Differential DNA Methylation Landscape in Skin Fibroblasts from African Americans with Systemic Sclerosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Primary Dermal Fibroblast Isolation and Culture
2.3. Reduced Representation Bisulfite Sequencing (RRBS)
2.4. Genome-Wide DNA Methylation Data Analysis
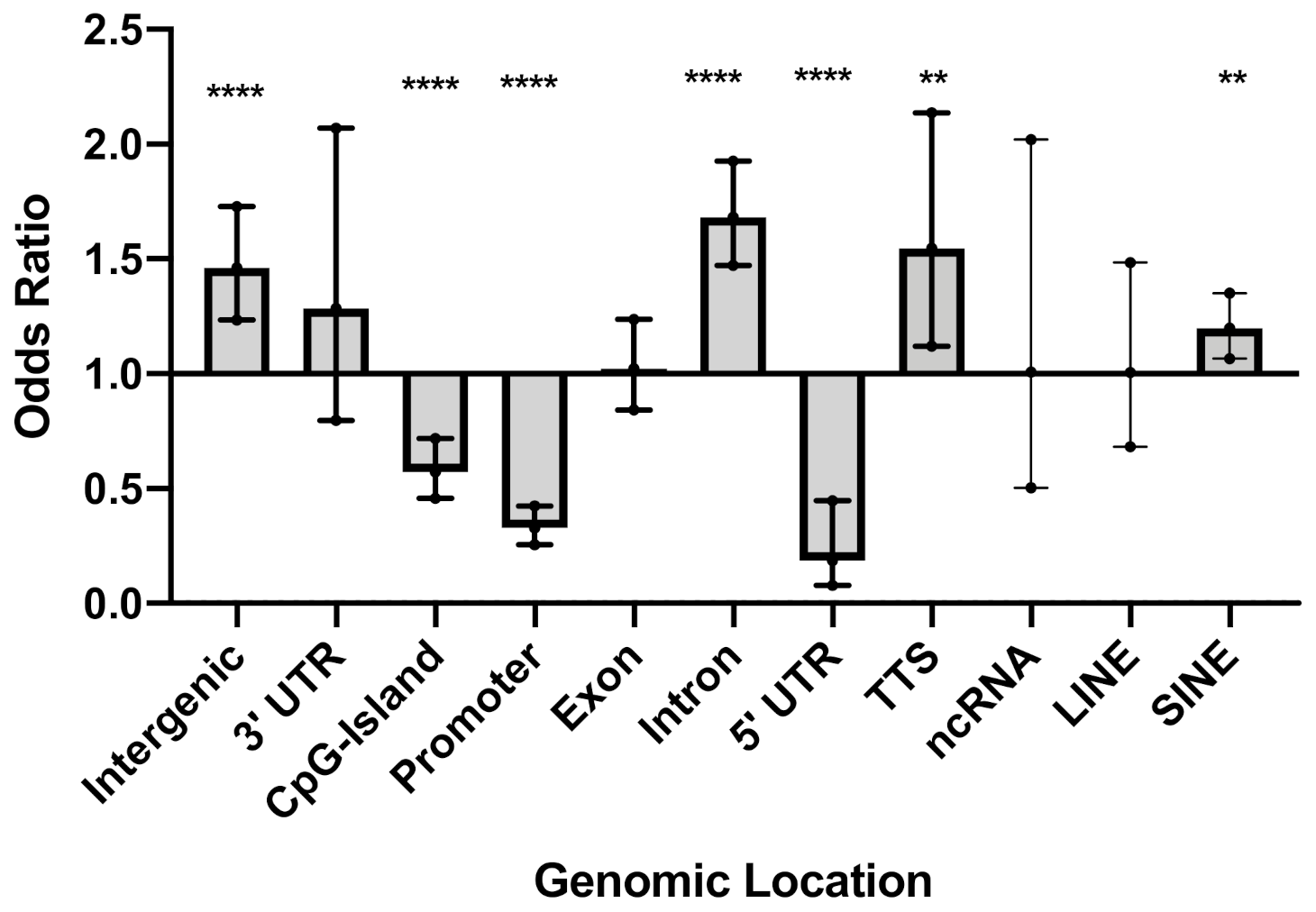
2.5. Genomic Annotation Enrichment Analysis
2.6. Gene Set Enrichment Analysis (GSEA)
2.7. Gene Ontology (GO) Enrichment Analysis
2.8. Gene Expression Analysis
3. Results
3.1. Subject Characteristics
3.2. Differentially Methylated Sites and Genes
3.3. Gene Set Enrichment Analysis (GSEA)
3.4. Gene Ontology (GO) Enrichment Analysis
3.5. Comparison of DNA Methylation with Previous Reports in Dermal Tissues
3.6. Gene Expression of Differentially Methylated Genes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Silver, R.M. Clinical aspects of systemic sclerosis (scleroderma). Ann. Rheum. Dis. 1991, 50 (Suppl. 4), 854–861. [Google Scholar] [CrossRef]
- Mayes, M.D.; Lacey, J.V., Jr.; Beebe-Dimmer, J.; Gillespie, B.W.; Cooper, B.; Laing, T.J.; Schottenfeld, D. Prevalence, incidence, survival, and disease characteristics of systemic sclerosis in a large US population. Arthritis Rheumatol. 2003, 48, 2246–2255. [Google Scholar] [CrossRef]
- Mendoza, F.; Derk, C.T. Systemic sclerosis mortality in the United States: 1999-2002 implications for patient care. J. Clin. Rheumatol. Pract. Rep. Rheum. Musculoskelet. Dis. 2007, 13, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Laing, T.J.; Gillespie, B.W.; Toth, M.B.; Mayes, M.D.; Gallavan, R.H., Jr.; Burns, C.J.; Johanns, J.R.; Cooper, B.C.; Keroack, B.J.; Wasko, M.C.; et al. Racial differences in scleroderma among women in Michigan. Arthritis Rheumatol. 1997, 40, 734–742. [Google Scholar] [CrossRef] [PubMed]
- Greidinger, E.L.; Flaherty, K.T.; White, B.; Rosen, A.; Wigley, F.M.; Wise, R.A. African-American race and antibodies to topoisomerase I are associated with increased severity of scleroderma lung disease. Chest 1998, 114, 801–807. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, E.; Furst, D.E. Systemic sclerosis mortality in the United States: 1979–1998. Eur. J. Epidemiol. 2005, 20, 855–861. [Google Scholar] [CrossRef] [PubMed]
- McNearney, T.A.; Reveille, J.D.; Fischbach, M.; Friedman, A.W.; Lisse, J.R.; Goel, N.; Tan, F.K.; Zhou, X.; Ahn, C.; Feghali-Bostwick, C.A.; et al. Pulmonary involvement in systemic sclerosis: Associations with genetic, serologic, sociodemographic, and behavioral factors. Arthritis Rheumatol. 2007, 57, 318–326. [Google Scholar] [CrossRef] [PubMed]
- Nietert, P.J.; Mitchell, H.C.; Bolster, M.B.; Shaftman, S.R.; Tilley, B.C.; Silver, R.M. Racial variation in clinical and immunological manifestations of systemic sclerosis. J. Rheumatol. 2006, 33, 263–268. [Google Scholar]
- Morgan, N.D.; Shah, A.A.; Mayes, M.D.; Domsic, R.T.; Medsger, T.A., Jr.; Steen, V.D.; Varga, J.; Carns, M.; Ramos, P.S.; Silver, R.M.; et al. Clinical and serological features of systemic sclerosis in a multicenter African American cohort: Analysis of the genome research in African American scleroderma patients clinical database. Medicine 2017, 96, e8980. [Google Scholar] [CrossRef]
- Angiolilli, C.; Marut, W.; van der Kroef, M.; Chouri, E.; Reedquist, K.A.; Radstake, T. New insights into the genetics and epigenetics of systemic sclerosis. Nat. Rev. Rheumatol. 2018, 14, 657–673. [Google Scholar] [CrossRef]
- Selmi, C.; Feghali-Bostwick, C.A.; Lleo, A.; Lombardi, S.A.; De Santis, M.; Cavaciocchi, F.; Zammataro, L.; Mitchell, M.M.; Lasalle, J.M.; Medsger, T., Jr.; et al. X chromosome gene methylation in peripheral lymphocytes from monozygotic twins discordant for scleroderma. Clin. Exp. Immunol. 2012, 169, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Matatiele, P.; Tikly, M.; Tarr, G.; Gulumian, M. DNA methylation similarities in genes of black South Africans with systemic lupus erythematosus and systemic sclerosis. J. Biomed. Sci. 2015, 22, 34. [Google Scholar] [CrossRef] [PubMed]
- Altorok, N.; Tsou, P.S.; Coit, P.; Khanna, D.; Sawalha, A.H. Genome-wide DNA methylation analysis in dermal fibroblasts from patients with diffuse and limited systemic sclerosis reveals common and subset-specific DNA methylation aberrancies. Ann. Rheum. Dis. 2015, 74, 1612–1620. [Google Scholar] [CrossRef] [PubMed]
- Ramos, P.S.; Zimmerman, K.D.; Haddad, S.; Langefeld, C.D.; Medsger, T.A., Jr.; Feghali-Bostwick, C.A. Integrative analysis of DNA methylation in discordant twins unveils distinct architectures of systemic sclerosis subsets. Clin. Epigenetics 2019, 11, 58. [Google Scholar] [CrossRef]
- Lu, T.; Klein, K.O.; Colmegna, I.; Lora, M.; Greenwood, C.M.T.; Hudson, M. Whole-genome bisulfite sequencing in systemic sclerosis provides novel targets to understand disease pathogenesis. BMC Med. Genom. 2019, 12, 144. [Google Scholar] [CrossRef]
- Li, T.; Ortiz-Fernandez, L.; Andres-Leon, E.; Ciudad, L.; Javierre, B.M.; Lopez-Isac, E.; Guillen-Del-Castillo, A.; Simeon-Aznar, C.P.; Ballestar, E.; Martin, J. Epigenomics and transcriptomics of systemic sclerosis CD4+ T cells reveal long-range dysregulation of key inflammatory pathways mediated by disease-associated susceptibility loci. Genome Med. 2020, 12, 81. [Google Scholar] [CrossRef]
- Michels, K.B.; Binder, A.M.; Dedeurwaerder, S.; Epstein, C.B.; Greally, J.M.; Gut, I.; Houseman, E.A.; Izzi, B.; Kelsey, K.T.; Meissner, A.; et al. Recommendations for the design and analysis of epigenome-wide association studies. Nat. Methods 2013, 10, 949–955. [Google Scholar] [CrossRef]
- Barfield, R.T.; Almli, L.M.; Kilaru, V.; Smith, A.K.; Mercer, K.B.; Duncan, R.; Klengel, T.; Mehta, D.; Binder, E.B.; Epstein, M.P.; et al. Accounting for population stratification in DNA methylation studies. Genet. Epidemiol. 2014, 38, 231–241. [Google Scholar] [CrossRef]
- Galanter, J.M.; Gignoux, C.R.; Oh, S.S.; Torgerson, D.; Pino-Yanes, M.; Thakur, N.; Eng, C.; Hu, D.; Huntsman, S.; Farber, H.J.; et al. Differential methylation between ethnic sub-groups reflects the effect of genetic ancestry and environmental exposures. eLife 2017, 6, e20532. [Google Scholar] [CrossRef]
- Husquin, L.T.; Rotival, M.; Fagny, M.; Quach, H.; Zidane, N.; McEwen, L.M.; MacIsaac, J.L.; Kobor, M.S.; Aschard, H.; Patin, E.; et al. Exploring the genetic basis of human population differences in DNA methylation and their causal impact on immune gene regulation. Genome Biol. 2018, 19, 222. [Google Scholar] [CrossRef]
- Quach, H.; Rotival, M.; Pothlichet, J.; Loh, Y.E.; Dannemann, M.; Zidane, N.; Laval, G.; Patin, E.; Harmant, C.; Lopez, M.; et al. Genetic Adaptation and Neandertal Admixture Shaped the Immune System of Human Populations. Cell 2016, 167, 643–656.e17. [Google Scholar] [CrossRef] [PubMed]
- Gopalan, S.; Carja, O.; Fagny, M.; Patin, E.; Myrick, J.W.; McEwen, L.M.; Mah, S.M.; Kobor, M.S.; Froment, A.; Feldman, M.W.; et al. Trends in DNA Methylation with Age Replicate across Diverse Human Populations. Genetics 2017, 206, 1659–1674. [Google Scholar] [CrossRef] [PubMed]
- Fagny, M.; Patin, E.; MacIsaac, J.L.; Rotival, M.; Flutre, T.; Jones, M.J.; Siddle, K.J.; Quach, H.; Harmant, C.; McEwen, L.M.; et al. The epigenomic landscape of African rainforest hunter-gatherers and farmers. Nat. Commun. 2015, 6, 10047. [Google Scholar] [CrossRef] [PubMed]
- Heyn, H.; Moran, S.; Hernando-Herraez, I.; Sayols, S.; Gomez, A.; Sandoval, J.; Monk, D.; Hata, K.; Marques-Bonet, T.; Wang, L.; et al. DNA methylation contributes to natural human variation. Genome Res. 2013, 23, 1363–1372. [Google Scholar] [CrossRef]
- Gu, H.; Bock, C.; Mikkelsen, T.S.; Jager, N.; Smith, Z.D.; Tomazou, E.; Gnirke, A.; Lander, E.S.; Meissner, A. Genome-scale DNA methylation mapping of clinical samples at single-nucleotide resolution. Nat. Methods 2010, 7, 133–136. [Google Scholar] [CrossRef]
- Bock, C.; Tomazou, E.M.; Brinkman, A.B.; Muller, F.; Simmer, F.; Gu, H.; Jager, N.; Gnirke, A.; Stunnenberg, H.G.; Meissner, A. Quantitative comparison of genome-wide DNA methylation mapping technologies. Nat. Biotechnol. 2010, 28, 1106–1114. [Google Scholar] [CrossRef]
- Atanelishvili, I.; Shirai, Y.; Akter, T.; Buckner, T.; Noguchi, A.; Silver, R.M.; Bogatkevich, G.S. M10, a caspase cleavage product of the hepatocyte growth factor receptor, interacts with Smad2 and demonstrates antifibrotic properties in vitro and in vivo. Transl. Res. 2016, 170, 99–111. [Google Scholar] [CrossRef]
- Krueger, F.; Andrews, S.R. Bismark: A flexible aligner and methylation caller for Bisulfite-Seq applications. Bioinformatics (Oxf. Engl.) 2011, 27, 1571–1572. [Google Scholar] [CrossRef]
- Assenov, Y.; Muller, F.; Lutsik, P.; Walter, J.; Lengauer, T.; Bock, C. Comprehensive analysis of DNA methylation data with RnBeads. Nat. Methods 2014, 11, 1138–1140. [Google Scholar] [CrossRef]
- Heinz, S.; Benner, C.; Spann, N.; Bertolino, E.; Lin, Y.C.; Laslo, P.; Cheng, J.X.; Murre, C.; Singh, H.; Glass, C.K. Simple combinations of lineage-determining transcription factors prime cis-regulatory elements required for macrophage and B cell identities. Mol. Cell 2010, 38, 576–589. [Google Scholar] [CrossRef]
- Subramanian, A.; Kuehn, H.; Gould, J.; Tamayo, P.; Mesirov, J.P. GSEA-P: A desktop application for Gene Set Enrichment Analysis. Bioinformatics 2007, 23, 3251–3253. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene set enrichment analysis: A knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef] [PubMed]
- Fabregat, A.; Jupe, S.; Matthews, L.; Sidiropoulos, K.; Gillespie, M.; Garapati, P.; Haw, R.; Jassal, B.; Korninger, F.; May, B.; et al. The Reactome Pathway Knowledgebase. Nucleic Acids Res. 2018, 46, D649–D655. [Google Scholar] [CrossRef] [PubMed]
- Irish, J.C.; Mills, J.N.; Turner-Ivey, B.; Wilson, R.C.; Guest, S.T.; Rutkovsky, A.; Dombkowski, A.; Kappler, C.S.; Hardiman, G.; Ethier, S.P. Amplification of WHSC1L1 regulates expression and estrogen-independent activation of ERalpha in SUM-44 breast cancer cells and is associated with ERalpha over-expression in breast cancer. Mol. Oncol. 2016, 10, 850–865. [Google Scholar] [CrossRef]
- Hardiman, G.; Savage, S.J.; Hazard, E.S.; Wilson, R.C.; Courtney, S.M.; Smith, M.T.; Hollis, B.W.; Halbert, C.H.; Gattoni-Celli, S. Systems analysis of the prostate transcriptome in African-American men compared with European-American men. Pharmacogenomics 2016, 17, 1129–1143. [Google Scholar] [CrossRef]
- Kuhn, K.; Bertling, W.M.; Emmrich, F. Cloning of a functional cDNA for human cytidine deaminase (CDD) and its use as a marker of monocyte/macrophage differentiation. Biochem. Biophys. Res. Commun. 1993, 190, 1–7. [Google Scholar] [CrossRef]
- Nibbs, R.J.; Graham, G.J. Immune regulation by atypical chemokine receptors. Nat. Reviews. Immunol. 2013, 13, 815–829. [Google Scholar] [CrossRef]
- Weiss, K.M.; Ruddle, F.H.; Bollekens, J. Dlx and other homeobox genes in the morphological development of the dentition. Connect. Tissue Res. 1995, 32, 35–40. [Google Scholar] [CrossRef]
- Skaug, B.; Khanna, D.; Swindell, W.R.; Hinchcliff, M.E.; Frech, T.M.; Steen, V.D.; Hant, F.N.; Gordon, J.K.; Shah, A.A.; Zhu, L.; et al. Global skin gene expression analysis of early diffuse cutaneous systemic sclerosis shows a prominent innate and adaptive inflammatory profile. Ann. Rheum. Dis. 2020, 79, 379–386. [Google Scholar] [CrossRef]
- Whitfield, M.L.; Finlay, D.R.; Murray, J.I.; Troyanskaya, O.G.; Chi, J.T.; Pergamenschikov, A.; McCalmont, T.H.; Brown, P.O.; Botstein, D.; Connolly, M.K. Systemic and cell type-specific gene expression patterns in scleroderma skin. Proc. Natl. Acad. Sci. USA 2003, 100, 12319–12324. [Google Scholar] [CrossRef]
- Gardner, H.; Shearstone, J.R.; Bandaru, R.; Crowell, T.; Lynes, M.; Trojanowska, M.; Pannu, J.; Smith, E.; Jablonska, S.; Blaszczyk, M.; et al. Gene profiling of scleroderma skin reveals robust signatures of disease that are imperfectly reflected in the transcript profiles of explanted fibroblasts. Arthritis Rheumatol. 2006, 54, 1961–1973. [Google Scholar] [CrossRef] [PubMed]
- Milano, A.; Pendergrass, S.A.; Sargent, J.L.; George, L.K.; McCalmont, T.H.; Connolly, M.K.; Whitfield, M.L. Molecular subsets in the gene expression signatures of scleroderma skin. PLoS ONE 2008, 3, e2696. [Google Scholar] [CrossRef]
- Assassi, S.; Swindell, W.R.; Wu, M.; Tan, F.D.; Khanna, D.; Furst, D.E.; Tashkin, D.P.; Jahan-Tigh, R.R.; Mayes, M.D.; Gudjonsson, J.E.; et al. Dissecting the heterogeneity of skin gene expression patterns in systemic sclerosis. Arthritis Rheumatol. 2015, 67, 3016–3026. [Google Scholar] [CrossRef] [PubMed]
- Derrett-Smith, E.C.; Martyanov, V.; Chighizola, C.B.; Moinzadeh, P.; Campochiaro, C.; Khan, K.; Wood, T.A.; Meroni, P.L.; Abraham, D.J.; Ong, V.H.; et al. Limited cutaneous systemic sclerosis skin demonstrates distinct molecular subsets separated by a cardiovascular development gene expression signature. Arthritis Res. 2017, 19, 156. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pendergrass, S.A.; Lemaire, R.; Francis, I.P.; Mahoney, J.M.; Lafyatis, R.; Whitfield, M.L. Intrinsic gene expression subsets of diffuse cutaneous systemic sclerosis are stable in serial skin biopsies. J. Investig. Dermatol. 2012, 132, 1363–1373. [Google Scholar] [CrossRef]
- Johnson, M.E.; Mahoney, J.M.; Taroni, J.; Sargent, J.L.; Marmarelis, E.; Wu, M.R.; Varga, J.; Hinchcliff, M.E.; Whitfield, M.L. Experimentally-derived fibroblast gene signatures identify molecular pathways associated with distinct subsets of systemic sclerosis patients in three independent cohorts. PLoS ONE 2015, 10, e0114017. [Google Scholar] [CrossRef]
- Rice, L.M.; Ziemek, J.; Stratton, E.A.; McLaughlin, S.R.; Padilla, C.M.; Mathes, A.L.; Christmann, R.B.; Stifano, G.; Browning, J.L.; Whitfield, M.L.; et al. A longitudinal biomarker for the extent of skin disease in patients with diffuse cutaneous systemic sclerosis. Arthritis Rheumatol. 2015, 67, 3004–3015. [Google Scholar] [CrossRef]
- Stifano, G.; Sornasse, T.; Rice, L.M.; Na, L.; Chen-Harris, H.; Khanna, D.; Jahreis, A.; Zhang, Y.; Siegel, J.; Lafyatis, R. Skin Gene Expression Is Prognostic for the Trajectory of Skin Disease in Patients With Diffuse Cutaneous Systemic Sclerosis. Arthritis Rheumatol. 2018, 70, 912–919. [Google Scholar] [CrossRef]
- Hinchcliff, M.; Huang, C.C.; Wood, T.A.; Matthew Mahoney, J.; Martyanov, V.; Bhattacharyya, S.; Tamaki, Z.; Lee, J.; Carns, M.; Podlusky, S.; et al. Molecular signatures in skin associated with clinical improvement during mycophenolate treatment in systemic sclerosis. J. Investig. Dermatol. 2013, 133, 1979–1989. [Google Scholar] [CrossRef]
- Walker, D.L.; Bhagwate, A.V.; Baheti, S.; Smalley, R.L.; Hilker, C.A.; Sun, Z.; Cunningham, J.M. DNA methylation profiling: Comparison of genome-wide sequencing methods and the Infinium Human Methylation 450 Bead Chip. Epigenomics 2015, 7, 1287–1302. [Google Scholar] [CrossRef]
- Schubeler, D. Function and information content of DNA methylation. Nature 2015, 517, 321–326. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez-Arcelus, M.; Ongen, H.; Lappalainen, T.; Montgomery, S.B.; Buil, A.; Yurovsky, A.; Bryois, J.; Padioleau, I.; Romano, L.; Planchon, A.; et al. Tissue-specific effects of genetic and epigenetic variation on gene regulation and splicing. PLoS Genet. 2015, 11, e1004958. [Google Scholar] [CrossRef] [PubMed]
- Ambrosi, C.; Manzo, M.; Baubec, T. Dynamics and Context-Dependent Roles of DNA Methylation. J. Mol. Biol. 2017, 429, 1459–1475. [Google Scholar] [CrossRef]
- Gutierrez-Arcelus, M.; Lappalainen, T.; Montgomery, S.B.; Buil, A.; Ongen, H.; Yurovsky, A.; Bryois, J.; Giger, T.; Romano, L.; Planchon, A.; et al. Passive and active DNA methylation and the interplay with genetic variation in gene regulation. eLife 2013, 2, e00523. [Google Scholar] [CrossRef] [PubMed]
- Bonder, M.J.; Luijk, R.; Zhernakova, D.V.; Moed, M.; Deelen, P.; Vermaat, M.; van Iterson, M.; van Dijk, F.; van Galen, M.; Bot, J.; et al. Disease variants alter transcription factor levels and methylation of their binding sites. Nat. Genet. 2017, 49, 131–138. [Google Scholar] [CrossRef]
- Bonder, M.J.; Kasela, S.; Kals, M.; Tamm, R.; Lokk, K.; Barragan, I.; Buurman, W.A.; Deelen, P.; Greve, J.W.; Ivanov, M.; et al. Genetic and epigenetic regulation of gene expression in fetal and adult human livers. BMC Genom. 2014, 15, 860. [Google Scholar] [CrossRef]
- Grundberg, E.; Meduri, E.; Sandling, J.K.; Hedman, A.K.; Keildson, S.; Buil, A.; Busche, S.; Yuan, W.; Nisbet, J.; Sekowska, M.; et al. Global analysis of DNA methylation variation in adipose tissue from twins reveals links to disease-associated variants in distal regulatory elements. Am. J. Hum. Genet. 2013, 93, 876–890. [Google Scholar] [CrossRef]
- Coit, P.; Ortiz-Fernandez, L.; Lewis, E.E.; McCune, W.J.; Maksimowicz-McKinnon, K.; Sawalha, A.H. A longitudinal and transancestral analysis of DNA methylation patterns and disease activity in lupus patients. JCI Insight 2020, 5, e143654. [Google Scholar] [CrossRef]
- Imgenberg-Kreuz, J.; Carlsson Almlof, J.; Leonard, D.; Alexsson, A.; Nordmark, G.; Eloranta, M.L.; Rantapaa-Dahlqvist, S.; Bengtsson, A.A.; Jonsen, A.; Padyukov, L.; et al. DNA methylation mapping identifies gene regulatory effects in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 2018, 77, 736–743. [Google Scholar] [CrossRef]
- Rice, L.M.; Stifano, G.; Ziemek, J.; Lafyatis, R. Local skin gene expression reflects both local and systemic skin disease in patients with systemic sclerosis. Rheumatology (Oxf.) 2016, 55, 377–379. [Google Scholar] [CrossRef][Green Version]
- Refaat, A.; Owis, M.; Abdelhamed, S.; Saiki, I.; Sakurai, H. Retrospective screening of microarray data to identify candidate IFN-inducible genes in a HTLV-1 transformed model. Oncol. Lett. 2018, 15, 4753–4758. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.K.; Zhou, X.; Mayes, M.D.; Gourh, P.; Guo, X.; Marcum, C.; Jin, L.; Arnett, F.C., Jr. Signatures of differentially regulated interferon gene expression and vasculotrophism in the peripheral blood cells of systemic sclerosis patients. Rheumatol. (Oxf.) 2006, 45, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Higgs, B.W.; Liu, Z.; White, B.; Zhu, W.; White, W.I.; Morehouse, C.; Brohawn, P.; Kiener, P.A.; Richman, L.; Fiorentino, D.; et al. Patients with systemic lupus erythematosus, myositis, rheumatoid arthritis and scleroderma share activation of a common type I interferon pathway. Ann. Rheum. Dis. 2011, 70, 2029–2036. [Google Scholar] [CrossRef] [PubMed]
- Higgs, B.W.; Zhu, W.; Richman, L.; Fiorentino, D.F.; Greenberg, S.A.; Jallal, B.; Yao, Y. Identification of activated cytokine pathways in the blood of systemic lupus erythematosus, myositis, rheumatoid arthritis, and scleroderma patients. Int. J. Rheum. Dis. 2012, 15, 25–35. [Google Scholar] [CrossRef]
- Brkic, Z.; van Bon, L.; Cossu, M.; van Helden-Meeuwsen, C.G.; Vonk, M.C.; Knaapen, H.; van den Berg, W.; Dalm, V.A.; Van Daele, P.L.; Severino, A.; et al. The interferon type I signature is present in systemic sclerosis before overt fibrosis and might contribute to its pathogenesis through high BAFF gene expression and high collagen synthesis. Ann. Rheum. Dis. 2016, 75, 1567–1573. [Google Scholar] [CrossRef]
- Wang, X.F.; Zhang, B.H.; Lu, X.Q.; Wang, R.Q. DLX5 gene regulates the Notch signaling pathway to promote glomerulosclerosis and interstitial fibrosis in uremic rats. J. Cell. Physiol. 2019, 234, 21825–21837. [Google Scholar] [CrossRef]
- Dees, C.; Zerr, P.; Tomcik, M.; Beyer, C.; Horn, A.; Akhmetshina, A.; Palumbo, K.; Reich, N.; Zwerina, J.; Sticherling, M.; et al. Inhibition of Notch signaling prevents experimental fibrosis and induces regression of established fibrosis. Arthritis Rheum. 2011, 63, 1396–1404. [Google Scholar] [CrossRef]
- Beyer, C.; Dees, C.; Distler, J.H. Morphogen pathways as molecular targets for the treatment of fibrosis in systemic sclerosis. Arch. Dermatol. Res. 2013, 305, 1–8. [Google Scholar] [CrossRef]
- Ramos, P.S. Epigenetics of scleroderma: Integrating genetic, ethnic, age, and environmental effects. J. Scleroderma Relat. Disord. 2019, 4, 238–250. [Google Scholar] [CrossRef]
- Beretta, L.; Caronni, M.; Vanoli, M.; Scorza, R. Systemic sclerosis after interferon-alfa therapy for myeloproliferative disorders. Br. J. Dermatol. 2002, 147, 385–386. [Google Scholar] [CrossRef]
- Solans, R.; Bosch, J.A.; Esteban, I.; Vilardell, M. Systemic sclerosis developing in association with the use of interferon alpha therapy for chronic viral hepatitis. Clin. Exp. Rheumatol. 2004, 22, 625–628. [Google Scholar] [PubMed]
- Tahara, H.; Kojima, A.; Hirokawa, T.; Oyama, T.; Naganuma, A.; Maruta, S.; Okada, K.; Ban, S.; Yoshida, K.; Takagi, H.; et al. Systemic sclerosis after interferon alphacon-1 therapy for hepatitis C. Intern. Med. 2007, 46, 473–476. [Google Scholar] [CrossRef] [PubMed]
- Powell, A.; Myles, M.L.; Yacyshyn, E. The development of systemic sclerosis in a female patient with multiple sclerosis following beta interferon treatment. Clin. Rheumatol. 2008, 27, 1467–1468. [Google Scholar] [CrossRef] [PubMed]
- Rezaei, R.; Mahmoudi, M.; Gharibdoost, F.; Kavosi, H.; Dashti, N.; Imeni, V.; Jamshidi, A.; Aslani, S.; Mostafaei, S.; Vodjgani, M. IRF7 gene expression profile and methylation of its promoter region in patients with systemic sclerosis. Int. J. Rheum. Dis. 2017, 20, 1551–1561. [Google Scholar] [CrossRef] [PubMed]
- Manetti, M.; Romano, E.; Rosa, I.; Guiducci, S.; Bellando-Randone, S.; De Paulis, A.; Ibba-Manneschi, L.; Matucci-Cerinic, M. Endothelial-to-mesenchymal transition contributes to endothelial dysfunction and dermal fibrosis in systemic sclerosis. Ann. Rheum Dis 2017, 76, 924–934. [Google Scholar] [CrossRef]
- Mendoza, F.A.; Piera-Velazquez, S.; Farber, J.L.; Feghali-Bostwick, C.; Jimenez, S.A. Endothelial Cells Expressing Endothelial and Mesenchymal Cell Gene Products in Lung Tissue From Patients with Systemic Sclerosis-Associated Interstitial Lung Disease. Arthritis Rheumatol. 2016, 68, 210–217. [Google Scholar] [CrossRef]
- Helmick, C.G.; Felson, D.T.; Lawrence, R.C.; Gabriel, S.; Hirsch, R.; Kwoh, C.K.; Liang, M.H.; Kremers, H.M.; Mayes, M.D.; Merkel, P.A.; et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheumatol. 2008, 58, 15–25. [Google Scholar] [CrossRef]
- Lim, U.; Song, M.A. Dietary and lifestyle factors of DNA methylation. Methods Mol. Biol. 2012, 863, 359–376. [Google Scholar]


| Patients (n = 15) | Controls (n = 15) | |
|---|---|---|
| Age at enrollment (mean ± SD) | 44.4 ± 9.7 | 45.6 ± 9.9 |
| Female, n (%) | 10 (67%) | 12 (80%) |
| dcSSc, n (%) | 14 (93%) | NA |
| ssSSc, n (%) | 1 (7%) | NA |
| Raynaud’s Phenomemon, n (%) | 15 (100%) | NA |
| Disease duration (mean ± SD) | 5.3 ± 5.2 | NA |
| mRSS (mean ± SD) 1 | 18.6 ± 9.1 | NA |
| ILD, n (%) | 3 (20%) | NA |
| PH/PAH, n (%) | 6 (40%) | NA |
| Overlap SLE, n (%) | 2 (13%) | NA |
| Anti-topoisomerase I, n (%) 2 | 5 (46%) | NA |
| Anti-RNA polymerase III, n (%) 3 | 1 (11%) | NA |
| Immunosuppressive medications, n (%) | 10 (67%) | NA |
| Smoker at enrollment, n (%) 4 | 1 (7%) | 1 (7%) |
| Symbol | Gene Type | Chr | Position (kb) | MDM | n Sites | Rank |
|---|---|---|---|---|---|---|
| Genes | ||||||
| RPL30P7 | Pseudogene | 5 | 10,489–10,489 | 0.31 | 1 | 25 |
| MGC12916 | RNA gene (lncRNA) | 17 | 14,207–14,209 | 0.23 | 31 | 60 |
| LINC01227 | RNA gene (ncRNA) | 16 | 80,601–80,607 | −0.21 | 1 | 80 |
| ENSG00000255342 | Uncategorized (lncRNA) | 11 | 123,007–123,007 | 0.22 | 11 | 81 |
| ENSG00000227930 | RNA gene | 7 | 23,931–23,937 | −0.2 | 2 | 84 |
| ENSG00000230104 | Uncategorized (lncRNA) | 2 | 173,539–173,540 | 0.22 | 1 | 131 |
| LOC102724927 | RNA gene (ncRNA) | 16 | 3998–4000 | −0.14 | 5 | 132 |
| ENSG00000229472 | - | 20 | 32,669–32,670 | 0.18 | 2 | 133 |
| MIR5587 | RNA gene (miRNA) | 16 | 585–585 | 0.22 | 2 | 141 |
| LOC105379365 | RNA gene (ncRNA) | 8 | 34,032–34,042 | 0.17 | 2 | 146 |
| LOC402634 | Pseudogene | 7 | 2433–2434 | 0.17 | 2 | 163 |
| NEK2P4 | Pseudogene | 2 | 131,935–131,937 | 0.2 | 7 | 173 |
| NCRNA00250 | RNA gene (ncRNA) | 8 | 135,850–135,855 | −0.19 | 1 | 180 |
| DLX5 | Protein coding | 7 | 96,650–96,654 | 0.17 | 114 | 192 |
| LOC101929882 | RNA gene (ncRNA) | 2 | 10,179–10,181 | 0.13 | 2 | 194 |
| FAM180B | Protein coding | 11 | 47,608–47,611 | 0.17 | 6 | 195 |
| LOC100652792 | Pseudogene | 15 | 93,306–93,307 | 0.16 | 4 | 197 |
| Promoters | ||||||
| CDA | Protein coding | 1 | 20,914–20,916 | −0.26 | 1 | 7 |
| TAF5LP1 | Pseudogene | 17 | 33,824–33,826 | −0.22 | 8 | 23 |
| LINC00619 | RNA gene (ncRNA) | 10 | 44,339–44,341 | −0.25 | 1 | 29 |
| RPL30P7 | Pseudogene | 5 | 10,487–10,489 | −0.31 | 1 | 45 |
| SNORA25 | RNA gene (snoRNA) | 13 | 106,549–106,551 | −0.23 | 3 | 72 |
| ENSG00000241456 | RNA gene | 7 | 151,123–151,125 | 0.17 | 3 | 74 |
| ENSG00000229974 | - | 7 | 134,832–134,834 | 0.18 | 2 | 94 |
| TMEM140 | Protein coding | 7 | 134,831–134,833 | 0.18 | 2 | 94 |
| LOC100420018 | Pseudogene | 11 | 35,990–35,992 | 0.37 | 1 | 101 |
| ACKR4 | Protein coding | 3 | 132,315–132,317 | −0.23 | 1 | 110 |
| ENSG00000255342 | Uncategorized (lncRNA) | 11 | 123,007–123,009 | −0.22 | 11 | 112 |
| Entity | Reactome Pathway Name | Size | ES | NES | p-Value | FDR q-Value |
|---|---|---|---|---|---|---|
| Genes | Glucuronidation | 15 | −0.84 | −2.13 | <0.001 | <0.001 |
| Chondroitin sulfate dermatan sulfate metabolism | 42 | −0.57 | −1.76 | 0.002 | 0.088 | |
| Gap junction trafficking | 25 | −0.59 | −1.65 | 0.002 | 0.228 | |
| G β: γ signalling through PI3Kgamma | 24 | −0.59 | −1.65 | 0.003 | 0.191 | |
| Activation of kainate receptors upon glutamate binding | 29 | −0.55 | −1.60 | 0.008 | 0.236 | |
| Immunoregulatory interactions between a lymphoid and a non-lymphoid cell | 58 | 0.31 | 1.49 | <0.001 | 0.246 | |
| Promoters | Regulation of β cell development | 27 | −0.64 | −1.92 | <0.001 | 0.022 |
| Regulation of gene expression in β cells | 17 | −0.64 | −1.75 | 0.002 | 0.200 | |
| Immunoregulatory interactions between a lymphoid and a non-lymphoid cell | 33 | 0.53 | 2.20 | <0.001 | 0.007 |
| ID | p-Value | Term | Region |
|---|---|---|---|
| Hypomethylated regions | |||
| GO:0060337 | 8.00 × 10−4 | type I interferon signaling pathway | genes |
| GO:0034340 | 9.00 × 10−4 | response to type I interferon | genes |
| GO:0070458 | 2.10 × 10−3 | cellular detoxification of nitrogen compound | genes |
| GO:0018916 | 2.80 × 10−3 | nitrobenzene metabolic process | genes |
| GO:0060708 | 2.80 × 10−3 | spongiotrophoblast differentiation | genes |
| GO:0032020 | 4.10 × 10−3 | ISG15-protein conjugation | genes |
| Hypermethylated regions | |||
| GO:0001837 | 1.00 × 10−4 | epithelial to mesenchymal transition | genes |
| GO:0072079 | 1.00 × 10−4 | nephron tubule formation | genes |
| GO:0048762 | 3.00 × 10−4 | mesenchymal cell differentiation | genes |
| GO:0060980 | 1.00 × 10−3 | cell migration involved in coronary vasculogenesis | genes |
| GO:0048729 | 1.70 × 10−3 | tissue morphogenesis | genes |
| GO:0035295 | 1.70 × 10−3 | tube development | genes |
| GO:0048864 | 1.80 × 10−3 | stem cell development | genes |
| GO:0003218 | 1.90 × 10−3 | cardiac left ventricle formation | genes |
| GO:0070172 | 1.90 × 10−3 | positive regulation of tooth mineralization | genes |
| GO:0072272 | 1.90 × 10−3 | proximal/distal pattern formation involved in metanephric nephron development | genes |
| GO:0072088 | 2.10 × 10−3 | nephron epithelium morphogenesis | genes |
| GO:0061333 | 2.20 × 10−3 | renal tubule morphogenesis | genes |
| GO:0060166 | 2.90 × 10−3 | olfactory pit development | genes |
| GO:0060021 | 2.90 × 10−3 | palate development | genes |
| GO:0060993 | 3.20 × 10−3 | kidney morphogenesis | genes |
| GO:0072080 | 3.20 × 10−3 | nephron tubule development | genes |
| GO:0045893 | 3.50 × 10−3 | positive regulation of transcription, DNA-templated | genes |
| GO:0003166 | 3.90 × 10−3 | bundle of His development | genes |
| GO:0072086 | 3.90 × 10−3 | specification of loop of Henle identity | genes |
| GO:0072513 | 3.90 × 10−3 | positive regulation of secondary heart field cardioblast proliferation | genes |
| GO:2000653 | 3.90 × 10−3 | regulation of genetic imprinting | genes |
| GO:1902680 | 3.90 × 10−3 | positive regulation of RNA biosynthetic process | genes |
| GO:0048598 | 4.20 × 10−3 | embryonic morphogenesis | genes |
| GO:0051891 | 4.80 × 10−3 | positive regulation of cardioblast differentiation | genes |
| GO:0072334 | 1.70 × 10−3 | UDP-galactose transmembrane transport | promoters |
| GO:0035524 | 3.30 × 10−3 | proline transmembrane transport | promoters |
| GO:0060166 | 3.30 × 10−3 | olfactory pit development | promoters |
| GO:2000097 | 3.30 × 10−3 | regulation of smooth muscle cell-matrix adhesion | promoters |
| GO:0001867 | 5.00 × 10−3 | complement activation, lectin pathway | promoters |
| GO:0015820 | 5.00 × 10−3 | leucine transport | promoters |
| GO:0019858 | 5.00 × 10−3 | cytosine metabolic process | promoters |
| GO:0038110 | 5.00 × 10−3 | interleukin-2-mediated signaling pathway | promoters |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baker Frost, D.; da Silveira, W.; Hazard, E.S.; Atanelishvili, I.; Wilson, R.C.; Flume, J.; Day, K.L.; Oates, J.C.; Bogatkevich, G.S.; Feghali-Bostwick, C.; et al. Differential DNA Methylation Landscape in Skin Fibroblasts from African Americans with Systemic Sclerosis. Genes 2021, 12, 129. https://doi.org/10.3390/genes12020129
Baker Frost D, da Silveira W, Hazard ES, Atanelishvili I, Wilson RC, Flume J, Day KL, Oates JC, Bogatkevich GS, Feghali-Bostwick C, et al. Differential DNA Methylation Landscape in Skin Fibroblasts from African Americans with Systemic Sclerosis. Genes. 2021; 12(2):129. https://doi.org/10.3390/genes12020129
Chicago/Turabian StyleBaker Frost, DeAnna, Willian da Silveira, E. Starr Hazard, Ilia Atanelishvili, Robert C. Wilson, Jonathan Flume, Kayleigh L. Day, James C. Oates, Galina S. Bogatkevich, Carol Feghali-Bostwick, and et al. 2021. "Differential DNA Methylation Landscape in Skin Fibroblasts from African Americans with Systemic Sclerosis" Genes 12, no. 2: 129. https://doi.org/10.3390/genes12020129
APA StyleBaker Frost, D., da Silveira, W., Hazard, E. S., Atanelishvili, I., Wilson, R. C., Flume, J., Day, K. L., Oates, J. C., Bogatkevich, G. S., Feghali-Bostwick, C., Hardiman, G., & Ramos, P. S. (2021). Differential DNA Methylation Landscape in Skin Fibroblasts from African Americans with Systemic Sclerosis. Genes, 12(2), 129. https://doi.org/10.3390/genes12020129






