Fucoxanthin Ameliorates Oxidative Stress and Airway Inflammation in Tracheal Epithelial Cells and Asthmatic Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
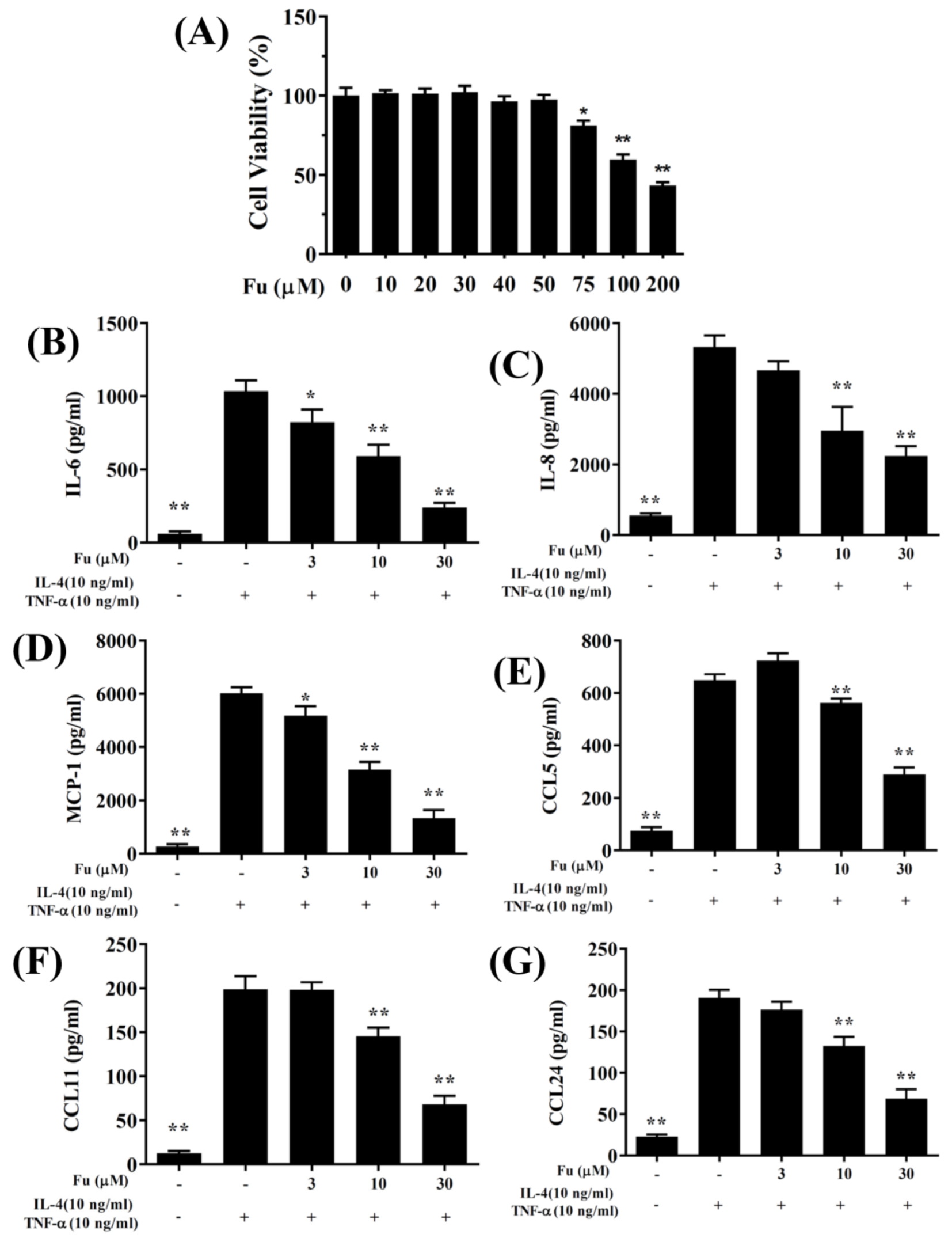
2.2. Cell Viability Assay
2.3. BEAS-2B Cell Culture and Fucoxanthin Treatment
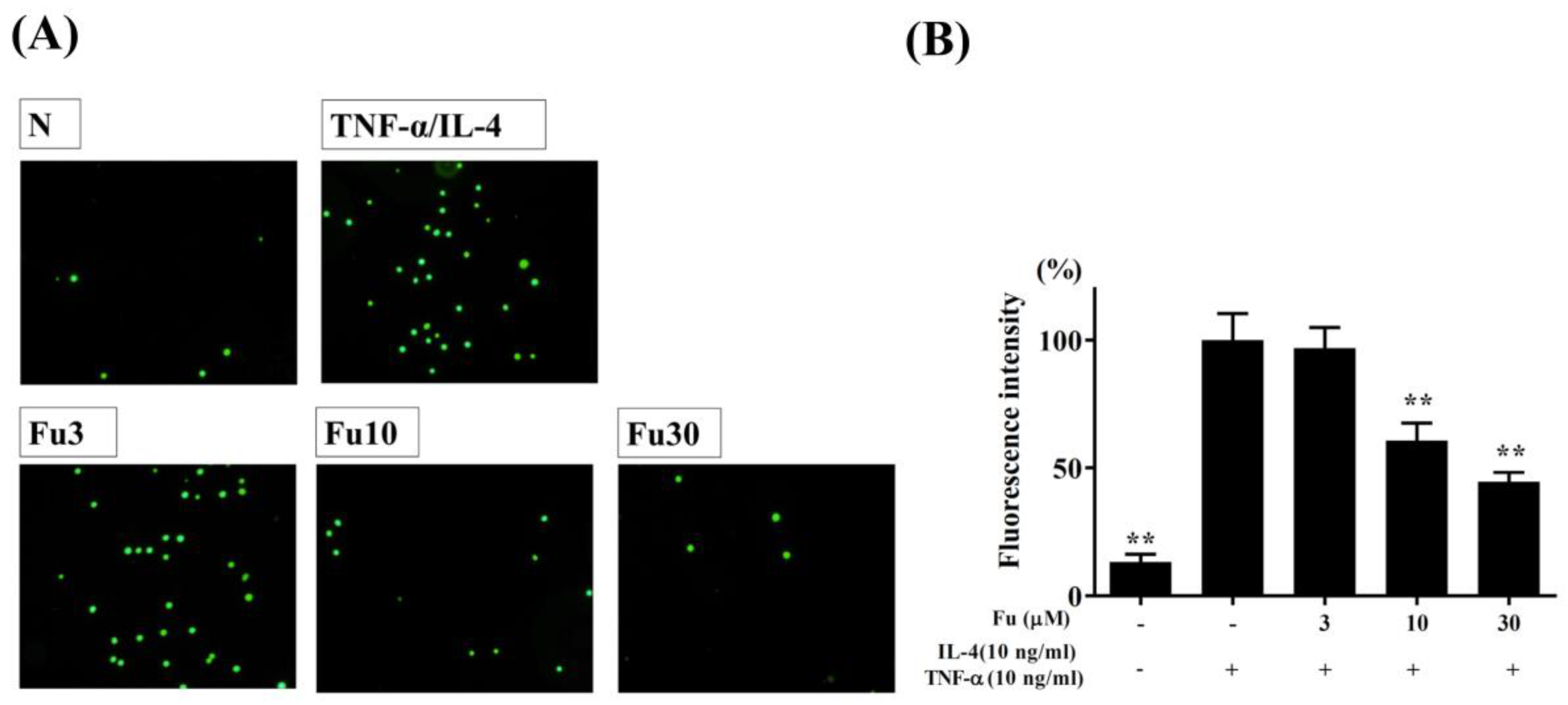
2.4. Cell-Cell Adhesion Assay
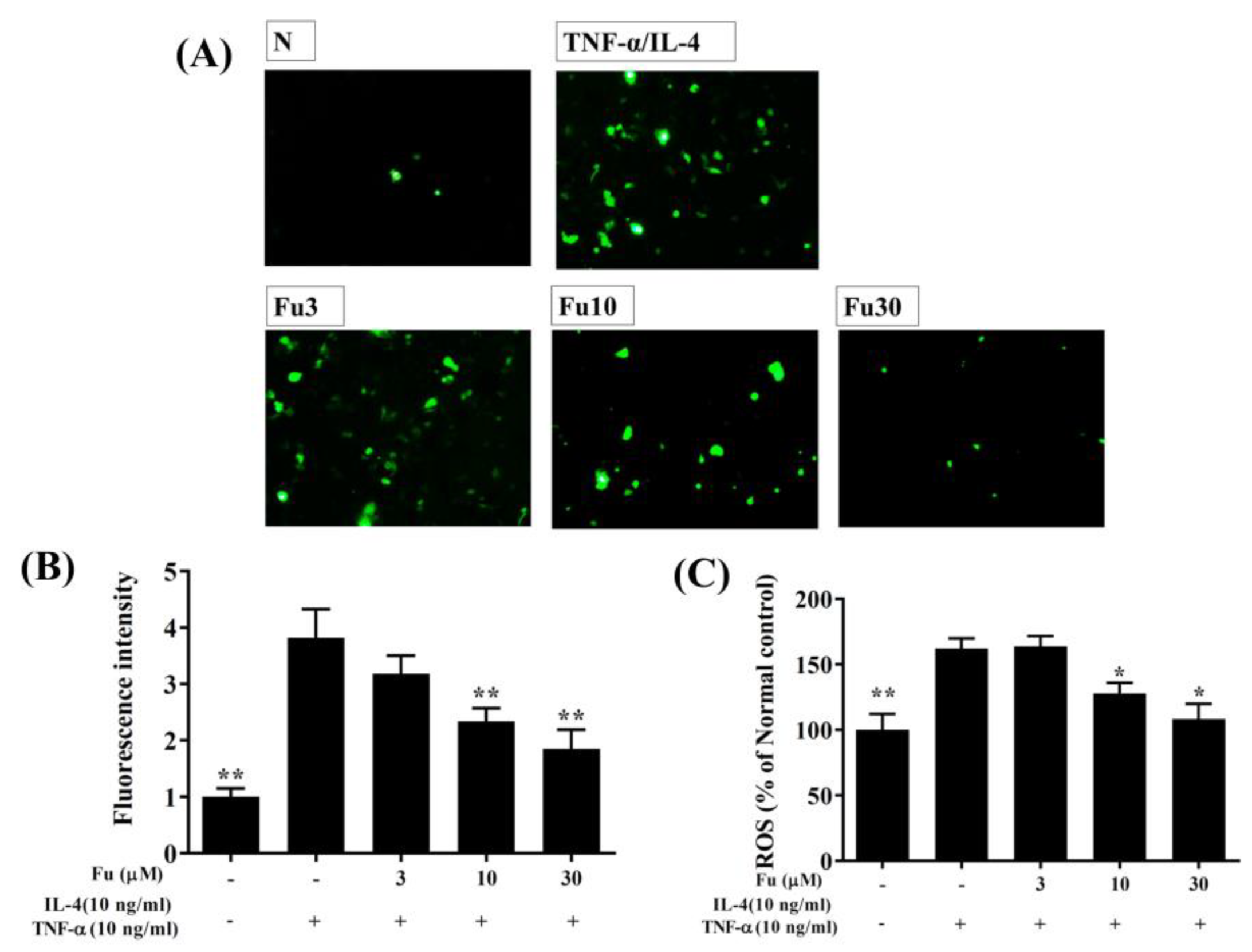
2.5. ROS Production in BEAS-2B Cells
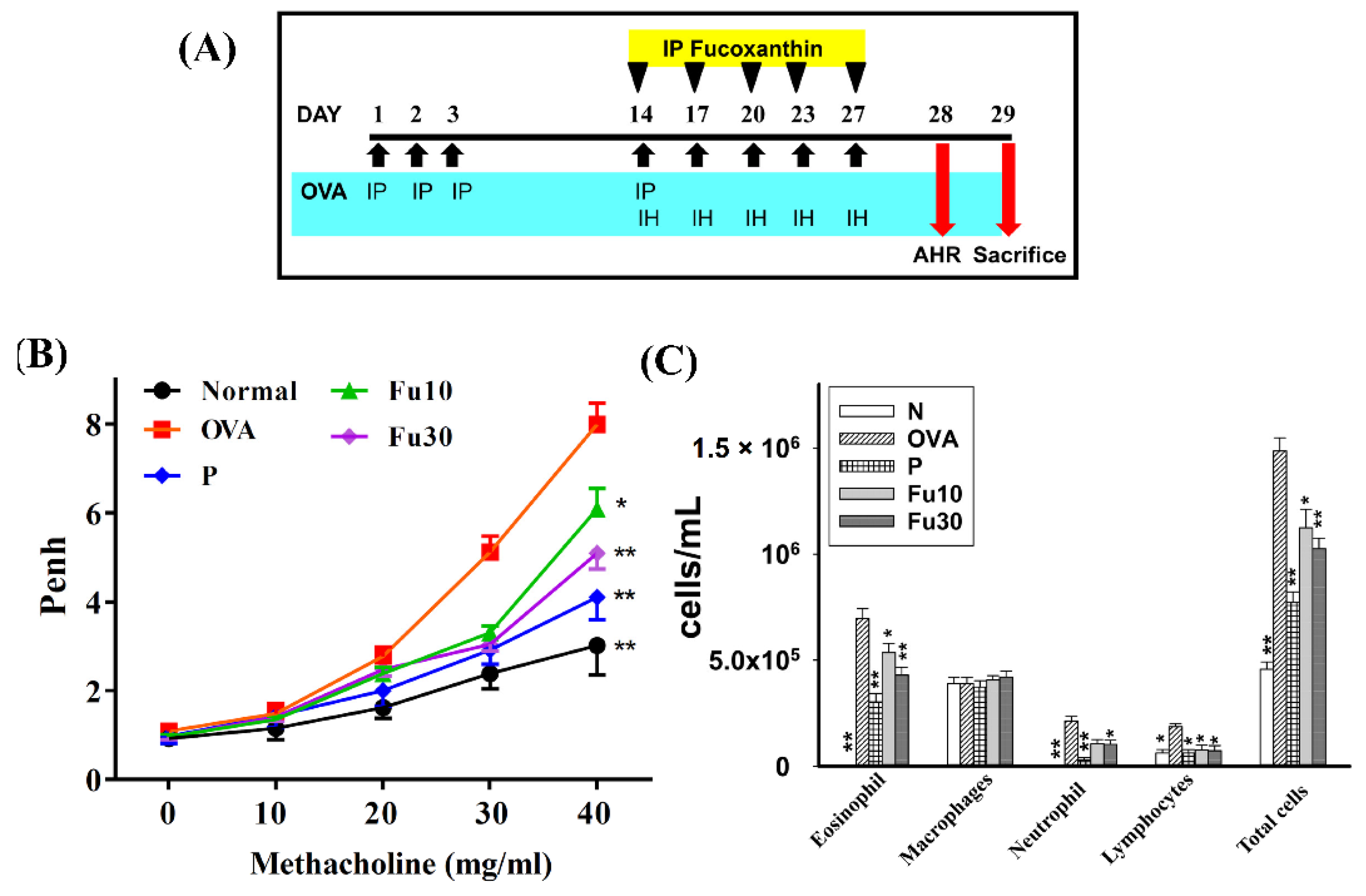
2.6. Animal Experiments
2.7. Mouse Sensitization and Administration of Fucoxanthin
2.8. Airway Hyperresponsiveness
2.9. Histological Analysis of Lung Tissue
2.10. Serum Collection and Splenocyte Culture
2.11. Bronchoalveolar Lavage Fluid and Cell Counting
2.12. RNA Isolation and Quantitative Real-Time PCR
2.13. ELISA
2.14. Immunohistochemical Staining
2.15. MDA Activity
2.16. Glutathione (GSH), Superoxide Dismutase (SOD), and Catalase (CAT) Assay
2.17. Statistical Analysis
3. Results
3.1. Fucoxanthin Reduced Inflammatory Mediators and Cell Adhesion in BEAS-2B Cells
3.2. Effect of Fucoxanthin on ROS Production
3.3. Fucoxanthin Attenuated AHR in Asthmatic Mice
3.4. Fucoxanthin Inhibited Eosinophils in the BALF
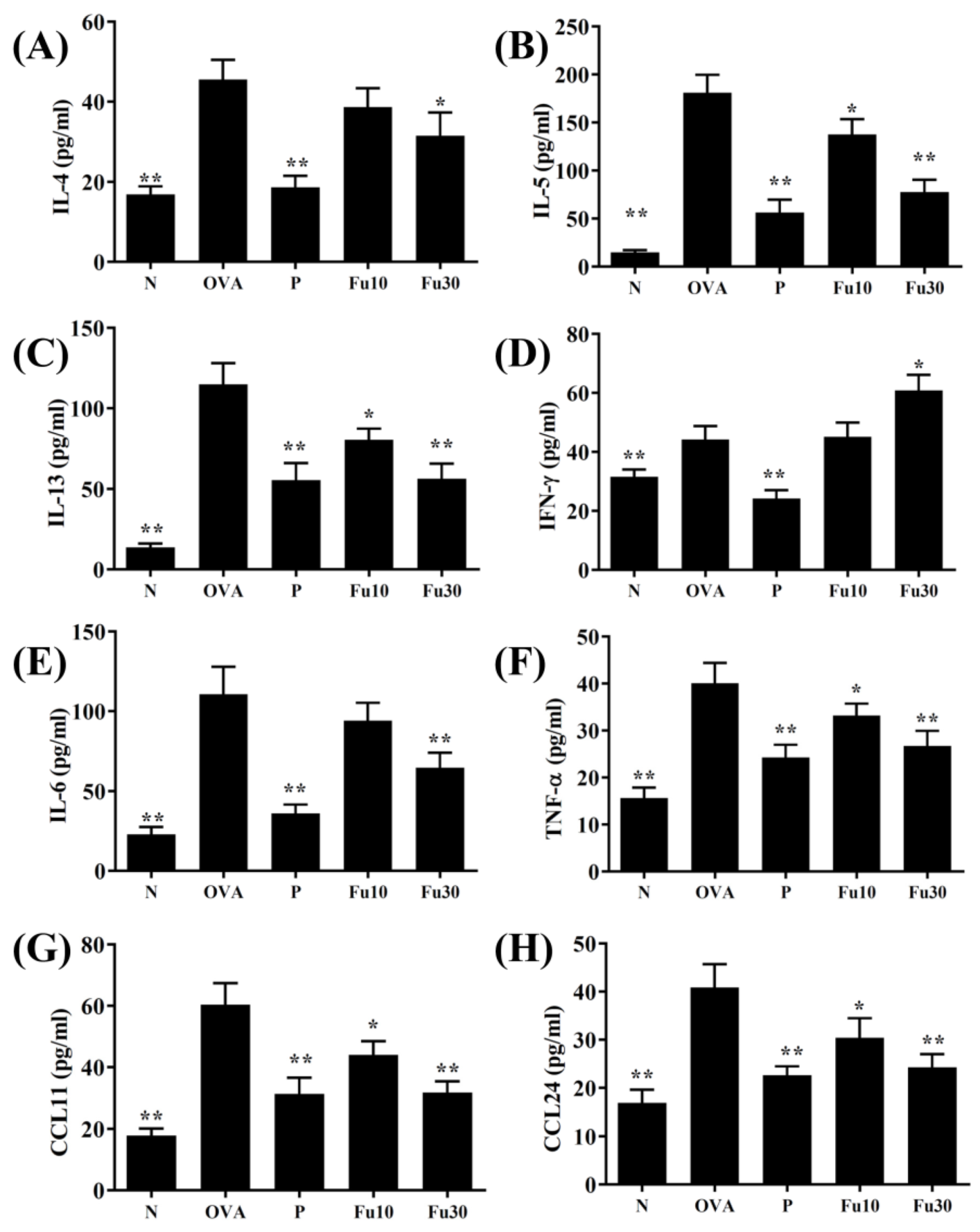
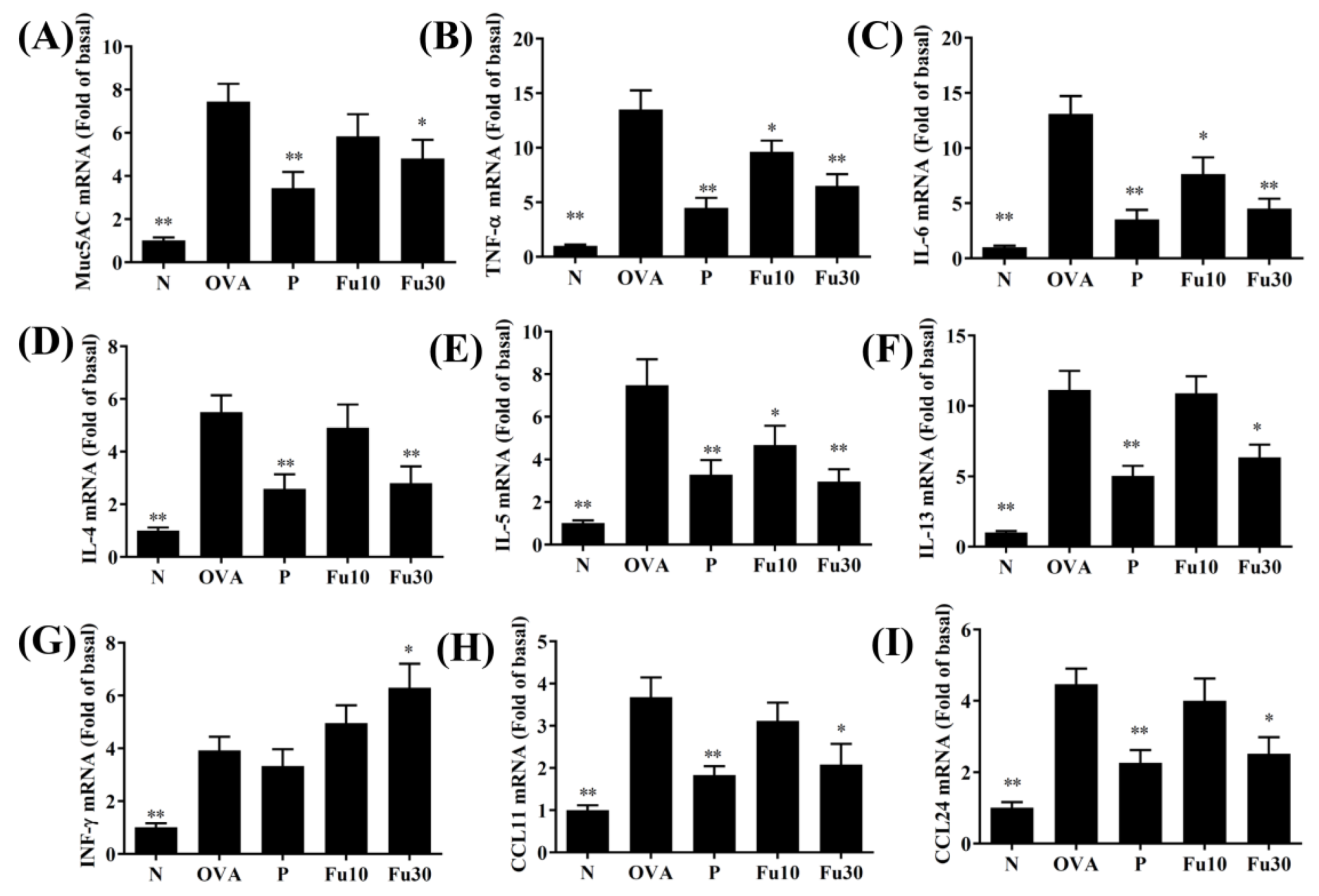
3.5. Fucoxanthin Modulated Chemokine and Cytokine Expression in the BALF and Lung Tissue
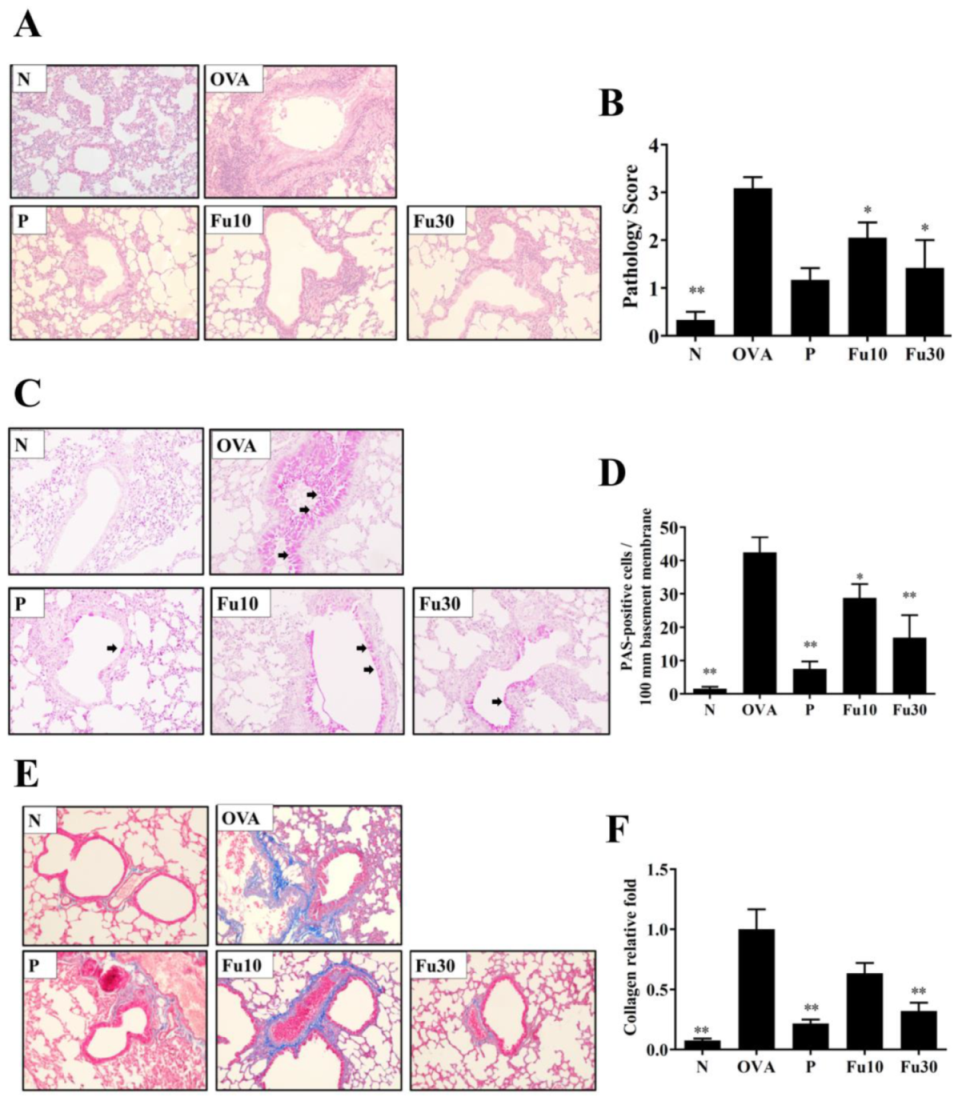
3.6. Fucoxanthin Reduced Goblet Cell Hyperplasia and Eosinophil Infiltration in Lung Tissue
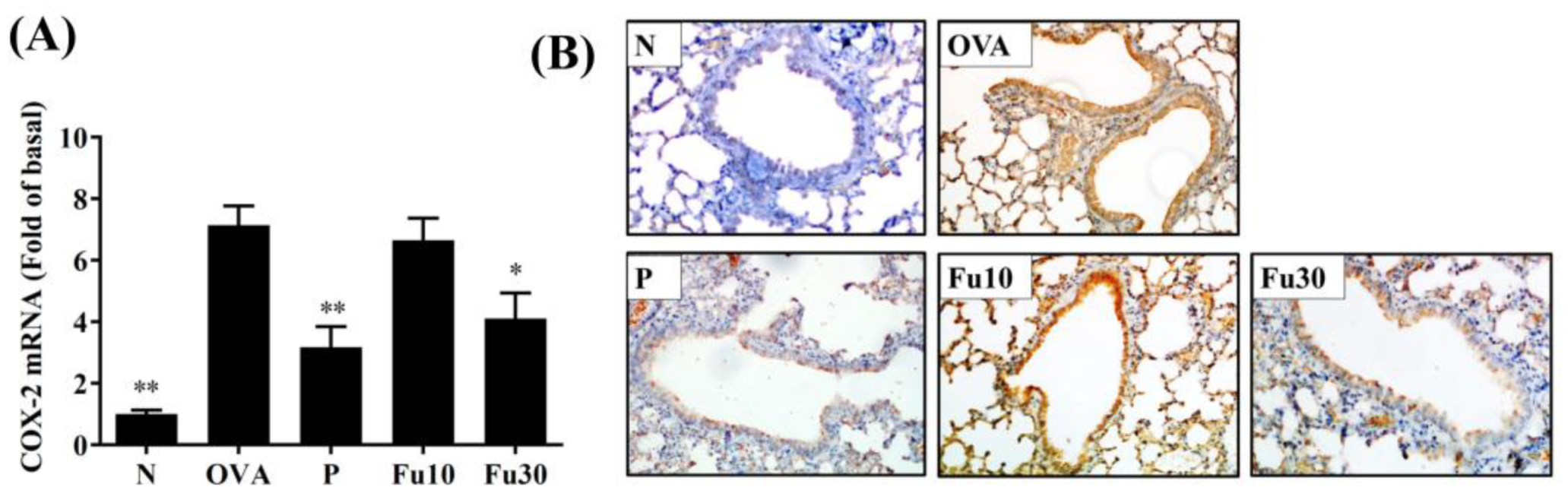
3.7. Fucoxanthin Reduced Collagen and COX-2 Expression in the Lungs
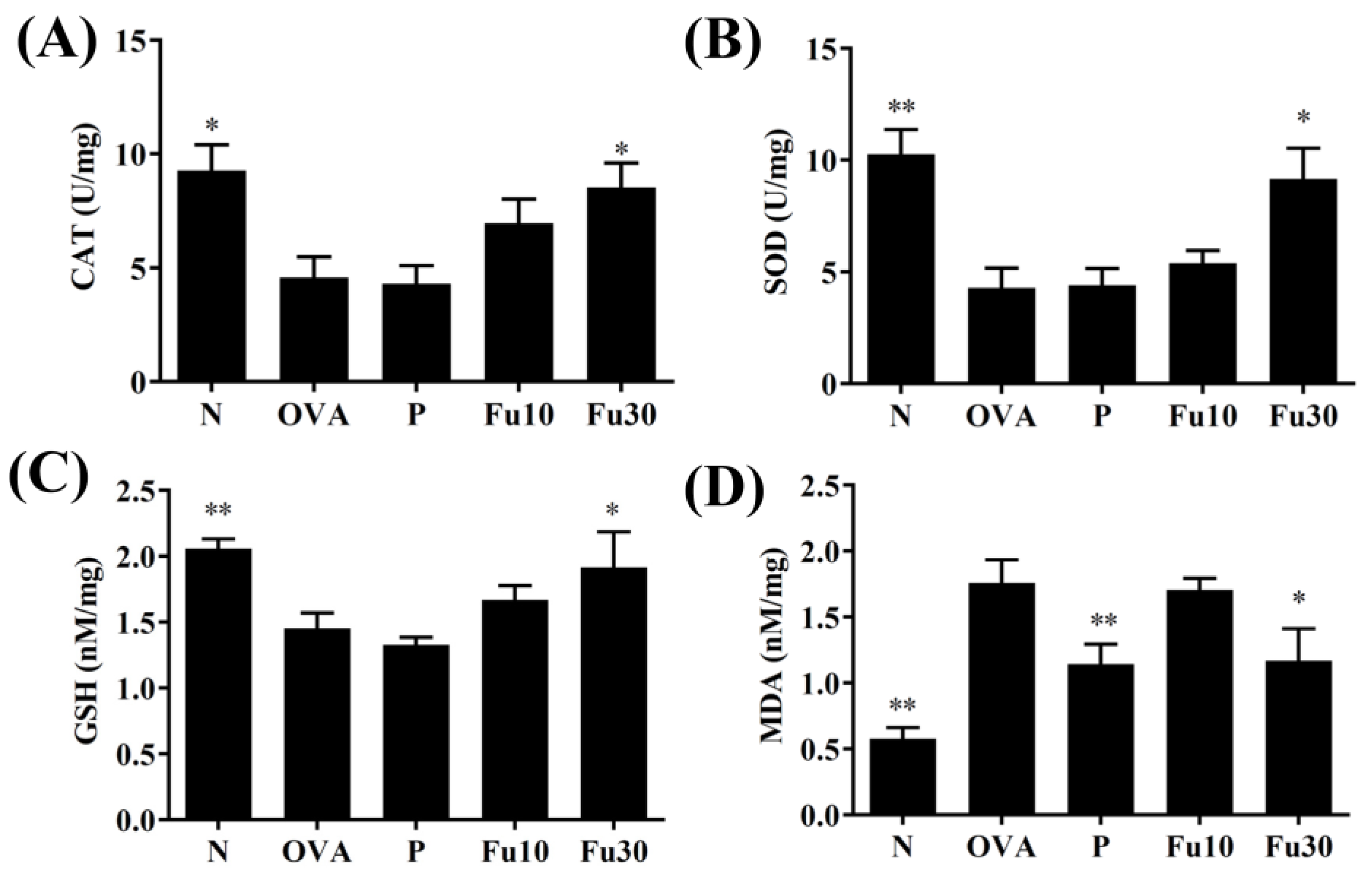
3.8. Fucoxanthin Modulated Antioxidant Enzyme Levels in the Lungs
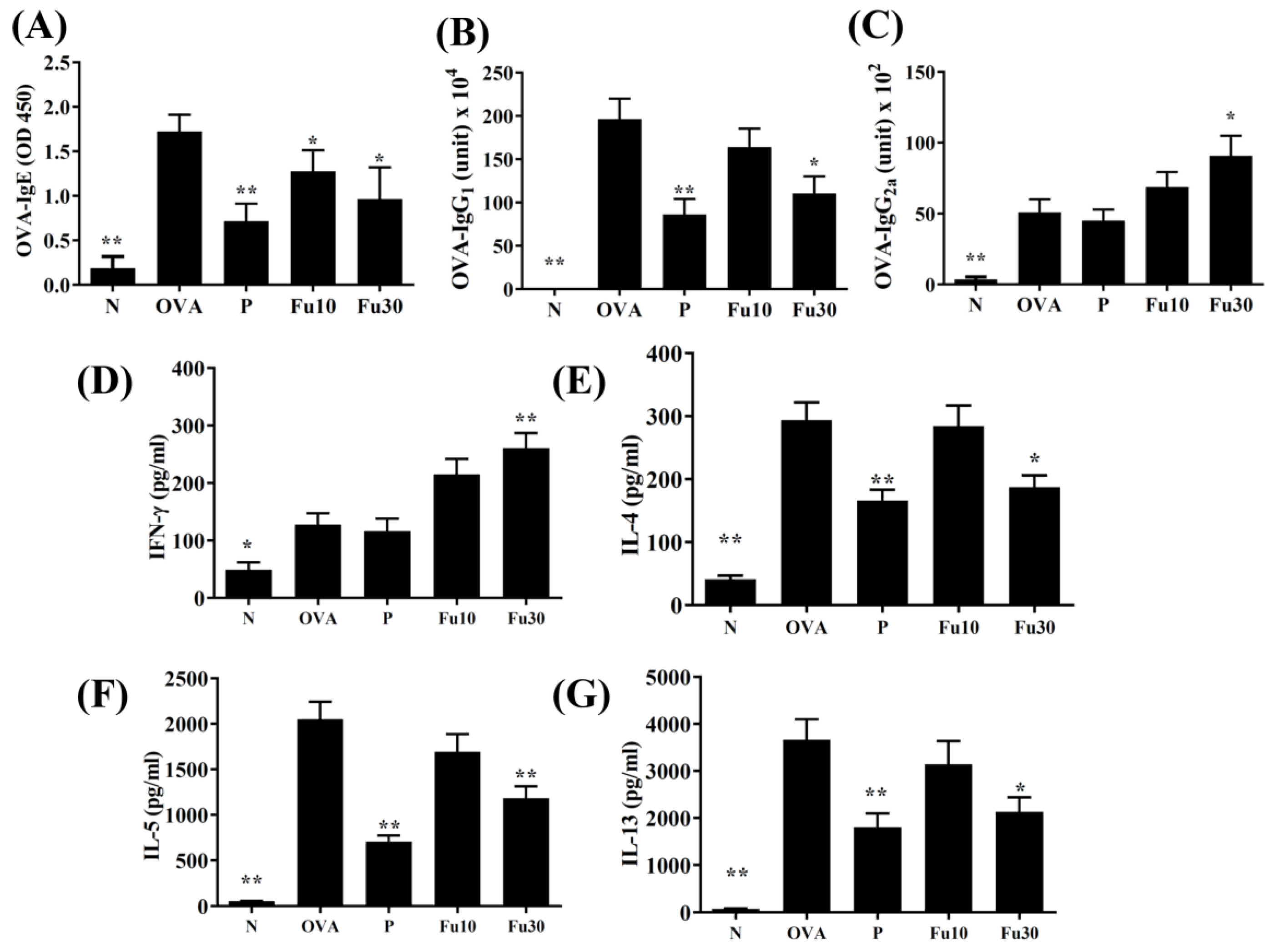
3.9. Fucoxanthin Modulated Serum Antibody and Splenocyte Cytokine Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nanda, A.; Wasan, A.N. Asthma in adults. Med. Clin. N. Am. 2020, 104, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Carpaij, O.A.; Burgess, J.K.; Kerstjens, H.A.M.; Nawijn, M.C.; van den Berge, M. A review on the pathophysiology of asthma remission. Pharmacol. Ther. 2019, 201, 8–24. [Google Scholar] [CrossRef] [PubMed]
- Song, W.J.; Lee, J.H.; Kang, Y.; Joung, W.J.; Chung, K.F. Future risks in patients with severe asthma. Allergy Asthma Immunol. Res. 2019, 11, 763–778. [Google Scholar] [CrossRef] [PubMed]
- Goleva, E.; Berdyshev, E.; Leung, D.Y. Epithelial barrier repair and prevention of allergy. J. Clin. Investig. 2019, 129, 1463–1474. [Google Scholar] [CrossRef]
- Lambrecht, B.N.; Hammad, H.; Fahy, J.V. The cytokines of asthma. Immunity 2019, 50, 975–991. [Google Scholar] [CrossRef]
- Coleman, J.M.; Naik, C.; Holguin, F.; Ray, A.; Ray, P.; Trudeau, J.B.; Wenzel, S.E. Epithelial eotaxin-2 and eotaxin-3 expression: Relation to asthma severity, luminal eosinophilia and age at onset. Thorax 2012, 67, 1061–1066. [Google Scholar] [CrossRef]
- Symowski, C.; Voehringer, D. Th2 cell-derived IL-4/IL-13 promote ILC2 accumulation in the lung by ILC2-intrinsic STAT6 signaling in mice. Eur. J. Immunol. 2019, 49, 1421–1432. [Google Scholar] [CrossRef]
- Bonser, L.R.; Erle, D.J. Airway mucus and asthma: The role of MUC5AC and MUC5B. J. Clin. Med. 2017, 6, 112. [Google Scholar] [CrossRef]
- Roan, F.; Obata-Ninomiya, K.; Ziegler, S.F. Epithelial cell-derived cytokines: More than just signaling the alarm. J. Clin. Investig. 2019, 129, 1441–1451. [Google Scholar] [CrossRef]
- Wadhwa, R.; Dua, K.; Adcock, I.M.; Horvat, J.C.; Kim, R.Y.; Hansbro, P.M. Cellular mechanisms underlying steroid-resistant asthma. Eur. Respir. Rev. 2019, 28, 190096. [Google Scholar] [CrossRef]
- Rodriguez, J.M.; Monsalves-Alvarez, M.; Henriquez, S.; Llanos, M.N.; Troncoso, R. Glucocorticoid resistance in chronic diseases. Steroids 2016, 115, 182–192. [Google Scholar] [CrossRef] [PubMed]
- Bielory, L. Complementary and alternative interventions in asthma, allergy, and immunology. Ann. Allergy Asthma Immunol. 2004, 93, S45–S54. [Google Scholar] [CrossRef]
- Fouladi, S.; Masjedi, M.; Ganjalikhani Hakemi, M.; Eskandari, N. The review of in vitro and in vivo studies over the glycyrrhizic acid as natural remedy option for treatment of allergic asthma. Iran. J. Allergy Asthma Immunol. 2019, 18, 1–11. [Google Scholar] [CrossRef]
- Li, J.; Zhang, F. The immunoregulatory effects of traditional chinese medicine on treatment of asthma or asthmatic inflammation. Am. J. Chin. Med. 2015, 43, 1059–1081. [Google Scholar] [CrossRef] [PubMed]
- Bae, M.; Kim, M.B.; Park, Y.K.; Lee, J.Y. Health benefits of fucoxanthin in the prevention of chronic diseases. Biochim. Biophys. Acta. Mol. Cell Biol. Lipids 2020, 10, 158618. [Google Scholar] [CrossRef]
- Muradian, K.; Vaiserman, A.; Min, K.J.; Fraifeld, V.E. Fucoxanthin and lipid metabolism: A minireview. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 891–897. [Google Scholar] [CrossRef] [PubMed]
- Gammone, M.A.; D’Orazio, N. Anti-obesity activity of the marine carotenoid fucoxanthin. Mar. Drugs 2015, 13, 2196–2214. [Google Scholar] [CrossRef]
- Satomi, Y. Antitumor and cancer-preventative function of fucoxanthin: A marine carotenoid. Anticancer Res. 2017, 37, 1557–1562. [Google Scholar] [CrossRef]
- Ma, S.Y.; Park, W.S.; Lee, D.S.; Choi, G.; Yim, M.J.; Lee, J.M.; Jung, W.K.; Park, S.G.; Seo, S.K.; Park, S.J.; et al. Fucoxanthin inhibits profibrotic protein expression in vitro and attenuates bleomycin-induced lung fibrosis in vivo. Eur. J. Pharmacol. 2017, 811, 199–207. [Google Scholar] [CrossRef]
- Robertson, R.C.; Guihéneuf, F.; Bahar, B.; Schmid, M.; Stengel, D.B.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. The anti-inflammatory effect of algae-derived lipid extracts on lipopolysaccharide (LPS)-stimulated human THP-1 macrophages. Mar. Drugs 2015, 13, 5402–5424. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, H.; Fan, Y.; Gao, Y.; Li, X.; Hu, Z.; Ding, K.; Wang, Y.; Wang, X. Fucoxanthin provides neuroprotection in models of traumatic brain injury via the Nrf2-ARE and Nrf2-autophagy pathways. Sci. Rep. 2017, 7, 46763. [Google Scholar] [CrossRef]
- Yang, X.; Guo, G.; Dang, M.; Yan, L.; Kang, X.; Jia, K.; Ren, H. Assessment of the therapeutic effects of fucoxanthin by attenuating inflammation in ovalbumin-induced asthma in an experimental animal model. J. Environ. Pathol. Toxicol. Oncol. 2019, 38, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, Y.; Veeraraghavan, V.P.; Mohan, S.K.; Ma, Y. Restorative effect of fucoxanthin in an ovalbumin-induced allergic rhinitis animal model through NF-κB p65 and STAT3 signaling. J. Environ. Pathol. Toxicol. Oncol. 2019, 38, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.H.; Chen, Y.L.; Huang, W.C.; Liou, C.J. Fucoxanthin attenuates fatty acid-induced lipid accumulation in FL83B hepatocytes through regulated Sirt1/AMPK signaling pathway. Biochem. Biophys. Res. Commun. 2018, 495, 197–203. [Google Scholar] [CrossRef]
- Huang, W.C.; Su, H.H.; Fang, L.W.; Wu, S.J.; Liou, C.J. Licochalcone A inhibits cellular motility by suppressing E-cadherin and MAPK signaling in breast cancer. Cells 2019, 8, 218. [Google Scholar] [CrossRef] [PubMed]
- Liou, C.J.; Chen, Y.L.; Yu, M.C.; Yeh, K.W.; Shen, S.C.; Huang, W.C. Sesamol alleviates airway hyperresponsiveness and oxidative stress in asthmatic mice. Antioxidants 2020, 9, 295. [Google Scholar] [CrossRef]
- Huang, W.C.; Liu, C.Y.; Shen, S.C.; Chen, L.C.; Yeh, K.W.; Liu, S.H.; Liou, C.J. Protective effects of licochalcone A improve airway hyper-responsiveness and oxidative stress in a mouse model of asthma. Cells 2019, 8, 617. [Google Scholar] [CrossRef]
- Huang, W.C.; Fang, L.W.; Liou, C.J. Phloretin attenuates allergic airway inflammation and oxidative stress in asthmatic mice. Front. Immunol. 2017, 8, 134. [Google Scholar] [CrossRef]
- Huang, W.C.; Chan, C.C.; Wu, S.J.; Chen, L.C.; Shen, J.J.; Kuo, M.L.; Chen, M.C.; Liou, C.J. Matrine attenuates allergic airway inflammation and eosinophil infiltration by suppressing eotaxin and Th2 cytokine production in asthmatic mice. J. Ethnopharmacol. 2014, 151, 470–477. [Google Scholar] [CrossRef]
- Liou, C.J.; Cheng, C.Y.; Yeh, K.W.; Wu, Y.H.; Huang, W.C. Protective effects of casticin from Vitex trifolia alleviate eosinophilic airway inflammation and oxidative stress in a murine asthma model. Front. Pharmacol. 2018, 9, 635. [Google Scholar] [CrossRef]
- Liou, C.J.; Huang, W.C. Dehydroepiandrosterone suppresses eosinophil infiltration and airway hyperresponsiveness via modulation of chemokines and Th2 cytokines in ovalbumin-sensitized mice. J. Clin. Immunol. 2011, 31, 656–665. [Google Scholar] [CrossRef]
- Sanjeewa, K.K.A.; Kim, E.A.; Son, K.T.; Jeon, Y.J. Bioactive properties and potentials cosmeceutical applications of phlorotannins isolated from brown seaweeds: A review. J. Photochem. Photobiol. B 2016, 162, 100–105. [Google Scholar] [CrossRef]
- Hu, L.; Chen, W.; Tian, F.; Yuan, C.; Wang, H.; Yue, H. Neuroprotective role of fucoxanthin against cerebral ischemic/reperfusion injury through activation of Nrf2/HO-1 signaling. Biomed. Pharmacol. 2018, 106, 1484–1489. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Ge, A.; Zhu, W.; Liu, Y.N.; Ji, N.F.; Zha, W.J.; Zhang, J.X.; Zeng, X.N.; Huang, M. Morin attenuates ovalbumin-induced airway inflammation by modulating oxidative stress-responsive MAPK signaling. Oxid. Med. Cell Longev. 2016, 2016, 5843672. [Google Scholar] [CrossRef]
- Kim, S.R.; Kim, D.I.; Kim, S.H.; Lee, H.; Lee, K.S.; Cho, S.H.; Lee, Y.C. NLRP3 inflammasome activation by mitochondrial ROS in bronchial epithelial cells is required for allergic inflammation. Cell Death Dis. 2014, 5, 460. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Corry, D.B.; Li, E. Mechanisms of allergy and adult asthma. Curr. Opin. Allergy Clin. Immunol. 2020, 20, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Rogers, L.K.; Cismowski, M.J. Oxidative stress in the lung—The essential paradox. Curr. Opin. Toxicol. 2018, 7, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Menzel, M.; Ramu, S.; Calven, J.; Olejnicka, B.; Sverrild, A.; Porsbjerg, C.; Tufvesson, E.; Bjermer, L.; Akbarshahi, H.; Uller, L. Oxidative stress attenuates TLR3 responsiveness and impairs anti-viral mechanisms in bronchial epithelial cells from COPD and asthma patients. Front. Immunol. 2019, 10, 2765. [Google Scholar] [CrossRef] [PubMed]
- Araujo, J.A.; Zhang, M.; Yin, F. Heme oxygenase-1, oxidation, inflammation, and atherosclerosis. Front. Pharmacol. 2012, 3, 119. [Google Scholar] [CrossRef]
- Charron, C.B.; Pakhalé, S. The role of airway hyperresponsiveness measured by methacholine challenge test in defining asthma severity in asthma-obesity syndrome. Curr. Opin. Allergy Clin. Immunol. 2016, 16, 218–223. [Google Scholar] [CrossRef]
- McGovern, T.K.; Chen, M.; Allard, B.; Larsson, K.; Martin, J.G.; Adner, M. Neutrophilic oxidative stress mediates organic dust-induced pulmonary inflammation and airway hyperresponsiveness. Am. J. Physiol. Lung Cell Mol. Physiol. 2016, 310, 6. [Google Scholar] [CrossRef] [PubMed]
- Nair, P.; Martin, J.G.; Cockcroft, D.C.; Dolovich, M.; Lemiere, C.; Boulet, L.P.; O’Byrne, P.M. Airway hyperresponsiveness in asthma: Measurement and clinical relevance. J. Allergy Clin. Immunol. Pr. 2017, 5, 649–659 e642. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Sivaprasad, U.; Gibson, A.M.; Ericksen, M.B.; Cunningham, C.M.; Bass, S.A.; Kinker, K.G.; Finkelman, F.D.; Wills-Karp, M.; Khurana Hershey, G.K. IL-13 receptor alpha2 contributes to development of experimental allergic asthma. J. Allergy Clin. Immunol. 2013, 132, 951–958 e951–956. [Google Scholar] [CrossRef]
- Chow, K.; Cosetti, M.K. Use of IL-5 inhibitor benralizumab as a novel therapy for eosinophilic otitis media: Clinical capsule and review of literature. Otol. Neurotol. 2020, 41, e238–e240. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, A.L.; Gama, C.S.; Rocha, N.P.; Teixeira, M.M. Revisiting the role of eotaxin-1/CCL11 in psychiatric disorders. Front. Psychiatry 2018, 9, 241. [Google Scholar] [CrossRef] [PubMed]
- Caminati, M.; Pham, D.L.; Bagnasco, D.; Canonica, G.W. Type 2 immunity in asthma. World Allergy Organ. J. 2018, 11, 13. [Google Scholar] [CrossRef]
- Persson, C. Airways exudation of plasma macromolecules: Innate defense, epithelial regeneration, and asthma. J. Allergy Clin. Immunol. 2019, 143, 1271–1286. [Google Scholar] [CrossRef] [PubMed]
- Moran, A.; Pavord, I.D. Anti-IL-4/IL-13 for the treatment of asthma: The story so far. Expert. Opin. Biol. 2020, 20, 283–294. [Google Scholar] [CrossRef]
- Kudo, M.; Ishigatsubo, Y.; Aoki, I. Pathology of asthma. Front. Microbiol. 2013, 4, 263. [Google Scholar] [CrossRef]
- Stewart, A.G.; Thomas, B.; Koff, J. TGF-beta: Master regulator of inflammation and fibrosis. Respirology (Carltonvic) 2018, 23, 1096–1097. [Google Scholar] [CrossRef]
- Royce, S.G.; Cheng, V.; Samuel, C.S.; Tang, M.L. The regulation of fibrosis in airway remodeling in asthma. Mol. Cell Endocrinol. 2012, 351, 167–175. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.J.; Chachi, L.; FitzGerald, J.M.; Backer, V.; Olivenstein, R.; Titlestad, I.L.; Ulrik, C.S.; Harrison, T.; Singh, D.; Chaudhuri, R.; et al. Effect of tralokinumab, an interleukin-13 neutralising monoclonal antibody, on eosinophilic airway inflammation in uncontrolled moderate-to-severe asthma (MESOS): A multicentre, double-blind, randomised, placebo-controlled phase 2 trial. Lancet Respir. Med. 2018, 6, 499–510. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, S.-J.; Liou, C.-J.; Chen, Y.-L.; Cheng, S.-C.; Huang, W.-C. Fucoxanthin Ameliorates Oxidative Stress and Airway Inflammation in Tracheal Epithelial Cells and Asthmatic Mice. Cells 2021, 10, 1311. https://doi.org/10.3390/cells10061311
Wu S-J, Liou C-J, Chen Y-L, Cheng S-C, Huang W-C. Fucoxanthin Ameliorates Oxidative Stress and Airway Inflammation in Tracheal Epithelial Cells and Asthmatic Mice. Cells. 2021; 10(6):1311. https://doi.org/10.3390/cells10061311
Chicago/Turabian StyleWu, Shu-Ju, Chian-Jiun Liou, Ya-Ling Chen, Shu-Chen Cheng, and Wen-Chung Huang. 2021. "Fucoxanthin Ameliorates Oxidative Stress and Airway Inflammation in Tracheal Epithelial Cells and Asthmatic Mice" Cells 10, no. 6: 1311. https://doi.org/10.3390/cells10061311
APA StyleWu, S.-J., Liou, C.-J., Chen, Y.-L., Cheng, S.-C., & Huang, W.-C. (2021). Fucoxanthin Ameliorates Oxidative Stress and Airway Inflammation in Tracheal Epithelial Cells and Asthmatic Mice. Cells, 10(6), 1311. https://doi.org/10.3390/cells10061311





