Doxorubicin and Cisplatin Modulate miR-21, miR-106, miR-126, miR-155 and miR-199 Levels in MCF7, MDA-MB-231 and SK-BR-3 Cells That Makes Them Potential Elements of the DNA-Damaging Drug Treatment Response Monitoring in Breast Cancer Cells—A Preliminary Study
Abstract
1. Introduction
| miRNA | Effect and Pathways Mediated in Breast Cancer | Ref. |
|---|---|---|
| miRNA-21 (-3p and -5p) | tumor growth, cancer cells proliferation, metastasis, invasion, sensitivity to chemotherapy, modulation of cancer-related gene expression | [9,10] |
| miRNA-106a-5p | cancer cell proliferation, colony-forming capacity, migration, invasion, breast cancer cell apoptosis and sensitivity to cisplatin, DNA damage response, suppression of the ATM-associated pathway | [11,12] |
| miRNA-126 (-3p and -5p) | cancer cell migration, tumor growth, proliferation, invasion and angiogenesis of triple-negative breast cancer cells | [13,14,15] |
| miRNA-155 (-3p and -5p) | inflammation, apoptosis, adhesion | [1,16,17] |
| miRNA-199b (-3p and -5p) | cancer aggressiveness, tumor growth, angiogenesis | [18,19,20] |
| miRNA-335 (-3p and -5p) | sensitivity of triple-negative breast cancer cells to paclitaxel, cisplatin and doxorubicin | [21] |
2. Materials and Methods
2.1. Cell Culture
2.2. Studied Drugs
2.3. MTT Cell Survival Assay
2.4. miRNA Isolation
2.5. qPCR Assessment of miRNAs
2.6. Statistical Analysis
3. Results
3.1. Doxorubicin and Cisplatin Cytotoxicity Evaluation
3.2. Target miRNAs Selection
3.3. Quantitative Assessment of the Basal Levels of Selected miRNAs
3.4. Cancer Cells Response to Drugs—Evaluation of the Potential Association between Drug Treatment and miRNA Levels
3.4.1. miRNA Alterations in MCF7 Cells
3.4.2. miRNA Alterations in MDA-MB-231 Cells
3.4.3. miRNA Alterations in SK-BR-3
4. Discussion
4.1. Clinical Relevance
4.2. Study Limitations
4.3. Potential Mechanism
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Faraoni, I.; Antonetti, F.R.; Cardone, J.; Bonmassar, E. miR-155 gene: A typical multifunctional microRNA. Biochim. Biophys. Acta Mol. Basis Dis. 2009, 1792, 497–505. [Google Scholar] [CrossRef]
- Esquela-Kerscher, A.; Slack, F. Oncomirs—microRNAs with a role in cancer. Nat. Rev. Cancer 2006, 6, 259–269. [Google Scholar] [CrossRef]
- Gao, Y.; Han, D.; Feng, J. MicroRNA in multiple sclerosis. Clin. Chim. Acta 2021, 516, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Clausen, A.R.; Durand, S.; Petersen, R.L.; Staunstrup, N.H.; Qvist, P. Circulating miRNAs as Potential Biomarkers for Patient Stratification in Bipolar Disorder: A Combined Review and Data Mining Approach. Genes 2022, 13, 1038. [Google Scholar] [CrossRef]
- Angelescu, M.A.; Andronic, O.; Dima, S.O.; Popescu, I.; Meivar-Levy, I.; Ferber, S.; Lixandru, D. miRNAs as Biomarkers in Diabetes: Moving towards Precision Medicine. Int. J. Mol. Sci. 2022, 23, 12843. [Google Scholar] [CrossRef] [PubMed]
- Koturbash, I.; Tolleson, W.H.; Guo, L.; Yu, D.; Chen, S.; Hong, H.; Mattes, W.; Ning, B. microRNAs as pharmacogenomic biomarkers for drug efficacy and drug safety assessment. Biomark. Med. 2015, 9, 1153–1176. [Google Scholar] [CrossRef] [PubMed]
- Uhr, K.; Prager-van der Smissen, W.J.C.P.; Heine, A.A.J.; Ozturk, B.; Van Jaarsveld, M.T.M.; Boersma, A.W.M.; Jager, A.; Wiemer, E.A.C.; Smid, M.; Foekens, J.A.; et al. MicroRNAs as possible indicators of drug sensitivity in breast cancer cell lines. PLoS ONE 2019, 14, e0216400. [Google Scholar] [CrossRef]
- Kahraman, M.; Röske, A.; Laufer, T.; Fehlmann, T.; Backes, C.; Kern, F.; Kohlhaas, J.; Schrörs, H.; Saiz, A.; Zabler, C.; et al. MicroRNA in diagnosis and therapy monitoring of early-stage triple-negative breast cancer. Sci. Rep. 2018, 8, 11584. [Google Scholar] [CrossRef] [PubMed]
- MiR-21-5p|BioVendor R&D. Available online: https://www.biovendor.com/mir-21-5p (accessed on 12 January 2023).
- Huang, S.; Fan, W.; Wang, L.; Liu, H.; Wang, X.; Zhao, H.; Jiang, W. Maspin inhibits MCF-7 cell invasion and proliferation by downregulating miR-21 and increasing the expression of its target genes. Oncol. Lett. 2020, 19, 2621–2628. [Google Scholar] [CrossRef]
- You, F.; Luan, H.; Sun, D.; Cui, T.; Ding, P.; Tang, H.; Sun, D. miRNA-106a Promotes Breast Cancer Cell Proliferation, Clonogenicity, Migration, and Invasion through Inhibiting Apoptosis and Chemosensitivity. DNA Cell Biol. 2019, 38, 198–207. [Google Scholar] [CrossRef]
- Szatkowska, M.; Krupa, R. Regulation of DNA Damage Response and Homologous Recombination Repair by microRNA in Human Cells Exposed to Ionizing Radiation. Cancers 2020, 12, 1838. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Lu, J.; Fan, B.; Sun, L. MicroRNA-126-5p Inhibits the Migration of Breast Cancer Cells by Directly Targeting CNOT7. Technol. Cancer Res. Treat. 2020, 19, 153303382097754. [Google Scholar] [CrossRef]
- MiR-126-3p|BioVendor R&D. Available online: https://www.biovendor.com/mir-126-3p (accessed on 12 January 2023).
- Hong, Z.; Hong, C.; Ma, B.; Wang, Q.; Zhang, X.; Li, L.; Wang, C.; Chen, D. MicroRNA-126-3p inhibits the proliferation, migration, invasion, and angiogenesis of triple-negative breast cancer cells by targeting RGS3. Oncol. Rep. 2019, 42, 1569–1579. [Google Scholar] [CrossRef]
- Ovcharenko, D.; Kelnar, K.; Johnson, C.; Leng, N.; Brown, D. Genome-Scale MicroRNA and Small Interfering RNA Screens Identify Small RNA Modulators of TRAIL-Induced Apoptosis Pathway. Cancer Res 2007, 67, 10782–10788. [Google Scholar] [CrossRef]
- Zhang, G.; Zhong, L.; Luo, H.; Wang, S. MicroRNA-155-3p promotes breast cancer progression through down-regulating CADM1. OncoTargets Ther. 2019, 12, 7993–8002. [Google Scholar] [CrossRef] [PubMed]
- Won, K.Y.; Kim, Y.W.; Kim, H.-S.; Lee, S.K.; Jung, W.-W.; Park, Y.-K. MicroRNA-199b-5p is involved in the Notch signaling pathway in osteosarcoma. Hum. Pathol. 2013, 44, 1648–1655. [Google Scholar] [CrossRef]
- Fang, C.; Wang, F.-B.; Li, Y.; Zeng, X.-T. Down-regulation of miR-199b-5p is correlated with poor prognosis for breast cancer patients. Biomed. Pharmacother. 2016, 84, 1189–1193. [Google Scholar] [CrossRef]
- Lin, X.; Qiu, W.; Xiao, Y.; Ma, J.; Xu, F.; Zhang, K.; Gao, Y.; Chen, Q.; Li, Y.; Li, H.; et al. MiR-199b-5p Suppresses Tumor Angiogenesis Mediated by Vascular Endothelial Cells in Breast Cancer by Targeting ALK1. Front. Genet. 2020, 10, 1397. [Google Scholar] [CrossRef] [PubMed]
- Ye, L.; Wang, F.; Wu, H.; Yang, H.; Yang, Y.; Ma, Y.; Xue, A.; Zhu, J.; Chen, M.; Wang, J.; et al. Functions and Targets of miR-335 in Cancer. OncoTargets Ther. 2021, 14, 3335–3349. [Google Scholar] [CrossRef]
- Sun, N.; Xu, H.N.; Luo, Q.; Li, L.Z. Potential Indexing of the Invasiveness of Breast Cancer Cells by Mitochondrial Redox Ratios. In Oxygen Transport to Tissue XXXVIII; Luo, Q., Li, L.Z., Harrison, D.K., Shi, H., Bruley, D.F., Eds.; Advances in Experimental Medicine and Biology; Springer International Publishing: Cham, Switzerland, 2016; Volume 923, pp. 121–127. ISBN 978-3-319-38808-3. [Google Scholar]
- Atalay, C.; Gurhan, I.D.; Irkkan, C.; Gündüz, U. Multidrug Resistance in Locally Advanced Breast Cancer. Tumor Biol. 2006, 27, 309–318. [Google Scholar] [CrossRef]
- Thorn, C.F.; Oshiro, C.; Marsh, S.; Hernandez-Boussard, T.; McLeod, H.; Klein, T.E.; Altman, R.B. Doxorubicin Pathways. Pharm. Genom. 2011, 21, 440–446. [Google Scholar] [CrossRef]
- Cox, J.; Weinman, S. Mechanisms of doxorubicin resistance in hepatocellular carcinoma. Hepatic Oncol. 2016, 3, 57–59. [Google Scholar] [CrossRef]
- Wang, H.; Guo, S.; Kim, S.-J.; Shao, F.; Ho, J.W.K.; Wong, K.U.; Miao, Z.; Hao, D.; Zhao, M.; Xu, J.; et al. Cisplatin prevents breast cancer metastasis through blocking early EMT and retards cancer growth together with paclitaxel. Theranostics 2021, 11, 2442–2459. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-H.; Chang, J.-Y. New Insights into Mechanisms of Cisplatin Resistance: From Tumor Cell to Microenvironment. Int. J. Mol. Sci. 2019, 20, 4136. [Google Scholar] [CrossRef] [PubMed]
- Romaniuk-Drapała, A.; Totoń, E.; Konieczna, N.; Machnik, M.; Barczak, W.; Kowal, D.; Kopczyński, P.; Kaczmarek, M.; Rubiś, B. hTERT Downregulation Attenuates Resistance to DOX, Impairs FAK-Mediated Adhesion, and Leads to Autophagy Induction in Breast Cancer Cells. Cells 2021, 10, 867. [Google Scholar] [CrossRef] [PubMed]
- Di, H.; Wu, H.; Gao, Y.; Li, W.; Zou, D.; Dong, C. Doxorubicin- and cisplatin-loaded nanostructured lipid carriers for breast cancer combination chemotherapy. Drug Dev. Ind. Pharm. 2016, 42, 2038–2043. [Google Scholar] [CrossRef]
- Lisiak, N.M.; Lewicka, I.; Kaczmarek, M.; Kujawski, J.; Bednarczyk-Cwynar, B.; Zaprutko, L.; Rubis, B. Oleanolic Acid’s Semisynthetic Derivatives HIMOXOL and Br-HIMOLID Show Proautophagic Potential and Inhibit Migration of HER2-Positive Breast Cancer Cells In Vitro. Int. J. Mol. Sci. 2021, 22, 11273. [Google Scholar] [CrossRef]
- Lou, G.; Ma, N.; Xu, Y.; Jiang, L.; Yang, J.; Wang, C.; Jiao, Y.; Gao, X. Differential distribution of U6 (RNU6-1) expression in human carcinoma tissues demonstrates the requirement for caution in the internal control gene selection for microRNA quantification. Int. J. Mol. Med. 2015, 36, 1400–1408. [Google Scholar] [CrossRef]
- Takele Assefa, A.; Vandesompele, J.; Thas, O. On the utility of RNA sample pooling to optimize cost and statistical power in RNA sequencing experiments. BMC Genom. 2020, 21, 312. [Google Scholar] [CrossRef]
- Xu, M.; Sizova, O.; Wang, L.; Su, D.-M. A Fine-Tune Role of Mir-125a-5p on Foxn1 During Age-Associated Changes in the Thymus. Aging Dis. 2017, 8, 277–286. [Google Scholar] [CrossRef]
- Khan, A.A.; Betel, D.; Miller, M.L.; Sander, C.; Leslie, C.S.; Marks, D.S. Transfection of small RNAs globally perturbs gene regulation by endogenous microRNAs. Nat. Biotechnol. 2009, 27, 549–555. [Google Scholar] [CrossRef]
- Zhao, S.; Li, J.; Feng, J.; Li, Z.; Liu, Q.; Lv, P.; Wang, F.; Gao, H.; Zhang, Y. Identification of Serum miRNA-423-5p Expression Signature in Somatotroph Adenomas. Int. J. Endocrinol. 2019, 2019, 8516858. [Google Scholar] [CrossRef]
- Gutiérrez-Vázquez, C.; Rodríguez-Galán, A.; Fernández-Alfara, M.; Mittelbrunn, M.; Sánchez-Cabo, F.; Martínez-Herrera, D.J.; Ramírez-Huesca, M.; Pascual-Montano, A.; Sánchez-Madrid, F. miRNA profiling during antigen-dependent T cell activation: A role for miR-132-3p. Sci. Rep. 2017, 7, 3508. [Google Scholar] [CrossRef]
- TargetScanHuman 8.0. Available online: https://www.targetscan.org/vert_80/ (accessed on 14 January 2023).
- GEPIA 2. Available online: http://gepia2.cancer-pku.cn/#index (accessed on 9 February 2023).
- UCSC Xena. Available online: https://xenabrowser.net/ (accessed on 9 February 2023).
- OncoLnc. Available online: http://www.oncolnc.org/ (accessed on 1 March 2023).
- Mar-Aguilar, F.; Mendoza-Ramírez, J.A.; Malagón-Santiago, I.; Espino-Silva, P.K.; Santuario-Facio, S.K.; Ruiz-Flores, P.; Rodríguez-Padilla, C.; Reséndez-Pérez, D. Serum Circulating microRNA Profiling for Identification of Potential Breast Cancer Biomarkers. Dis. Markers 2013, 34, 163–169. [Google Scholar] [CrossRef]
- Loh, H.-Y.; Norman, B.P.; Lai, K.-S.; Rahman, N.M.A.N.A.; Alitheen, N.B.M.; Osman, M.A. The Regulatory Role of MicroRNAs in Breast Cancer. Int. J. Mol. Sci. 2019, 20, 4940. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, A.M.; Salvatore, M.; Incoronato, M. miRNA-Based Therapeutics in Breast Cancer: A Systematic Review. Front. Oncol. 2021, 11, 668464. [Google Scholar] [CrossRef] [PubMed]
- Gironella, M.; Seux, M.; Xie, M.-J.; Cano, C.; Tomasini, R.; Gommeaux, J.; Garcia, S.; Nowak, J.; Yeung, M.L.; Jeang, K.-T.; et al. Tumor protein 53-induced nuclear protein 1 expression is repressed by miR-155, and its restoration inhibits pancreatic tumor development. Proc. Natl. Acad. Sci. USA 2007, 104, 16170–16175. [Google Scholar] [CrossRef]
- Gao, Y.; Liu, Z.; Ding, Z.; Hou, S.; Li, J.; Jiang, K. MicroRNA-155 increases colon cancer chemoresistance to cisplatin by targeting forkhead box O3. Oncol. Lett. 2018, 15, 4781–4788. [Google Scholar] [CrossRef] [PubMed]
- Lv, L.; An, X.; Li, H.; Ma, L. Effect of miR-155 knockdown on the reversal of doxorubicin resistance in human lung cancer A549/dox cells. Oncol. Lett. 2016, 11, 1161–1166. [Google Scholar] [CrossRef] [PubMed]
- MiR-155-5p|BioVendor R&D. Available online: https://www.biovendor.com/mir-155-5p (accessed on 12 January 2023).
- Dan, T.; Shastri, A.A.; Palagani, A.; Buraschi, S.; Neill, T.; Savage, J.E.; Kapoor, A.; DeAngelis, T.; Addya, S.; Camphausen, K.; et al. miR-21 Plays a Dual Role in Tumor Formation and Cytotoxic Response in Breast Tumors. Cancers 2021, 13, 888. [Google Scholar] [CrossRef]
- Meng, F.; Henson, R.; Wehbe–Janek, H.; Ghoshal, K.; Jacob, S.T.; Patel, T. MicroRNA-21 Regulates Expression of the PTEN Tumor Suppressor Gene in Human Hepatocellular Cancer. Gastroenterology 2007, 133, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Chen, Y.; Tian, R.; Li, J.; Li, H.; Lv, T.; Yao, Q. miRNA-21 enhances chemoresistance to cisplatin in epithelial ovarian cancer by negatively regulating PTEN. Oncol. Lett. 2017, 14, 1807–1810. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Zou, F.; Zhang, X.; Li, H.; Dulak, A.; Tomko, R.J., Jr.; Lazo, J.S.; Wang, Z.; Zhang, L.; Yu, J. microRNA-21 Negatively Regulates Cdc25A and Cell Cycle Progression in Colon Cancer Cells. Cancer Res. 2009, 69, 8157–8165. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Gatti, R.A. MicroRNAs: New players in the DNA damage response. J. Mol. Cell Biol. 2011, 3, 151–158. [Google Scholar] [CrossRef]
- Luo, L.; Xia, L.; Zha, B.; Zuo, C.; Deng, D.; Chen, M.; Hu, L.; He, Y.; Dai, F.; Wu, J.; et al. miR-335-5p targeting ICAM-1 inhibits invasion and metastasis of thyroid cancer cells. Biomed. Pharmacother. 2018, 106, 983–990. [Google Scholar] [CrossRef]
- Tomasetti, M.; Monaco, F.; Manzella, N.; Rohlena, J.; Rohlenova, K.; Staffolani, S.; Gaetani, S.; Ciarapica, V.; Amati, M.; Bracci, M.; et al. MicroRNA-126 induces autophagy by altering cell metabolism in malignant mesothelioma. Oncotarget 2016, 7, 36338–36352. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Ma, Q. MicroRNA-106a inhibits cell proliferation and induces apoptosis in colorectal cancer cells. Oncol. Lett. 2018, 15, 8941–8944. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.-J.; Wei, L.-L.; Wu, X.-J.; Huo, F.-C.; Mou, J.; Pei, D.-S. MiR-106a-5p inhibits the cell migration and invasion of renal cell carcinoma through targeting PAK5. Cell Death Dis. 2017, 8, e3155. [Google Scholar] [CrossRef]
- Lai, Y.; Quan, J.; Hu, J.; Chen, P.; Xu, J.; Guan, X.; Xu, W.; Lai, Y.; Ni, L. miR-199b-5p serves as a tumor suppressor in renal cell carcinoma. Exp. Ther. Med. 2018, 16, 436–444. [Google Scholar] [CrossRef]
- Gao, J.; Zhang, Q.; Xu, J.; Guo, L.; Li, X. Clinical significance of serum miR-21 in breast cancer compared with CA153 and CEA. Chin. J. Cancer Res. 2013, 25, 743–748. [Google Scholar] [CrossRef]
- Najjary, S.; Mohammadzadeh, R.; Mokhtarzadeh, A.; Mohammadi, A.; Kojabad, A.B.; Baradaran, B. Role of miR-21 as an authentic oncogene in mediating drug resistance in breast cancer. Gene 2020, 738, 144453. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Tan, Z.; Hu, H.; Liu, H.; Wu, T.; Zheng, C.; Wang, X.; Luo, Z.; Wang, J.; Liu, S.; et al. microRNA-21 promotes breast cancer proliferation and metastasis by targeting LZTFL1. BMC Cancer 2019, 19, 738. [Google Scholar] [CrossRef]
- Valeri, N.; Gasparini, P.; Braconi, C.; Paone, A.; Lovat, F.; Fabbri, M.; Sumani, K.M.; Alder, H.; Amadori, D.; Patel, T.; et al. MicroRNA-21 induces resistance to 5-fluorouracil by down-regulating human DNA MutS homolog 2 (hMSH2). Proc. Natl. Acad. Sci. USA 2010, 107, 21098–21103. [Google Scholar] [CrossRef]
- You, F.; Li, J.; Zhang, P.; Zhang, H.; Cao, X. miR106a Promotes the Growth of Transplanted Breast Cancer and Decreases the Sensitivity of Transplanted Tumors to Cisplatin. Cancer Manag. Res. 2020, 12, 233–246. [Google Scholar] [CrossRef] [PubMed]
- Mayr, C. Regulation by 3′-Untranslated Regions. Annu. Rev. Genet. 2017, 51, 171–194. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Paranjape, T.; Ullrich, R.; Nallur, S.; Gillespie, E.; Keane, K.; Esquela-Kerscher, A.; Weidhaas, J.B.; Slack, F.J. The mir-34 microRNA is required for the DNA damage response in vivo in C. elegans and in vitro in human breast cancer cells. Oncogene 2009, 28, 2419–2424. [Google Scholar] [CrossRef] [PubMed]
- Lukashchuk, N.; Vousden, K.H. Ubiquitination and Degradation of Mutant p53. Mol. Cell. Biol. 2007, 27, 8284–8295. [Google Scholar] [CrossRef]
- Habbe, N.; Koorstra, J.-B.M.; Mendell, J.T.; Offerhaus, G.J.; Ryu, J.K.; Feldmann, G.; Mullendore, M.E.; Goggins, M.G.; Hong, S.-M.; Maitra, A. MicroRNA miR-155 is a biomarker of early pancreatic neoplasia. Cancer Biol. Ther. 2009, 8, 340–346. [Google Scholar] [CrossRef]
- Zhang, C.-M.; Zhao, J.; Deng, H.-Y. MiR-155 promotes proliferation of human breast cancer MCF-7 cells through targeting tumor protein 53-induced nuclear protein 1. J. Biomed. Sci. 2013, 20, 79. [Google Scholar] [CrossRef]
- Luo, W.; Zhang, H.; Liang, X.; Xia, R.; Deng, H.; Yi, Q.; Lv, L.; Qian, L. DNA methylation-regulated miR-155-5p depresses sensitivity of esophageal carcinoma cells to radiation and multiple chemotherapeutic drugs via suppression of MAP3K10. Oncol. Rep. 2020, 43, 1692–1704. [Google Scholar] [CrossRef]
- Gasparini, P.; Lovat, F.; Fassan, M.; Casadei, L.; Cascione, L.; Jacob, N.K.; Carasi, S.; Palmieri, D.; Costinean, S.; Shapiro, C.L.; et al. Protective role of miR-155 in breast cancer through RAD51 targeting impairs homologous recombination after irradiation. Proc. Natl. Acad. Sci. USA 2014, 111, 4536–4541. [Google Scholar] [CrossRef]
- Chiu, Y.-C.; Tsai, M.-H.; Chou, W.-C.; Liu, Y.-C.; Kuo, Y.-Y.; Hou, H.-A.; Lu, T.-P.; Lai, L.-C.; Chen, Y.; Tien, H.-F.; et al. Prognostic significance of NPM1 mutation-modulated microRNA−mRNA regulation in acute myeloid leukemia. Leukemia 2016, 30, 274–284. [Google Scholar] [CrossRef]
- Cao, W.; Gao, W.; Liu, Z.; Hao, W.; Li, X.; Sun, Y.; Tong, L.; Tang, B. Visualizing miR-155 To Monitor Breast Tumorigenesis and Response to Chemotherapeutic Drugs by a Self-Assembled Photoacoustic Nanoprobe. Anal. Chem. 2018, 90, 9125–9131. [Google Scholar] [CrossRef]
- Wu, A.; Chen, Y.; Liu, Y.; Lai, Y.; Liu, D. miR-199b-5p inhibits triple negative breast cancer cell proliferation, migration and invasion by targeting DDR1. Oncol. Lett. 2018, 16, 4889–4896. [Google Scholar] [CrossRef]
- Fornari, F.; Milazzo, M.; Chieco, P.; Negrini, M.; Calin, G.A.; Grazi, G.L.; Pollutri, D.; Croce, C.M.; Bolondi, L.; Gramantieri, L. MiR-199a-3p Regulates mTOR and c-Met to Influence the Doxorubicin Sensitivity of Human Hepatocarcinoma Cells. Cancer Res. 2010, 70, 5184–5193. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, N.; Minemura, C.; Asai, S.; Kikkawa, N.; Kinoshita, T.; Oshima, S.; Koma, A.; Kasamatsu, A.; Hanazawa, T.; Uzawa, K.; et al. Identification of miR-199-5p and miR-199-3p Target Genes: Paxillin Facilities Cancer Cell Aggressiveness in Head and Neck Squamous Cell Carcinoma. Genes 2021, 12, 1910. [Google Scholar] [CrossRef]
- Scarola, M.; Schoeftner, S.; Schneider, C.; Benetti, R. miR-335 Directly Targets Rb1 (pRb/p105) in a Proximal Connection to p53-Dependent Stress Response. Cancer Res 2010, 70, 6925–6933. [Google Scholar] [CrossRef] [PubMed]
- Hao, J.; Lai, M.; Liu, C. Expression of miR-335 in triple-negative breast cancer and its effect on chemosensitivity. J. BUON 2019, 24, 1526–1531. [Google Scholar] [PubMed]
- Hajibabaei, S.; Sotoodehnejadnematalahi, F.; Nafissi, N.; Zeinali, S.; Azizi, M. Aberrant promoter hypermethylation of miR-335 and miR-145 is involved in breast cancer PD-L1 overexpression. Sci. Rep. 2023, 13, 1003. [Google Scholar] [CrossRef]
- The Cancer Genome Atlas Program-NCI. Available online: https://www.cancer.gov/about-nci/organization/ccg/research/structural-genomics/tcga (accessed on 1 March 2023).
- Mavrommati, I.; Johnson, F.; Echeverria, G.V.; Natrajan, R. Subclonal heterogeneity and evolution in breast cancer. NPJ Breast Cancer 2021, 7, 155. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Z. MicroRNAs as Oncogenes and Tumor Suppressors. N. Engl. J. Med. 2005, 353, 1768–1771. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Fu, L.L.; Wen, X.; Liu, B.; Huang, J.; Wang, J.H.; Wei, Y.Q. Oncogenic and tumor suppressive roles of microRNAs in apoptosis and autophagy. Apoptosis 2014, 19, 1177–1189. [Google Scholar] [CrossRef] [PubMed]
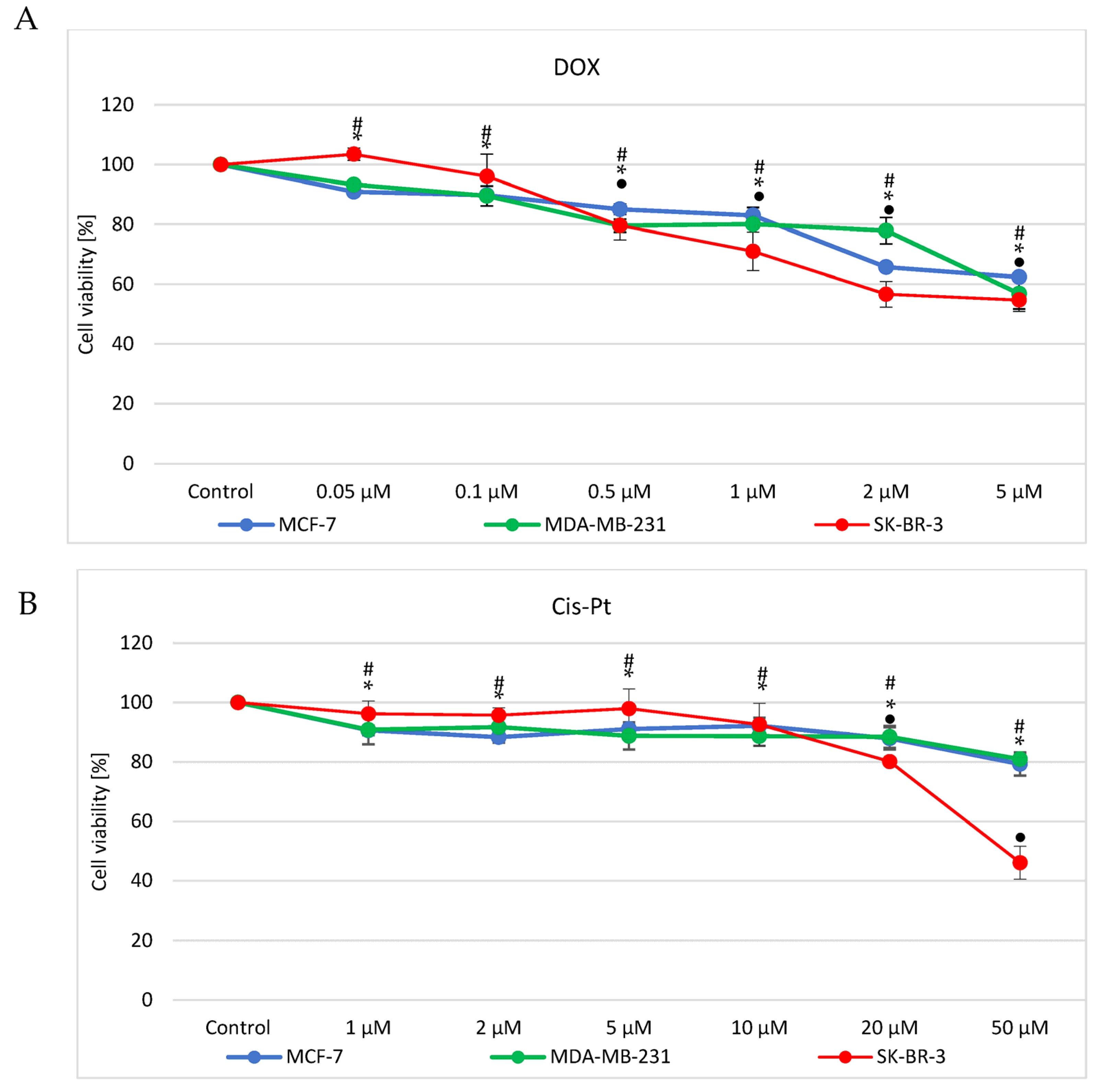
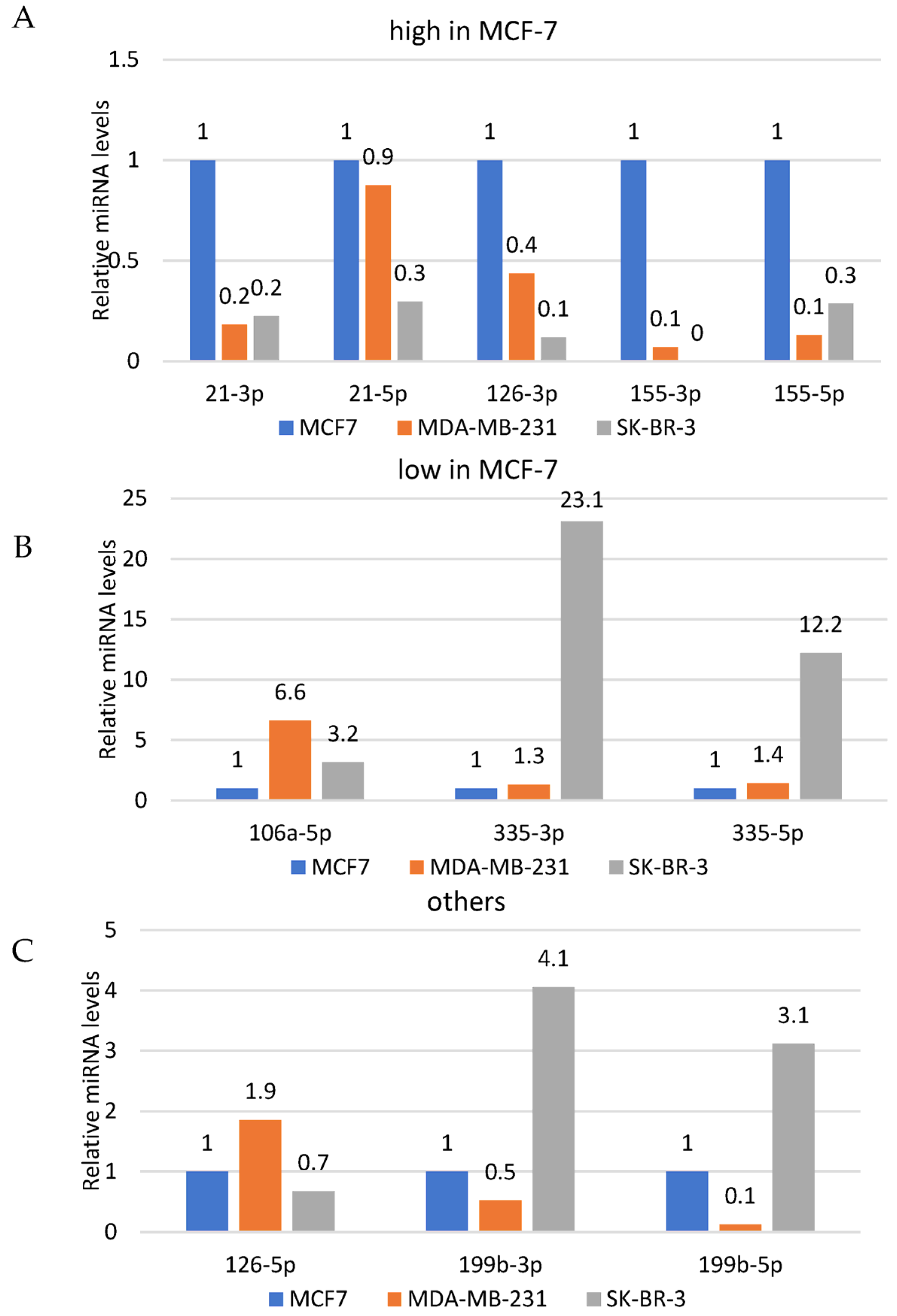
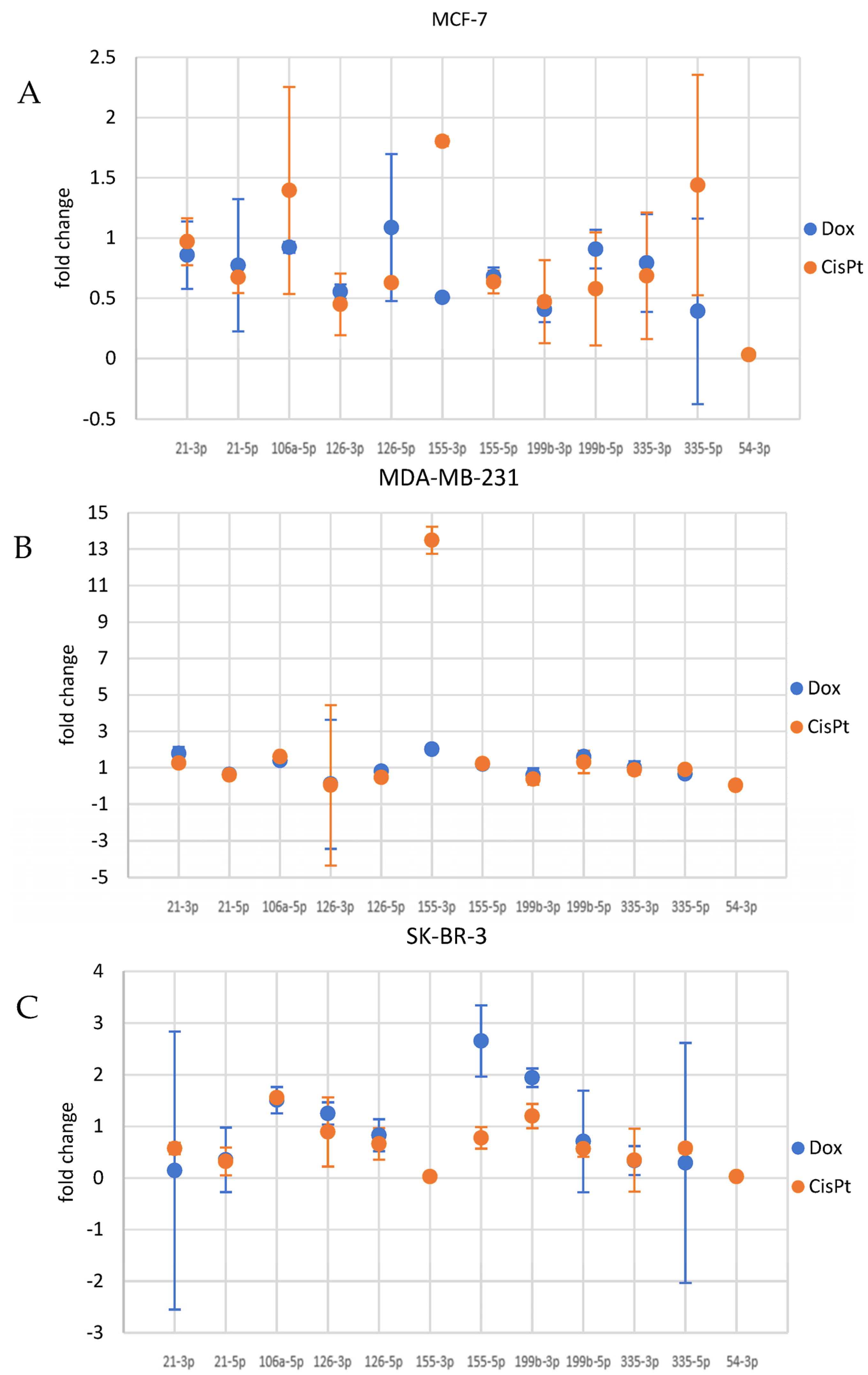
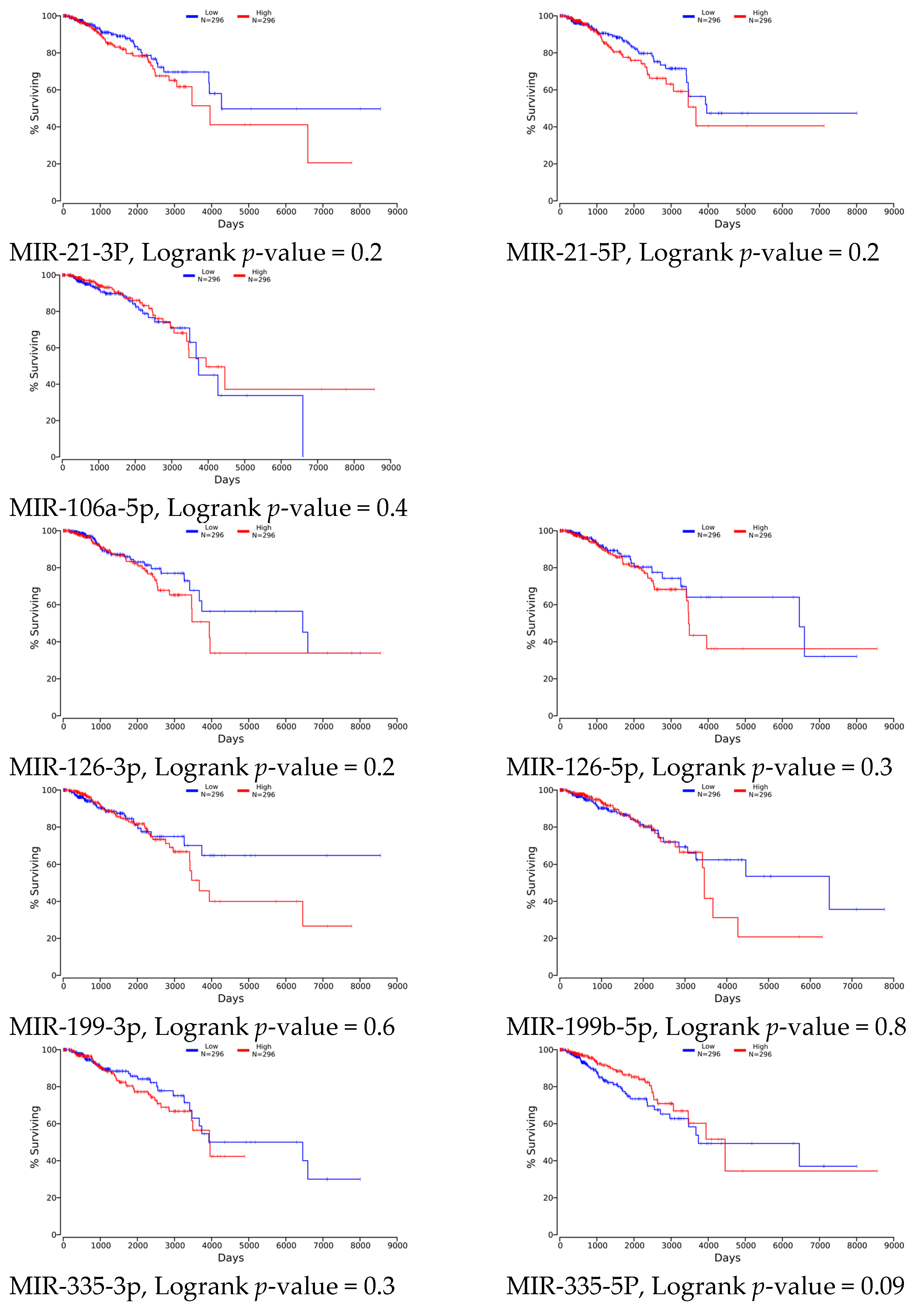
| Drug | IC50 [µM] | ||
|---|---|---|---|
| Cell Line | DOX | Cis-Pt | |
| MCF7 | 27.4 ± 0.9 | >50 | |
| MDA-MB-231 | 12.9 ± 1.8 | >50 | |
| SK-BR-3 | 5.8 ± 0.7 | 44.3 ± 2.6 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mizielska, A.; Dziechciowska, I.; Szczepański, R.; Cisek, M.; Dąbrowska, M.; Ślężak, J.; Kosmalska, I.; Rymarczyk, M.; Wilkowska, K.; Jacczak, B.; et al. Doxorubicin and Cisplatin Modulate miR-21, miR-106, miR-126, miR-155 and miR-199 Levels in MCF7, MDA-MB-231 and SK-BR-3 Cells That Makes Them Potential Elements of the DNA-Damaging Drug Treatment Response Monitoring in Breast Cancer Cells—A Preliminary Study. Genes 2023, 14, 702. https://doi.org/10.3390/genes14030702
Mizielska A, Dziechciowska I, Szczepański R, Cisek M, Dąbrowska M, Ślężak J, Kosmalska I, Rymarczyk M, Wilkowska K, Jacczak B, et al. Doxorubicin and Cisplatin Modulate miR-21, miR-106, miR-126, miR-155 and miR-199 Levels in MCF7, MDA-MB-231 and SK-BR-3 Cells That Makes Them Potential Elements of the DNA-Damaging Drug Treatment Response Monitoring in Breast Cancer Cells—A Preliminary Study. Genes. 2023; 14(3):702. https://doi.org/10.3390/genes14030702
Chicago/Turabian StyleMizielska, Anna, Iga Dziechciowska, Radosław Szczepański, Małgorzata Cisek, Małgorzata Dąbrowska, Jan Ślężak, Izabela Kosmalska, Marta Rymarczyk, Klaudia Wilkowska, Barbara Jacczak, and et al. 2023. "Doxorubicin and Cisplatin Modulate miR-21, miR-106, miR-126, miR-155 and miR-199 Levels in MCF7, MDA-MB-231 and SK-BR-3 Cells That Makes Them Potential Elements of the DNA-Damaging Drug Treatment Response Monitoring in Breast Cancer Cells—A Preliminary Study" Genes 14, no. 3: 702. https://doi.org/10.3390/genes14030702
APA StyleMizielska, A., Dziechciowska, I., Szczepański, R., Cisek, M., Dąbrowska, M., Ślężak, J., Kosmalska, I., Rymarczyk, M., Wilkowska, K., Jacczak, B., Totoń, E., Lisiak, N., Kopczyński, P., & Rubiś, B. (2023). Doxorubicin and Cisplatin Modulate miR-21, miR-106, miR-126, miR-155 and miR-199 Levels in MCF7, MDA-MB-231 and SK-BR-3 Cells That Makes Them Potential Elements of the DNA-Damaging Drug Treatment Response Monitoring in Breast Cancer Cells—A Preliminary Study. Genes, 14(3), 702. https://doi.org/10.3390/genes14030702








