Simple Summary
Hepatoblastoma is the most common malignant pediatric tumor of the liver. Unlike hepatocellular carcinoma (HCC) which has been associated with hepatitis B virus infection or cirrhosis, the etiology of hepatoblastoma remains vague. Genetic syndromes, including familial adenomatous polyposis (FAP), Beckwith-Wiedemann syndrome (BWS), and trisomy 18 syndrome, have been associated with hepatoblastoma. BWS is an overgrowth syndrome which exhibits an alteration of genomic imprinting on chromosome 11p15.5. N6-Methyladenosine (M6A) is an RNA modification with rampant involvement in the metabolism of cells and malignant diseases. It has been observed to impact the development of various cancers via its governance of gene expression. Here, we explore the role of m6A and its genetic associates in promoting HB, and the impact this may have on our future management of the disease.
Abstract
Hepatoblastoma (HB) is a rare primary malignancy of the developing fetal liver. Its course is profoundly influenced by genetics, in the context of sporadic mutation or genetic syndromes. Conventionally, subtypes of HB are histologically determined based on the tissue type that is recapitulated by the tumor and the direction of its differentiation. This classification is being reevaluated based on advances on molecular pathology. The therapeutic approach comprises surgical intervention, chemotherapy (in a neoadjuvant or post-operative capacity), and in some cases, liver transplantation. Although diagnostic modalities and treatment options are evolving, some patients experience complications, including relapse, metastatic spread, and suboptimal response to chemotherapy. As yet, there is no consistent framework with which such outcomes can be predicted. N6-methyladenosine (m6A) is an RNA modification with rampant involvement in the normal processing of cell metabolism and neoplasia. It has been observed to impact the development of a variety of cancers via its governance of gene expression. M6A-associated genes appear prominently in HB. Literature data seem to underscore the role of m6A in promotion and clinical course of HB. Illuminating the pathogenetic mechanisms that drive HB are promising additions to the understanding of the clinically aggressive tumor behavior, given its potential to predict disease course and response to therapy. Implicated genes may also act as targets to facilitate the evolving personalized cancer therapy. Here, we explore the role of m6A and its genetic associates in the promotion of HB, and the impact this may have on the management of this neoplastic disease.
1. Introduction
Hepatoblastoma (HB) is a rare and heterogeneous malignancy deriving from embryonic liver parenchyma. Despite affecting fewer than two children in a million, it represents over a third of primary tumors of the pediatric liver [1,2,3,4]. Its genesis and progression are highly genetically mediated, indicated by sporadic forms adjacent to a strong syndromic association, but also intimately associated with the frequent occurrence of extramedullary hematopoiesis, as identified in early studies on the developing intrahepatic biliary system of the fetal liver [5].
Subtypes are described histologically, based on the ratio of epithelial to mesenchymal tissues present and their stage of aberrant differentiation [6]. Fetal-type tumors are further differentiated than their more immature embryonal counterparts. Due to the architectural and cytological complexity of tumors, and compounded by their rarity, consensus between experts is not always established. This is particularly observed amongst subtypes such as small cell undifferentiated, which unfortunately traverses a less favorable clinical course [2,4,6]. The 5-year survival rate of patients with HB approaches 80% due to advances in early detection and multidisciplinary therapy, however, it remains challenging to predict the clinical course. Relapse of disease, metastatic spread, and variable response to chemotherapy are grave concerns that affect many patients [4,7].
N6-Methyladenosine (m6A) is an extraordinarily prevalent modification of eukaryote RNA [8,9]. Its presence governs multiple facets of normal cellular processing, differentiation, and cell cycle regulation. It is widely implicated in malignant diseases [10].
Epigenetics has been recognized as a novel concept pinpointing the epigenetic trait as a firmly heritable phenotype, which arises from modifications in a chromosome devoid of alterations in the DNA sequence. There are more than one hundred chemical modifications (of which m6A is one) that have been identified on RNAs. Such changes harbor significant biological functions in living organisms using their involvement in intervening in epigenetic regulation. Such a role has been detected mainly in the origin and progression of hematologic malignancies [11]. At present, epitranscriptomics, represented by m6A changes, has grown into a major research topic. This development is in part linked to second-generation sequencing tackling epigenetic modification at the transcriptome level. At its core, m6A methylation changes can arbitrate the post-transcriptional control of gene expression devoid of altering base sequences. In adding together to this concept, RNA m6A change is (1) reversible and (2) dynamically modulated by modifying molecules or modifiers (e.g., writers, readers, and erasers). They have presently been demonstrated to play a crucial role in regulating not only mRNA decay and stability, but also splicing, translation efficiency, and localization. Further, m6A sites have also been recognized in long non-coding RNAs as well as non-coding RNAs, such as microRNAs. The development of high throughput m6A sequencing technology has enormously accelerated in recent years. There is accumulated evidence that m6A and its related factors are involved in HB [12,13,14]. It seems that the regulation consists of the self-renewal, proliferation, and differentiation of the neoplastic cell.
Some antibody-dependent (such as methylated RNA immunoprecipitation sequencing (MeRIP-seq) and m6A individual-nucleotide-resolution cross-linking and immunoprecipitation (miCLIP)) and antibody-independent (such as MAZTER-seq deploying a sequence-specific, methylation-sensitive, single-stranded ribonuclease MazF, m6A-sensitive RNA-endoribonuclease–facilitated sequencing, and deamination adjacent to RNA modification targets sequencing or DART-seq) are terrific tools for detecting and measuring RNA modifications [15]. Next-generation sequencing (NGS) methods have been developed for m6A sequencing, making high-resolution detection of m6A epitranscriptomes in diverse cell contexts a reality [16]. Indeed, the advent of such NGS-based methods advanced our understanding of this epigenetic marker.
Here, we review the advancement of research on the biological characteristics of m6A methylation in HB. We hope to provide a basis for developing molecularly targeted therapies established on the aberrant m6A modifications in potentially related hematologic neoplasms.
2. The Form and Function of m6A
Modifications frequently occur within the mRNA of eukaryotic organisms and have been implicated in a plethora of normal cellular processes, as well as in the governance of cancers [10,17]. The most numerous of these modifications is m6A, a post-transcriptional methylation occurring at the N6-position of the adenosine base. In particular, m6A is highly conserved across normal body tissues and in malignant cell populations, implying its durability and stability, as well as its wide-reaching function. Simply put, it affects every aspect of RNA metabolism [10].
The process of m6A methylation is dynamic and reversible, akin to other forms of epigenetic regulation, supporting the proposal that it falls under this umbrella. The formation of m6A occurs through a methyltransferase complex comprising a methyltransferase-like (METTL) 3 and METTL14 heterodimer in a catalytic core and a multi-protein regulatory subunit, prominently featuring Wilms’ tumor 1-associated protein (WTAP) [12,18]. The effects of m6A are breathtaking in their diversity; fittingly, they are mediated by an expanding list of biological components, broadly grouped into readers, writers, and erasers according to the way in which they interact with m6A (Figure 1).
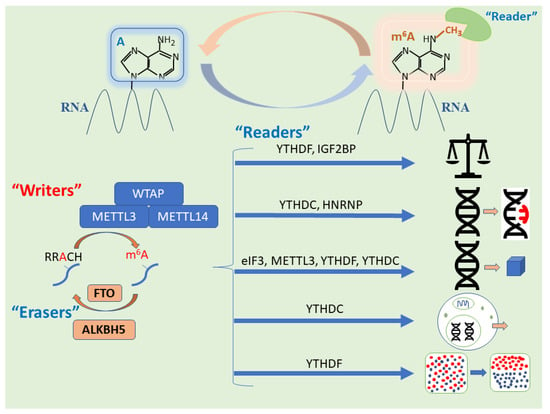
Figure 1.
The m6A methylation is catalyzed by the writer complex, including METTL3, METTL14, METTL16, WTAP. The m6A modification is erased by demethylases, including FTO and ALKBH5, where FTO is preferentially responsible for the demethylation of N6,2′-O-dimethyladenosine (m6Am) at the 7-methylguanosine cap. The m6A-modified RNA reader proteins include YTHDF1/2/3, YTHDC1/2, IGF2BP1/2/3, and HNRNPC/A2B1. M6A modification modulates miRNA biogenesis, inactivation, m6A switch, RNA translocation, pre-mRNA splicing, RNA translation, RNA decay, and RNA stability. WTAP, Wilms’ tumor 1-associated protein; eIF3, eukaryotic initiation factor 3; YTHDF, YT521-B homology domain family; YTHDC, YT521-B homology domain-containing protein; IGF2BP, insulin-like growth factor 2 mRNA-binding protein; HNRNP, heterogeneous nuclear ribonucleoproteins; FTO-CTD, FTO C-terminal domain.
The behavior of certain cancers, including their genesis and progression, has long been linked to the expression of tumor-related genes. In this context, the modulation of heritable gene expression without DNA sequence changes is often stimulated by alteration of m6A levels [8]. The sequence in which m6A methylation takes place is catalyzed by a host of genes that make up the writer complex, an elaborate conversation in which substrates are installed into the m6A methylation prior to the determination of downstream RNA fate by other reader proteins [9]. Regulators of m6A RNA methylation are involved in various human diseases through the effects they exert and thus provide potential targets for cancer therapy [8].
Characterization of the genetic and epigenetic promotion of rare malignancies such as HB is of great clinical importance. Histologic subtyping suggests that some patterns experience worse outcomes than others, from aggressive growth and early spread to resistance to current chemotherapeutic mechanisms. Poorly differentiated subtypes, such as the small cell undifferentiated histologic subtype, represent a diagnostic and management challenge due to their rarity and recalcitrant behavior. The acquisition of mesenchymal features, even in small quantities, correlates with chemoresistance [1]. Troublesome subtypes consistently show the arrest of normal cellular development at an earlier embryologic stage [19]. Processes of genetic modification that impact the differentiation of embryologic tissues, such as those governed by m6A, have been of particular interest. In addition to identifying therapeutic targets, understanding the mechanism by which the disease propagates may identify those who are predisposed outside of the established genetic syndromes, stratify risk, and offer clarity around patient outcomes [7,8,10].
The m6A methylation processing and its biological functions is depicted in Figure 1, Figure 2, Figure 3 and Figure 4. Figure 1 displays m6A writers (methyltransferase) methylating RNA in the adenine nucleobase of the amino group at N6 position. The consensus motif of METTL3 is RRA*CH (R = A/G; A* = methylated A; H = A/C/U). Once m6A is deposited on RNA, m6A reader proteins are recruited and determine RNA fates, such as splicing, stability, and translation efficiency, ultimately affecting gene expression. Also, m6A is removed from RNA through demethylation by eraser proteins.
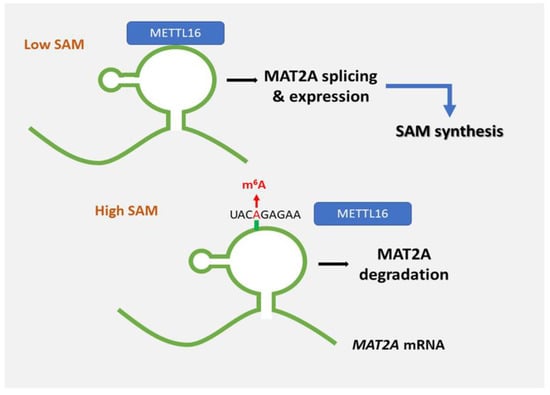
Figure 2.
METTL16 methylates a stem-loop structure in the 3′ untranslated region (UTR) of S-adenosyl methionine (SAM) synthase, methionine adenosyltransferase 2A (MAT2A). In SAM-repleted conditions, MAT2A is methylated and degraded. Conversely, in SAM-depleted conditions, METTL16 induces splicing and expression of MAT2A.
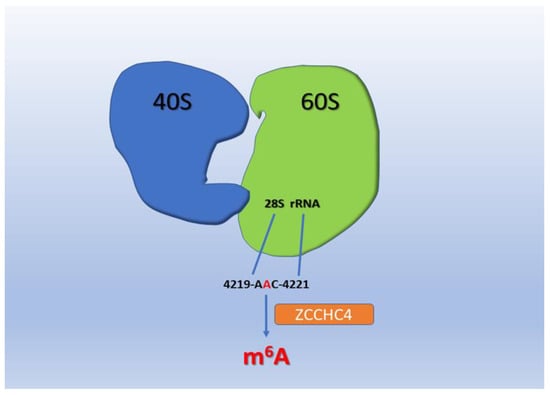
Figure 3.
Methylation of A4220 in 28S ribosomal RNA (rRNA) by zinc finger CCHC domain-containing protein 4 (ZCCHC4) promotes ribosome assembly and translation.
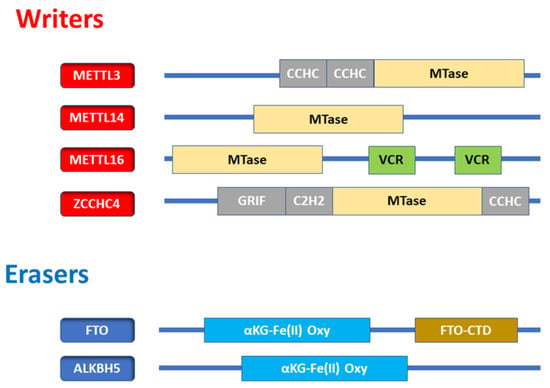
Figure 4.
The domain composition of m6A enzymes. (Top, writers) m6A writers contain methyltransferase (MTase) domains. METTL3 contains Cys-Cys-Cys-His (CCCH) zinc finger motifs. METTL16 has two vertebrate-conserved regions (VCR).
Zinc finger CCHC domain-containing protein 4 (ZCCHC4) possesses several zinc finger motifs, including Gly-Arg-Phe (GRF), Cys2-His2 (C2H2), and Cys-Cys-His-Cys (CCHC) domains. Methyltransferase-like 5 (METTL5), acts as a conserved methyltransferase, specifically catalyzing m6A at the 18S A1832 motif. METTL5 allows S-adenosyl-L-methionine binding activity in addition to rRNA (adenine-N6-)-methyltransferase activity. METTL5 is involved in the positive regulation of translation and rRNA methylation [20]. Fat mass and obesity-associated protein (FTO) and alkb homolog 5 (ALKBH5) contain KGFe(II)-dependent dioxygenase domains conserved in dioxygenase family enzymes.
FTO’s role as an m6A eraser is controversial since several studies showed that FTO is solely responsible for the demethylation of m6A on snoRNA 5′ cap [21,22,23]. Essentially, FTO preferentially demethylates m6Am (N6,2′-O-dimethyladenosine) detected in 5′-caps of mRNAs, rather than m6A [23]. Several m6A detection technologies unintentionally map m6Am changes as m6A sites. Overall, the thought of reversible m6A remains controversial. The biological role and evidence of its removal are still poorly understood. It, obviously, adds a layer of complexity when trying to associate m6A with neoplastic progression. Dominissini et al. introduced the human and mouse m6A modification landscape in a transcriptome-wide manner based on antibody-mediated capture and parallel sequencing [24]. They identified over 12,000 m6A sites showing a typical consensus in the transcripts of more than 7000 human genes. Two distinct landmarks appeared, the one around stop codons and the other within long internal exons. Both are highly conserved between humans and mice [22,23,24,25,26,27]. Additionally, Meyer et al. found that m6A is massively enriched in the three prime untranslated regions (3′ UTRs) as well as near the stop codon in mature mRNA [28]. Figure 2 displays that METTL16 methylates a stem-loop structure in the 3′ untranslated region (UTR) of S-adenosyl methionine (SAM) synthase, methionine adenosyltransferase 2A (MAT2A). In SAM-repleted conditions, MAT2A is methylated and degraded. Conversely, in SAM-depleted conditions, METTL16 induces splicing and expression of MAT2A. Figure 3 displays the methylation of A4220 in 28S ribosomal RNA (rRNA), ZCCHC4 promotes ribosome assembly and translation.
Figure 4 exhibits the domain composition of m6A enzymes. In addition, m6A writers contain methyltransferase (MTase) domains. METTL3 contains CCCH zinc finger motifs. METTL16 has two vertebrate-conserved regions (VCR) or domains.
3. Key Genetic Players in Hepatoblastoma
Many cytogenetic changes have been cataloged in HB. Most of these changes result from highly penetrant somatic mutations in undifferentiated or minimally differentiated fetal hepatocyte precursors. Such modifications often involve the Wnt signaling pathway, influencing the fate of the transcriptional cofactor beta-catenin [29,30,31]. Beta-catenin is encoded by CTNNB1, a proto-oncogene with a pivotal role in cell-to-cell communication and adhesion [12,32].
As a critical component of the Wnt/beta-catenin signaling pathway, CTNNB1 is mutated in 50–90% of HB [12]. CTNNB1 is consistently upregulated in HB tumor cells and, when knocked down, reduces their viability and induces apoptosis [12]. Furthermore, there is a statistically significant correlation between the expression of CTNNB1 and METTL3, which speaks to an intimate relationship between the two components. The m6A modulation of METTL3 in turn regulates the processing of CTNNB1, sharing its responsibility in driving HB [32]. TERT is a regulator of telomerase, an enzyme related to cell immortality, and a significant player in Wnt signal activation [33]. Its expression is enhanced by MYC, which is itself an activated target gene of Wnt signaling. In this way, a vicious cycle can occur, by which upregulation of the Wnt signaling pathway further stimulates its drivers [34].
METTL3 is implicated in malignant disease and normal physiological processes, such as regulating hematopoietic stem cell differentiation [18]. In addition to its function as the catalytic core of the m6A writer complex, METTL3 can act explicitly as an oncogene, promoting HB proliferation while spurring tumor growth by virtue of its role in m6A modification [12,18].
Much like other genes involved in the modification of m6A, WTAP exhibits diverse and complex biological functions. It has been established as tumorigenic in many cancers, including hepatocellular carcinoma (HCC) and osteosarcoma, particularly, through its impact on m6A [35,36]. In the governance of normal embryological development and cellular processing, WTAP is a key player, engaging in transcriptional and post-transcriptional regulation of cellular genes to guide the organization of biological structures such as the genitourinary system and in various facets of cell-cycle regulation [37,38]. Intrinsic to normal biology and pathology alike, WTAP demonstrates a similar duality to m6A itself and many of its associated genes.
Regarding its role as a risk factor for HB, Zhuo et al. propose that single nucleotide polymorphisms of WTAP potentially predispose to HB development, acting in this context as a genetic modifier [7]. Upregulated expression levels of the WTAP gene appeared to increase the risk of developing HB in children. Certain genotypes were observed to modify the risk in specific sub-populations: children under seventeen months of age, female children, and progression in those with stage I or II tumors [7].
The YT521-B homology (YTH) domain family members, including YTHDC1 and YTHDF2, are widespread and highly conserved within eukaryotic cells [35]. They function to specifically bind m6A and mediate its interaction with mRNAs; this established function places them under scrutiny for their role in m6A dependent cancers [18]. However, whilst each has been linked to pathological processes in diverse body tissues, their contribution to the development of HB has only recently been investigated.
YTHDC1 is a nuclear m6A reader, regulating RNA splicing in a concentration-dependent manner [35]. Its role in mRNA processing has been implicated in numerous cancer-driving processes, such as angiogenesis, growth factor signaling, metastatic spread, apoptosis, and genetic instability [35]. Polymorphisms of m6A-associated genes play an essential role in HB, and preliminary investigation suggests that YTHDC1 is no different [32]. While polymorphisms selected by Chen et al. did not contribute to HB susceptibility, stratification analysis within the same study did detect the potential contribution of YTHDC1 to HB risk [35,39]. This finding begs further study in a larger cohort to tease out additional polymorphisms of interest.
YTHDF2 also functions as a crucial reader, usually modifying the degradation of m6A-modified mRNAs [39]. The study of its relationship to HB is in its infancy. Still, preliminary work by Cui et al. suggests that its role in developing HB may confer unfavorable clinical outcomes [13].
4. The Impact of m6A on Tissue Development
The proliferation of HB is governed by the aberrant differentiation of hepatocytes in early life [6,29]. Whether malignant transformation occurs during the earliest phases of differentiation or through gene mutations that impact an already differentiated tissue, abnormal methylation is ubiquitous in implicated genes. The disseminated effects of losing m6A modification agents, such as METTL3, impact the developmental stages of a variety of tissues [8]. Loss of METTL3 in animal models, such as in the murine fetal liver and in zebrafish embryogenesis, can activate downstream signaling pathways in hematopoietic stem cells and progenitor cells and influence Notch-dependent signaling [18].
Specifically, m6A methylation exhibits a dynamic pattern of downstream effects in the developing liver. To catalogue effects and pursue an underlying mechanism, a transcriptome-wide investigation of porcine liver at different developmental stages has been performed, emphasizing the relationship between m6A methylation and gene expression6. Genes involved with hepatocyte differentiation and liver development are consistently highly m6A methylation dependent. Interruption of the normal function of this process can reasonably be suspected in many cases in which fetal hepatocytes undergo aberrant development [40].
5. Promotion of Hepatoblastoma via the m6A Pathway
The promotion of cancer via m6A modification is achieved through various mechanisms. As we have described, the biological effects of m6A are diverse and associated with normal eukaryote physiology as much as they are implicated in disease. When physiological processes such as DNA damage responses and pluripotency deviate from normal, aberrant m6A modification is often the culprit [41]. The m6A RNA methylation promotes cancer via two overarching roles: a tumor promotor or a suppressor of innate anti-tumor mechanisms. An example of the former is the modification of m6A methylation by METTL3 in its role as a proto-oncogene, enhancing m6A modification and degradation of SOCS2 with the subsequent development of HCC. As a suppressor of protective biological functions, METTL3 downregulation by m6A modulates in the context of endometrial cancer, leading to increased cell proliferation and tumorigenicity [28]. Other familiar characters come to light when investigating the clinical significance of genes associated with m6A modification in a pathological context, chiefly: CTNNB1, METTL3, WTAP, YTHDC1, and YTHDF2 [12,13]. Preliminary studies have identified polymorphisms in several of these genes, however, such studies are bound by confounding factors that must be acknowledged: firstly, sample sizes are often small due to the rarity of HB in all populations, a problem that is amplified when subgroups are stratified. Secondly, it can be problematic to extrapolate from genetic studies in isolation. Finally, HB is a profoundly heterogeneous disease and therefore should be considered alongside environmental factors. As a nod to its biological heterogeneity, m6A does not function solely to propagate cancer. Instead, it demonstrates a more nuanced dual role, in some instances inhibiting tumor progression. For example, in glioblastoma multiforme (GBM), the downregulation of m6A leads to decreased levels of the ADAM19 gene, enhancing its expression [10]. The enhanced expression of this gene promotes cellular self-renewal and tumorigenesis. A similar phenomenon is observed in METTL3 within animal embryologic models, in which knockdown of METTL3 is associated with reduced self-renewal10. HB exhibits this complex relationship well, owing to its propagation of a flurry of m6A-mediated promotions and cellular circuit breaks. The overexpression of m6A has been a ubiquitous observation in HB. Since the Wnt/beta-catenin signaling pathway primarily drives HB, it can be reasonably extrapolated that upregulation in m6A causes tumor growth by influencing genes common to this system. Liu et al. showed that genes such as CTNNB1, CCND1, and NKD1 showed increased m6A methylation compared with non-tumor tissues [12]. There is a consistently observed tendency for m6A modification to aggregate in HB, emphasizing its role in the malignant process. METTL3 is frequently identified as a critical player in HB development machinery [32]. In addition to its impact, METTL3 influences CTNNB1 as a downstream target. CTNNB1 is an infamous proto-oncogene encoding beta-catenin, a protein mutated in most HB cases [4]. In this context, the mutated form of beta-catenin is degradation resistant, accumulating in the nucleus of hepatocyte precursors and binding to the transcription factor TCF4/LEF-1; in doing so, it activates target genes such as c-MYC and Cyclin-D1 [42]. The impaired degradation of beta-catenin and its associated impact on downstream genes is a common theme in proposed mechanisms of HB development. Loss of function somatic mutations in the tumor suppressor genes AXIN1 and AXIN2 impair beta-catenin degradation, augmenting the above carcinogenic pathway [43,44].
The workhorse of m6A formation is the METTL3/METTL14 heterodimer and its regulatory subunit, which features WTAP in prominence [21,45]. The genesis and propagation of HB are linked to abnormal m6A modification. An elevated level of m6A is identified in tumor cells, alongside upregulation of a familiar cohort of genes, including METTL3, WTAP, etc. FTO and YTHFD2 [8,45,46,47,48]. Interestingly, no upregulation of METTL14 is identified within tumor cells compared with background liver [8]. Liu et al. investigated the functional role of m6A affiliated genes in HB. Knockdown of the above mentioned, overexpressed genes markedly suppressed HB cell proliferation and caused HB cells to undergo apoptosis [12]. This translates clinically to a reduction in tumor size and weight, when METTL3 is knocked out stably in a murine model10. In keeping with the sequence of downstream effects of METTL3 in vitro, the transcription factor of CTNNB1 (TCF4/LEF-1) and target gene (Cyclin-D1) were downregulated in the knockout murine HB cells [12]. From this observation, it is not too far a stretch to propose the downregulation of CTNNB1 itself and subsequently of the beta-catenin product in vivo, in a METTL3 knockout model. METTL3 has been isolated as the main factor in aberrant m6A modification in the context of HB, above that of WTAP, its co-catalytic associate. The mechanism by which WTAP influences the development and progression of HB itself remains undetermined. Zhuo et al. highlight the ability of certain single nucleotide polymorphisms to enhance neighboring critical genes. For example, TCP1 is enhanced by a functional genotype of WTAP and is known to be implicated in several solid organ malignancies. The door is open for the study of additional single nucleotide polymorphisms of WTAP, which may be of functional significance.
6. The Role of m6A Associated Genes as Diagnostic and Prognostic Biomarkers
There is a significant correlation between an increased protein expression level of m6A modifiers and HB. This prompts important clinical questions regarding the utility of associated genes as diagnostic and prognostic biomarkers. Cui et al. illustrate the function of METTL3 as an oncogene in HB, regulated by the micro-RNA miR-186, to propagate HB via the Wnt/beta-catenin signaling pathway [13]. The axis of interaction between METTL3 and its micro-RNA modifier may represent a therapeutic target or prognostic biomarker for patients with HB. In addition, polymorphisms of METTL3 may affect the occurrence of HB; this idea is extrapolated to suggest that certain genotypes can incur a greater risk of HB development [12,32]. Prediction of susceptibility and clinical course is of great importance for a disease in which early detection and treatment correlate strongly with survival. Complementing this, Liu et al. explored the relationship between METTL3 expression and the clinicopathological characteristics of patients with HB. Increased METTL3 expression was associated with frequent recurrence and poor survival [32]. Increased expression of YTHDF2, the m6A reader of a different mechanism, is also significantly associated with poor overall survival rates in HB [13]. These observations add to existing histologic descriptions, forming a helpful constellation of features that predict poor outcomes. While undoubtedly in its early stages, such research offers promise for the diagnosis, prognosis, and risk stratification of susceptible or existing patients10. Early exploration of single nucleotide polymorphisms of WTAP hint at the potential to discover additional functional genotypes, which may elucidate the mechanisms that drive HB. This understanding will enable a more accurate catalog of mutation sequences in the evolution of HB. It will perhaps inform how aberrant METTL3 expression is understood under their shared role in m6A modification. The identification of HB with high malignant potential is a step towards personalized, precise management, supplementing clinical predictors such as alpha-fetoprotein level (AFP), age at diagnosis and pre-treatment extent at disease (PRETEXT) classification [49]. Description of the pathogenesis of HB and the illumination of key genetic players has not always correlated with tumor behavior and risk stratification, which is of immediate utility to clinicians and patients. Meaningful correlation has been hampered by small patient sample sizes and the sheer range of molecular and genetic targets analyzed. Nagae et al. describe a relationship between age at diagnosis, and the vista of known genetic aberrations [49]. A changing pattern of gene mutation was identified as the age at diagnosis increased. Amongst patients younger than eight years old, mutation of CTNNB1 formed the overwhelming majority [49]. Patients in which TERT is frequently mutated tend to be older, approaching young adolescence [49]. TERT mutation has been implicated in particularly aggressive tumors, which is in keeping with the increase in discordant tumor behavior with advancing age at diagnosis [49,50,51]. The changing genetic landscape of tumors with patient age also raises the possibility of discrete tumor biology governing clinical expression [49].
The idea that tumor biology may vary according to clinical behavior is further supported by the description of different epigenetic hallmarks within the HB cohort [52]. Such hallmarks are qualified by the degree of hyper- or hypomethylation of DNA, leading to discriminable effects on the transcriptome [52]. Since the transcriptome influences the molecular behavior of the tumor, it follows that teasing out such epigenetic profiles may be of value to prognostication and risk stratification [52]. An example of this idea in practice is the repression of tumor suppressors in aggressive HB, by defined epigenetic hypermethylation: CpG island hypermethylation. By extrapolating advances in the understanding of HB epigenetics, Carrillo-Reixach et al. proposed the utility of a molecular risk stratification system, which may augment existing clinical models [52].
Moving beyond prognostication to suggest mechanisms of treatment, genes implicated in the immune microenvironment of tumors are of particular interest. The tumor suppressor gene AXIN1, alongside other aberrantly methylated genes LAMB1 and NOTUM, were deemed by Zhang et al. to be closely related to immune cells in hepatoblastoma tissues, in which they were upregulated [43]. This finding is of great potential utility as NOTUM knockdown subsequently attenuated growth, migration, and invasion of tumor cells in a murine model [43]. Inhibition of the gene CHKA, involved in membrane biosynthesis in the context of normal cellular function, has arrested tumor growth in a selection of tumor types, including HB, HCC, breast, lung, and prostate [52]. This finding emphasizes the implicitly shared pathways in tumorigenesis across different body sites and suggests CHKA as a target for future HB treatment [52]. As aberrantly methylated and expressed genes are cataloged in droves, their potential therapeutic roles can be investigated by describing the interplay between genes and other cancer-modulating mechanisms such as the host immune response.
Recently, specific inhibitors of both m6A eraser and writers have been developed. The design of 20 molecules with low micromolar IC50′s and specificity toward FTO over ALKBH5 confirmed two competitive inhibitors, FTO-02 and FTO-04. Remarkably, FTO-04 prevented neurosphere formation in patient-derived glioblastoma stem cells (GSCs) without preventing the growth of healthy neural stem cell-derived neurospheres. FTO-04 increased m6A and m6Am levels in GSCs consistent with FTO inhibition as well. This data may quite strongly support FTO-04 as a potential new lead for the therapeutic handling of glioblastoma [53]. Besides, other FTO inhibitors such as FB23/FB23-2, MA/MA2, MO-I-500, and, intriguingly, Entacapone have been delineated to harbor a binding ability toward FTO with the aim to reduce the RNA demethylase activity, which should have anti-cancer effects other than anti-obesity effects [54]. Also, METTL3 inhibitors and FTO inhibitors have been proposed to harbor an immunotherapeutic potential in controlling acute myeloid leukemia [55].
7. Detection of RNA Modification by Nanopore Sequencing
There are several methods to detect RNA modifications, including antibody immunoprecipitation (e.g., MeRIP-seq, miCLIP) [56,57] and chemical-based modification, required to convert RNA to complementary DNA (cDNA). It has been noted that cDNA-based methods through reverse transcription or amplification are vulnerable to bias [58]. These concerns can be aggravated by the concurrent use of old-fashioned short-read sequencing technologies, which are known to exhibit GC bias. These technologies rely on available antibodies or known enzymes, such as ligases, which indicate a preference for specific sequences or nucleotides [56]. These techniques are often unable to identify the underlying RNA molecule that is modified, for example, base modifications that are known to have a role in controlling the activity and stability of RNA [58]. Thus, the large-scale application of these technologies is challenged by the requirement for complex protocols.
To address these limitations, a direct RNA sequencing platform provided by Oxford Nanopore Technologies (ONT) is proposed as a substitute technology to determine sites of alteration in the native RNA molecule [58]. Direct RNA nanopore sequencing has been used to analyze m6A in Arabidopsis [59], yeast [58,60], RNA virus genomes [61], and human cells [62,63,64,65,66]. In particular, Workman et al. focused on the m6A methyltransferase-binding motif, a short recurring pattern in its architecture with presumed biological function [64]. An ionic current change was demonstrated within the motif, attributable to m6A. This signal difference was subsequently validated utilizing data from the synthetic RNA. Fascinatingly, m6A-modified motifs in isoforms of the same gene were identified by using the ionic current difference. Whilst Workman et al. describe a relatively higher error rate of ONT when compared with cDNA sequencing, this technology directly detects post-transcriptional modifications, providing valuable insight into the mechanics of associated disease [64].
More recently, a computational method known as xPore has been developed by Pratanwanich et al. [62]. This technique enables differential RNA modifications from direct RNA sequencing data to be retained. This technology was tested on direct RNA sequencing data across six genetically distinct human cell lines covering liver cancer cells (HEPG2), colon cancer cells (HCT116), breast cancer cells (MCF7), lung adenocarcinoma cells (A549), and leukemia cells (K562), and HEK293T-KO cells. Between 800 and 2000 differentially modified sites were identified for all five cancer cell lines; most sites conformed to m6A DRACH motif. These findings indicated that RNA modifications could be observed across conditions, even when samples have a diverse genetic background. In the same study, the authors identified the dynamics of m6A by investigating the different tissues represented by the cell lines. Profoundly, the authors found that many m6A sites are preserved across multiple cell lines, with most positions being shared [62]. A significant advantage of xPore is that it is suited for detecting m6A with direct RNA-seq data from clinical cancer samples, even in the context of limited RNA (e.g., 2.5 micrograms) [62]. Since xPore does not rely on strict case-control comparisons, it offers essential flexibility in analyzing primary tissues and patient samples. False-positive results are primarily avoided by stringent filtering, which, in conjunction with the large data handling capacity of the method, opens new opportunities for larger-scale analysis of clinical patient data [62]. Furthermore, Jenjaroenpun et al. employed native RNA sequencing on lung cancer cell lines H460 and CEPH1463 [66,67,68]. They detected m6A in the RNA using epitranscriptomal landscape inferring from glitches on ONT signals (ELIGOS), in which caught error signatures are used to elucidate the properties of RNA. While it accurately determines RNA modifications’ specific locations, ELIGOS technology relies upon other sequencing methods to provide the proxy of detection. Therefore, traditional sequencing methods are required to understand the nature of the modification ELIGOS reveals [66]. Although still in its infancy, direct RNA sequencing has the propensity to detect RNA base modifications. Nanopore sequencing and its adjuncts such as ELIGOS will serve to update our current knowledge of the epitranscriptome in cancer.
8. Conclusions
HB represents the majority share of a rare disease cohort, with an incidence that appears to be increasing. Abnormal activation of a constellation of genes occurs to govern its inception and clinical course, many of which are linked to aberrant methylation of RNA. Although the investigation of HB’s genetic and epigenetic basis is in its relative infancy, there is a healthy precedent linking these genes to the trajectory of other solid organ malignancies. In keeping with its behavior in other biological systems, m6A appears to be a centerpiece in the complex genetic promotion of HB. Sequencing technologies and their adjuncts serve to elucidate the location and nature of m6A modifications within RNA and reveal the scale of their role across human tissues and diseases. The future will undoubtedly shed further light on the consequences of this modification for the patient journey. In this way, predictive and management decisions about the individual patient will rightly remain at the forefront of consideration.
Author Contributions
Conceptualization, C.M.S., methodology, C.M.S.; formal analysis, C.M.S., F.M.A.; investigation, F.M.A.; resources, C.M.S.; data curation, F.M.A.; writing—original draft preparation, F.M.A.; writing—review and editing, F.M.A., C.M.S., R.L., F.S.; visualization, F.S.; supervision, C.M.S.; project administration, C.M.S.; funding acquisition, C.M.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research has been funded by Consolato Sergi, Medical Professional Corporation and the generosity of the Children’s Hospital of Eastern Ontario, Ottawa, Ontario, and the Stollery Children’s Hospital Foundation and supporters of the Lois Hole Hospital for Women through the Women and Children’s Health Research Institute (WCHRI, Grant ID #: 2096), Natural Science Foundation of Hubei Province for Hubei University of Technology (100-Talent Grant for Recruitment Program of Foreign Experts Total Funding: Digital PCR and NGS-based diagnosis for infection and oncology, 2017–2022), Österreichische Krebshilfe Tyrol (Krebsgesellschaft Tirol, Austrian Tyrolean Cancer Research Institute, 2007 and 2009–“DMBTI and cholangiocellular carcinomas” and “Hsp70 and HSPBP1 in carcinomas of the pancreas”), Austrian Research Fund (Fonds zur Förderung der wissenschaftlichen Forschung, FWF, Grant ID L313-B13), Canadian Foundation for Women’s Health (“Early Fetal Heart-RES0000928”), Cancer Research Society (von Willebrand factor gene expression in cancer cells), Canadian Institutes of Health Research (Omega-3 Fatty Acids for Treatment of Intestinal Failure Associated Liver Disease: A Translational Research Study, 2011–2014, CIHR 232514), and the Saudi Cultural Bureau, Ottawa, Canada. The funders had no role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Acknowledgments
This work is dedicated to the memory of Julie Johnstone, who very sadly passed away suddenly on 8th February 2022. She worked tirelessly for the Pathological Society of Great Britain and Ireland for more than 18 years. She was a hardworking, dedicated, helpful and kind colleague, who is very sadly missed.
Conflicts of Interest
C. M. Sergi receives royalties from Springer and Nova publishers. All royalties go to pediatric charities.
References
- Marin, J.J.G.; Cives-Losada, C.; Asensio, M.; Lozano, E.; Briz, O.; Macias, R.I.R. Mechanisms of Anticancer Drug Resistance in Hepatoblastoma. Cancers 2019, 11, 407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morgan Auld, F.; Sergi, C.M. Surgical Pathology Diagnostic Pitfalls of Hepatoblastoma. Int. J. Surg. Pathol. 2022, 10668969211070178. [Google Scholar] [CrossRef] [PubMed]
- Lachance, E.; Mandziuk, J.; Sergi, C.M.; Bateman, J.; Low, G. Radiologic-Pathologic Correlation of Liver Tumors. In Liver Cancer; Sergi, C.M., Ed.; Exon Publications: Brisbane, QLD, Australia, 2021. [Google Scholar]
- Hager, J.; Sergi, C.M. Hepatoblastoma. In Liver Cancer; Sergi, C.M., Ed.; Exon Publications: Brisbane, QLD, Australia, 2021. [Google Scholar]
- Sergi, C.; Adam, S.; Kahl, P.; Otto, H.F. The remodeling of the primitive human biliary system. Early Hum. Dev. 2000, 58, 167–178. [Google Scholar] [CrossRef]
- Ranganathan, S.; Lopez-Terrada, D.; Alaggio, R. Hepatoblastoma and Pediatric Hepatocellular Carcinoma: An Update. Pediatr. Dev. Pathol. 2020, 23, 79–95. [Google Scholar] [CrossRef]
- Zhuo, Z.J.; Hua, R.X.; Chen, Z.; Zhu, J.; Wang, M.; Yang, Z.; Zhang, J.; Li, Y.; Li, L.; Li, S.; et al. WTAP Gene Variants Confer Hepatoblastoma Susceptibility: A Seven-Center Case-Control Study. Mol. Ther. Oncolytics 2020, 18, 118–125. [Google Scholar] [CrossRef]
- Jiang, X.; Liu, B.; Nie, Z.; Duan, L.; Xiong, Q.; Jin, Z.; Yang, C.; Chen, Y. The role of m6A modification in the biological functions and diseases. Signal. Transduct. Target Ther. 2021, 6, 74. [Google Scholar] [CrossRef]
- He, S.; Wang, H.; Liu, R.; He, M.; Che, T.; Jin, L.; Deng, L.; Tian, S.; Li, Y.; Lu, H.; et al. mRNA N6-methyladenosine methylation of postnatal liver development in pig. PLoS ONE 2017, 12, e0173421. [Google Scholar] [CrossRef]
- Sun, T.; Wu, R.; Ming, L. The role of m6A RNA methylation in cancer. Biomed. Pharmacother. 2019, 112, 108613. [Google Scholar] [CrossRef]
- Zhao, Y.; Peng, H. The Role of N(6)-Methyladenosine (m(6)A) Methylation Modifications in Hematological Malignancies. Cancers 2022, 14, 332. [Google Scholar] [CrossRef]
- Liu, L.; Wang, J.; Sun, G.; Wu, Q.; Ma, J.; Zhang, X.; Huang, N.; Bian, Z.; Gu, S.; Xu, M.; et al. m(6)A mRNA methylation regulates CTNNB1 to promote the proliferation of hepatoblastoma. Mol. Cancer 2019, 18, 188. [Google Scholar] [CrossRef] [Green Version]
- Cui, X.; Wang, Z.; Li, J.; Zhu, J.; Ren, Z.; Zhang, D.; Zhao, W.; Fan, Y.; Zhang, D.; Sun, R. Cross talk between RNA N6-methyladenosine methyltransferase-like 3 and miR-186 regulates hepatoblastoma progression through Wnt/beta-catenin signalling pathway. Cell Prolif. 2020, 53, e12768. [Google Scholar] [CrossRef] [Green Version]
- Li, G.; Deng, L.; Huang, N.; Cui, Z.; Wu, Q.; Ma, J.; Pan, Q.; Sun, F. m(6)A mRNA Methylation Regulates LKB1 to Promote Autophagy of Hepatoblastoma Cells through Upregulated Phosphorylation of AMPK. Genes 2021, 12, 1747. [Google Scholar] [CrossRef]
- Yang, C.; Hu, Y.; Zhou, B.; Bao, Y.; Li, Z.; Gong, C.; Yang, H.; Wang, S.; Xiao, Y. The role of m(6)A modification in physiology and disease. Cell Death Dis. 2020, 11, 960. [Google Scholar] [CrossRef]
- Huang, H.; Weng, H.; Chen, J. m(6)A Modification in Coding and Non-coding RNAs: Roles and Therapeutic Implications in Cancer. Cancer Cell 2020, 37, 270–288. [Google Scholar] [CrossRef]
- Zaccara, S.; Ries, R.J.; Jaffrey, S.R. Reading, writing and erasing mRNA methylation. Nat. Rev. Mol. Cell Biol. 2019, 20, 608–624. [Google Scholar] [CrossRef]
- Liu, S.; Zhuo, L.; Wang, J.; Zhang, Q.; Li, Q.; Li, G.; Yan, L.; Jin, T.; Pan, T.; Sui, X.; et al. METTL3 plays multiple functions in biological processes. Am. J. Cancer Res. 2020, 10, 1631–1646. [Google Scholar]
- Lopez-Terrada, D.; Alaggio, R.; de Davila, M.T.; Czauderna, P.; Hiyama, E.; Katzenstein, H.; Leuschner, I.; Malogolowkin, M.; Meyers, R.; Ranganathan, S.; et al. Towards an international pediatric liver tumor consensus classification: Proceedings of the Los Angeles COG liver tumors symposium. Mod. Pathol. 2014, 27, 472–491. [Google Scholar] [CrossRef] [Green Version]
- Ignatova, V.V.; Stolz, P.; Kaiser, S.; Gustafsson, T.H.; Lastres, P.R.; Sanz-Moreno, A.; Cho, Y.L.; Amarie, O.V.; Aguilar-Pimentel, A.; Klein-Rodewald, T.; et al. The rRNA m(6)A methyltransferase METTL5 is involved in pluripotency and developmental programs. Genes Dev. 2020, 34, 715–729. [Google Scholar] [CrossRef]
- Chen, X.Y.; Zhang, J.; Zhu, J.S. The role of m(6)A RNA methylation in human cancer. Mol. Cancer 2019, 18, 103. [Google Scholar] [CrossRef] [Green Version]
- Esteve-Puig, R.; Bueno-Costa, A.; Esteller, M. Writers, readers and erasers of RNA modifications in cancer. Cancer Lett. 2020, 474, 127–137. [Google Scholar] [CrossRef]
- Mauer, J.; Luo, X.; Blanjoie, A.; Jiao, X.; Grozhik, A.V.; Patil, D.P.; Linder, B.; Pickering, B.F.; Vasseur, J.J.; Chen, Q.; et al. Reversible methylation of m(6)Am in the 5’ cap controls mRNA stability. Nature 2017, 541, 371–375. [Google Scholar] [CrossRef] [Green Version]
- Dominissini, D.; Moshitch-Moshkovitz, S.; Schwartz, S.; Salmon-Divon, M.; Ungar, L.; Osenberg, S.; Cesarkas, K.; Jacob-Hirsch, J.; Amariglio, N.; Kupiec, M.; et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature 2012, 485, 201–206. [Google Scholar] [CrossRef]
- Moshitch-Moshkovitz, S.; Dominissini, D.; Rechavi, G. The epitranscriptome toolbox. Cell 2022, 185, 764–776. [Google Scholar] [CrossRef]
- Kortel, N.; Ruckle, C.; Zhou, Y.; Busch, A.; Hoch-Kraft, P.; Sutandy, F.X.R.; Haase, J.; Pradhan, M.; Musheev, M.; Ostareck, D.; et al. Deep and accurate detection of m6A RNA modifications using miCLIP2 and m6Aboost machine learning. Nucleic Acids Res. 2021, 49, e92. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, J.; Li, X.; Xiong, X.; Wang, J.; Zhou, Z.; Zhu, X.; Gu, Y.; Dominissini, D.; He, L.; et al. N(1)-methyladenosine methylation in tRNA drives liver tumourigenesis by regulating cholesterol metabolism. Nat. Commun. 2021, 12, 6314. [Google Scholar] [CrossRef]
- Meyer, K.D.; Saletore, Y.; Zumbo, P.; Elemento, O.; Mason, C.E.; Jaffrey, S.R. Comprehensive analysis of mRNA methylation reveals enrichment in 3’ UTRs and near stop codons. Cell 2012, 149, 1635–1646. [Google Scholar] [CrossRef] [Green Version]
- Shen, G.; Shen, H.; Zhang, J.; Yan, Q.; Liu, H. DNA methylation in Hepatoblastoma-a literature review. Ital. J. Pediatr. 2020, 46, 113. [Google Scholar] [CrossRef]
- Zhang, W.; Meyfeldt, J.; Wang, H.; Kulkarni, S.; Lu, J.; Mandel, J.A.; Marburger, B.; Liu, Y.; Gorka, J.E.; Ranganathan, S.; et al. beta-Catenin mutations as determinants of hepatoblastoma phenotypes in mice. J. Biol. Chem. 2019, 294, 17524–17542. [Google Scholar] [CrossRef]
- Sha, Y.L.; Liu, S.; Yan, W.W.; Dong, B. Wnt/beta-catenin signaling as a useful therapeutic target in hepatoblastoma. Biosci. Rep. 2019, 39, BSR20192466. [Google Scholar] [CrossRef]
- Chen, H.; Duan, F.; Wang, M.; Zhu, J.; Zhang, J.; Cheng, J.; Li, L.; Li, S.; Li, Y.; Yang, Z.; et al. Polymorphisms in METTL3 gene and hepatoblastoma risk in Chinese children: A seven-center case-control study. Gene 2021, 800, 145834. [Google Scholar] [CrossRef]
- Czauderna, P.; Garnier, H. Hepatoblastoma: Current understanding, recent advances, and controversies. F1000Res 2018, 7, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Czauderna, C.; Poplawski, A.; O’Rourke, C.J.; Castven, D.; Perez-Aguilar, B.; Becker, D.; Heilmann-Heimbach, S.; Odenthal, M.; Amer, W.; Schmiel, M.; et al. Epigenetic modifications precede molecular alterations and drive human hepatocarcinogenesis. JCI Insight 2021, 6, 146196. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, Y.; Li, L.; Zhu, J.; Yang, Z.; Zhang, J.; Li, S.; Xin, Y.; Xia, H.; He, J. YTHDC1 gene polymorphisms and hepatoblastoma susceptibility in Chinese children: A seven-center case-control study. J. Gene Med. 2020, 22, e3249. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Peng, C.; Chen, J.; Chen, D.; Yang, B.; He, B.; Hu, W.; Zhang, Y.; Liu, H.; Dai, L.; et al. WTAP facilitates progression of hepatocellular carcinoma via m6A-HuR-dependent epigenetic silencing of ETS1. Mol. Cancer 2019, 18, 127. [Google Scholar] [CrossRef] [Green Version]
- Spector, D.L.; Lamond, A.I. Nuclear speckles. Cold Spring Harb. Perspect. Biol. 2011, 3, 646. [Google Scholar] [CrossRef] [Green Version]
- Little, N.A.; Hastie, N.D.; Davies, R.C. Identification of WTAP, a novel Wilms’ tumour 1-associating protein. Hum. Mol. Genet. 2000, 9, 2231–2239. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Xie, H.; Ying, Y.; Chen, H.; Yan, H.; He, L.; Xu, M.; Xu, X.; Liang, Z.; Liu, B.; et al. YTHDF2 mediates the mRNA degradation of the tumor suppressors to induce AKT phosphorylation in N6-methyladenosine-dependent way in prostate cancer. Mol. Cancer 2020, 19, 152. [Google Scholar] [CrossRef]
- Sergi, C.M. Pathology of Childhood and Adolescence: An Illustrated Guide, 1st ed.; Springer: Berlin-Heidelberg, Germany, 2020; p. 1617. [Google Scholar]
- An, Y.; Duan, H. The role of m6A RNA methylation in cancer metabolism. Mol. Cancer 2022, 21, 14. [Google Scholar] [CrossRef]
- Qu, Y.; Dang, S.; Hou, P. Gene methylation in gastric cancer. Clin. Chim. Acta 2013, 424, 53–65. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhang, T.; Yin, Q.; Luo, H. Development and validation of genomic and epigenomic signatures associated with tumor immune microenvironment in hepatoblastoma. BMC Cancer 2021, 21, 1156. [Google Scholar] [CrossRef]
- Mazzoni, S.M.; Fearon, E.R. AXIN1 and AXIN2 variants in gastrointestinal cancers. Cancer Lett. 2014, 355, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Shen, F.; Huang, W.; Qin, S.; Huang, J.T.; Sergi, C.; Yuan, B.F.; Liu, S.M. Glucose Is Involved in the Dynamic Regulation of m6A in Patients With Type 2 Diabetes. J. Clin. Endocrinol. Metab. 2019, 104, 665–673. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Yang, F.; Gao, M.; Gong, R.; Jin, M.; Liu, T.; Sun, Y.; Fu, Y.; Huang, Q.; Zhang, W.; et al. miR-149-3p Regulates the Switch between Adipogenic and Osteogenic Differentiation of BMSCs by Targeting FTO. Mol. Ther. Nucleic Acids 2019, 17, 590–600. [Google Scholar] [CrossRef] [Green Version]
- Zhao, N.N.; Dong, G.P.; Wu, W.; Wang, J.L.; Ullah, R.; Fu, J.F. FTO gene polymorphisms and obesity risk in Chinese population: A meta-analysis. World J. Pediatr. 2019, 15, 382–389. [Google Scholar] [CrossRef]
- Shen, F.; Huang, W.; Huang, J.T.; Xiong, J.; Yang, Y.; Wu, K.; Jia, G.F.; Chen, J.; Feng, Y.Q.; Yuan, B.F.; et al. Decreased N(6)-methyladenosine in peripheral blood RNA from diabetic patients is associated with FTO expression rather than ALKBH5. J. Clin. Endocrinol. Metab. 2015, 100, E148–E154. [Google Scholar] [CrossRef] [Green Version]
- Nagae, G.; Yamamoto, S.; Fujita, M.; Fujita, T.; Nonaka, A.; Umeda, T.; Fukuda, S.; Tatsuno, K.; Maejima, K.; Hayashi, A.; et al. Genetic and epigenetic basis of hepatoblastoma diversity. Nat. Commun. 2021, 12, 5423. [Google Scholar] [CrossRef]
- Czauderna, P. Is it worth completely resecting hepatoblastoma at diagnosis? Lancet Oncol. 2019, 20, 614–615. [Google Scholar] [CrossRef]
- Aronson, D.C.; Weeda, V.B.; Maibach, R.; Czauderna, P.; Dall’Igna, P.; de Ville de Goyet, J.; Branchereau, S.; Perilongo, G.; Brock, P.; Zsiros, J.; et al. Microscopically positive resection margin after hepatoblastoma resection: What is the impact on prognosis? A Childhood Liver Tumours Strategy Group (SIOPEL) report. Eur. J. Cancer 2019, 106, 126–132. [Google Scholar] [CrossRef]
- Carrillo-Reixach, J.; Torrens, L.; Simon-Coma, M.; Royo, L.; Domingo-Sabat, M.; Abril-Fornaguera, J.; Akers, N.; Sala, M.; Ragull, S.; Arnal, M.; et al. Epigenetic footprint enables molecular risk stratification of hepatoblastoma with clinical implications. J. Hepatol. 2020, 73, 328–341. [Google Scholar] [CrossRef]
- Huff, S.; Tiwari, S.K.; Gonzalez, G.M.; Wang, Y.; Rana, T.M. m(6)A-RNA Demethylase FTO Inhibitors Impair Self-Renewal in Glioblastoma Stem Cells. ACS Chem. Biol. 2021, 16, 324–333. [Google Scholar] [CrossRef]
- Lan, N.; Lu, Y.; Zhang, Y.; Pu, S.; Xi, H.; Nie, X.; Liu, J.; Yuan, W. FTO—A Common Genetic Basis for Obesity and Cancer. Front. Genet. 2020, 11, 559138. [Google Scholar] [CrossRef]
- Kumar, S.; Nagpal, R.; Kumar, A.; Ashraf, M.U.; Bae, Y.S. Immunotherapeutic Potential of m6A-Modifiers and MicroRNAs in Controlling Acute Myeloid Leukaemia. Biomedicines 2021, 9, 9060690. [Google Scholar] [CrossRef]
- Helm, M.; Motorin, Y. Detecting RNA modifications in the epitranscriptome: Predict and validate. Nat. Rev. Genet. 2017, 18, 275–291. [Google Scholar] [CrossRef]
- Li, X.; Xiong, X.; Yi, C. Epitranscriptome sequencing technologies: Decoding RNA modifications. Nat. Methods 2016, 14, 23–31. [Google Scholar] [CrossRef]
- Garalde, D.R.; Snell, E.A.; Jachimowicz, D.; Sipos, B.; Lloyd, J.H.; Bruce, M.; Pantic, N.; Admassu, T.; James, P.; Warland, A.; et al. Highly parallel direct RNA sequencing on an array of nanopores. Nat. Methods 2018, 15, 201–206. [Google Scholar] [CrossRef]
- Parker, M.T.; Knop, K.; Sherwood, A.V.; Schurch, N.J.; Mackinnon, K.; Gould, P.D.; Hall, A.J.; Barton, G.J.; Simpson, G.G. Nanopore direct RNA sequencing maps the complexity of Arabidopsis mRNA processing and m(6)A modification. Elife 2020, 9, 49658. [Google Scholar] [CrossRef]
- Xu, L.; Seki, M. Recent advances in the detection of base modifications using the Nanopore sequencer. J. Hum. Genet. 2020, 65, 25–33. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.; Lee, J.Y.; Yang, J.S.; Kim, J.W.; Kim, V.N.; Chang, H. The Architecture of SARS-CoV-2 Transcriptome. Cell 2020, 181, 914–921.e910. [Google Scholar] [CrossRef]
- Pratanwanich, P.N.; Yao, F.; Chen, Y.; Koh, C.W.Q.; Wan, Y.K.; Hendra, C.; Poon, P.; Goh, Y.T.; Yap, P.M.L.; Chooi, J.Y.; et al. Identification of differential RNA modifications from nanopore direct RNA sequencing with xPore. Nat. Biotechnol. 2021, 39, 1394–1402. [Google Scholar] [CrossRef]
- Leger, A.; Amaral, P.P.; Pandolfini, L.; Capitanchik, C.; Capraro, F.; Miano, V.; Migliori, V.; Toolan-Kerr, P.; Sideri, T.; Enright, A.J.; et al. RNA modifications detection by comparative Nanopore direct RNA sequencing. Nat. Commun. 2021, 12, 7198. [Google Scholar] [CrossRef]
- Workman, R.E.; Tang, A.D.; Tang, P.S.; Jain, M.; Tyson, J.R.; Razaghi, R.; Zuzarte, P.C.; Gilpatrick, T.; Payne, A.; Quick, J.; et al. Nanopore native RNA sequencing of a human poly(A) transcriptome. Nat. Methods 2019, 16, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, D.A.; Sathe, S.; Einstein, J.M.; Yeo, G.W. Direct RNA sequencing enables m(6)A detection in endogenous transcript isoforms at base-specific resolution. RNA 2020, 26, 19–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenjaroenpun, P.; Wongsurawat, T.; Wadley, T.D.; Wassenaar, T.M.; Liu, J.; Dai, Q.; Wanchai, V.; Akel, N.S.; Jamshidi-Parsian, A.; Franco, A.T.; et al. Decoding the epitranscriptional landscape from native RNA sequences. Nucleic Acids Res. 2021, 49, e7. [Google Scholar] [CrossRef] [PubMed]
- Jenjaroenpun, P.; Wanchai, V.; Ono-Moore, K.D.; Laudadio, J.; James, L.P.; Adams, S.H.; Prior, F.; Nookaew, I.; Ussery, D.W.; Wongsurawat, T. Two SARS-CoV-2 Genome Sequences of Isolates from Rural U.S. Patients Harboring the D614G Mutation, Obtained Using Nanopore Sequencing. Microbiol. Resour. Announc. 2020, 10, e01109–e01120. [Google Scholar] [CrossRef]
- Jenjaroenpun, P.; Wongsurawat, T.; Udaondo, Z.; Anderson, C.; Lopez, J.; Mohan, M.; Tytarenko, R.; Walker, B.; Nookaew, I.; Ussery, D.; et al. Complete Genome Sequences of Four Isolates of Vancomycin-Resistant Enterococcus faecium with the vanA Gene and Two Daptomycin Resistance Mutations, Obtained from Two Inpatients with Prolonged Bacteremia. Microbiol. Resour. Announc. 2020, 9, e01380–e01419. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).