Abstract
Autism spectrum disorder (ASD) is an early-onset neurodevelopmental disorder in which genetics play a major role. Molecular diagnosis may lead to a more accurate prognosis, improved clinical management, and potential treatment of the condition. Both copy number variations (CNVs) and single nucleotide variations (SNVs) have been reported to contribute to the genetic etiology of ASD. The effectiveness and validity of clinical targeted panel sequencing (CTPS) designed to analyze both CNVs and SNVs can be evaluated in different ASD cohorts. CTPS was performed on 573 patients with the diagnosis of ASD. Medical records of positive CTPS cases were further reviewed and analyzed. Additional medical examinations were performed for a group of selective cases. Positive molecular findings were confirmed by orthogonal methods. The overall positive rate was 19.16% (109/569) in our cohort. About 13.89% (79/569) and 4.40% (25/569) of cases had SNVs only and CNVs only findings, respectively, while 0.9% (5/569) of cases had both SNV and CNV findings. For cases with SNVs findings, the SHANK3 gene has the greatest number of reportable variants, followed by gene MYT1L. Patients with MYT1L variants share common and specific clinical characteristics. We found a child with compound heterozygous SLC26A4 variants had an enlarged vestibular aqueduct syndrome and autistic phenotype. Our results showed that CTPS is an effective molecular diagnostic tool for ASD. Thorough clinical and genetic evaluation of ASD can lead to more accurate diagnosis and better management of the condition.
1. Introduction
Autism spectrum disorder (ASD) is an early-onset neurodevelopmental disorder characterized by social communication deficits and repetitive sensory-motor behaviors that affect about 1% of children worldwide [1]. The reported sex ratio ranges from 2:1–5:1 (approximately 4:1) between males and females [2,3]. Genetic variants have been considered to be a major cause of pathogenesis. 64–93% ASD risk is heritable [4,5]. Studies of ASD siblings showed that 7–20% of subsequent children suffered ASD diagnosis after their elder brother/sister [6]. Risk also increased in families with a higher diagnostic rate of ASD.
Identifying genetic etiologies of ASD provides useful information for clinicians and families. Genetic testing keeps marching forward owing to advances in sequencing technologies. It is currently considered that ASD genetic variants are highly heterogeneous and individualized. Over 1000 genes have been reported to be associated with ASD [7]. Chromosomal aberrations, copy number variations (CNVs), and single nucleotide variations (SNVs) both play a role in the pathology of ASD and have led to progress in the understanding of the complex genetic background of the disease. Chromosomal microarray (CMA) analysis for CNV detection is recommended as the most appropriate initial test for the etiologic evaluation of ASD patients [8,9,10]. In recent years, whole-exome sequencing (WES) based on next-generation sequencing (NGS) technology has been applied for further etiological evaluation of patients without CMA findings. It allows for the identification of SNVs, including pathogenic substitutions, insertions, or deletions, which have been associated with ASD [8,11,12]. Whole-genome sequencing (WGS) has also added value as a diagnostic test for ASD [13]. Clinical heterogeneity is much more widely recognized in ASD children sharing core features [14]. Improving the skills of distinguishing key clinical features may increase the rate of recognition for patients harboring relevant genetic variants. In addition, children with certain syndromes may exhibit autism-like behaviors. Genetic testing can help diagnosis and evaluate prognosis.
Overall, finding a diagnostic etiology helps patients and families obtain more information about co-occurring medical problems and prognosis, acquire effective interventions and connect families to specific support groups. Therefore, a study using NGS containing CNVs and SNVs analysis for ASD cohort is ideal for clinical and research practice. In this study, we report the effectiveness and validity of clinical targeted panel sequencing (CTPS) comprising 2742 genes. Additionally, we report the yield and specific founding of CTPS cases and understand the correlations between genotype and phenotype of ASD.
2. Materials and Methods
2.1. Study Participants and Case Review
Patients receiving a diagnosis of ASD in the Department of Child Health Care, Children’s Hospital of Fudan University, were included consecutively from January 2019 to December 2020. The inclusion criteria were as follows: all the patients met the criteria of ASD diagnosed by developmental-behavioral pediatricians using the Diagnostic and Statistical Manual of Mental Disorders, fifth edition (DSM-V)) [15], medical records of positive CTPS results were reviewed. Additional medical examinations were performed for the selected cases.
2.2. Clinical Targeted Panel Sequencing, Data Processing, and Variant Classification
Genomic DNA of every patient was extracted from peripheral blood samples in EDTA-coated Vacutainers according to standard procedure. CTPS was applied for enrolled ASD patients using the Agilent ClearSeq Inherited Disease panel kit (Santa Clara, CA, USA) for enrichment based on NGS [16,17]. The CTPS included 2742 genes. Sequencing was performed on an Illumina HiSeq X10 (Illumina, San Diego, CA, USA) with 150 bp pair-end sequencing. The average on-target sequencing depth was 200× for CTPS and average reads mapping rate was 99.8%. The fraction of the targeted region with at least 10× and 20× were 99.1% and 97.2%.
Details of the variant calling, filtration, and annotation can be found in our previously published papers [16]. Briefly, for SNV and small insertion/deletion calling, GATK best practice pipeline was applied, including sequence alignment to the hg19 reference genome by BWA (V.05.9-r16), sorting by Samtools (v.1.8) and the duplication removed by Picard (v.2.20.1), with default settings. Variants were annotated by VEP (v.104.2) [18] and ANNOVAR (v.2019-10-24) [19] with basic gene-based annotation (RefSeq, Ensembl), damaging prediction (SIFT, PolyPhen2), function annotation (OMIM), and pathogenicity annotation (ClinVar, HGMD). Then, variants were filtered according to the following criteria: (1) variants out of the capture region (exon region extended by 15 bp); (2) high allele frequency in public gnomAD databases; (3) zygosity not match; (4) variants from genes with AD inheritance model not inherited from healthy parents; (5) low-quality variants except reported pathogenic variants; (6) clinical phenotype matching by a computational phenotype filtering process.
For CNV calling, two read-depth-based algorithms, CANOES and HMZDelFinder, were applied at the exon level and combined at the region-level. The PICNIC (Pipeline for clinical NGS-involved CNV detection) and AnnotSV (online version) were used for the following CNV filtration and annotation [20]. Variants were annotated with gene-based annotation (RefSeq), region-based annotation (DGV) and function annotation (OMIM). Then, variants were filtered, mainly considering the frequency of deletions or duplications in the internal samples, the region size and also the matching of clinical phenotypes.
For the combination of SNV and CNV, an SCI strategy [16] was additionally applied to consider the complicated compound heterozygous condition. Finally, our internal automatic variant processing pipelines can leave an average of approximately a dozen genes (median 40) per sample for further manual review.
We made the diagnosis considering both CNVs and SNVs based on the ACMG guidelines but with some adjustments, as published studies have described variant classification criteria [16,17]. The criteria of “diagnostic rate/positive rate” is the proportion of cases with positive findings, where we can find disease-causing SNVs or CNVs that can explain the patient’s phenotype with matched inheritance model. The “overall positive rate” is the proportion of cases finding both SNVs and CNVs. For the diagnostic SNVs, Sanger sequencing was used for variant confirmation, qPCR/MLPA was used for the diagnostic CNVs, and Mutation Surveyor software (SoftGenetics version5.0) was used to analyze the data for both patient samples and their parents.
3. Results
3.1. Demographics and Clinical Files of Patients
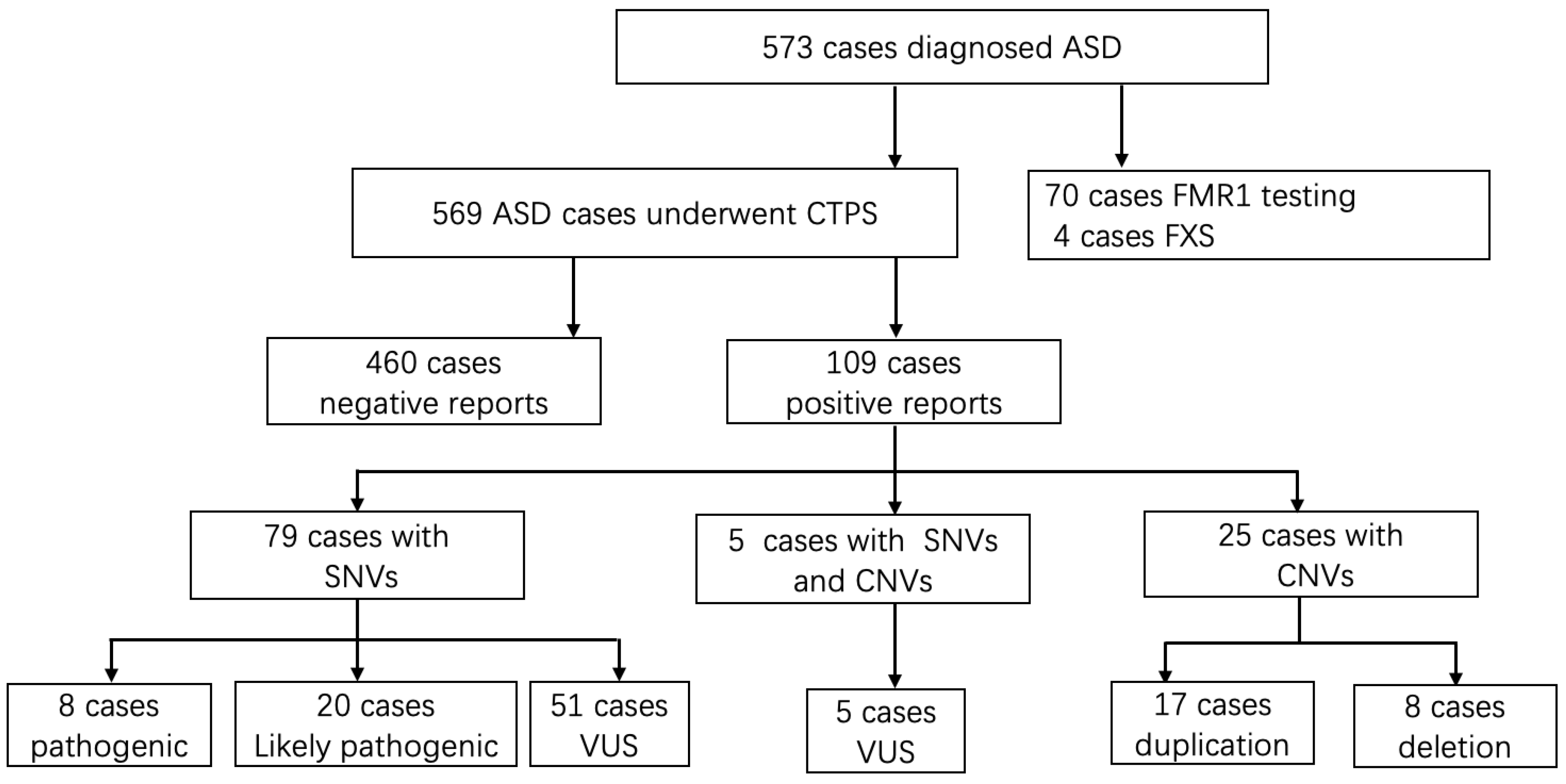
A total of 573 patients receiving a diagnosis of ASD were enrolled in the cohort following the inclusion criteria. The sex ratio was 4.03:1 (459 Males to 114 females). The mean age of these patients was 3.6 years old, from 16 months to 12.8 years. A diagnosis of ASD was made by qualified developmental-behavioral specialists using DSM-V [15] and Autism Diagnostic Observation Schedule, second edition (ADOS-2) [21]. Figure 1 showed the flow of study.
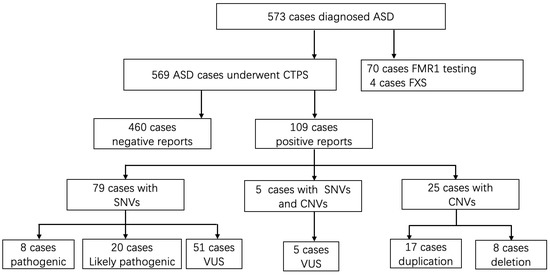
Figure 1.
The flow of genetic evaluation of 573 cases with autism spectrum disorders in genetic clinics. ASD, autism spectrum disorder; CTPS: clinical targeted panel sequencing; FXS: Fragile X syndrome; SNVs: single nucleotide variations; CNVs, copy number variations; VUS, variant of unknown significance.
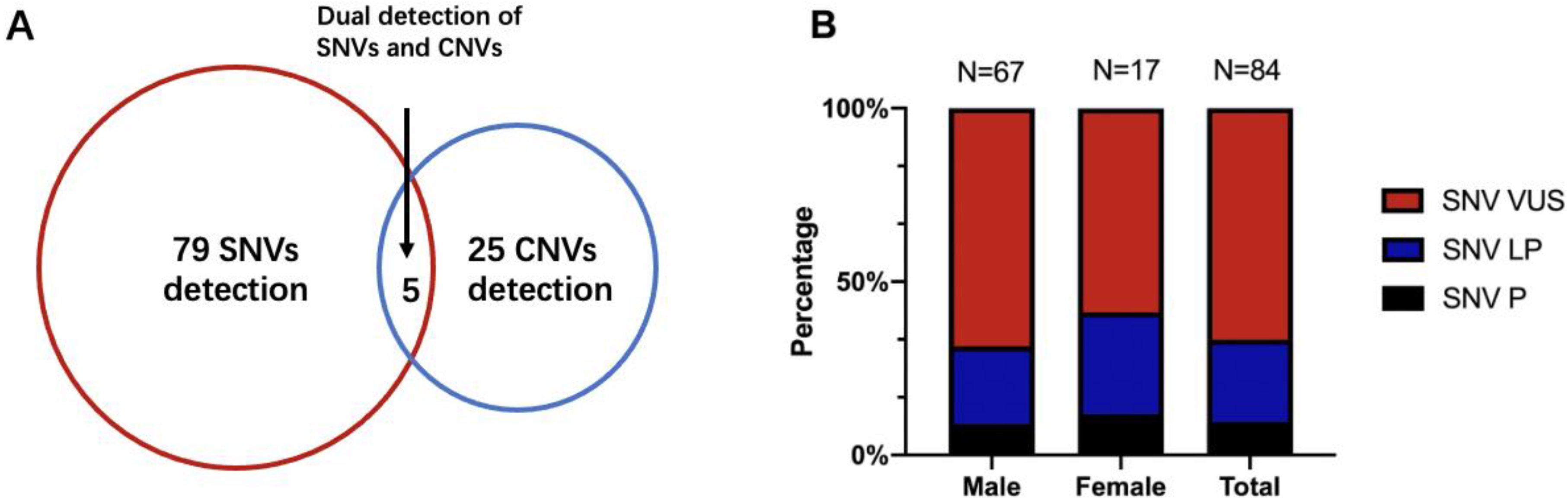
Seventy patients underwent extra FMR1 testing in addition to CTPS, who had prominent clinical features especially the facial characteristics such as long face, prominent ears, and prominent jaw, and 4 out of 70 (5.71%) obtained positive results and were lately diagnosed with Fragile X syndrome. Analyzing SNVs and CNVs simultaneously, an overall diagnostic yield of 19.16% (109/569) was reached (Table 1 and Table 2). SNVs alone were detected in 13.89% of the cases (79/569), 25 patients with CNVs alone accounted for 4.40% (25/569) of the detection rate, and the remaining 0.88% (5/569) had both SNV and CNV findings (Figure 2A).

Table 1.
SNVs identified from ASD patients from CTPS.

Table 2.
CNVs identified from ASD patients from CTPS.
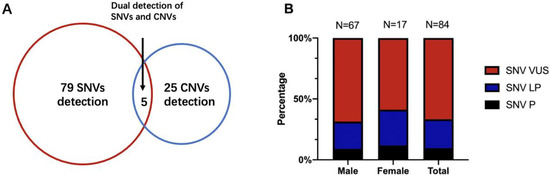
Figure 2.
(A) The detection yield of SNV and CNV of CTPS in 569 patients. (B) The percentage of SNVs diagnostic variations between males and females. SNVs, single nucleotide variations; CNVs, copy number variations; P, pathogenic; LP, likely pathogenic; VUS, variant of unknown significance.
For SNVs, 62 patients had missense mutations, 10 patients had frameshift mutations, 1 patient had a nonframeshift mutation, 4 had splicing mutations, and 7 had more than one kind of variant. We found that 5 patients had SHANK3 variants, which was the greatest number of reportable variants, leading to the diagnostic yield of 0.88% (5/569). Four children had MYT1L variants. MECP2, DIP2B, DYRK1A, FOXP1, and PHIP variants were found in 3 patients, respectively (Table 1). For CNVs, 18 were duplication variants (interestingly, we found that 6 patients had 15q11-13 duplications in 20% of CNV cases), and 12 were deletion variants (all were heterozygous deletions), which are shown in Table 2 (for gene impacted and frequency of CNVs detailed in Table S1).
Of 109 patients who had molecular abnormalities, 84 were males and 25 were females. For diagnostic SNVs in males, a total of 67 cases were included, of which “pathogenic” SNVs accounted for 8.96% (6/67), “likely pathogenic” SNVs accounted for 22.39% (15/67), and the remaining 68.66% (46/67) were “variants of unknown significance (VUS)”. CNVs were found in 21 male patients (including 4 patients with SNVs), of which 8 patients were also validated by CMA. In 25 females with genetic abnormalities, SNVs were found in 17 females and CNVs could be found in 9 females (5 had CMA validation), with one having both CNV and SNV. Meanwhile, the percentage of pathogenic SNVs in females was 11.76% (2/17), “likely pathogenic” was 29.41% (5/17), and “VUS” was 58.82% (10/17). There were no significant differences between the proportions of SNVs in males and females (Fisher’s exact p = 0.63) (Figure 2B). Thirty-four cases also conducted parental tests (both father and mother) by Sanger. Fourteen SNVs were de novo (41.18%, 14/34), 17 were inherited (50.00%, 17/34), and 3 had variants that were both de novo and inherited (8.82%, 3/34), leaving 50 unavailable to be tested.
3.2. Representative Cases from CTPS
3.2.1. Summarized Cases: Analysis of Clinical Characteristics Classify Genetic Problems
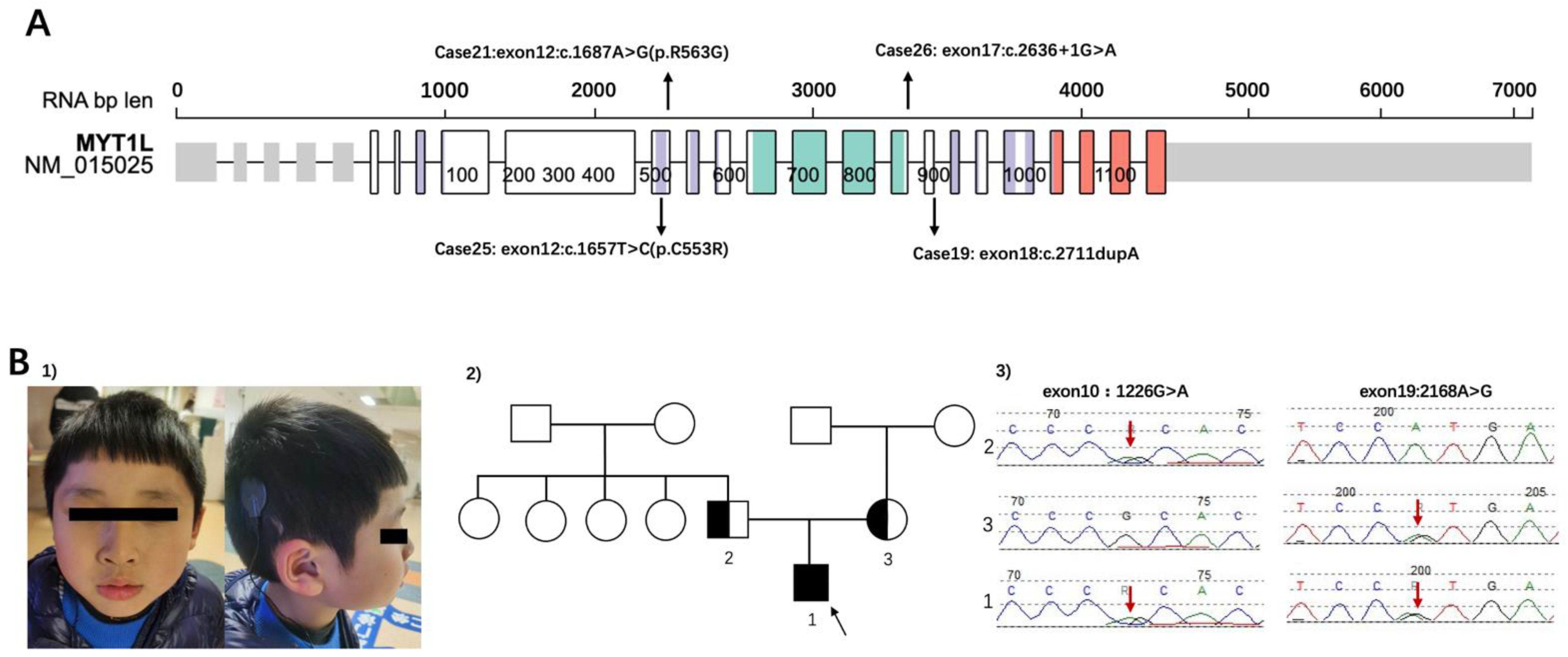
Among ASD patients who had identified SNVs, SHANK3 was the most common variant (n = 5). The second on the list was MYT1L, a myelin transcription factor, a transcription factor enabling fibroblast-to-neuron conversions. The inheritance of Cases 19/21/25/26 was de novo (Figure 3A). Tracing the developmental milestones, they all had obvious delayed motor developments. The independent walking for 4 children ranged from 20 to 30 months. Additionally, all children had significant language delay, most of them could speak less than 5 words even though they were over 3 years old. Abnormal sensory processing, such as biting, touching, or smelling objects was common in these children. Three patients (Case 19/21/25) were overweight, and ptyalism still existed owing to hypotonia. Case 21 also had strabismus. It was specifically observed that Cases 21/25/26 with MYT1L variants had apparent stereotypic hand movements. Analysis of characteristics in these patients can better screen and classify them in the clinic.
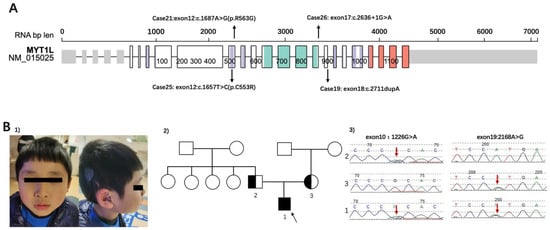
Figure 3.
(A) The locations of variants of patients in the study. Representation based on Protein Paint (Wisteria color: zf-C2HC domain, Turquoise color: MYT1 domain, Orange color: Smc domain) (https://proteinpaint.stjude.org/, accessed on 1 March 2022). The cases correspond with Table 1. (B) (1) The dysmorphic features of case 6 (shown for 1). (2) The Pedigree of family 1. 2 was the father of the patient; 3 was the mother of the patient. The black arrow showed the patient of case 6. (3) SLC26A4 sequence results of patient and his parents. Heterozygous variants of SLC26A4 were identified in the proband (red arrows in patient 1). Both his parents were in a heterozygous state for the variant (paternal: red arrow of 2 for c.1226G>A (p. R409H) in exon 10, maternal: red arrow of 3 for c.2168A>G(p. H723R) in exon 19).
3.2.2. Renewed Case: Genetic Diagnosis Should Also Focus on Clinical Manifestations
Case 6 was a boy with compound heterozygous variants of SLC26A4 (Figure 3B). The patient failed postnatal hearing screening and was inaudible to high-pitched sounds. He was diagnosed with bilateral profound sensorineural hearing loss and enlarged vestibular aqueduct syndrome soon afterwards. Right cochlear implants were performed when the child was 1 year old. He has been undertaking language training ever since. The patient was referred to our clinic at 4 years old because he behaved abnormally and undisciplined in kindergarten. His mother reported her child had a short attention span and hyperactivity. The Wechsler Preschool and Primary Scale of Intelligence (WPPSI) [22] showed that the child had developmental delay both in verbal and nonverbal intelligence (his IQS of Verbal Scale was 48, and IQS of Performance Scales was 62, producing a Full Scale IQ of 50). Compound heterozygous variants of SLC26A4 (c.1226G>A(p. R409H), c.2168A>G(p. H723R)) were detected in this patient by CTPS. The parents were validated by Sanger sequence. The results showed that his missense variants were both inherited by his parents (paternal: c.1226G>A (p. R409H) in exon 10, maternal: c.2168A>G(p. H723R) in exon 19). The patient had poor social communication and was suspected autistic, however, no reports have shown the correlations between the SLC26A4 gene and autism. Neither did he had interventions related to ASD, nor regularly followed up at the clinic. Atomoxetine hydrochloride was taken irregularly until school age. He had a re-examination at 7 years old. Maintaining Back-and-forth conversation was hard for him. He displayed poor social reciprocity and exhibited repetitive patterns of behavior (running back and forth repeatedly and watching traffic lights consistently) and sensory perception problems such as counting numbers and biting or smelling objects. His ADOS score was above the cutoff (social affect 5, repetitive score 3, severity 4). He was eventually diagnosed with ASD. The WES of the core family and CMA also performed according to his parents’ requirements but did not reveal any other causative variants.
4. Discussion
4.1. Clinical Benefits and Limits of CTPS
Genetics have a large contribution to ASD. Identifying a genetic etiology improving accuracy of counseling for patients and their families. Information about prognosis and recurrence risk is the most important benefit of genetic testing. The benefits also include preventing co-occurring medical conditions and avoiding unnecessary tests and harmful treatments [8]. It is necessary to advise patients and their families to undergo genetic evaluation.
Both rare inherited and de novo variations are relevant to ASD during early neurodevelopment. Kim et al. [23] provided evidence that rare inherited variations have a functional relationship with ASD in the developing brain. Sanders et al. [24] detected 2591 families from SSC and revealed that de novo variants were strongly associated with ASD, leading to a total of 6 risk loci (1q21.1, 3q29, 7q11.23, 16p11.2, 15q11.2-13, 22q11.2), and 65 ASD risk genes (additional 2 loci: NRXN1 (2p16.3), SHANK3(22q13.3) were included in the list of risk genes). The CNVs detected in our study overlapped with their observation in 3q29, 16p11.2, 15q11.2-13, 22q11.2, and 22q13.3. These CNVs had a high risk of developmental delay [25]. Satterstrom et al.‘s work [26] undertook the largest exome sequencing, identified 102 ASD risk genes, most of which had effects on the regulation of gene expression or neuronal communication. Of these genes, 19 of them overlapped with our study (ARID1B, ASXL3, BCL11A, CHD8, CTNNB1, DEAF1, FOXP1, FOXP2, KDM6B, MED13L, PPPP2R5D, PTEN, SCN2A, SETD5, SHANK2, SHANK3, SKI, STXBP1, TCF20), whereas they had not analyzed de novo mutations on chromosome X. For high-confidence risk genes reported in Choi’s study [27], we had 16 genes overlapped (ASXL3, BCL11A, CHD8, CTNNB1, DDX3X, DEAF1, FOXP1, FOXP2, KDM6B, MYT1L, PTEN, SCN2A, SETD5, SHANK3, SKI, TCF20). It’s worth noting that the contribution of de novo non-coding variants may not be as high as that of coding regions [28].
For now, CMA and Fragile X testing are recommended as the first tier for ASD patients. CMA tests abnormalities of chromosomal structure and duplications or deletions in chromosomal regions. The diagnostic yield of CNV is 5.4–14% (median 9%) in ASD patients [29,30]. When CMA does not find an etiology, the next step recommended for etiologic evaluation of ASD is WES. WES identified SNVs that have been confirmed as ASD risk genes. The reported diagnostic yield is 8–25.8% [29,31]. This process takes much expenditure and is a waste of time. CTPS, comprising 2742 genes, is able to analyze SNVs and CNVs simultaneously to provide an etiological diagnosis for children and patients. The potential of CTPS is that it can discover variants effectively and costs less. Reducing the cost of time and price is much needed in developing countries, especially in China. Families will easily accept and be willing to undergo genetic testing and finally benefit from it. As research progresses, genetic testing may contribute to identifying effective interventions related to specific etiologies.
Based on the results of CTPS in this study, we had an overall diagnostic yield of 19.16% for 569 ASD patients, including both SNVs and CNVs. A meta-analysis in 2020 identified 14 ASD studies across 1530 patients using targeted gene panel sequencing or WES, and the diagnostic yield was 17.1% (95% CI, 11–25%) [32]. Rossi et al. [31] recruited 163 ASD/autistic patients, all of whom had additional clinical features such as intellectual disabilities (ID)/developmental delays (DD) (92.6%) and epilepsy/seizures (38.7%). They found that the diagnostic rate of their ASD cohort was 25.8% using WES (42 of 163). Aspromonte et al. [33] developed a next-generation sequencing gene panel of 74 selected genes. They analyzed 150 individuals with ID and/or ASD, and a confident diagnosis was reached in 41 patients (27%). In our study, the diagnostic yield was slightly lower than reported previously. The possible reason is that it was a single-center analysis, which may have selection bias in the sample. The children were referred to our department with fewer comorbidities, such as epilepsy or multiple other malformations. CTPS detected selected genes, not including the whole exome/genome. Additionally, a small number of CNVs were not covered in the panel [16]. The CTPS was designed for children with syndrome and developmental abnormalities, not a targeted panel specialized for ASD children. However, it had comprehensive candidate genes other than ASD genes to avoid missing suspected disease-causing genes. The restriction could explain the lower rate of our study.
Clinicians should focus more on the clinical features of children and find more valuable clues to the cause of the disease. CTPS offers a better choice for patients and clinicians as an effective molecular diagnostic tool.
4.2. Implications for Representative Cases from CTPS
Myelin transcription factor 1-like (MYT1L), mapping to a region of human chromosome 2, encodes the MYT1L protein, which contains 6 zinc fingers (an N-terminal zinc finger, 2 tandem central zinc fingers, and 3 C-terminal zinc fingers) [34]. The MYT1L gene is co-expressed with other ASD-related genes, including NRXN1, TCF4, and BCL11A in the prefrontal cortex during mid-fetal development, higher in prenatal expression [35], and transcription may persist in the brains of children. It plays a crucial role in neurogenesis, helps neural stem cells transform into neurons, and is expressed in oligodendrocyte linage cells during myelination and remyelination [36]. We found 4 children with de novo MYT1L variants. Coursimault et al. [37] reported 40 patients with MYT1L-associated neurodevelopmental disorder and reviewed 22 patients in published data. In their reports, many patients with MYT1L variants had language delay (95%), ID (70%), ASD (43%), motor delay (78%), and hypotonia (47%). Consistent with the previous literature, all of our 4 patients with ASD had developmental delays, overweight/obesity were common in these patients. Table 3 shows the clinical symptoms of our patients and public literature. All of our patients had abnormal sensory processing, whereas few studies mentioned it. Notably, 3 of our patients had stereotypic hand movements, which were not detailed in Juliette et al.’s work. De Rocker et al. [38] showed 3 patients with rigid hand movements, which was also manifested in our patients. These children had special hand phenotypes (typical repetitive, purposeless hand movements such as rubbing, tapping, wringing, or clapping) without rapid regression of acquired skills and stagnation. Therefore, a detailed history is particularly important. Our patients had obvious sensory abnormalities and stereotypic hand movements, which may contribute to the phenotype of MYT1L.

Table 3.
Clinical symptoms of MYT1L patients and published literature.
Solute carrier family 26 member 4 (SLC26A4), encoding pendrin (an anion transporter) [44], causes Pendred syndrome and an enlarged vestibular aqueduct. It is expressed mostly in the thyroid and is also expressed at some levels in the prostate, kidney, urinary bladder, and brain [45]. Pedred Syndrome is a common disorder with hereditary hearing loss, abnormal development of the cochlea, and diffuse thyroid enlargement. The patient in our study had SLC26A4 heterozygous variants. Due to the lack of literature connecting SLC26A4 with autism, CMA and WES were performed. The patient had typical autistic features but found no cause other than SLC26A4 variants. He had a complex phenotype beyond Pendred syndrome presentation, hence, our study reported an ASD child with SLC26A4 variants. It is hard for us to explain his sensory perception abnormalities and repetitive behaviors with hearing problems. SLC26A4 variants may cause his autistic phenotype, while early social competence may be overlooked by deafness. Nevertheless, further studies and case reports are required to show the relationships between the SLC26A4 gene and phenotype with ASD. After all, some of the variants of uncertain significance may be determined as pathogenic in the future [8]. It is hoped that more animal research will be conducted to examine the effect of scl26a4 on autism and on the gene-phenotype associations in neurodevelopment.
5. Conclusions
In summary, CTPS is an effective tool for genetic testing of ASD patients, a valuable strategy suitable for patients with neurodevelopmental disorders in China. It is important to identify the key clinical features of patients diagnosed with ASD that allow more accurate genetic diagnosis and harbor relevant genetic variants. It is expected that genetic research will allow the development of better treatments and interventions for ASD children within the next decade, thus guiding further family planning and social support.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/genes13061010/s1, Table S1: title Detailed CNVs of ASD patients from CTPS.
Author Contributions
Q.X. and C.H. conceived and designed the study; L.H. and G.Z. carried out sample collections, C.H., Y.D., H.L., K.Z., D.L., B.W., X.X., and Q.X. identified patients; C.H. wrote the manuscript, Q.X. and X.X. modified the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded in part by the National Natural Science Foundation of China (NSFC, No. 82171540), the Key Subject Construction Project of Shanghai Municipal Health Commission (No. shslczdzk02903) and the Young Clinical Scientist Program of Children’s Hospital of Fudan university (No. 2022LCKXJ03).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Ethics Committee of the Children’s Hospital of Fudan University (Approval number: 2016-77, approval date: 25 February 2016).
Informed Consent Statement
Written informed consent was obtained from patients or parents giving consent on behalf of their minor children.
Data Availability Statement
The data presented in this study are available on request from the corresponding author. The confidential patient data will not be shared.
Acknowledgments
We thank our colleagues at the Child Health Care of Children’s Hospital of Fudan University for their help. We thank Xia Wang from Ailife Diagnostics for his comments and Xinran Dong from Clinical Genetic Center, Children’s Hospital of Fudan University for her help on the writing of the Material and Methods Section.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lord, C.; Elsabbagh, M.; Baird, G.; Veenstra-Vanderweele, J. Autism Spectrum Disorder. Lancet 2018, 392, 508–520. [Google Scholar] [CrossRef]
- Brugha, T.S.; Spiers, N.; Bankart, J.; Cooper, S.A.; McManus, S.; Scott, F.J.; Smith, J.; Tyrer, F. Epidemiology of Autism in Adults across Age Groups and Ability Levels. Br. J. Psychiatry 2016, 209, 498–503. [Google Scholar] [CrossRef] [Green Version]
- Lord, C.; Brugha, T.S.; Charman, T.; Cusack, J.; Dumas, G.; Frazier, T.; Jones, E.J.H.; Jones, R.M.; Pickles, A.; State, M.W.; et al. Autism Spectrum Disorder. Nat. Rev. Dis. Primers 2020, 6, 5. [Google Scholar] [CrossRef] [PubMed]
- Tick, B.; Bolton, P.; Happe, F.; Rutter, M.; Rijsdijk, F. Heritability of Autism Spectrum Disorders: A Meta-Analysis of Twin Studies. J. Child Psychol. Psychiatry 2016, 57, 585–595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woodbury-Smith, M.; Scherer, S.W. Progress in the Genetics of Autism Spectrum Disorder. Dev. Med. Child Neurol. 2018, 60, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Sandin, S.; Lichtenstein, P.; Kuja-Halkola, R.; Larsson, H.; Hultman, C.M.; Reichenberg, A. The Familial Risk of Autism. JAMA 2014, 311, 1770–1777. [Google Scholar] [CrossRef]
- He, X.; Sanders, S.J.; Liu, L.; de Rubeis, S.; Lim, E.T.; Sutcliffe, J.S.; Schellenberg, G.D.; Gibbs, R.A.; Daly, M.J.; Buxbaum, J.D.; et al. Integrated Model of De Novo and Inherited Genetic Variants Yields Greater Power to Identify Risk Genes. PLoS Genet. 2013, 9, e1003671. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hyman, S.L.; Levy, S.E.; Myers, S.M.; Section On Developmental Council On Children With Disabilities, and Pediatrics Behavioral. Identification, Evaluation, and Management of Children with Autism Spectrum Disorder. Pediatrics 2020, 145. [Google Scholar] [CrossRef] [Green Version]
- Volkmar, F.; Siegel, M.; Woodbury-Smith, M.; King, B.; McCracken, J.; State, M. Child American Academy of, and Issues Adolescent Psychiatry Committee on Quality. Practice Parameter for the Assessment and Treatment of Children and Adolescents with Autism Spectrum Disorder. J. Am. Acad. Child Adolesc. Psychiatry 2014, 53, 237–257. [Google Scholar] [CrossRef] [Green Version]
- Schaefer, G.B.; Mendelsohn, N.J. Professional Practice and Guidelines Committee. Clinical Genetics Evaluation in Identifying the Etiology of Autism Spectrum Disorders: 2013 Guideline Revisions. Genet. Med. 2013, 15, 399–407. [Google Scholar] [CrossRef] [Green Version]
- O’Roak, B.J.; Vives, L.; Fu, W.; Egertson, J.D.; Stanaway, I.B.; Phelps, I.G.; Carvill, G.; Kumar, A.; Lee, C.; Ankenman, K.; et al. Multiplex Targeted Sequencing Identifies Recurrently Mutated Genes in Autism Spectrum Disorders. Science 2012, 338, 1619–1622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Srivastava, S.; Love-Nichols, J.A.; Dies, K.A.; Ledbetter, D.H.; Martin, C.L.; Chung, W.K.; Firth, H.V.; Frazier, T.; Hansen, R.L.; Prock, L.; et al. Correction: Meta-Analysis and Multidisciplinary Consensus Statement: Exome Sequencing Is a First-Tier Clinical Diagnostic Test for Individuals with Neurodevelopmental Disorders. Genet. Med. 2020, 22, 1731–1732. [Google Scholar] [CrossRef] [PubMed]
- Lowther, C.; Valkanas, E.; Giordano, J.L.; Wang, H.Z.; Currall, B.B.; O’Keefe, K.; Collins, R.L.; Zhao, X.; Austin-Tse, C.A.; Evangelista, E.; et al. Systematic Evaluation of Genome Sequencing as a First-Tier Diagnostic Test for Prenatal and Pediatric Disorders. bioRxiv 2020. [Google Scholar] [CrossRef]
- Coury, D. Medical Treatment of Autism Spectrum Disorders. Curr. Opin. Neurol. 2010, 23, 131–136. [Google Scholar] [CrossRef] [PubMed]
- APAd American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Dong, X.; Liu, B.; Yang, L.; Wang, H.; Wu, B.; Liu, R.; Chen, H.; Chen, X.; Yu, S.; Chen, B.; et al. Clinical Exome Sequencing as the First-Tier Test for Diagnosing Developmental Disorders Covering Both Cnv and Snv: A Chinese Cohort. J. Med. Genet. 2020, 57, 558–566. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Kong, Y.; Dong, X.; Hu, L.; Lin, Y.; Chen, X.; Ni, Q.; Lu, Y.; Wu, B.; Wang, H.; et al. Clinical and Genetic Spectrum of a Large Cohort of Children with Epilepsy in China. Genet. Med. 2019, 21, 564–571. [Google Scholar] [CrossRef]
- McLaren, W.; Pritchard, B.; Rios, D.; Chen, Y.; Flicek, P.; Cunningham, F. Deriving the Consequences of Genomic Variants with the Ensembl Api and Snp Effect Predictor. Bioinformatics 2010, 26, 2069–2070. [Google Scholar] [CrossRef]
- Wang, K.; Li, M.; Hakonarson, H. Annovar: Functional Annotation of Genetic Variants from High-Throughput Sequencing Data. Nucleic Acids Res. 2010, 38, e164. [Google Scholar] [CrossRef]
- Qian, Q.I.N.; Bo, L.I.U.; Lin, Y.A.N.G.; Bing-bing, W.U.; Hui-jun, W.A.N.G.; Xin-Ran, D.O.N.G.; Yu-lan, L.U.; Wen-Hao, Z.H.O.U. Application of Copy Number Variation Screening Analysis Process Based on High Throughput Sequencing Technology. Chin. J. Evid. Based Pediatrics 2018, 13, 275–279. [Google Scholar]
- Lord, C.M.; Rutter, P.D.; Labore, S.; Risi, K.; Gotham, B.S. Autism Diagnostic Observation Schedule, (Ados-2), 2nd ed.; Western Pschological Services: Los Angeles, CA, USA, 2012. [Google Scholar]
- David, W. Wechsler Preschool and Primary Scale of Intelligence, 4th ed.; Pearson: London, UK, 2012. [Google Scholar]
- Kim, Y.; An, J.Y. Spatio-Temporal Roles of Asd-Associated Variants in Human Brain Development. Genes 2020, 11, 535. [Google Scholar] [CrossRef]
- Sanders, S.J.; He, X.; Willsey, A.J.; Ercan-Sencicek, A.G.; Samocha, K.E.; Cicek, A.E.; Murtha, M.T.; Bal, V.H.; Bishop, S.L.; Dong, S.; et al. Insights into Autism Spectrum Disorder Genomic Architecture and Biology from 71 Risk Loci. Neuron 2015, 87, 1215–1233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, J.Y.; Sanders, S.J. Appreciating the Population-Wide Impact of Copy Number Variants on Cognition. Biol. Psychiatry 2017, 82, 78–80. [Google Scholar] [CrossRef] [PubMed]
- Satterstrom, F.K.; Kosmicki, J.A.; Wang, J.; Breen, M.S.; De Rubeis, S.; An, J.Y.; Peng, M.; Collins, R.; Grove, J.; Klei, L.; et al. Large-Scale Exome Sequencing Study Implicates Both Developmental and Functional Changes in the Neurobiology of Autism. Cell 2020, 180, 568–584.e23. [Google Scholar] [CrossRef]
- Choi, L.; An, J.Y. Genetic Architecture of Autism Spectrum Disorder: Lessons from Large-Scale Genomic Studies. Neurosci. Biobehav. Rev. 2021, 128, 244–257. [Google Scholar] [CrossRef]
- Werling, D.M.; Brand, H.; An, J.Y.; Stone, M.R.; Zhu, L.; Glessner, J.T.; Collins, R.L.; Dong, S.; Layer, R.M.; Markenscoff-Papadimitriou, E.; et al. An Analytical Framework for Whole-Genome Sequence Association Studies and Its Implications for Autism Spectrum Disorder. Nat. Genet. 2018, 50, 727–736. [Google Scholar] [CrossRef]
- Tammimies, K.; Marshall, C.R.; Walker, S.; Kaur, G.; Thiruvahindrapuram, B.; Lionel, A.C.; Yuen, R.; Uddin, M.; Roberts, W.; Weksberg, R.; et al. Molecular Diagnostic Yield of Chromosomal Microarray Analysis and Whole-Exome Sequencing in Children with Autism Spectrum Disorder. JAMA 2015, 314, 895–903. [Google Scholar] [CrossRef]
- Ho, K.S.; Wassman, E.R.; Baxter, A.L.; Hensel, C.H.; Martin, M.M.; Prasad, A.; Twede, H.; Vanzo, R.J.; Butler, M.G. Chromosomal Microarray Analysis of Consecutive Individuals with Autism Spectrum Disorders Using an Ultra-High Resolution Chromosomal Microarray Optimized for Neurodevelopmental Disorders. Int. J. Mol. Sci. 2016, 17, 2070. [Google Scholar] [CrossRef] [Green Version]
- Rossi, M.; El-Khechen, D.; Black, M.H.; Hagman, K.D.F.; Tang, S.; Powis, Z. Outcomes of Diagnostic Exome Sequencing in Patients with Diagnosed or Suspected Autism Spectrum Disorders. Pediatr. Neurol. 2017, 70, 34–43.e2. [Google Scholar] [CrossRef] [Green Version]
- Stefanski, A.; Calle-López, Y.; Leu, C.; Pérez-Palma, E.; Pestana-Knight, E.; Lal, D. Clinical Sequencing Yield in Epilepsy, Autism Spectrum Disorder, and Intellectual Disability: A Systematic Review and Meta-Analysis. Epilepsia 2021, 62, 143–151. [Google Scholar] [CrossRef]
- Aspromonte, M.C.; Bellini, M.; Gasparini, A.; Carraro, M.; Bettella, E.; Polli, R.; Cesca, F.; Bigoni, S.; Boni, S.; Carlet, O.; et al. Characterization of Intellectual Disability and Autism Comorbidity through Gene Panel Sequencing. Hum. Mutat. 2020, 41, 1183. [Google Scholar] [CrossRef]
- Kim, J.G.; Armstrong, R.C.; Agoston, D.V.; Robinsky, A.; Wiese, C.; Nagle, J.; Hudson, L.D. Myelin Transcription Factor 1 (Myt1) of the Oligodendrocyte Lineage, Along with a Closely Related Cchc Zinc Finger, Is Expressed in Developing Neurons in the Mammalian Central Nervous System. J. Neurosci. Res. 1997, 50, 272–290. [Google Scholar] [CrossRef] [Green Version]
- Werling, D.M.; Pochareddy, S.; Choi, J.; An, J.-Y.; Sheppard, B.; Peng, M.; Li, Z.; Dastmalchi, C.; Santpere, G.; Sousa, A.M.; et al. Whole-Genome and Rna Sequencing Reveal Variation and Transcriptomic Coordination in the Developing Human Prefrontal Cortex. Cell Rep. 2020, 31, 107489. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Shao, Q.; Li, Z.; Gonzalez, G.A.; Lu, F.; Wang, D.; Pu, Y.; Huang, A.; Zhao, C.; He, C.; et al. Myt1l Promotes Differentiation of Oligodendrocyte Precursor Cells and Is Necessary for Remyelination after Lysolecithin-Induced Demyelination. Neurosci. Bull. 2018, 34, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Coursimault, J.; Guerrot, A.M.; Morrow, M.M.; Schramm, C.; Zamora, F.M.; Shanmugham, A.; Liu, S.; Zou, F.; Bilan, F.; Le Guyader, G.; et al. Myt1l-Associated Neurodevelopmental Disorder: Description of 40 New Cases and Literature Review of Clinical and Molecular Aspects. Hum. Genet. 2021, 141, 65–80. [Google Scholar] [CrossRef] [PubMed]
- De Rocker, N.; Vergult, S.; Koolen, D.; Jacobs, E.; Hoischen, A.; Zeesman, S.; Bang, B.; Béna, F.; Bockaert, N.; Bongers, E.M.; et al. Refinement of the Critical 2p25.3 Deletion Region: The Role of Myt1l in Intellectual Disability and Obesity. Genet. Med. 2015, 17, 460–466. [Google Scholar] [CrossRef] [Green Version]
- Windheuser, I.C.; Becker, J.; Cremer, K.; Hundertmark, H.; Yates, L.M.; Mangold, E.; Peters, S.; Degenhardt, F.; Ludwig, K.U.; Zink, A.M.; et al. Nine Newly Identified Individuals Refine the Phenotype Associated with Myt1l Mutations. Am. J. Med. Genet. A 2020, 182, 1021–1031. [Google Scholar] [CrossRef] [Green Version]
- Blanchet, P.; Bebin, M.; Bruet, S.; Cooper, G.M.; Thompson, M.L.; Duban-Bedu, B.; Gerard, B.; Piton, A.; Suckno, S.; Deshpande, C.; et al. Myt1l Mutations Cause Intellectual Disability and Variable Obesity by Dysregulating Gene Expression and Development of the Neuroendocrine Hypothalamus. PLoS Genet. 2017, 13, e1006957. [Google Scholar] [CrossRef] [Green Version]
- Carvalho, L.M.; D’Angelo, C.S.; Mustacchi, Z.; da Silva, I.T.; Krepischi, A.C.V.; Koiffmann, C.P.; Rosenberg, C. A Novel Myt1l Mutation in a Boy with Syndromic Obesity: Case Report and Literature Review. Obes. Res. Clin. Pract. 2021, 15, 124–132. [Google Scholar] [CrossRef]
- Loid, P.; Makitie, R.; Costantini, A.; Viljakainen, H.; Pekkinen, M.; Makitie, O. A Novel Myt1l Mutation in a Patient with Severe Early-Onset Obesity and Intellectual Disability. Am. J. Med. Genet. A 2018, 176, 1972–1975. [Google Scholar] [CrossRef]
- Al Tuwaijri, A.; Alfadhel, M. Myt1l Mutation in a Patient Causes Intellectual Disability and Early Onset of Obesity: A Case Report and Review of the Literature. J. Pediatr. Endocrinol. Metab. 2019, 32, 409–413. [Google Scholar] [CrossRef]
- Everett, L.A.; Glaser, B.; Beck, J.C.; Idol, J.R.; Buchs, A.; Heyman, M.A.; Adawi, F.; Hazani, E.; Nassir, E.; Baxevanis, A.D.; et al. Pendred Syndrome Is Caused by Mutations in a Putative Sulphate Transporter Gene (Pds). Nat. Genet. 1997, 17, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Fagerberg, L.; Hallström, B.M.; Oksvold, P.; Kampf, C.; Djureinovic, D.; Odeberg, J.; Habuka, M.; Tahmasebpoor, S.; Danielsson, A.; Edlund, K.; et al. Analysis of the Human Tissue-Specific Expression by Genome-Wide Integration of Transcriptomics and Antibody-Based Proteomics. Mol. Cell. Proteom. 2014, 13, 397–406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).