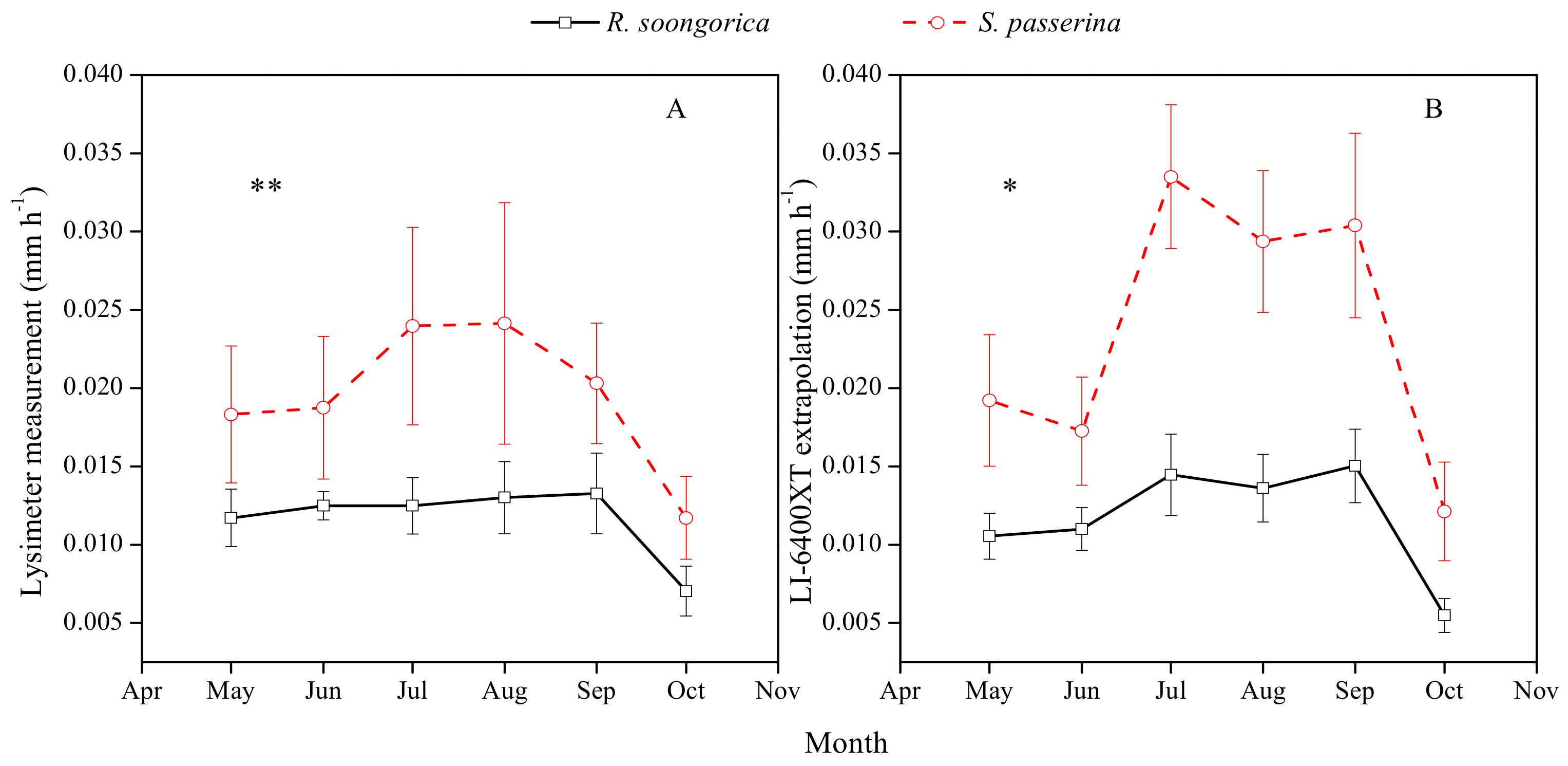
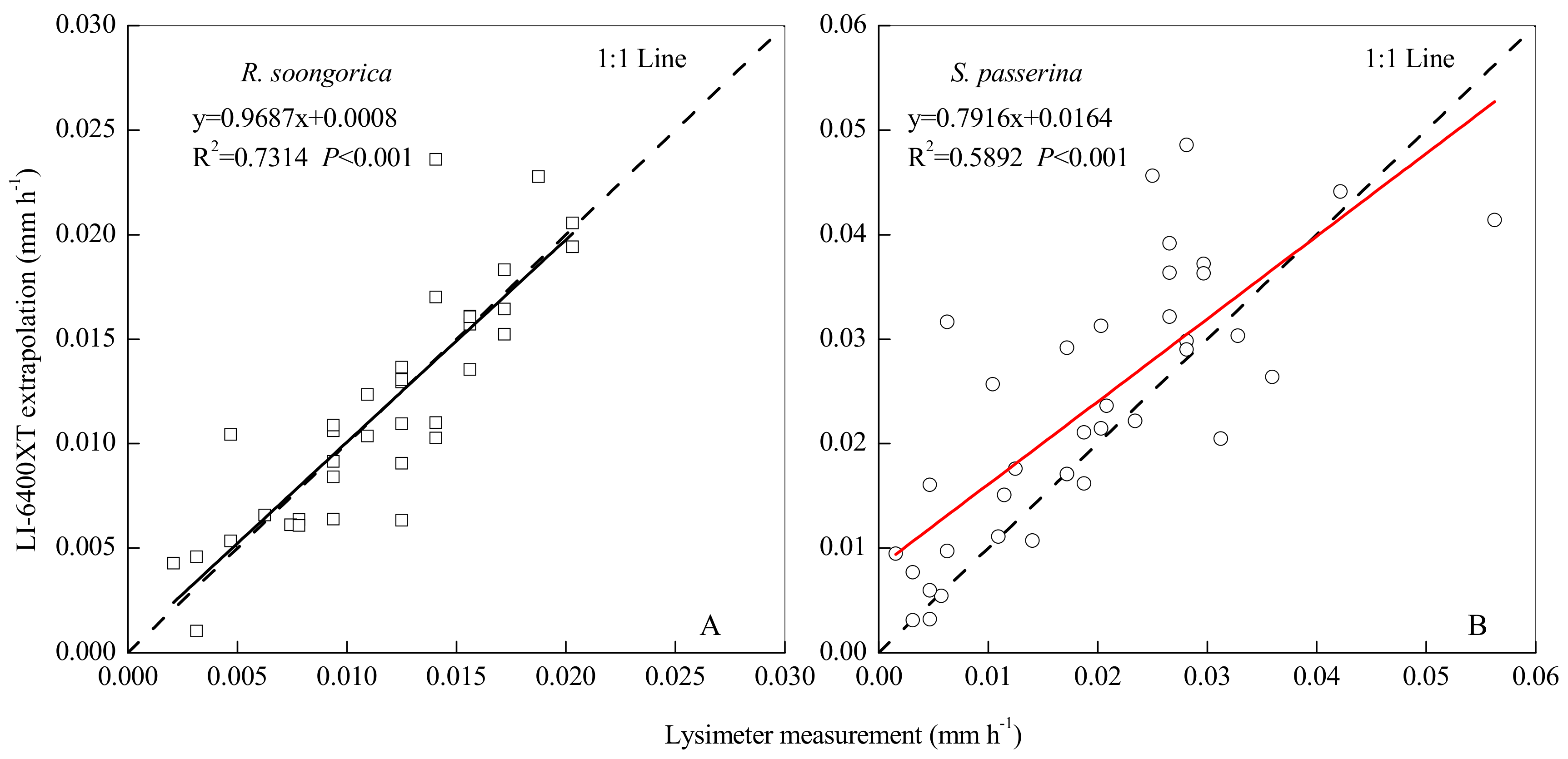
3.5. Scaling Up Transpiration from Leaf to Canopy Level
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Schlesinger, W.H.; Jasechko, S. Transpiration in the global water cycle. Agric. For. Meteorol. 2014, 189–190, 115–117. [Google Scholar] [CrossRef]
- Jasechko, S.; Sharp, Z.D.; Gibson, J.J.; Birks, S.J.; Yi, Y.; Fawcett, P.J. Terrestrial water fluxes dominated by transpiration. Nature 2013, 496, 347. [Google Scholar] [CrossRef] [PubMed]
- Good, S.P.; Noone, D.; Bowen, G. Hydrologic connectivity constrains partitioning of global terrestrial water fluxes. Science 2015, 349, 175–177. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, J.F.; Kemp, P.R.; Tenhunen, J.D. Effects of long-term rainfall variability on evapotranspiration and soil water distribution in the chihuahuan desert: A modeling analysis. Plant Ecol. 2000, 150, 145–159. [Google Scholar] [CrossRef]
- Ham, J.M.; Heilman, J.L.; Lascano, R.J. Determination of soil water evaporation and transpiration from energy balance and stem flow measurements. Agric. For. Meteorol. 1990, 52, 287–301. [Google Scholar] [CrossRef]
- Evett, S.R.; Howell, T.A.; Schneider, S.; Arland, D.; Copeland, K.S.; Dusek, D.A.; Brauer, D.K.; Tolk, J.A.; Marek, G.W.; Marek, T.M.; et al. The bushland weighing lysimeters: A quarter century of crop et investigations to advance sustainable irrigation. Trans. ASABE 2016, 59, 163. [Google Scholar] [CrossRef]
- Meinzer, F.C. Stomatal control of transpiration. Trends Ecol. Evol. 1993, 8, 289–294. [Google Scholar] [CrossRef]
- Huang, L.; Zhang, Z.S.; Li, X.R. The extrapolation of the leaf area-based transpiration of two xerophytic shrubs in a revegetated desert area in the Tengger Desert, China. Hydrol. Res. 2015, 46, 389–399. [Google Scholar] [CrossRef]
- Ivanova, L.A.; Ivanov, L.A.; Ronzhina, D.A.; Tserenkhand, G.; Tsoog, S.; Bazha, S.N.; Gunin, P.D. Leaf and biomass traits of mongolian forest-steppe shrubs linking to their ecological properties. Arid Ecosyst. 2012, 2, 45–53. [Google Scholar] [CrossRef]
- Barbour, M.M.; Hunt, J.E.; Walcroft, A.S.; Rogers, G.N.D.; McSeveny, T.M.; Whitehead, D. Components of ecosystem evaporation in a temperate coniferous rainforest, with canopy transpiration scaled using sapwood density. New Phytol. 2005, 165, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Herbst, M.; Kappen, L.; Thamm, F.; Vanselow, R. Simultaneous measurements of transpiration, soil evaporation and total evaporation in a maize field in northern germany. J. Exp. Bot. 1996, 47, 1957–1962. [Google Scholar] [CrossRef]
- Ávila-Lovera, E.; Zerpa, A.J.; Santiago, L.S. Stem photosynthesis and hydraulics are coordinated in desert plant species. New Phytol. 2017, 216, 1119–1129. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.W.; Zhao, W.Z. Canopy transpiration obtained from leaf transpiration, sap flow and fao-56 dual crop coefficient method. Hydrol. Process. 2015, 29, 2983–2993. [Google Scholar] [CrossRef]
- Karam, F.; Breidy, J.; Stephan, C.; Rouphael, J. Evapotranspiration, yield and water use efficiency of drip irrigated corn in the bekaa valley of lebanon. Agric. Water Manag. 2003, 63, 125–137. [Google Scholar] [CrossRef]
- Baker, J.T.; Gitz, D.C.; Payton, P.; Wanjura, D.F.; Upchurch, D.R. Using leaf gas exchange to quantify drought in cotton irrigated based on canopy temperature measurements. Agron. J. 2007, 99, 637–644. [Google Scholar] [CrossRef]
- Jones, H.G. Use of thermography for quantitative studies of spatial and temporal variation of stomatal conductance over leaf surfaces. Plant Cell Environ. 1999, 22, 1043–1055. [Google Scholar] [CrossRef]
- Gitz, D.C., III; Baker, J.T.; Lascano, R.J. Scaling leaf measurements to estimate whole canopy gas exchanges of cotton. Am. J. Plant Sci. 2016, 7, 1952–1963. [Google Scholar] [CrossRef]
- Gitz, D.C., III; Baker, J.T.; Lascano, R.J. Relating xylem cavitation to gas exchange in cotton. Am. J. Plant Sci. 2015, 6, 10. [Google Scholar] [CrossRef]
- Kim, J.; Verma, S.B. Modeling canopy photosynthesis: Scaling up from a leaf to canopy in a temperate grassland ecosystem. Agric. For. Meteorol. 1991, 57, 187–208. [Google Scholar] [CrossRef]
- Irmak, S.; Mutiibwa, D.; Irmak, A.; Arkebauer, T.J.; Weiss, A.; Martin, D.L.; Eisenhauer, D.E. On the scaling up leaf stomatal resistance to canopy resistance using photosynthetic photon flux density. Agric. For. Meteorol. 2008, 148, 1034–1044. [Google Scholar] [CrossRef]
- Infante, J.M.; Rambal, S.; Joffre, R. Modelling transpiration in holm-oak savannah: Scaling up from the leaf to the tree scale. Agric. For. Meteorol. 1997, 87, 273–289. [Google Scholar] [CrossRef]
- Van der Zande, D.; Mereu, S.; Nadezhdina, N.; Cermak, J.; Muys, B.; Coppin, P.; Manes, F. 3d upscaling of transpiration from leaf to tree using ground-based lidar: Application on a mediterranean holm oak (quercus ilex l.) tree. Agric. For. Meteorol. 2009, 149, 1573–1583. [Google Scholar] [CrossRef]
- Zhang, Z.S.; Zhao, Y.; Li, X.R.; Huang, L.; Tan, H.J. Gross rainfall amount and maximum rainfall intensity in 60-minute influence on interception loss of shrubs: A 10-year observation in the tengger desert. Sci. Rep. 2016, 6, 26030. [Google Scholar] [CrossRef] [PubMed]
- Loik, M.E.; Breshears, D.D.; Lauenroth, W.K.; Belnap, J. A multi-scale perspective of water pulses in dryland ecosystems: Climatology and ecohydrology of the western USA. Oecologia 2004, 141, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Zhang, Z.S. Effect of rainfall pulses on plant growth and transpiration of two xerophytic shrubs in a revegetated desert area: Tengger Desert, China. Catena 2016, 137, 269–276. [Google Scholar] [CrossRef]
- Li, X.R.; Xiao, H.L.; Zhang, J.G.; Wang, X.P. Long-term ecosystem effects of sand-binding vegetation in the Tengger Desert, Northern China. Restor. Ecol. 2004, 12, 376–390. [Google Scholar] [CrossRef]
- Li, X.R.; Kong, D.S.; Tan, H.J.; Wang, X.P. Changes in soil and vegetation following stabilisation of dunes in the southeastern fringe of the Tengger Desert, China. Plant Soil 2007, 300, 221–231. [Google Scholar] [CrossRef]
- Zhang, Y.F.; Wang, X.P.; Hu, R.; Pan, Y.X. Throughfall and its spatial variability beneath xerophytic shrub canopies within water-limited arid desert ecosystems. J. Hydrol. 2016, 539, 406–416. [Google Scholar] [CrossRef]
- Yin, H.X.; Yan, X.; Shi, Y.; Qian, C.J.; Li, Z.H.; Zhang, W.; Wang, L.R.; Li, Y.; Li, X.Z.; Chen, G.X.; et al. The role of East Asian monsoon system in shaping population divergence and dynamics of a constructive desert shrub Reaumuria soongarica. Sci. Rep. 2015, 5, 15823. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.B.; Liu, M.L.; Li, X.R.; Cao, B.; Ma, X.F. Identification of differentially expressed genes in leaf of Reaumuria soongorica under peg-induced drought stress by digital gene expression profiling. PLoS ONE 2014, 9, e94277. [Google Scholar] [CrossRef] [PubMed]
- He, M.Z.; Zhang, K.; Tan, H.J.; Hu, R.; Su, J.Q.; Wang, J.; Huang, L.; Zhang, Y.F.; Li, X.R. Nutrient levels within leaves, stems, and roots of the xeric species Reaumuria soongorica in relation to geographical, climatic, and soil conditions. Ecol. Evol. 2015, 5, 1494–1503. [Google Scholar] [CrossRef] [PubMed]
- Su, P.X.; Yan, Q.D.; Xie, T.T.; Zhou, Z.J.; Gao, S. Associated growth of C3 and C4 desert plants helps the C3 species at the cost of the C4 species. Acta Physiologiae Plantarum 2012, 34, 2057–2068. [Google Scholar] [CrossRef]
- Yang, H.T.; Li, X.R.; Liu, L.C.; Jia, R.L.; Wang, Z.R.; Li, X.J.; Li, G. Biomass allocation patters of four shrubs in desert grassland. J. Desert Res. 2013, 33, 1340–1348. [Google Scholar] [CrossRef]
- Youssef, T.; Awad, M.A. Mechanisms of enhancing photosynthetic gas exchange in date palm seedlings (phoenix dactylifera l.) under salinity stress by a 5-aminolevulinic acid-based fertilizer. J. Plant Growth Regul. 2007, 27, 1–9. [Google Scholar] [CrossRef]
- Berry, J.A.; Downton, W.J.S. 9:-Environmental regulation of photosynthesis. In Photosynthesis; Academic Press: Cambridge, MA, USA, 1982; pp. 263–343. [Google Scholar]
- Kidron, G.J. The effect of shrub canopy upon surface temperatures and evaporation in the Negev Desert. Earth Surf. Process. Landf. 2009, 34, 123–132. [Google Scholar] [CrossRef]
- Zhao, W.Z.; Liu, B.; Chang, X.X.; Yang, Q.Y.; Yang, Y.T.; Liu, Z.L.; Cleverly, J.; Eamus, D. Evapotranspiration partitioning, stomatal conductance, and components of the water balance: A special case of a desert ecosystem in China. J. Hydrol. 2016, 538, 374–386. [Google Scholar] [CrossRef]
- Irmak, S.; Howell, T.A.; Allen, R.G.; Payero, J.O.; Martin, D.L. Standardized ASCE penman-monteith: Impact of sum-of-hourly vs. 24-hour timestep computations at reference weather station sites. Trans. ASABE 2005, 48, 1063. [Google Scholar] [CrossRef]
- Wang, X.P.; Zhang, Y.F.; Hu, R.; Pan, Y.X.; Xu, H.J.; Shi, W.; Jin, Y.X.; Yasuda, H. Revisit of event-based rainfall characteristics at shapotou area in northern china. Sci. Cold Arid Reg. 2016, 8, 0477–0484. [Google Scholar] [CrossRef]
- Pearcy, R.W.; Ehleringer, J. Comparative ecophysiology of C3 and C4 plants. Plant Cell Environ. 1984, 7, 1–13. [Google Scholar] [CrossRef]
- Haworth, M.; Elliott-Kingston, C.; McElwain, J.C. Stomatal control as a driver of plant evolution. J. Exp. Bot. 2011, 62, 2419–2423. [Google Scholar] [CrossRef] [PubMed]
- Ehleringer, J.R.; Sage, R.F.; Flanagan, L.B.; Pearcy, R.W. Climate change and the evolution of C4 photosynthesis. Trends Ecol. Evol. 1991, 6, 95–99. [Google Scholar] [CrossRef]
- Yan, W.M.; Zhong, Y.Q.W.; Shangguan, Z.P. A meta-analysis of leaf gas exchange and water status responses to drought. Sci. Rep. 2016, 6, 20917. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.D. High hopes for c4 plants. Nature 1994, 367, 322. [Google Scholar] [CrossRef]
- Drake, B.G.; Raschke, K.; Salisbury, F.B. Temperature and transpiration resistances of leaves as affected by air temperature, humidity, and wind speed. Plant Physiol. 1970, 46, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Pieruschka, R.; Huber, G.; Berry, J.A. Control of transpiration by radiation. Proc. Natl. Acad. Sci. USA 2010, 107, 13372–13377. [Google Scholar] [CrossRef] [PubMed]
- Vico, G.; Porporato, A. Modelling c3 and c4 photosynthesis under water-stressed conditions. Plant Soil 2008, 313, 187–203. [Google Scholar] [CrossRef]
- Porporato, A.; Laio, F.; Ridolfi, L.; Rodriguez-Iturbe, I. Plants in water-controlled ecosystems: Active role in hydrologic processes and response to water stress: III. Vegetation water stress. Adv. Water Resour. 2001, 24, 725–744. [Google Scholar] [CrossRef]
- Davidson, E.A.; Belk, E.; Boone, R.D. Soil water content and temperature as independent or confounded factors controlling soil respiration in a temperate mixed hardwood forest. Glob. Change Biol. 1998, 4, 217–227. [Google Scholar] [CrossRef]
- Abd El Rahman, A.A.; Batanouny, K.H. Transpiration of desert plants under different environmental conditions. J. Ecol. 1965, 53, 267–272. [Google Scholar] [CrossRef]
- Amthor, J.S. Scaling CO2-photosynthesis relationships from the leaf to the canopy. Photosynth. Res. 1994, 39, 321–350. [Google Scholar] [CrossRef] [PubMed]
| Month | Shrubs | Height (cm) | Canopy Projection Area (×100 cm) | New Shoot Height (cm) | LAI (m2 m−2) |
|---|---|---|---|---|---|
| May | S.passerina | 20.13 ± 1.13 | 6.91 ± 0.76 | 1.84 ± 0.16 | 1.51 ± 0.36 |
| R.soongorica | 17.80 ± 3.03 | 7.92 ± 0.84 | 2.65 ± 0.37 | 1.00 ± 0.19 | |
| Jun. | S.passerina | 22.68 ± 1.21 | 8.77 ± 0.83 | 5.91 ± 1.07 | 1.71 ± 0.42 |
| R.soongorica | 21.68 ± 2.35 | 10.70 ± 1.04 | 6.88 ± 1.19 | 1.33 ± 0.19 | |
| Jul. | S.passerina | 26.15 ± 1.50 | 10.80 ± 1.47 | 7.20 ± 0.86 | 1.85 ± 0.46 |
| R.soongorica | 23.30 ± 3.51 | 11.87 ± 1.13 | 7.74 ± 1.39 | 1.51 ± 0.20 | |
| Aug. | S.passerina | 29.78 ± 2.23 | 13.56 ± 2.24 | 10.67 ± 1.23 | 1.81 ± 0.35 |
| R.soongorica | 24.08 ± 2.99 | 12.92 ± 1.25 | 7.86 ± 1.44 | 1.56 ± 0.26 | |
| Sep. | S.passerina | 28.48 ± 2.75 | 14.71 ± 1.18 | 11.13 ± 1.10 | 1.72 ± 0.33 |
| R.soongorica | 23.65 ± 3.51 | 12.12 ± 1.91 | 7.51 ± 1.42 | 1.33 ± 0.20 | |
| Qct. | S.passerina | 28.88 ± 2.42 | 14.44 ± 1.14 | 10.95 ± 0.69 | 1.56 ± 0.37 |
| R.soongorica | 23.75 ± 3.30 | 10.86 ± 0.70 | 7.23 ± 1.27 | 1.26 ± 0.22 |
| Month | gs | T1 | Tleaf | RH1 | PAR | VPD1 | A |
|---|---|---|---|---|---|---|---|
| May | 0.2077 | 0.7857 *** | 0.7952 *** | −0.2137 | 0.7104 *** | 0.6960 *** | −0.1373 |
| Jun. | 0.7149 *** | −0.2673 | −0.2874 | 0.5154 * | 0.1098 | −0.5996 ** | 0.8189 *** |
| Jul. | 0.5572 ** | 0.0441 | 0.0737 | 0.4360 * | 0.5456 ** | −0.0287 | 0.5101 * |
| Aug. | 0.4580 * | 0.5642 ** | 0.5631 ** | −0.3076 | 0.6200 ** | 0.4411 * | 0.7516 *** |
| Sep. | 0.3530 | 0.7056 *** | 0.7205 *** | −0.5480 ** | 0.5785 ** | 0.6820 *** | 0.7556 *** |
| Qct. | 0.7081 *** | 0.8440 *** | 0.8629 *** | −0.7627 *** | 0.8169 *** | 0.8539 *** | 0.8845 *** |
| Month | gs | T1 | Tleaf | RH1 | PAR | VPD1 | A |
|---|---|---|---|---|---|---|---|
| May | 0.7807 *** | 0.8234 *** | 0.8264 *** | −0.3549 | 0.8397 *** | 0.8381 *** | 0.7071 *** |
| Jun. | 0.6717 *** | 0.6453 *** | 0.6654 *** | −0.3065 | 0.4962 * | 0.5554 ** | 0.7154 *** |
| Jul. | 0.7225 *** | 0.5963 ** | 0.6242 ** | −0.0541 | 0.7028 *** | 0.4980 * | 0.5786 ** |
| Aug. | 0.6735 *** | 0.7353 *** | 0.7447 *** | −0.4890 * | 0.7152 *** | 0.6142 ** | 0.7395 *** |
| Sep. | 0.2939 | 0.7605 *** | 0.7685 *** | −0.5593 ** | 0.7855 *** | 0.6872 *** | 0.7084 *** |
| Qct. | 0.6213 ** | 0.8892 *** | 0.8920 *** | −0.6171 ** | 0.6077 ** | 0.8639 *** | 0.8969 *** |
| Shrubs | Measurement Methods | Rn (W m−2) | Wind Speed (m s−1) | Tair (°C) | RH (%) | VPD (kPa) |
|---|---|---|---|---|---|---|
| R. soongorica | Lysimeter | 0.6549 *** | −0.0297 | 0.4058 * | −0.0762 | 0.2026 |
| LI-6400XT | 0.5430 *** | −0.2128 | 0.4177 * | −0.0347 | 0.2182 | |
| S. passerina | Lysimeter | 0.7106 *** | 0.4763 ** | 0.6000 *** | −0.5324 | 0.6923 *** |
| LI-6400XT | 0.7514 *** | 0.1864 | 0.6220 *** | −0.5623 | 0.6972 *** |
| Shrubs | Accumulated Rainfall (mm) | Canopy Height (cm) | Canopy Projection Area (×100 cm2) | New Shoot Length (cm) | LAI (m2 m−2) |
|---|---|---|---|---|---|
| R. soongorica | 0.9226 ** | 0.9042 * | 0.7423 | 0.7935 | 0.5070 |
| S. passerina | 0.9558 ** | 0.9275 ** | 0.9842 *** | 0.9585 ** | 0.1482 |
| Shrubs | Soil Matric Potential (pF) | Soil Temperature (°C) |
|---|---|---|
| R. soongorica | −0.6948 | 0.3171 |
| S. passerina | 0.5977 | 0.9171 ** |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, Y.; Wang, X.; Zhang, Y.; Pan, Y.; Hu, R. Extrapolation of Leaf Measurements to Obtain the Whole-Canopy Transpiration of C3 and C4 Xerophytic Shrubs. Water 2018, 10, 813. https://doi.org/10.3390/w10060813
Jin Y, Wang X, Zhang Y, Pan Y, Hu R. Extrapolation of Leaf Measurements to Obtain the Whole-Canopy Transpiration of C3 and C4 Xerophytic Shrubs. Water. 2018; 10(6):813. https://doi.org/10.3390/w10060813
Chicago/Turabian StyleJin, Yanxia, Xinping Wang, Yafeng Zhang, Yanxia Pan, and Rui Hu. 2018. "Extrapolation of Leaf Measurements to Obtain the Whole-Canopy Transpiration of C3 and C4 Xerophytic Shrubs" Water 10, no. 6: 813. https://doi.org/10.3390/w10060813
APA StyleJin, Y., Wang, X., Zhang, Y., Pan, Y., & Hu, R. (2018). Extrapolation of Leaf Measurements to Obtain the Whole-Canopy Transpiration of C3 and C4 Xerophytic Shrubs. Water, 10(6), 813. https://doi.org/10.3390/w10060813




