A New Immortalized Human Lacrimal Gland Cell Line
Abstract
1. Introduction
2. Materials and Methods
2.1. Surgical Removal and Patient Description Germany
2.2. Surgical Removal and RNA Sequencing from Lacrimal Glands
2.3. Electroporation
2.4. Tissue Preparation and Cell Culture
2.5. Collagen Coating
2.6. RNA Purification and Sequencing
2.7. RNA Sequencing and Data Analysis
2.8. cDNA Synthesis
2.9. Reverse Transcription (RT)-PCR and Gel Electrophoresis
2.10. Semiquantitative RT-PCR Analysis
2.11. Fluorescence Immunostaining of PAX6,FOXC1/2, CLDN5, KRT19 and OCLN
2.12. Fluorescence Immunostaining of SV40
2.13. Fluorescein Isothiocyanate (FITC)-Dextran Permeability Assay
2.14. Transepithelial Electrical Resistance (TEER) Measurement
2.15. Spheroid Culture
2.16. Histological Staining
2.17. Spheroid Analysis Area, Perimeter, Circularity, and Roundness
2.18. Three-Dimensional Culture in Extracellular Matrix Environment
2.19. Transmission Electron Microscopy
3. Results
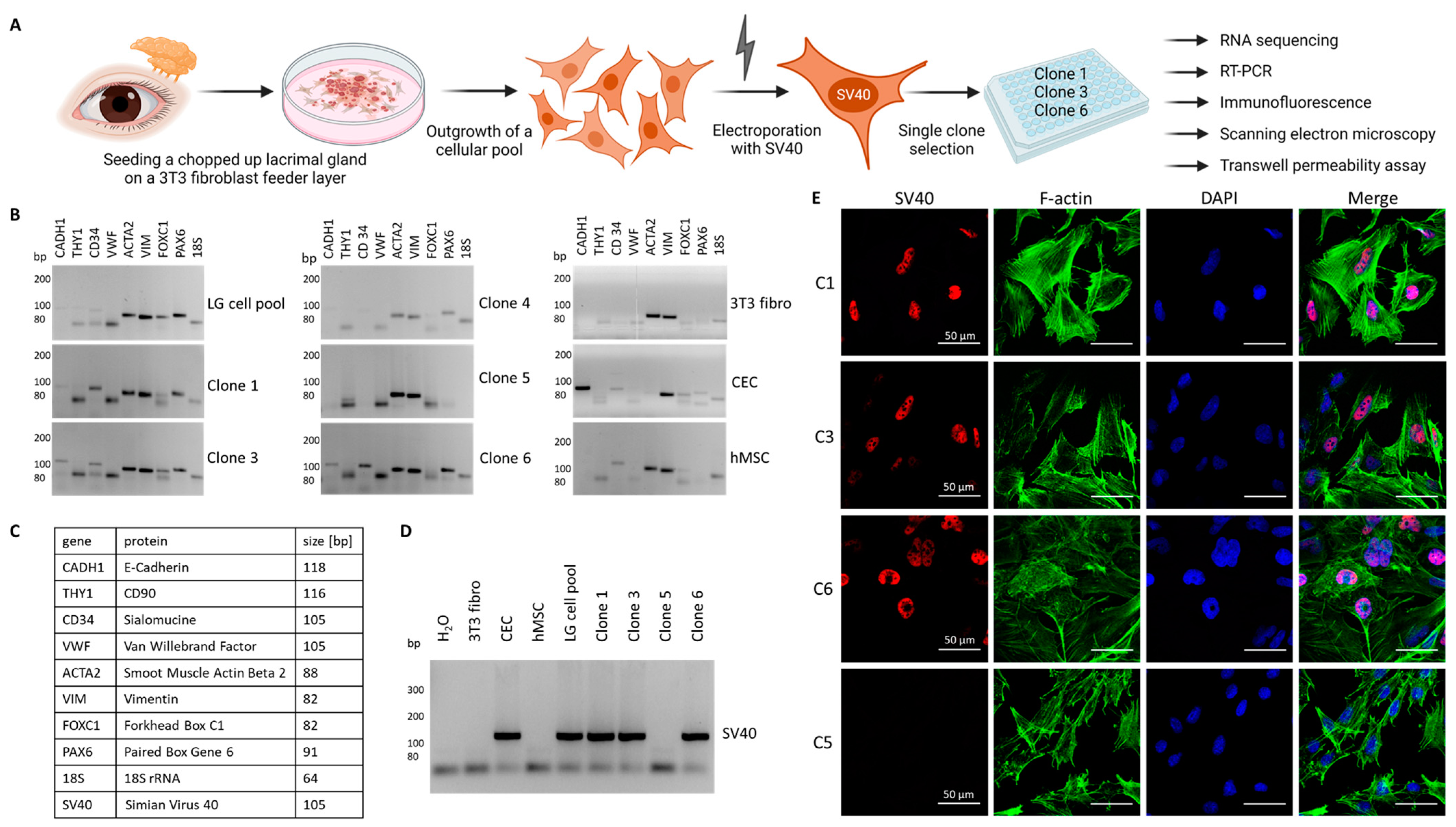
3.1. Generation of Immortalized Lacrimal Gland Cell Lines
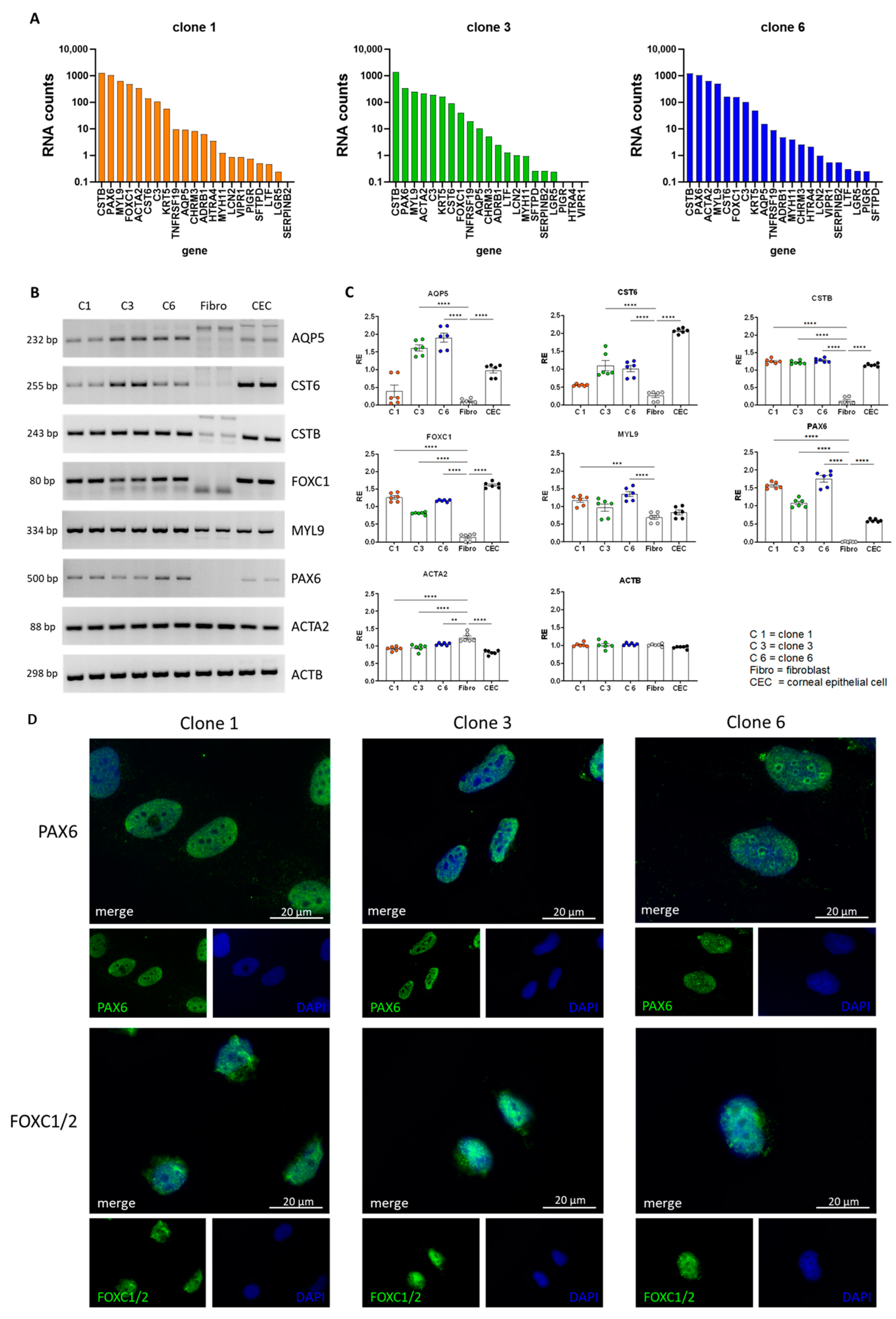
3.2. Total RNAseq of Immortalized Human Lacrimal Gland Cells
3.3. Validation of Lacrimal Gland Markers at the Transcriptional and Protein Levels
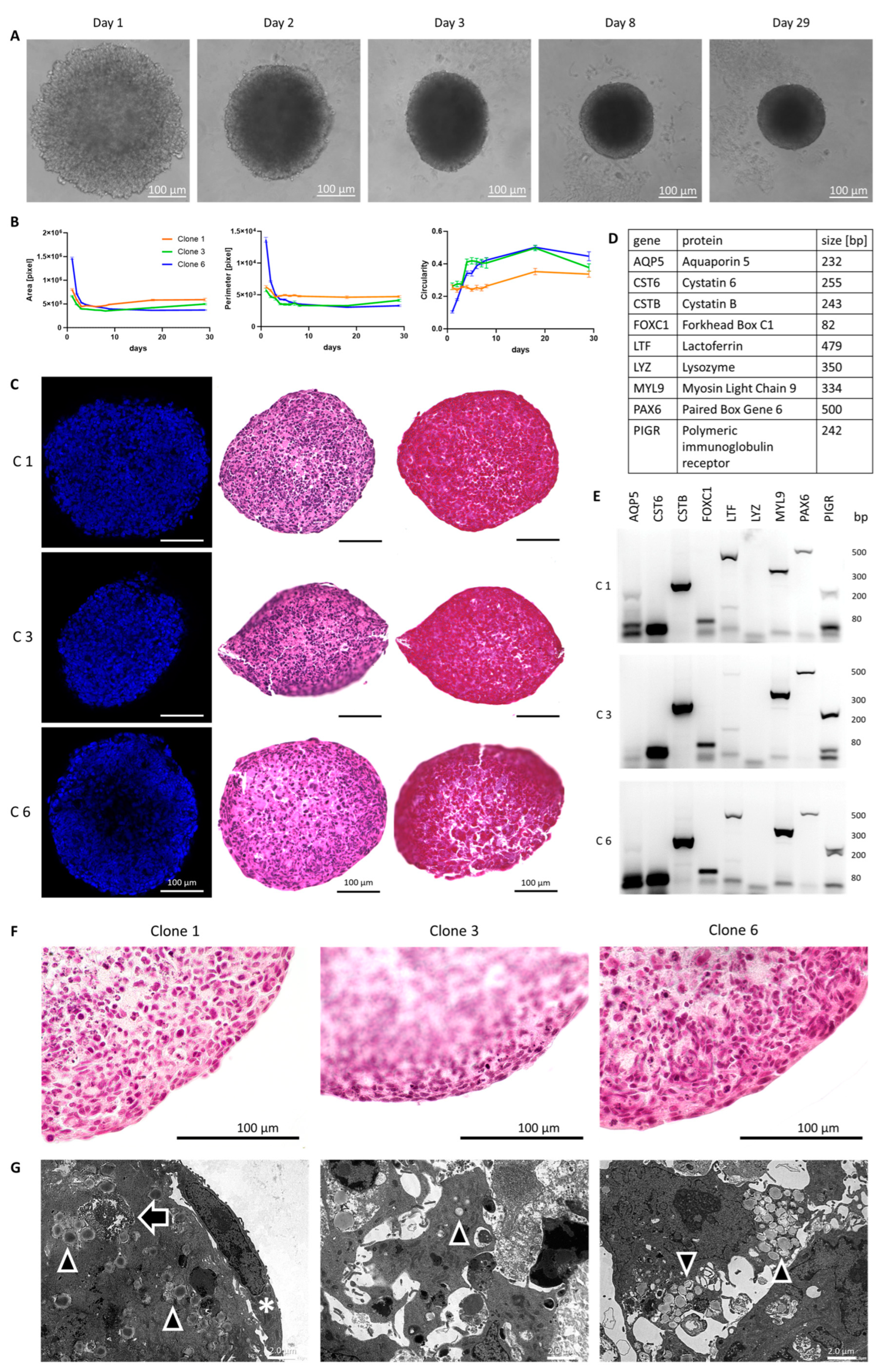
3.4. Utilization of Immortalized Cells in a 3D Spheroid Model
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Craig, J.P.; Nelson, J.D.; Azar, D.T.; Belmonte, C.; Bron, A.J.; Chauhan, S.K.; de Paiva, C.S.; Gomes, J.A.; Hammitt, K.M.; Jones, L.; et al. TFOS DEWS II Report Executive Summary. Ocul. Surf. 2017, 15, 802–812. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, F.; Alves, M.; Bunya, V.Y.; Jalbert, I.; Lekhanont, K.; Malet, F.; Na, K.-S.; Schaumberg, D.; Uchino, M.; Vehof, J.; et al. TFOS DEWS II epidemiology report. Ocul. Surf. 2017, 15, 334–365. [Google Scholar] [CrossRef] [PubMed]
- Belmonte, C.; Nichols, J.J.; Cox, S.M.; Brock, J.A.; Begley, C.G.; Bereiter, D.A.; Dartt, D.A.; Galor, A.; Hamrah, P.; Ivanusic, J.J.; et al. TFOS DEWS II pain and sensation report. Ocul. Surf. 2017, 15, 404–437. [Google Scholar] [CrossRef] [PubMed]
- Garg, A.; Zhang, X. Lacrimal gland development: From signaling interactions to regenerative medicine. Dev. Dyn. 2017, 246, 970–980. [Google Scholar] [CrossRef] [PubMed]
- Rocha, E.M.; Alves, M.; Rios, J.D.; Dartt, D.A. The aging lacrimal gland: Changes in structure and function. Ocul. Surf. 2008, 6, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Dartt, D.A. Dysfunctional Neural regulation of lacrimal gland secretion and its role in the pathogenesis of dry eye syndromes. Ocul. Surf. 2004, 2, 76–91. [Google Scholar] [CrossRef]
- Paulsen, F. Functional anatomy and immunological interactions of ocular surface and adnexa. Dev. Ophthalmol. 2008, 41, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Zhang, Y. The lacrimal gland: Development, wound repair and regeneration. Biotechnol. Lett. 2017, 39, 939–949. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Arnold, P.; Paulsen, F. Human lacrimal gland cell line: Potential techniques and translational implications. Exp. Eye Res. 2023, 229, 109430. [Google Scholar] [CrossRef]
- Schechter, J.E.; Warren, D.W.; Mircheff, A.K. A Lacrimal gland is a lacrimal gland, but rodent’s and rabbit’s are not human. Ocul. Surf. 2010, 8, 111–134. [Google Scholar] [CrossRef]
- Bannier-Hélaouët, M.; Post, Y.; Korving, J.; Bustos, M.T.; Gehart, H.; Begthel, H.; Bar-Ephraim, Y.E.; van der Vaart, J.; Kalmann, R.; Imhoff, S.M.; et al. Exploring the human lacrimal gland using organoids and single-cell sequencing. Cell Stem Cell 2021, 28, 1221–1232.e7. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Bothra, N.; Ali, M.J. Lacrimal gland targeted therapies for refractory epiphora: Quantitative changes in the gland activity. Orbit 2023, 43, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data. 2010. Available online: http://www.bioinformatics.babraham.ac.uk/projects/fastqc/ (accessed on 12 July 2023).
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Dobin, A.; Davis, C.A.; Schlesinger, F.; Drenkow, J.; Zaleski, C.; Jha, S.; Batut, P.; Chaisson, M.; Gingeras, T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics 2013, 29, 15–21. [Google Scholar] [CrossRef]
- Wang, L.; Wang, S.; Li, W. RSeQC: Quality control of RNA-seq experiments. Bioinformatics 2012, 28, 2184–2185. [Google Scholar] [CrossRef]
- Broad Institute. “Picard Toolkit”. Broad Institute, GitHub Repository. 2018. Available online: http://broadinstitute.github.io/picard/ (accessed on 12 July 2023).
- Okonechnikov, K.; Conesa, A.; García-Alcalde, F. Qualimap 2: Advanced multi-sample quality control for high-throughput sequencing data. Bioinformatics 2016, 32, 292–294. [Google Scholar] [CrossRef]
- Liao, Y.; Smyth, G.K.; Shi, W. feature Counts: An efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 2014, 30, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Kolde, R.K. R Package, version 1.7; Package ‘pheatmap’; R Core Team: Vienna, Austria, 2015. [Google Scholar]
- Wickham, H. ggplot2. Wiley Interdiscip. Rev. Comput. Stat. 2011, 3, 180–185. [Google Scholar] [CrossRef]
- Kassambara, A. R package, version 0.1 7; ggpubr: “ggplot2” Based Publication Ready Plots; R Core Team: Vienna, Austria, 2018. [Google Scholar]
- Foggetti, A.; Baccini, G.; Arnold, P.; Schiffelholz, T.; Wulff, P. Spiny and Non-spiny Parvalbumin-Positive Hippocampal Interneurons Show Different Plastic Properties. Cell Rep. 2019, 27, 3725–3732.e5. [Google Scholar] [CrossRef]
- Xu, S.; Ma, L.; Evans, E.; Okamoto, C.T.; Hamm-Alvarez, S.F. Polymeric immunoglobulin receptor traffics through two distinct apically targeted pathways in primary lacrimal gland acinar cells. J. Cell Sci. 2013, 126, 2704–2717. [Google Scholar] [CrossRef]
- Klećkowska-Nawrot, J.; Nowaczyk, R.; Goździewska-Harłajczuk, K.; Krasucki, K.; Janeczek, M. Histological, histochemical and fine structure studies of the lacrimal gland and superficial gland of the third eyelid and their significance on the proper function of the eyeball in alpaca (Vicugna pacos). Folia Morphol. 2015, 74, 195–205. [Google Scholar] [CrossRef]
- Araki-Sasaki, K.; Ohashi, Y.; Sasabe, T.; Hayashi, K.; Watanabe, H.; Tano, Y.; Handa, H. An SV40-immortalized human corneal epithelial cell line and its characterization. Investg. Ophthalmol. Vis. Sci. 1995, 36, 614–621. [Google Scholar]
- Hirayama, M.; Ko, S.B.; Kawakita, T.; Akiyama, T.; Goparaju, S.K.; Soma, A.; Nakatake, Y.; Sakota, M.; Chikazawa-Nohtomi, N.; Shimmura, S.; et al. Identification of transcription factors that promote the differentiation of human pluripotent stem cells into lacrimal gland epithelium-like cells. NPJ Aging Mech. Dis. 2017, 3, 1. [Google Scholar] [CrossRef]
- Makarenkova, H.P.; Ito, M.; Govindarajan, V.; Faber, S.C.; Sun, L.; McMahon, G.; Overbeek, P.A.; Lang, R.A. FGF10 is an inducer and Pax6 a competence factor for lacrimal gland development. Development 2000, 127, 2563–2572. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Tanzil, G.; Arnold, P.; Gögele, C.; Hahn, J.; Breier, A.; Meyer, M.; Kohl, B.; Schröpfer, M.; Schwarz, S. SV40 Transfected Human Anterior Cruciate Ligament Derived Ligamentocytes—Suitable as a Human In Vitro Model for Ligament Reconstruction? Int. J. Mol. Sci. 2020, 21, 593. [Google Scholar] [CrossRef]
- Pongkorpsakol, P.; Turner, J.R.; Zuo, L. Culture of Intestinal Epithelial Cell Monolayers and Their Use in Multiplex Macromolecular Permeability Assays for In Vitro Analysis of Tight Junction Size Selectivity. Curr. Protoc. Immunol. 2020, 131, e112. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.M.; Choi, M.E.; Jeon, E.J.; Park, J.-M.; Kim, S.; Park, J.E.; Oh, S.W.; Choi, J.-S. Cell-derived vesicles from adipose-derived mesenchymal stem cells ameliorate irradiation-induced salivary gland cell damage. Regen. Ther. 2022, 21, 453–459. [Google Scholar] [CrossRef] [PubMed]
- Lin, G.C.; Smajlhodzic, M.; Bandian, A.-M.; Friedl, H.-P.; Leitgeb, T.; Oerter, S.; Stadler, K.; Giese, U.; Peham, J.R.; Bingle, L.; et al. An In Vitro Barrier Model of the Human Submandibular Salivary Gland Epithelium Based on a Single Cell Clone of Cell Line HTB-41: Establishment and Application for Biomarker Transport Studies. Biomedicines 2020, 8, 302. [Google Scholar] [CrossRef]
- Saito, T.; Dai, T.; Asano, R. The hyaluronan synthesis inhibitor 4-methylumbelliferone exhibits antitumor effects against mesenchymal-like canine mammary tumor cells. Oncol. Lett. 2013, 5, 1068–1074. [Google Scholar] [CrossRef]
- Anbazhagan, R.; Bartek, J.; Monaghan, P.; Gusterson, B.A. Growth and development of the human infant breast. Am. J. Anat. 1991, 192, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Hayward, M.-K.; Allen, M.D.; Gomm, J.J.; Goulding, I.; Thompson, C.L.; Knight, M.M.; Marshall, J.F.; Jones, J.L. Mechanostimulation of breast myoepithelial cells induces functional changes associated with DCIS progression to invasion. NPJ Breast Cancer 2022, 8, 109. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Su, Y.; Fassl, A.; Hinohara, K.; Qiu, X.; Harper, N.W.; Huh, S.J.; Bloushtain-Qimron, N.; Jovanović, B.; Ekram, M.; et al. Perturbed myoepithelial cell differentiation in BRCA mutation carriers and in ductal carcinoma in situ. Nat. Commun. 2019, 10, 182. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.Y.; Kim, C.-H.; Lee, W.-S.; Kim, H.-N.; Song, K.-S.; Yoon, J.-H. Ciliary and secretory differentiation of normal human middle ear epithelial cells. Acta Otolaryngol. 2002, 122, 270–275. [Google Scholar] [CrossRef] [PubMed]
- Lagies, S.; Schlimpert, M.; Neumann, S.; Wäldin, A.; Kammerer, B.; Borner, C.; Peintner, L. Cells grown in three-dimensional spheroids mirror in vivo metabolic response of epithelial cells. Commun. Biol. 2020, 3, 246. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Eglen, R.M. Three-Dimensional Cell Cultures in Drug Discovery and Development. SLAS Discov. 2017, 22, 456–472. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Matsumoto, K.; Lish, S.R.; Cartagena-Rivera, A.X.; Yamada, K.M. Budding epithelial morphogenesis driven by cell-matrix versus cell-cell adhesion. Cell 2021, 184, 3702–3716.e30. [Google Scholar] [CrossRef]
- Puliafito, A.; Hufnagel, L.; Neveu, P.; Streichan, S.; Sigal, A.; Fygenson, D.K.; Shraiman, B.I. Collective and single cell behavior in epithelial contact inhibition. Proc. Natl. Acad. Sci. USA 2012, 109, 739–744. [Google Scholar] [CrossRef]

| Gene | Primer Sequence | Product Size (Base Pairs) | Annealing Temperature [°C] |
|---|---|---|---|
| ACTA2 | GCCGAGATCTCACCGACTAC TCACGGACAATCTCACGCTC | 88 | 60 |
| ACTB | GATCCTCACCGAGCGCGGCTACA GCGGATGTCCACGTCACACTTCA | 298 | 60 |
| AQP5 | TCCATTGGCCTGTCTGTCAC CTTTGATGATGGCCACACGC | 232 | 63 |
| CADH1 | AGGGGTTAAGCACAACAGCA ACGACGTTAGCCTCGTTCTC | 118 | 60 |
| CD34 | TAGACTGTGCAGTGATGTGGT GGCAGACTTGGCTAAAGGTCC | 105 | 60 |
| CD90 | TGGATTAAGGATGAGGCCCG GGGGAGGTGCAGTCTGTATT | 116 | 60 |
| CST6 | GGCAGCAACAGCATCTACTACTT ACAGTTGTGCTTTAGGAGCTGAG | 255 | 59 |
| CSTB | CGTGTCATTCAAGAGCCAGG CGCTCTGGTAGACGGAGGAT | 243 | 59 |
| FOXC1 | TCGGCTTGAACAACTCTCCA GACGTGCGGTACAGAGACTG | 82 | 60 |
| hu18S | GGAGCCTGAGAAACGGCTA TCGGGAGTGGGTAATTTGC | 64 | 60 |
| LTF | CAGACCGCAGACATGAAACT TTCAAGAATGGACGAAGTGT | 479 | 60 |
| LYZ | CTCTCATTGTTCTGGGGC ACGGACAACCCTCTTTGC | 350 | 60 |
| MYL9 | ACCCACCAGAAGCCAAGATG GCGTTGCGAATCACATCCTC | 334 | 63 |
| PAX6 | TAACCTGCCTATGCAACCCC ATAACTCCGCCCATTCACCG | 91 | 60 |
| PAX6 | AGTTCTTCGCAACCTGGCTA TGAACGTGCTGCTGATAGGA | 500 | 57 |
| PIGR | AATGCTGACCTCCAAGTGCTAAAG ATCACCACACTGAATGAGCCATCC | 242 | 60 |
| VIM | GCTTCAGAGAGAGGAAGCCG AAGGTCAAGACGTGCCAGAG | 82 | 60 |
| VWF | AGAACAGATGTGTGGCCCTG CTTCCGGTCCTGACAGACAC | 113 | 60 |
| Antibody | Host Species | Dilution | Company |
|---|---|---|---|
| PAX6 | Sheep | 1:50 | R&D Systems, Minneapolis, MN, USA |
| FOXC1/2 | Rabbit | 1:500 | Cohesion, Suzhou, China |
| SV40 | Mouse | 1:50 | Merck-Millipore, Darmstadt, Germany |
| F-Actin | Mouse | 1:200 | Invitrogen, Waltham, MA, USA |
| CLDN5 | Rabbit | 1:100 | Invitrogen, Waltham, MA, USA |
| KRT19 | Mouse | 1:50 | DAKO, Santa Clara, CA, USA |
| OCLN | Mouse | 1:250 | Thermo Fisher Scientific, Waltham, MA, USA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gleixner, S.; Zahn, I.; Dietrich, J.; Singh, S.; Drobny, A.; Schneider, Y.; Schwendner, R.; Socher, E.; Blavet, N.; Bräuer, L.; et al. A New Immortalized Human Lacrimal Gland Cell Line. Cells 2024, 13, 622. https://doi.org/10.3390/cells13070622
Gleixner S, Zahn I, Dietrich J, Singh S, Drobny A, Schneider Y, Schwendner R, Socher E, Blavet N, Bräuer L, et al. A New Immortalized Human Lacrimal Gland Cell Line. Cells. 2024; 13(7):622. https://doi.org/10.3390/cells13070622
Chicago/Turabian StyleGleixner, Sophie, Ingrid Zahn, Jana Dietrich, Swati Singh, Alice Drobny, Yanni Schneider, Raphael Schwendner, Eileen Socher, Nicolas Blavet, Lars Bräuer, and et al. 2024. "A New Immortalized Human Lacrimal Gland Cell Line" Cells 13, no. 7: 622. https://doi.org/10.3390/cells13070622
APA StyleGleixner, S., Zahn, I., Dietrich, J., Singh, S., Drobny, A., Schneider, Y., Schwendner, R., Socher, E., Blavet, N., Bräuer, L., Gostian, A.-O., Balk, M., Schulze-Tanzil, G., Günther, C., Paulsen, F., & Arnold, P. (2024). A New Immortalized Human Lacrimal Gland Cell Line. Cells, 13(7), 622. https://doi.org/10.3390/cells13070622








