Roles of Resveratrol in Improving the Quality of Postovulatory Aging Oocytes In Vitro
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Feeding
2.2. Oocyte Collection and Blastocyst Culture
2.3. In Vitro Fertilization
2.4. POA In Vitro and Resveratrol Treatment
2.5. Sperm Binding Assay
2.6. Immunofluorescent Staining
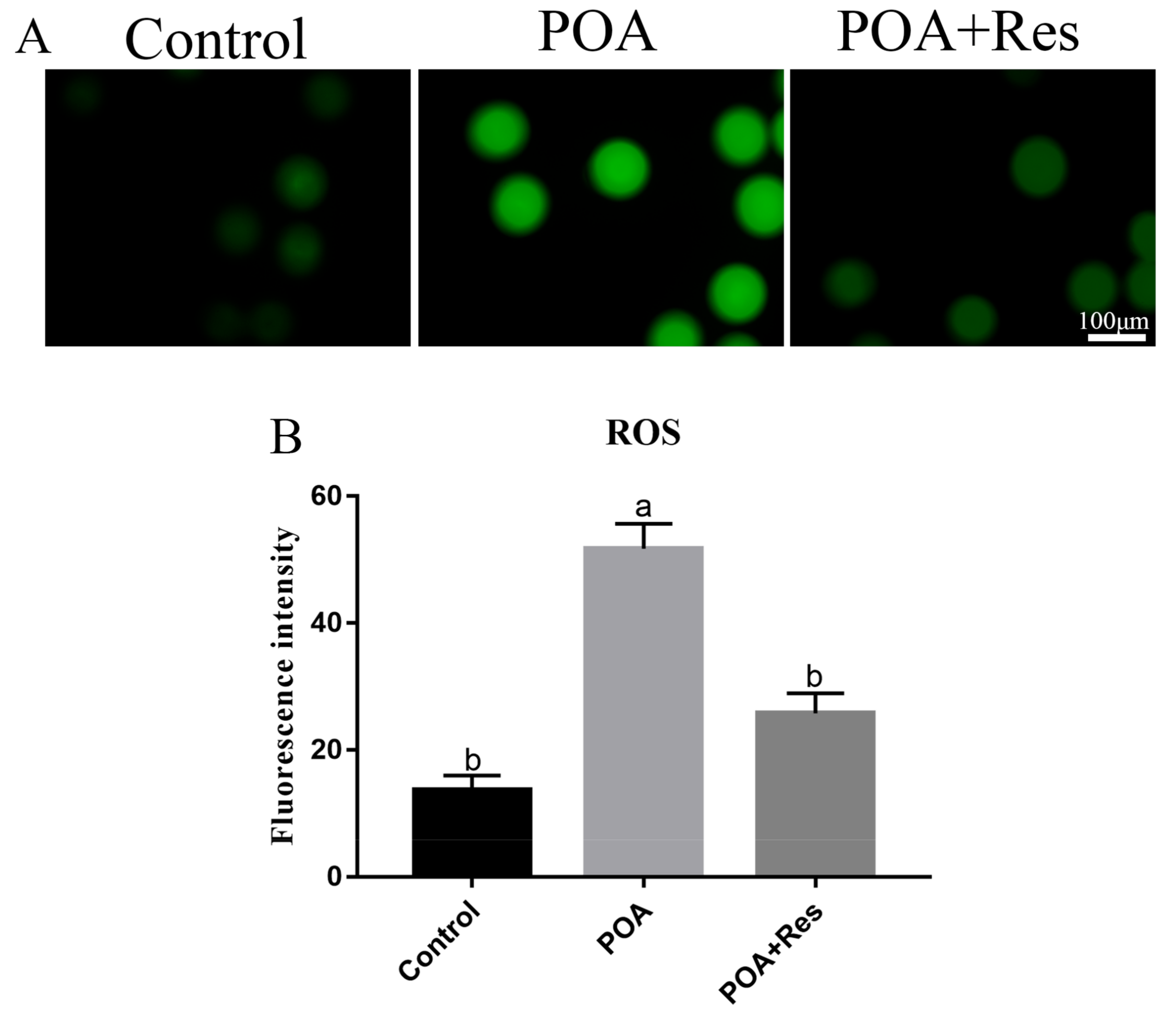
2.7. Detection of ROS Level
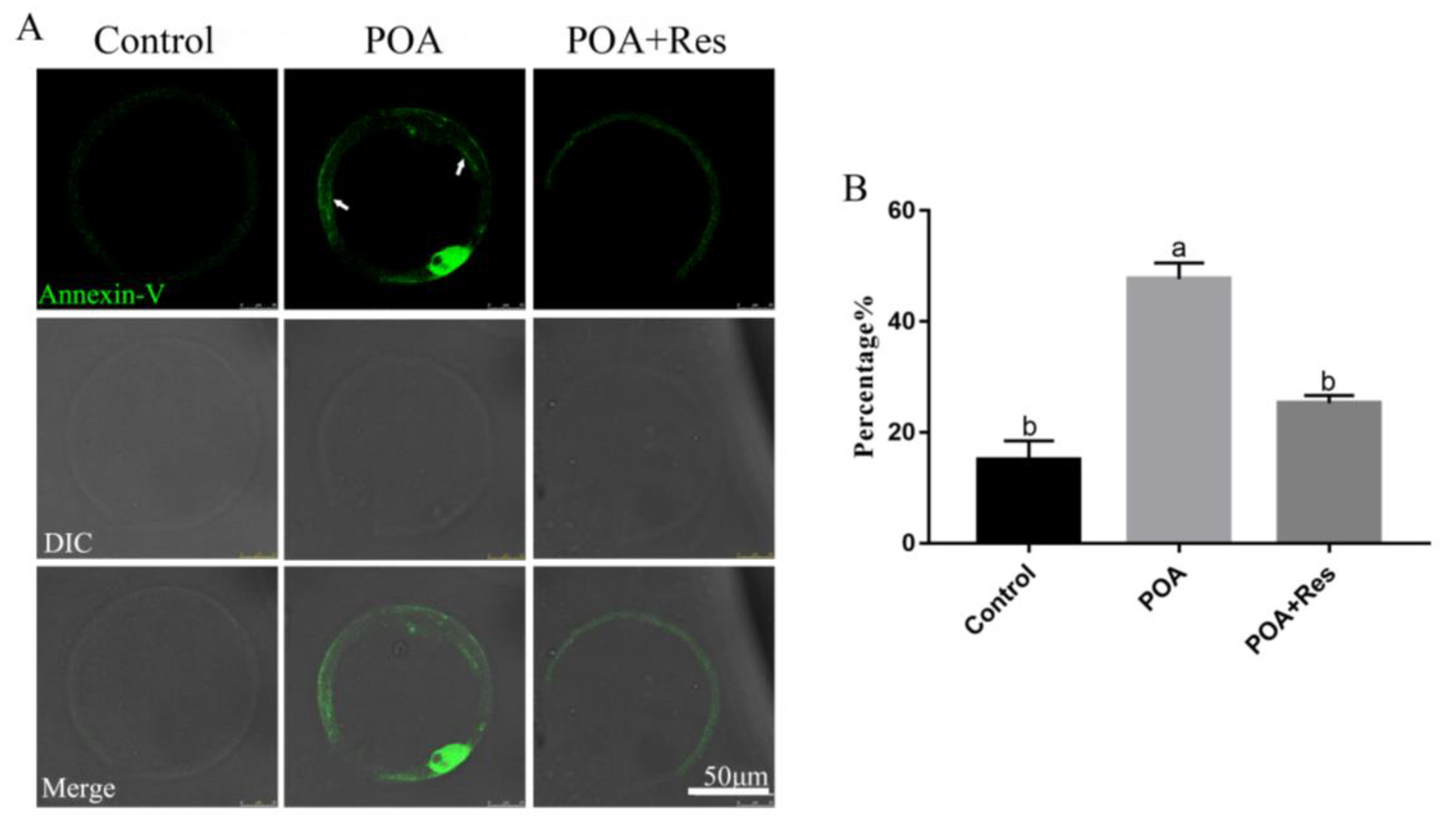
2.8. Annexin-V Staining
2.9. Mitochondrion Distribution
2.10. Fluorescence Intensity Analysis
2.11. Statistical Analysis
3. Results
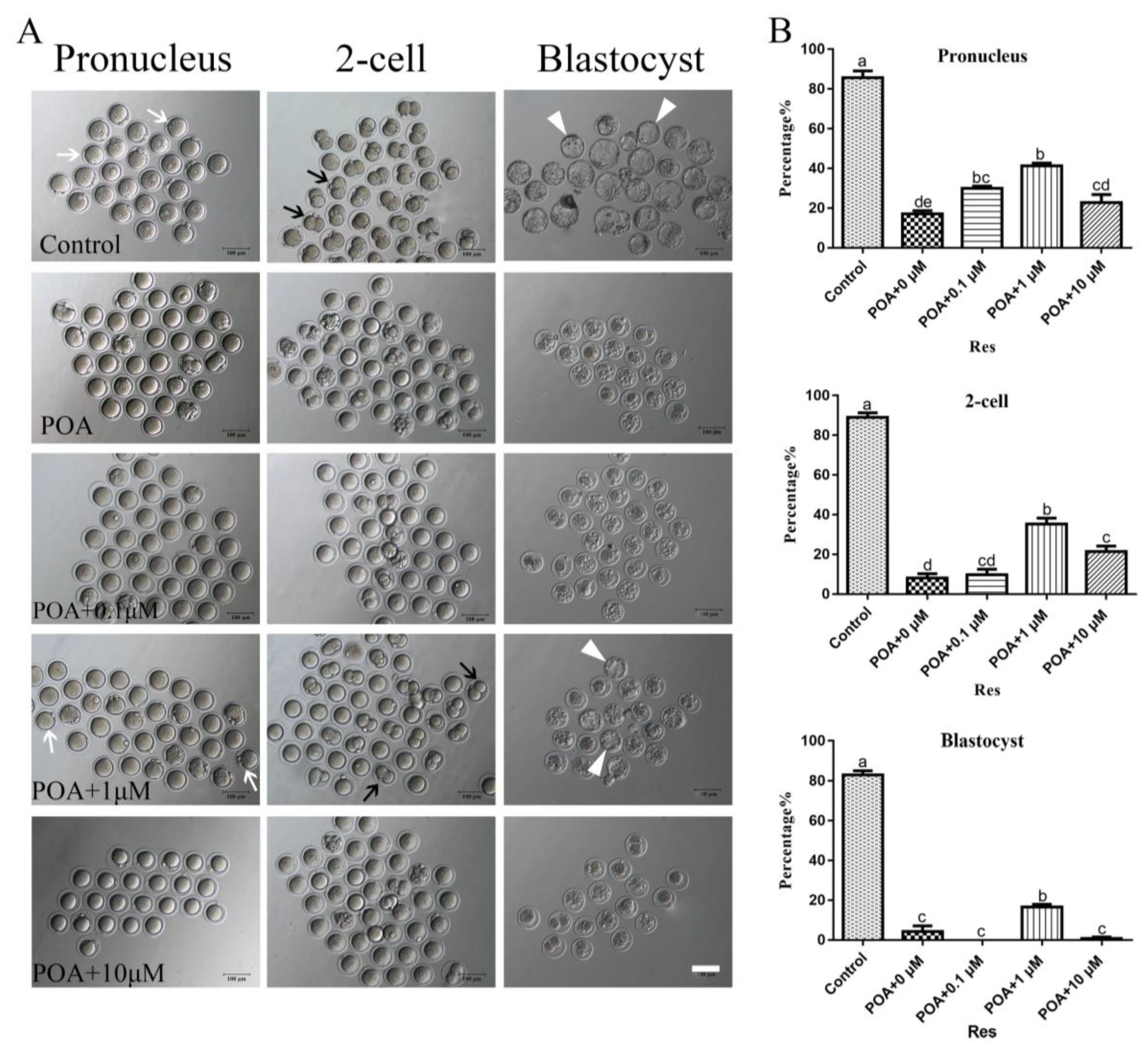
3.1. Effects of POA In Vitro on the Rates of IVF and Early Embryo Development
3.2. Resveratrol Improves the Developmental Potential of POA Oocytes
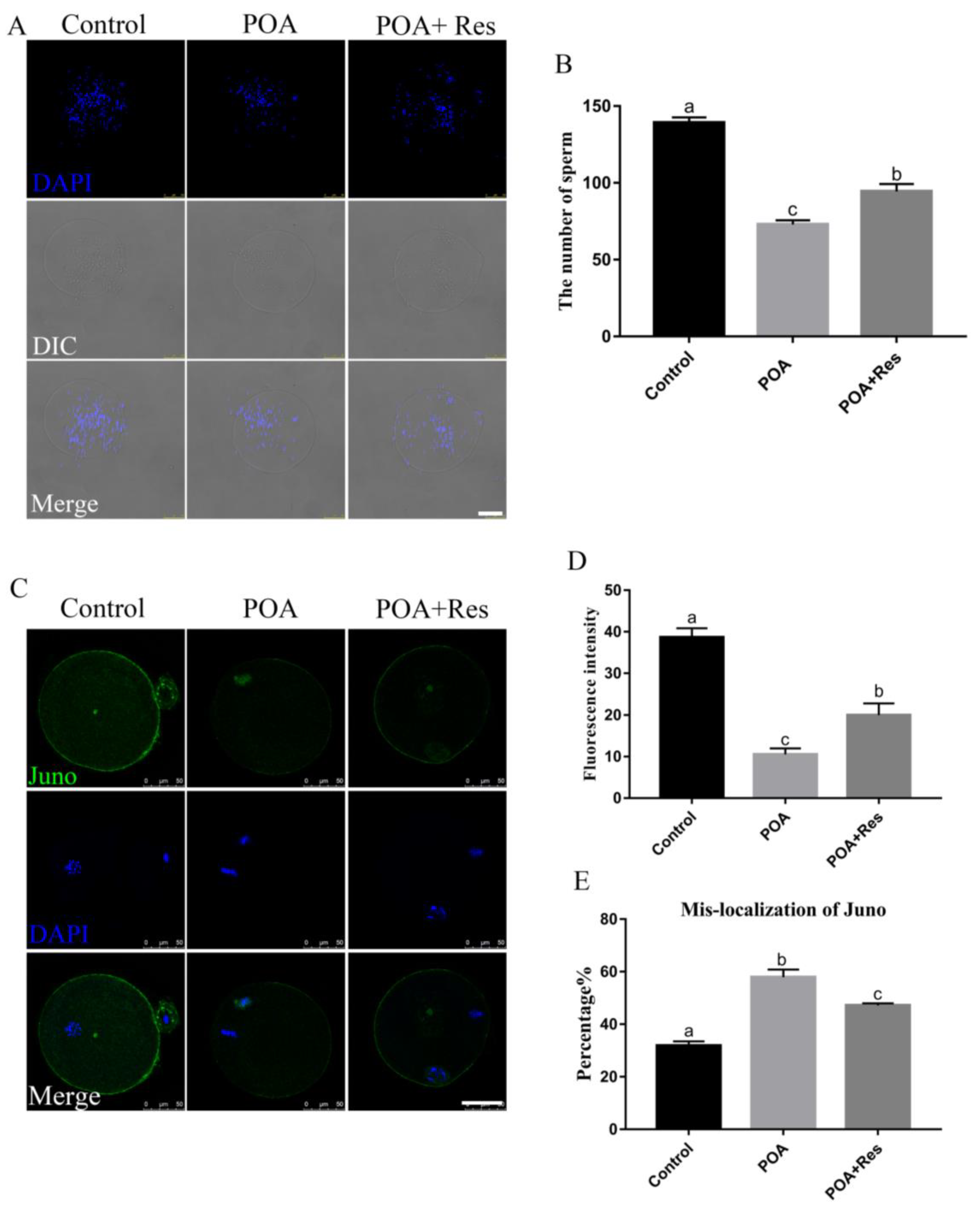
3.3. Resveratrol Improves the Sperm Binding Ability and Restores the Juno Expression of POA Oocytes
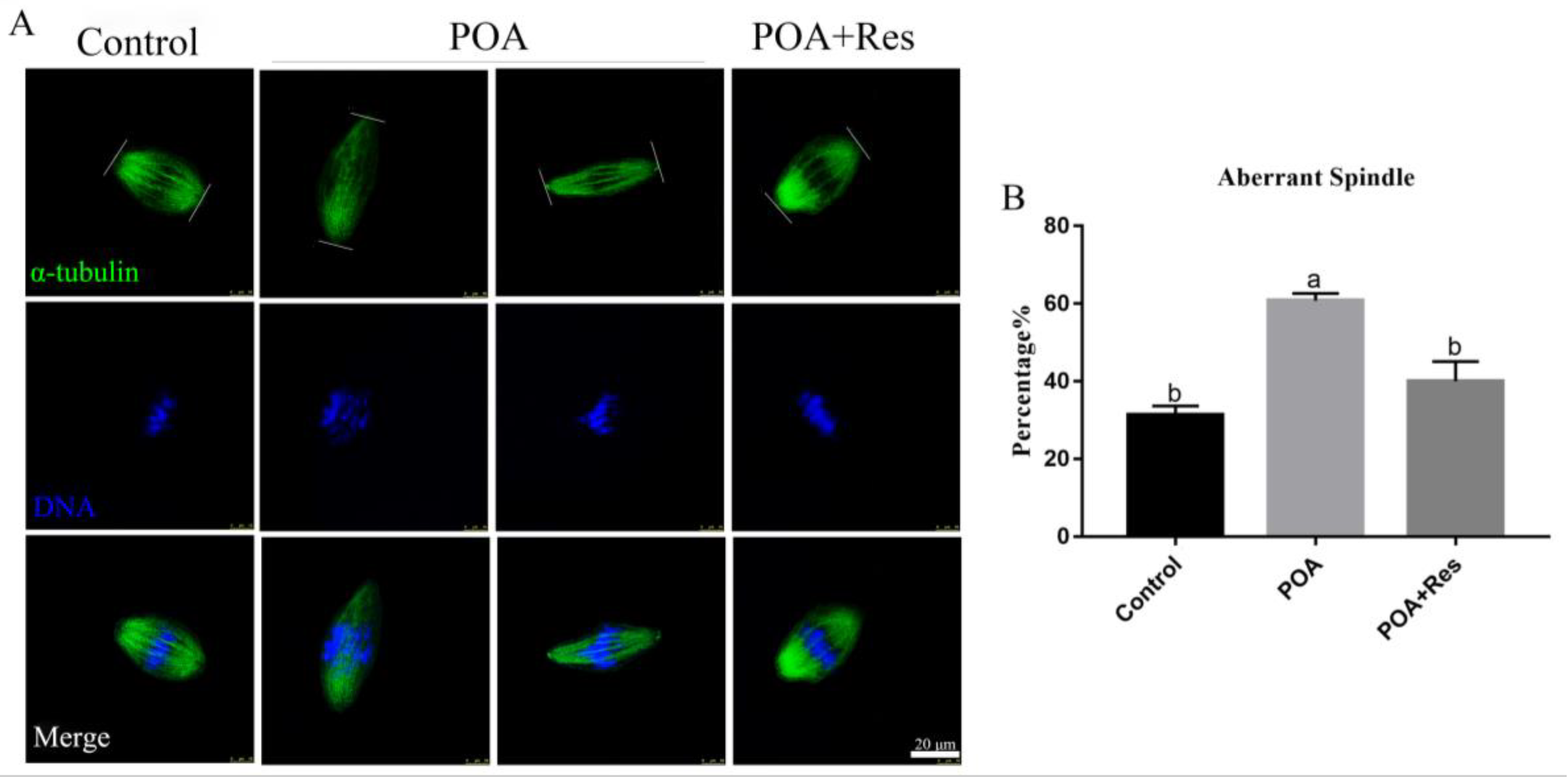
3.4. Resveratrol Maintains Correct Spindle Assembly and Morphology in POA Oocytes
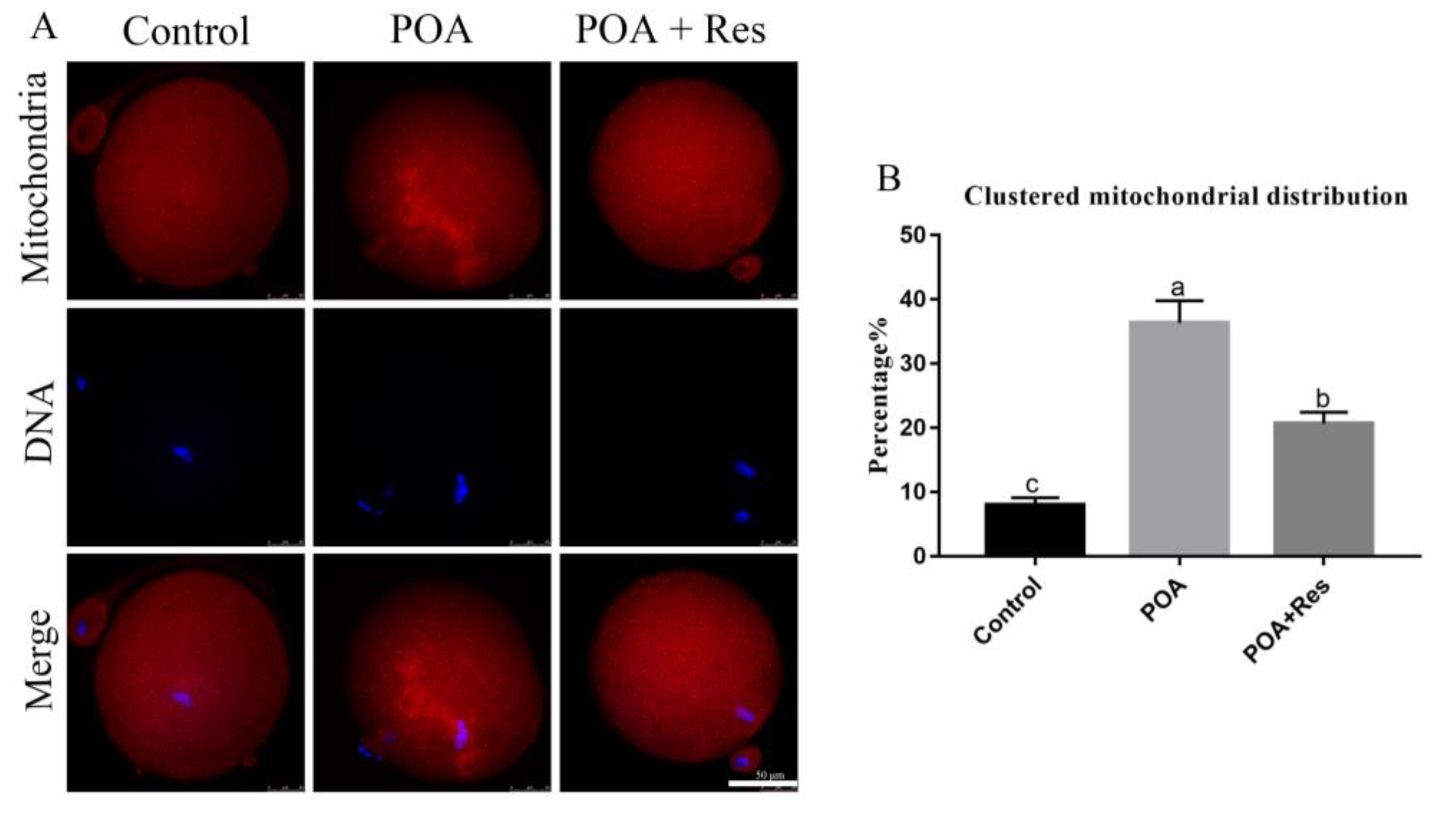
3.5. Resveratrol Reduces the Clustered Mitochondrion Distribution in POA Oocytes
3.6. Resveratrol Decreases the Intracellular ROS Accumulation in POA Oocytes
3.7. Resveratrol Alleviates the Early Apoptosis of POA Oocytes
3.8. Resveratrol Restores the Abnormal Level of H3K9me2 in POA Oocytes
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Broekmans, F.J.; Soules, M.R.; Fauser, B.C. Ovarian aging: Mechanisms and clinical consequences. Endocr. Rev. 2009, 30, 465–493. [Google Scholar] [CrossRef] [PubMed]
- Kocourkova, J.; Burcin, B.; Kucera, T. Demographic relevancy of increased use of assisted reproduction in European countries. Reprod Health 2014, 11, 37. [Google Scholar] [CrossRef] [PubMed]
- Butcher, R.L. Pre-ovulatory and post-ovulatory overripeness. Int. J. Gynaecol. Obstet. 1976, 14, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Sun, H.H.; Ji, C.L.; Zhang, J.; Yuan, H.J.; Luo, M.J.; Liu, X.Y.; Tan, J.H. Role of calcium-sensing receptor in regulating spontaneous activation of postovulatory aging rat oocytes. Biol. Reprod. 2018, 98, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Miao, Y.; Zhou, C.; Cui, Z.; Zhang, M.; ShiYang, X.; Lu, Y.; Xiong, B. Postovulatory aging causes the deterioration of porcine oocytes via induction of oxidative stress. FASEB J. 2018, 32, 1328–1337. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.X.; Cui, W.; Zhang, M.; Zhang, J.; Wang, T.Y.; Zhu, J.; Jiao, G.Z.; Tan, J.H. Role of Na+/Ca2+ exchanger (NCX) in modulating postovulatory aging of mouse and rat oocytes. PLoS ONE 2014, 9, e93446. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.; Martin, J.H.; Aitken, R.J. Accumulation of electrophilic aldehydes during postovulatory aging of mouse oocytes causes reduced fertility, oxidative stress, and apoptosis. Biol. Reprod. 2015, 92, 33. [Google Scholar] [CrossRef]
- Premkumar, K.V.; Chaube, S.K. Increased level of reactive oxygen species persuades postovulatory aging-mediated spontaneous egg activation in rat eggs cultured in vitro. In Vitro Cell. Dev. Biol. Anim. 2016, 52, 576–588. [Google Scholar] [CrossRef]
- Tatone, C.; Carbone, M.C.; Gallo, R.; Delle Monache, S.; Di Cola, M.; Alesse, E.; Amicarelli, F. Age-associated changes in mouse oocytes during postovulatory in vitro culture: Possible role for meiotic kinases and survival factor BCL2. Biol. Reprod. 2006, 74, 395–402. [Google Scholar] [CrossRef]
- Prasad, S.; Koch, B.; Chaube, S.K. Maturation promoting factor destabilization facilitates postovulatory aging-mediated abortive spontaneous egg activation in rat. Dev. Growth Differ. 2016, 58, 293–302. [Google Scholar] [CrossRef]
- Prasad, S.; Koch, B.; Chaube, S.K. RO-3306 prevents postovulatory aging-mediated spontaneous exit from M-II arrest in rat eggs cultured in vitro. Biomed. Pharmacother. 2016, 78, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Romero-Bueno, R.; de la Cruz Ruiz, P.; Artal-Sanz, M.; Askjaer, P.; Dobrzynska, A. Nuclear Organization in Stress and Aging. Cells 2019, 8, 664. [Google Scholar] [CrossRef] [PubMed]
- Tarin, J.J.; Ten, J.; Vendrell, F.J.; Cano, A. Dithiothreitol prevents age-associated decrease in oocyte/conceptus viability in vitro. Hum. Reprod. 1998, 13, 381–386. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Winston, N.J.; Braude, P.R.; Johnson, M.H. Are failed-fertilized human oocytes useful? Hum. Reprod. 1993, 8, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Liu, L. Mechanism of resveratrol in improving ovarian function in a rat model of premature ovarian insufficiency. J. Obstet. Gynaecol. Res. 2018, 44, 1431–1438. [Google Scholar] [CrossRef]
- Busch, F.; Mobasheri, A.; Shayan, P.; Lueders, C.; Stahlmann, R.; Shakibaei, M. Resveratrol modulates interleukin-1beta-induced phosphatidylinositol 3-kinase and nuclear factor kappaB signaling pathways in human tenocytes. J. Biol. Chem. 2012, 287, 38050–38063. [Google Scholar] [CrossRef]
- Kizmazoglu, C.; Aydin, H.E.; Sevin, I.E.; Kalemci, O.; Yuceer, N.; Atasoy, M.A. Neuroprotective Effect of Resveratrol on Acute Brain Ischemia Reperfusion Injury by Measuring Annexin V, p53, Bcl-2 Levels in Rats. J. Korean Neurosurg. Soc. 2015, 58, 508–512. [Google Scholar] [CrossRef]
- Seo, K.; Seo, S.; Han, J.Y.; Ki, S.H.; Shin, S.M. Resveratrol attenuates methylglyoxal-induced mitochondrial dysfunction and apoptosis by Sestrin2 induction. Toxicol. Appl. Pharmacol. 2014, 280, 314–322. [Google Scholar] [CrossRef]
- Bagul, P.K.; Katare, P.B.; Bugga, P.; Dinda, A.K.; Banerjee, S.K. SIRT-3 Modulation by Resveratrol Improves Mitochondrial Oxidative Phosphorylation in Diabetic Heart through Deacetylation of TFAM. Cells 2018, 7, 235. [Google Scholar] [CrossRef]
- Antognelli, C.; Moretti, S.; Frosini, R.; Puxeddu, E.; Sidoni, A.; Talesa, V.N. Methylglyoxal Acts as a Tumor-Promoting Factor in Anaplastic Thyroid Cancer. Cells 2019, 8, 547. [Google Scholar] [CrossRef] [PubMed]
- Braga, A.; Ferreira, P.; Oliveira, J.; Rocha, I.; Faria, N. Heterologous production of resveratrol in bacterial hosts: Current status and perspectives. World J. Microbiol. Biotechnol. 2018, 34, 122. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, M.; Chen, Z.J.; Du, Y. Resveratrol promotes the embryonic development of vitrified mouse oocytes after in vitro fertilization. In Vitro Cell. Dev. Biol. Anim. 2018, 54, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Jo, Y.J.; Oh, J.S.; Kim, N.H. Quercetin delays postovulatory aging of mouse oocytes by regulating SIRT expression and MPF activity. Oncotarget 2017, 8, 38631–38641. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Lu, Y.; Zhang, M.; Miao, Y.; Zhou, C.; Cui, Z.; Xiong, B. Melatonin improves the fertilization ability of post-ovulatory aged mouse oocytes by stabilizing ovastacin and Juno to promote sperm binding and fusion. Hum. Reprod. 2017, 32, 598–606. [Google Scholar] [CrossRef] [PubMed]
- He, Y.T.; Wang, W.; Shen, W.; Sun, Q.Y.; Yin, S. Melatonin protects against Fenoxaprop-ethyl exposure-induced meiotic defects in mouse oocytes. Toxicology 2019, 425, 152241. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Luo, S.M.; Ma, J.Y.; Shen, W.; Yin, S. Cytotoxicity and DNA Damage Caused from Diazinon Exposure by Inhibiting the PI3K-AKT Pathway in Porcine Ovarian Granulosa Cells. J. Agric. Food Chem. 2019, 67, 19–31. [Google Scholar] [CrossRef]
- Yang, L.L.; Zhao, Y.; Luo, S.M.; Ma, J.Y.; Ge, Z.J.; Shen, W.; Yin, S. Toxic effects and possible mechanisms of hydrogen sulfide and/or ammonia on porcine oocyte maturation in vitro. Toxicol. Lett. 2018, 285, 20–26. [Google Scholar] [CrossRef]
- Li, W.D.; Yu, S.; Luo, S.M.; Shen, W.; Yin, S.; Sun, Q.Y. Melatonin defends mouse oocyte quality from benzo[ghi]perylene-induced deterioration. J. Cell Physiol. 2019, 234, 6220–6229. [Google Scholar] [CrossRef]
- Lu, Z.; Zhang, C.; Han, C.; An, Q.; Cheng, Y.; Chen, Y.; Meng, R.; Zhang, Y.; Su, J. Plasticizer Bis(2-ethylhexyl) Phthalate Causes Meiosis Defects and Decreases Fertilization Ability of Mouse Oocytes in Vivo. J. Agric. Food Chem. 2019, 67, 3459–3468. [Google Scholar] [CrossRef]
- Giritharan, G.; Talbi, S.; Donjacour, A.; Di Sebastiano, F.; Dobson, A.T.; Rinaudo, P.F. Effect of in vitro fertilization on gene expression and development of mouse preimplantation embryos. Reproduction 2007, 134, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Aydin, H.; Sultana, A.; Li, S.; Thavalingam, A.; Lee, J.E. Molecular architecture of the human sperm IZUMO1 and egg JUNO fertilization complex. Nature 2016, 534, 562–565. [Google Scholar] [CrossRef] [PubMed]
- Grayson, P. Izumo1 and Juno: The evolutionary origins and coevolution of essential sperm-egg binding partners. R. Soc. Open Sci. 2015, 2, 150296. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, E.M.; Vireque, A.A.; Adona, P.R.; Meirelles, F.V.; Ferriani, R.A.; Navarro, P.A. Cytoplasmic maturation of bovine oocytes: Structural and biochemical modifications and acquisition of developmental competence. Theriogenology 2009, 71, 836–848. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.Y.; Wu, G.M.; Lai, L.; Park, K.W.; Cabot, R.; Cheong, H.T.; Day, B.N.; Prather, R.S.; Schatten, H. Translocation of active mitochondria during pig oocyte maturation, fertilization and early embryo development in vitro. Reproduction 2001, 122, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Lai, F.N.; Ma, J.Y.; Liu, J.C.; Wang, J.J.; Cheng, S.F.; Sun, X.F.; Li, L.; Li, B.; Nyachoti, C.M.; Shen, W. The influence of N-acetyl-l-cysteine on damage of porcine oocyte exposed to zearalenone in vitro. Toxicol. Appl. Pharmacol. 2015, 289, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.; Aitken, R.J. Oxidative stress and ageing of the post-ovulatory oocyte. Reproduction 2013, 146, R217–R227. [Google Scholar] [CrossRef] [PubMed]
- Lord, T.; Nixon, B.; Jones, K.T.; Aitken, R.J. Melatonin prevents postovulatory oocyte aging in the mouse and extends the window for optimal fertilization in vitro. Biol. Reprod. 2013, 88, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.; Ma, J.; Schatten, H.; Sun, Q. Epigenetic changes associated with oocyte aging. Sci. China Life Sci. 2012, 55, 670–676. [Google Scholar] [CrossRef] [PubMed]
- Manosalva, I.; Gonzalez, A. Aging changes the chromatin configuration and histone methylation of mouse oocytes at germinal vesicle stage. Theriogenology 2010, 74, 1539–1547. [Google Scholar] [CrossRef] [PubMed]
- Lienert, F.; Mohn, F.; Tiwari, V.K.; Baubec, T.; Roloff, T.C.; Gaidatzis, D.; Stadler, M.B.; Schubeler, D. Genomic prevalence of heterochromatic H3K9me2 and transcription do not discriminate pluripotent from terminally differentiated cells. PLoS Genet. 2011, 7, e1002090. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Skutt-Kakaria, K.; Davison, J.; Ou, Y.L.; Choi, E.; Malik, P.; Loeb, K.; Wood, B.; Georges, G.; Torok-Storb, B.; et al. G9a/GLP-dependent histone H3K9me2 patterning during human hematopoietic stem cell lineage commitment. Genes Dev. 2012, 26, 2499–2511. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, Y.; Inoue, K.; Oikawa, M.; Kamimura, S.; Ogonuki, N.; Kodama, E.N.; Ohkawa, Y.; Tsukada, Y.; Ogura, A. Histone chaperone CAF-1 mediates repressive histone modifications to protect preimplantation mouse embryos from endogenous retrotransposons. Proc. Natl. Acad. Sci. USA 2015, 112, 14641–14646. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, Y.-L.; Tang, S.-B.; Shen, W.; Yin, S.; Sun, Q.-Y. Roles of Resveratrol in Improving the Quality of Postovulatory Aging Oocytes In Vitro. Cells 2019, 8, 1132. https://doi.org/10.3390/cells8101132
Sun Y-L, Tang S-B, Shen W, Yin S, Sun Q-Y. Roles of Resveratrol in Improving the Quality of Postovulatory Aging Oocytes In Vitro. Cells. 2019; 8(10):1132. https://doi.org/10.3390/cells8101132
Chicago/Turabian StyleSun, Yan-Li, Shou-Bin Tang, Wei Shen, Shen Yin, and Qing-Yuan Sun. 2019. "Roles of Resveratrol in Improving the Quality of Postovulatory Aging Oocytes In Vitro" Cells 8, no. 10: 1132. https://doi.org/10.3390/cells8101132
APA StyleSun, Y.-L., Tang, S.-B., Shen, W., Yin, S., & Sun, Q.-Y. (2019). Roles of Resveratrol in Improving the Quality of Postovulatory Aging Oocytes In Vitro. Cells, 8(10), 1132. https://doi.org/10.3390/cells8101132




